Abstract
Previous studies have indicated that ErbB receptors for neuregulins play an important role in cardiac development and muscle spindle formation during embryogenesis; however, little is known about their functions in adulthood. Recent reports indicate that breast cancer therapy with humanized monoclonal ErbB2 antibody induces cardiomyopathy, suggesting that ErbB2 serves as a crucial signaling receptor, even in the adult heart. Here, we examine ErbB2 expression and localization in both cardiac and skeletal muscle of adult mice via immunoblot and immunohistochemistry. ErbB2 was detected as a band ~185 kD molecular mass in each cardiac and skeletal muscle extraction. Confocal images of double labeling showed that ErbB2 was colocalized with caveolin-3 in cardiac muscle and with dihydropyridine receptor in skeletal muscle, suggesting that ErbB2 was localized at the T-tubule. In addition, immunoelectron micrographs clearly demonstrated that ErbB2 was located at the T-tubule in both types of muscle. Taken together, the results of the present study suggest that neuregulin-ErbB2 signaling plays a role in the physiological function of cardiac and skeletal muscle, even in adulthood.
In the present study, we clearly demonstrate that ErbB2 is localized at the T-tubule not only in the cardiac muscle but also in the skeletal muscle of adult mice.
Materials and Methods
Animals
Adult ddY mice used in this study were cared for and handled in compliance with the guidelines of the Institution of Experimental Animals at Shinshu University School of Medicine.
Antibodies
Rabbit polyclonal ErbB2 (sc-284) antibody was purchased from Santa Cruz Biotechnology (Santa Cruz, CA); mouse monoclonal caveolin-3 (610420) antibody was obtained from BD Biosciences (Lexington, KY). Mouse monoclonal ryanodine receptor (RyR; MA3-925), and mouse monoclonal dihydropyridine receptor (DHPR; MA3-920) antibodies were purchased from Affinity Bioreagents, Inc. (Golden, CO). Mouse monoclonal myosin slow-type heavy chain (MHC-S) was purchased from Ylem S.R.L (West Nightcott Dulverton, United Kingdom). Immunofluorescence antibodies were purchased from Molecular Probes Inc., (Eugene, OR) including Alexa 488 or 594 conjugated to streptavidin, goat anti-mouse Ig or goat anti-rabbit Ig.
Immunohistochemistry
Immunohistochemistry was performed as in our previous study (Ueda et al. 2004). Briefly, adult mice anesthetized with sodium pentobarbiturate were perfused with 4% paraformaldehyde. Subsequently, the quadriceps muscles were removed and immersed in the fixative overnight at 4C. After being rinsed in phosphate-buffered saline (PBS), they were immersed in 30% sucrose for cryoprotection overnight at 4C, embedded in optimal cutting temperature compound and sectioned at 10-μm thickness in a cryostat. Cryosections were treated with 1% Triton X-100 in PBS and 10% goat serum, incubated with primary (1:100) and secondary (1:500) antibodies, and observed with a confocal laser scanning microscope (LSM510, Carl Zeiss Japan Co.; Tokyo, Japan). For the control, we used PBS instead of primary antibodies following the procedure described above.
Immunoelectron Microscopy
Cryosections at 10-μm thickness fixed with 4% paraformaldehyde were treated with 1% Triton X-100 and 1% H2O2 in PBS for 30 min. Subsequently, the sections were incubated with 10% goat serum for 30 min and rabbit polyclonal ErbB2 antibody (1:100) overnight at 4C, followed by incubation with biotin-conjugated goat anti-rabbit Ig and streptavidin coupled with horseradish peroxidase using the Histofine Kit according to the manufacturer's protocol (Nichirei; Tokyo, Japan). After being rinsed in PBS, they were treated with metal-enhanced DAB substrate (Amersham Biosciences Corp.; Piscataway, NJ) and 1% osmium tetroxide for 30 min, dehydrated in a graded series of ethanol and acetone, and embedded in Epoxi resin by the inverted gelatin capsule method. Ultrathin sections at 70-nm thickness were counterstained with only uranyl acetate and were observed with a JEM-1200 electron microscope (Nihondenshi Co.; Tokyo, Japan).
Immunoblotting
Cardiac and quadriceps muscles were chopped into small pieces and homogenized in 10 mM N-(2-hydroxylethyl) piperazine-2′-2-ethanesulfonic acid (HEPES) buffer at pH 7.4, containing 150 mM NaCl, 1 mM EDTA, 1% IGEPAL-630, 1 mM PMSF, 1 μg/ml pepstatin, and 2 μg/ml aprotinin at 4C (10% w/v), respectively. The homogenates were centrifuged at 10,000 × g for 10 min at 4C and the supernatant used as the postnuclear fraction. Total protein concentrations were determined using the Lowry method. Thirty micrograms of each protein in the postnuclear fraction were resolved on 7% SDS-PAGE under reducing conditions with 5% 2-mercaptoethanol and electrotransferred to a nitrocellulose sheet for 30 min at 15 V. After blocking with 5% skim milk, the sheets were incubated with polyclonal ErbB2 antibody (1:500) overnight at 4C. They were then incubated with peroxidase conjugated to goat anti-rabbit Ig, and visualized using an electrochemiluminescence immunoblotting detecting system (Amersham Pharmacia Biotech Inc.; Buckinghamshire, United Kingdom).
Results
First, we examined whether cardiac and skeletal muscle express ErbB2 with immunoblotting. Figure 1 shows that an immunoreactive band was recognized at ~185 kD molecular mass in each extraction. These findings indicate that ErbB2 is constitutively expressed in adult cardiac and skeletal muscle. Double-immunolabeling experiments were conducted to explore ErbB2 subcellular localization in cardiac muscle. Confocal images labeled with ErbB2 and MHC-S demonstrated that ErbB2 localization was exclusively distinct from that of MHC-S (Figure 2A-C). Furthermore, caveolin-3 was colocalized with ErbB2, suggesting that ErbB2 is located at the T-tubule (Figure 2D-F). Endocardial cells were also labeled with ErbB2 (data not shown). On the other hand, skeletal muscle was examined by double immunolabeling with ErbB2 and RyR or DHPR. Figures 3A and 3D clearly show that ErbB2 was found in a stripe fashion, but not detected in the plasma membrane. In addition, ErbB2 was found between RyR striations (Figure 3A-C), while ErbB2 was almost overlapped in DHPR labeling (Figures 3D and 3E). Muscle spindle was weakly labeled with ErbB2, which is consistent with previous studies (Andrechek et al. 2002; Leu et al. 2003), but the neuromuscular junction (NMJ) was not recognized (data not shown). Immunocontrol sections showed ErbB2 labeling in neither cardiac muscle nor skeletal muscle (data not shown). Taken together with these findings, it is likely that ErbB2 is localized at the T-tubule in both cardiac and skeletal muscle in adulthood. Immunoelectron microscopic examinations were performed to determine whether ErbB2 is localized at the T-tubule. Immunoreactive deposits were found in endocardial cells at a low-power field (Figure 4A). At high-power fields, ErbB2 was seen around the T-tubule in cardiac muscle (Figure 4B) and skeletal muscle (Figure 4C).
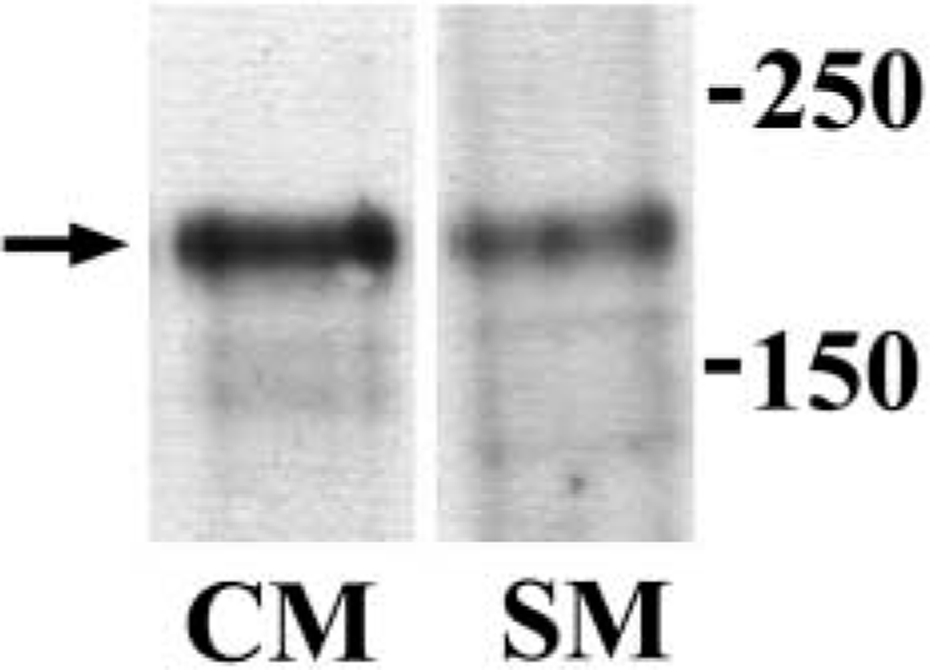
Immunoblotting of ErbB2 in cardiac and skeletal muscle extractions. The arrow indicates an immunoreactive band ~185 kD molecular mass in each lane. Numbers on the right-hand side indicate molecular mass. CM, cardiac muscle; SM, skeletal muscle.
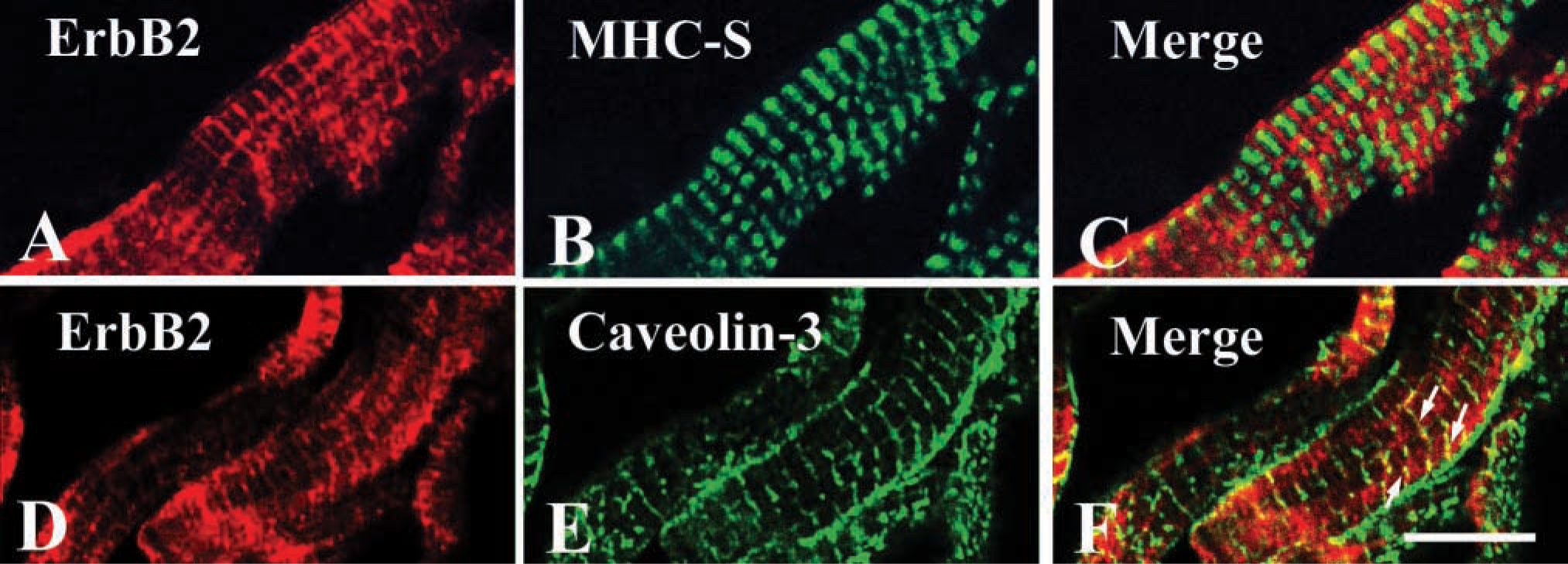
Confocal images of cardiac muscle labeled with (
Discussion
The present study clearly demonstrates that ErbB2 is localized at the T-tubule in adult murine cardiac as well as skeletal muscle. These findings support the hypothesis that neuregulin-ErbB2 receptor signaling plays a role in physiological function, even in adulthood.
It has been widely accepted that ErbB2 overexpression in mammary carcinomas yields a poor prognosis (Slamon et al. 2001). Humanized mouse monoclonal antibody [trastuzumab (Hereceptin)] is an effective agent for the regulation of ErbB2 receptor activity in metastatic breast carcinoma, but it promotes the development of dilated cardiomyopathy in combination with anthracycline (Slamon et al. 2001; Yarden and Sliwkowski 2001). Conditional ErbB2 mutants survived to adulthood but developed severe dilated cardiomyopathy arising usually in the second month of age (Crone et al. 2002; Ozcelik et al. 2002). Ozcelik et al. (2002) first demonstrated that ErbB2 was localized at the T-tubule in cardiac muscle as well as skeletal muscle using confocal microscopy, an observation that is supported by immunoelectron microscopy experiments in the present study. ErbB receptors are known as regulators of many signaling cascades, involving ras/mitogen-activated protein kinase, phosphatidylinositol-3 (PI-3) kinase/Akt, rho/rac, and phospholipid C (Yarden and Sliwkowski 2001). Many studies have shown that PI-3/Akt kinases activate to produce NO through endothelial NO synthase (Albrecht et al. 2003; Massion and Balligand 2003; Mungrue et al. 2003), and Lemmens et al. (2004) recently reported that neuregulin-1 has a negative inotropic effect and activates endothelial NO synthase in isolated rabbit cardiomyocytes. Accordingly, it is likely that neuregulin-ErbB2 plays a physiological role in adult cardiac muscle at the T-tubule, probably through NO production.
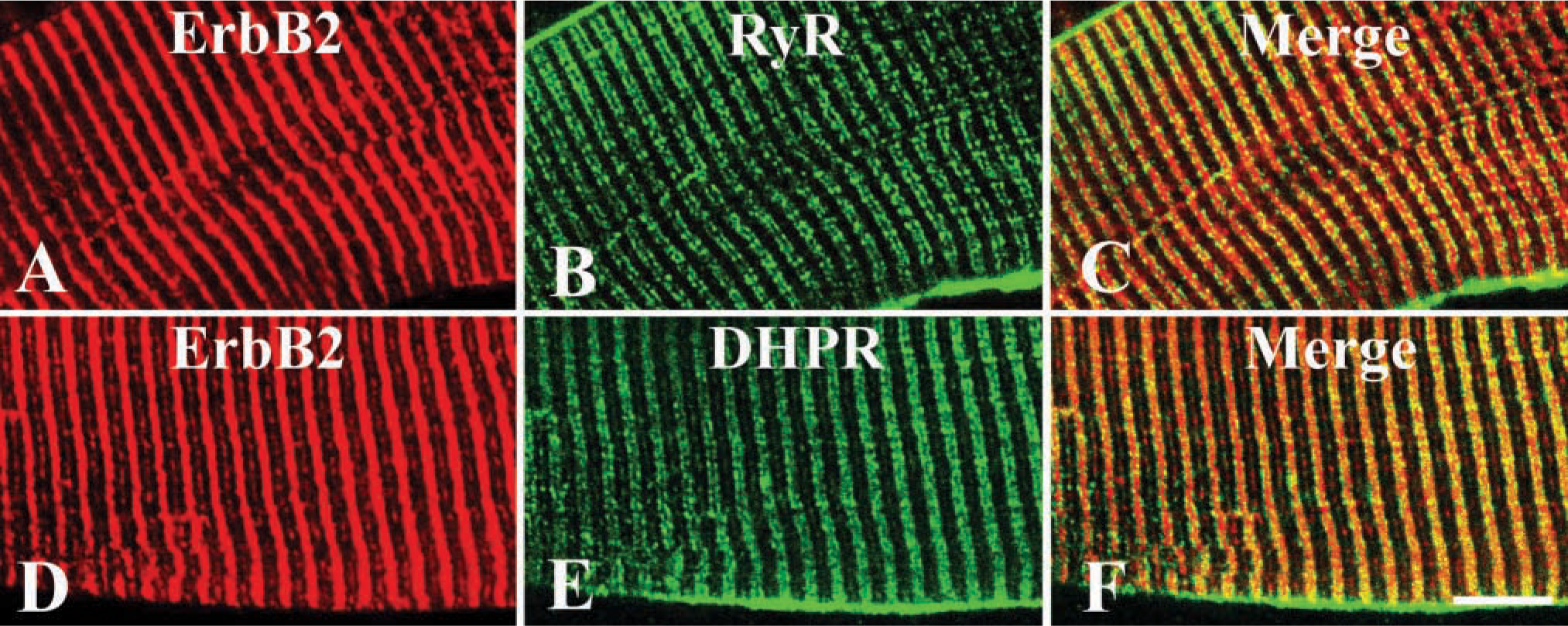
Confocal images of skeletal muscle labeled with (
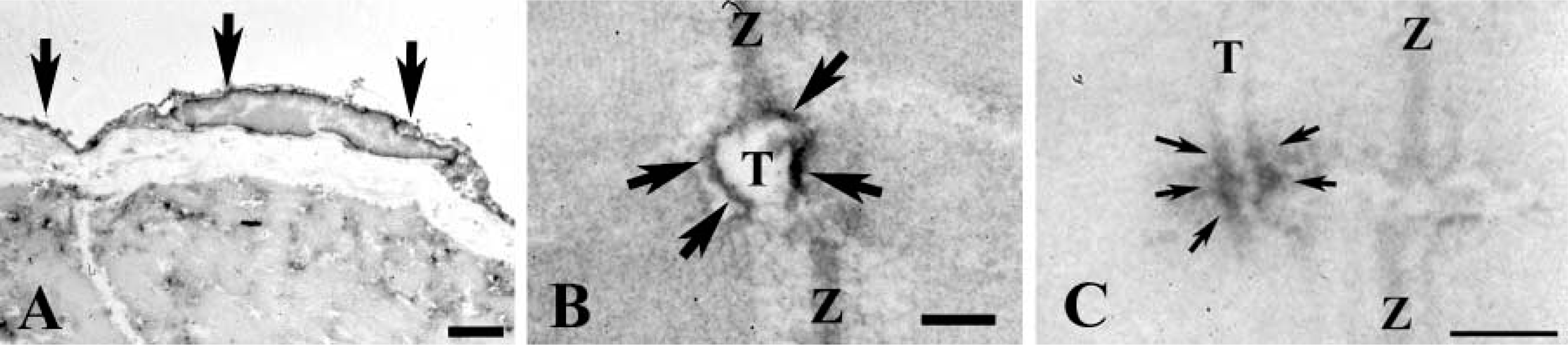
Immunoelectron micrographs labeled with ErbB2. At low magnification, immunoreactive deposits are found in the endocardial cell (arrows in
It has been suggested that neuregulin-1, which is produced in the endocardium, signals in a paracrine manner to the ErbB2/ErbB4 receptor heteromers present in the juxtaposed myocardium during heart development (Meyer and Birchmeier 1995). Additionally, exogenous neuregulin-1 was shown to convert working cardiac muscle to specialized cardiac muscle between 8.5 and 10.5 days postcoitum (Rentschler et al. 2002). Although we did not determine whether ErbB2 was found in specialized cardiac muscle in the adult, it was interesting that endocardial cells expressed ErbB2 (present study). Presently, we have no idea why the endocardial cell expresses ErbB2, but it should be borne in mind that specialized cardiac muscle has no T-tubule. There might be a distinct correlation between the adult and embryonic heart with regard to neuregulin signaling between endocardial cells and specialized cardiac muscle, because it is unlikely that the signaling is attributed to the development of the cardiac conduction system in adulthood. We favor the conclusion described by Crone et al. (2002) that the role of ErbB2 in the adult heart is distinct from its role during embryogenesis.
On the other hand, where is ErbB2 localized, and how does it function in skeletal muscle? Altiok et al. (1995) reported that ErbB2 was concentrated at the NMJ, and Lin et al. (2000) concluded that ErbB2-deficient mice died at birth because of abnormal NMJ development at the diaphragm. These studies indicate that ErbB2 is necessary for NMJ formation during embryogenesis. However, transgenic mice with inactivated ErbB2 produced by gene ablation have shown normal NMJ formation, although the acetylcholine receptor level was decreased (Leu et al. 2003). In our present study, we could not find NMJ labeled with ErbB2. At present, we do not have a good explanation for the discrepancy between our findings and those of previous studies (Altiok et al. 1995; Lin et al. 2000), but the possibility that ErbB2 expression in NMJ might be reduced in adult skeletal muscle cannot be excluded. Andrechek et al. (2002) reported that mice devoid of ErbB2 in skeletal muscle had proprioceptive defects caused by a lack of muscle spindle, and induced apoptosis in myoblasts upon differentiation to myofibers. Indeed, we were able to find muscle spindles labeled with ErbB2, but their labeling was not as strong as that of the T-tubule. This finding suggests that ErbB2 expression decreases in adult muscle spindles. Taken together, and as documented in cardiac muscle, these results indicate that it is probable that ErbB2 function in adult skeletal muscle is different from that in the embryo.
In conclusion, we have demonstrated that ErbB2 localizes at the T-tubule not only in cardiac muscle but also in skeletal muscle of adult mice. These findings support the hypothesis that neuregulin-ErbB2 signaling plays a role in the muscle contraction-relaxation mechanism. Further studies may shed light on a new therapeutic approach to cardiac and skeletal muscle diseases.
Footnotes
Acknowledgements
This study was supported in part by a grant from the Ministry of Education, Science, Sports, and Culture, Japan (HU; grant number 13470363).
