Abstract
We investigated the intracellular distribution of the mRNAs corresponding to the three non-allelic CaM genes in cultured hippocampal cells by in situ hybridization with digoxigenin-labeled gene-specific riboprobes. In neurons the perikaryon was heavily stained and strong dendritic mRNA targeting was detected for all three CaM genes. The color labeling exhibited a punctate distribution, suggesting that CaM mRNAs are transported in RNA granules. Immunocytochemistry for S100 demonstrated that glial cells express CaM mRNAs at a very low level. A minority of the cultured cells were negative for either labeling.
Keywords
C
In rodent brain, the CaM mRNAs are heavily distributed in the neuronal cell somata (Palfi et al. 1999). Berry and Brown (1996) demonstrated that the CaM I mRNAs are transiently targeted into the dendritic compartment in cortical and hippocampal pyramidal and cerebellar Purkinje cells in the newborn brain. Previously, we demonstrated by quantitative, radioactive in situ hybridization that mRNAs transcribed from all three CaM genes are significantly more abundant in dendrite-rich than in axon-rich regions of rat brain (Palfi et al. 1999; Kortvely et al. 2002). However, the mRNA concentrations were consistently different. In all the dendrite-rich areas, the highest and lowest levels were those of the CaM I and CaM II genes, respectively. These results suggest that a significant amount of CaM mRNAs, transcribed not only from the CaM I but also from the CaM II and CaM III genes, is translocated into dendrites in a gene-specific manner. The number of known dendritically targeted mRNA species in mammalian neurons is growing (Wells and Fallon 2000), and the evidence indicates that the mRNAs in dendrites are translated there. The cells can thereby alter the dendritic protein concentrations locally, in response to various cues.
In the present study, we carried out CaM gene-specific, non-radioactive in situ hybridization (ISH) cytochemistry on rat primary hippocampal cultures. This system was selected for the following reasons: (a) unlike many immortalized cell lines, it may represent the nearest approximation to in vivo CNS neurons; (b) it mimics several in vivo characteristics of these cells (e.g., dendrites and axons are present, synapse formation takes place); (c) its monolayer structure allows resolution of the subcellular compartments; and (d) notably, in vivo hippocampal pyramidal cells have directly been demonstrated to target CaM I mRNAs into dendrites (e.g., Blichenberg et al. 1999).
On embryonic day 18, rat hippocampi were trypsinized, cells were dissociated by trituration, and plated onto poly-
A majority of the cultured cells exhibited neuronal morphology (Brewer et al. 1993; Brewer 1997) and prominent CaM gene expression (Figures 1A–1C, 1E, and 1F), whereas the remaining population was not stained at all. No differences were found among the three CaM genes as regards the ratio of labeled/nonlabeled cells. In neurons, intense labeling was detected in the entire cytoplasm but the nucleus remained unstained (Figures 1A–1C, 1E, and 1F). Even in their distal portions, dendrites were also labeled for the mRNAs corresponding to all three CaM genes. The labeling intensity of the dendrites, compared to that of the somata, was similar for the three antisense probes. At higher magnification, the hybridization signal displayed a punctate intracellular distribution (Fig. 1A, 1C, and 1E). Hybridization with sense probes revealed very faint nonspecific reactivity comparable to that of the background (Figure 1D).
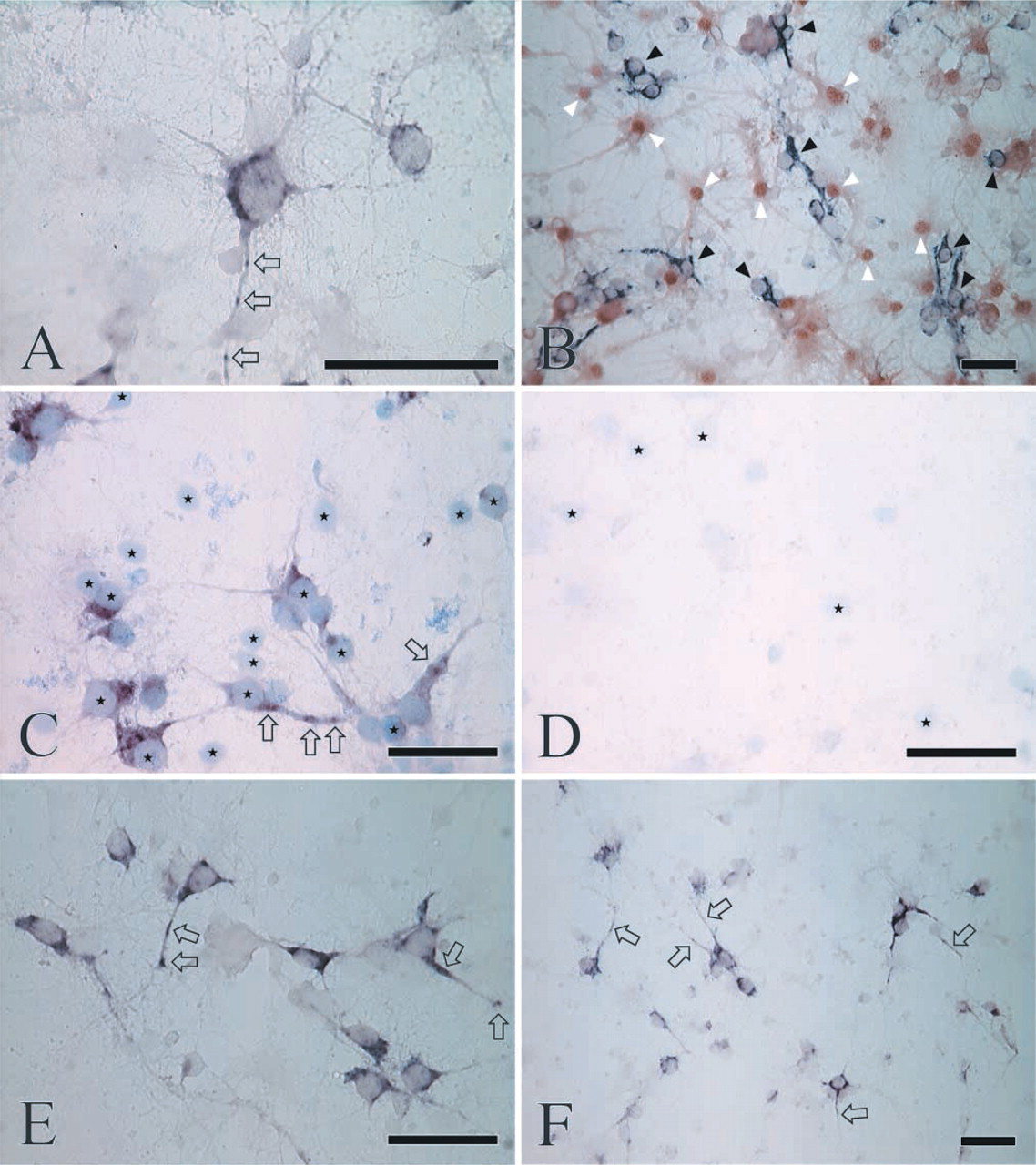
Distribution of CaM mRNAs in primary hippocampal cells, as revealed by color ISH cytochemistry using CaM I (
To identify the cells with nondetectable CaM expression, glia-specific S100 immunocytochemistry was carried out after the ISH protocol. Because the S100 protein is mainly confined to the nucleus, whereas the CaM mRNAs are localized in the cytoplasm, the parallel use of the two detection systems would feasibly allow discrimination among CaM-positive/S100-negative or CaM-negative/S100-positive cells. The vast majority of the CaM-negative cells proved to be S100-positive, whereas the neurons expressing the CaM genes did not express the S100 marker (Figure 1B). Moreover, a relatively small number of cells were negative for both the CaM mRNAs and the S100 protein. It should be noted, however, that “negative” can simply mean a very low but still appropriate expression level for the cell. Nevertheless, the identity of the “double-negative” cells remains unknown.
Here, by utilizing nonradioactive ISH cytochemistry, we demonstrate directly that mRNAs transcribed from each CaM gene are subjected to dendritic trans-location in the hippocampal neurons in primary culture. In addition, a punctate localization of the CaM mRNAs was revealed, which is consistent with previous findings demonstrating that mRNAs translocate in granular structures. Direct in vivo evidence of the dendritic CaM mRNA pool has been obtained only for the CaM I mRNAs, and exclusively in pyramidal and Purkinje neurons of the developing rat brain (Berry and Brown 1995,1996). However, our indirect data acquired from quantitative ISH experiments (Palfi et al. 1999,2002; Kortvely et al. 2002) hinted that the mRNAs of the other two CaM genes are also directed towards the dendritic compartment. Our previous data also indicated that CaM mRNAs might translocate to dendrites uniformly and to a greater extent in the early postnatal period (Kortvely et al. 2002), whereas their targeting may become more restricted and differential in the adult (Palfi et al. 1999). The present study strongly supports the idea of a widespread dendritic trafficking of all CaM mRNAs, thus more closely resembling the initial in vivo postnatal period than adulthood. Because primary neurons may preserve many immature properties, their CaM expression could reflect the stages of early neuronal development, when the mRNAs corresponding to the three CaM genes are relatively similar in their ability to translocate dendritically. Targeting of the CaM mRNAs into the dendritic compartment may be involved in local dendritic/ postsynaptic activity-dependent CaM protein synthesis. Our recent findings are not in full agreement with previous in vitro data obtained on PC12 cells, where only the CaM I and II mRNAs were present in both the somata and the processes, whereas the CaM III signals were restricted to the cell bodies (Zhang et al. 1993). However, PC12 cells are derived from peripheral neurons that are different in many aspects from those in the CNS, explaining this discrepancy reasonably.
This study demonstrates that, at least in primary hippocampal neurons, (a) the capacity for dendritic translocation is not restricted to CaM I transcripts but is a more general characteristic of the CaM transcripts, (b) CaM mRNA transport most probably occurs in granular units, and (c) S100-positive (glial) cells do not express CaM in a detectable amount. Further studies are needed to clarify the significance of these findings under in vivo conditions.
Footnotes
Acknowledgements
Supported by grants from the National Scientific Research Fund, Hungary (OTKA T034621) and the Ministry of Health, Hungary (57/2000) to KG.
The skillful technical assistance of Ms Zsuzsa Ambrus is highly appreciated.
