Abstract
We have recently shown the presence of E-cadherin and of α- and γ-catenins in human and baboon corpora lutea. These are components of adherens junctions between cells. The cytoplasmic catenins link the cell membrane-associated cadherins to the actin-based cytoskeleton. This interaction is necessary for the functional activity of the E-cad-herins. Our aim therefore was to determine the presence of α-actin in the baboon corpus luteum, to further establish whether the necessary components for E-cadherin activity are present in this tissue. An antibody specific for the smooth muscle isoform of actin, α-actin, was used for these studies. The results using immunohistochemistry show that (a) α-actin is present in steroidogenic cells of the active corpus luteum, theca externa of the corpus luteum, cells of the vasculature, and the tunica albuginea surrounding the ovary. The intensity of immunoreactivity for α-actin varied, with the cells of the vasculature reacting more intensely than the luteal cells. A difference in intensity of immunoreactivity was also observed among the luteal cells, with the inner granulosa cells showing stronger immunoreactivity than the peripheral theca lutein cells. There was no detectable immunoreactivity in the steroidogenic cells of the atretic corpus luteum. However, in both the active and atretic corpora lutea, α-actin-positive vascular cells were dispersed within the tissue. (b) Total α-actin (luteal and non-luteal), as determined by Western blot analyses, does not change during the luteal phase and subsequent corpus luteum demise (atretic corpora lutea). (c) hCG stimulated the expression of α-actin and progesterone secretion by the early luteal phase (LH surge + 1–5 days) and midluteal phase (LH surge + 6–10 days) cells in culture, but only progesterone in the late luteal phase (LH surge + 11–15 days). The data show that α-actin is present in luteal cells and that its expression is regulated by hCG, thus suggesting that E-cadherin may form functional adherens junctions in the corpus luteum.
Keywords
T
The function of the cell adhesion- and tight junction-related molecules is determined by their association with the cytoplasmic cytoskeletal proteins, particularly actin. These interactions are important in the organization of the cytoskeleton, regulation of adhesion between cells, signal transduction, cell motility (Gumbiner, 1993; Loewenstein, 1981; Farquhar and Palade, 1963), and regulation of steroidogenesis (Hall and Almahbobi, 1992). Although the cytoskeletal components, such as the microfilaments, intermediate filaments and microtubules, have been extensively examined in rat granulosa cells (Amsterdam and Rotmensch, 1987), little is known about the presence or regulation of the cytoskeletal components in the human or subhuman primate corpus luteum.
We therefore sought to determine (a) the presence of α-actin in the baboon corpora lutea of the menstrual cycle, (b) the levels of the protein through various stages of corpus luteum development, and (c) the effect of hCG on actin expression and progesterone production in cultured luteal cells.
Materials and Methods
Tissues
Baboon ovaries and corpora lutea were obtained from female cycling baboons (Papio anubis) with well-defined menstrual cycles and weighing 14–17 kg. The study was approved by the Review Board for Animal Experimentation, University of Texas Health Science Center at Houston, and was carried out in accordance with the principles and procedures described in the guide for the care and use of laboratory animals as approved by the National Institutes of Health (NIH 80–23). The baboons were kept in individual cages and in air-conditioned rooms. They were fed monkey chow and water ad libitum. Their menstrual cycles were monitored daily by the appearance and score of their perineal tumescence and serum progesterone after deturgescence (Khan-Dawood et al., 1988). The day of perineal deturgescence was equated to the day of the LH surge and thus to corpus luteum development. Days LH surge + 1–5 days, LH surge + 6–10 days, and LH surge + 11–15 days were designated as the early, mid-, and late luteal phases.
Under general anesthesia induced with ketamine HCl (10–12 mg/kg body weight) and maintained with endotracheal intubation and nitrous oxide:oxygen 1:1, by volume with 3–4% halothane as necessary, a laparotomy was carried out through a lower abdominal midline incision. The ovaries were examined to locate the corpus luteum. A lutectomy was performed as previously described (Khan-Dawood et al., 1988). A total of 15 corpora lutea were obtained for the present set of experiments (n = 5 in each phase) and a total of six ovaries were obtained by ovariectomy (n = 2 in each phase).
Chemicals
The antiserum for anti-human smooth muscle α-actin was obtained from Dako (Carpinteria, CA). The immunogen used to generate the monoclonal antiserum was against the N-terminal decapeptide of smooth muscle α-actin, clone 1A4 (Skalli et al., 1986) and is highly specific for this antigen. This antiserum reacts with smooth muscle cells of vessels and different parenchymes, but with different intensity according to the amount of smooth muscle α-actin present. The antibody does not react with actin from fibroblasts (β and γ-cytoplasmic) and striated muscle (α-sarcomeric) (Skalli et al., 1986). The secondary antibody, goat anti-mouse IgG, an affinity-purified antibody, was obtained from Transduction Laboratories (Lexington, KY). Dulbecco's modified Eagle's medium (DMEM), Ham's F-12 nutrient medium, and Hepes (N[2-hydroxyethyl] piperazine-N 1[2-ethanesulfonic acid]) were obtained from Gibco Laboratories (Grand Island, NY). Bouin's solution, sodium bicarbonate, collagenase, DNAse, culture grade bovine serum albumin (BSA), and fetal bovine serum (FBS) were obtained from Sigma (St Louis, MO). Matrigel and ITS+ premix (providing insulin 5.0 mg/liter, transferrin 5.0 mg/liter, selenic acid 5.0 μg/liter, and linoleic acid 5.23 μg/liter) were obtained from Collaborative Research (Lexington, MA). Human chorionic gonadotropin (hCG) with a potency of 9000 IU/mg was a gift from Serono Laboratories (Randolph, MA). 2,3,6,7[H3]-progesterone (specific activity 90–100 Ci/mmol) was obtained from Amersham (Arlington Heights, IL). Nonradioactive progesterone was obtained from Sigma. Reagents for electrophoresis were obtained from Bio-Rad (Richmond, CA).
Immunohistochemical Localization Procedures
Tissues fixed in Bouin's solution (24 hr at 22°C) were dehydrated and embedded in Surgipath paraffin (Surgipath Medical Industries; Grayslake, IL) as previously described (Khan-Dawood, 1987). Five-μm paraffin sections of corpora lutea, cardiac muscle, and uterine myometrium were deparaffinized and immunostained. Briefly, the immunostaining consisted of incubation with normal horse serum for 10 min and 60 min each with the monoclonal primary antibody at 2 μg/ml (1:500) diluted in PBS (pH 7.2) containing 2% normal mouse serum, followed by the secondary antibody (1:250 dilution). 3,3'Diaminobenzidine tetrahydrochloride dissolved at 0.5 mg/ml in 0.05 Tris-HCl, pH 7.6, containing 3.3 μl of 3.0% hydrogen peroxide was used as the chromogen, which was detected as brown reaction products. Endogenous peroxidases were removed by treatment with 2% hydrogen peroxide in 0.05 M Tris-HCl buffer, pH 7.6. Sections were stained with hematoxylin (Gill et al., 1974). Negative controls for immunostaining were performed by substituting the primary antibody with PBS or preimmune serum. Positive controls consisted of uterine tissue and cardiac tissue with known immunoreactivity. These were treated similarly to the test tissues.
Western Analyses
Analysis of Tissue Concentrations of α-actin. Western blot analyses were performed as described (Khan-Dawood et al., 1996b) according to Laemmli (1970). Briefly, luteal and non-luteal tissues were homogenized in 5 volumes of sucrose-3-N-morpholino-propane sulfonic acid buffer (MOPS), pH 7.2, consisting of 250 mM MOPS, 20 mM sucrose, 1 mM phenylmethyl sulfonyl fluoride, 2 mM dithiothreitol, 2 mM CaCl2, and 20 μg/ml trypsin inhibitor, using a polytron P-10 homogenizer (Tekmar; Cincinnati, OH). After centrifugation at 1000 × g for 10 min, the supernatant was re-centrifuged at 10,000 X g for 60 min in a Beckman L7 ultracentrifuge (Beckman Instruments; Palo Alto, CA). The pellet was resuspended in sample buffer consisting of 0.5 M sucrose, 15% sodium dodecyl sulfate (SDS), 0.3 M Tris, and 0.01 M EDTA. After protein determination (Lowry et al., 1954), 25 μg of the supernatant protein was resolved by electrophoresis on a 12% SDS separating gel and a 4% stacking gel. Molecular weight markers used were prestained SDS-PAGE standards. After transfer onto nitrocellulose membranes (Hybond-C Super; Amersham) in a Bio-Rad apparatus at 90 V for 60 min, the membranes were treated with 5% dry milk in PBS (pH 7.4) for 60 min at room temperature and incubated with the α-actin antibody at a dilution of 1 μg/ml (1:1000 dilution) for 16 hr at 4°C. The reaction products were detected using the Renaissance chemiluminescence procedure essentially as described by the manufacturer (Dupont NEN; Boston, MA).
Effect of hCG on Expression of α-actin
Luteal cells from each phase (early, mid-, and late) of corpus luteum development were cultured for 4 days as previously described (Khan-Dawood et al., 1988). Tissue was dispersed using collagenase (1.0 mg/ml), DNAse (0.015 mg/ml), BSA (1 mg/ml), and Hepes (4.7 mg/ml) for 2 hr at 37°C in a shaking water bath. Cell viability was determined by the Trypan blue exclusion test (Tennant, 1964). Cells (100,000/well) were plated in culture dishes coated with the basement membrane (Matrigel) diluted 1:3, 40 μl/well, cultured at 37°C in 5% CO2 and air, in DMEM and Ham's F-12 (1:1 v/v) with added ITS+ premix (providing insulin at 5.0 μg/liter transferrin at 5.0 μg/liter, selenic acid 5.0 μg/liter, and linoleic acid 5.23 μg/liter) containing 10% fetal bovine serum (FBS). After 24 hr the medium was replaced with DMEM and Ham's F-12 (1:1 v/v) without FBS and the cells were cultured for a further 24 hr. Cells were treated with hCG (10 IU/mL) for 2 days, with medium being replaced every 24 hr. The cells were trypsinized (0.05% trypsin: EDTA), an aliquot was removed for protein determination (Lowry et al., 1954), and the remainder were processed for electrophoresis.
The cells were lysed in buffer (O'Farrell, 1975) after trypsin treatment. The buffer contained 1 mM MgCl2, 0.01 U/ml aprotinin, 5 mM phenylmethyl sulfonyl fluoride, and 1 mM CaCl2. After sonication for 20 sec, samples were boiled for 5 min and 10-μg protein samples were electrophoresed on a 12% SDS polyacrylamide gel. After transfer to nitrocellulose membranes and reaction with the primary antibody at a dilution of 1 μg/ml (1:1000 dilution), the reaction products were detected as described above. Initial experiments determined the appropriate concentration of hCG to be used. Thus, 1, 10, 50, or 100 IU of hCG was added to midluteal phase cells (100,000/well) in FBS-free medium after 24 hr of treatment with FBS containing DMEM: Ham's F-12 (1:1, v/v) medium. Media concentrations of progesterone indicated that 10 IU of hCG was appropriate for these experiments.
Radioimmunoassay of Progesterone
Progesterone in the media was assayed in non-extracted samples by a previously described radioimmunoassay (Khan-Dawood et al., 1988). The antiserum was raised against progesterone-11-oxime-BSA conjugate. Insignificant crossreaction with 17α-hydroxyprogesterone, 20α-dihydro-progesterone, and corticosterone is a characteristic of this antibody. The sensitivity of the assay is 5 pg/tube and media blanks have no detectable progesterone. Inter- and intra-assay coefficients of variation are 6.0 ± 1.2% and 7.0 ± 0.5%, respectively. Progesterone production in ng/100,000 cells/24 hr was expressed as a mean of triplicate samples assayed.
Analyses of Data
The Western blots of five tissue samples in the early, mid-, and late luteal phase were analyzed by determining band intensities of the electrophoresed samples using a Phosphoimager Series 400 (Molecular Dynamics; Sunnyvale, CA) and Image Quant 3.2 software. The densitometric values obtained were statistically analyzed using a one-way ANOVA to compare samples from each stage of luteal development, followed by the Student's Newman-Keuls test for determination of significance. Differences were considered to be significant at p ≤ 0.001.
Results
Smooth muscle α-actin was localized in cardiac tissue (Figure 1), uterine tissue (not shown) used as positive controls, and ovarian tissue. In cardiac tissue in which the primary antibody was omitted (Figure 2) and in ovarian tissue that was treated with preadsorbed antiserum for the smooth muscle α-actin (Figure 3), no positive immunoreactivity was observed. Smooth muscle α-actin was detected at various intensities in the ovarian tissue. Intense immunoreactivity was observed in the theca externa (TE) surrounding the luteal cells as well as in the blood vessels (BV) that form a significant component of the corpus luteum. Comparatively lower levels of immunoreactivity were seen in the luteal cells, with the inner granulosa lutein cells (GC) showing stronger immunoreactivity compared to the peripheral theca lutein cells (TL) (Figure 4). The luteal cells of the early, mid-, and late corpus luteum (Figures 4-6) showed stronger immunoreactivity than the luteal cells of the atretic corpus luteum (CL) (Figure 7). The single layer of surface epithelial cells (SE) of the ovary did not show the presence of α-actin. However, a layer of cells immediately below the surface epithelium forming the tunica albuginea (TA) was strongly positive, and positive cells were present and dispersed in the ovarian stroma. The oocyte (O) and the surrounding follicular cells (F) were not positive for α-actin (Figure 7).
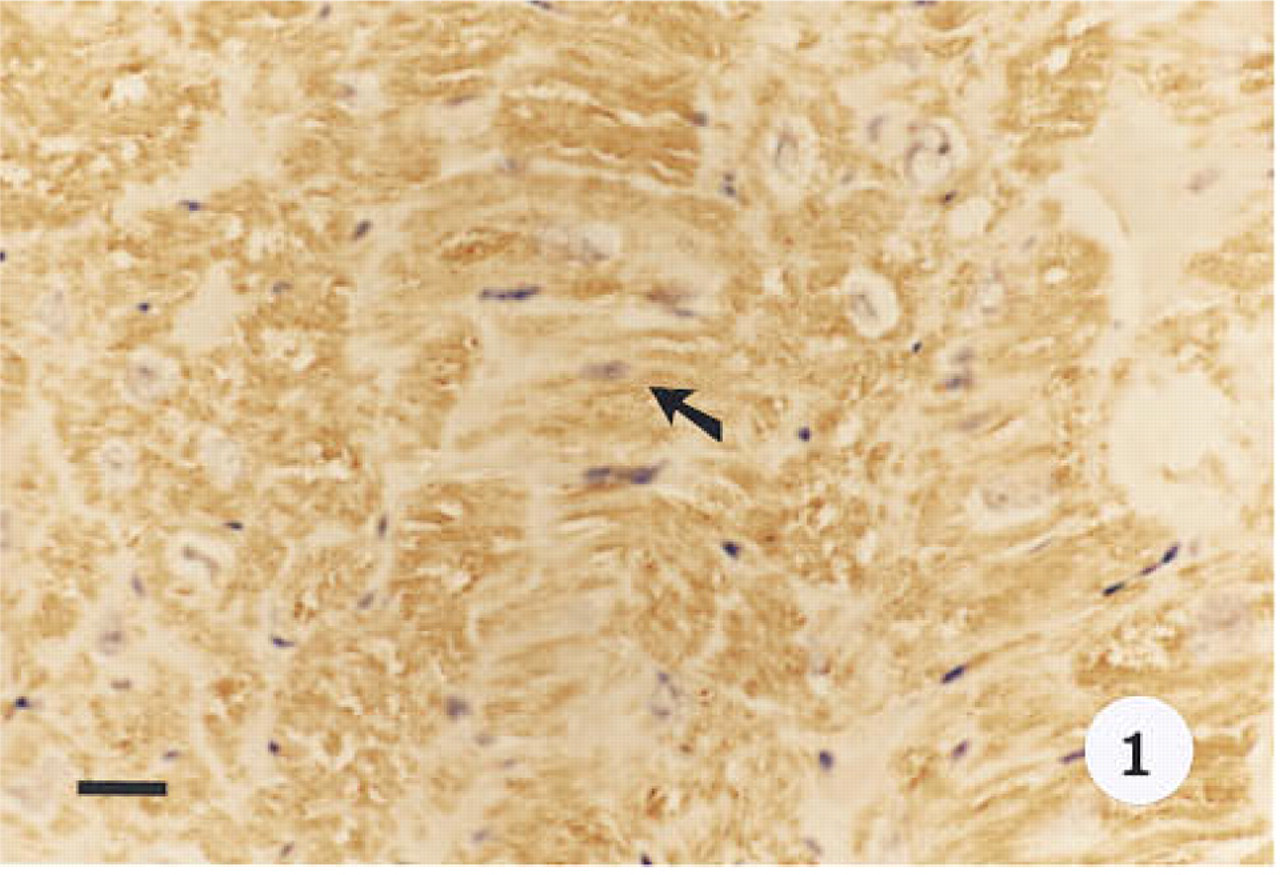
Immunohistological localization of smooth muscle α-actin in baboon cardiac tissue used as a positive control. Tissue was stained with hematoxylin. α-Actin is localized to the cytoplasm of the cells (arrow). Original magnification x 40. Bar = 20 μm.
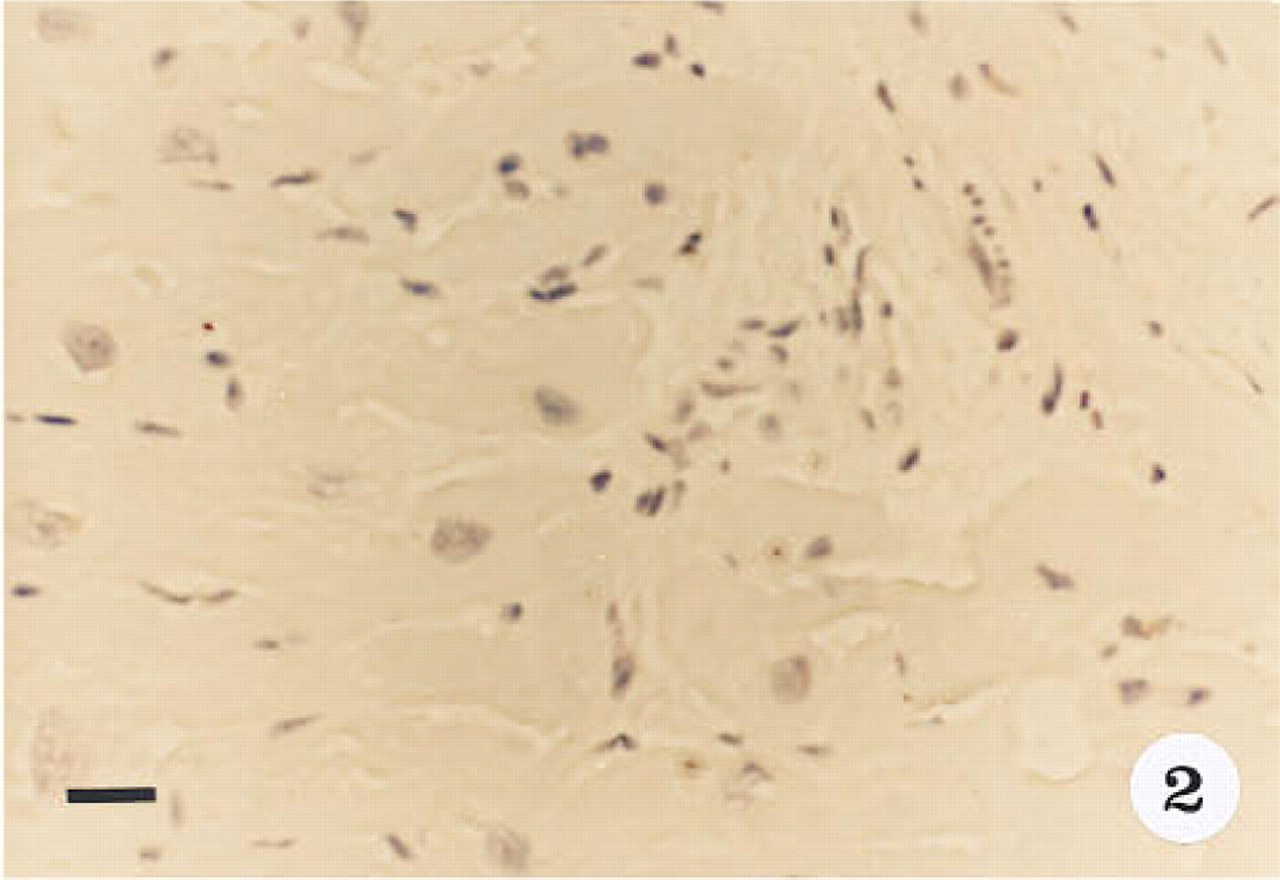
Cardiac muscle control in which the primary antibody was omitted. Sections stained with hematoxylin. Original magnification x 40. Bar = 20 μm.
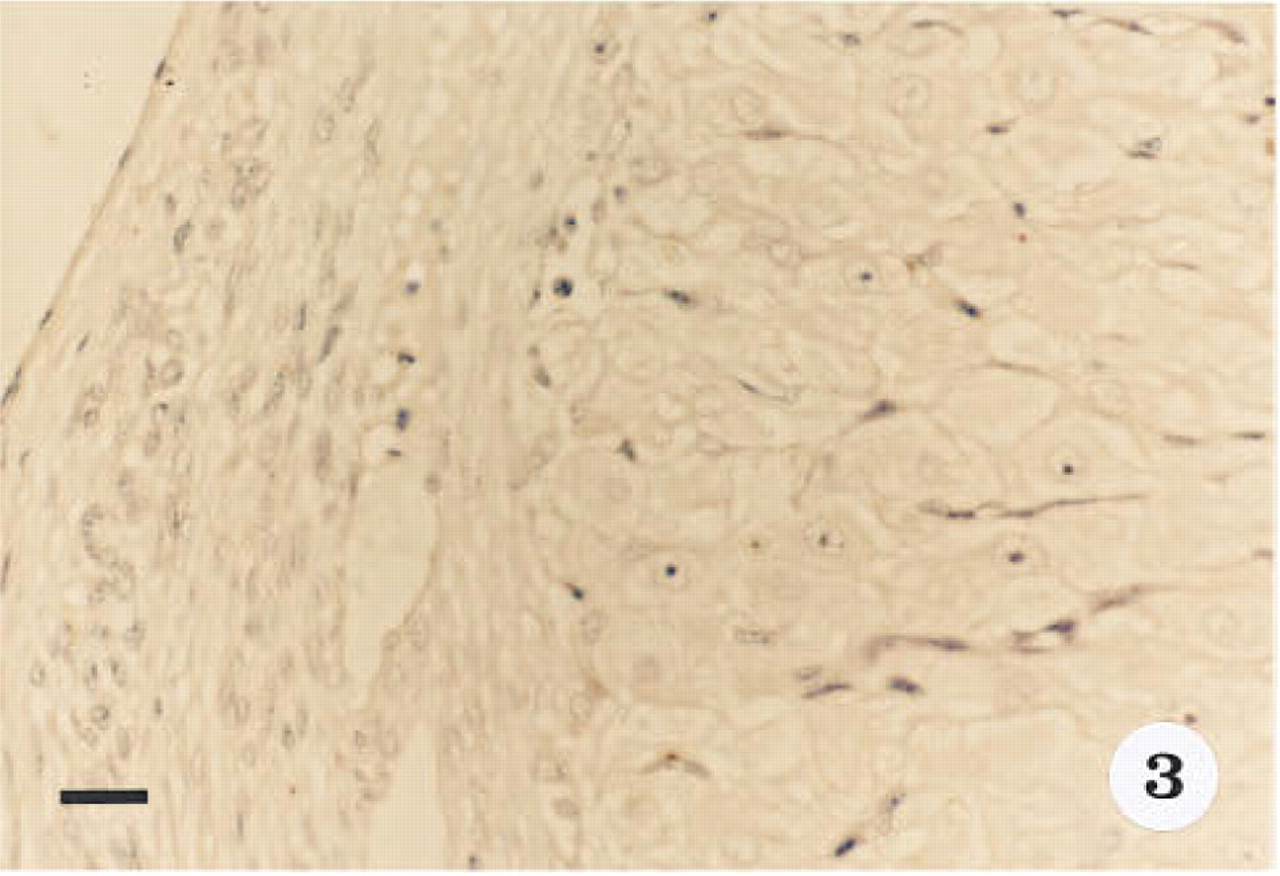
Immunoreactivity for smooth muscle α-actin is absent from corpus luteum tissue from the early luteal phase in which the primary antibody was preadsorbed with α-actin before tissue treatment. Section stained with hematoxylin. Original magnification x 40. Bar = 20 μm.
Comparison of levels of smooth muscle α-actin (densitometric units) in early, mid-, and late luteal phase corpus luteum cells cultured for 48 hr and progesterone levels in media (ng/100,000 cells).
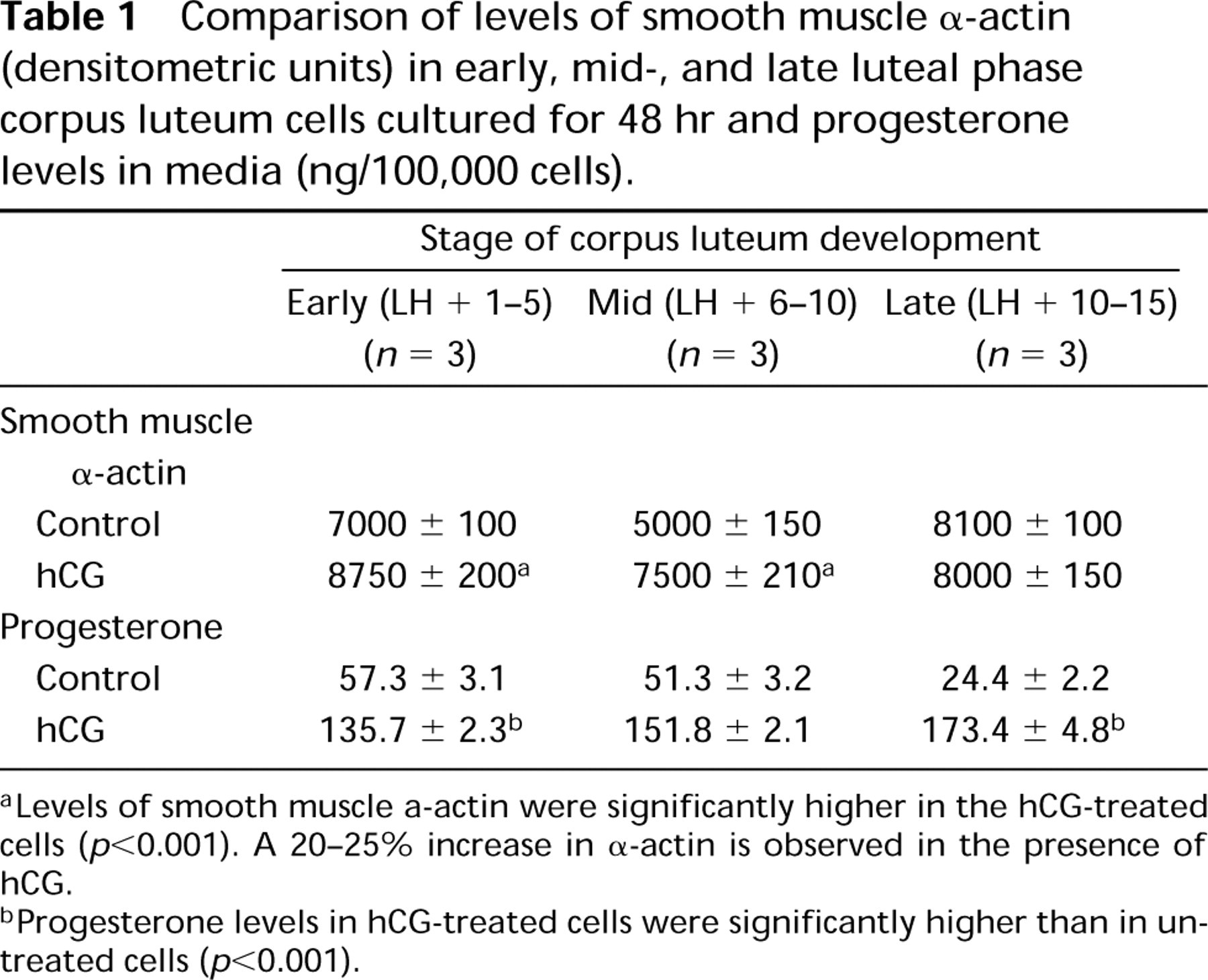
aLevels of smooth muscle α-actin were significantly higher in the hCG-treated cells (p<0.001). A 20–25% increase in α-actin is observed in the presence of hCG.
bProgesterone levels in hCG-treated cells were significantly higher than in untreated cells (p<0.001).
Western blot analyses of 25 μg of the membrane fraction indicated that the overall level of protein in the early, mid-, late and atretic corpora lutea does not change significantly (Figure 8, Lanes A-D). The level of α-actin in luteal tissue was less than in cardiac tissue (Figure 8, Lane E). Cultured luteal cells in all three stages showed the presence of α-actin (Figures 9a-9c, Lane A). Examination of the effect of hCG on the expression of the protein indicated that hCG stimulates the expression of α-actin in the cells of the early (Figure 9a, Lanes B and C) and mid-luteal (Figure 9b, Lanes B and C) phase cells but not those of the late luteal phase (Figure 9c, Lanes B and C; Table 1). Under the experimental conditions, a 20 ± 3% increase in the early luteal phase and a 22 ± 4% increase in the mid-luteal phase, both of which were significantly different from the control, were observed. However, progesterone levels were significantly increased by hCG in all three luteal stages (Table 1).
Discussion
In the ovary, smooth muscle α-actin was differentially expressed in the various compartments of this organ. The high level of α-actin in the tunica albuginea, the walls of the blood vessels, and the theca externa surrounding the corpus luteum is probably involved in the contractile function of this protein. Both actin and myosin have been identified in the ovary of the rat (Callebaut and Van Nassauw, 1987; Amsterdam et al., 1977). The intermediate filament protein desmin is present in abundance in the arteries around the corpus luteum and the vasculature of the ovary (Selstam et al., 1993; Wiltbank et al., 1990; Callebaut and Van Nassauw, 1987). Therefore, it is likely (Wiltbank et al., 1990) that these proteins are involved in the regulation of ovarian vascular resistance.
The cytoskeletal protein actin has been extensively examined in the differentiation of both the rat and human granulosa cells (Kranen et al., 1993; Amsterdam et al., 1989; Ben-Ze'ev, 1990; Ben-Ze'ev and Amsterdam, 1986, 1987, 1989; Carnegie and Tsang, 1988; Amsterdam and Rotmensch, 1987; Ben-Ze'ev et al., 1987), but little is known about its presence or role in the cells constituting the corpus luteum. In our study, smooth muscle α-actin was localized in the steroidogenic cells of the corpus luteum at different intensities, suggesting different levels of the protein in these cells. Thus, the inner region of the corpus luteum, composed mainly of granulosa lutein cells that form the “large cell” population of the corpus luteum parenchyma, appears to have higher levels of α-actin. In contrast, the surrounding layer of theca lutein cells, which form the “small cell” population, have lower levels of this protein. In most species examined, only one cell type responds to endocrine regulators. In the human and the baboon (Khan-Dawood et al., 1996a), only the small cells respond to LH by increasing progesterone secretion several-fold, but both cell types secrete progesterone under basal conditions. That the large and small cells in contact in culture secrete significantly more progesterone than when they are cultured separately (Khan-Dawood et al., 1996a) indicates or suggests that “communication” or “synergy” must occur between the cells for enhanced secretory capacity of these cells. Several paracrine/autocrine factors have been implicated in this communication process. Gap junctions have been recognized in the corpus luteum (Khan-Dawood et al., 1996b; Redmer et al., 1991) and bovine luteal cells are able to communicate via these junctions (Redmer et al., 1991). We have recently shown the presence of gap junctions and of adherent and tight junctions in the corpora lutea of women and baboons (Khan-Dawood et al., 1996b,c,d) and have suggested that communication between the cells of the corpus luteum may occur via these junctions as in the follicular granulosa cells (Amsterdam and Rotmensch, 1987). The presence in luteal cells of α-actin, which is an integral component of these junctions (Madara, 1988; Hirano et al., 1987), further supports this hypothesis.
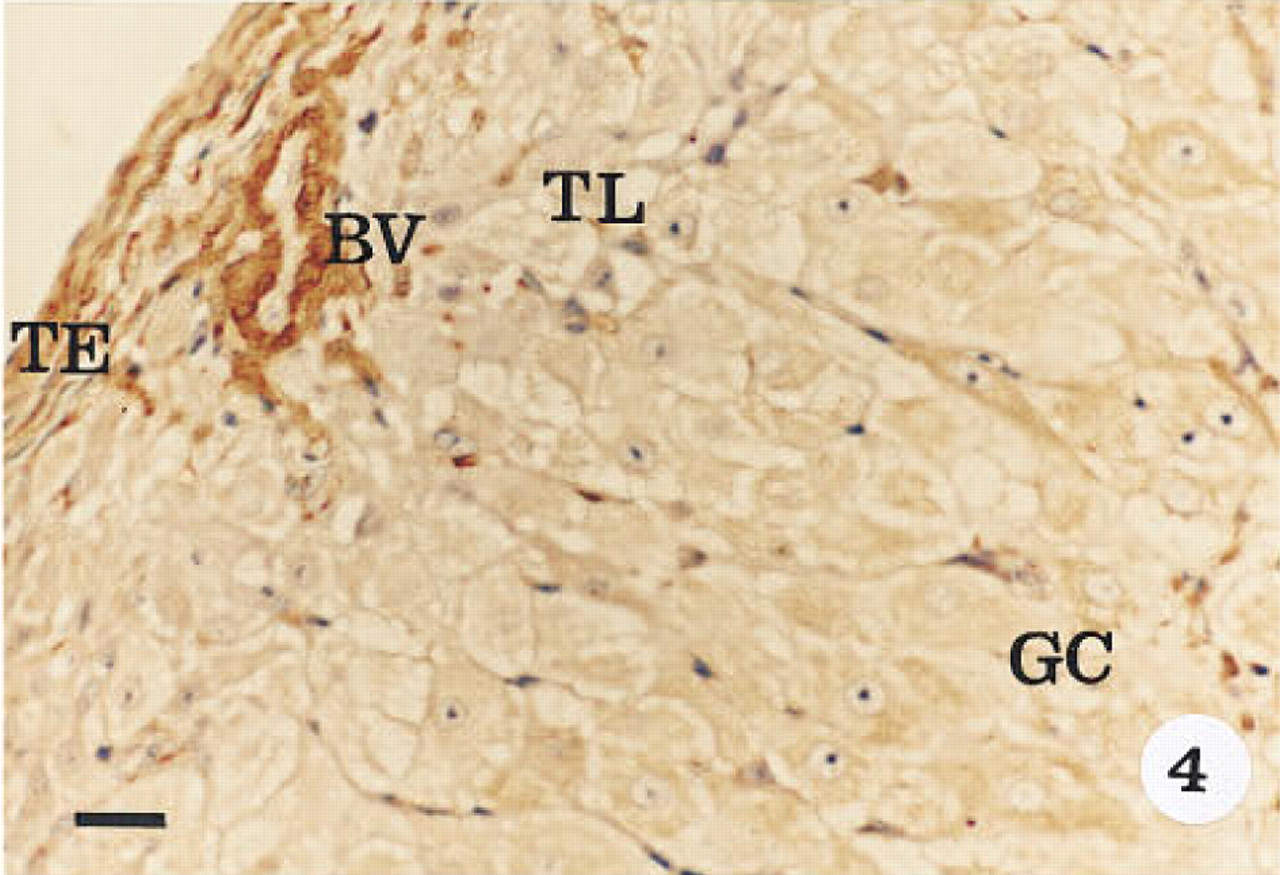
Immunohistological localization of α-actin in an early corpus luteum (LH surge + 1–5 days). Tissue was stained with hematoxylin. Intense immunoreactivity is observed in the theca externa (TE) and the blood vessel walls (BV). Immunoreactivity is also observed in the peripheral theca lutein (TL) and inner granulosa lutein cells (GC). The immunoreactivity in the granulosa lutein cells is more intense than in the theca lutein cells. Original magnification x 40. Bar = 20 μm.
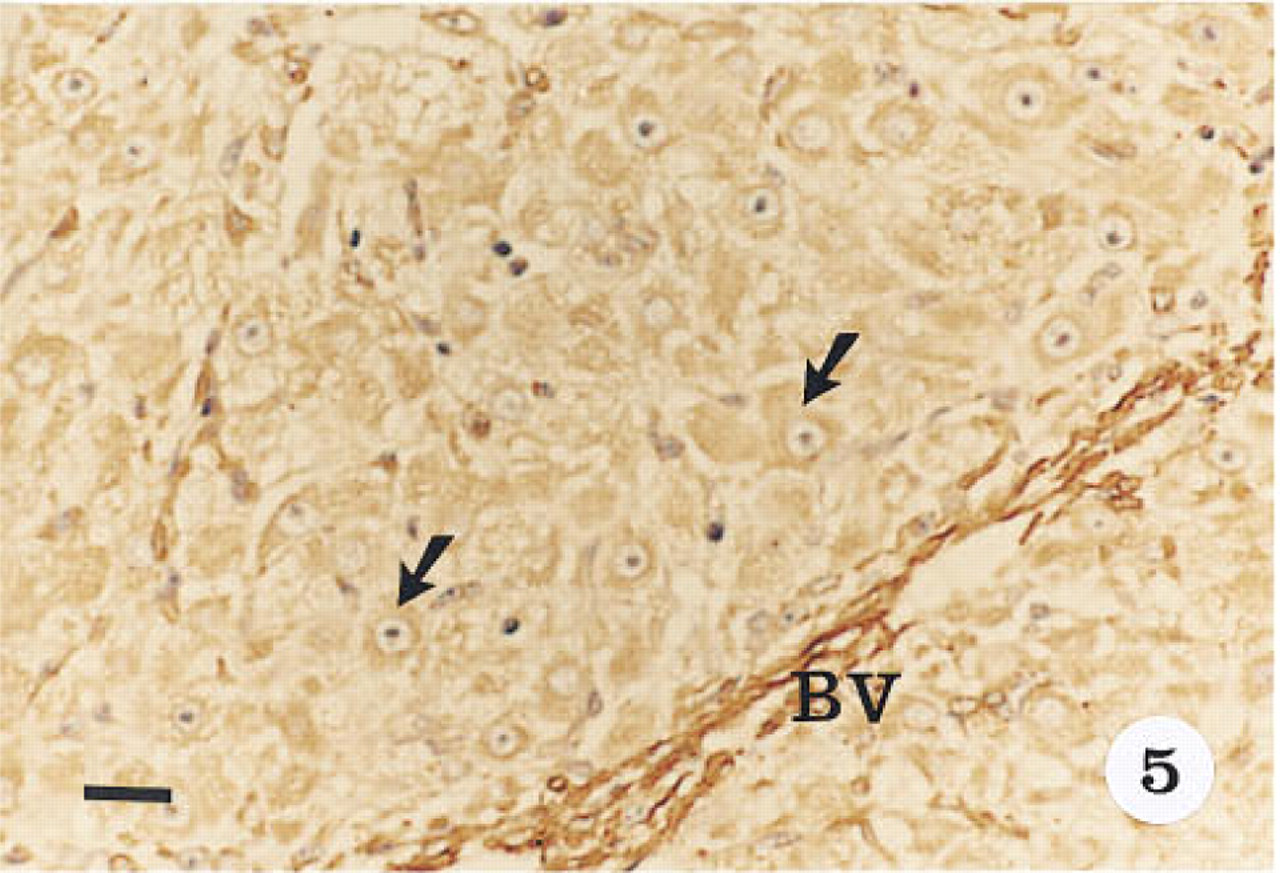
Immunohistological localization of α-actin in the steroidogenic parenchyma of the mid-luteal phase corpus luteum. Tissue was stained with hematoxylin. Immunoreactivity is localized to the cytoplasm of the luteal cells (arrows). Cells of the blood vessels (BV) reacted more intensely than the luteal cells. Original magnification x 40. Bar = 20 μm.
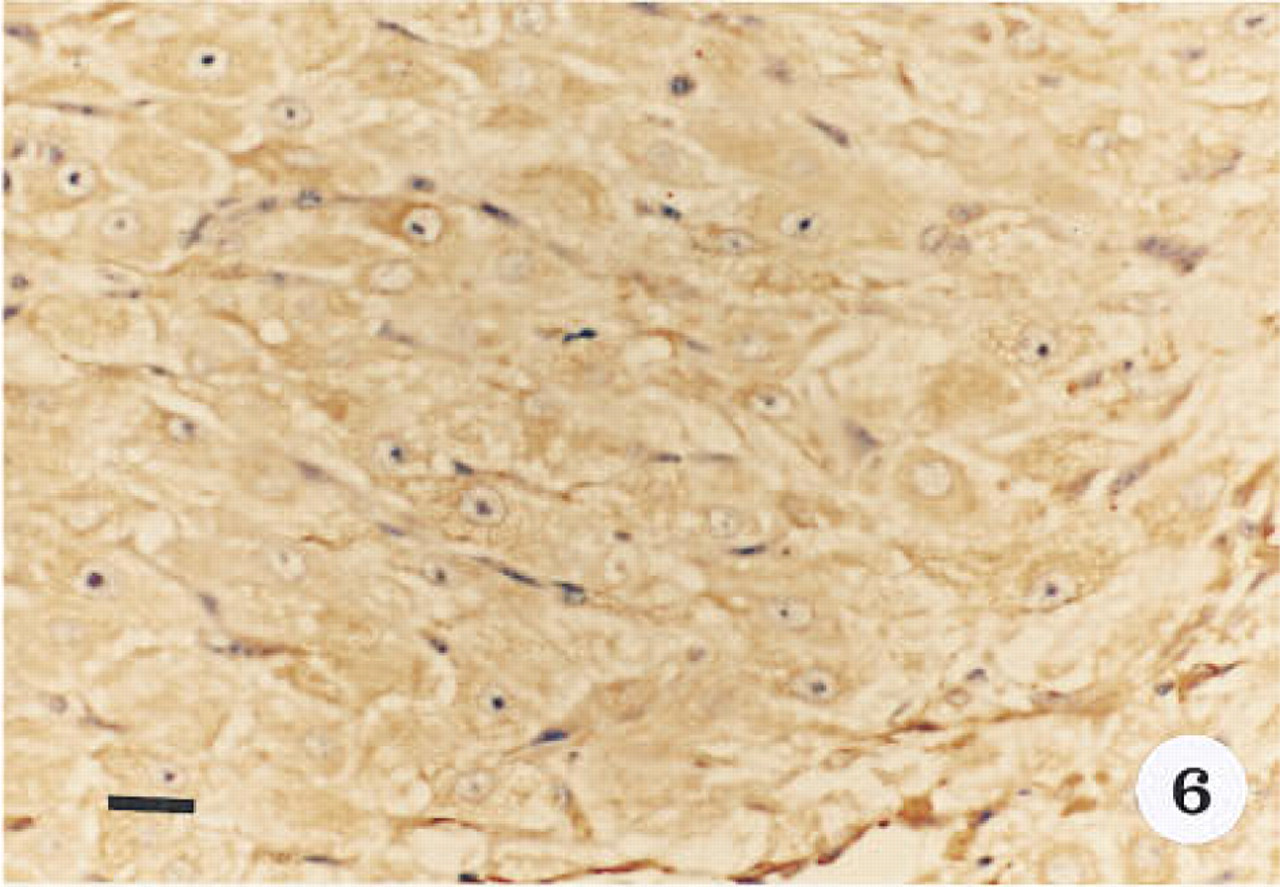
Immunohistological localization of α-actin in the late luteal phase corpus luteum, showing the cytoplasmic immunoreactivity in the inner granulosa lutein cells. Tissue was stained with hematoxylin. Original magnification x 40. Bar = 20 μm.
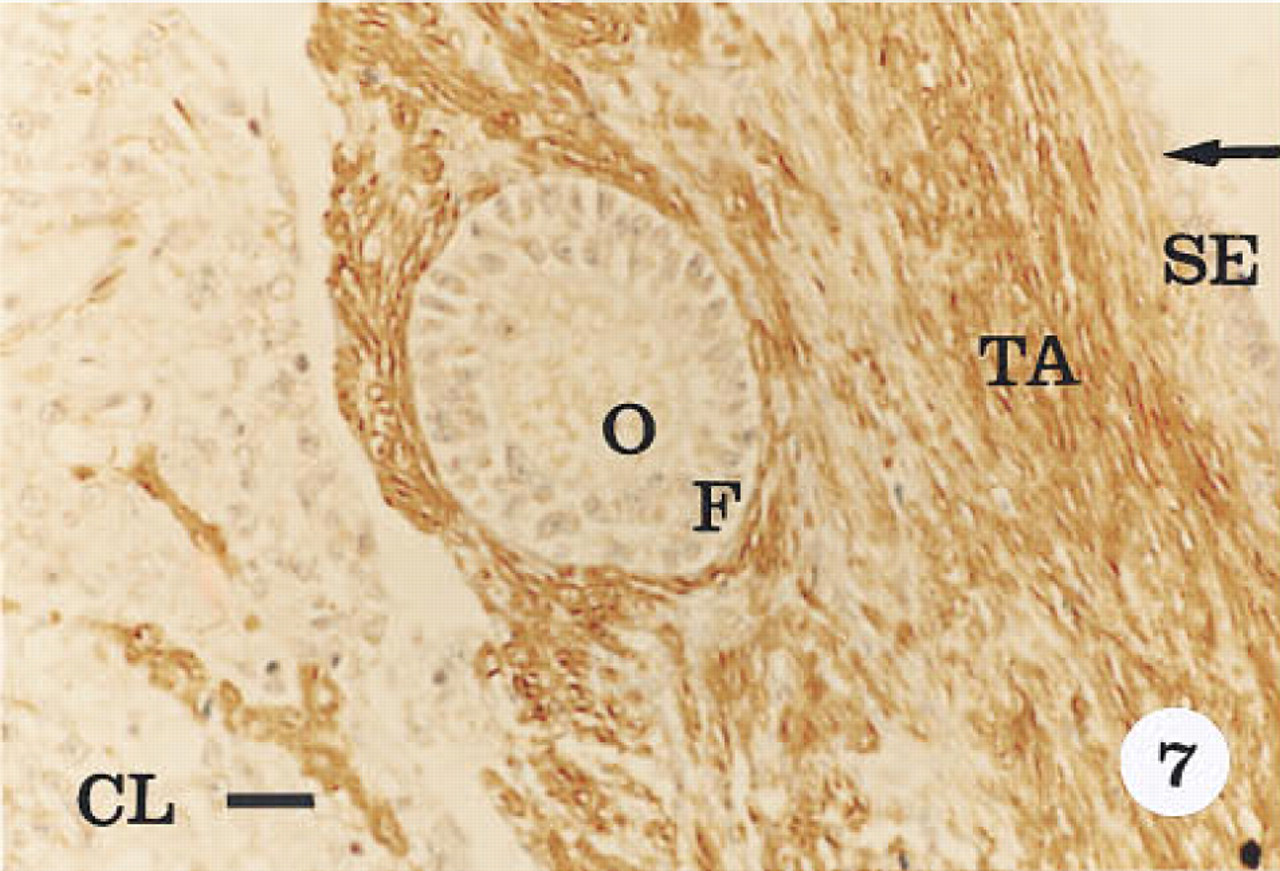
Immunohistological localization of α-actin in the atretic corpus luteum (CL). Tissue was stained with hematoxylin. The intensity of immunoreactivity is less compared to the early (Figure 4), mid-luteal (Figure 5), and late luteal phase corpus luteum (Figure 6). Immunoreactivity is not present in the surface epithelium (SE), oocyte (O), or the follicular cells (F) surrounding it. The tunica albuginea (TA) is positive for α-actin. Original magnification x 40. Bar = 20 μm.
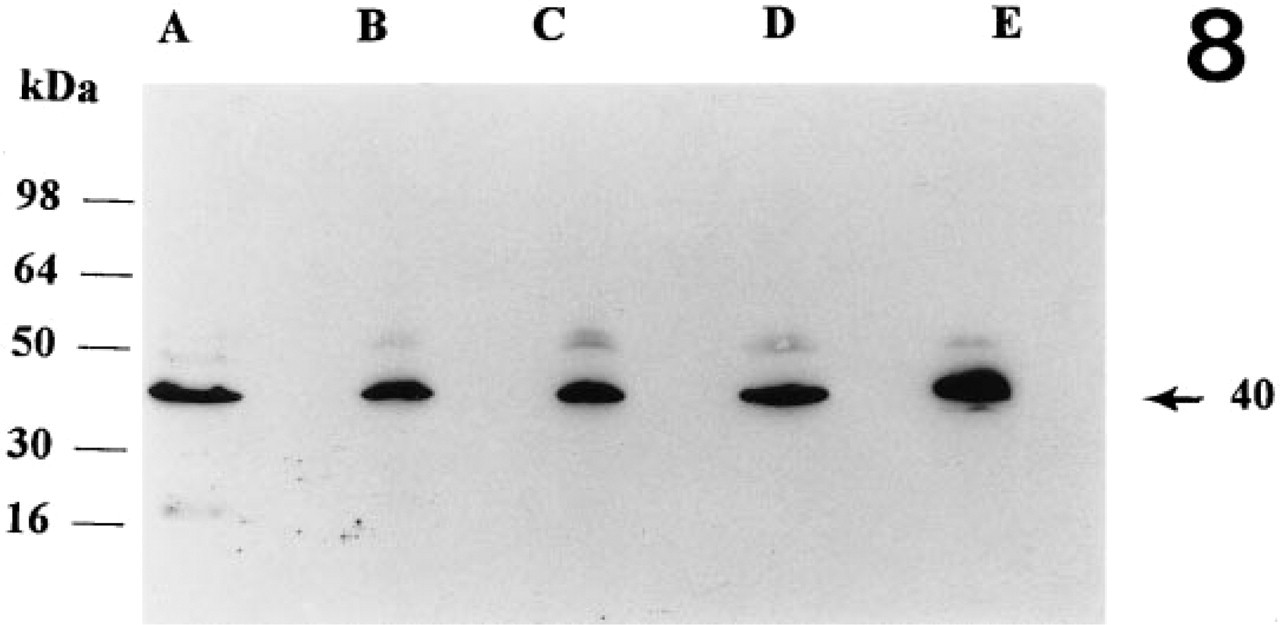
Western analyses of 25 μg of protein from baboon corpora lutea. Lane A, early luteal phase corpus luteum (LH surge + 1–5 days); Lane B, mid-luteal phase corpus luteum (LH surge + 6–10 days); Lane C, late luteal phase corpus luteum (LH surge + 11–15 days); Lane D, atretic corpus luteum; Lane E, baboon cardiac tissue. Densitometric analysis did not show significant differences between tissues examined.
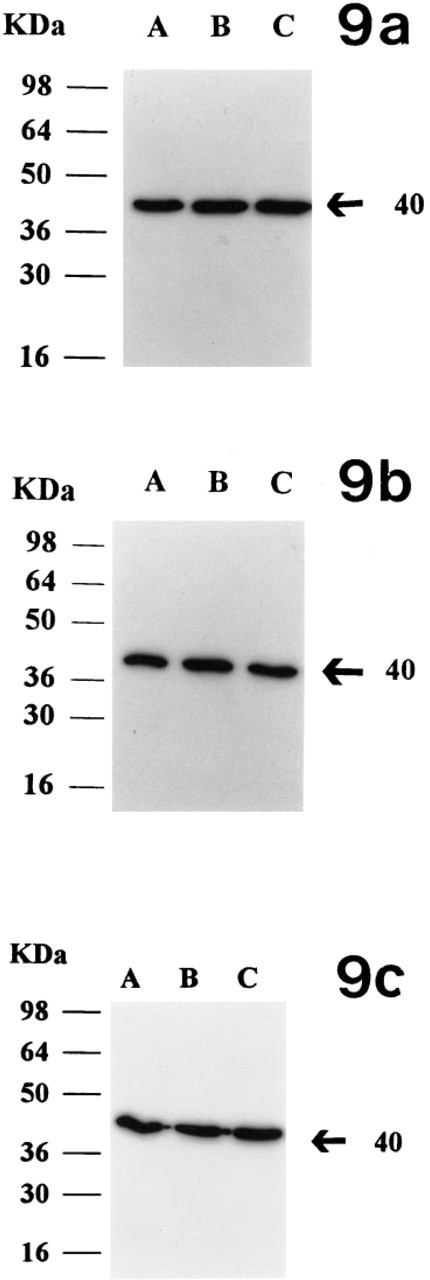
Examination of the effect of hCG (10 IU) on expression of α-actin in luteal cells in culture (for details see text). Lane A, expression without hCG; Lanes B and C (duplicates), expression with 10 IU of hCG. (
The total actin levels in the corpus luteum do not appear to change during luteal development. However, α-actin in the steroidogenic luteal cells is stimulated by LH/hCG, as reflected by increased α-actin levels from early and mid-luteal phase cultured luteal cells in the presence of hCG. hCG also stimulated progesterone production in these cells. However, hCG failed to significantly increase α-actin by luteal cells from the late luteal phase, in spite of increased progesterone output.
In summary, we have demonstrated the presence of α-actin in the ovary and in various stages of corpus luteum development in the baboon. The differential immunoreactive pattern observed suggests different levels of the protein in the various compartments of the ovary. The presence of the peptide in the luteal cells suggests that previously identified junctional proteins may be functional in this tissue and that hCG/LH may regulate its expression.
Footnotes
Acknowledgements
Supported in part by a National Institutes of Health Grant HD24928.
