Abstract
Mucin O-glycosylation in cancer is characterized by aberrant expression of immature carbohydrate structures leading to exposure of simple mucin-type carbohydrate antigens and peptide epitopes. Glycosyltransferases controlling the initial steps of mucin O-glycosylation are responsible for the altered glycosylation observed in cancer. We studied the expression in gastric cell lines of six UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferases (GalNAc-T1, T2, T3, T4, T6, T11) that catalyze the initial key step in the regulation of mucin O-glycosylation, the transfer of GalNAc from UDP-GalNAc to serine and threonine residues. We also studied the expression of ST6GalNAc-I, the enzyme responsible for the synthesis of Sialyl-Tn antigen (NeuAcα2,6GalNAc) and the ST3Gal-I, the enzyme responsible for the synthesis of Sialyl-T antigen (NeuAcα2,3Galβ1,3GalNAc). This study was done using specific monoclonal antibodies, enzymatic assays, and RT-PCR. Our results showed that GalNAc-T1, -T2, and -T3 have an ubiquitous expression in all gastric cell lines, whereas GalNAc-T4, -T6, and -T11 show a restricted expression pattern. The immunoreactivity with MAb VU-2-G7 suggests that, apart from GalNAc-T4, another GalNAc transferase is involved in the glycosylation of the Thr in the PDTR region of the MUC1 tandem repeat. The expression of ST3Gal-I correlates with the expression of the Sialyl-T antigen in gastric cell lines and in the control cell lines studied. The expression of ST6GalNAc-I is low in gastric cell lines, in accordance with the low/absent expression of the Sialyl-Tn antigen.
Keywords
MUC1 is a cell membrane-associated mucin with a large, highly glycosylated extracellular domain consisting of multiple tandem repeats of 20 amino acids (Gendler and Spicer 1995). MUC1 is expressed in the apical surface of most normal glandular epithelial cells. In most carcinomas, MUC1 is overexpressed and displays an altered glycosylation, resulting in tumor-specific exposure of immature carbohydrate structures and peptide epitopes within the tandem repeat (Carraway et al. 1992; Taylor–Papadimitriou et al. 1999). These alterations make the MUC1 mucin a promising tumor antigen with diagnostic and therapeutic potential in the treatment of cancer (Apostolopoulos and McKenzie 1994; Finn et al. 1995; Miles and Taylor– Papadimitriou 1999).
One mechanism for alteration of mucin glycosylation, including MUC1, is the differential expression of the enzymes that initiate O-glycosylation: UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferases (GalNAc-transferases) (Clausen and Bennett 1996). These enzymes control the first level of complexity of mucin glycosylation, i.e., the sites and density of O-glycan occupancy of the mucin tandem repeat (Clausen and Bennett 1996). This initial key step is performed by a family of GalNAc-transferases that catalyze the transfer of GalNAc from UDP-GalNAc to serine and threonine residues (Clausen and Bennett 1996). The molecular processes governing the specificity and kinetics of the initiation step of O-glycosylation are not fully understood. It is becoming clear, however, that the substrate specificities of GalNAc-transferases are different and may determine the sites of O-glycan attachments. To date, 12 distinct members of the mammalian GalNAc-transferase family have been identified and characterized (Homa et al. 1993; White et al. 1995; Bennett et al. 1996,1998,1999a,b; Hagen et al. 1997; Ten Hagen et al. 1998,1999,2001; Guo et al. 2002; Schwientek et al. 2002). The finding that the GalNAc-transferases genes are differentially expressed in human tissues (Bennett et al. 1996; Hagen et al. 1997; Sutherlin et al. 1997; Mandel et al. 1999), and that the GalNAc-transferase family members have different kinetic properties and substrate specificities, suggest that these enzymes may have different functions and that the repertoire of GalNAc-transferases expressed in a given cell may determine the O-glycan attachment pattern (Wandall et al. 1997). Studies in vitro have shown that GalNAc-transferases display site specificity and different kinetic properties towards O-glycosylation sites in the MUC1 tandem repeat (Wandall et al. 1997). These studies showed that only GalNAc-T4 is able to complement other GalNAc-transferases in the O-glycosylation of MUC1 producing a fully glycosylated tandem repeat, including the Thr in the PDT∗R immunodominant region of the MUC1 repeat (Figure 1A) (Bennett et al. 1998; Hassan et al. 2000). This high density of glycosylation has been associated with MUC1 from breast cancer cells (Muller et al. 1997,1999; Muller and Hanisch 2002).
Recent studies have shown altered expression of GalNAc-transferases with malignant transformation. Variation of expression pattern has been described in oral squamous cell carcinoma, in which decreased expression of GalNAc-T1 and increased expression of GalNAc-T2 and -T3 have been reported compared to the expression pattern in normal oral mucosa (Mandel et al. 1999). Higher expression of GalNAc-T1, GalNAc-T2, and GalNAc-T3 has also been described in colorectal carcinoma compared with normal colon epithelium (Kohsaki et al. 2000; Shibao et al. 2002), suggesting that the differential expression of GalNAc-transferases underlies the mechanism responsible for altered carbohydrate antigen expression observed in epithelial cancers.
A second level of complexity in mucin O-glycosylation is the processing of the carbohydrate chains by other glycosyltransferases. Altered expression of glycosyltransferases responsible for elongation, branching, and termination of carbohydrate chains (Brockhausen et al. 1995,2001; Dalziel et al. 2001) leads to the formation of immature carbohydrate structures: GalNAc-T/S (Tn-antigen), NeuAcα2-6GalNAc-T/S (Sialyl-Tn-antigen), Galβ1-3GalNAc-T/S (T-antigen), and NeuAcα2-3Galβ1-3GalNAc-T/S (Sialyl-T-antigen) (Figure 1B). The aberrant expression of key sialyl-transferases ST6GalNAc-I and ST3Gal-I may determine the expression of the simple mucin-type carbohydrate antigens Sialyl-Tn and Sialyl-T (Brockhausen et al. 1995,2001; Dalziel et al. 2001).
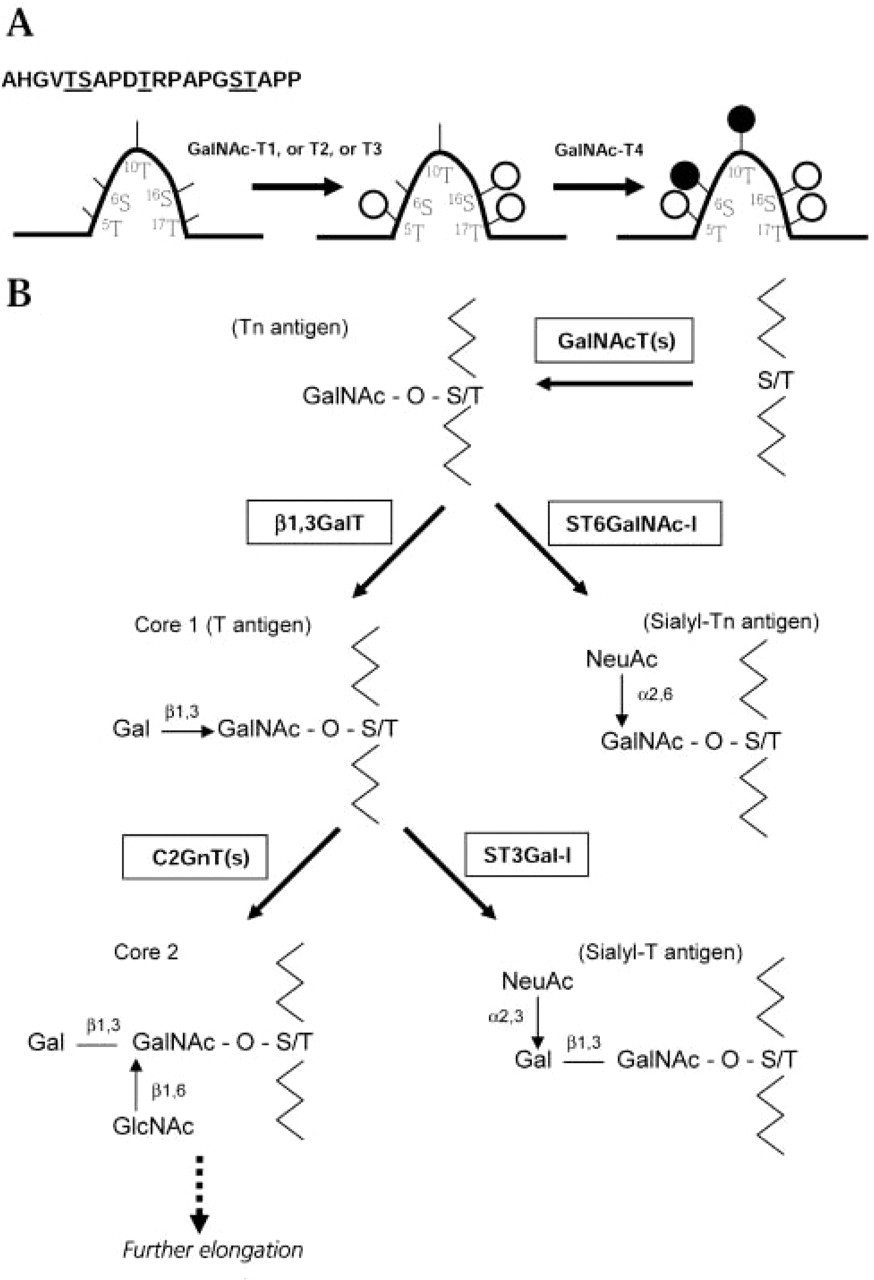
Initial steps of mucin glycosylation. (
In this study we have characterized the expression of six GalNAc-transferases (GalNAc-T1, -T2, -T3, -T4, -T6, -T11), and the expression of sialyltransferases ST6GalNAc-I, and the ST3Gal-I, in gastric carcinoma cell lines. We evaluated whether the expression of these glycosyltransferases in gastric carcinoma cell lines determines the expression of the simple mucin-type carbohydrate antigens Tn, Sialyl-Tn, T, and Sialyl-T. We have further evaluated whether the expression of different cancer-associated MUC1 glycoforms is determined by a specific pattern of expression of glycosyltransferases in gastric carcinoma cell lines.
Materials and Methods
Cell Lines and Cell Culture
Four gastric carcinoma cell lines (MKN45, KATOIII, GP202, and GP220) were studied. The colon carcinoma cell line HT-29 and the hematopoietic cell line K562 were used as controls for expression of enzymes, carbohydrate antigens, and MUC1. GP202 and GP220 cell lines were established in our laboratory (Gartner et al. 1996). MKN45, KATOIII, HT-29, and K562 are commercially available cell lines (American Type Culture Collection; Rockville, MD). The pathologic differentiation of the original tumors of the gastric cell lines is as follows: MKN45, poorly differentiated (Tamura et al. 1996); GP202, GP220, and KATOIII, signetring cell (Takeshima et al. 1991; Gartner et al. 1996). Cell lines were grown in RPMI 1640 with Glutamax, supplemented with 10% inactive fetal bovine serum, 10 U/ml penicillin, 10 μg/ml streptomycin, 10 U/ml nystatin, and 50 μg/ ml gentamicin. Cultures were maintained at 37C in a humidified 5% CO2 atmosphere.
Monoclonal Antibodies and Immunofluorescence
The monoclonal antibodies (MAbs) specific for glycosyl-transferases, carbohydrate antigens, and the MUC1 mucin are listed in Table 1.
Cell lines were fixed in acetone for 5 min before immunofluorescence. Samples designed for neuraminidase treatment were incubated with neuraminidase from Clostridium perfringens type IV (Sigma Chemical; St Louis, MO) diluted in 0.1 M sodium acetate buffer (pH 5.5) to a final concentration of 0.1 U/ml. The incubation was carried out for 2 hr at 37C and was followed by three washes in cold water.
Single immunofluorescence was performed as follows. Samples were rinsed twice in Tris-buffered saline (TBS, pH 7.6) and incubated for 20 min with rabbit non-immune serum diluted 1:5 in TBS containing 5% of BSA. Sections were rinsed in TBS and incubated overnight at 4C with MAbs (Table 1) diluted in TBS containing 5% BSA. Sections were washed three times for 5 min in TBS and incubated with FITC-conjugated rabbit anti-mouse immunoglobulin diluted 1:70 in TBS (code F-261; Dako, Glostrup, Denmark). Sections were washed three times for 5 min in TBS and mounted in glycerol containing p-phenylene-diamine. Double-labeling immunofluorescence was performed on both neuraminidase-treated and non-treated sections. The Sialyl-T-antigen was evaluated based on the anti-T MAb reactivity after neuraminidase treatment. Samples were rinsed twice in TBS for 5 min and incubated with mouse MAb 4B10 (IgG1; Table 1) for 2 hr at room temperature. Sections were washed twice for 5 min in TBS and bound MAbs were detected with FITC-conjugated donkey anti-mouse IgG (Jackson Immunoresearch; West Grove, PA) diluted 1:50 in TBS. Sections were washed twice for 5 min in TBS and blocked with non-immune goat serum diluted 1:10 in TBS. Sections were incubated with HH8 or HB-T (both IgM; Table 1) overnight at 4C. Sections were washed three times for 5 min with TBS and incubated with Texas red-conjugated goat anti-mouse IgM (Jackson Immunoresearch) diluted 1:100 in TBS. Sections were washed three times for 5 min in TBS and mounted in glycerol containing p-phenylene-diamine. The samples were examined in fluorescence microscopes (Olympus BH-2 and Leica DMIRE2).
Monoclonal antibodies used for immunocytochemistry
A semiquantitative approach was used to score the immunofluorescence labeling. Percentage of positive cells was scored as <25%, 25-50%, 50-75%, and >75%, based on the analysis of approximately 1 × 104 cells. Intensity of immunofluorescence labeling was graded as -, negative; +, weakly positive; + +, moderately positive; + + +, strongly positive. Results are based on three independent assays analyzed by two independent observers.
RNA and Multiplex RT-PCR
RNA was extracted from the different cell lines using the Tripure isolation reagent kit (Boehringer Mannheim; Indianapolis, IN) according to the manufacturer's protocol. RNA yield and quality were determined spectrophotometrically and 1.0–1.7 μg of total RNA of each cell line was reverse-transcribed using the Superscript II RNase H–Reverse Transcriptase kit (Life Technologies; Carlsbad, CA) according to the manufacturer's instructions. To control the yield of cDNA synthesis, as well as any genomic DNA contamination, a PCR with primers designed in different exons for amplification of the hypoxanthine phosphoribosyltransferase (HRPT) mRNA was performed.
Human ST6GalNAc-I was amplified using the forward primer, 5'-GCA ACC ACA GCC AAG ACG CTC ATT CCC AA-3’ and the reverse primer, 5'-TGT CAC GAC CTT CTG CAC CAA GGA GTA GT-3’ (amplicon length 455 bp). These primers were designed to amplify both the short and long forms of the human ST6GalNAc-I, as previously described (Ikehara et al. 1999). HRPT was amplified using the forward primer, 5'-TTC CTC CTC CTG AGC AGT CAG C-3’ and the reverse primer, 5'-GCG ATG TCA ATA GGA CTC CAG AT-3’ (amplicon length 756 bp).
For the specific HRPT fragment amplification, after preheating for 4 min at 94C, 30 cycles were performed, each cycle comprising 30 sec at 92C, 1 min at 65C, and 1 min at 72C, followed by a final extension of 5 min at 72C. PCR reaction mixture consisted of 2.0 μl sample cDNA, 2.5 μl PCR buffer (500 mM KCl, 15 mM MgCl2, 100 mM Tris-HCl, pH 9.0), 3.0 μl of 10 mM dNTPs (Promega; Madison, WI), 0.2 μl Taq DNA polymerase (Amersham Biosciences; Piscataway, NJ), 4.0 μl of each 3 pM HRPT-specific primers and H2O at 25 μl.
The multiplex PCR of ST6GalNAc-I and HRPT was done as follows. After preheating for 4 min at 94C, 35 cycles were performed with each cycle of 30 sec at 92C, 45 sec at 64C, and 2 min at 72C, followed by a final extension of 10 min at 72C. PCR reaction mixture contained 4.0 μl sample cDNA, 5.0 μl PCR buffer (500 mM KCl, 15 mM MgCl2, 100 mM Tris-HCl, pH 9.0), 3.0 μl of 10 mM dNTPs (Promega), 0.4 μl Taq DNA polymerase (Amersham Biosciences), 2.0 μl of each 3 pM HRPT-specific primers, 4.0 μl of each 3 pM ST6GalNAc-I-specific primers, and H2O to a total volume of 25 μl. In all experiments, negative control reactions were performed by replacing cDNA template with sterile water.
Reaction products obtained were submitted to electrophoresis in 2% agarose gels containing ethidium bromide. Fluorescence measurements were made using Gel Doc 1000 (BioRad; Hercules, CA). For semiquantitative analysis of the multiplex RT-PCR data, fluorescence of each sample was compared with that of HRPT, co-amplified within the same tube, by calculating the ratio (fluorescence units of ST6GalNAc-I band/fluorescence units of HRPT band)X 100. mRNA expression was determined in two independent experiments.
Gel bands corresponding to the hST6GalNAc-I fragments were extracted using QIAquick Gel Extraction Kit (QIAGEN; Hilden, Germany). Identity and sequence of the amplicons were confirmed by automated sequencing (ABI Prism 377; Applied Biosystems, Foster City, CA) in the forward and reverse sense using the ST6GalNAc-I-specific primers.
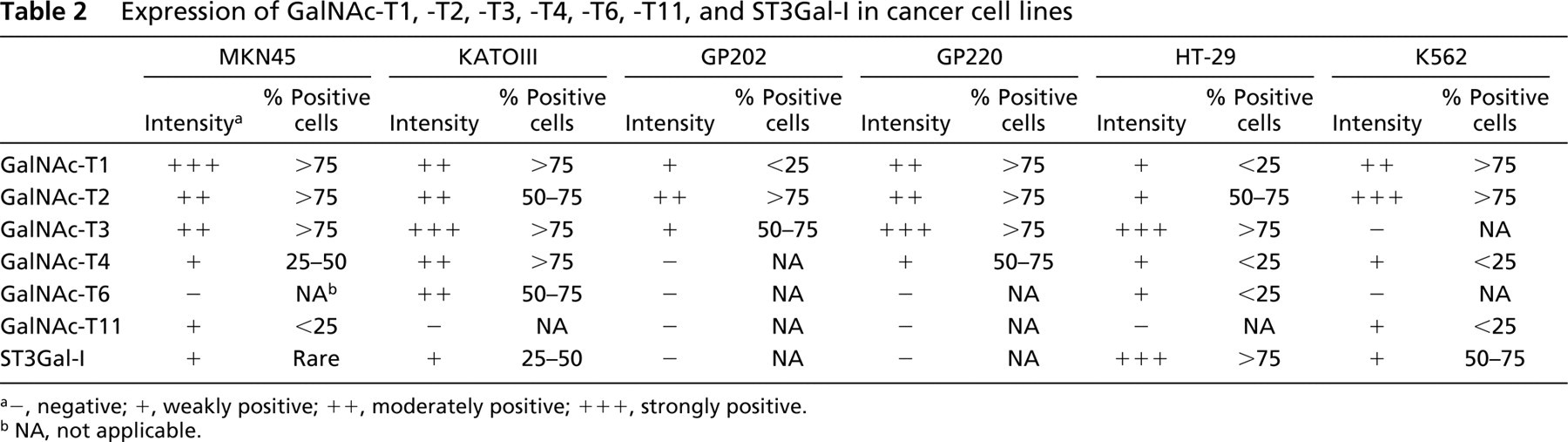
Expression of GalNAc-T1, -T2, -T3, -T4, -T6, -T11, and ST3Gal-I in cancer cell lines
a-, negative; +, weakly positive; ++, moderately positive; +++, strongly positive.
b NA, not applicable.
GalNAc-tranferase Assay
Cell pellets were washed in PBS and resuspended in 50 mM Mops buffer, pH 7.5, containing 1% Triton X-100, to obtain 2 × 106 cells/100 μl buffer.
GalNAc-T4 transferase activity was assayed using the MUC1 glycopeptide GalNAc4TAP25 (T∗APPAHGVT∗SAP-DTRPAPGS∗T∗APPA; asterisk indicates site with GalNAc) as substrate. Glycopeptide GalNAc4TAP25 was synthesized chemoenzymatically and characterized as previously described (Hassan et al. 2000). Enzymatic assays were performed in reaction mixtures of 50 μl containing 25 mM cacodylate (pH 7.4), 10 mM MnCl2, 0.25% Triton X-100, 200 μM UDP-[14C]-GalNAc (1000 cpm/nmol), 250 μM of acceptor substrate, and 5 μl of the cell lysates. Products were quantified by scintillation counting after chromatography on a Dowex column as previously described (Bennett et al. 1998).
Results
Expression of GalNAc-transferases GalNAc-T1, -T2, -T3, -T4, -T6, and -T11 in Gastric Cell Lines
The results obtained in the immunofluorescence study of GalNAc-transferases are summarized in Table 2. The expression of GalNAc-transferases in the gastric cell lines was variable. MKN45 showed a high level of expression of GalNAc-T1, moderate levels of expression of GalNAc-T2, -T3, and low levels of expression of GalNAc-T4 and -T11. GalNAc-T6 was the only absent enzyme in this cell line. KATOIII showed high expression of GalNAc-T3 (Figure 2A) and moderate expression of GalNAc-T1, -T2, -T4, and -T6. KATOIII was the gastric cell line showing the highest level of expression of GalNAc-T4, and the expression was observed in more than 75% of the cells (Figure 2B). GP202 showed moderate expression of GalNAc-T2, low expression of GalNAc-T1 and -T3 (Figure 2H), and no expression of GalNAc-T4 (Figure 2I), -T6, and -T11. GP220 showed high expression of GalNAc-T3, moderate expression of GalNAc-T1 and -T2, and low levels of expression of GalNAc-T4.
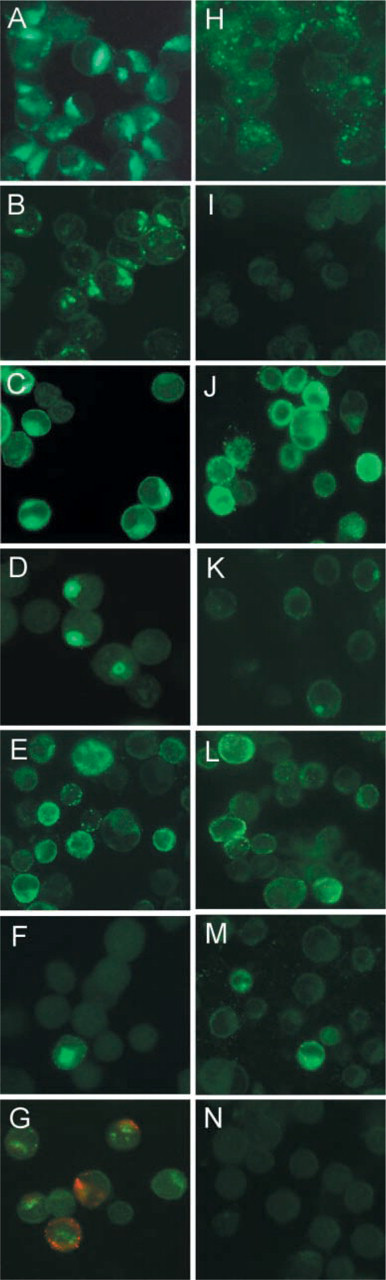
Immunofluorescence study of the expression of glycosyl-transferases, MUC1, and simple mucin-type carbohydrate antigens. Gastric cell lines KATOIII (
The control cell line, HT-29, showed high expression of GalNAc-T3 and weak expression of GalNAc-T1, -T2, -T4, and -T6. The control hematopoietic cell line K562 showed high expression of GalNAc-T2, moderate expression of GalNAc-T1, and low expression of GalNAc-T4 and -T11.
The fluorescent signal observed for every GalNAc-transferase was perinuclear in all the cell lines, characteristic of Golgi localization.
Enzymatic Activity of GalNAc-T4 in Gastric Cancer Cell Lines
The enzymatic activity of GalNAc-T4 was measured in an assay using the MUC1 glycopeptide GalNAc4 TAP25. This acceptor substrate allows discrimination between GalNAc-T4 activity and other known GalNAc-transferase activities (Bennett et al. 1998; Hassan et al. 2000; Schwientek et al. 2002). As shown in Figure 3, the gastric cell lines showed different levels of enzymatic activity. KATOIII showed the higher level of activity, with MKN45 and GP220 also showing significant levels of GalNAc-T4 activity. GP202 showed no GalNAc-T4 enzymatic activity. The control cell lines HT-29 and K562 showed very low enzymatic activity.
Expression of GalNAc-α2,6-sialyltransferase ST6GalNAc-I in Gastric Cell Lines
The expression of ST6GalNAc-I was evaluated by multiplex RT-PCR due to lack of an antibody for immunodetection of the enzyme.
Low expression of ST6GalNAc-I mRNA was detected in every gastric cell line, with ST6GalNAc-I/HRPT ratio values ranging between 11.3% and 22.2% when compared to the control cell line K562 (Figure 4). The identity of the amplified sequence (band of 455 bp) was confirmed by sequence analysis (data not shown) to be the long-form previously described (Ikehara et al. 1999). The spliced-variant shorter form, described by Ikehara et al. and expected to be of 220 bp, was not detected in any of the cell lines tested.
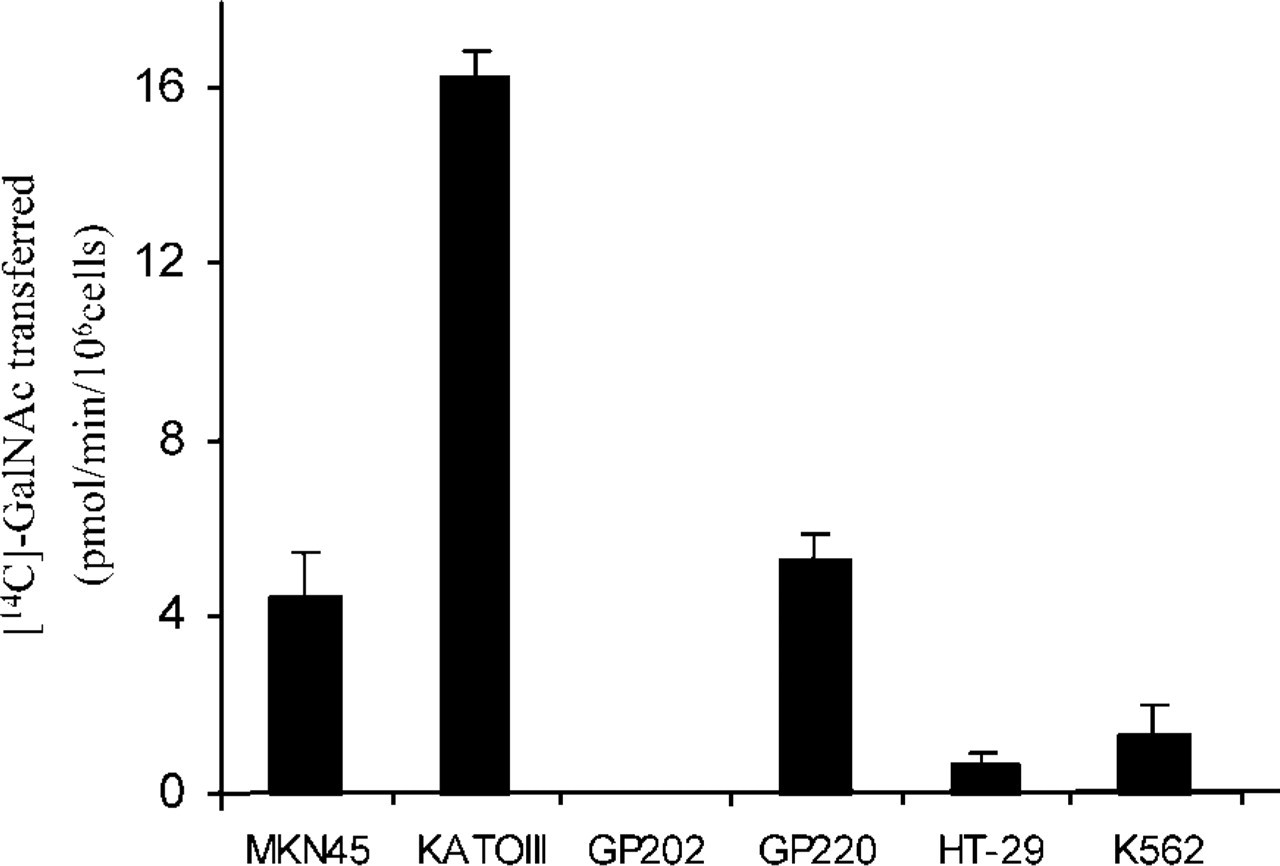
Enzymatic activity of GalNAc-T4. Enzymatic assays were performed using a specific substrate for GalNAc-T4, MUC1 glycopeptide GalNAc4TAP25. Results are expressed in pmol of GalNac tranferred per minute per 106 cells.
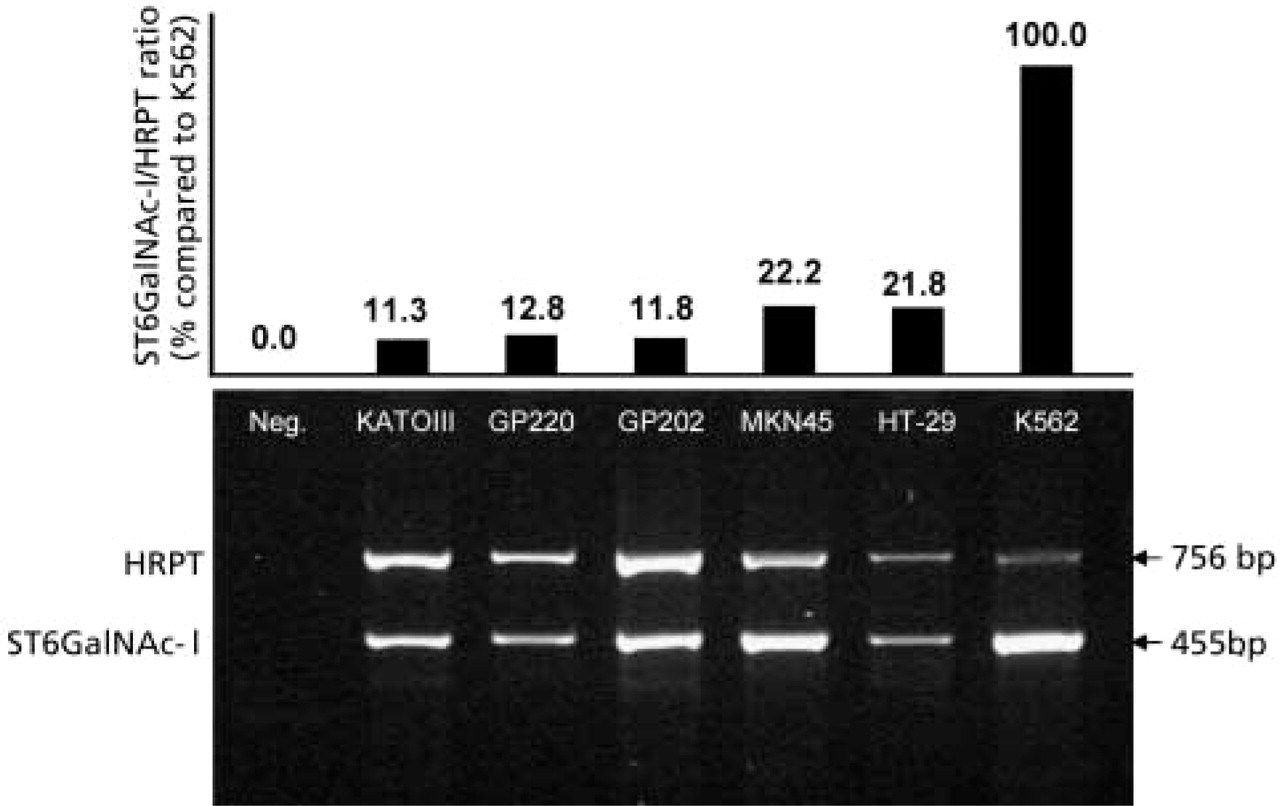
ST6GalNAc-I expression evaluated by multiplex PCR. (
Expression of Gal-α2,3-sialyltransferase ST3Gal-I in Gastric Cell Lines
ST3Gal-I was weakly expressed by KATOIII in 25–50% of the cells (Figure 2G, green) and in MKN45 in rare cells. The fluorescence signal was perinuclear in all positive cells. The cell lines GP202 (Figure 2N) and GP220 showed no expression of ST3Gal-I. The control cell line HT-29 showed a strong perinuclear signal in more than 75% of the cells, and K562 showed a weak staining in 50–75% of the cells (Table 2). Double labeling was performed to check for co-expression of ST3Gal-I and Sialyl-T-antigen (Figure 2G).
Expression of Carbohydrate Antigens in Gastric Cell Lines
The expression of carbohydrate antigens is summarized in Table 3. Antigen Tn was expressed in all gastric cell lines at either moderate or high levels. Membrane staining was observed in all the cell lines, although diffuse cytoplasmic or a perinuclear staining was also observed in MKN45, KATOIII (Figure 2E), and GP202 (Figure 2L). Low levels of expression of Sialyl-Tn-antigen were detected in rare KATOIII cells (Figure 2F) and in <25% of GP202 cells (Figure 2M). Control cell line HT-29 showed moderate perinuclear/cytoplasmic staining of Sialyl-Tn in <25% of the cells, while K562 showed a strong membrane/cytoplasmic staining in 50–75% of the cells. Expression of both T-and Sialyl-T-antigen was observed in KATOIII in <25% of the cells (Figure 2G, red). Sialyl-T was also observed in rare MKN45 cells. HT-29 showed rare cells expressing T-antigen and high expression of Sialyl-T. K562 also showed high expression of Sialyl-T-antigen in >75% of the cells.
Expression of mucin-type carbohydrate antigens in cancer cell lines
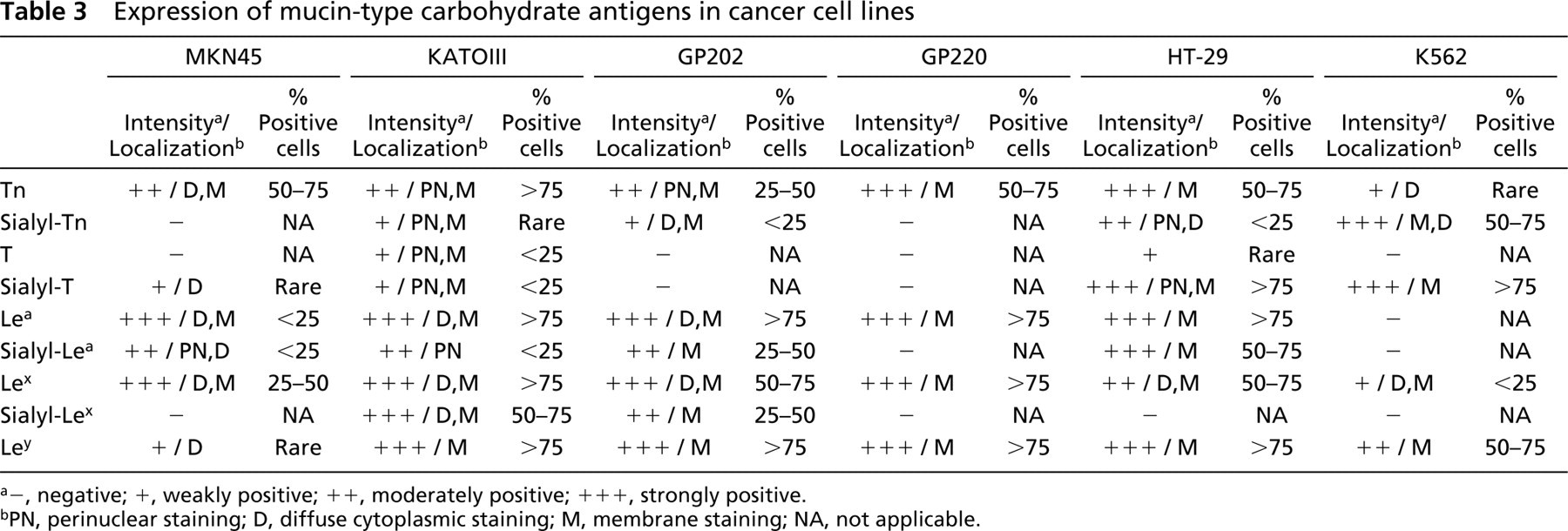
a-, negative; +, weakly positive; ++, moderately positive; + + +, strongly positive.
bPN, perinuclear staining; D, diffuse cytoplasmic staining; M, membrane staining; NA, not applicable.
All the gastric cell lines showed expression of type 1 and type 2 chain Lewis antigens (Table 3). Lea and Lex antigens were expressed in all the gastric cell lines. Ley was expressed in all the gastric cell lines in >75% of the cells with the exception of MKN45, which showed only rare positive cells. Sialyl-Lea was expressed in all gastric cell lines with the exception of GP220, whereas expression of Sialyl-Lex in gastric cell lines was observed only in KATOIII and GP202 (Table 3).
Expression of MUC1 in Gastric Cancer Cell Lines
Expression of MUC1 is summarized in Table 4. All gastric cell lines expressed MUC1 as detected by HMFG1 (Figures 2C and 2J). Under-glycosylated MUC1 detected by MAb SM3 was observed at similar levels of expression in the gastric cell lines. Immunoreactivity with MAb VU-2-G7 was detected in KATOIII in 25–50% of the cells, with a strong perinuclear staining pattern (Figure 2D). VU-2-G7 also reacted with <25% of GP202 (Figure 2K) and GP 220 cells. Immunoreactivity with MAb VU-4-H5 was observed in 25–50% of GP202 and GP220 cells, as well as in <25% of KATOIII cells, with a diffuse cytoplasmic and membrane staining pattern. Immunoreactivity with MAb 5E5 was observed in all gastric cell lines. The control cell lines HT-29 and K562 showed low levels of MUC1 expression.
Discussion
In this study we have characterized the expression of six members of the polypeptide GalNAc-transferase family in gastric carcinoma cell lines. GalNAc-T1, -T2, and -T3 were expressed in every gastric cell line. The expression of GalNAc-T4 was very heterogeneous among the cell lines, with KATOIII showing the highest level of expression as shown by ICC and enzymatic assay. GalNAc-T6 was expressed only in KATOIII, whereas GalNAc-T11 was observed only in MKN45. These results show that the repertoire and the levels of expression of GalNAc-transferases are different in the gastric cell lines. These observations are in accordance with the variable pattern of expression of GalNAc-transferases previously described in pancreatic, mammary, and colon cell lines and carcinomas (Sutherlin et al. 1997; Mandel et al. 1999; Kohsaki et al. 2000). Interestingly, GalNAc-T11 was solely expressed in the poorly differentiated gastric cell line MKN45 (Tamura et al. 1996). Correlation with differentiation status was not observed for other GalNAc-transferases, including GalNAc-T3 as previously described (Sutherlin et al. 1997). These differences may stem from tissue-specific variation because we observed similar levels of GalNAc-T3 expression in every histomorphological type of gastric carcinoma (unpublished data).
Studies in vitro have shown that GalNAc-transferases display site specificity and different kinetic properties towards O-glycosylation sites in mucins, such as the MUC1 tandem repeat (Wandall et al. 1997; Hassan et al. 2000). These characteristics of GalNAc-transferases suggest that the members of this family of enzymes have different functions and that the repertoire of GalNAc-transferases expressed in a given cell may determine the pattern of O-glycan attachment, especially the density of glycosylation of the MUC1 tandem repeat (Wandall et al. 1997). In fact, recent studies have shown that thus far only GalNAc-T4 is able to complement other GalNAc-transferases in the O-glycosylation of MUC1, producing a tandem repeat with the highest density of O-glycan attachment (Bennett et al. 1998). This density of glycosylation, characterized by five sites glycosylated out of five putative sites, is characteristic of the cancer-associated form of MUC1 from breast carcinoma (Muller et al. 1997,1999; Muller and Hanisch 2002). In the present study we determined whether the expression of GalNAc-transferases was associated with different glycosylation forms of the MUC1 tandem repeat in gastric carcinoma cell lines. This study was performed using MAbs that recognize specific MUC1 glycoforms. Our results showed that the four gastric cell lines studied expressed MUC1, as detected by antibody HMFG1. However, the expression of the different glycoforms of the MUC1 tandem repeat was heterogeneous, as summarized in Table 4. MAb VU-4-H5 was used to recognize the PDTR region when Thr is not glycosylated (Schol et al. 1998), and MAb VU-2-G7 was used to recognize the PDTR region when Thr is glycosylated with GalNAc (Ryuko et al. 2000). These antibodies were used to characterize the glycosylation status of the immunodominant PDTR region of the MUC1 tandem repeat (Mensdorff-Pouilly et al. 2000). Our results showed that VU-2-G7 reacts strongly with KATOIII, which is the cell line with higher levels of expression of GalNAc-T4 as detected by ICC and by enzymatic activity. This result suggests that the GalNAc glycosylated PDTR region of the MUC1 tandem repeat is present in this cell line due to the action of GalNAc-T4, which is the only GalNAc-transferase thus far identified capable of glycosylating the Thr in the PDTR (Bennett et al. 1998). Despite this observation, GP202, which is devoided of GalNAc-T4 as determined by ICC (Figure 2I), exhibits few cells with low reactivity for VU-2-G7 (Figure 2K). This observation was confirmed by enzymatic assay for GalNAc-T4 (Figure 3). The presence of VU-2-G7 staining and low levels of VU-4-H5 indicates that there is some glycosylation of the PDTR region independent of GalNAc-T4 expression, suggesting that there is another GalNAc-transferase capable of such activity towards the MUC1 tandem repeat. Identification of all the members of the GalNAc-transferase family, as well as the detailed characterization of their in vivo sites of O-glycosylation, should clarify this issue.
Expression of MUC1 in cancer cell lines
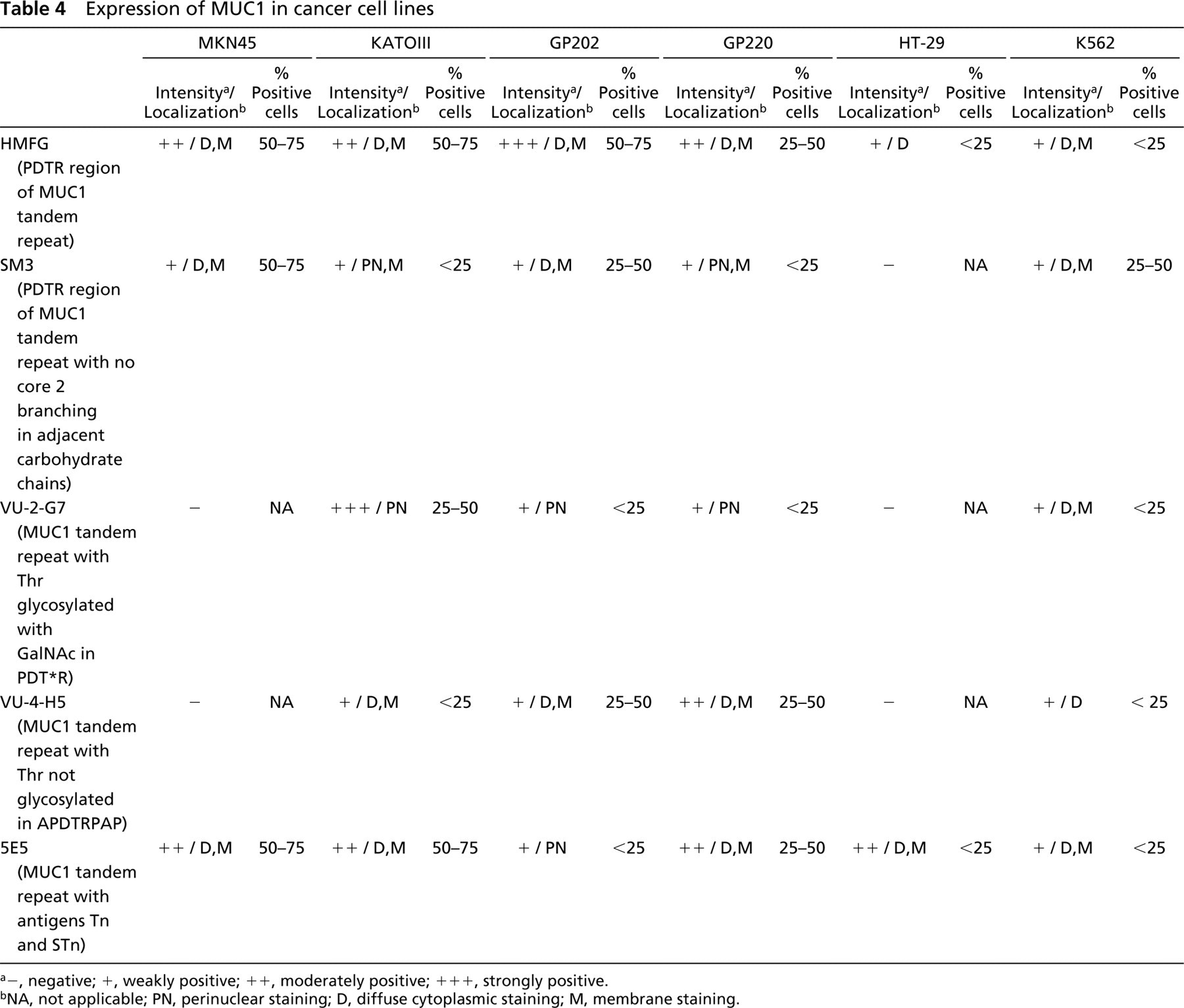
a-, negative; +, weakly positive; ++, moderately positive; + + +, strongly positive.
bNA, not applicable; PN, perinuclear staining; D, diffuse cytoplasmic staining; M, membrane staining.
The perinuclear staining pattern observed with the antibody VU-2-G7 suggests that the GalNAc glycosylated PDTR region is only available in immature MUC1 molecules that are undergoing glycosylation in the Golgi. Further glycosylation leads to either complex carbohydrate chains or simple sialylated GalNAc structures (Sialyl-Tn). For the KATOIII cell line, Sialyl-Tn-antigen was observed in rare cells and therefore does not explain the lack of reactivity of VU-2-G7 in 50% of KATOIII cells. On the contrary, complex carbohydrate antigens are highly expressed in KATOIII, as assessed with MAbs to Lewis antigens (see Table 3) and may contribute to masking of the VU-2-G7 epitope in some of the KATOIII cells.
The expression of Sialyl-T-antigen was observed in KATOIII as well as in the control cell lines HT-29 and K562. We observed a complete overlap between the expression of Sialyl-T-antigen and the expression of ST3Gal-I, supporting the role of ST3Gal-I in the formation of Sialyl-T (NeuAcα2,3Galβ1,3GalNAc) in gastric, colon HT-29, and hematopoietic K562 cell lines. Our observations are in agreement with recent studies with breast cancer cell lines showing that the sialyltransferase ST3Gal-I is the key enzyme in the formation of the Sialyl-T-antigen (NeuAcα2,3Galβ1,3GalNAc) (Dalziel et al. 2001).
In the present study we observed low levels of expression of mRNA of ST6GalNAc-I in MKN45, KATOIII, GP202, GP220 and high levels in control cell line K562, which is the unique cell line expressing high levels of Sialyl-Tn (Figure 4). Among the sialyltransferases identified thus far, ST6GalNAc-I is considered to be the enzyme responsible for the synthesis of the Sialyl-Tn structure (Ikehara et al. 1999). Our results show that the low expression of ST6GalNAc-I in gastric cell lines correlates with very low/absent detection of the Sialyl-Tn-antigen. The incomplete overlap between ST6GalNAc-I mRNA detection and the product of the activity of this sialyltransferase, the Sialyl-Tn-antigen, may be explained by competition for the GalNAc substrate between ST6GalNAc-I and other glycosyltransferases, such as Core1 Gal-transferases and Core2 GlcNAc-transferases (Brockhausen et al. 2001). The Golgi subcompartment in which ST6GalNAc-I resides has not been defined (Ikehara et al. 1999), and this should be examined in future studies to determine whether these glycosyltransferases can physiologically compete for the substrate in gastric cancer cells.
In a previous study, Ikehara et al. (1999) have identified two splice variants of the ST6GalNAc-I cDNA clones: one clone of 2.46 kb, encoding an active enzyme of 600 amino acids, and a 2.23-kb cDNA splice-variant of the longer form, encoding an inactive enzyme. The authors propose an alternative splicing model for regulation of expression of ST6GalNAc-I and consequent expression of Sialyl-Tn antigen. In the present study the primers used were designed to amplify both the short and the long form described by Ikehara et al. (1999). Our results showed the absence of the short form and presence of the long form of the ST6GalNAc-I even in the cell lines depleted of Sialyl-Tn-antigen. The observation that the cell lines studied in the present work did not show the alternative splicing model of ST6GalNAc-I suggests that this is not a major mechanism of regulation of Sialyl-Tn-antigen expression.
The Sialyl-Tn-antigen is frequently observed in gastric carcinoma (David et al. 1992; Yonezawa et al. 1992; Bresalier et al. 1996) and in intestinal metaplasia of the human stomach (David et al. 1992), a precursor lesion of gastric carcinoma (Correa and Shiao 1994). The characterization of the expression of ST6GalNAc-I in gastric tissues and gastric carcinoma cell lines should contribute to clarification of the role of this enzyme in the aberrant expression of Sialyl-Tn-antigen in gastric carcinogenesis.
In summary, we have shown that (a) GalNAc-T1, -T2, and -T3 have a ubiquitous expression in all gastric cell lines, although with variable expression levels, whereas GalNAc-T4, -T6, and -T11 show a restricted expression pattern. (b) The immunoreactivity with MAb VU-2-G7 suggests that, apart from GalNAc-T4, another GalNAc-transferase is involved in the glycosylation of the Thr in the PDTR region of the MUC1 tandem repeat. (c) The expression of ST3Gal-I is associated with the expression of the Sialyl-T-antigen in gastric cell lines, and in the control cell lines studied. (d) The expression of ST6GalNAc-I is low in gastric cell lines, in accordance with the low/absent expression of the Sialyl-Tn-antigen. Our results contribute to understanding of the molecular mechanisms that underlie aberrant expression of simple mucin-type carbohydrate antigens and of particular MUC1 glycoforms in gastric carcinoma cells.
Footnotes
Acknowledgements
Supported by Fundação para a Ciěncia e a Tecnologia, Programa Operacional Ciěncia, Tecnologia, Inovação do Quadro Comunitário de Apoio III, Project POCTI 36376/99, and the Danish Cancer Society.
We thank Dr Joy Burchell and Prof Joyce Taylor– Papadimitriou for providing antibodies HMFG1 and SM3.
