Abstract
We have proposed to divide intestinal metaplasia (IM) into two categories, i.e., a mixed gastric and intestinal (GI) type, and a solely intestinal (I) type, based on the residual gastric phenotype cells. The GI-mixed-type IM can be identified by the presence of both cells with either gastric or intestinal phenotypes in a single gland. This study is conducted to elucidate whether cells in the GI-mixed-type IM glands can simultaneously present both gastric and intestinal phenotypes. MUC5AC, MUC2, CD10 and villin expressions were investigated in 20 samples from five gastric cancer cases, directly using either AlexaFluor 488- or 568-labeled specific monoclonal antibodies and observed by fluorescent microscopy and confocal laser-scanning microscopy. GI-mixed IM glands comprise a population expressing MUC5AC and MUC2, MUC5AC and villin, and MUC5AC and CD10. MUC2 and villin expressions were reciprocally increased with decreasing MUC5AC expression, while CD10 expression was limited to cells with only a residual MUC5AC expression or no expression. These results suggest that a heterogeneous cell population with both gastric and intestinal phenotypes would develop into a single intestinal phenotype, as reflected in the progression of intestinal metaplasia from GI-mixed-type- to I-type IM-type glands.
According to this classification, I-type IM glands are solely comprised of intestinal phenotypic cells, whereas GI-mixed-type IM glands also contain gastric phenotypic cells. Interestingly, although the number of gastric cells varies as much as those in a gastric-predominant or an intestinal-predominant type, GI-mixed-type IM glands appear to gradually become I-type IM glands (Inada et al. 1997; Tatematsu et al. 2003). The exact mechanisms by which these two kinds of phenotype cells or phenotype shifts come to be produced have yet to be determined, but at least two explanations are possible. The first is that aberrantly expressed Cdx1 and/or Cdx2 transcriptional factors, mammalian homologs of the caudal-related homeobox genes, may play an important role in such processes (Silberg et al. 1997; Mizoshita et al. 2001; Tsukamoto et al. 2003; Yuasa 2003). The other would be that some genetic alterations such as methylation occur in stem cells leading them to supply such various cell types, especially as seen in GI-mixed-type IM in our classification, resulting in the phenotype change caused by their accumulation (Kang et al. 2001,2003a,b; Kim et al. 2004; Lee et al. 2004).
Immunohistochemical techniques are widely used to identify intestinal and gastric cell differentiation for the classification of gastric cancers and IM (Inada et al. 1997; Reis et al. 1999; Inada et al. 2001; Jass and Walsh 2001; Mizoshita et al. 2001; Silva et al. 2002; Kawachi et al. 2003; Tatematsu et al. 2003). To date, a preferred method to evaluate IM and gastric cancers utilizes the anti-mucin core proteins 5AC (MUC5AC) and 6 (MUC6) together with anti-CD10, anti-villin, and anti-MUC2 antibodies. Mucin core proteins comprise an expanded gene family consisting of at least 19 members (Tanaka et al. 1991; Gum et al. 2002; Chen et al. 2003; Ringel and Lohr 2003), of which MUC5AC, MUC6 and MUC2 genes are homologous to each other and are localized at chromosome 11p15.5 within a 400-kbp gene span (Pigny et al. 1996; Winterford et al. 1999). Their expressions might be differentially regulated by the restricted MUC5AC presence on surface epithelial cells (Reis et al. 1997), MUC6 on cells in the glandular compartment of pyloric mucosa (Ho et al. 1995; Reis et al. 2000), and MUC2 in the goblet cells of small and large intestines, and on IM (Jass 2000). A secreting endopeptidase, CD10 (Landry et al. 1994; Sezaki et al. 2003), and one of the actin-binding cytoskeletal proteins, villin (Landry et al. 1994; MacLennan et al. 1999; Pinto et al. 1999), are also observed in intestinal cells, whose expressions indicate absorptive-cell differentiation in IM (Landry et al. 1994). Expressions of these molecules are widely used to evaluate gastric cancers whether the differentiation direction is toward gastric or intestinal phenotype.
Using an adaptation of this approach to investigate IM glands, it has been demonstrated that small populations of MUC2-positive cells containing either MUC5AC or MUC6 are present in IM glands (Ho et al. 1995; Inada et al. 1997,2001; Reis et al. 1999,2000; Tatematsu et al. 2003). However, it remains unclear whether these glands, probable GI-mixed IM glands, are composed of dual phenotype cells with intestinal and gastric differentiation. In the present study we evaluated the co-expression of MUC5AC and MUC2, MUC5AC and villin, and MUC5AC and CD10 in GI-mixed-type IM glands using multiple immunofluorescent staining techniques at the single-cell level. The combined use of these markers succeeded in providing evidence of cells with both gastric and intestinal phenotypes in IM.
Materials and Methods
Tissue Samples
Twenty normal stomach tissue samples were obtained from five patients with gastric cancer who underwent gastrectomy at Aichi Cancer Center Hospital. They were cut from normal areas more than 10 cm away from the cancer, immediately frozen in Tissue-Tek OCT (Optimal Cutting Temperature) Compound (Sakura Finetechnical Co. Ltd.; Tokyo, Japan) with liquid nitrogen and then stored at −80C until use. Four-μm-thick frozen sections prepared with a cryostat were fixed in cold methanol and dried at room temperature for use in immunohistochemical analysis.
Antibodies
Table 1 shows the characteristics of mouse monoclonal antibodies (MAbs) used in this study. To specifically detect the immune reactions with two respective mouse MAbs, we employed Zenon Mouse IgG-labeling kits to directly label the MAbs with either AlexaFluor 488 or AlexaFluor 568 (Molecular Probes; Eugene, OR). Fluor-labeled MAbs were prepared immediately prior to use, according to the suppliers' protocols. The optimal concentrations of primary MAbs were determined empirically, and the final concentrations were 1:100 of anti-MUC5AC, 1:100 of anti-CD10, and 1:5000 of anti-villin MAbs. In all cases, isotype-matched monoclonal antibodies were used as a negative control.
Immunofluorescent Staining
Four μm-thick frozen sections were fixed in cold methanol for 10 min, then air-dried at room temperature for 30 min and rehydrated in PBS for 15 min at room temperature. To reduce nonspecific bindings, the sections were incubated with a blocking reagent (PBS containing 0.2% Triton X-100, 0.2% BSA, and 5% heat-inactivated normal goat serum) for 30 min at room temperature and then reacted with a mixture of two primary antibodies labeled with either AlexaFluor 488 or 568 for 2 hr at room temperature. After washing twice with PBS containing 0.2% Triton X-100 for 15 min, the sections were incubated with 4′,6-diamidino-2-phenylindole (DAPI, Molecular Probes) to stain the nucleus for 1 min at a dilution of 1:20000. After washing with PBS, the sections were mounted in Mowiol 4–88 Reagent (CALBIOCHEM; San Diego, CA). To remove O-linked glycosylation on MUC2 for Ccp58 anti-MUC2 antibody to bind the epitope, we performed alkali-catalyzed β-elimination on the frozen section, the same as previously reported (Hong and Kim 2000).
Antibodies used in this study

Immunofluorescence and Confocal Laser-scanning Fluorescence Microscopy
Multicolor-stained tissues with AlexaFluor 488, AlexaFluor 568, and DAPI were observed using an Olympus BH5 fluorescence microscope (Olympus; Tokyo, Japan) equipped with a xenon arc lamp and an appropriate filter set. Confocal laser scanning was performed with the Radiance 2100 K-3 system (BioRad; Clinisciences S.A., Montrouge, France) that employs optical fibers both in the illumination source and the detection aperture. This system was equipped with a 50-mW Crypton-Argon laser and filters allowing excitation with both a 488-nm and a 560-nm laser line. Two channels were available for simultaneous data acquisition: Channel 1 (displayed as green) could use a 510–550-nm bandpass filter and either a 515-nm or a 530-nm longpass filter, while Channel 2 (displayed as red) could include either a 550-nm or 590-nm longpass filter.
Image Analysis
All images were recorded by a digital video camera and converted to TIFF files. Merged images were made using Adobe Photoshop software (Adobe Systems; San Jose, CA).
Results
Since all of the monoclonal antibodies used in this study were IgG1, we employed a Zenon antibody-labeling kit to detect two antigens simultaneously. This technique makes it possible to individually detect several co-expressed antigens when using the same isotype antibodies. We conjugated anti-MUC2 mAb, anti-villin mAb and anti-CD10 MAb with AlexaFluor 488, while anti-MUC5AC was conjugated with AlexaFluor 568 (anti-CD10 MAb was also conjugated with AlexaFluor 568 in some cases). Their specific bindings (Table 1) were not modified by the Zenon-labeling procedure.
Co-expression of MUC2 and MUC5AC on GI-mixed-type IM
As shown in Figure 1, intestinal and gastric differentiation makers were detected in IM glands. In the first series of experiments using such specimens, we examined the expression of MUC2 as a marker for intestinal goblet cells and that of MUC5AC as a marker for gastric-surface columnar cells. Surface columnar epithelial cells exhibited MUC5AC without MUC2 expression in normal gastric mucosa, while goblet cells of the intestinal or I-type IM exhibited MUC2 without MUC5AC, as demonstrated in previous studies (Figures 2A and 2C) (Inada et al. 1997,2001; Tatematsu et al. 2003). GI-mixed-type IM glands, which were identified by the presence of both MUC5AC and MUC2 in a single gland (Figure 2B), demonstrated both the antigens in glands with a differential positive cell ratio. MUC2-positive cells showed a goblet cell-like feature with MUC5AC expression that included the MUC2 epitope in their cytoplasm of determined GI-mixed-type IM glands (Figure 2B). This result indicates that GI-mixed-type IM glands include cells sharing both gastric and intestinal phenotypes. Interestingly, the co-expressed MUC2 and MUC5AC epitope was not completely colocalized in their cytoplasm. To examine the differential cellular localization between MUC2 and MUC5AC in GI-mixed-type IM cells in more detail, we used a confocal laser-scanning microscope. MUC2 was clearly observed at the center of the cytoplasm, which was surrounded by the MUC5AC-expressing area (Figures 3A-3F), suggesting that MUC2 and MUC5AC are sorted in a differential manner.
Furthermore, MUC2 antigen appeared in the supra-nuclear region of goblet cells in I-type IM glands (Figure 2C), whereas its distribution appeared more diffusely in the mucous vesicle of GI-mixed type IM cells (Figure 2B). The alteration in MUC2 distribution seems to arise from the difference between I-type IM and GI-mixed-type IM cells. To elucidate the possibility that higher glycosylation reduced antibody binding in goblet cells of I-type IM the same as in the colonic mucosa previously described (Hong and Kim 2000), alkali-catalyzed β-elimination was performed to remove glycosylation. Following the procedure, increased anti-MUC2 MAb reactivity was observed, which was MUC2 staining similar to that in GI-mixed-type IM glands (Figures 3G and 3H). These results suggest that MUC2 in I-type IM cells is more abundantly glycosylated than in GI-mixed-type IM. The cellular distribution of MUC5AC showed no apparent changes between GI-mixed-type IM cells and normal gastric surface columnar cells.
Co-expression of MUC5AC and Villin/CD10 in GI-mixed-type IM
In a second set of experiments, we examined MUC5AC as a marker for gastric surface columnar cells and either villin or CD10 as the other markers for intestinal cells to verify whether the GI-mixed-type IM glands exhibit both intestinal and gastric phenotypes. Both villin and CD10 have been widely used as specific markers to identify intestinal absorptive cells and are exclusively expressed at the brush borders of cells solely in I-type IM glands but not in gastric glands (Figures 4 and 5). GI-mixed-type IM glands were identified by the presence of MUC5AC and villin in individual glands (Figure 4). Villin was detected on the apical end of MUC5AC-positive cells in GI-mixed-type IM glands, whose expressed cell numbers increased with diminishing MUC5AC expression in the glands. It is noteworthy that CD10 expression was not always detected with villin at the brush border in GI-mixed IM glands (Figure 6) but was also occasionally localized in the cytoplasm (Figure 5). Its inclusion bodylike appearance was the same as that reported in familial microvillus-inclusion body disease (Groisman et al. 2002). This inclusion body-like CD10 staining pattern is more apparent in GI-mixed-type IM glands with more abundant MUC5AC-expressing cells, but it disappears with declining MUC5AC expression.
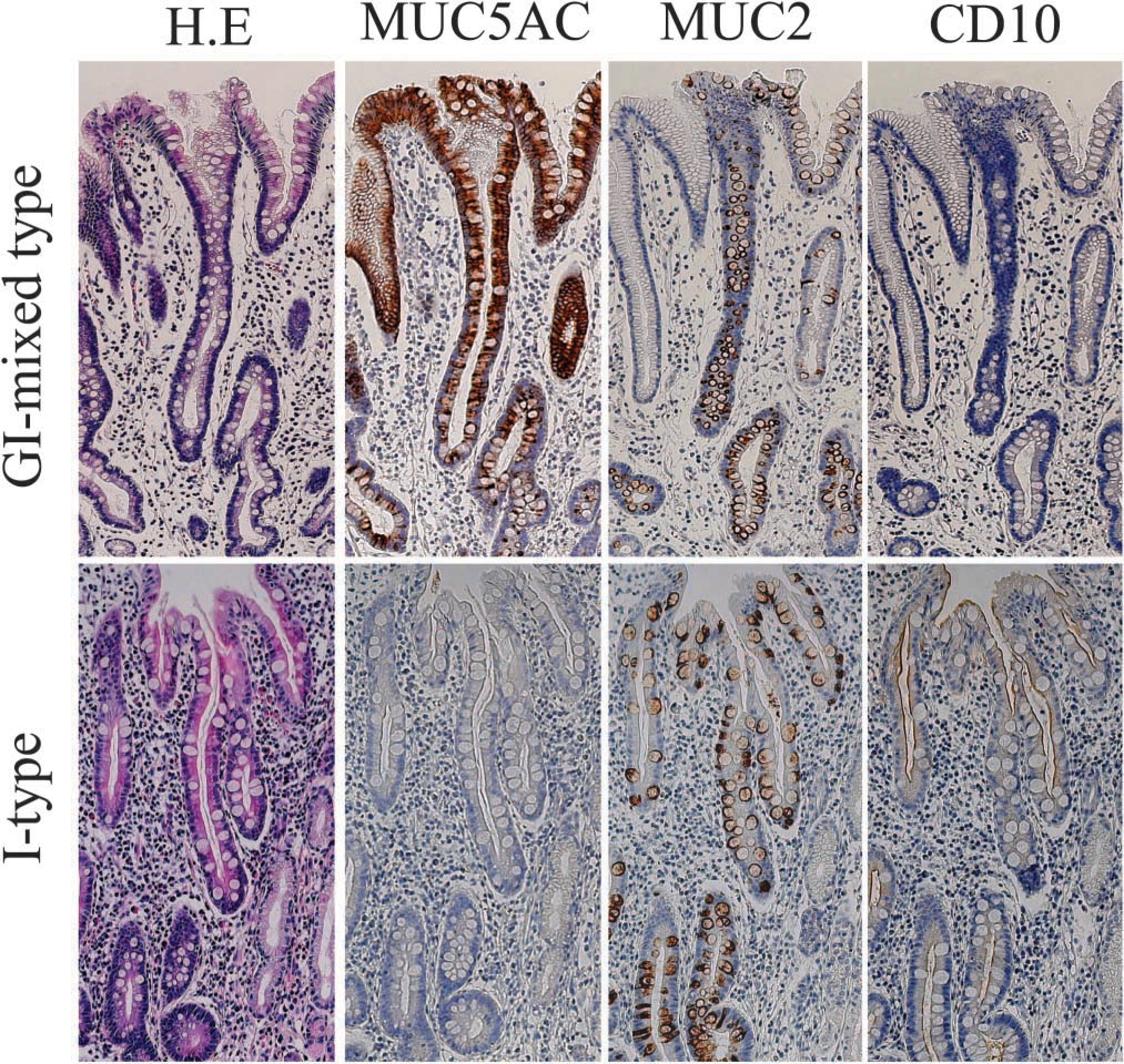
Immunnohistochemical analysis for IM glands with sequential sections. IM glands were detected by the expression of either CD10 or MUC2. Solely I-type IM glands express the intestinal markers without MUC5AC expression (lower panel, ×40), while GI-mixed-type IM glands express the intestinal markers with MUC5AC (upper panel, ×40).
Discussion
To date, several immunohistochemical studies have demonstrated an intestinal or a gastric phenotype by the detection of specific molecular expressions in IM glands using serial sections (Ho et al. 1995; Inada et al. 1997, 2001; Reis et al. 1999; Jass 2000; Shaoul et al. 2000; Silva et al. 2002; Tatematsu et al. 2003). These studies have attempted to demonstrate that some cells are either intestinal or gastric phenotypic markers, suggesting limited possibilities for the same cells to have both gastric- and intestinal-marker antigens (Reis et al. 1999, 2000; Lopez-Ferrer et al. 2000,2001; Shaoul et al. 2000). In the present study, we were able to overcome such limitations by employing Zenon antibody-labeling technology, thus successfully demonstrating the co-expression of both gastric and intestinal molecular markers in the same cells in GI-mixed-type IM glands, in which MUC5AC clearly existed with MUC2, or villin and/or CD10. These findings suggest that mixed gastric and intestinal type metaplasia is formed by cells with dual differentiation and are consistent with the previously demonstrated evidence that some metaplastic cells have both intestinal and gastric differentiation-specific structures (Goldman and Ming 1968).
The cells in GI-mixed-type IM glands exhibited MUC2 with MUC5AC in their cytoplasm (Figures 2 and 3), while the cells in I-type IM glands exhibited MUC2 in the peri-nuclear Golgi apparatus area. The subcellular localization of MUC2 appeared to shift from secretion vesicles to the Golgi apparatus area with a histological alteration from GI-mixed-type IM to I-type IM. A glycosylation change might be one of the probable explanations, since the MUC 2 antibody used in this study could detect only underglycosylated MUC2 core protein for the epitope as discussed in previous studies (Hong and Kim 2000; Shaoul et al. 2000). In the present study we observed the enhanced anti-MUC2 antibody staining in I-type IM glands by alkali-catalyzed β-elimination, which was similar to the MUC2 staining on GI-mixed-type IM glands. This result indicates that O-linked glycosylation limited the detection of MUC2 expression in goblet cells in I-type IM glands, suggesting that the difference between the higher glycosylated MUC2 in I-type IM glands and the lower glycosylated MUC2 in the goblet cells in the GI-mixed-type IM glands would be the other maturation indicator for goblet cells in IM glands.
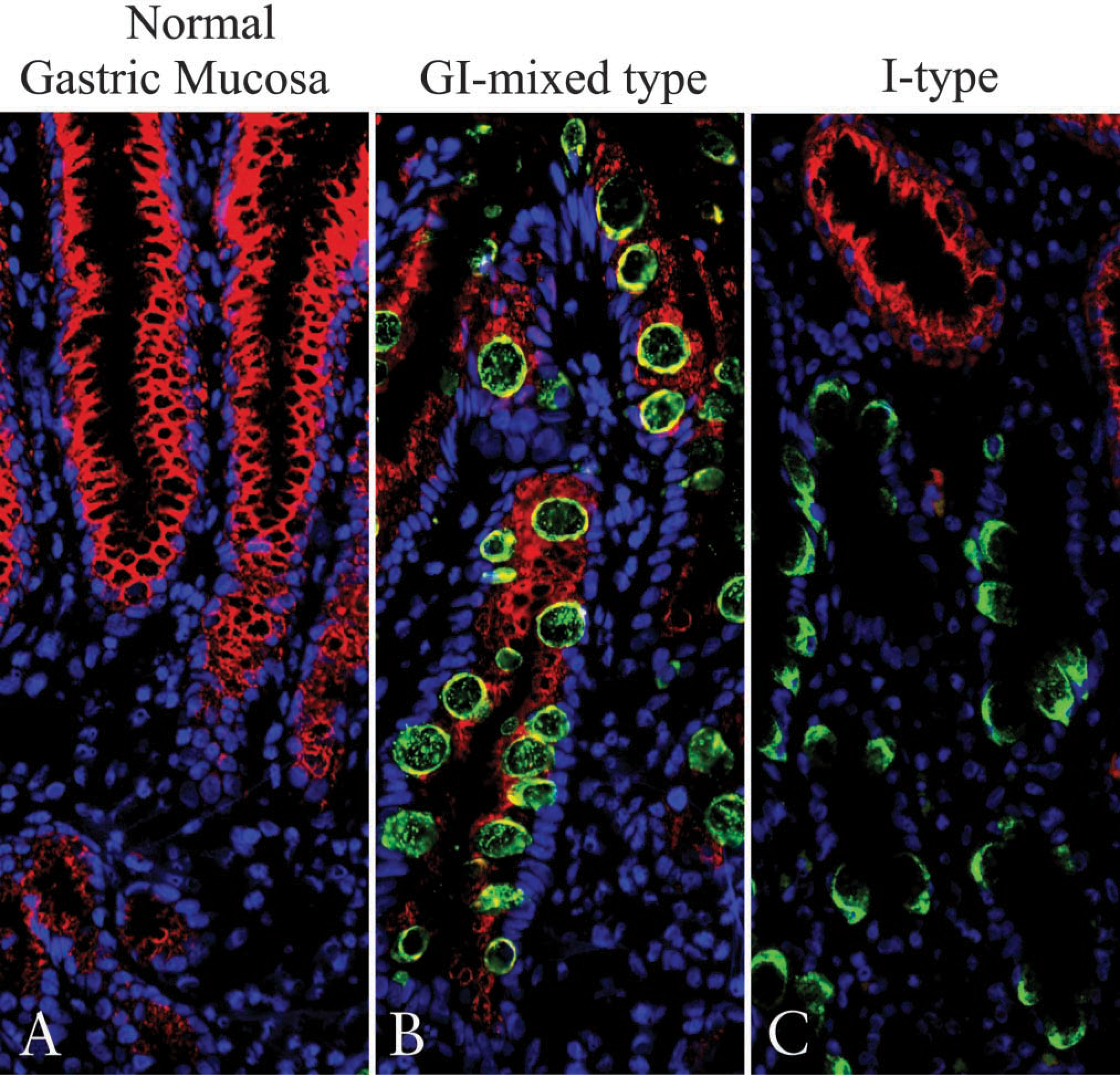
Localization of MUC5AC and MUC2 mucin core protein in normal gastric glands and IM glands. Frozen sections were stained with two mouse MAbs against MUC5AC directly labeled with AlexaFluor 568 and MUC2 with AlexaFluor 488 as described in Materials and Methods. Nuclei were counter-stained with DAPI (blue) and observed by fluorescence microscopy. MUC5AC (red) and MUC2 (green) mucin core protein expressions were observed in the cytoplasm of foveolar epithelial cells in normal gastric mucosa (
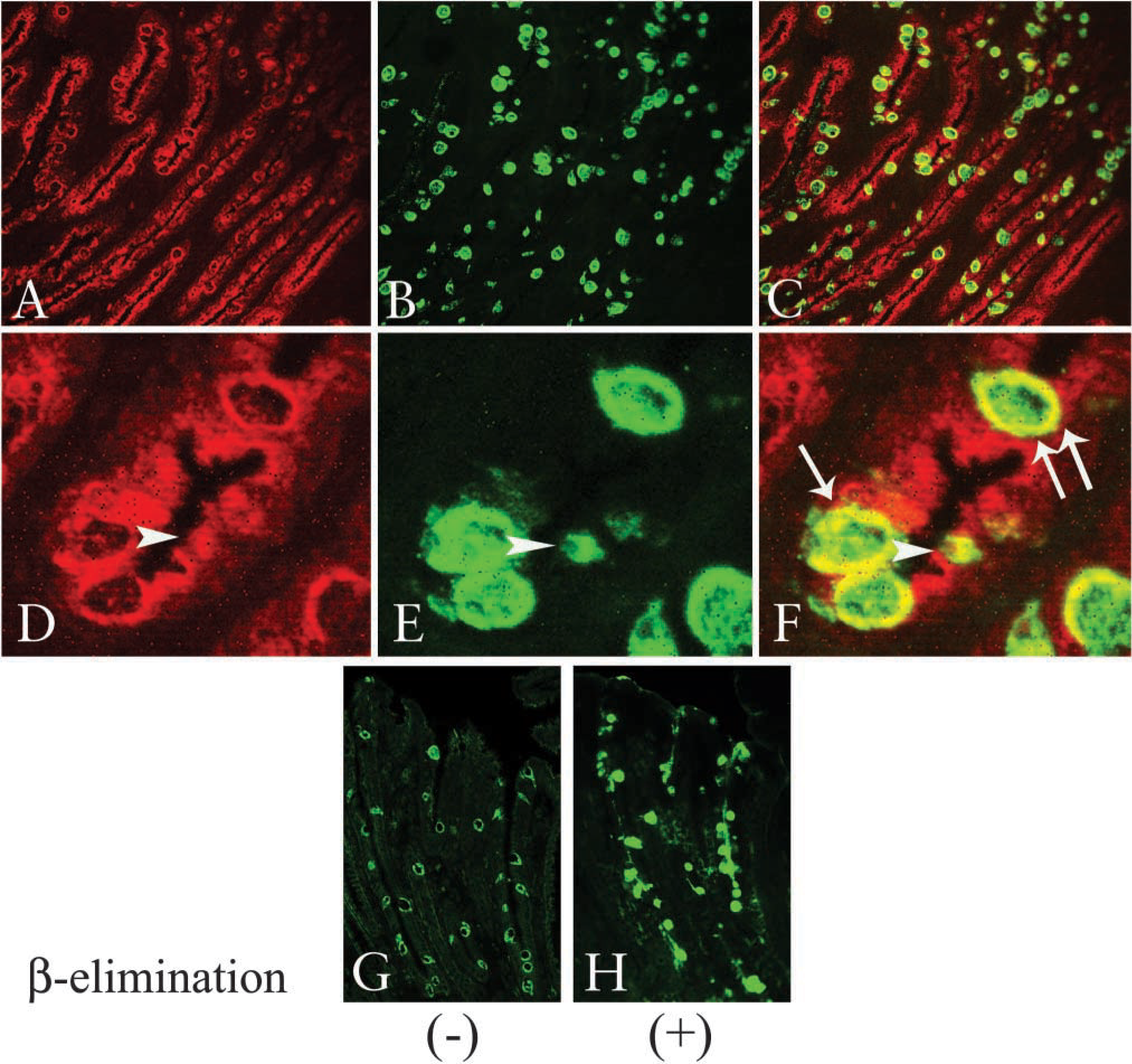
Colocalization of MUC5AC and MUC2 mucin core proteins in GI-mixed-type IM glands. Frozen sections were stained by the double-immunofluorescence method using two mouse MAbs against MUC5AC and MUC2 directly labeled with AlexaFluor 568 or 488. MUC5AC (red) and MUC2 (green) expressions in GI-mixed-type IM glands were observed by confocal laser scanning microscopy (
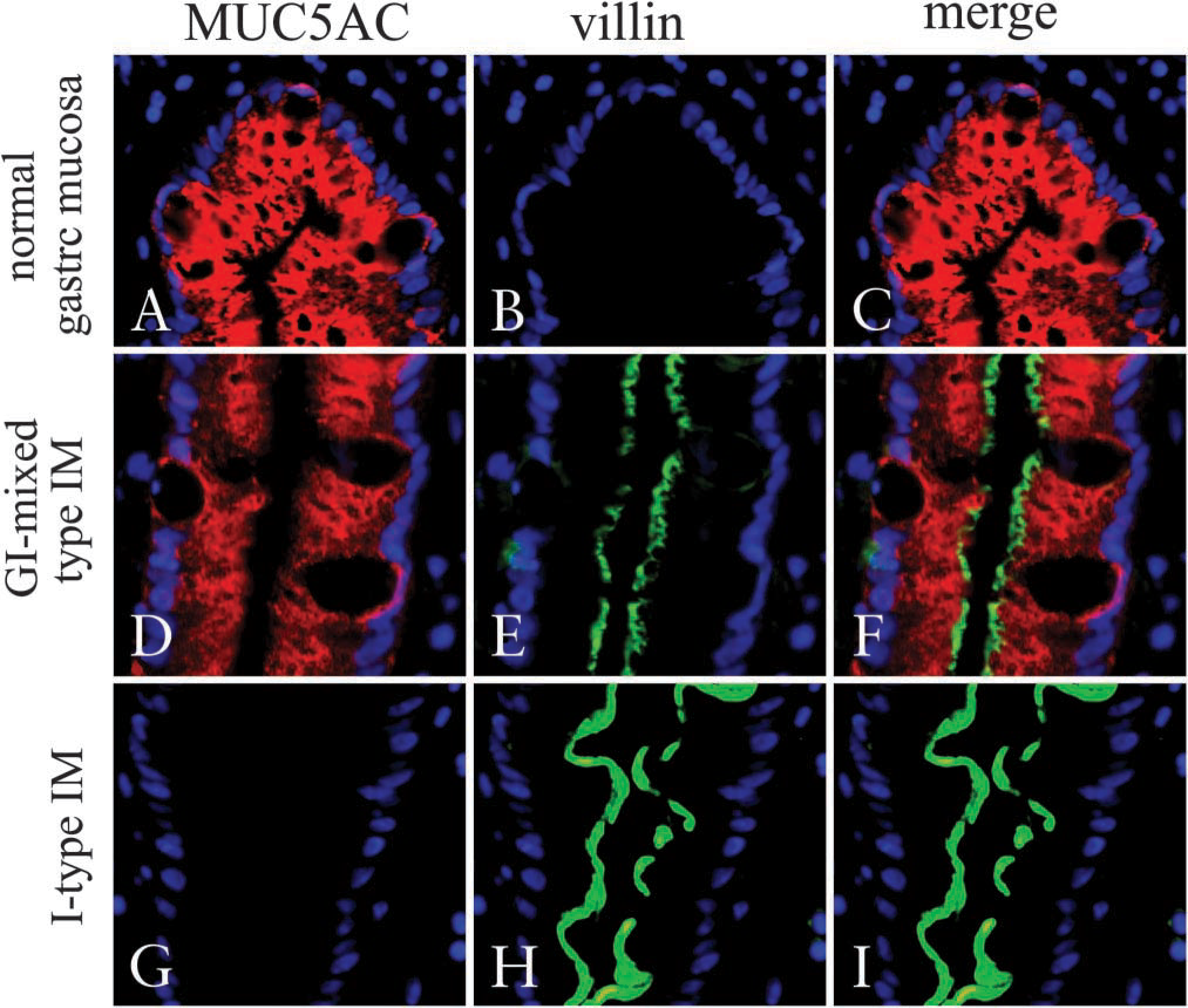
Expression of MUC5AC and villin in normal gastric glands and GI-mixed-type IM glands. Frozen sections were stained by the double-immunofluorescence method using two mouse MAbs against villin and MUC5AC directly labeled with AlexaFluor 488 or 568. Nuclei were counter-stained by DAPI (blue) and observed by fluorescence microscopy. (
The columnar epithelial cells, similar to the intestinal absorptive cells, were seen in GI-mixed-type IM glands that exhibited MUC5AC and either villin or CD10. Compared with villin-positive cells, CD10 was preferentially found in the faint MUC5AC-preserved columnar cells, suggesting that CD10 exists in cells of the more intestinalized GI-mixed-type IM glands (Figure 6). Analogously, in the development of fetal mouse small intestine, villin appears on the brush border prior to CD10 (Landry et al. 1994; Montgomery et al. 1999). Villin is an actin-binding cytoskeletal protein essential for brush border formation in normal epithelial cells of the intestine, while CD10 is a brush border-associated neutral peptidase. Adapting these findings to human IM, the structural accomplishment of IMs such as villin expression might precede by functional maturations such as digestive enzyme, CD10 expression. Moreover, it may be reasonable to assume that villin-positive and CD10-negative cells in GI-mixed-type IM are functionally immature absorptive cells, and that the phenotype shift from GI-mixed-type IM to solely I-type IM is a kind of maturation. Furthermore, the expression of CD10 in the cytoplasm of columnar cells in GI-mixed IM (Figure 5) might support this idea, because it has been found in the surface enterocytes of familial microvillus inclusion disease due to the immaturity of CD10 (Groisman et al. 2002).
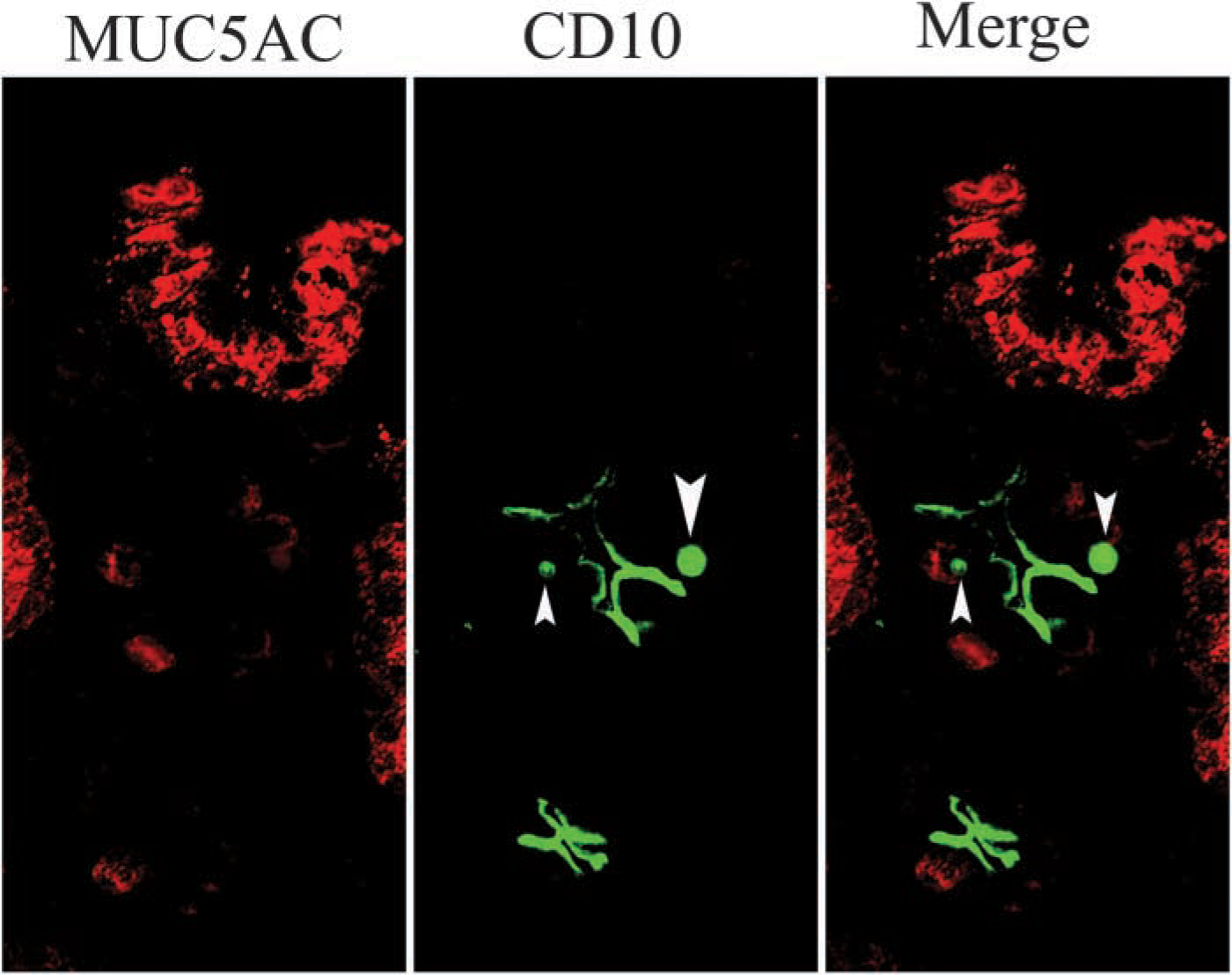
Cellular localization of CD10 in GI-mixed IM glands. Frozen sections were stained by the double-immuno-fluorescence method using two mouse MAbs against MUC5AC and CD10 directly labeled with AlexaFluor 568 and 488, respectively. This figure was obtained by fluorescence microscopy. CD10 (green) was expressed in the microvilli of columnar cells without MUC5AC expression (red), while CD10 stained as inclusion body-like signals in MUC5AC-positive cells. Arrowheads indicate CD10-positive inclusion bodies (X60).
Co-expression ratios of MUC5AC to MUC2, villin, or CD10 varied greatly in terms of establishing a dominant MUC5AC type or a MUC2/villin/CD10 dominant type. Based on these findings, the cells in mixed-type IM glands seem to be about to free themselves from the gastric phenotype, finally becoming solely I-type IM cells. As villin, CD10, and MUC2 expressions progressively increase, MUC5AC expression reciprocally diminishes. Gastric-type cells are gradually reduced and finally replaced by intestinal-type cells, leading to solely I-type IM. These results indicate that GI-mixed-type IM cells are multi-phenotypic, suggesting that the phenotype shift from a GI-mixed type to a solely I-type IM should occur in each cell over time (Figure 7).
In conclusion, we demonstrated that intestinalization occurs in individual cells with MUC5AC expression in GI-mixed-type IM glands. The cells with an intestinal phenotype in GI-mixed type IM glands are morphologically and functionally less mature than those in I-type IM glands. As they are midway between gastric and intestinal phenotypic cells with varying degrees of differentiation and maturation, the GI-mixed-type IM glands consist of a heterogeneous population of cells. Based on observations in this study, we hypothesize that the cells in GI-mixed-type IM glands remain out of some regulations on intestinal differentiation and subsequent functional maturation toward becoming intestinal type cells. Furthermore, considering that stem cells are also present in GI-mixed-type IM, such unstable phenotypes might be induced at stem cells by either transcriptional factors or DNA methylation. To clarify and confirm these possibilities, further studies based on molecular biological techniques and applying our findings will have to be initiated starting with the identification of GI-mixed IM cells.
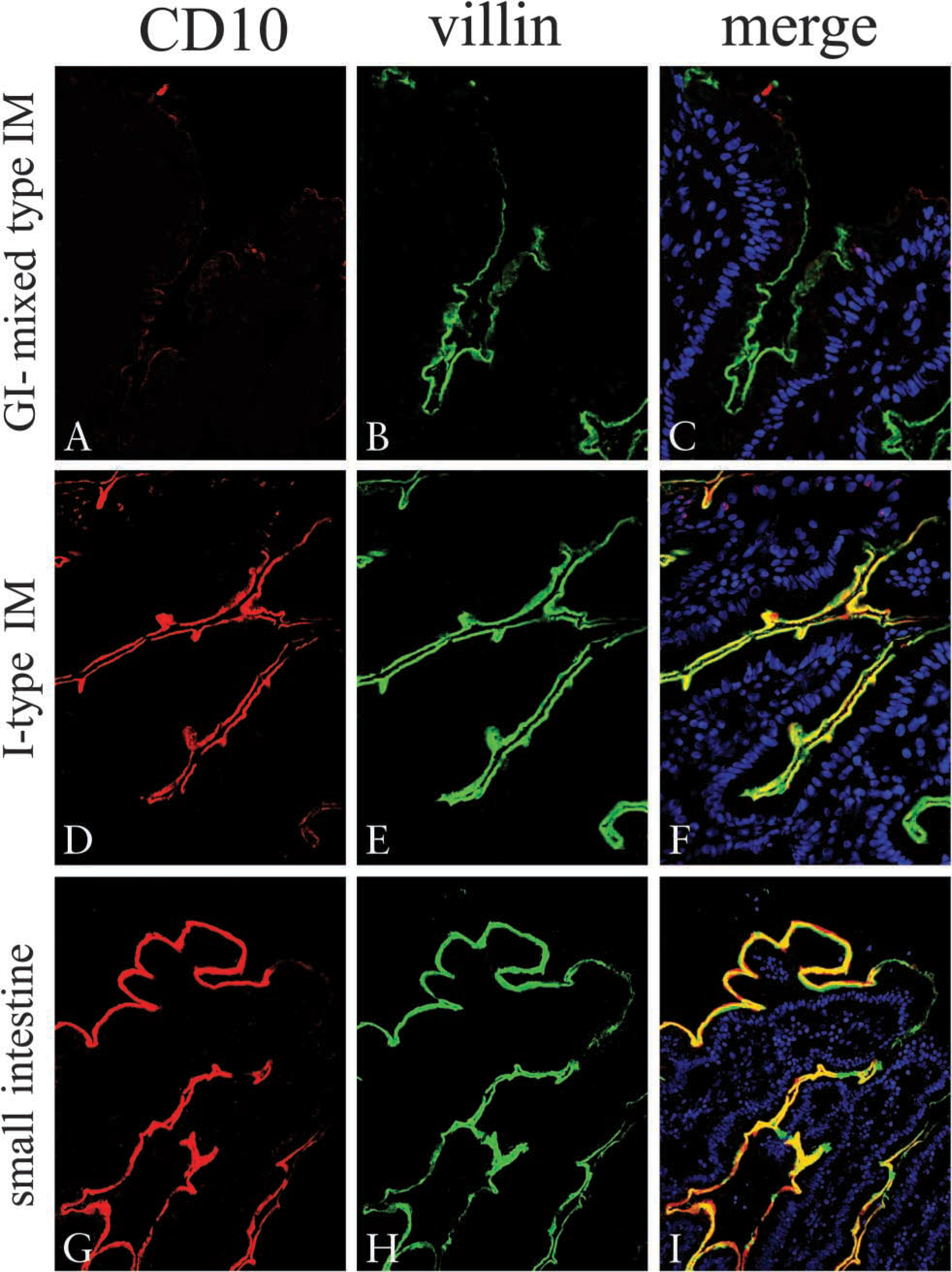
Expression of villin and CD10 in GI-mixed IM glands. Frozen sections were stained by the double-immunofluorescent method using two mouse MAbs against villin and CD10 directly labeled with AlexaFluor 488 and 568, respectively. Nuclei were counter-stained by DAPI (blue) and observed by fluorescence microscopy. CD10 (red;
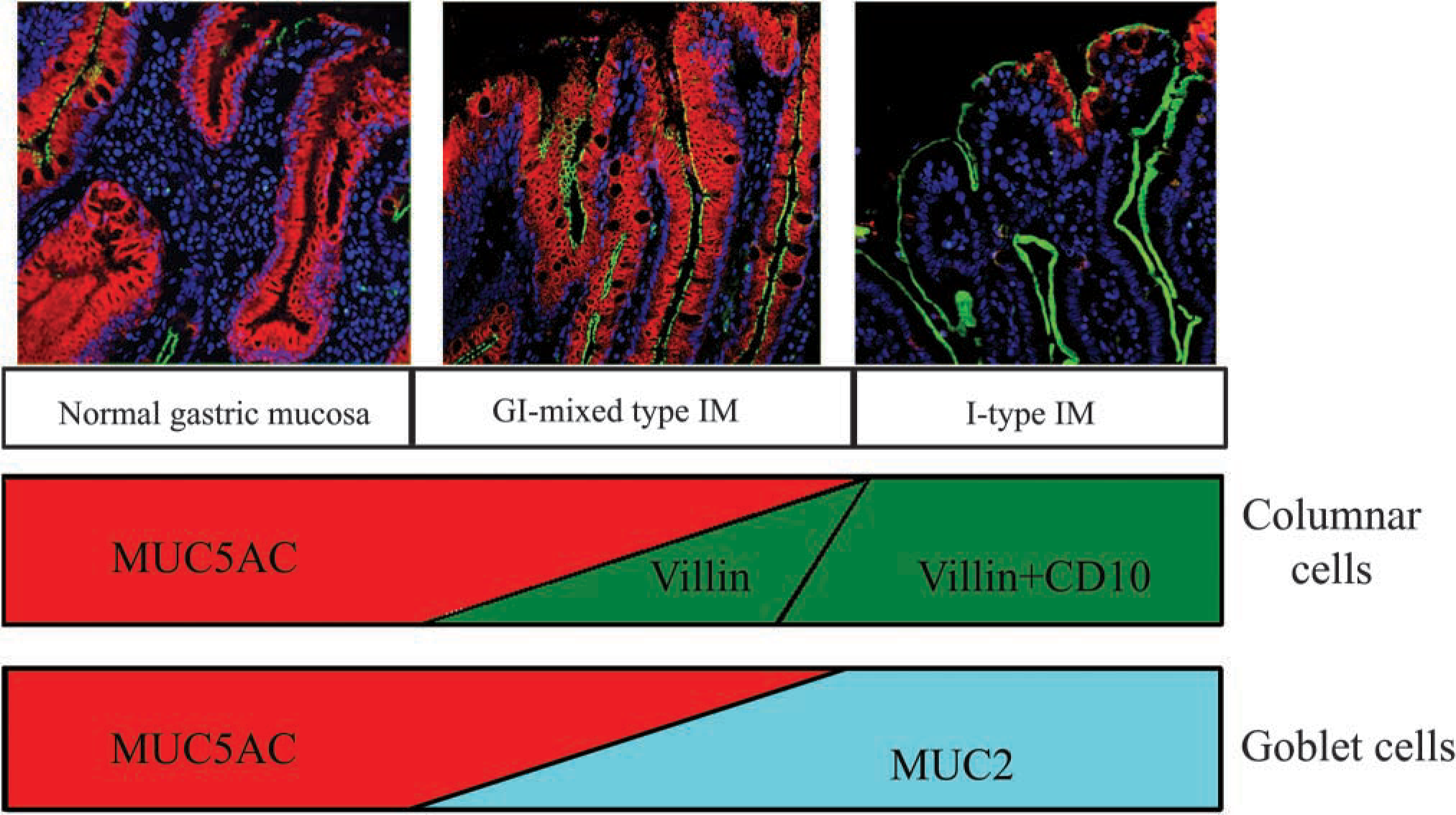
Schematic representation of phenotypic shift of differentiation markers from GI-mixed-type IM to I-type IM glands. Gastric foveolar epithelial cells express only MUC5AC. IM begins from such foveolar epithelial cells, along two different cellular pathways. One begins with an aberrant expression of villin and subsequent CD10 on columnar epithelial cells, resulting in absorptive-like intestinal cells. The other starts from an ectopic expression of MUC2 and then accumulates in mucous vesicles, developing into goblet-like intestinal cells. As villin, CD10, and MUC2 expressions progressively increase, MUC5AC expression reciprocally diminishes. Gastric-type cells are gradually reduced and are finally replaced by intestinal-type cells, leading to solely I-type IM (upper panel, ×20).
Footnotes
Acknowledgements
Supported by a Grant-in-Aid for Scientific Research on Priority Area (16790196) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
