Abstract
We used the NADPH-diaphorase histochemical method as a potential marker for nitric oxide synthase (NOS)-containing nerve fibers innervating the pineal gland of the sheep. Nerve fibers containing NADPH-diaphorase activity provide dense innervation of the sheep pineal gland. The nerve fibers were located in the pineal capsule, in the connective tissue septae separating the lobuli of the gland, and penetrating between the pinealo-cytes. The nerve fibers were either smooth or endowed with boutons en passant. After bilateral removal of the superior cervical ganglion, the dense network of NADPH-diaphorase-positive fibers was still present in the gland. Ganglionectomy affected neither the distribution nor the appearance of the NADPH-diaphorase-positive fibers. Most of the NADPH-diaphorase-positive fibers also contained peptide histidine isoleucine and vasoactive intestinal polypeptide, and a comparatively smaller fraction contained neuropeptide Y. Pinealocytes never exhibited NADPH-diaphorase activity. These results demonstrate a major neural input to the sheep pineal gland with NADPH-diaphorase-positive nerve fibers of nonsympathetic origin.
Keywords
T
The final neural input to the pineal gland is diversified (Korf and M⊘ller 1984). Sympathetic nerve fibers from perikarya located in the superior cervical ganglia (Zhang et al. 1991) have been demonstrated in every mammalian species investigated (see M⊘ller 1992). However, pinealopetal neuronal projections from para-sympathetic ganglia, as well as from perikarya located in the brain reaching the pineal gland via the pineal stalk, have been described anatomically (for review see M⊘ller 1992) and electrophysiologically (Semm and De-maine 1984).
The sheep is a highly photoperiodic animal (Ravault et al. 1989; Matthews et al. 1993) with prominent cir-cadian rhythms (Tessoneaud et al. 1995). This, together with a large pineal gland, makes it a model of choice for anatomic and biochemical investigations. This species is endowed with prominent innervation of the pineal gland with nerve fibers of extrasympa-thetic origin, which have been shown to contain vaso-active intestinal peptide (VIP), peptide histidine isoleucine (PHI) (Cozzi et al. 1994), and neuropeptide Y (NPY) (Cozzi et al. 1992).
Recently, nitric oxide (NO) was shown to be synthesized in the mammalian pineal gland (Spessert et al. 1993; Lin et al. 1994; Maronde et al. 1995). This labile intercellular messenger molecule has been shown to play a role in many cellular physiological processes (Bredt and Snyder 1992). In the pinealocyte, adrenergic cGMP stimulation has been shown to be mediated by NO activation of guanylate cyclase (Spessert et al. 1993; Guerrero et al. 1994; Schaad et al. 1995).
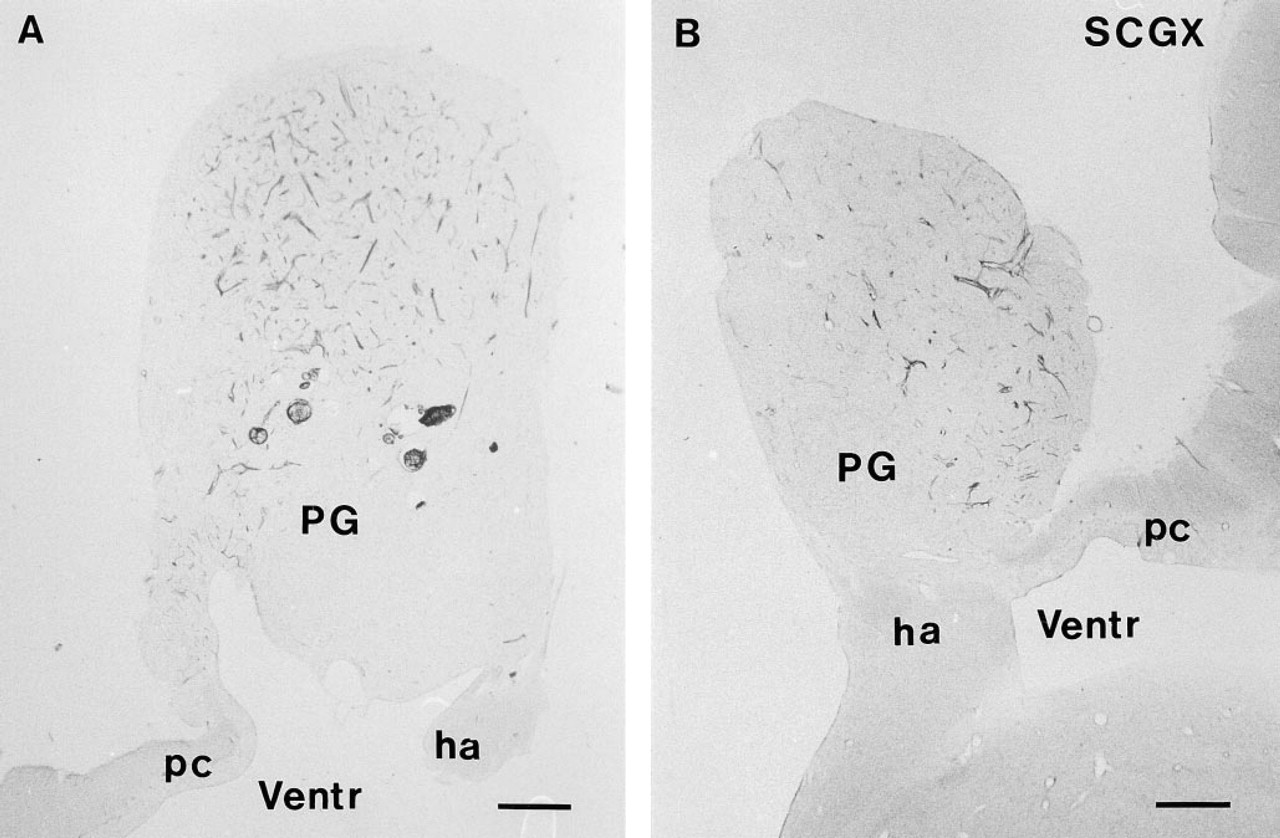
NADPH-diaphorase activity in the pineal gland (PG) of a control sheep. The pineal gland, which is on top of the third ventricle (Ventr), connects through the pineal stalk with the habenular nucleus (ha) rostrally and to the posterior commissure (pc) caudally. Note the large number of fibers in the ventrocaudal part of the pineal.
Because NADPH is a co-substrate for the NO-synthesizing enzyme nitric oxide synthase (NOS) (Nathan and Xie 1994), most NOS-containing neurons also exhibit NADPH-diaphorase (NADPH-d) activity (Dawson et al. 1991). However, other enzymes, such as cytochrome P450 reductase, also exibit NADPH-d activity (Tracey et al. 1993).
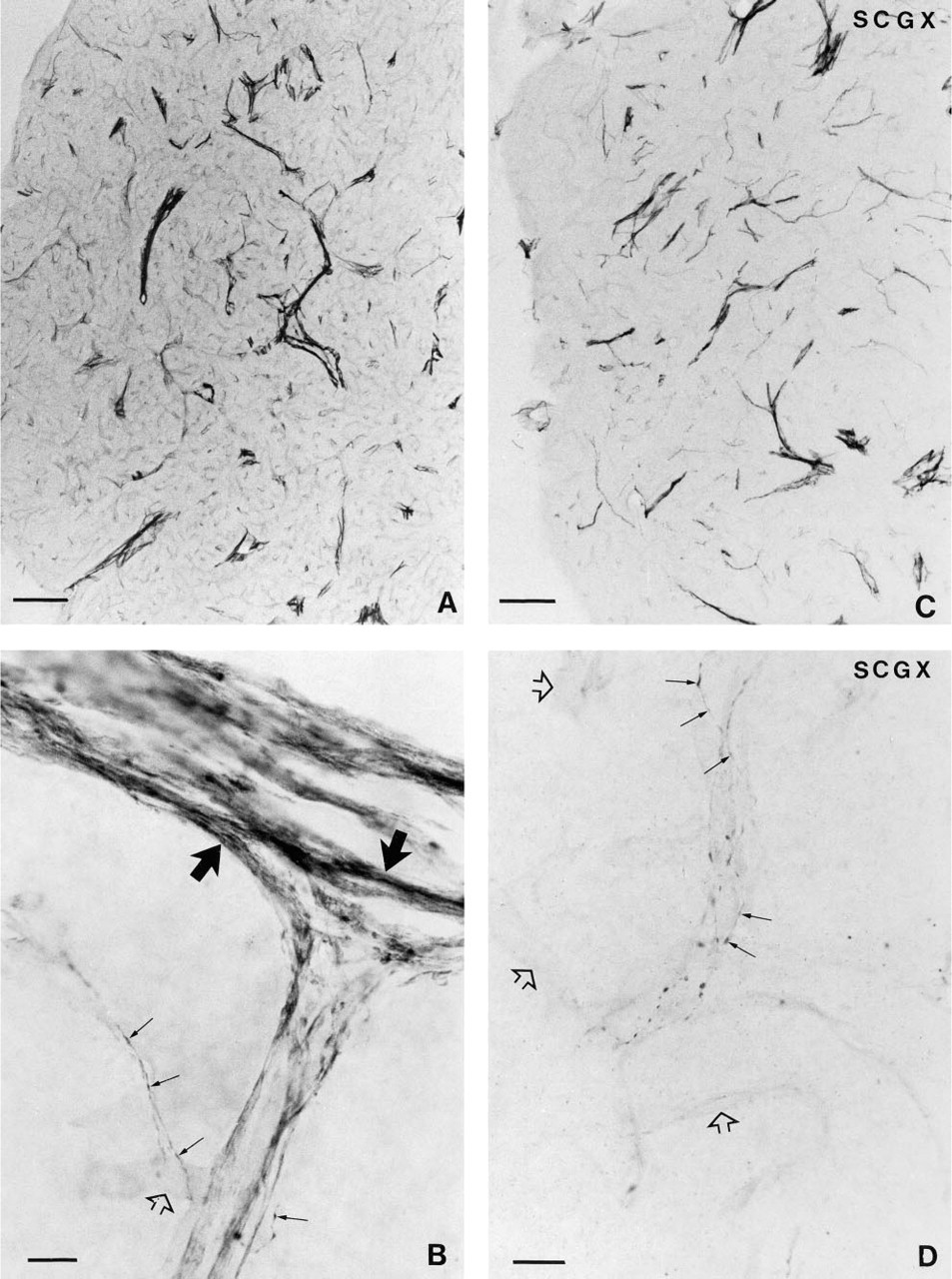
Photomicrograph of a section through the pineal gland of a control sheep, showing the presence of NADPH-d activity. NADPH-d-positive fibers cluster around blood vessels and intraparenchymally. Bar = 250 μm.
In the present study of the sheep pineal gland, the NADPH-d histochemical method was employed, revealing dense innervation of the gland. The nerves were still present after removal of both superior cervical ganglia. Furthermore, co-localization of NADPH-d activity with neuropeptides VIP and PHI was demonstrated in the nerve fibers. Some fibers also contained NPY. Therefore, this study clearly confirms the presence of prominent nonsympathetic innervation of the sheep pineal gland, which might use NO in its neurotransmission.
Materials and Methods
Animals
Eight Ile France rams, weighing 80 kg, were used. The rams were exposed to natural photoperiods in Nouzilly, France, and given free access to food and water. Four animals were subjected to superior cervical ganglionectomy (SGGX) and four animals were used as controls.
Superior Cervical Ganglionectomy
The rams were deeply anesthetized with sodium pentobarbital (Sanofi, Paris, France; 0.8 g/animal iv) and given atropine (40 mg/animal). After intubation, the anesthesia was maintained with 4% halothane and 2 liters of oxygen/min during the surgical procedure. Authorization for the surgical procedures was obtained by INRA, Nouzilly, France. The ganglion on the left side of the neck was removed first, followed by removal of the right ganglion 1 week later. After bilateral removal of the ganglia the rams exhibited ptosis as well as miosis. After the latter surgery, the animals were allowed to recover for 1 month.
Fixation of the Brains
The rams were sacrificed by decapitation performed by a licensed butcher. The heads were immediately perfused with 2 liters of PBS, pH 7.4, containing 1% sodium nitrite, followed by 4 liters of 4% paraformaldehyde in PBS. The skulls were opened and the brains carefully removed and postfixed in the same fixative for 48 hr at 4C. The epithalami were isolated by dissection and washed in PBS for 24 hr, followed by cryoprotection in a solution of 20% sucrose in PBS for 48 hr. Serial sagittal sections 40 μm thick were obtained in a cryostat and transferred to wells with PBS.
NADPH-diaphorase Histochemistry
The free-floating sections were washed for 10 min in PBS, followed by permeabilization of the membranes with a solution containing 0.1% Triton X-100 (TX) in PBS for 10 min at 37C. The sections were incubated for 60 min at 37C in a solution containing (1 mg/ml) β-nicotinamide adenine dinu-cleotide phosphate, reduced form, tetrasodium salt (β-NADPH, Sigma #N1630; St Louis, MO) (0.1 mg/ml) nitroblue tetra-zolium (NBT; Sigma #N6876), and 0.3% TX in 0.1 M PBS (pH 7.4). The reaction was terminated by washing the sections in PBS. After a double rinse in PBS, the sections were mounted, dried, and coverslipped with Depex. Control reaction was performed by omission of β-NADPH.
Double Labeling
After 15-min incubation in the NADPH-d solution, sections were washed three times for 10 min in PBS. Then they were incubated in a solution containing 5% swine serum, 1% bovine serum albumin (BSA), 0.3% TX in PBS for 20 min at room temperature (RT), followed by incubation for 48 hr at 4C in a solution containing primary rabbit polyclonal antibodies against VIP (diluted 1:100, #8086; a generous gift from Professor J. Fahrenkrug, Copenhagen), or PHI (diluted 1:8,000, #3668–5; from Professor J. Fahrenkrug, Copenhagen), or NPY (diluted 1:1,000, #8182–5; raised in our laboratory) in 0.3% TX and 1% BSA in PBS. Next, the sections were washed three times for 10 min in PBS, followed by incubation in FITC-conjugated swine anti-rabbit serum (diluted 1:20; Dako, Copenhagen, Denmark) for 1 hr at RT. Finally, the sections were washed in PBS, mounted, and coverslipped with Glycergel.
Immunohistochemical Absorption Controls
When the VIP and PHI antisera were preabsorbed for 24 hr with the specific peptide (50 μg/ml diluted antiserum), all staining was abolished.
Results
A large number of nerve fibers positive for NADPH-d were found in the pineal gland of the sheep (Figure 1A). The density of this neuronal network was low in the dorsorostral and high in the ventrocaudal portion of the gland (Figures 1A and 1B). The fibers were distributed in the capsule, from which they penetrated the pineal gland primarily via the connective tissue septae, often located close to blood vessels (Figures 2A, 2C, and 2D). Single fibers were observed intra-parenchymally in the pineal lobules between the pine-alocytes (Figure 2B). The appearance of the NADPH-d nerve fibers was either smooth or with boutons en passant (Figures 2B and 2D). NADPH-d-positive nerve fibers were observed in both the rostral and the caudal portion of the pineal stalk (Figure 1A).
After SCGX, neither the distribution nor the appearance of the NADPH-d-positive nerve fibers in the pineal was affected (Figure 1B). A decrease in the number of fibers was not observed in any of the SCGX animals (Figures 2A and 2C).
VIP (Figure 3A) and PHI immunoreactivity (Figure 3C) were observed in many nerve fibers exhibiting NADPH-d activity (Figures 3B and 3D). A few NADPH-d-positive fibers, in both control (Figure 4B) and superior cervical ganglionectomized animals, also contained NPY (Figure 4A).
In control as well as SCGX animals, the endothelia of blood vessels exhibited weak NADPH-d activity. NADPH-d histochemistry did not reveal any positive cells in sheep pineal gland.
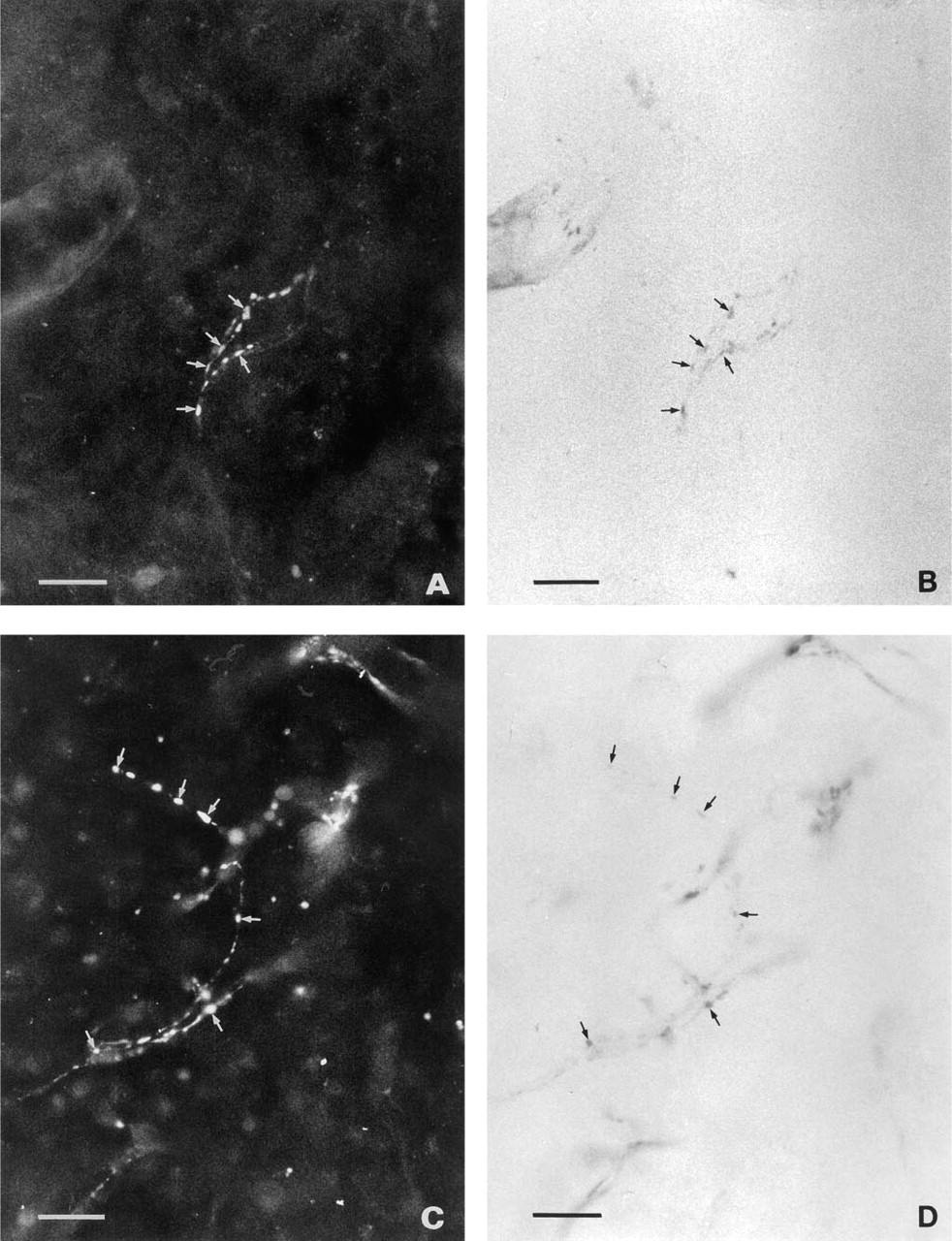
Double-stained sheep pineal sections. Some but not all of the NADPH-d-positive nerve fibers (arrows)
Discussion
The presence of NADPH-d-reactive nerve fibers, demonstrated in the present study of the sheep pineal gland, is of interest because of the recent demonstration of the NO-synthesizing enzyme NOS in the mammalian pineal gland (Spessert et al. 1992; Lin et al. 1994; Schaad et al. 1994; Maronde et al. 1995; Spes-sert et al. 1995; White and Klein 1995; L⊘pez-Figueroa and M⊘ller 1996; L⊘pez-Figueroa et al. 1996). It has been suggested that NO is involved in the adrenergic stimulation of cGMP via a stimulation by NO of gua-nylyl cyclase (Guerrero et al. 1994; Maronde et al. 1995; Schaad et al. 1995).
The NADPH-d histochemical reaction is often used as a marker for NOS-containing neurons (Dawson et al. 1991), although not all NADPH-d-reactive neurons contain NOS (Wörl et al. 1994). However, recent immunohistochemical stainings (L⊘pez-Figueroa et al. 1996) in our laboratory have shown NOS-immunore-active fibers in the sheep pineal gland, confirming the presence of this enzyme in presynaptic intrapineal fibers of this species.
It must be emphasized that in our study of the sheep pineal gland, NADPH-d was found only presyn-aptically in nerve fibers. Pinealocytes never exhibited this enzyme activity. In contrast, in the rat pineal gland, a few NADPH-d-reactive nerve fibers have been observed (Vincent and Kimura 1992), a finding questioned in other studies (Sato 1992; Maronde et al. 1995). However, we have recently confirmed, in an extensive series of rat pineal glands stained for NADPH-d, the presence of this enzyme in both nerve fibers and some (but few) pinealocytes (L⊘pez-Figueroa and M⊘ller 1996).
In the sheep, the location of NADPH-d activity indicates a presynaptic function of NO, e.g., an influence on the release of VIP/PHI and NPY, which were shown to be present in the NADPH-d reactive fibers in this study. However, NO is a very labile molecule, and diffusion of NO from a presynaptic synthesis to a postsynaptic location, where it may stimulate the gua-nylyl cyclase, cannot be excluded.
The presence of NADPH-d-reactive nerve fibers close to the blood vessels may suggest an influence on the regulation of pineal blood flow. The brain vascula-ture is richly innervated by nerve fibers containing NADPH-d (Suzuki et al. 1993) and NOS (Nozaki et al. 1993; Zhang et al. 1995). NO is a potent vasodilator (Gaw et al. 1991), and in the sheep the VIP-induced relaxation of endothelium-denuded cerebral arteries is blocked by inhibition of NOS (Gaw et al. 1991). We have recently shown, by use of an antibody against en-dothelial NOS, the presence of this enzyme in the vas-culature of the sheep (L⊘pez-Figueroa et al. 1996). Production of NOS by the endothelial cells is therefore likely.
The origin of the NADPH-d fibers shown in the present study is enigmatic. However, a parasympa-thetic origin from the sphenopalatine or otic ganglion is possible. We have previously demonstrated a prominent nonsympathetic innervation of the sheep pineal gland by nerve fibers containing VIP/PHI (Cozzi et al. 1994) and NPY (Cozzi et al. 1992; M⊘ller et al. 1994). The VIP/PHI-containing nerve fibers probably originate in the sphenopalatine ganglia or otic ganglia, because perikarya containing VIP/PHI are richly present in this location (Minami et al. 1994). In the same ganglia, VIP neurons also often contain NOS (Nozaki et al. 1993; Yamamoto et al. 1993; Ceccatelli et al. 1994). Accordingly, we hypothesize that the majority of the NADPH-d-reactive nerve fibers, demonstrated in the present study of the sheep, originate from perikarya in these ganglia.
In both control and ganglionectomized animals, a few NADPH-d-reactive nerve fibers contained NPY. Nerve cell bodies containing NPY (Ceccatelli et al. 1994) and NADPH-d (Suzuki et al. 1993) are present in the sphenopalatine ganglion of the rat. However, a few perikarya containing NADPH-d activity have also been described in the superior cervical ganglion (Dun et al. 1995). Therefore, this ganglion cannot be totally excluded as the source for some NADPH-d-reactive nerve fibers in the sheep pineal gland.
Fibers exhibiting NADPH-d activity were present in the sheep pineal stalk. An additional possibility for the origin of these fibers might be from a nucleus in the forebrain or brainstem.
In summary, this investigation demonstrates a prominent innervation of the sheep pineal gland with nerve fibers containing NADPH-d activity. The fibers are of nonsympathetic character and the majority of these fibers contain neuropeptide VIP or PHI. A few nerve fibers also contain NPY.
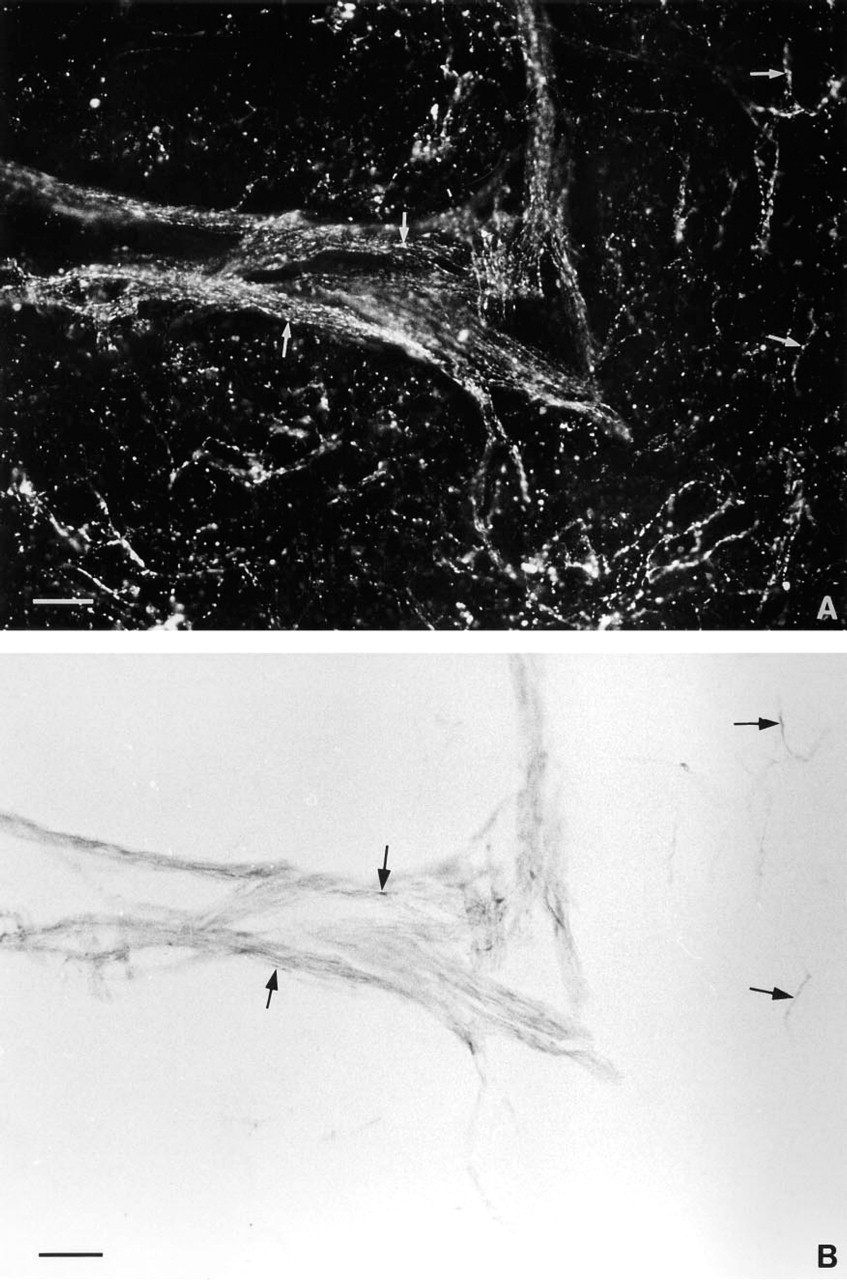
Fluorescence micrograph
Footnotes
Acknowledgements
Supported by grants from Novo's Fonds Komite, The Danish Research Foundation (#12–0236, #12–1486–1, and #12–1642–1), and by a grant from the Danish Research Foundation to The Biotechnology Center for Signal Peptide Research.
We are grateful to Ms Ursula Rentzmann for technical assistance and to Ms Grazyna Hahn for photographic assistance.
