Abstract
Galectin, an animal lectin that recognizes β-galactosides of glycoconjugates, is involved in multiple biological functions such as cell growth, differentiation, apoptosis, and signal transduction. The present study using in situ hybridization revealed the predominant expression of galectin-1 and galectin-3 in the mouse ovary. Galectin-1 mRNA was diffusely expressed in the ovarian stroma, including the interstitial glands and theca interna, and intensely expressed in the corpus luteum (CL) at particular stages of regression. Transcripts of galectin-3 were restricted to CL and always coincident to the expression of 20α-hydroxysteroid dehydrogenase (20α-HSD), a progesterone degradation enzyme. In the non-pregnant ovary, signals for both galectin-1 and −3 were intense in the old, regressing CL formed at previous estrous cycles. In the newly formed CL, the signal intensity of galectin-1 first increased at the starting point of regression followed by increasing galectin-3/20α-HSD expressions. Under gestation with active progesterone production, signals for both galectin-1 and −3 in CL completely disappeared. At the perinatal stage, intense expressions of galectin-3/20α-HSD recovered in the remaining CL of gestation with the temporal expression of galectin-1 and continued until weaning. These findings suggest that galectin-1 and −3 may mediate progesterone production and metabolism in luteal cells via different mechanisms.
G
Galectins are widely distributed in the mammalian body but show cell/tissue-specific expressions. The digestive tract is one of the organs rich in galectin, as it expresses at least six subtypes (galectin-2, −3, 4/6, −7, and −9) in the epithelium with region-dependent and cell-specific patterns (Nio et al. 2005). On the other hand, the major subtype in the urinary system is galectin-3, which exists predominantly in epithelia from the kidney to the distal end of the urethra (Nio et al. 2006). Previous studies on the genital system have detected significant expressions of galectin-1, −3, and −7. Galectin-1, a stromal type of galectin, is ubiquitously expressed in a variety of tissues and is especially intense in the lymphoid tissues and muscles. In the female genital system, the uterus and ovary express galectin-1, as shown by Northern and Western blot analyses (Choe et al. 1997). Although cultured porcine granulosa cells were reported to contain galectin-1 (Walzel et al. 2004), cell types expressing galectin-1 in the ovary in vivo have not been identified. Sato et al. (2002) detected galectin-7, an epidermal type of galectin, in the ovary by Western blot analysis and immunohistochemistry, suggesting that stromal cells were a cellular source of galectin-7. The uterus and placenta express galectin-1 as well as galectin-3. As galectin-1 and −3 are important mediators of inflammation (Almkvist and Karlsson 2004), many researchers have studied their expressions in the uterus in relation to endometrial function and implantation (Phillips et al. 1996; Choe et al. 1997; Lee et al. 1998; von Wolff et al. 2005).
Regarding the function of galectin in the genital organ, its intimate relation to steroid hormones is worth noting. Choe et al. (1997) described the expression of galectin-1 in the uterus as being differentially regulated by the ovarian steroids, progesterone and estrogen. On the other hand, selective expression of galectin-1 in ovarian cells and testicular Leydig cells suggests their involvement in steroid hormone production (Wollina et al. 1999; Timmons et al. 2002; Dettin et al. 2003; Martinez et al. 2004). Addition of galectin-1 to cultured porcine granulosa cells in vitro inhibited follicle-stimulating hormone (FSH)-stimulated progesterone synthesis (Walzel et al. 2004). Similarly, addition of galectin-1 to cultured rat Leydig cells caused the reduction of testosterone production (Martinez et al. 2004). These studies imply that galectin-1 may act as an inhibitory regulator of steroid hormone production; however, identification of cells expressing galectin in the ovary in vivo and involvement of galectin in the production of progesterone are still unclear.
In the present study we investigated the cellular expression of eight major subtypes of galectin (galectin-1, −2, −3, −4/6, −7, −8, and −9) in the mouse ovary under non-pregnant, pregnant, and postpartum conditions, with special reference to the production and activity of progesterone in the corpus luteum (CL).
Materials and Methods
Animals and Tissue Sampling
Ten-week-old adult female ddY mice were supplied by Japan SLC (Shizuoka, Japan). Non-pregnant mice at met-estrus and at di-estrus (estrous cycle was determined by vaginal smears), pregnant mice (day 15.5 and day 18.5 after observation of vaginal plugs), and postpartum mice (from postpartum day 0.5 until weaning) were used in this study. Tissue samplings were performed in the morning. Mice under deep anesthesia with pentobarbital were killed by blood letting from the heart, and the ovaries were dissected out. Tissues were embedded into optimal cutting temperature compound (OCT; Sakura Finetechnical Co., Ltd. Tokyo, Japan) and quickly frozen in liquid nitrogen. This study was approved by the Animal Care and Use Committee of Hokkaido University.
In Situ Hybridization
Two non-overlapping 45-mer antisense oligonucleotide probes for each galectin subtype mRNAs (galectin-1 through −9, except for galectin-5) were synthesized as reported previously (Nio et al. 2005). Antisense probes were also designed to nucleotide residues 291–335 and 610–654 of a progesterone synthesis enzyme, Δ5-3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase I (3β-HSD) (GenBank accession no. M58567) and nucleotide residues 80–124 and 795–839 of a progesterone degradation enzyme, 20α-hydroxysteroid dehydrogenase (20α-HSD) (GenBank accession no. AB059565). All probes were labeled with 33P-dATP using terminal deoxynucleotidyl transferase (Promega; Madison, WI). The procedure for in situ hybridization has been described previously (Nio et al. 2005,2006). In brief, 10-μm-thick fresh frozen sections were fixed with 4% paraformaldehyde in 0.1 M phosphate buffer for 15 min and acetylated with 0.25% acetic anhydride in 0.1 M triethanolamine-HCl (pH 8.0) for 10 min. Hybridization was performed at 42C for 10 hr by adding 10,000 cpm/μl 33P-labeled oligonucleotide probes. Sections were then rinsed at room temperature for 30 min in 2X SSC (1X SSC: 150 mM sodium chloride, 15 mM sodium citrate) containing 0.1% sarkosyl, twice at 55C for 40 min in 0.1X SSC containing 0.1% sarkosyl, dehydrated through a graded series of ethanol, and air dried. Sections were either exposed to BioMax MR films (Kodak; Rochester, NY) for 1 day (for 3β-HSD and 20α-HSD) or for 10 days (for galectins) or dipped in autoradiographic emulsion (NTB-2; Kodak) at 4C for 8 weeks and counterstained with hematoxylin after development. Hybridized sections used for exposure on X-ray films were stained with hematoxylin and eosin (H/E).
In situ hybridization using two non-overlapping antisense probes exhibited consistent labeling in all tissues examined. Specificity of the hybridization was also confirmed by the disappearance of signals with an excess dose of unlabeled antisense probes.
Results
Overview of Galectin Expression in the Ovary
We first examined mRNA expressions of eight major galectin subtypes (galectin-1, −2, −3, −4/6, −7, −8, and −9) in the ovary under non-pregnant (at met-estrus and diestrus), pregnant (at day 15.5), and postpartum (at day 3.5) conditions. Significant signals for galectin-1, −3, and galectin-7 could be detected on X-ray films of serially cut hybridized sections. Signals for other subtypes were undetectable in the present in situ hybridization. Transcripts of galectin-1 were broadly distributed in the ovarian stroma under all conditions (Figures 1A, 1D, 1G, and 1J) and also found in some of the CL of the non-pregnant ovary (asterisks in Figures 1A and 1D). On the other hand, expression of galectin-3 was restricted to CL in the non-pregnant and postpartum ovaries (Figures 1B, 1E, and 1K). Signals for galectin-7 appeared on the surface of the ovary under all conditions (data not shown).
Galectin expression in CL changed during the estrous cycle and by gestation. At met-estrus, some CL showed intense signals for galectin-3 and less intense signals for galectin-1 (asterisks in Figures 1A and 1B), whereas the other CL lacked signals for both subtypes as indicated by arrows in Figures 1A and 1B. At diestrus, all CL expressed galectin-1 and −3 with various intensities (Figures 1D and 1E). In the pregnant ovary, signals for both galectin-1 and −3 in CL completely disappeared (Figures 1G and 1H). After parturition, some CL returned to express transcripts of galectin-3 (asterisks in Figure 1K) and intermingled with CL lacking any signals for galectin-3 (arrows in Figure 1K). All CL in the postpartum ovary weakly expressed galectin-1 (Figure 1J).
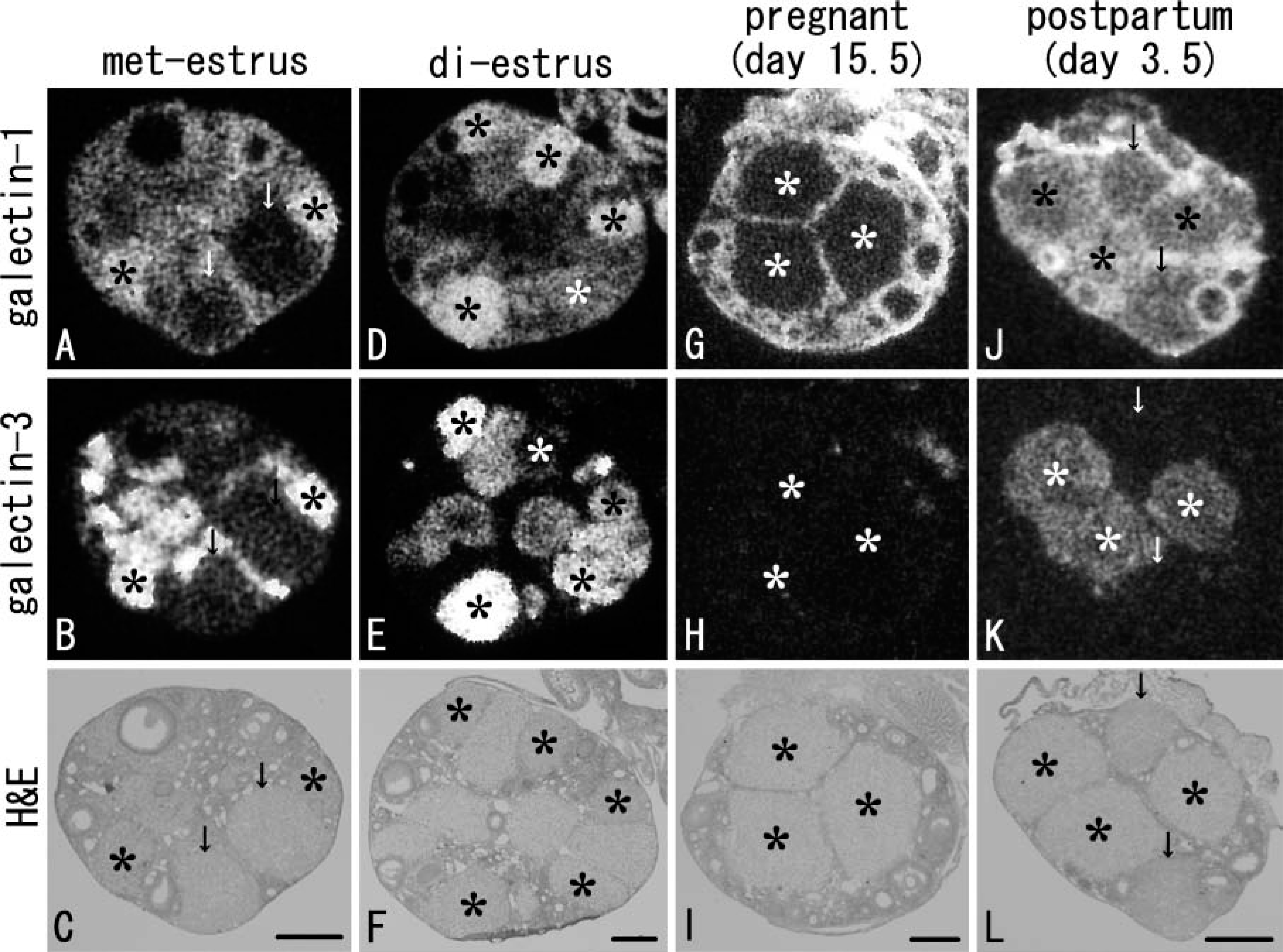
X-ray observations of galectin expressions on adjacent sections of the ovary under non-pregnant (at met-estrus,
Galectin Expression and Progesterone Production in the Non-pregnant Ovary
Light microscopic observation of hybridized sections revealed detailed expression patterns of galectin mRNAs. In the non-pregnant ovary at met-estrus, galectin-1, a stromal type of galectin, was expressed in various cells including the interstitial glands, theca interna, and the surface epithelium; however, oocytes and granulosa cells lacked signals for galectin-1 (Figure 2). In addition, some CL at met-estrus expressed galectin-1 with moderate intensity (double asterisks in Figure 2), and these CL simultaneously showed selective expression of galectin-3 (Figure 2). The other CL at met-estrus, possibly the newly formed CL, possessed faint signals for both galectin subtypes (single asterisks in Figures 2A and 2B). At di-estrus, all CL expressed galectin-1 and −3 mRNAs with various intensities (Figures 3A and 3B), some containing extremely intense signals for galectin-1 (single asterisks in Figures 3A). Signal intensities of two galectins at di-estrus tended to be complementary: CL with intense signals for galectin-1 weakly expressed galectin-3 (single asterisks in Figures 3A and 3B).
Because CL is a predominant tissue of galectin production in the ovary, expressions of key enzymes for progesterone synthesis and degradation, 3β-HSD and 20α-HSD, were examined on serial sections. Signal intensities of 3β-HSD and 20α-HSD in each CL reversed, and expression of 20α-HSD correlated well with that of galectin-3 (Figures 2B-2D and Figures 3B-3D). CL with intense signals for 3β-HSD and weak signals for 20α-HSD, which were actively producing progesterone and observed only at met-estrus, were identifiable as new CL formed after the last ovulation (single asterisks in Figures 2C and 2D). In this type of CL, transcripts of both galectin-1 and −3 were faint at met-estrus (single asterisks in Figures 2A and 2B). At di-estrus, however, the newly formed CL entered a regression stage as characterized by the decreased 3β-HSD and increased 20α-HSD expressions (single asterisks in Figures 3C and 3D) and changed to express galectin-1 intensely, whereas expression of galectin-3 was still weak (single asterisks in Figures 3A and 3B). The ovary at met-estrus and di-estrus contained CL formed at previous estrous cycles. These old CL were in a process of regression and expressed 20α-HSD intensely and 3β-HSD weakly (Figures 2C and 2D and Figures 3C and 3D). The old, regressing CL contained sufficient transcripts of both galectin-1 and −3 (Figures 2A and 2B and Figures 3A and 3B). Some that expressed 20α-HSD more intensely and 3β-HSD more weakly corresponded to CL formed before the last estrous cycle and contained abundant transcripts of galectin-1 (double asterisks in Figure 2 and Figure 3).
These findings indicate that (1) CL at regression stage is always accompanied by galectin expressions, and galectins are more intensely expressed in the old, regressing CL formed at previous estrous cycles; (2) galectin-3 expression is habitually synchronized with the synthesis of 20α-HSD, a progesterone degradation enzyme; and (3) newly formed CL shows a sharp increase in galectin-1 expression at the starting point of regression at di-estrus.
Galectin Expression and Progesterone Production in Association With Gestation
To clarify the change of galectin expression associated with gestation, mRNA expressions of galectins, 3β-HSD, and 20α-HSD were investigated in serial sections of the ovary under gestational and postpartum conditions. In the ovary at day 15.5 of gestation, both signals for galectin-1 and −3 completely disappeared from the CL (asterisks in Figures 4A and 4B), although the surface epithelium and stromal tissues maintained expression of galectin-1 (Figure 4). Signals for 3β-HSD were intense in all CL, whereas these for 20α-HSD were extremely weak or undetectable (asterisks in Figures 4C and 4D). In the postpartum ovary at day 3.5, all CL still intensely expressed 3β-HSD (Figure 5) but changed to express galectin-1, although not intensely (Figure 5). Galectin-3 and 20α-HSD were selectively and concomitantly expressed in some CL (asterisks in Figures 5B and 5D), intermingling with CL without galectin-3/20α-HSD expressions. These two types of CL observed in the postpartum ovary were investigated in detail below.
In the ovary after parturition, CL of menstruation (spurious CL) is newly formed after the postpartum estrus. Therefore, the remaining veritable CL of gestation intermingles with CL of menstruation in the postpartum ovary, and both types of CL continue during lactation (Takiguchi et al. 2004). To characterize the two types of CL, namely, CL with or without galectin-3/20α-HSD expressions, mRNA expressions of galectins, 20α-HSD, and 3β-HSD were investigated on X-ray films exposed to serial sections of the ovary from the perinatal stage until weaning. Expressions of galectins and 20α-HSD in CL disappeared during pregnancy as described above. At day 18.5 of pregnancy (the day before parturition), however, expression of galectin-3 recovered in all CL of gestation that intensely expressed 20α-HSD (Figures 6A and 6B). The recovered expression of galectin-3 and 20α-HSD in CL of gestation became more intense at postpartum day 0.5 (Figures 6F and 6G) and quickly decreased in intensity at postpartum day 2.5 (asterisks in Figures 6K and 6L). CL without expressions for galectin-3/20α-HSD but expressing 3β-HSD intensely first appeared at day 2.5 after parturition, being identifiable as CL of menstruation formed after the postpartum estrus (arrows in Figures 6K-6N). Thus, CL expressing galectin-3 and 20α-HSD at postpartum day 3.5 (see Figure 5) were identified as the remaining CL of gestation. Expressions of galectin-3 and 20α-HSD in the remaining CL of gestation continued during lactation (data not shown).
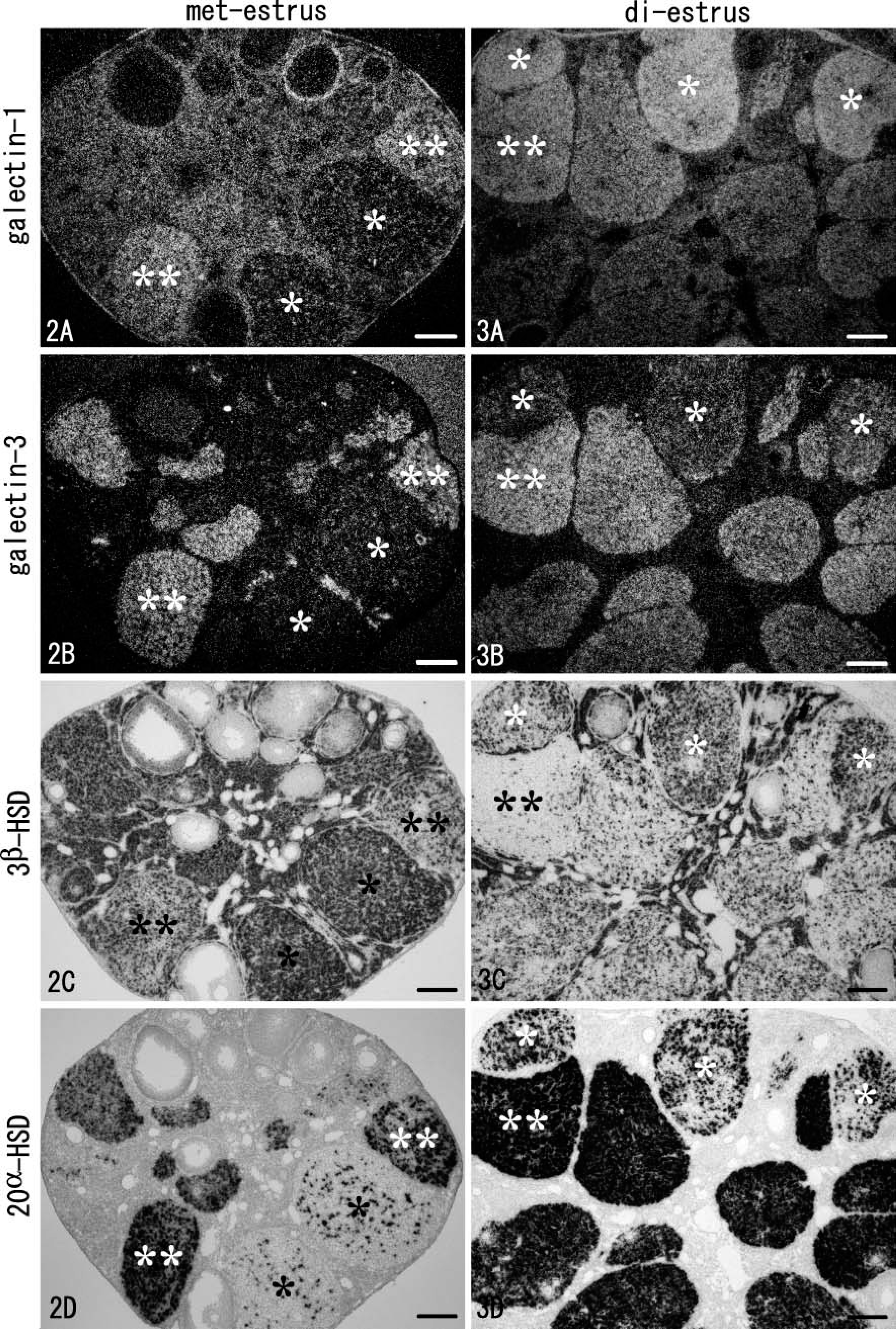
Light microscopic observations of hybridized sections in the non-pregnant ovary at met-estrus (Figure 2) and at di-estrus (Figure 3). Four serial sections are hybridized for mRNA expressions of galectin-1, −3, 3β-HSD, and 20α-HSD and observed under dark fields (
Galectin-1 expression in the remaining CL of gestation sharply increased in intensity at postpartum day 0.5 (Figure 6H), although on the day before parturition (day 18.5 of pregnancy) these CL still lacked signals for galectin-1 (asterisks in Figure 6). At day 2.5 after parturition, signals for galectin-1 in the remaining CL of gestation conspicuously decreased (asterisks in Figure 6M), whereas signals for galectin-1 became intense in the newly formed CL of menstruation, which intensely expressed 3β-HSD but lacked galectin-3/20α-HSD expressions (arrows in Figures 6K-6N). Noteworthily, the two types of CL were easily distinguished in H/E staining (Figure 6O). At day 3.5 after parturition, galectin-1 in CL of menstruation decreased in expression level (see Figure 5), and all CL changed to weakly express galectin-1 until weaning (data not shown).
Galectin expression and progesterone production in CL associated with pregnancy and parturition are summarized in Table 1. Expression patterns of galectins were characterized as follows. Intense expressions of galectin-1 and −3 occur at the perinatal stage; their expressions do not synchronize but rather are complementary. Gestational CL and the postpartum CL of menstruation express predominantly galectin-3 and −1, respectively. As observed in the non-pregnant ovary, expression of galectin-3 is coincident to the expression of 20α-HSD in the remaining CL of gestation and continues until weaning. Postpartum expression of galectin-1 in both CL of gestation and menstruation is transient and sharply decreases in intensity until postpartum day 3.5.
Discussion
Expression of Galectin Subtypes in the Ovary
Previous studies have reported the existence of galectin-1 and −7 in the ovary (Choe et al. 1997; Sato et al. 2002). In this study we revealed that the predominant galectin subtypes in the mouse ovary are galectin-1 and −3 throughout non-pregnant, pregnant, and postpartum conditions. Although Sato et al. (2002) immuno-histochemically reported expression of galectin-7 in ovarian stromal cells of mice, the present in situ hybridization detected its signals only in the surface epithelium of the ovary.
Galectin-1 is diffusely expressed in the connective tissues, muscles, and lymphoid tissues (Pace and Baum 1997). Previous Northern blot and Western blot analyses reported expression of galectin-1 in the ovary and uterus of mice (Choe et al. 1997). Although cultured porcine granulosa cells were shown to contain galectin-1 (Walzel et al. 2004), the present in situ hybridization analysis failed to detect any signals for galectin-1 in murine granulosa cells. This study revealed the consistent and diffuse expression of galectin-1 in the ovarian stroma, including the interstitial glands and theca interna, and in the surface epithelium. Another expression site of galectin-1 was CL, in which expression levels depended on the estrous cycle and gestation. On the other hand, galectin-3 is known to exist in epithelial cells of the skin, gastrointestinal tract, and urinary tract (Nio et al. 2005,2006) as well as in immune cells including macrophages and mast cells (Cherayil et al. 1989; Woo et al. 1990; Craig et al. 1995). No study is available about the expression of galectin-3 in the ovary. The present study revealed the intense and selective expression of galectin-3 in CL of the non-pregnant and postpartum ovary. Galectin-3 in CL was characterized by a concomitant expression with 20α-HSD, a progesterone degradation enzyme. In CL with active progesterone production, for example, in CL of gestation, expression of galectin-3 and also galectin-1 completely disappeared. Furthermore, expressions of both galectin subtypes in the non-pregnant ovary were more intense in the old, regressing CL formed at previous estrous cycles than those in the newly formed CL. These findings strongly support the proposition that galectins are involved in luteolysis.
Expression of Galectin-3 and −1 in Regressing CL
In animals with incomplete estrous cycle, such as rodents, CL have a very limited life span during the estrous cycle in which it enters the regressing process immediately after a short functional stage with active progesterone production. The regression process of CL in the mouse ovary is classified into two phases: the functional regression stage and the following structural regression stage (McCracken et al. 1999). At the former stage, 20α-HSD is expressed in luteal cells to catalyze progesterone to a biologically inactive form (20α-dihydroxyprogesterone). The latter stage induces apoptosis of luteal cells, resulting in atrophy and disappearance of CL (Komatsu et al. 2003). Because galectin-3 was simultaneously expressed with 20α-HSD in the regressing CL but not expressed in CL with a high activity of progesterone production, galectin-3 may be involved in the functional regression of CL. Galectin-1 in CL was more intensely expressed at di-estrus than at met-estrus, and the highest expression level was found in the newly formed CL at the starting point of the functional regression stage with an increasing expression of 20α-HSD (and also galectin-3). When changes of galectin expression were examined in association with weaning, galectin-1 expression always preceded these of galectin-3/20α-HSD in CL at beginning of luteolysis (Nio and Iwanaga, unpublished data). This finding suggests that galectin-1 may play a leading role at the initiation of the functional regression in CL.
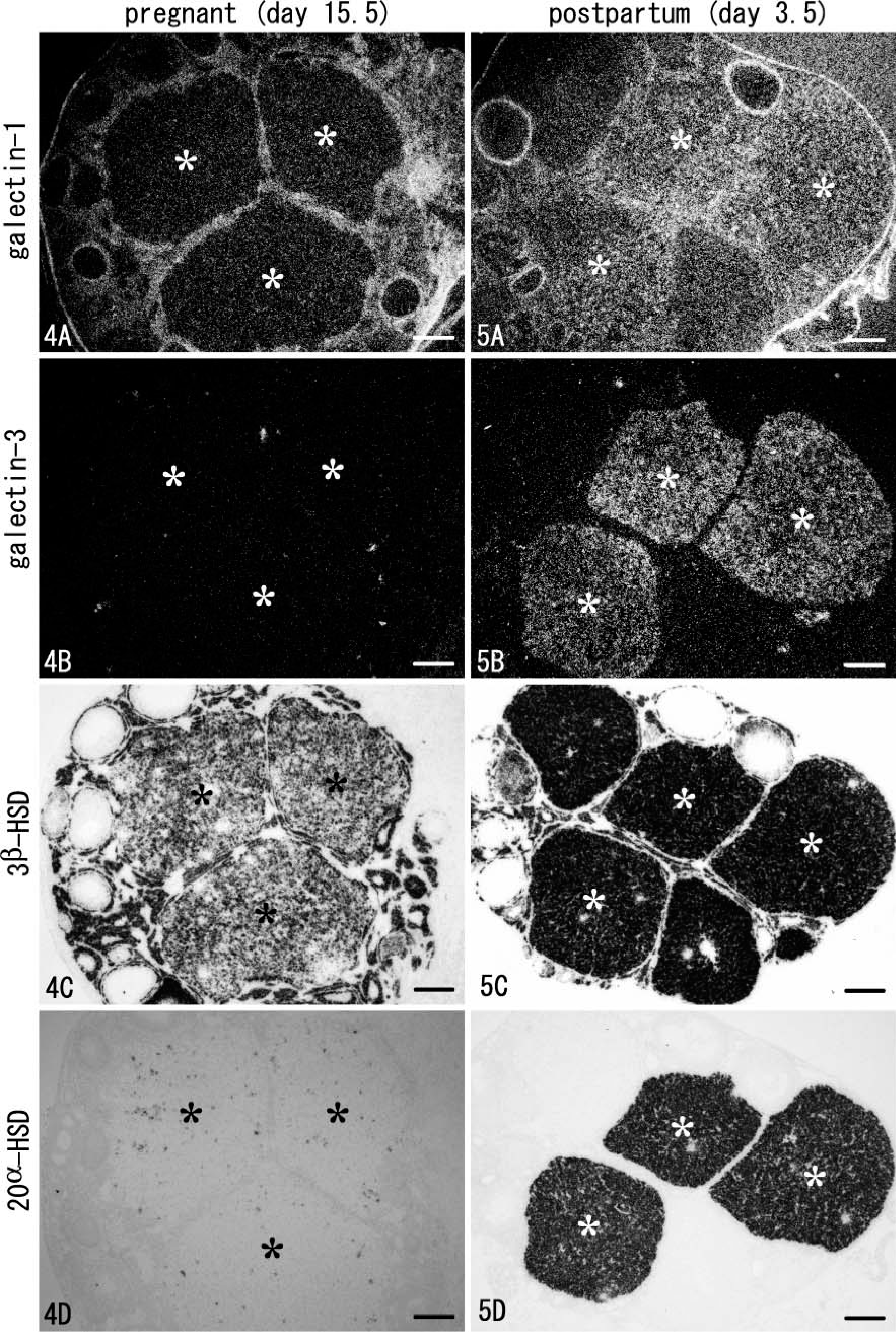
Light microscopic observation of four hybridized serial sections from the ovary under pregnant (day 15.5) (Figure 4) and postpartum (day 3.5) (Figure 5) conditions. The sections for galectins were darkfield images (
Galectin expression and progesterone production in corpus luteum associated with gestation
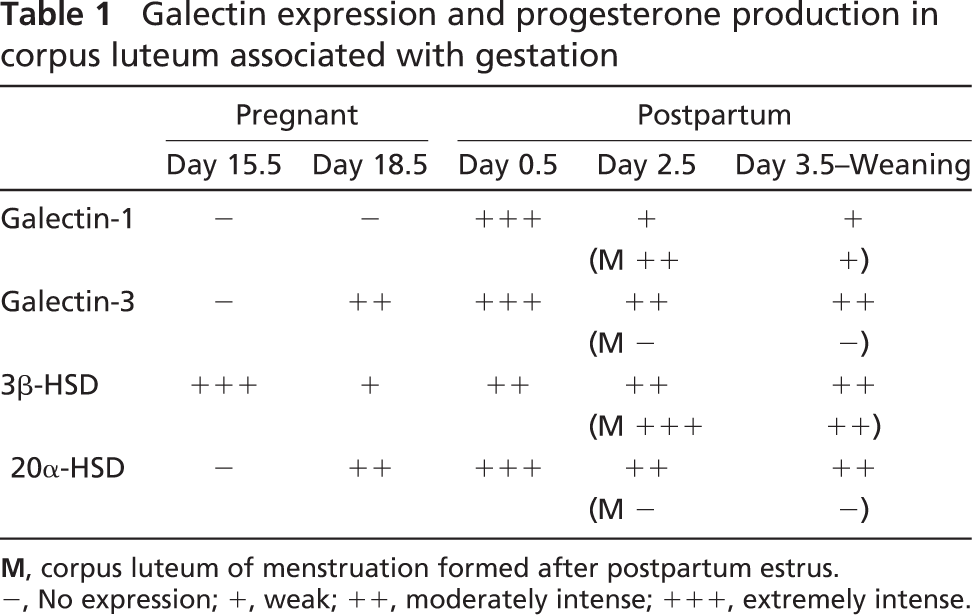
M, corpus luteum of menstruation formed after postpartum estrus.
−, No expression; +, weak; ++, moderately intense; +++, extremely intense.
Galectin-1 is known to cause apoptosis of thymocytes and activated T cells in the lymphoid tissues (Perillo et al. 1995,1997), whereas galectin-3 exerts anti-apoptotic activity on a variety of cells (Liu et al. 2002). To examine the involvement of galectin-1 and −3 in the structural regression of CL, we compared apoptotic events (TUNEL staining) and galectin expressions on serial sections from the ovary at di-estrus. Immuno-reactivities for galectin-1 and −3 were less intense in the TUNEL-positive CL, which had entered the structural regression stage, suggesting a non-direct role for galectins in the structural regression of CL (Nio and Iwanaga, unpublished data). However, because a moderately intense immunoreactivity for galectin-1 was found in the TUNEL-positive CL, this preliminary examination does not rule out the involvement of galectin-1 in the structural regression of CL.
Galectin and Progesterone Synthesis
Synchronized expression of galectin-3 and 20α-HSD strongly supports the idea that galectin-3 might act as a negative regulator of progesterone activity. However, no information is available on the relationship between galectin-3 and progesterone activity. For galectin-1, some studies have dealt with the regulation of steroid hormone synthesis by galectin. Martinez et al. (2004) observed inhibition of testosterone synthesis by galectin-1 using cultured rat Leydig cells and concluded that apoptosis of Leydig cells induced by galectin-1 was responsible for the reduction in steroidogenesis. Another in vitro study using porcine granulosa cells showed that addition of galectin-1 to the culture medium downregulated the FSH-stimulated expression of both 3β-HSD and cytochrome P450-dependent cholesterol side-chain cleavage enzymes necessary for progesterone synthesis (Walzel et al. 2004). In the present study, CL with an intense expression of galectin-1 showed only a faint expression of 3β-HSD in agreement with the idea that galectin-1 downregulates progesterone production. Although expressions of galectin-1 and −3 were not synchronized in CL, both galectins may act as negative regulators of progesterone by inhibiting progesterone production and accelerating progesterone degradation through the regulation of 3β-HSD and 20α-HSD expressions, respectively. The mechanism for the inhibitory regulation of steroid synthesis by galectin remains to be elucidated. We conducted a preliminary examination of mRNA expressions of receptors for FSH, luteinizing hormone, progesterone, and estrogen; however, no significant relationship could be found in the expression patterns of these receptors and galectins (Nio and Iwanaga, unpublished data). Further investigation into the regulatory molecules of galectin expressions is in progress in our laboratory.
In conclusion, the present study revealed galectin-1 and −3 expressions in the non-pregnant, pregnant, and postpartum ovaries of mice. Galectin-1 and −3 were intensely expressed in CL at particular regression stages, and expression of galectin-3 synchronized with the expression of 20α-HSD, a degradation enzyme of progesterone. Galectins in luteal cells may mediate progesterone production and metabolism via different mechanisms, and CL represents favorable material for research on the regulation of steroid production by galectin.
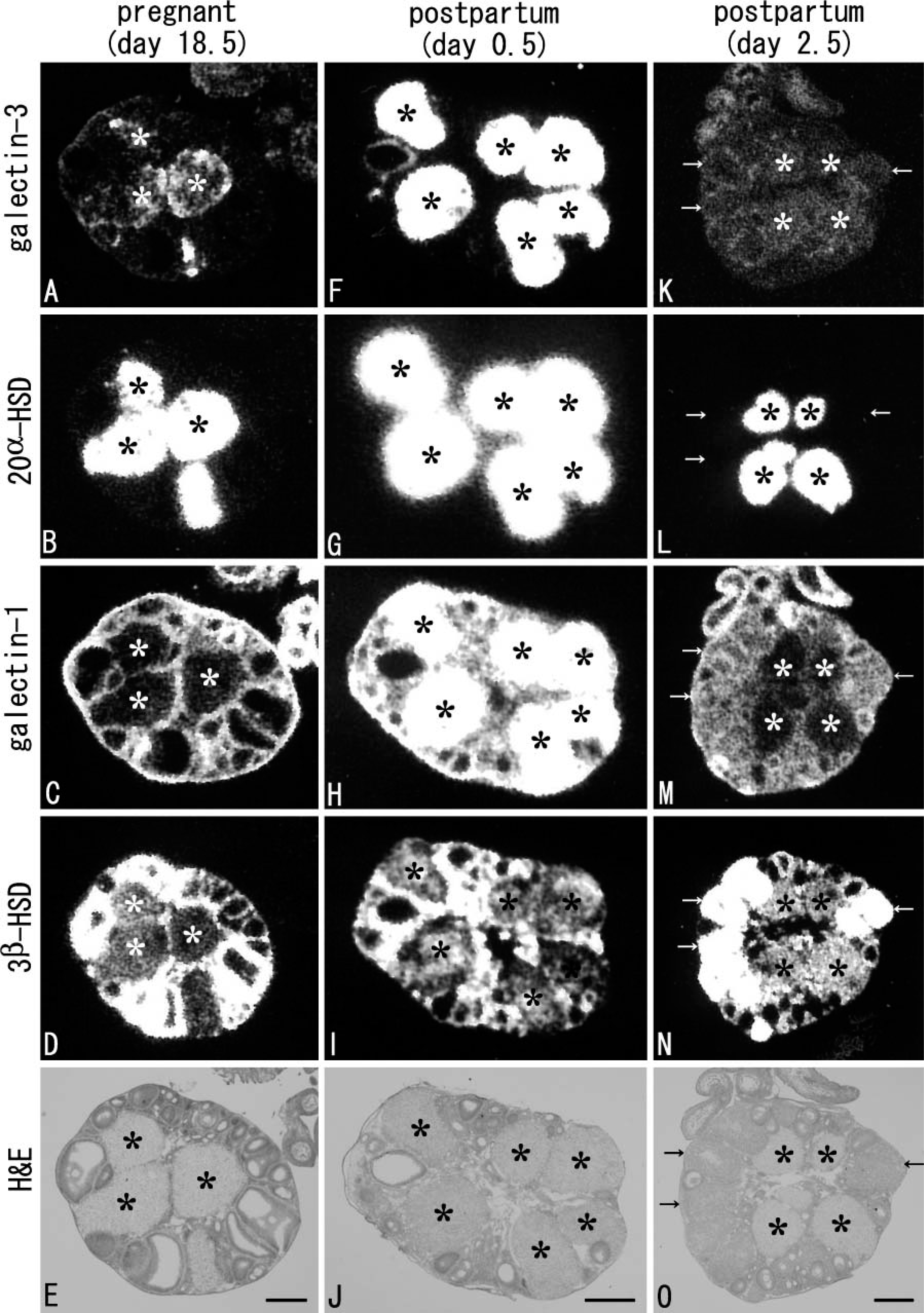
X-ray film images of the expressions of galectins, 20α-HSD, and 3β-HSD in serial sections of the ovary at perinatal stage (gestational day 18.5:
Footnotes
Acknowledgements
This work was supported by a grant from the Research Fellow of the Japan Society for the Promotion of Science (#17.9019 to JN) and a grant from the Ministry of Education, Science, Sport, and Culture, Japan [Grant-in-Aid for Scientific Research (B) #15390052 to TI].
