Abstract
Hyaluronan (HA) is expressed by most tissues, including skin. Localization of HA in the skin is assessed by histology with HA-binding protein (HABP) serving as the probe. Reports have suggested that HA expression in skin is altered in a number of diseases. However, interlaboratory variations in HABP staining profiles, even in normal skin, suggest a need to standardize methods and/or identify new probes. We report the staining patterns of a HA-binding peptide (termed “Pep-1”) in human and mouse skin. After acetone fixation, Pep-1 stained HA in the intercellular spaces of the epidermis, whereas staining in the dermis was weak and diffuse in both human and mouse skin. HABP staining of the epidermis and dermis were comparable in human skin but failed to stain the vital epidermis of mouse skin. In human skin, Pep-1 stained the basal, spinous, and granular layers, whereas HABP failed to stain the basal layer. Precipitation of HA in situ resulted in dermal staining but weak staining in the epidermis for HABP and Pep-1. Our results may suggest that Pep-1 is sensitive to HA conformation. Furthermore, Pep-1 may represent a new probe to study HA expression in the skin.
H
Cutaneous HA metabolism is normally a tightly regulated process. However, abnormally high concentrations of HA in the skin have been reported for a number of diseases including psoriasis (Wells et al. 1991; Tammi et al. 1994), scleroderma (Sondergaard et al. 1997; Passos et al. 2003), amyotrophic lateral sclerosis (Ono et al. 1996), lichen sclerosus et atrophicus (Kaya et al. 2000), and acute cutaneous graft vs host disease (GVHD) (Milinkovic et al. 2004). Similarly, pharmacologic agents such as glycolic acid (Bernstein et al. 2001), substance K (Kim et al. 2004), and estrogens (Gendimenico et al. 2002) have been shown to markedly increase HA expression in the skin. In contrast, diabetic patients with severely restricted joint mobility of the hands showed a significant loss of HA in the basement membrane zone of the skin (Bertheim et al. 2002), whereas topical application of steroids also reduced HA expression (Zhang et al. 2000). Thus, enhanced or reduced concentrations of HA may contribute to the dermatopathologies of various diseases as well as treatments. Lastly, HA may provide a useful marker to monitor disease progression.
Analyses of the concentration and polymer size of HA are typically determined by biochemical methods such as chromatography (Okamoto et al. 1999; Chaidedgumjorn et al. 2002; Karlsson and Bergman 2003), gel electrophoresis (Ikegami-Kawai and Takahashi 2002), and ELISA-like methods (Stern and Stern 1992). On the other hand, HA localization studies are based primarily on histology. Classic histochemical studies of HA are based on nonspecific cationic dyes such as Alcian blue. Ripellino et al. (1985) developed a more specific technique for staining HA in brain sections using the HA-specific probe, HA-binding protein (HABP). HABP is a complex of aggrecan and link protein and is derived from bovine cartilage (Tengblad 1979). Tammi and colleagues (1988) evaluated the distribution of HA in human skin using HABP as their probe. They found weak dermal staining but intense staining in the basal and spinous layers of the epidermis. Others have reported that HABP more strongly stained the dermis compared with the epidermis (Wells et al. 1991; Lin et al. 1997). Differences in the epidermal staining pattern have also been reported, with weak reactivity in the basal layer and more intense staining in the spinous and granular layers (Lin et al. 1997). Discrepancies in HABP staining profiles make comparisons among studies difficult to interpret and suggest the need to standardize current techniques and/or develop alternative probes for HA.
Recently, we have developed a HA-binding peptide (termed Pep-1) using the phage display technique (Mummert et al. 2000). Analyses of the Pep-1 amino acid sequence showed that it did not resemble known HA-binding domain sequences of CD44 or the B(X)7B motif, where B represents a basic amino acid residue and X any non-acidic residue (Entwistle et al. 1996; Mummert et al. 2000). Alanine scanning studies showed that the critical amino acids for HA binding were contiguous residues at positions 4, 5, and 6 (W, Q, F) and positions 9, 10, and 11 (L, T, V) (Mummert et al. 2000). Finally, in vitro studies showed that Pep-1 binds HA in immobilized, cell-associated, and soluble forms. Thus, we decided to investigate the potential utility of Pep-1 to serve as a probe for the qualitative and quantitative assessment of HA in the skin. Herein, we report the HA staining profiles of Pep-1 and HABP in human foreskin and mouse ear skin.
Materials and Methods
Skin Samples
Human skin samples were from discarded foreskins following circumcisions. Mouse ear skin was from C57BL/6 mice maintained in our breeding colonies. Animal experiments were approved by the Institutional Review Board at University of Texas Southwestern Medical Center.
Peptides and HABP
Pep-1 (GAHWQFNALTVR) and a scrambled peptide control (WRHGFALTAVNQ) were synthesized by Invitrogen (Carlsbad, CA) using standard fMOC chemistry as previously described (Mummert et al. 2000). Briefly, an amidated and biotinylated lysine residue was included at the C terminus of the linker sequence (GGGS) to facilitate detection of peptides. Stock solutions were prepared by dissolving peptides to 1 mg/ml in dH2O and were stored at −20C. HABP was purchased from Seikagaku Corporation (Tokyo, Japan) and biotin labeled with N-hydroxysuccinimido-biotin (Pierce Chemical Co; Rockford, IL) as described by the manufacturer.
Sample Preparation and Staining Procedure
Mouse ear skin and human foreskin were embedded in Optimal Cutting Temperature compound (OCT; Sakura Finetek, Torrance, CA), snap frozen in liquid N2, and sectioned at 8-μm thickness. Skin samples were fixed for 10 min in acetone and blocked for 10 min in 100 mM glycine followed by a 30-min incubation with PBS containing 1% BSA. Slides were washed with PBS and treated for 16 hr at 4C with Streptomyces hyaluronidase at 100 U/ml (Sigma-Aldrich; St Louis, MO) or mock treated with buffer alone (100 mM sodium acetate, pH 5.0). Biotinylated Pep-1 (5 μg/ml), scrambled peptide control (5 μg/ml), or HABP (1 μg/ml) in PBS containing 1% BSA were incubated on slides for 5 hr at 37C. In some experiments, biotin-conjugated peptides were used at 50 μg/ml to ascertain the impact of peptide concentration on the HA staining patterns. After washing in PBS containing 0.05% Tween 20, slides were developed by a 30-min incubation with fluorescein 5 isothiocyanate (FITC)-conjugated streptavidin (Jackson ImmunoResearch Laboratories; West Grove PA) diluted 1:200 in PBS containing 1% BSA. Skin sections were evaluated under an Olympus BX60 fluorescence microscope (Olympus; Melville, NY) equipped with a Sensys digital camera system (Photometrics; Tucson, AZ) and Metaview software (Universal Imaging Corp.; Downington, PA).
Glycosaminoglycan Inhibition Assay
To determine the specificity of Pep-1 for HA moieties in skin, we tested the inhibitory potential of HA oligomers and polymers as well as two HA-related glycosaminoglycans (chondroitin sulfate A and dermatan sulfate). Briefly, biotinylated Pep-1 (5 μg/ml) was incubated overnight in the presence of HA polymers, HA oligomers, chondroitin sulfate A, or dermatan sulfate in PBS containing 1% BSA at 37C. All of the inhibitors were used at a concentration of 1.5 mg/ml. Peptide solutions containing the various glycosaminoglycan inhibitors were then added to skin sections and developed as above.
Other Fixation Techniques
Mouse ear skin was embedded in OCT, snap frozen in liquid N2, and sectioned at 8-μm thickness. Skin samples were fixed for 10 min in PBS containing 3% paraformaldehyde and 0.5% cetylpyridinium chloride (CPC). After washing in PBS, sections were blocked for 10 min in 100 mM glycine followed by a 30-min incubation with PBS containing 1% BSA. Staining of paraformaldehyde/CPC-fixed samples was performed exactly as described for acetone fixation.
Permanent sections were prepared by embedding acetone-fixed ear skin in paraffin. Sections were cut to 8-μm thickness, deparaffinized, blocked with glycine and BSA as above, and stained with Pep-1 or the scrambled peptide after pretreatment with Streptomyces hyaluronidase or mock treated with buffer alone. Skin sections were stained with Pep-1 and the scrambled peptide control as above.
Quantitative Analysis
Integrated fluorescence intensities in 20–30 fields per skin section (n = 3) were obtained with Metaview software (Universal Imaging Corp.) from images captured at x200 magnification. Mean values were compared with a two-tailed Student's t-test and differences considered significant for p<0.05.
Results
Staining Profiles of HABP and Pep-1 in Human Foreskin
Significant variations in HABP staining profiles of human skin have been reported. Moreover, there are discrepancies between biochemical findings (i.e., high concentrations of dermal HA) (Sakai et al. 2000) and histologic observations (i.e., weak staining of the dermis by HABP) (Tammi et al. 1988). Thus, we assessed the ability of Pep-1 to serve as a probe for HA histology.
We found that HABP stained the dermis of human foreskin uniformly and with high intensity (Figure 1). In contrast, HABP predominantly stained HA in the spinous and granular layers of the epidermis, whereas staining in the basal layer was relatively weak or absent. No staining was detected in the stratum corneum. Quantitative analyses showed insignificant differences (p>0.05) between the dermal and epidermal (spinous and granular layers) integrated fluorescence intensities (Figure 2). Pretreatment of cryostat sections with Streptomyces hyaluronidase, an enzyme that specifically degrades HA, significantly (p<0.01) reduced HABP staining in both the epidermis and dermis, showing HA specificity (Figure 1 and Figure 2). Unlike HABP, Pep-1 showed diffuse and relatively weak staining of HA in the dermal compartment (Figure 1). With the exception of the stratum corneum, Pep-1 stained all of the epidermal layers, although the intensity of the staining appeared to be in the order basal layer > spinous layer > granular layer. However, integrated fluorescence intensities were not significantly different among the various epidermal layers as assessed by ANOVA (p>0.05) (data not shown). On the other hand, comparisons of integrated fluorescence intensities between the epidermis and dermis showed that Pep-1 stained the epidermis significantly better than the dermis (Figure 2; p<0.01). We next evaluated the specificity of Pep-1 staining by pretreatment of slides with Streptomyces hyaluronidase. Qualitative (Figure 1) and quantitative (Figure 2) analyses showed that digestion of HA from skin sections nearly completely abrogated Pep-1 staining, indicating HA specificity (Figure 1 and Figure 2).
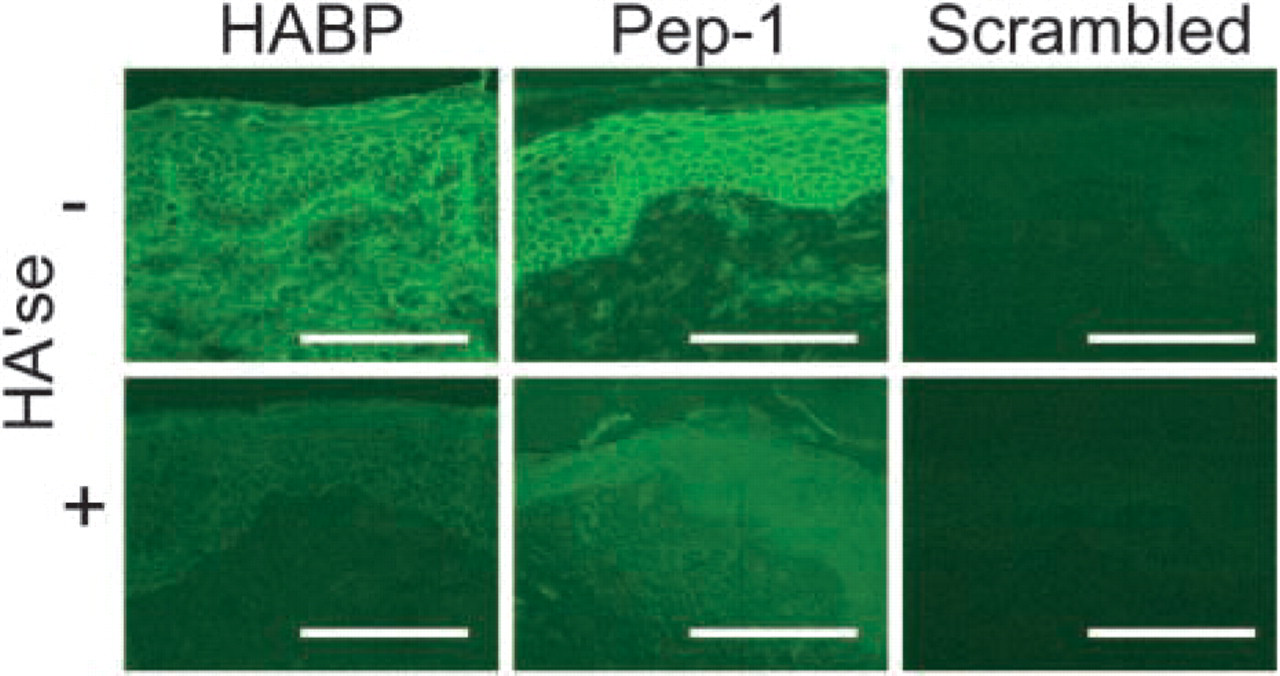
HA expression profiles in human foreskin. Cryostat sections were stained for HA with HABP, Pep-1, and the scrambled peptide control. Images represent x200 magnification. Some sections were pretreated with Streptomyces hyaluronidase (+HA'se) before staining to show HA specificity. Mock-treated (-HA'se) samples were treated in buffer alone (100 mM sodium acetate, pH 5.0). Note that Pep-1 detected HA associated with keratinocytes in the epidermis but only modestly stained HA in the dermal extracellular matrix. In contrast, HABP bound HA in both the epidermis and dermis. Data shown are the representative fields. Bars = 100 μm.
Staining Profiles of HABP and Pep-1 in Mouse Skin
Mice remain important models for many human diseases including selected dermatologic disorders (i.e., atopic dermatitis) (Yagi et al. 2002). Therefore, we compared the HA staining profiles of HABP and Pep-1 in mouse skin to determine if Pep-1 can be applied to experimental systems.
HABP stained the dermis of mouse ear skin uniformly and with high intensity (Figure 3 and Figure 4). On the other hand, HABP stained the epidermis mainly in the stratum corneum and basal lamina with relatively weak or no binding in the vital epidermis. Pretreatment of slides with Streptomyces hyaluronidase significantly reduced the intensity of staining in the dermis, whereas the stratum corneum and basal lamina was partially resistant to enzymatic digestion (Figure 3 and Figure 4). Finally, preincubation of HABP with HA polymers (1.5 mg/ml) significantly reduced HA staining, further showing the specificity of HABP for HA (data not shown).
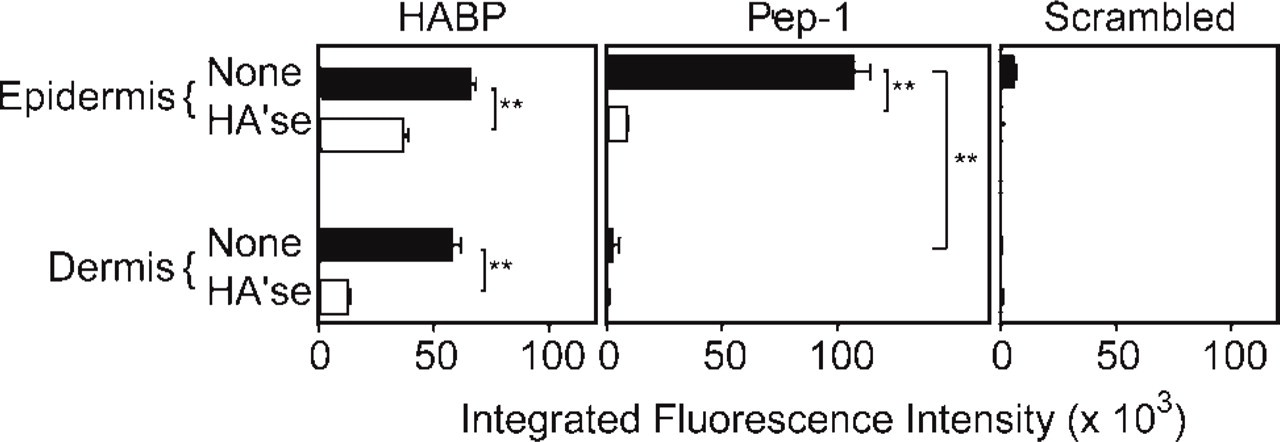
Integrated fluorescence intensities of HABP, Pep-1, and the scrambled peptide control in human foreskin. Fluorescence intensities were measured in the epidermis and dermis in 20–30 fields from three independent samples. Skin sections were mock treated (closed bars) or treated with Streptomyces hyaluronidase (HA'se, open bars) to assess HA specificity. Values are expressed as means ± SD, and statistically significant differences are indicated by asterisks (∗∗ p<0.01).
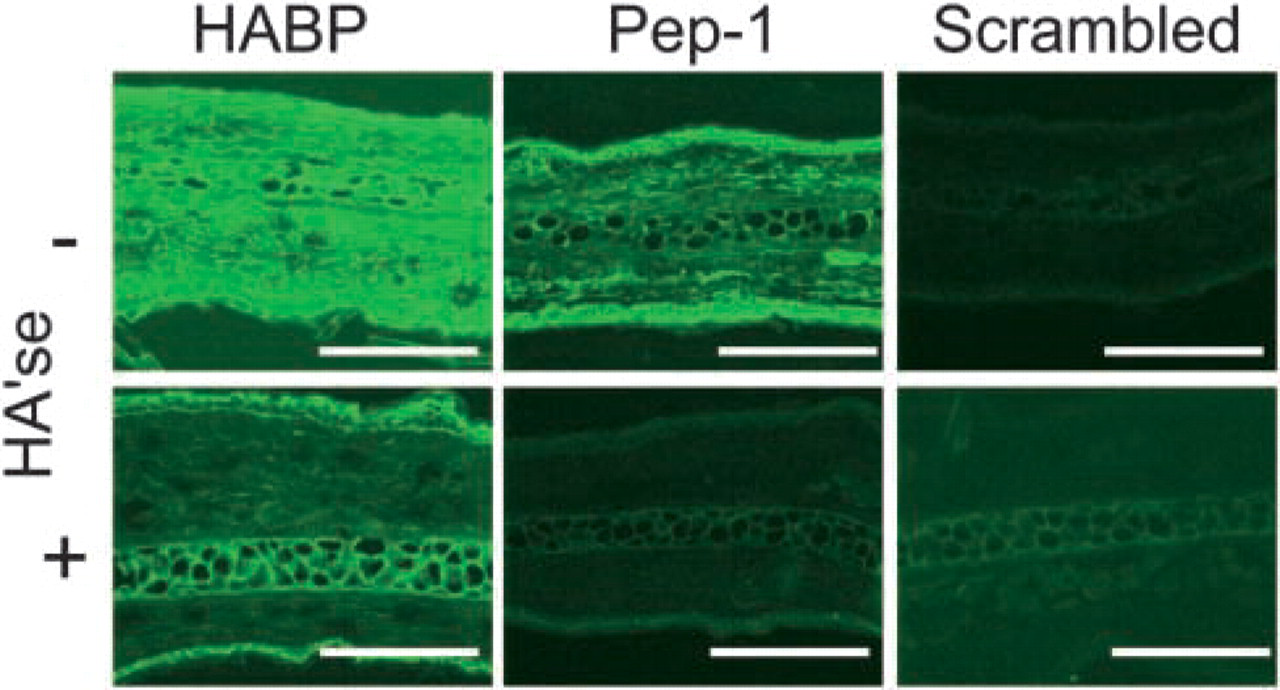
HA expression profiles in mouse ear skin. Cryostat sections were stained for HA with HABP, Pep-1, and the scrambled peptide control. Images represent x200 magnification. Some sections were pretreated with Streptomyces hyaluronidase (HA'se) before staining to show HA specificity. Note that Pep-1 detected HA associated with keratinocytes in the epidermis but diffusely stained HA in the dermal extracellular matrix. As with human foreskin, HABP stained both the epidermis and dermis. Data shown are the representative fields. Bars = 100 μm.
Pep-1 showed a staining pattern similar to human skin, with diffuse and relatively weak staining in the dermis and high-intensity staining in the epidermis (Figure 3 and Figure 4). Evaluation of the epidermal and dermal integrated fluorescence intensities showed that the Pep-1 staining was significantly weaker in the dermis compared with the epidermis (Figure 4; p<0.01). Again, the scrambled peptide control failed to bind cryostat sections, showing the requirement of the Pep-1 sequence for molecular interaction with HA. Finally, Streptomyces hyaluronidase treatment significantly reduced Pep-1 intensities showing HA specificity. We should note that Pep-1 staining patterns were not significantly altered when hyaluronidase digestions were performed in the presence of protease inhibitors (Ripellino et al. 1985) (data not shown). These results indicated that reduced Pep-1 staining after Streptomyces hyaluronidase digestion was not due to contaminating proteases in the hyaluronidase preparation.
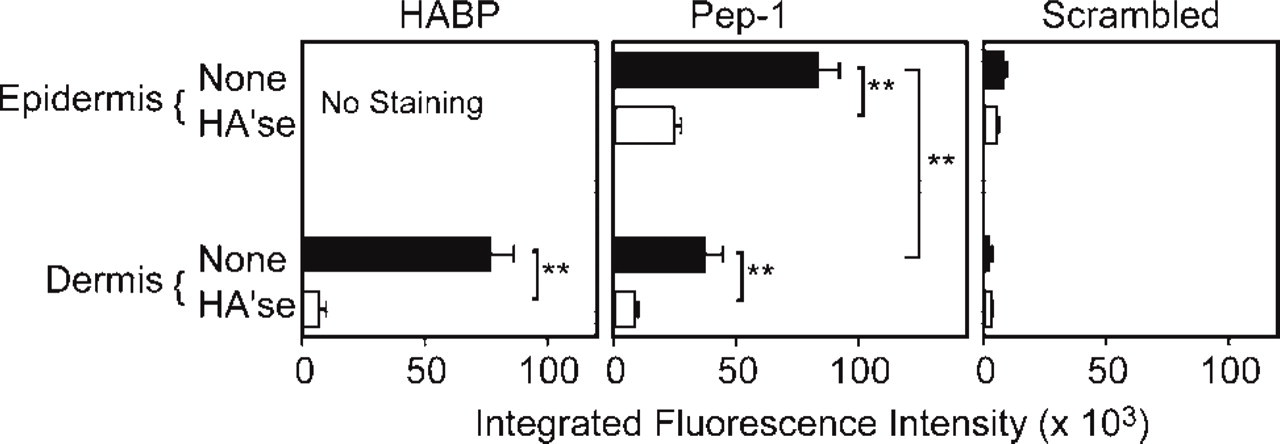
Integrated fluorescence intensities of HABP, Pep-1, and the scrambled peptide control in mouse ear skin. Fluorescence intensities were measured in the epidermis and dermis in 20–30 fields from three independent samples. Skin sections were mock treated (closed bars) or treated with Streptomyces hyaluronidase (HA'se, open bars) to determine HA specificity. Values are expressed as means ± SD, and statistically significant differences are indicated by asterisks (∗∗ p<0.01).
Effect of Pep-1 Concentration on HA Staining Patterns
Because the observed staining pattern of Pep-1 could have been the result of the peptide concentration, we assessed the impact of higher Pep-1 concentrations on the staining profiles in human and mouse skin. As shown in Figure 5, a 10-fold higher concentration (50 μg/ml) of Pep-1, but not the scrambled peptide control, markedly enhanced the fluorescence intensities in both human and mouse skin. However, higher concentrations of Pep-1 did not result in a HABP-like staining pattern. These results show that Pep-1 staining profiles are not concentration dependent.
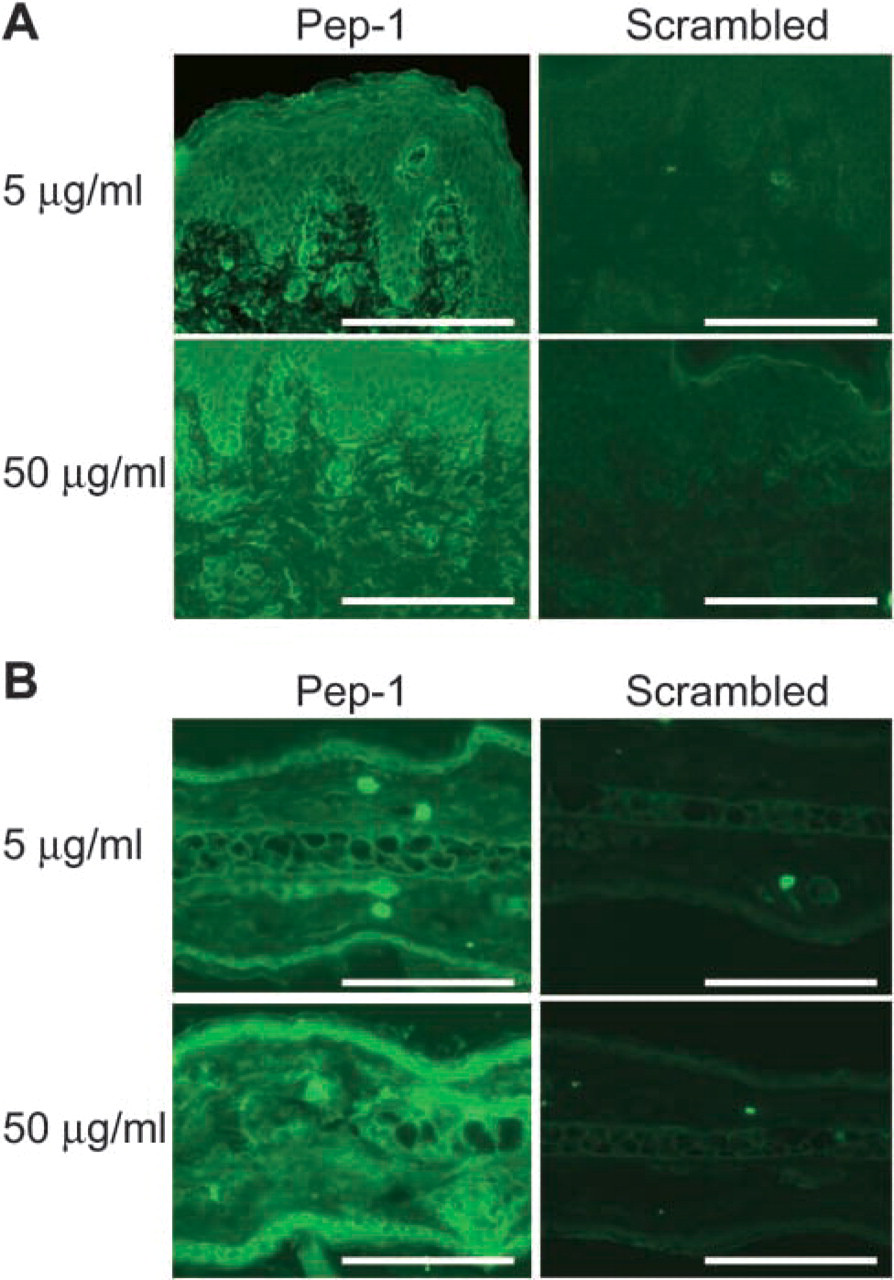
Impact of peptide concentrations on HA staining profiles. (
Specificity of Pep-1 for HA
To assess the specificity of Pep-1 for HA, we conducted inhibition assays using HA polymers and oligomers, chondroitin sulfate A, and dermatan sulfate. As shown in Figure 6, HA oligomers and polymers nearly completely abrogated binding of Pep-1 to cryostat sections. In contrast, two related glycosaminoglycans (chondroitin sulfate A and dermatan sulfate) failed to significantly inhibit Pep-1 binding to skin when used at the same inhibitor concentration. These results suggest that Pep-1 binds specifically to HA among the potentially available glycosaminoglycans.
Impact of Fixation on HA Staining Patterns
Because the method of fixation has been shown to alter HABP binding patterns in skin, we assessed the impact of paraformaldehyde/CPC fixation on Pep-1 binding to mouse ear skin. Paraformaldehyde/CPC fixation markedly altered the binding pattern of Pep-1. In contrast to acetone-fixed specimens, Pep-1 stained with high intensity throughout the dermis but showed weak to no binding in the epidermis. On the other hand, the scrambled peptide control showed little or no staining throughout the skin (Figure 7A). Interestingly, pretreatment of skin sections with Streptomyces hyaluronidase showed only partial abrogation of Pep-1 binding. Similarly, HABP binding to paraformaldehyde/CPC-fixed sections was only partially sensitive to pretreatment with Streptomyces hyaluronidase (Figure 7A). The failure of hyaluronidase pretreatment to completely remove Pep-1 and HABP staining may reflect the inability to completely remove CPC-precipitated HA from tissue sections (Lin et al. 1997). In sum, these results show that the method of fixation can dramatically impact the Pep-1 staining pattern.
Because paraffin sections retain details of tissue morphology better than frozen sections, we also ascertained the capacity of Pep-1 to stain deparaffinized skin. As shown in Figure 7B Pep-1, but not the scrambled peptide control, bound throughout the epidermis but diffusely in the dermis. However, the intensity of Pep-1 staining was less than for cryostat sections. Again, Pep-1 binding was reduced after pretreatment with Streptomyces hyaluronidase showing HA specificity.
Discussion
Localization of HA in skin and other tissues is conventionally determined using HABP as the standard probe. A number of studies using HABP have been reported for normal skin as well as skin from patients with local or systemic diseases. As discussed in the introduction, significant variations in HABP staining patterns even in normal skin have been reported.
In an effort to develop alternative HA probes for histology, we have compared the staining profiles of HABP and Pep-1 in human and mouse skin. Perhaps the most striking finding was that HABP uniformly stained the dermis with high intensity, whereas Pep-1 showed diffuse dermal staining with relatively weak intensity after acetone fixation. In contrast, fixation of tissues with paraformaldehyde/CPC resulted in intense dermal staining for both HABP and Pep-1.
How can we explain the discordant staining profiles observed in these studies? One possible reason for differences in dermal staining between HABP and Pep-1 after acetone fixation is that Pep-1 may have a markedly slower diffusion rate in the dermal compartment compared with HABP. However, given the relatively small size of Pep-1 compared with HABP, one would expect Pep-1 to more easily diffuse into the tissue for staining, not vice versa.
Recent biophysical studies have shown that HA adopts distinct conformations based on polymer length (Scott and Heatley 2002), degree of hydration (Cowman et al. 2005), and its association with membrane surfaces (Ionov et al. 2004). Although biophysical analyses have been based exclusively on in vitro systems, Milinkovic et al. (2004) have evaluated the adhesion of non-stimulated lymphocytes to skin biopsies from patients with acute GVHD. Although HA was expressed throughout GVHD skin (i.e., epidermis and dermis), the lymphocytes bound almost exclusively to HA expressed on endothelial cells of dermal vessels. These authors concluded that HA expressed by dermal endothelium was specialized to support lymphocyte adherence under sheer stress (Milinkovic et al. 2004). We suggest that differences between HABP and Pep-1 staining profiles may reflect differences in HA tertiary structures and/or HA lattice meshwork in different layers of the skin.
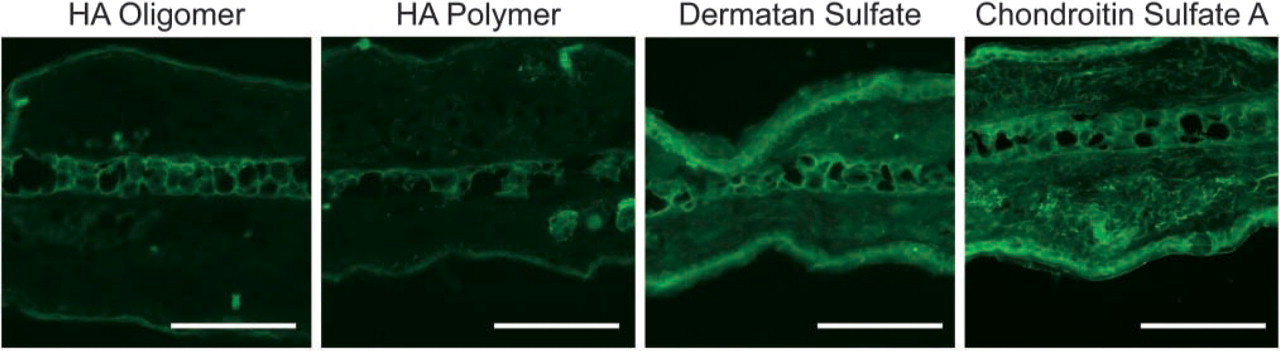
Pep-1 specificity for HA in situ. Biotin-labeled Pep-1 was pretreated with HA oligomers, HA polymers, dermatan sulfate, and chondroitin sulfate A. Solutions of Pep-1 containing glycosaminoglycan inhibitors were added to cryostat sections of mouse skin and inhibition assessed by staining intensities. Note that HA oligomers and polymers significantly inhibited Pep-1 binding to skin sections, whereas dermatan sulfate and chondroitin sulfate A had little or no effect. Images represent x400 magnification. Data shown are the representative fields. Bars = 100 μm.
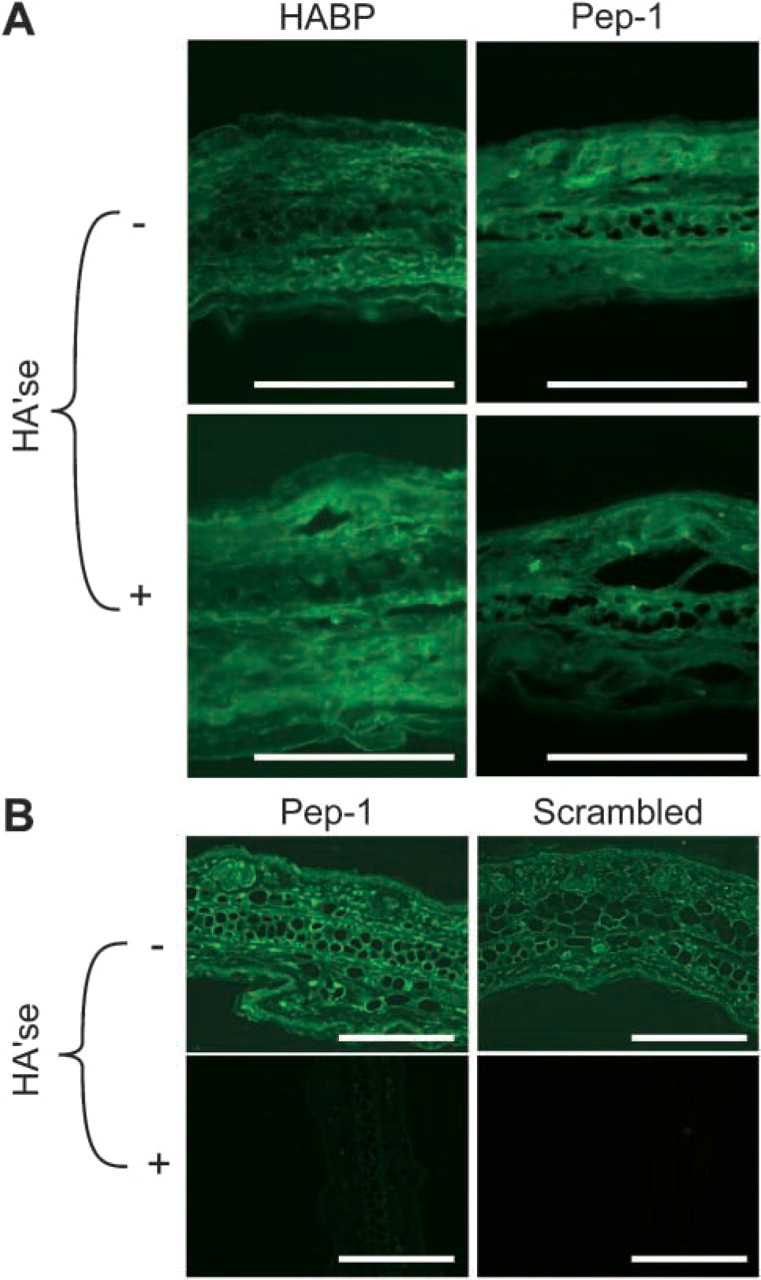
Impact of fixation on HA staining patterns in mouse skin. (
Can differences in HA conformations explain why Pep-1 stains the dermis after paraformaldehyde/CPC fixation? Recently, Cowman et al. (2005) evaluated the conformations of HA by tapping mode atomic force microscopy. When HA was deposited on the structured water layer of prehydrated mica, an extended conformation was favored. In contrast, HA deposited on mica lacking structured water favored a weakly helical, coiled conformation. Thus, the degree of hydration significantly impacts the conformation of HA polymers. Because precipitation likely alters the hydration of HA polymers in situ, we suggest that precipitated dermal HA (paraformaldehyde/CPC fixed) adopts a different conformation than non-precipitated dermal HA (acetone fixed). Interestingly, Streptomyces hyaluronidase abrogated dermal staining by HABP after acetone fixation but only partially reduced dermal HABP staining after paraformaldehyde/CPC fixation. Failure of Streptomyces hyaluronidase to completely remove HA staining for both HABP and Pep-1 may suggest that hyaluronidase cannot digest all HA conformations and/or lattice meshwork assemblies.
In terms of utility, Pep-1 may allow expression of epidermal HA to be studied in a number of pathologic states in fine detail after acetone fixation. Pep-1 may be of special value for studying epidermal HA expression in experimental conditions in mice, where the thin epidermis can be especially difficult to distinguish from the underlying dermis. Lastly, comparisons of Pep-1 and HABP staining profiles in conjunction with biophysical and biochemical analyses may shed new light on the relationship between HA structure and function in situ. Based on our experimental observations, we suggest that Pep-1 may represent a unique reagent to probe HA conformation.
Finally, we should emphasize that the synthetic nature of Pep-1 may represent an advantage over HABP, which is derived from a complex biological material (i.e., cartilage) (Tengblad 1979). For example, Armstrong and Bell (2002) showed that the fraction of link proteins in HABP preparations can significantly impact the size of the HA polymers detected in solution. As a synthetic peptide, it should be possible to synthesize Pep-1 in a more reproducible fashion, which may reduce experimental variation.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health Grant R01 AR-48840.
We would like to thank Patricia Adcock for her secretarial assistance and Dr. Akira Takashima for his thoughtful comments and suggestions.
