Abstract
Synthesized oligonucleotides are used in anti-sense and anti-gene technology to control gene expression. Because cells do not easily take up oligonucleotides, cationic liposomes have been employed to facilitate their transport into cells. Athough cationic liposomes have been used in this way for several years, the precise mechanisms of the delivery of oligonucleotides into cells are not known. Because no earlier reports have been published on the liposomal delivery of oligonucleotides at the ultrastructural level, we performed a study, using electron microscopy, on the cellular uptake and intracellular distribution of liposomal digoxigenin-labeled oligodeoxynucleotides (ODNs) at several concentrations (0.1, 0.2, and 1.0 μM) in CaSki cells. Two cationic lipids (10 μM) were compared for transport efficiency: polycationic 2,3-dioleoyloxy-N-[2(sperminecarboxamido)ethyl]-N,N-dimethyl-1-propanaminium trifluoroacetate (DOSPA) and monocationic dimethyl-dioctadecylammonium bromide (DDAB). Both liposomes contained dioleoyl-phosphatidylethanolamine (DOPE) as a helper lipid. Endocytosis was found to be the main pathway of cellular uptake of liposomal ODNs. After release from intracellular vesicles, ODNs were carried into the perinuclear area. The nuclear membrane was found to be a barrier against the penetration of ODNs delivered by liposomes into the nucleus. Release from vesicles and transport into the nuclear area was faster when the oligo-DDAB/DOPE complex had a positive net charge (0.1 and 0.2 μM ODN concentrations), and only under this condition were some ODNs found in nucleoplasm. Although DOSPA/DOPE could also efficiently deliver ODNs into the cytosol, no ODNs were found in nucleoplasm. These findings suggest that both the type of liposome and the charge of the oligo-liposome complex are important for determination of the intracellular distribution of ODNs.
Keywords
O
Oligonucleotide-based techniques involve several problems, especially those associated with the stability, affinity, specificity, and cellular delivery of the oligos (Wagner, 1994). For example, unmodified phosphodiester antisense oligodeoxynucleotides (AS-ODNs) are rapidly degraded by nucleases (Scanlon et al., 1995). To solve this problem, several modifications of AS-ODNs have been developed, but these modifications have often resulted in reduced cellular uptake (e.g., using peptide nucleic acids) (Wittung et al., 1995; Hanvey et al., 1992) or nonspecific effects (e.g., using phosphorothioate ODNs) (Perez et al., 1994). Nevertheless, the worst problem is ineffective cellular uptake. Not only longer DNA or RNA but also shorter oligonucleotides often depend on a carrier system for effective delivery into cells.
As for oligonucleotides, cationic liposomes have been widely used because of their several advantages over conventional delivery methods, such as spontaneous complex formation with negatively charged nucleic acids (Smith et al., 1993). Cationic liposomes are also less toxic than traditional in vitro transfection methods (e.g., calcium phosphate precipitation, DEAE and polybrene-mediated transfection and electroporation) (Farhood et al., 1995; Smith et al., 1993). Positively charged liposomes not only improve the transfer of oligonucleotides through cellular barriers but also protect against enzymatic degradation of oligonucleotides in cell culture (Lappalainen et al., 1994b; Capaccioli et al., 1993) and in human serum (Capaccioli et al., 1993). The disadvantage of cationic liposomes is that they may accumulate in the phagocytic cells of the reticuloendothelial system (RES) after iv injection (Lasic and Papahadjopoulos, 1995; Smith et al., 1993) and therefore do not deliver oligonucleotides to all target sites in vivo. However, local application to the skin, mucosa, and eye, inhalation into the respiratory tract and lungs, and injections into cavities, tumors, and other targets are possible. In addition, blood cells, and naturally RES, are suitable targets for the delivery of oligonucleotides by cationic liposomes. At present, three companies based on the anti-sense technology are working on oligonucleotide delivery mechanisms using cationic liposomes (Gura, 1995).
Although cationic liposomes have been successfully used as oligonucleotide carriers, the cellular uptake mechanisms and intracellular fate of liposomal oligonucleotides are not fully understood. Results from our previous studies suggest that liposomal ODNs are transported into the nucleus (Lappalainen et al., 1994b). In addition, Bennett et al. (1992) and Wagner et al. (1993) have found that ODNs delivered with cationic liposomes are localized within the nucleus. However, these observations were made at the light microscopic level. Electron microscopy has shown that most non-liposomal ODNs (Tarrasón et al., 1995), as well as liposomal plasmid DNA (Zabner et al., 1995; Zhou and Huang, 1994), remain inside the lysosomal compartment and only a small proportion of nucleic acids is released into the cytosol. On the other hand, using both cell fractionation and electron microscopy, Beltinger et al. (1995) showed a high amount of phosphorothioate ODNs (without carriers) in the cytosol and nucleus. Although there can be differences in the uptake between the cell lines, the findings may also partly result from methodological differences. Therefore, the present study was carried out at the electron microscopic level to further evaluate the transfer process and intracellular fate of ODNs combined with polycationic DOSPA/DOPE or monocationic DDAB/DOPE liposomes. AS-ODNs against HPV 16 E7 oncogene were used in CaSki cells, which are an HPV 16-positive cell line.
Materials and Methods
Cell Culture
Adherent CaSki cells (a human cervical cancer cell line known to contain 600 copies of human papilloma virus 16 DNA) (Breitburd, 1987) were grown in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum (Gibco BRL; Paisley, UK) at 37°C in 5% CO2. For light and electron microscopy, 1.2 × 105 cells/well were grown in 24-well plates (Nunc; Roskilde, Denmark) with and without round glass microscope slides, respectively. The CaSki cells were cultured for 12 hr before addition of ODNs and liposomes to the culture medium.
ODNs and Liposomes
Phosphodiester oligodeoxynucleotides purchased from Genosys (Genosys Biotechnologies; Cambridge, UK) had digoxigenin (DIG-ODNs) incorporated into the 5’ end during synthesis and were purified by HPLC. The (anti-sense) sequence of DIG-ODN (16-mer) was 5'GTGTATCTCCATGCAT3', which is complementary to the initiation site of the E7 oncogene of HPV 16 (from nt 562). The cellular uptake of a random sequence, 5'CATCTGTATTGGATCG3', was also investigated.
A commercial polycationic transfection reagent, LipofectAMINE (Gibco BRL) containing 2,3-dioleoyloxy-N-[2 (sperminecarboxamido)ethyl]-N,N-dimethyl-1-propanaminium trifluoroacetate (DOSPA) and dioleoyl-phosphatidylethanolamine (DOPE) (3/1 w/w) or a monocationic liposomal preparation containing dimethyldioctadecylammonium bromide (DDAB) and DOPE (2/5 w/w) was used. The liposomes containing DDAB and DOPE were prepared as described by Rose et al. (1991). The size distribution of the lipid-oligonucleotide dispersions was determined by quasielastic light scattering (Nicomp Submicron Particle Sizer, Model 370; Santa Barbara, CA) three times from 400 μl of samples, which contained DDAB/DOPE at the concentration of 10 μM. Table 1 shows the charge ratio and mean diameter of the liposomes and the oligo-liposome complexes.
Incubation of DIG-ODNs and Cationic Liposomes in CaSki Cells
On the basis of our earlier studies (Lappalainen et al., 1994a), DDAB and DOSPA were used at a final concentration of 10 μM in culture medium. The final concentrations of DIG-ODNs were 0.1, 0.2, and 1.0 μM, with or without liposomes. The ODNs were incubated with cationic liposomes in H2O for 15 min at room temperature before adding them to the culture medium. For light and electron microscopy, DIG-ODNs, with or without liposomes, were incubated with CaSki cells for 20 min, 1.5, 4, and 24 hr. Light microscopy was also used at 5 min.
Particle size, ODN/liposome ratio (-/+) and net charge of the oligo-lipid complexes a

aMean particle diameter (and SD) of liposomes and ODN/liposome complexes were measured after incubation in H2O using quasi-elastic light scattering. The final concentrations of cationic lipid (DOSPA or DDAB) were 10 μM and ODNs 0.1, 0.2, or 1.0 μM. The charges of one DOSPA, DDAB, and ODN (16-mer) molecule were +5, +1, and -16, respectively.
bPresumably micellar structures were too small to be accurately measured by quasi-elastic light scattering.
Immunocytochemistry
Detection of DIG-labeled ODNs was based on a direct immunocytochemical technique using horseradish peroxidase-labeled sheep polyclonal antibody against digoxigenin (anti-DIG-POD).
Processing of CaSki Cells. After incubation with DIG-ODNs and cationic liposomes, CaSki cells were fixed in 3% paraformaldehyde (EM grade; TAAB, Berks, UK) and 0.5% glutaraldehyde (EM grade; TAAB) in 0.1 M phosphate buffer (PB, pH 7.4) for 15 min. After washings, the cells were treated with 1% NaBH4 for 5 min to reduce free aldehyde groups and double bonds. After extensive washings, CaSki cells were immersed in 25% sucrose in 0.05 M PB for 5 min and freeze-thawed three times using liquid nitrogen to increase the penetration of the antisera during immunostaining.
Immunostaining. The samples were washed three times using 0.05 M Tris-buffered saline (TBS, pH 7.4). At the first washing, 0.2% Triton X-100 in TBS was used for 5 min. After incubation in 10% normal goat serum (NGS, 15 min) in TBS and washing in 1% NGS in TBS, the samples were incubated with horseradish peroxidase-conjugated anti-DIG antibody Fab fragments (anti-DIG-POD, 1:500; Boehringer Mannheim, Pentzberg, Germany) in 1% NGS in TBS at 4°C overnight. The immunoperoxidase reaction was developed using 3,3'-diaminobenzidine hydrochloride (DAB) as chromogen.
Controls. Three controls were used: (a) cells without DIG-ODNs and liposomes; (b) cells with cationic liposomes, but without oligonucleotides; and (c) cells treated with liposomal DIG-ODNs, omitting anti-DIG incubation during immunostaining.
Sample Preparation for Light Microscopy. After immunostaining, the immunoperoxidase reaction product was intensified using 0.02% OsO4<PS8fi in PB for 4 min. The samples were dehydrated in ethanol and treated with xylene for 5 min before mounting in DePex (Gurr; BDH, Poole, UK). At the light microscopic level the experiments were repeated three times in duplicate.
Sample Preparation for Electron Microscopy (EM). After immunostaining, the samples were treated with 1% OsO4 in PB for 1 hr. After staining with 2% uranyl acetate and dehydration, the samples were embedded in Epon (Fluka; Buchs, Switzerland). Ultrathin sections were cut on copper grids and stained with uranyl acetate and lead citrate. Some sections were left unstained for detection of any immunoperoxidase reaction in the nucleus. The sections were analyzed using JEOL JEM-1200 EX transmission EM. The experiments were performed twice in duplicate for electron microscopy. Many sections were cut from the samples for screening the intracellular localization of immunopositivity.
Results
Immunopositive Signals on Plasma Membrane After 5 Min of Incubation
On light microscopy, immunopositive signals for DIG-ODNs delivered by DDAB/DOPE were already seen on the plasma membrane and in the cytoplasm after 5 and 20 min (Figure 1B) of incubation, respectively. After 1.5 and 4 hr (Figure 1E) of incubation, immunopositivity for DIG-ODNs with DDAB/DOPE was localized to the perinuclear or nuclear area.
The immunopositive signals were strongest after 1.0 μM DIG-ODN and DDAB/DOPE treatment. Signals from DIG-ODNs delivered by DDAB/DOPE were more intense earlier (5 min, 20 min, 1.5 hr) than those delivered by DOSPA/DOPE, as seen with CaSki cells after 20 min of incubation with liposomal DIG-ODNs in Figures 1B and 1C. Most cells (over 95%) contained immunopositivity after 1.5-hr incubation of 1.0 μM DIG-ODN with DDAB/DOPE (Figure 1E). However, some immunopositivity from DIG-ODNs delivered using DOSPA/DOPE was also seen in the cytoplasm and perinuclear (or nuclear) area after 20 min (Figure 1C) and 1.5 hr of incubation, respectively. The difference disappeared after 4 hr (Figures 1E and 1F), and after 24 hr the signals were slightly more intense from DIG-ODNs delivered using DOSPA/DOPE (Figure 1I) than from those delivered using DDAB/DOPE (Figure 1H). At ODN concentrations of 0.1 and 0.2 μM, only weak immunopositivity was seen with DOSPA/DOPE in CaSki cells, whereas intense immunoreactivity was seen with DDAB/DOPE. When cationic liposomes were not used, DIG-ODNs showed only weak immunoreactivity at all concentrations and observation times. Immunopositivity for liposomal random DIG-ODN was similar to that for liposomal anti-sense DIG-ODN.
Immunoreactivity in Intracellular Vesicles
At the ultrastructural level, liposomal DIG-ODNs were taken up into small intracellular vesicles, which were sometimes fused into large multivesicular bodies in the cytoplasm (Figures 2C-2E). In addition, immunopositive plasmalemmal vesicles, open at the cell surface, could often be observed (Figure 2E). Irrespective of the oligo concentration or the type of liposome, the oligo-liposome complexes were similarly internalized. In the cytosol, liposomes were localized in the vicinity of immunoreactive signals for DIG-ODNs (Figure 2F). Interestingly, the plasma membrane and the envelopes of the intracellular vesicles frequently showed high immunoreactivity (Figures 2C-2E). Although light microscopy showed possible weak immunoreactivity in a few CaSki cells treated with DIG-ODNs without liposomes (Figure 1D), no immunopositive signals were detected at the ultrastructural level. By contrast, cells treated with liposomes but without DIG-ODNs (Figure 1G) or cells without anti-DIG incubation were found immunonegative on light and electron microscopy.
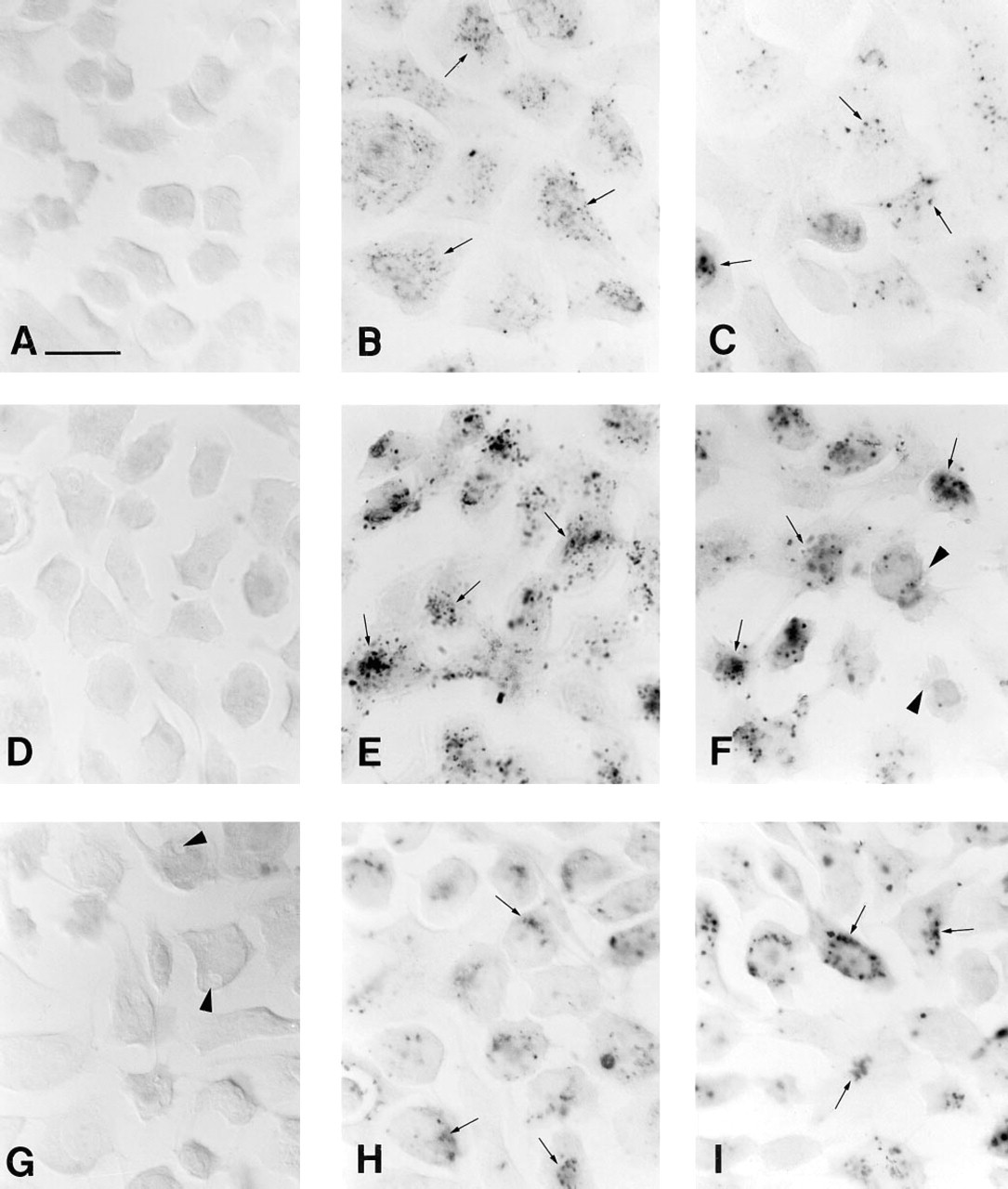
Light photomicrographs of CaSki cells immunostained for DIG. Some of the punctate immunoreactivity is indicated by arrows.
Differences in Intracellular Distribution
After 1.5 hr of incubation of CaSki cells treated with DIG-ODNs at a concentration of 1.0 μM and DDAB/DOPE (when the oligo-lipid complex had a negative net charge; Table 1), most immunoreactive signals were still located in the intracellular vesicles (Figure 2E). With a longer incubation time (4 hr), most immunopositivity was found in the cytosol (Figure 2F). Although immunopositivity was detected in the perinuclear area, no immunoreactivity was seen within the nucleus (Figure 2F). After 24 hr of incubation, high immunoreactivity was still detected in the cytosol and nuclear envelope, but no immunopositivity was seen in the nucleoplasm (Figure 2G). The possible positive signals from the immunoperoxidase reaction in the nucleus had to be confirmed using ultrathin sections unstained by uranyl acetate and lead citrate (Figure 2H), since the chromatin of the nucleus is intensively stained with these agents (Figure 2A), hindering identification of any immunoperoxidase reaction product in the nucleus.
CaSki cells treated with DIG-ODNs at concentrations of 0.1 or 0.2 μM and DDAB/DOPE (when the oligo-lipid complexes were positive; Table 1) showed immunopositivity in their cytosol and perinuclear areas after only 1.5 hr of incubation (Figure 2I). The important finding was that although most immunopositivity was located on the nuclear envelope, some was also seen in the nucleoplasm after 4 hr of incubation (Figure 2J). After 24 hr of incubation, staining was less intense and no immunopositivity was seen in the nucleoplasm (Figures 2K and 2L). Treatment of cells with ODNs (0.1, 0.2 or 1.0 μM) and DOSPA/DOPE (when the oligo-lipid complexes were positive; Table 1) did not produce any immunoreactivity in the nucleoplasm at any observation time. By contrast, immunoreactivity in the cytosol and nuclear membrane weakened with decreasing oligo concentrations and was similar to that in the DDAB/DOPE delivery system.
General Morphological Observations
In light microscopy there were no differences in cellular uptake between random and anti-sense oligos, regardless of the carrier system or oligo concentration used. At the EM level, random and anti-sense oligos were compared at the concentration of 1.0 μM after 1.5-hr incubation and no differences were observed. More foaming (bubbling) in the cytosol (Figure 1G) and blebbing of the plasma membrane (Figure 1F) occurred in cells treated with cationic liposomes than in control cells. However, cell membranes and nuclei remained intact after liposome treatment. The changes were more frequent with DOSPA/DOPE than DDAB/DOPE. Liposomes and liposomal DIG-ODNs also caused a slight reduction in cell growth after 24-hr incubation. No effects on cell growth or cell shape could be observed after DIG-ODN treatment without liposomes. Shrunken and rounded cells phagocytosed by adjacent cells (indicating possible apoptosis) were observed both in the control samples and in the samples treated with liposomes and DIG-ODNs. These changes were observed both in light and electron microscopy and are in agreement with the finding of Arens et al. (1995), who showed that HPV 16-positive cells have a high apoptosis rate.
Discussion
The most important findings of the present study were as follows: (a) liposomal ODNs are efficiently taken up by endocytosis irrespective of the charge of the ODN/liposome complex; (b) the charge of the ODN-DDAB/DOPE complex and the type of liposome affected intracellular distribution; and (c) the nuclear envelope was a barrier against the penetration of ODNs delivered by liposomes into the nucleus of CaSki cells.
Our study showed that both negatively and positively charged oligo-liposome complexes were rapidly and effectively taken up during endocytosis, released into the cytosol, and transported into the perinuclear area. Both polycationic and monocationic liposomes transferred ODNs along the endocytotic pathway. This is in agreement with recent studies in which the main route of liposomal DNA was found to be endocytosis and thereafter its release into cytosol (Wrobel and Collins, 1995; Zhou and Huang, 1994). Instead, our results contradict the theory that the net charge of the nucleic acid-liposome complex has to be positive for effective transport of nucleic acids into cells (Farhood et al., 1995; Zhou and Huang, 1994). However, the direct plasma membrane fusion mechanism cannot be entirely excluded because the immunoreactivity was also observed in the plasma membrane of CaSki cells.
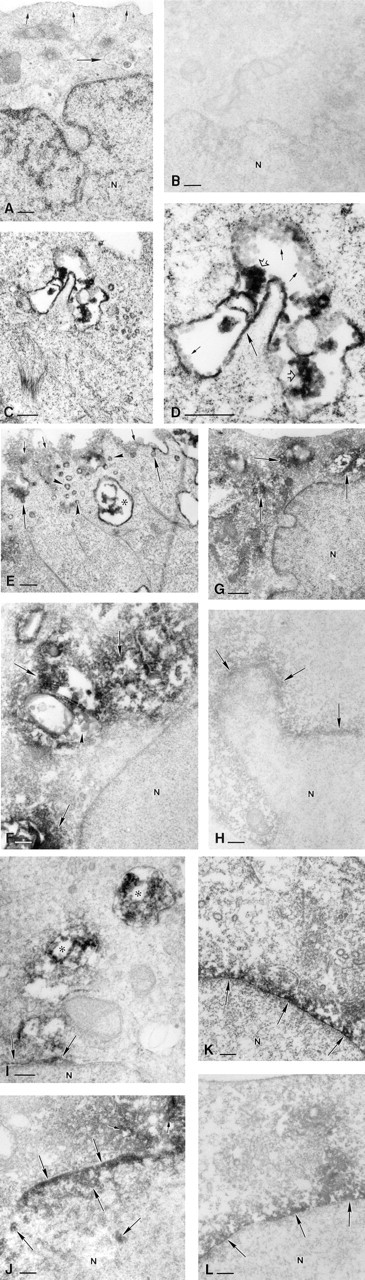
Electron photomicrographs of CaSki cells immunostained for DIG. N, nucleus.
In the presence of DDAB/DOPE, more ODNs were seen inside CaSki cells when ODNs were used at the highest concentration (1.0 μM). By contrast, at lower concentrations (0.1 and 0.2 μM), ODNs were released from intracellular vesicles into the cytosol and carried more rapidly into the perinuclear area. This suggests that ODNs are released more rapidly from intracellular vesicles into the cytosol if the oligo-liposome complex has a positive net charge, as shown with model membranes (Jääskeläinen et al., 1994). A recent study by Zabner et al. (1995) showed that plasmid DNA remained in intracellular vesicles when cationic liposomes were used. However, only one DNA/ liposome ratio (1/5, w/w), found optimal in previous studies, was investigated at the ultrastructural level. Another study (Zhou and Huang, 1994) has shown that DNA is released into the cytosol after destabilization of endosomes. Our results do not permit direct comparison with these results because of differences in cationic liposomes and DNA size. These two factors can affect the physicochemical characteristics and the intracellular fate of nucleic acid-liposome complexes.
ODNs were delivered more rapidly into cells with DDAB/DOPE than with DOSPA/DOPE. At lower ODN concentrations (0.1 and 0.2 μM), DDAB/DOPE delivered ODNs more effectively than DOSPA/DOPE did, although no remarkable differences occurred at higher concentration. Because our immmunocytochemical method is only semiquantitative, no firm conclusions can be drawn about the amount of intracellular ODNs. One explanation for the difference in delivery effectiveness may be that the amount of the helper lipid DOPE was lower in the DOSPA/DOPE preparation than in the DDAB/DOPE preparation. According to our measurements, particles were smaller in DOSPA/ DOPE than DDAB/DOPE. The small particle size (<5 nm) has been reported to correspond with micelles rather than with liposomes (Solodin et al., 1995). Fusogenic DOPE is needed for stabilizing most types of cationic lipids in a lipid bilayer and may affect cell penetration function (Farhood et al., 1995). Previous studies have shown that a DNA/liposome complex containing DOPE destabilizes endosomes, allowing the complexes to escape into cytosol (Farhood et al., 1995; Zhou and Huang, 1994). According to the manufacturer, DOSPA/DOPE is up to 30-fold as effective as conventional monocationic liposomes (including DDAB/DOPE) in the transfection of plasmid DNA (Hawley-Nelson et al., 1993). Again, the length of nucleic acids (oligonucleotide vs plasmid DNA) may explain the difference between results. According to Hawley-Nelson et al. (1993), the cell type also contributes to the difference between DOSPA/DOPE and monocationic liposomes.
Our EM study showed no ODNs inside CaSki cells incubated without cationic liposomes. However, the weak immunoreactivity in CaSki cells detected in light microscopy after incubation with ODNs but without liposomes suggests that some non-liposomal ODNs can be taken up by CaSki cells. Recently, a study at the ultrastructural level demonstrated non-liposomal ODNs in intracellular vesicles, indicating that the cellular uptake of ODNs also depends on endocytosis (Beltinger et al., 1995; Tarrasón et al., 1995).
Using EM, our study is the first to show that the nuclear envelope can form a barrier against the penetration of liposomal ODNs into the nucleus of CaSki cells. This might be explained by the persisting association of ODNs with liposomes after release into the cytosol (Singhal and Huang, 1994), and the complexes as such may enter the nucleus (Friend et al., 1990). This may cause the size-limited entry of ODN- liposome complexes into the nucleus. Some ODNs were located in the nucleoplasm when the oligo- DDAB/DOPE complexes were positively charged. Instead, ODNs could not penetrate through the nuclear envelope into the nucleoplasm if DOSPA/DOPE was used, even when the charge ratio of ODN-DOSPA/DOPE was the same as that of the oligo-DDAB/DOPE complex (0.32). To confirm that the anti-DIG antibody could also detect any nuclear signals, we used several freeze-thawing steps as well as Triton X-100 to facilitate antibody penetration. Taken together, our results suggest that not only the net charge of the complex but also the type of the liposome influences the penetration of ODNs into the nucleus, at least in CaSki cells. This can be a limitation in the anti-gene technique, in which ODNs have to be transported into the nucleus before the biological effects of ODNs may be more easily applied to the anti-sense technique, in which gene expression can be controlled in the cytoplasm during the translation process.
Only a few studies have investigated the nuclear localization of ODNs at the EM level (Beltinger et al., 1995; Tarrasón et al., 1995; Zamecnik et al., 1994). Two of these studies (Beltinger et al., 1995; Zamecnik et al., 1994) have shown successful entry of ODNs into the nucleus. Our results are in agreement with those of Tarrasón et al. (1995), who found only a small proportion of ODNs located in the nucleus. The results of these studies are difficult to compare because not only different detection methods and carriers but also different cell types were used. The digoxigenin label was used in our experiments because it is not present in most mammalian cells (or tissues) and therefore does not lead to unspecific signals which may occur with “natural” labels, such as biotin (Höltke et al., 1992). Recently, digoxigenin-labeled oligonucleotides have been used to study the cellular uptake of ODNs (Plenat et al., 1995; Tarrasón et al., 1995; Yee et al., 1994). Furthermore, Tarrasón et al. (1995) showed that the intracellular distribution of digoxigenin-labeled ODNs is similar to that of fluo-rescein isothiocyanate-labeled ODNs, which are widely used in AS-ODN experiments.
Because the intracellular distribution may vary according to the cell type, each delivery system should be tested in the cells that would be the target cells in vivo. The CaSki cells used in the present study are known to contain human papillomavirus (HPV) DNA. Several molecular biological and epidemiological data have, in turn, provided evidence for the role of HPV infection in cervical cancer (Lowy et al., 1994; zur Hausen, 1991). Because HPV is the ideal target for local liposomal oligonucleotide-based therapy, the findings made in the present study are useful, especially when specific anti-sense or anti-gene therapies are designed against HPV infections.
