Abstract
Galectin-1 and galectin-3, β-galactoside–binding lectins, are predominantly expressed in the regressing corpus luteum (CL) of mouse ovary. This study revealed the expression patterns and cellular localizations of galectins during CL formation and regression by ISH and IHC. Galectin-1 mRNA expression temporarily increased in active CL, preceding the expression of progesterone degradation enzyme 20α-hydroxysteroid dehydrogenase (20α-HSD), which represents functional luteolysis. The expressions of both galectin-1 and galectin-3 remarkably increased in the structurally regressing CL, which vigorously expressed 20α-HSD and contained abundant apoptotic luteal cells. Ultrastructurally, galectin-1– and galectin-3–immunoreactive cells were identified as fibroblasts and infiltrating macrophages, respectively. In addition, some populations of luteal cells themselves expressed galectin-3 in regressing CL and formed unique demarcation membranes in the cytoplasm, showing a non-typical apoptotic feature. Ovary of adult mice with repeated estrus cycles contained CL of three different generations. Among them, the old CL formed during previous estrus cycles consisted of galectin-3–positive luteal cells. The galectin-3–positive old CL was resistant to apoptosis and seemed to be eliminated by a mechanism different from apoptosis. The stage- and cell-specific expression of galectin in CL suggests its differential contribution to luteolysis, and this expression may be mediated by major regulatory molecules of CL function, prolactin and/or prostaglandin F2α.
G
To clarify the involvement of galectin in CL function and the regulatory mechanism of galectin expression in CL, the cellular and subcellular expressions of galectin throughout the estrus cycle are essential. Recently, Kim et al. (2008) and Lohr et al. (2008) reported that galectin-3 was predominantly expressed in regressing CL of cows and mice, but the cellular localization of galectins, especially of galectin-1, in CL is still unclear. In this study, we demonstrated the differential localization of galectin-1 and galectin-3 in regressing CL of mice at mRNA and protein levels, with special reference both to the expression of receptors for prolactin (PRL) and prostaglandin (PG) F2α, which are the main regulatory molecules of CL function, and to the involvement of galectin in luteal cell elimination.
Materials and Methods
Animals and Tissue Sampling
Female ddY mice were purchased from Japan SLC (Shizuoka, Japan). Mice were maintained under controlled light (12 hr light/dark, lights on at 8:30 AM) and controlled temperatures (20–22C) and had free access to chow and tap water. All animals were treated according to the laboratory animal control guidelines of Hokkaido University (Approval number 08-0054), which conform to the Guide for Care and Use of Laboratory Animals of US Institute for Laboratory Animal Research.
Ovulation was compulsorily started by hormonal induction at 4 weeks postpartum, before the first natural ovarian cycle. To stimulate follicular growth, 5 IU of equine chorionic gonadotropin (eCG; Serotropin; ASKA Pharmaceutical, Tokyo, Japan) was injected SC and followed by 5 IU of human chorionic gonadotropin (hCG; Gonatropin; ASKA Pharmaceutical) after 48 hr (day 0). In the morning from the next day to the fourth day after hCG injection (days 1–4), mice were killed by draining blood from the heart, and fresh ovaries were collected. Mature females at the age of 8 weeks were treated with the same procedure of hormone injection to synchronize the ovarian cycle. For ISH, the obtained ovaries were embedded in a frozen medium (OCT compound; Sakura Finetechnical, Tokyo, Japan), frozen in liquid nitrogen, and stored at −30C until used. For IHC, the mice were perfusion-fixed with a physiological saline followed with 4% paraformaldehyde. Other mice were perfusion-fixed with 2.5% glutaraldehyde for electron microscopic observation. The ovaries obtained from perfusion-fixed mice were immersed into the same fixatives at 4C for an additional 12 hr.
In Situ Hybridization
Two non-overlapping 45-mer antisense oligonucleotide probes for galectin-1, galectin-3, 3β-HSD, and 20α-HSD were synthesized as reported previously (Nio and Iwanaga 2007). Antisense probes were also designed for nucleotide residues 661–705 and 1501–1545 of luteinizing hormone receptor (LH-R; GenBank accession number NM_013582), 671–715 and 931–975 of PRL receptor (PRL-R; GenBank accession number NM_011169), and 376–420 and 661–705 of PGF2α receptor (PGF2α-R; GenBank accession number AB094411). All probes were labeled with 33P-dATP using terminal deoxynucleotidyl transferase (Invitrogen; Carlsbad, CA). The procedure for ISH has been described previously (Nio et al. 2005). In brief, the fresh frozen sections (10-μm thick) were fixed with 4% paraformaldehyde in 0.1 M phosphate buffer for 15 min and acetylated with 0.25% acetic anhydride in 0.1 M triethanolamine-HCl (pH 8.0) for 10 min. Hybridization was performed at 42C for 12 hr by adding 10,000 cpm/μl 33P-labeled oligonucleotide probes. Then the sections were rinsed at room temperature for 30 min in 2x saline sodium citrate (SSC; 1 × SSC: 150 mM sodium chloride and 15 mM sodium citrate) containing 0.1% sarkosyl, immersed twice at 55C for 40 min in 0.1x SSC containing 0.1% sarkosyl, dehydrated through a graded series of ethanol, and air-dried. The sections were exposed to BioMax MR films (Kodak; Rochester, NY) for 1 day (for 3β-HSD and 20α-HSD), for 3 days (for PGF2α-R), or for 10 days (for galectins, LH-R, and PRL-R). Other sections were dipped in autoradiographic emulsion (NTB-2; Kodak) and exposed at 4C for 8 weeks. The sections were counterstained with hematoxylin after exposure.
ISH using two non-overlapping antisense probes exhibited consistent labeling in all tissues examined. Specificity of the hybridization was also confirmed by the disappearance of the signals, with an excess dose of unlabeled antisense probes.
Immunohistochemistry
The paraformaldehyde-fixed tissues were dehydrated through a graded series of ethanol and embedded in paraffin according to the conventional method. The sections were cut at 5-μm thickness and mounted on gelatin-coated glass slides. They were deparaffinized and rinsed in PBS (pH 7.2). After preincubation with normal goat serum for 30 min, the sections were incubated overnight with rabbit anti-galectin-1 polyclonal antibody (1.0 μg/ml; Nio-Kobayashi et al. 2009), rabbit anti-galectin-3 polyclonal antibody (1.0 μg/ml; Santa Cruz Biotechnology, Santa Cruz, CA), or rabbit anti-20α-HSD polyclonal antibody (1:750 in dilution; a kind gift from Prof. G. Gibori, University of Illinois). The sections were rinsed with PBS and incubated with biotinylated goat antibody against rabbit IgG followed with peroxidase-labeled streptavidin by using Histofine Kit (Nichirei; Tokyo, Japan). Then the antigen–antibody reactions were visualized with 0.01% DAB in 0.05 M Tris–HCl buffer (pH 7.6) containing 0.003% H2O2 and were counterstained with hematoxylin. The immunoreactions completely disappeared when PBS was used instead of the first antibodies. The specificity of antibodies used in this study was also confirmed by antigen absorption test in both Western blotting and IHC, as described in our previous study (Nio-Kobayashi et al. 2009).
Double Staining of Apoptosis and IHC for Galectin-3
Apoptotic cells in the sections were visualized using ApopTag Fluorescein In Situ Apoptosis Detection Kit (Millipore; Billerica, MA) on the basis of terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling (TUNEL) reaction. The sections used for TUNEL staining were then immunostained with the anti-galectin-3 antibody, followed with Cy3-labeled anti-rabbit IgG (Jackson ImmunoResearch Laboratories; West Grove, PA), and observed under a confocal laser microscope (FV300; Olympus, Tokyo, Japan).
Transmission Electron Microscopic Observation For IHC at the electron microscopic level, paraformaldehyde-fixed ovaries were processed for the preembedding silver-intensified immunogold method as described previously (Nio et al. 2006). The frozen sections (15-μm thick) were immunostained with anti-galectin and 20α-HSD antibodies and subsequently reacted with goat anti-rabbit immunoglobulins covalently linked to 1.4 nm gold particles (1:200 in dilution; BBInternational, Cardiff, UK). After silver enhancement (HQ silver; Nanoprobes, Yaphank, NY), the sections were postfixed with 1% OsO4 for 30 min, dehydrated through a graded series of ethanol, and directly embedded in Quetol 812 (Nisshin EM; Tokyo, Japan) on glass slides. After the embedded sections were detached from the glass slides, ultrathin sections were prepared and stained with uranyl acetate and lead citrate for observation under a transmission electron microscope (TEM; H-7100; Hitachi, Tokyo, Japan).
For membrane-enhanced conventional TEM observation, glutaraldehyde-fixed samples were postfixed in 1% OsO4 containing 0.018 g/ml potassium ferrocyanide at 4C for 1.5 hr. They were dehydrated through a graded series of ethanol and embedded in Quetol 812 according to the conventional method. Ultrathin sections were prepared from these blocks, stained using the same method described earlier, and observed under a TEM.
Statistical Analysis
The number of TUNEL-positive apoptotic cells per 1 mm2 of CL was expressed as mean ± SEM. The statistical difference was determined by two-sided Student's t-test for comparisons between the two types of CL. Difference with p<0.05 was considered to be statistically significant.
Results
In rodents, the functional CL that actively produces progesterone has a very short lifespan and immediately starts to regress unless fertilization occurs. The initiation of functional luteolysis is characterized by the expression of a progesterone degradation enzyme (20α-HSD), which catalyzes progesterone into an inactive form (20α-dihydroxyprogesterone). The subsequent “structural luteolysis” accompanies morphological changes of luteal cells and the gradual involution of CL to form a small scar composed of fibrous tissue (McCracken et al. 1999). In rodents, complete regression of CL takes over several estrus cycles, resulting in the formation of interstitial glands in the ovarian stroma instead of the corpus albicans. Because the ovary of mature mice, which has repeated estrus cycles, contains CL formed via at least three different estrus cycles, it is not easy to clarify the exact expression of galectin during CL formation and regression. Thus, we used immature mice for which we could compulsorily induce and control the ovarian cycle by hormonal treatment.
Changes of Galectin mRNA Expressions During CL Formation and Regression
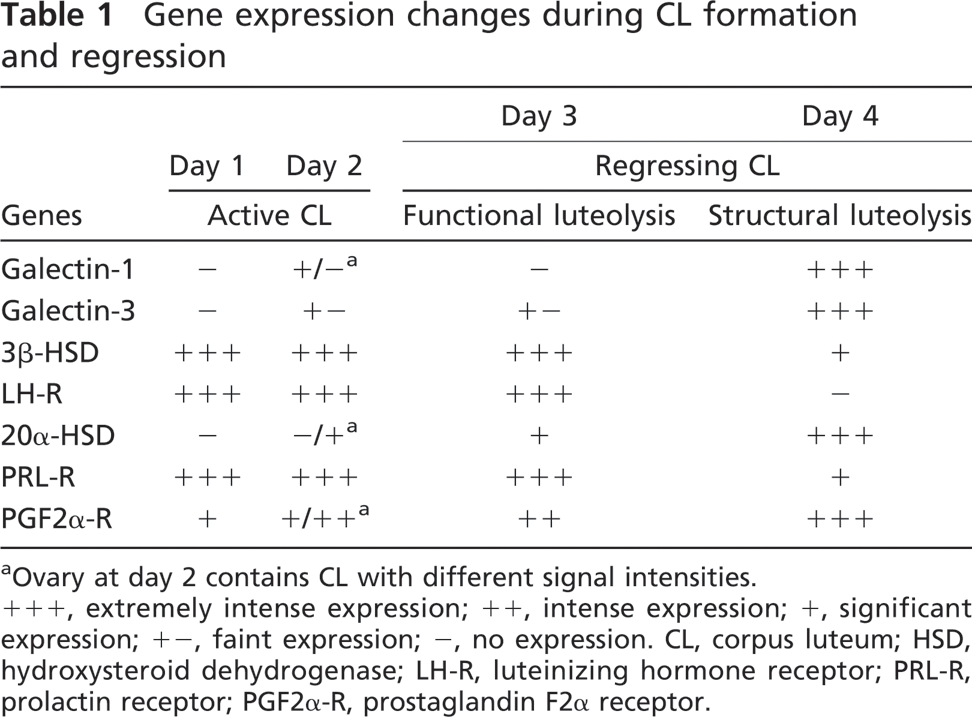
The mRNA expressions of galectin-1, galectin-3, and related molecules were analyzed using X-ray film images (Figures 1A–1U) and light microscope images (Figures 1V–1X) of serial sections from the ovaries. When the first ovulation was induced in immature mice by a combined administration of eCG and hCG, more than 10 CL were newly formed on the next day after hCG injection (day 1). The newly formed CL intensely expressed 3β-HSD and LH-R mRNAs, but lacked the mRNA expression for the progesterone degradation enzyme (20α-HSD), indicating that all CL actively produced progesterone (Figures 1C–1E). Galectin-1 was diffusely expressed in the ovarian stroma, but all CL lacked signals for galectin-1 and galectin-3 (Figures 1A and 1B). At day 2 after the injection, transcripts for galectin-1 were increased in several CL (asterisks in Figures 1H, 1V, and 1W), but the signals for galectin-3 were faint in all CL (Figure 1I). Signals for both 3β-HSD and LH-R were still intense in all CL at day 2 (Figures 1K and 1L). All the CL at this stage expressed PRL-R intensely (Figure 1M), but expressed PGF2α-R mRNA with different intensities (Figure 1N). The CL that more abundantly expressed luteolytic PGF2α-R corresponded to the CL lacking galectin-1 mRNA (compare CL indicated by arrows in Figures 1H and 1N). These CL contained some cells with an intense expression of 20α-HSD, representing the start of functional luteolysis (arrows in Figures 1J and 1V–1X). This finding implies that the start of functional luteolysis is not exactly synchronized in all CL even if ovulation is compulsorily induced by hormonal treatment, and that the expression of galectin-1 mRNA disappeared immediately before the start of functional luteolysis (arrows in Figure 1X). On day 3 after the injection (data not shown but noted in Table 1), all CL expressed 20α-HSD at a significant level, keeping an abundant expression of 3β-HSD and LH-R mRNAs. Expression of galectin-1 temporarily disappeared in all CL at this stage (Table 1). At day 4, expressions of both galectins and 20α-HSD were extremely intense in all CL (Figures 1O–1Q), whereas the signals for LH-R sharply disappeared (Figure 1S). The transcripts for PGF2α-R were observed abundantly, whereas the expression of 3β-HSD and PRL-R became weak in the CL at day 4 (Figures 1R, 1T, and 1U). The change of mRNA expressions during CL formation and regression is summarized in Table 1.
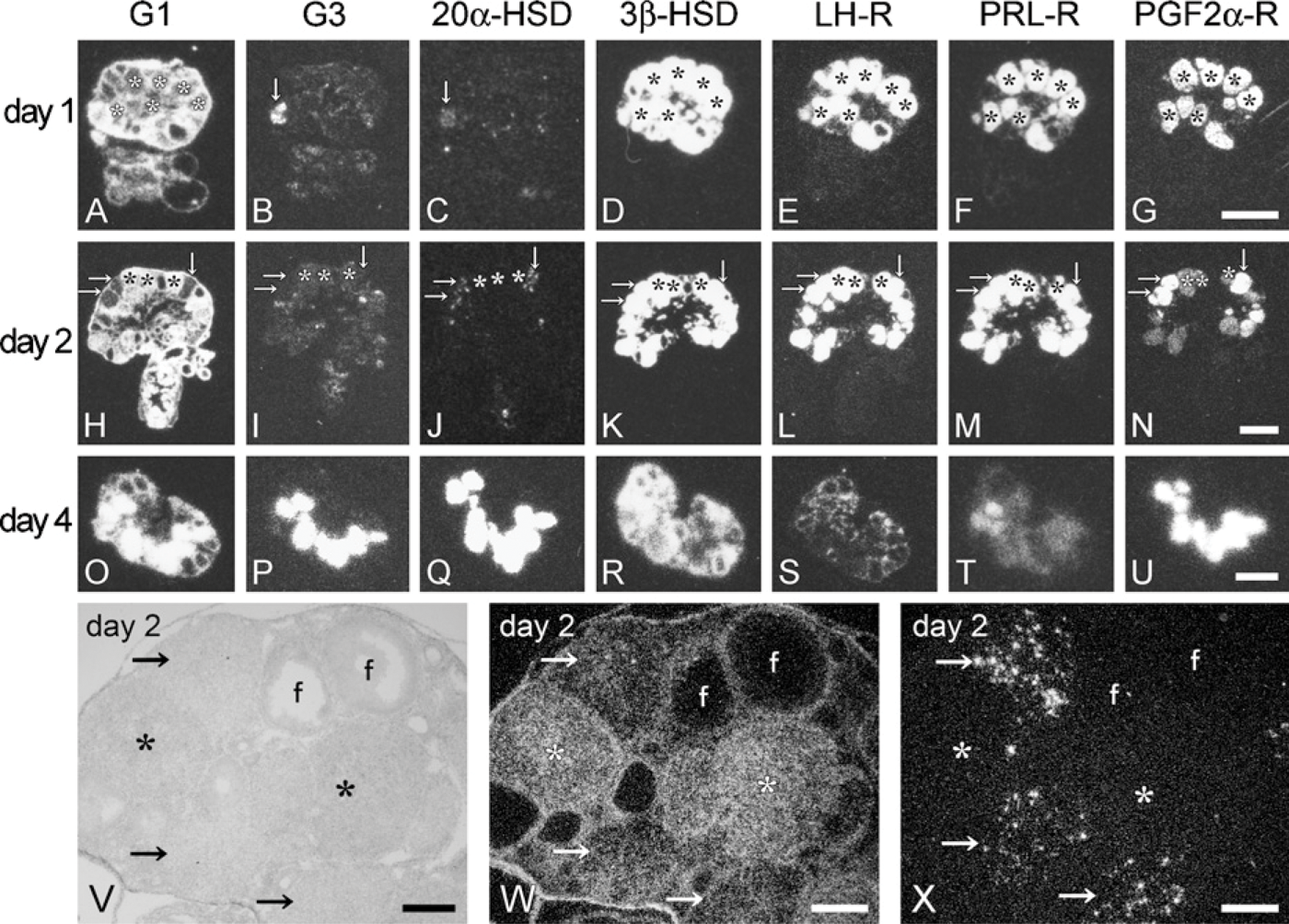
Gene expression patterns during corpus luteum (CL) formation and regression revealed by ISH (
Gene expression changes during CL formation and regression
Ovary at day 2 contains CL with different signal intensities.
+++, extremely intense expression; ++, intense expression; +, significant expression; +-, faint expression; -, no expression. CL, corpus luteum; HSD, hydroxysteroid dehydrogenase; LH-R, luteinizing hormone receptor; PRL-R, prolactin receptor; PGF2α-R, prostaglandin F2α receptor.
Immunohistochemical Localization of Galectin in the Ovary of Immature Mice
An immunohistochemical analysis was performed in the ovary of eCG- and hCG-treated immature mice to identify the galectin-expressing cells. The immunoreactivity for galectin-1 was found in fibroblast-like cells scattered in active CL at day 2 after hCG injection (Figure 2A). The galectin-1–immunoreactive cells tended to gather around the capillary vessels, but neither endothelial cells nor luteal cells were immunoreactive for galectin-1. Galectin-1–immunoreactive fibroblast-like cells projected fine, dendritic processes along the luteal cells (Figure 2A). A small number of galectin-3–positive cells, identified as infiltrating macrophages, were observed in active CL at day 2 (Figure 2B). At day 4 after hCG injection, both types of galectin-positive cells (fibroblasts-like cells and macrophages) increased in number throughout the CL (Figures 2C and 2D). Intense immunoreaction for galectin-3 was localized in the cytoplasm of infiltrating macrophages, which possessed large vacuoles in the cytoplasm (arrows in Figure 2D). The cytoplasm of luteal cells at this stage intensely expressed 20α-HSD (Figure 2E) and was moderately immunoreactive for galectin-3 (asterisks in Figure 2D). Apoptotic luteal cells with condensed or fragmented nuclei were found in regressing CL at day 4 after hCG injection (arrows in Figure 2C).
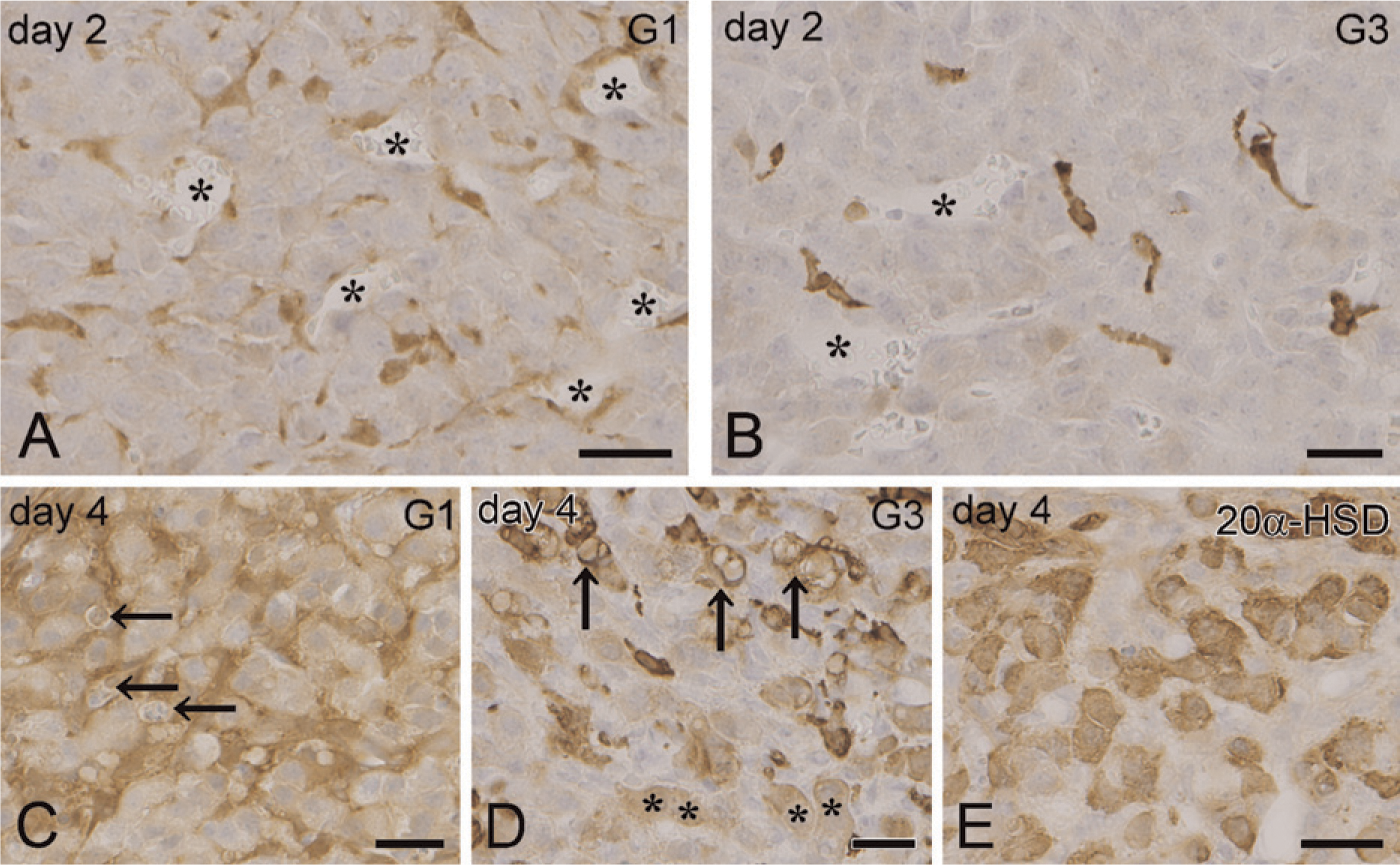
IHC of galectin in CL of hormone-treated immature mice. Galectin-1 immunoreactivity in active CL at day 2 is found in fibroblast-like cells distributed among luteal cells
Electron Microscopic Localization of Galectin
Electron microscopic observation confirmed the types of galectin-expressing cells and clearly revealed their morphological changes in both active (at day 2 after hCG injection) and regressing CL (at day 4 after hCG injection) of immature mice. Gold particles showing the presence of galectin-1 immunoreactivity were diffusely distributed in the cytoplasm of fibroblasts but not in endothelial cells or luteal cells in active CL (Figure 3A). On the other hand, gold particles for galectin-3 were localized in the cytoplasm of infiltrating macrophages in active CL (Figure 3B). In regressing CL at day 4, the galectin-3–positive macrophages contained abundant vacuoles in their cytoplasm (Figure 3C). The cytoplasm of some luteal cells in regressing CL was also immunolabeled with galectin-3 antibody (Figure 3D). These galectin-3–positive regressing luteal cells showed invaginated plasma membranes, and their cytoplasm was fragmented to form small debris (arrows in Figure 3D), but had intact nuclei. The regressing luteal cells possessed an intense immunoreactivity for 20α-HSD in their cytoplasm (Figure 3E). Observation under a membrane-enhanced conventional TEM showed the regressing luteal cells at this stage forming demarcation membranes and pinching off the cytoplasm (arrows in Figure 3F). Some population of luteal cells exhibited typical apoptotic cell death at day 4 after hCG injection; however, the other population of luteal cells characterized by galectin-3 expression were in a regressing process different from apoptosis, resulting in the formation of down-sized luteal cells.
Galectin Immunolocalization in the Ovary of Adult Mice
In adult mice with repeated estrus cycles, the ovary contained CL formed via at least three different estrus cycles (classified as new, previous, and oldest CL). When the ovaries from adult mice were immunostained for galectin-3, old CL (previous and oldest CL) was more intensely immunolabeled (CL labeled as 2 and 3 in Figure 4A) than the new CL formed at a current estrus cycle (CL labeled as 1 in Figure 4A). Galectin-3–positive macrophages were more numerous in old CL than in new CL (Figures 4A and 4B). In addition, the cytoplasm of luteal cells in old CL was moderately immunolabeled with galectin-3 antibody (CL labeled as 2 and 3 in Figures 4A and 4B). The number of galectin-1–positive fibroblasts also increased in old CL (data not shown). Thus, the old CL consisted of small-sized luteal cells, which abundantly contained galectin-3, a great number of galectin-1–positive fibroblasts, and galectin-3–positive macrophages. The double staining for galectin-3 and TUNEL showed that TUNEL-positive apoptotic luteal cells were more abundant in new CL, whereas they were scant in old CL, with intense immunoreactivities for galectin-3 in both macrophages and luteal cells (Figure 4C). At a higher magnification of new CL, luteal cells with galectin-3 immunoreactivity in the cytoplasm had escaped from TUNEL reaction (Figure 4D). Some of the TUNEL-positive luteal cells in new CL were engulfed by the galectin-3–positive macrophages (Figure 4E). Statistically, the number of apoptotic luteal cells was significantly less in old CL than in new CL (Figure 4F). The differences in the galectin-3 immunoreaction and the number of apoptotic cells between new CL and old CL are summarized in Table 2.
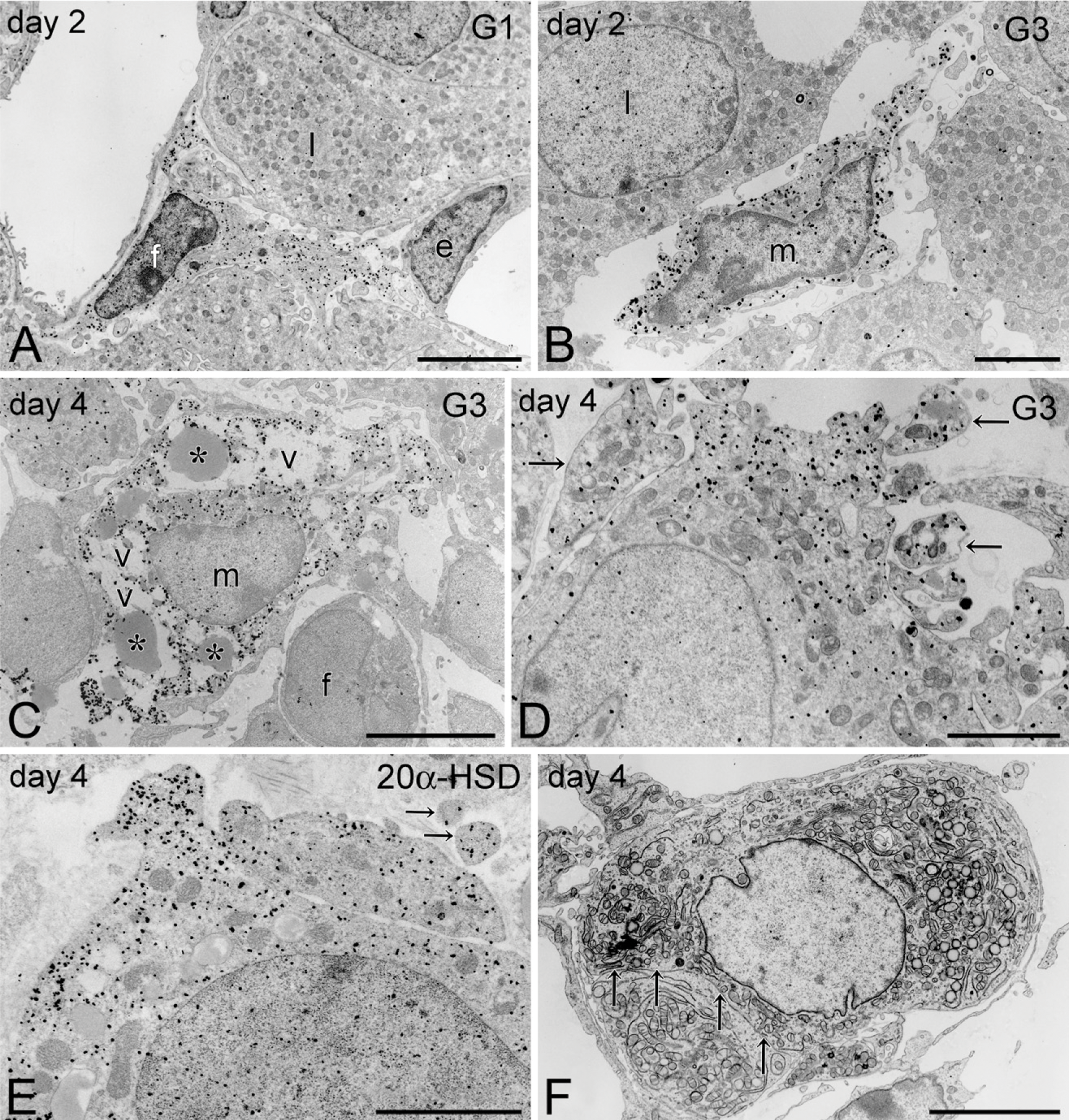
Transmission electron microscopic (TEM) observation of galectin-expressing cells. Gold particles representing the existence of galectin-1 are distributed in the cytoplasm of fibroblasts (f) but not in either endothelial cells (e) or luteal cells (l) in active CL at day 2
Discussion
The ovary, one of the organs expressing abundant galectin, is further characterized by the dynamic change of the expression patterns. This histochemical study revealed a stage- and cell-specific localization of galectin-1 and galectin-3 in the CL of mouse ovary, with comparison to the expression of related molecules essential for CL function. Both galectins were intensely expressed in regressing CL, but their expressions in active CL were limited to an extremely low level, confirming our previous finding concerning the intimate relationship of galectin to luteolysis (Nio and Iwanaga 2007). This study is the first to immunohistochemically reveal that galectin-1 is localized in the cytoplasm of fibroblasts in CL, in agreement with the general idea that galectin-1 is a stromal type of galectin (Barondes et al. 1994). Furthermore, galectin-1 showed a biphasic expression pattern during the estrus cycle. A temporal increase in the expression of galectin-1 occurred in CL before the expression of 20α-HSD, which represents the beginning of functional luteolysis. The preceding expression of galectin-1 was also observed in another experiment, showing that compulsory weaning in mice induced a temporal expression of galectin-1 in CL before the expression of 20α-HSD (Nio-Kobayashi et al., unpublished data). These findings suggest that galectin-1 plays a leading role in luteolysis. After CL ceased to express galectin-1 (at day 3 after hCG injection), the expression of galectin-1 mRNA again increased in CL under structural luteolysis, which contained abundant numbers of apoptotic luteal cells (at day 4). In the ovary of adult mature mice with repeated estrus cycles, old CL formed during previous estrus cycles contained galectin-1–positive fibroblasts more abundantly, supporting the idea of the contribution of galectin-1 to the remodeling of CL to the interstitial glands.
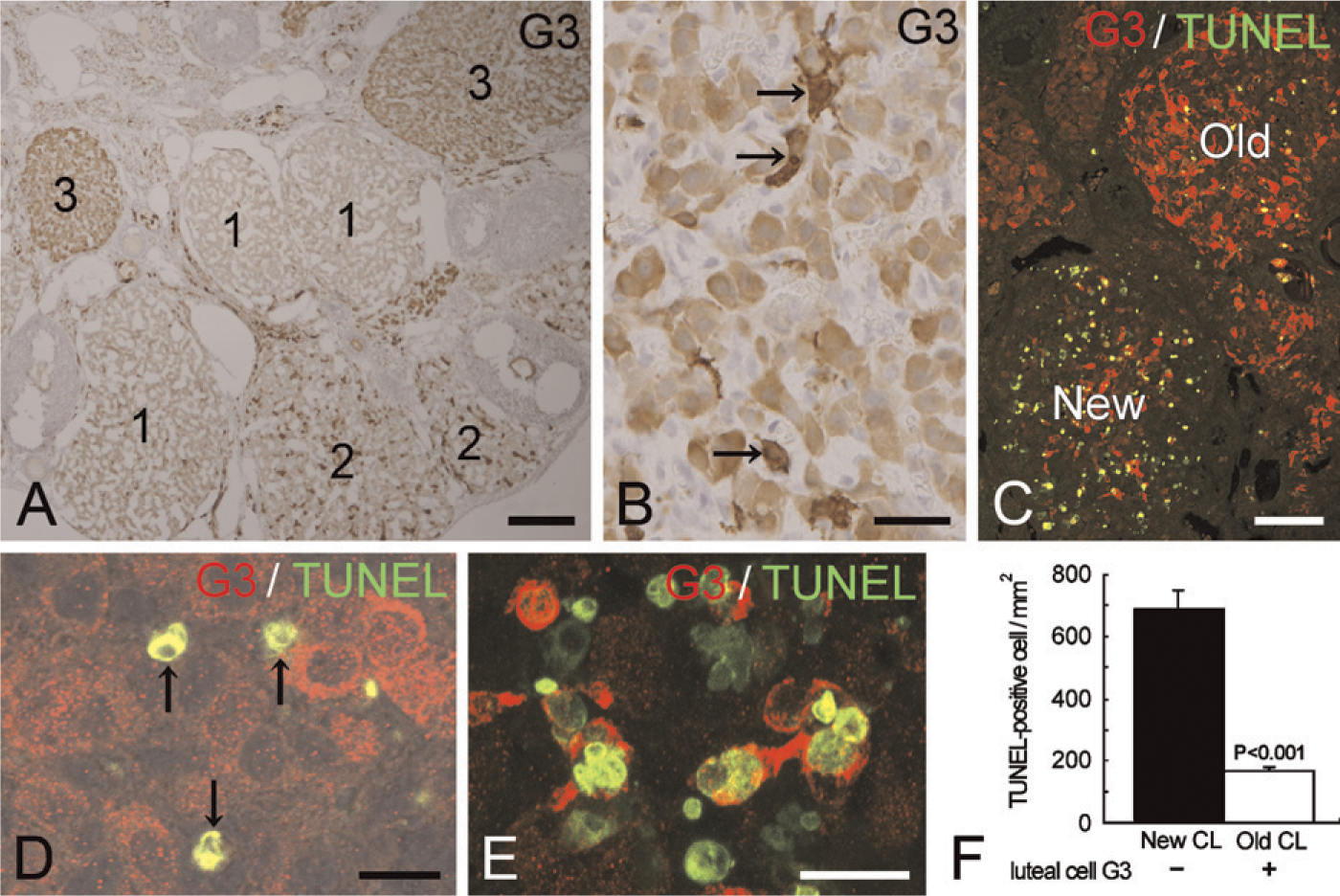
Galectin-3 immunolocalization in the ovary of adult mice. The immunoreactivities for galectin-3 are more intense in old CL formed during previous estrus cycles (2 and 3 in
Galectin-3 immunoreactivities and number of apoptotic cells in new CL and old CL
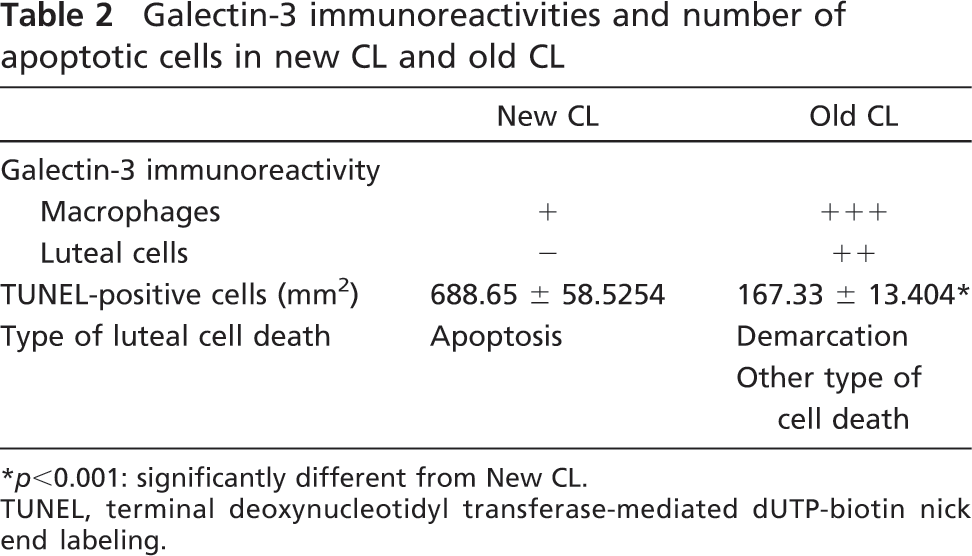
p<0.001: significantly different from New CL.
TUNEL, terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling.
Galectin-3 was consistently and intensely expressed in regressing CL with a concomitant expression of 20α-HSD, whereas it was almost absent in active CL. The galectin-3 immunoreactivity was localized in the cytoplasm of macrophages distributed in regressing CL, in agreement with a recent study by Lohr et al. (2008). Infiltrating macrophages with galectin-3 immunoreactivity increased in number parallel with the aging of CL. There is increasing evidence that macrophages play a pivotal role in luteolysis (Lei et al. 1991; Bukovský et al. 1995). We demonstrated here the accumulation of galectin-3–positive macrophages in CL under structural luteolysis, which contained abundant numbers of apoptotic luteal cells (at day 4 after hCG injection). In contrast, Komatsu et al. (2003) have reported the infiltration of macrophages in CL under the functional luteolysis—but not the structural luteolysis—of the mouse ovary, which possessed 20α-HSD enzyme activity, by immunostaining using anti-mouse macrophage IgG (Cedarlane; Burlington, ON, Canada). This discrepancy may be due to the specificity and characterization of antibodies used for the staining of macrophages. The galectin-3-positive infiltrating macrophages observed in this study contained numerous vacuoles in their cytoplasm, suggesting its accelerated phagocytotic activity. This finding coincides with the fact that galectin-3 is involved in phagocytosis (Dong and Hughes 1997; Sano et al. 2003; Rotshenker 2009): galectin-3–deficient macrophages exhibited a reduced phagocytotic activity.
Cytoplasmic galectin-3 is known to function as an inhibitor of apoptosis (Yang et al. 1996; Nangia-Makker et al. 2007). Galectin-3 binds to various antiapoptotic proteins such as Bcl-2, activated K-Ras, Alix/A1P1, and synexin to inhibit the intrinsic apoptotic pathways. The intracellular galectin-3 can also regulate extrinsic apoptotic pathways induced through death receptors such as Fas (CD95/apo-1), tumor necrosis factor receptor-1 (TNFR-1/p55/CD120), and TRAIL (TNF-related apoptosis inducing ligand/Apo2-L; Nangia-Makker et al. 2007). This study revealed that luteal cells themselves were immunoreactive for galectin-3 in old CL formed at previous estrus cycles. The TUNEL-positive apoptotic cells were remarkably fewer in old CL composed of galectin-3–positive luteal cells than in new CL composed of galectin-3–negative luteal cells, supporting the idea that the intracellular galectin-3 may function as an anti-apoptotic factor also in luteal cells. Even if the galectin-3–containing luteal cells of old CL were resistant to apoptosis, they must be eliminated by some process. This electron microscopic study detected galectin-3 in the cytoplasm of regressing luteal cells and a unique fragmentation of their cytoplasm by demarcation membranes. These cells continued to possess intact nuclei, unlike the typical features of apoptotic cell (Kerr and Harmon 1991). The unique fragmentation of luteal cells at luteolysis was previously demonstrated in the ovary of marmosets (Fraser et al. 1999): for the vast majority of regressing luteal cells, cell death was processed by a non-apoptotic mechanism, namely “cell atrophy with phagocytosis of cytoplasmic debris.” Thus, it is considered that the demarcation is a process of luteal cell elimination instead of apoptosis especially in old CL and that it may be regulated by the cytoplasmic galectin-3. By this process, the luteal cells of old CL are downsized via several estrus cycles and finally result in the formation of the interstitial glands.
The main regulatory molecules of CL function in rodents are PRL and PGF2α released from the pituitary gland and the uterus, respectively. PRL and PGF2α have opposing luteotrophic or luteolytic effects on CL (Stocco et al. 2001). PRL mainly functions luteotrophically on CL by upregulating the expressions of 3β-HSD and LH-R, whereas PGF2α always acts luteolytically by inducing 20α-HSD expression. PRL and PGF2α regulate ovarian functions through their receptors expressed mainly in luteal cells. This study clearly demonstrated the expression pattern of receptors for the two main signals of CL, PRL-R and PGF2α-R, in association with galectin expression. The expression of PRL-R was very high in active CL; however, it was sharply reduced in regressing CL, with an intense expression of PGF2α-R. It is well known that PGF2α induces 20α-HSD expression in CL at the beginning of functional luteolysis (Stocco et al. 2007). Galectin-3 and 20α-HSD were expressed in regressing CL, with an intense expression of PGF2α-R, suggesting that the expression of galectin-3 is linked with the release of PGF2α from the uterus. This idea is supported by our preliminary study showing that the administration of PGF2α induces galectin-3 expression in CL. On the other hand, our preliminary examination showed that the continuous administration of bromocriptine, which suppresses the secretion of PRL from the pituitary gland, caused an extensive expression of galectin-1 in CL. Furthermore, compulsory weaning in mice reduced PRL secretion from the pituitary gland due to the lack of sucking reflex, inducing galectin-1 expression in CL (Nio-Kobayashi et al., unpublished data). Because galectin-1 expression was also induced by PGF2α administration (Nio-Kobayashi et al., unpublished data), the first increase in galectin-1 expression in active CL may be caused by the reduction of the plasma concentration of PRL, and the second increase in galectin-1 expression in regressing CL may be affected by PGF2α as well as galectin-3/20α-HSD. Taken together, these findings indicate that PRL and PGF2α may be the key molecules that directly or indirectly regulate galectin expression in CL. To elucidate the function and regulatory mechanism of galectin expression by PRL and PGF2α, further investigations—including the identification of ligand carbohydrates for galectin in the ovary—are required.
In conclusion, the stage-specific expression and cellular localization of galectin-1 and galectin-3 in regressing CL suggest their differential contribution to luteolysis. Galectin-1 may function as an initiator of luteolysis and may be regulated by plasma PRL concentrations. Involvement of galectin-3 in regressing luteal cells differs between new CL, which is first exposed to luteolytic signals, and old CL, which receives repeated luteolytic signals during several estrus cycles. Thus, galectin-3 may play a role in the formation of the interstitial gland and luteal cell elimination.
Footnotes
Acknowledgements
The authors thank Prof. Geula Gibori and Dr. Shehu Aurora, University of Illinois at Chicago, for their kind gift of the antibody for 20α-HSD. We are also grateful to Prof. Kiyoshi Okuda, Okayama University, for his kind advice.
