Abstract
Laminin 5 is a trimeric glycoprotein involved in cell adhesion in the epidermal basement membrane. To determine the precise orientation of laminin 5 in adult human skin, we used plural epitope-specific monoclonal antibodies, a polyclonal antiserum, and postembedding immunogold electron microscopy (IEM). Immunogold labeling distances from the basal keratinocyte plasma membrane (PM) were measured for each gold particle (>200 particles) and the mean distance (nm) calculated. Antibodies included BM165 (recognizing the α3-chain first globular domain) that was measured at 35.40 ± 2.20 nm from the keratinocyte PM, K140 (recognizing a region adjacent to the β3-chain globular domain IV) that measured 45.20 ± 3.60 nm from the PM, and an anti-laminin 5 polyclonal antiserum that was 43.43 ± 6.28 nm from the PM. The laminin 5 γ2-chain short arm hinge domain was previously localized to the lower lamina densa (LD) at approximately 56.30 ± 1.65 nm from the keratinocyte PM. Taken together with previous γ2-chain data and the distribution of the polyclonal antisera, we estimate that the long axis of laminin 5 is oriented at an angle of approximately 27° from the horizontal lamina lucida (LL)/LD border and propose that the γ2-chain lies farthest from the PM. This novel orientation, with the majority of the laminin 5 molecule lying obliquely along the LL/LD border and not perpendicularly, as was first thought, sheds new light on the organization of the basement membrane and likely molecular interactions.
L
Previously, immunogold electron microscopy (IEM) has shown that laminin 5 is present in the lower lamina lucida (LL) (McGrath et al. 1994; Rousselle and Aumailley 1994). McGrath et al. used pre-embedding immunolabeling methods followed by routine EM processing to localize the GB3 laminin 5 γ2-chain antibody to the lower LL. However, this technique relies on good antibody-tissue penetration, which is not always possible with antibodies attached to colloidal gold particles or when inaccessible areas, including the basement membrane LD, are labeled. In such cases, postembedding labeling is a more reliable technique. We have reported, using postembedding methods, that laminin 5 localizes to the lower LL/LD border with the majority of labeling in the upper lamina densa (LD), including the γ2-chain labeling that was restricted to the mid-LD (Masunaga et al. 1996; Bedane et al. 1997; Kirtschig et al. 1998).
Laminin is believed to be involved in epidermal cell adhesion, polarity, and migration, and to act as a developmental signal for hemidesmosomal assembly (Aumailley and Rousselle 1999). The laminin 5 cell binding domain for the HD α6β4 integrin is believed to be located within globular domains of the α3-chain (Niessen et al. 1994; Aumailley and Rousselle 1999). Regions on the laminin 5 β3 short arm also bind to the fibronectin-like repeats on the noncollagenous 1 (NC-1) domain of collagen VII and fibulin 2 (Utani et al. 1997; Chen et al. 1999,2002). Therefore, the predicted laminin 5 orientation is with the long axis of the α3-chain perpendicular to the LD with the five globular domains in closest apposition to the keratinocyte (see Figures 3a and bi). We aimed to determine and compare a more precise distribution and position of laminin 5 globular domains of the α3-chain with a β3-chain domain-specific antibody and antiserum against the whole molecule using an IEM technique.
Materials and Methods
Skin Samples
Samples of normal skin from nonspecialized sites (abdomen and thigh from two individuals n = 2; n = 4 in total) were obtained at routine surgical procedures and were used for postembedding immunogold electron microscopy.
Indirect IEM
Samples of normal human skin were cryofixed and processed for postembedding IEM according to the methods previously described (Shimizu et al. 1989,1990). The samples were washed in PBS and cryoprotected in 20% glycerol (in PBS) for 1 hr at 4C. Cryofixation was in liquid propane at −190C using a Leica CPC freeze plunge apparatus (Leica; Cambridge, UK), freeze-substitution over 3 days at −80C in methanol using the AFS apparatus (Leica), and the samples were embedded in Lowicryl K11M resin (Ladd Research Industries; Burlington, VT) over 4 days at −60C. The temperature was gradually raised (by 5C/hr) while the resin was polymerized under UV light and liquid nitrogen vapor at 10C. Ultrathin sections were then cut and collected on pioloform-coated nickel grids. Sections were subsequently stained with uranyl acetate (15 min) and observed in a Hitachi H-7100 transmission electron microscope (Tokyo, Japan) at 75 kV. Blocks showing good ultrastructure were selected for immunolabeling experiments.
The sections were pre-incubated in buffer containing PBS with 5% normal goat serum (NGS), 1% bovine serum albumen (BSA), and 0.1% gelatin. The sections were then incubated with the mouse monoclonal antibody BM165 directed against the first globular (G1) domain of the laminin 5 α3-chain (1:50) (Rousselle et al. 1991b; Rousselle and Aumailley 1994; Champliaud et al. 1996; McMillan et al. 1997); K140 (also previously known as 6F12) directed against the laminin 5 β3-chain adjacent to domain IV (Marinkovich et al. 1992; McMillan et al. 1997), and a rabbit polyclonal serum directed against the whole laminin 5 molecule (1:200; see below) (see Table 1) or normal rabbit serum (1:500). The rabbit laminin 5 antiserum was produced by immunizing rabbits with sodium dodecyl sulfate (SDS)-extracted laminin 5 from third-passage cultured primary human keratinocytes. The anti-laminin 5 antiserum used in this study was tested on severe lethal (Herlitz) junctional epidermolysis bullosa (EB) skin (n = 3) and showed severely reduced or absent dermal-epidermal junction staining in all cases (data not shown). In addition, this anti-laminin 5 serum failed to react by indirect immunofluorescence on 6-week human fetal skin but showed staining from 9 weeks and onwards after laminin 5 expression had started and hemidesmosome assembly had begun. This confirms the specificity of this antiserum to laminin 5 and not any other laminin variants present in the human dermal-epidermal junction. The two laminin 5 monoclonal antibodies had also previously been described and used to stain EB patients' skin (n = 11) (McMillan et al. 1997).
Primary antibodies or antisera were all diluted in PBS buffer containing 1% NGS, 1% BSA, and 0.1% gelatin and incubated at 37C for 2 hr. The sections were then washed in a drop of PBS buffer four times for 5 min each and placed on a drop of secondary linker antibody, again diluted in PBS buffer (for 2 hr at 37C). Secondary antisera against mouse anti-rabbit IgG or rabbit anti-mouse IgG (Dako; Ely, UK) were diluted 1:500. Then the sections were incubated with a third antibody, 5-nm gold-conjugated labeled goat anti-rabbit or goat anti-mouse antibody (Biocell; Cardiff, UK) diluted 1:500 in Tris-buffered saline (TBS) for 2 hr at 37C. The sections were washed twice in TBS buffer and twice in distilled water (5 min each). After staining with 15% alcoholic uranyl acetate (15 min) and lead citrate (1 min), the sections were observed in a Hitachi H-7100 transmission electron microscope (Tokyo, Japan).
Immunogold Distribution Assessment
The techniques for ultrastructural measurements were similar to those previously performed (Masunaga et al. 1996; Bedane et al. 1997; McMillan et al. 2003). Electron micrographs were taken at a standard magnification (30K) and were enlarged by a standard factor ×2.08. The final magnification (×62,500) was checked using electron micrographs taken of a carbon diffraction grating. For purposes of standardization, all the measurements were made by one observer (J.R.M.). All measurements were in nm from the keratinocyte plasma membrane (values were positive for measurements below the plasma membrane and negative if above the plasma membrane). The number of gold particles measured was at least 200 (between 200 and 375) for each antibody or antiserum (Table 1). Approximately 20 individual micrographs covering more than 40 μm of basement membrane were scanned and the gold particle analyses performed for each epitope. Each antibody was labeled in duplicate using different skin samples (n=2) from two individuals and the data checked for differences and then pooled.
Immunogold laminin subunit labeling reveals the orientation and distribution of laminin chains in adult human epidermal basement membrane
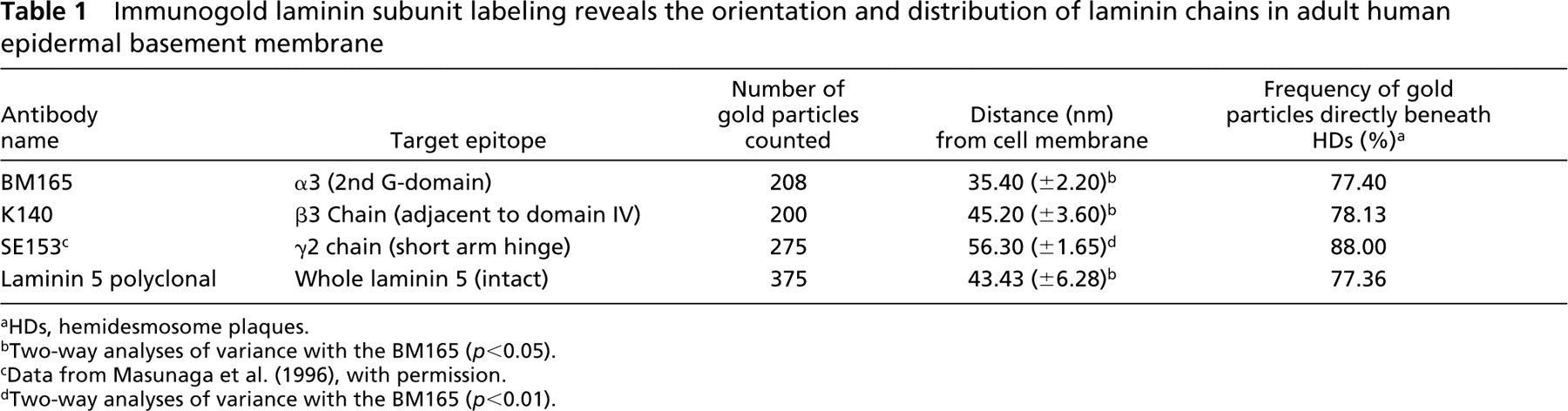
HDs, hemidesmosome plaques.
Two-way analyses of variance with the BM165 (p<0.05).
Data from Masunaga et al. (1996), with permission.
Two-way analyses of variance with the BM165 (p<0.01).
Only non-obliquely (perpendicular) sectioned areas of BMZ were included in the assessment with clearly defined LL and LD. The basement membrane beneath melanocytes or damaged areas was excluded from this study. The distance from the plasma membrane to the center of each gold particle was measured and recorded (Table 1). The mean distance of each gold particle from the plasma membrane was calculated in μm ± SEM (Table 1) using the Minitab statistical package (Minitab; University of Pennsylvania, State College, PA). Gold particles that appeared clumped or associated with any deposit were also excluded. Each gold particle was also scored for its location immediately beneath a hemidesmosomal plaque and the percentage of labeling beneath the HD for each antibody or antiserum was calculated and expressed as a percentage of the total basement membrane labeling (Table 1). Three statistical analyses tests were performed using the Minitab software package. Two-way analysis of variance, Student' t-test, and the Mann–Whitney confidence interval test were performed (at 95% and 99% confidence intervals) to compare the respective mean antigen distances.
Results
Normal Control Skin Labeling
Immunogold labeling for laminin 5 antibodies and antiserum showed strong signals in the basement membrane (Figures 1a–f). Immunogold particles (5 nm in diameter) were observed along the LL/LD boundary for the first α3-chain globular domain-specific antibody (BM165). This labeling showed an extended distribution, slightly higher in the mid-LL (Figures 1a and 1b) in contrast to the β3-chain antibody (K140) that showed relatively sparse labeling, the majority of which was restricted to the LD but also extended to the LL/LD border (Figures 1c and 1d). The polyclonal antiserum showed abundant labeling over the majority of the LD but also extended to the lower quarter of the LL (Figures 1e and 1f) in a similar fashion to BM165 (Figures 1a and 1b). The average values for both laminin 5 antibodies and the antiserum were calculated from measurements of the distances from gold particles to the plasma membrane (Table 1). The α3-chain first globular domain antibody was located some 10 nm above the β3-chain (35.4 ± 2.20 nm from the plasma membrane vs 45.2 ± 3.60 nm) and the polyclonal antiserum was located between these two values at 43.4 ± 6.28 nm below the keratinocyte plasma membrane (Table 1).
The distribution of all laminin 5 labeling is represented in graphic form for BM165 (Figure 2a), K140 (Figure 2b), and polyclonal laminin 5 (Figure 2c). The α3-chain globular domain showed a more restricted distribution range (15–75 nm) than both the β3-chain (K140; Figures 1c and 1d) and the polyclonal antiserum (Figures 1e and 1f). Estimates of the size of laminin 5 come from measurements made from rotary shadowing experiments (Marinkovich et al. 1992; Vailly et al. 1994) showing that it was approximately 110 nm in length and 45 nm from the β3 short arm to domain IV. Given the difference in the relative ultrastructural localization between the α3- and β3-chains and knowing the approximate dimensions of laminin 5, we used the Pythagorean theorem and simple triangle geometry to calculate the approximate angle of the long axis of laminin 5 relative to the LL (to approximately 27°) (Figure 3bii), as follows: right-angled triangle one side: a1 = 110 nm, b1 = 45 nm, c1 = 118 nm with the smallest angle θ1 = 22°; right-angled triangle two sides: a2 = 117 nm, b2 = 10 nm, c2 = 118 nm with the smallest angle θ2 = 5°.
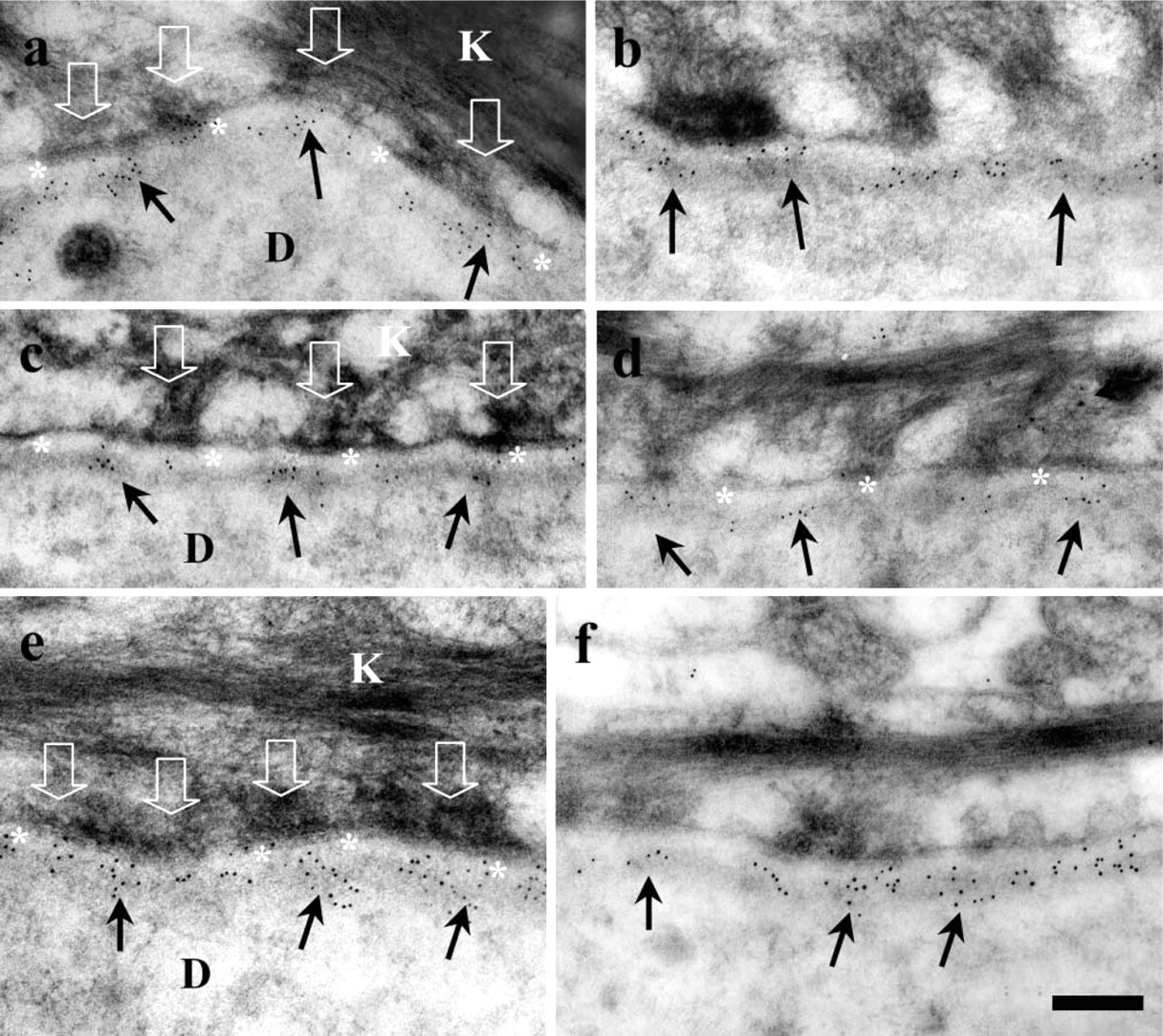
Postembedding IEM reveals a distinct labeling pattern for the α3 first globular (G1) domain (BM165;
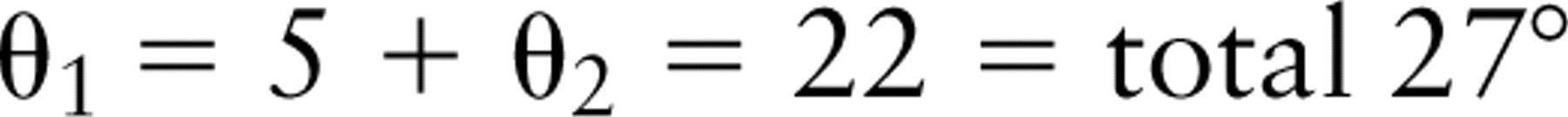
The samples of normal skin used in this and previous studies have demonstrated that the majority (75–88%) of laminin 5 labeling was restricted to immediately beneath the hemidesmosome plaque (Table 1). The two α3- and β2-chain antibodies and one serum showed a remarkably similar percentage labeling associated with the anchoring filament complex beneath hemidesmosomes, ranging between 77 and 78% (Table 1). There was no difference in the laminin 5 labeling distance between the samples from different body sites. Statistical analyses revealed differences between BM165 and both K140 and the whole polyclonal laminin 5 antiserum (p<0.05) and a highly significant difference between BM165 and SE153 (p<0.01; Table 1). K140 and the whole laminin 5 polyclonal serum were different from the SE153 staining (p<0.05).
There was no difference between K140 staining and the whole laminin 5 polyclonal serum (p>0.05).
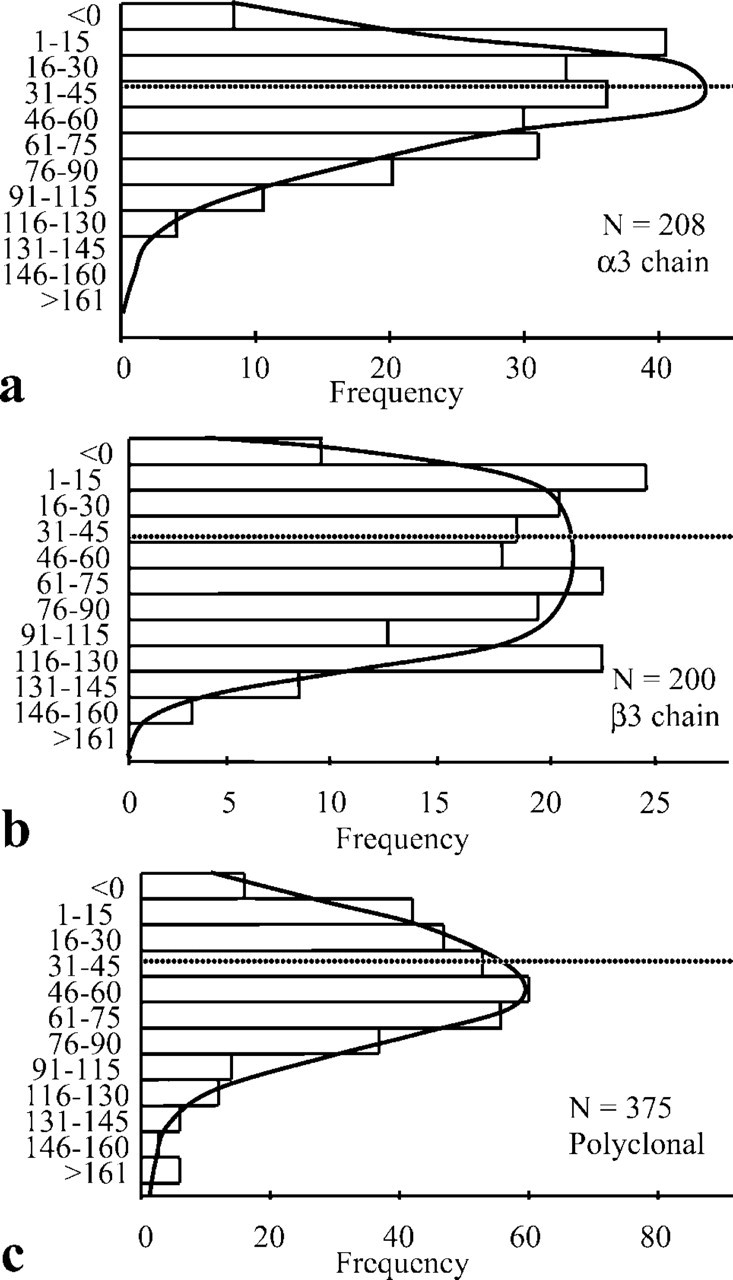
Distribution of laminin 5 labeling (nm) from the epidermal plasma membrane for different anti-laminin chain antibodies and antiserum (BM165,
Discussion
We have demonstrated in detail the localization of specific α3 and β3 domains and furthermore have suggested a new orientation of laminin 5 in the human basement membrane. The globular domains of the α3-chain of laminin 5 are localized to the lower quarter of the LL, closer to the epidermis than the majority of the laminin 5 molecule (using the γ2-chain and polyclonal antiserum for reference). Masunaga et al. (1996) also used a similar postembedding IEM method to localize the majority (66%) of all laminin 5 chain labeling to the LD. The γ2-chain short arm was measured at 56 nm from the plasma membrane (Masunaga et al. 1996) and an α3-chain epitope was mapped 38 nm from the plasma membrane (similar to our findings). We therefore hypothesize that laminin 5 is oriented with the α3-chain globular domains some 35–40 nm from the keratinocyte plasma membrane. The γ2-chain is even farther from the plasma membrane, in the lower LD. These data and previous reports (Rousselle et al. 1997) support the theory that the α3-chain long axis is obliquely aligned within the LL and LD.
Previous reports of the laminin 5 molecular shape using rotary metal shadowing of molecules isolated from cultured cells suggest that the α3-chain long axis is approximately 110 nm in length and that the length of the two β3 and γ2 short arms is between 60 and 70 nm (Rousselle et al. 1991b; Marinkovich et al. 1992; Vailly et al. 1994). Using careful measurement of the dimensions of laminin 5, the sites of epitope binding, and the Pythagorean theorem, it is possible to estimate the approximate angle at which the long axis of this molecule lies. We estimate that the long axis of the α3-chain of laminin 5 is at an angle of approximately 27° to the LD border, much more obliquely aligned than was previously thought (Figure 3). However, caution should be exercised in interpreting this data due to the potential flexibility of laminin 5 in vivo or the possibility that it exists in a shape other than the proposed cruciform shape as seen using rotary shadowing.
This hypothesis supposes that laminin 5 maintains a rigid cruciform structure but, as rotary metal shadowing studies can demonstrate, there may be parts of the laminin 5 molecule with considerable flexibility (Rousselle et al. 1991a; Marinkovich et al. 1992; Vailly et al. 1994; Aumailley and Rousselle 1999). Indeed, the flexibility observed in the short arms of laminin 5 may explain the wider range of labeling observed for the short arm of the β3-chain compared with the α3 or γ2 domains that may be stably anchored to the hemidesmosomal integrin α6β4 (Figures 1c and 1d vs Figures 1a and 1b). Taken together with the previous data (Masunaga et al. 1996) of the γ2-chain labeling deeper within the LD (Table 1) and the β3-chain labeling in the lower LL, this suggests that some molecular flexibility may be involved. Alternatively, laminin 5 may have a rigid structure but may pivot about one or more fixed points that may include the α3-chain α6β4 integrin cell binding globular domains or about a separate collagen VII, nidogen or fibulin 2 binding domain (Chen et al. 1997,2002; Utani et al. 1997). A close association between laminin 5 and the extracellular portion of the bullous pemphigoid antigen 2 (BP180) has also been proposed (Hopkinson et al. 1998; Sonnenberg et al. 1999; Zillikens and Giudice 1999; Hopkinson and Jones 2000; Nonaka et al. 2000; Leverkus et al. 2001).
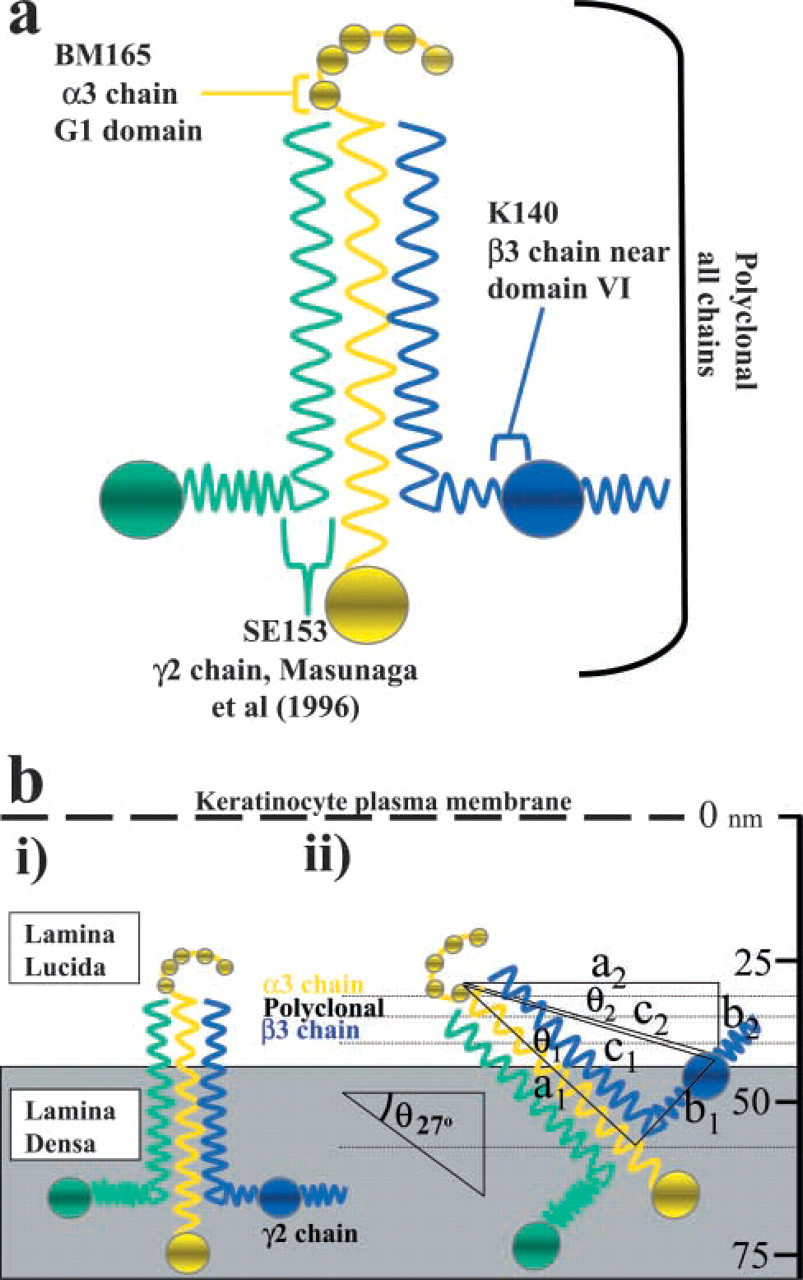
Schematic diagram showing the previously proposed and the new suggested laminin 5 orientation and antibody binding sites for the laminin 5 α3-, β3-, and γ2-chains. (
The average LL width in the human epidermal basement membrane was measured at 45–50 nm (Briggaman et al. 1971; Briggaman and Wheeler 1975; Masunaga et al. 1996; Bedane et al. 1997). Our data confirm that the LL width beneath K11M-embedded, cryofixed, and cryosubstituted human epidermis is approximately 40–45 nm. It is still possible to make a comparison of labeling, even if measurements were not made in previous studies by examining the positions of the labeling relative to the LL and LD, if clearly visible on the electron microscopic images (Rousselle et al. 1991a; McGrath et al. 1994). Previously, McGrath et al. (1994) used pre-embedding and cryosectioning immunolabeling methods followed by routine electron microscopic processing to localize laminin 5 (GB3) and kalinin antibodies to the lower LL border, findings that broadly support our data. However, the pre-embedding technique relies on good antibody tissue penetration, which is not always possible using antibodies attached to large colloidal gold particles to label LD antigens.
Various laminin forms are expressed in the epidermal basement membrane, including laminin 5 (α3β3γ2) and laminin 10 (α5β1γ1), and some less abundant forms may also be present, including laminin 1 (α1β1γ1), laminin 6 (α3β1γ1), and laminin 7 (α3β2γ1), previously identified in amnion, as well as laminin 11 (α5β2γ1) (Aumailley and Rousselle 1999; Pouliot et al. 2002). Our α3-chain data may include antibody measurements from laminins 5, 6, and possibly 7. The presence and relative expression levels of intact laminins 6/7 beneath normal epidermis have not yet been confirmed. However, from our data, particularly the narrow peak for the α3-chain (see Figure 2a), we suggest that either that laminin 6/7 is not abundantly expressed in normal skin or that it shares a limited α3 domain localization similar to laminin 5. The expression of laminin 6/7 was first identified in vitro and in amnion (Champliaud et al. 1996), and it has been hypothesized to act as an anchor or bridge between laminin 5 and the other BMZ molecules by virtue of its long multidomain β1 and γ1 arm domains (Chan et al. 1997). However, it is generally accepted that laminin 5 is one of the major and most important ligands for the α6β4 integrin (Niessen et al. 1994; Champliaud et al. 1996) and that it binds the NC-1 domain of collagen VII (Chen et al. 1999). The exact extent and position of the α6β4 in the LL space have yet to be measured, but estimates using other methods have suggested that the α6-subunit may extend as far as the mid-LL (McGrath et al. 1994).
The short arms of the β3- or γ2-chain of laminin 5 interact with the noncollagenous (NC-1) fibronectin-like domains of collagen VII (Chen et al. 1999). We have previously determined the localization of the large NC-1 domain of collagen VII (using the LH7:2 monoclonal antibody) to be approximately 58 nm from the plasma membrane within the upper LD (Shimizu et al. 1997; McMillan et al. 2003), which closely matches the β3-chain (40–80 nm, average 45 nm) and the previously studied γ2-chain (56 nm) (data from Masunaga et al. 1996, with permission). Given the large size and complex multidomain structure of the NC-1 domain, both β3- and γ2-chains remain likely candidates for laminin 5-collagen VII interactions. The laminin 5 γ2-chain (domain IV) has also recently been implicated in binding of nidogen and fibulin 2 (Utani et al. 1997; Sasaki et al. 2001), putative bridging molecules between the laminin and collagen networks. The position of the γ2-chain at the lowest position within the lamina densa would facilitate such interactions (Ghohestani et al. 2001).
In conclusion, the position of the laminin 5 α3 first globular domain within the lower LL and the relative position of the γ2 and β3 short arms in the LD suggest the need for a revised orientation of this molecule, which will help to shed new light on the organization laminin complexes and possible laminin interaction partners. Our findings, together with advances in antibody production and characterization, pave the way for detailed examination of other epidermal basement membrane antigens and their likely interactions.
Footnotes
Acknowledgements
Supported by a grant-in-aid of Scientific Research A (13357008, H.S.), a Health and Labor Sciences Research Grant (Research into specific diseases H13–02), by a grant from the Japanese Society for the Promotion of Science (JSPS, grant #00345, J.R.M.), by a grant-in-aid for JSPS fellows' research expenses (#00345) and by an award from the Japanese Society for Investigative Dermatology International Fellowship (Shiseido) (2000).
We gratefully acknowledge the technical support of Mr. H. Nakamura and Drs F. Iwao and H. Nakamura for supplying the normal control skin samples, and M. P. Marinkovich for the generous gift of antibodies and antiserum used in this study. We also thank Dr T. Masunaga for kindly providing the data from the γ2 chain studies.
