Abstract
Endothelial nitric oxide synthase (eNOS), originally found in the endothelium of vascular tissue, also exists in other cell types, including ciliated epithelia of airways. The eNOS is ultrastructurally localized to the basal body of the microtubules of the cilia, and nitric oxide (NO) stimulates ciliary beat frequency (CBF). We examined whether the expression of eNOS is present in ciliated cells of other organs. Western blotting analysis revealed that eNOS was expressed in the rat cerebrum, lung, trachea, testis, and oviduct. Immunohistochemical staining showed that eNOS was localized in the ciliated epithelia of airways, oviduct, testis, and ependymal cells of brain in addition to the endothelium and smooth muscle of the vasculature. To confirm the activation of eNOS in the ciliated epithelia, we examined the effect of
N
In mammals, ciliated epithelial cells line the respiratory tract, part of the reproductive tract, and ventricles of the central nervous system. These cilia contribute to the movement and clearance of mucus (Houtmeyers et al. 1999), and transport of ova (Talbot et al. 1999) and cerebrospinal fluid (CSF) (Scott et al. 1974) via ciliary beating and electrolyte transport mechanisms. In cilia, the motion is achieved by coupling the hydrolysis of ATP by dynein to microtubule sliding (Murad 1994). Both cAMP and cGMP increase dynein-like ATPase activity (Walczak and Nelson 1994). Wyatt et al. (1998) demonstrated that both cAMP-dependent protein kinase (PKA) and cGMP-dependent protein kinase (PKG) are involved in the regulation of CBF in bovine bronchial epithelial cells.
Recently, eNOS, soluble guanylyl cyclase (sGC), and PKG I-β have been identified in the ciliated epithelia of airways (Zhan et al. 1999a,b). Moreover, both the NO donor NOC-12 and GSNO increase CBF (Li et al. 2000), and
Materials and Methods
Reagents and Antibodies
Minimal essential medium Eagle (MEME) was from Mediatech (Herndon, VA).
Mouse monoclonal antibodies (MAbs) against eNOS and iNOS, and a rabbit polyclonal antibody against brain NOS (bNOS), were purchased from Transduction Laboratories (Lexington, KY). Rabbit polyclonal antibody against the α1-and β1-subunits of sGC was obtained from Cayman Chemical (Ann Arbor, MI). Rabbit polyclonal anti-PKG I-α and PKG I-β antibodies were purchased from StressGen (Victoria, BC, Canada). Goat anti-mouse or anti-rabbit IgG conjugated with horseradish peroxidase (HRP) and nitrocellulose membrane were from Bio-Rad (Hercules, CA). Biotinylated anti-mouse antibody, avidin-biotin-HRP complex, and diaminobenzidine (DAB) kit were from Vector Labs (Burlingame, CA). Normal mouse IgG was obtained from DAKO (Carpinteria, CA). ECL blotting detection reagents were obtained from Amersham.
Tissue Preparation
All rats were obtained from Hilltop Laboratory Animals (Scottdale, PA) and were treated in accordance with NIH guidelines. Seven female adult (six for IHC and one for Western blotting analysis) and 16 male adult (six for IHC, one for Western blotting analysis, and nine for NOx measurement) Sprague-Dawley rats weighing 260–300 g were used. Animals were sacrificed with an overdose of isoflurane (Ohmeda PPD; Liberty Corner, NJ). Then the lung, trachea, cerebrum, testis, and oviducts were rapidly removed.
For Western blotting analysis, the tissues were homogenized in ice-cold 50 mM Tris-HCl (pH 7.4) containing 0.1 mM EDTA, 0.1 mM EGTA, 0.1 μM phenylmethylsulfonyl fluoride (PMSF), 2 μM leupeptin, 1 μM pepstatin, and 0.1% 2-mercaptoethanol. The homogenates were centrifuged at 15,000 X g for 30 min at 4C and the pellet was discarded. The protein of the supernatant was used.
For IHC, the specimens were immersed in 4% paraformaldehyde in 0.1 M PBS, pH 7.4, at room temperature (RT). After 3 hr of fixation, the specimens were dehydrated in ethanol for paraffin embedding. Serial sections 5-μm thick were cut and mounted for immunostaining.
For preparation of ciliated epithelia, rat tracheal mucosa explants were cut into small pieces (4–5 mm3) and rinsed several times with PBS.
Western Blotting Analysis
Protein samples were loaded (50 μg each) and separated on 7.5% SDS-PAGE, followed by blotting of the proteins to nitrocellulose membrane. The blot was blocked with a buffer consisting of 10 mM Tris-HCl (pH 7.4), 0.15 M NaCl, 2% nonfat milk, 2% bovine serum albumin, and 0.1% Tween-20, for 1 hr at RT. The blots were then incubated with primary antibody (1:1000 dilution) for 1 hr at RT. Then the blots were incubated with a secondary goat anti-mouse or anti-rabbit IgG conjugated with HRP and detected with ECL. After detection, the bands were quantified with a densitometer and Imagequant software (Molecular Dynamics; Sunnyvale, CA).
Immunohistochemistry
After deparaffinization, the slides were treated with 3% H2O2 in PBS for 20 min to quench endogenous peroxidase activity. After blocking of nonspecific sites with 10% goat serum in PBS for 1 hr, tissue sections were incubated with mouse anti-eNOS MAb (diluted 1:500 for sections from oviduct, airway, and testis and 1:1000 for sections from cerebrum) at 4C overnight. Unbound primary antibody was washed off with PBS and the sections were incubated with biotinylated anti-mouse antibody (1:200 dilution) for 1 hr. Specific binding was detected using an avidin-biotin-HRP complex (1:100 dilution) for 1 hr and a substrate solution of H2O2 and DAB. Negative controls were carried out with normal mouse IgG.
Measurement of NOx (NO− 2/NO− 3) in the Media
The measurement of NO and its oxidation products (NOx) was carried out using a chemiluminescence analyzer (NOA 280; Sievers Instruments, Boulder, CO), reduction of nitrite and nitrate using vanadium (III) and hydrochloric acid at 90C. Ten μl of samples was injected into the glass purge vessel containing 5 ml of vanadium chloride (0.1 M in 1 N HCl) and 100 μl of diluted (1:30) antifoaming agent. NOx were sparged from the vessel and carried in nitrogen gas to the analyzer. The output from the analyzer was integrated to determine total peak area. The nitrite/nitrate concentrations were determined by comparison with the calibration curve (1–25 μM) of NaNO2 in distilled water.
Statistical Analysis
The results are expressed as means ± SE. One-factor ANOVA was used to compare the difference among groups. The value of
Results
Western Blotting Analysis
eNOS antibody reacted with a protein of 135-kD mass in all tissues of rat brain, lung, trachea, testis, and oviduct (Figure 1). The antibody did not crossreact with bNOS and iNOS proteins because there were no bands at 160 kD or 130 kD. bNOS antibody reacted only in cerebrum and testis; iNOS reacted weakly in cerebrum, lung, and trachea. The results indicated that only eNOS was localized in all these cilia-containing tissues. The NO-cGMP signal pathway proteins sGC α1, sGC β1, PKG I-α, and PKG I-β were also detected in all these cilia-containing tissues (Figure 2). sGC α1 was dominant in cerebrum, testis, and oviduct; sGC β1 was dominant in lung and trachea.
Immunoreactivity of eNOS in Airway, Cerebrum, Oviduct, and Testis
On the basis of our previous work showing the physical association of eNOS, but not iNOS or bNOS, with the basal body of the microtubules of lung epithelial cilia (Xue et al. 1996a,b) and because only eNOS was detectable in all the cilia-containing tissues, as examined by Western analysis, we focused on eNOS for IHC. In conduit vessels, the endothelial cells were strongly positive (Figure 3A, blue arrows) for eNOS staining. Negative controls showed no background staining (Figure 3B).
eNOS antibody gave a strong and specific signal in ciliated epithelia of trachea (Figure 3C, blue arrows), bronchus (Figure 3D, blue arrows), and bronchiole (Figure 3E, blue arrows), with prominent staining of eNOS in Clara cells (Figure 3E, red arrows) and in type II cells (Figure 3E, green arrows) of alveoli. Nonciliated epithelia in trachea (Figure 3C, red arrows) and bronchus (Figure 3D, red arrows) were not stained with eNOS.
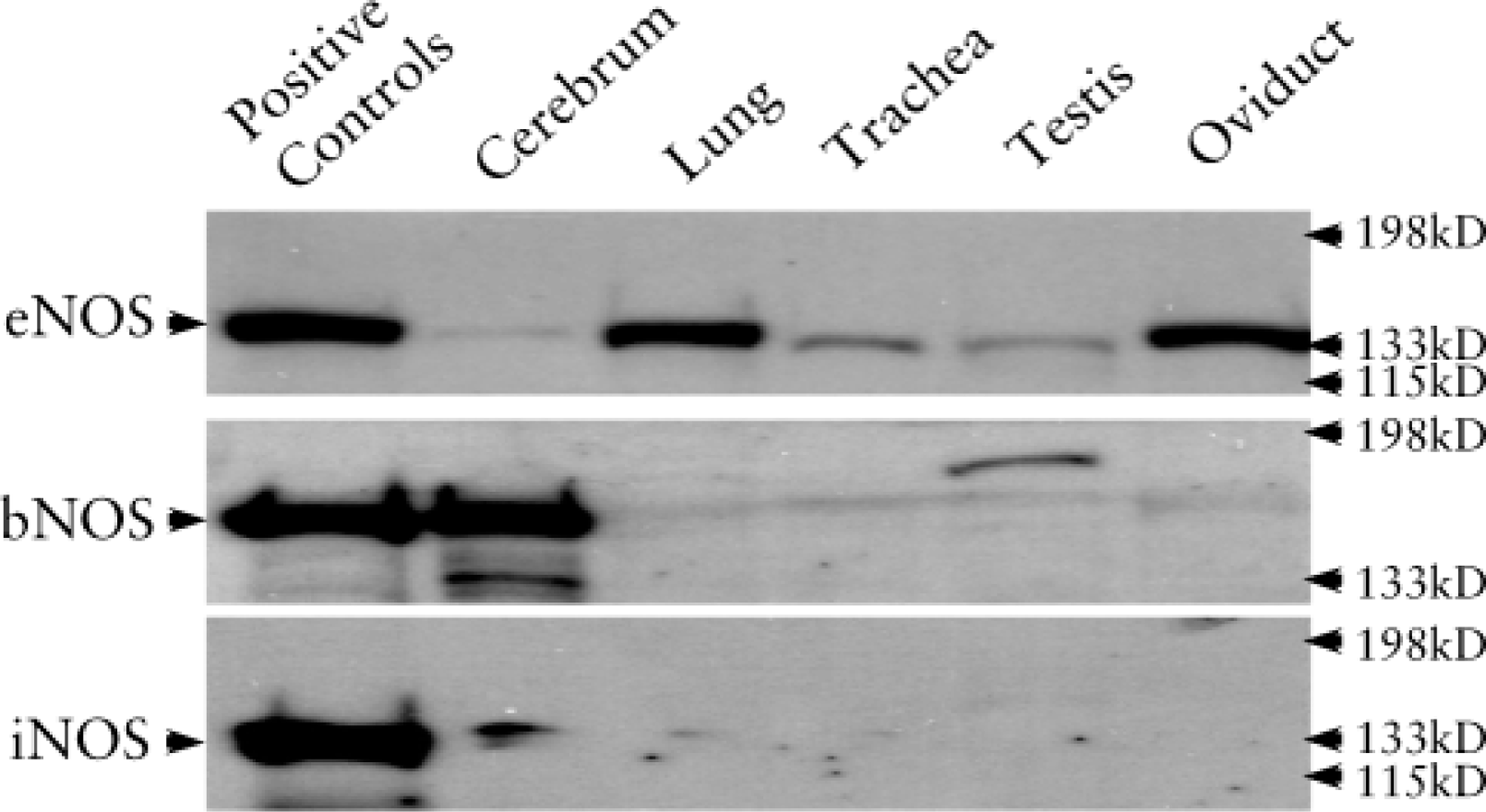
Western blotting analysis of NOS isoforms. eNOS (135 kD) was detected in all the tissues of cerebrum, lung, trachea, testis, and oviduct. bNOS (160 kD) was detected in cerebrum and testis. In the testis, the band of bNOS was at approximately 180 kD. iNOS (130 kD) was weakly detected in the cerebrum and lung.
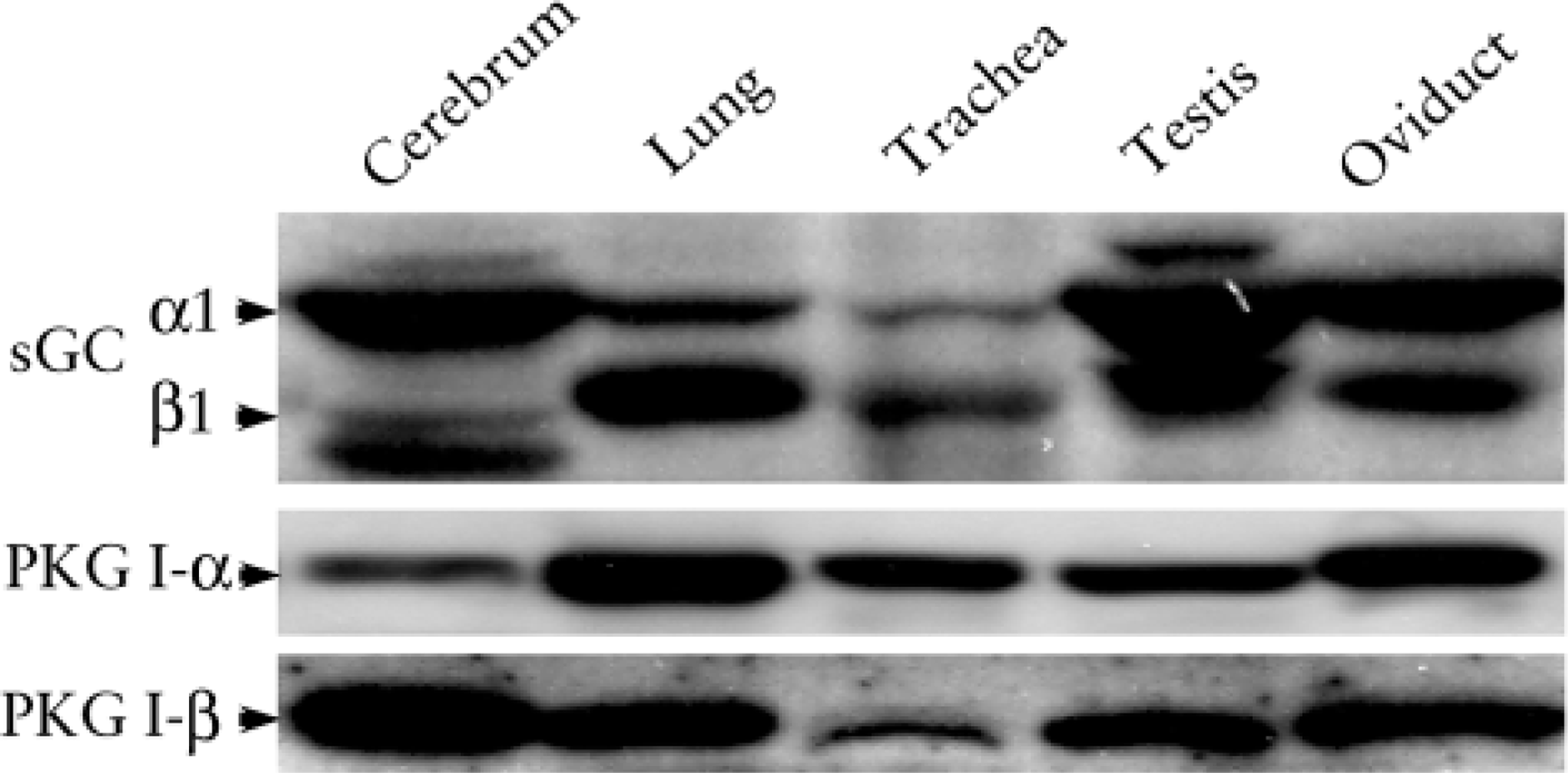
Western blotting analysis of sGC and PKG I isoforms. sGC α1, sGC β1, PKG I-α, and PKG I-β were all detected in the cerebrum, lung, trachea, testis, and oviduct. The sGC α1 was dominant in the cerebrum, testis, and oviduct. The sGC β1 was dominant in the lung and trachea.
Positive immunostaining for eNOS was detected in the ciliated epithelium (Figure 3F, blue arrows; ampulla) and the endothelia and smooth muscle in the vasculature of the oviduct (data not shown). Nonciliated epithelia were not stained by eNOS (Figure 3F, red arrow).
In the testis, eNOS IHC staining was positive in type B spermatogonia (S
The ependymal cells of the lateral ventricles exhibited a highly positive IHC expression of eNOS (Figure 3H). There was also a strong staining in the epithelia of the choroid plexus (data not shown).
Effect of l -arginine on NOx Concentration
In explants exposed to
Discussion
In mammals, ciliated epithelial cells line the respiratory tract, part of the reproductive tract, and ventricles of the central nervous system. The presence of NO in the respiratory tract was suggested by the discovery of NO in exhaled air (Gustafsson et al. 1991) and by eNOS localization in the epithelia of airway and lung (Le Cras et al. 1996; Xue and Johns 1996b; Xue et al. 1996a; Dweik et al. 1998). Here we detected eNOS in ciliated epithelia outside the lung in tissues including trachea, bronchus, oviduct, testis, and cerebrum.
Kartagener's syndrome, or primary ciliary dyskinesia, is an autosomal dominant disease in which cilia lack motility as a result of the absence of dynein arms in the cilia (Palmblad et al. 1984). Patients with this disease demonstrate the absence of NO in nasal air (Lundberg et al. 1994). Moreover, low levels of nasal NO correlate with impaired mucociliary function in the upper airways of patients suffering from respiratory tract diseases (Lindberg et al. 1997). A recent report demonstrated that primary ciliary dyskinesia is associated with a missense mutation (G894→T) in exon 7 of eNOS and with a bi-allelic tetranucleotide polymorphism in the promoter of the iNOS gene (Storm van's Gravesande et al. 2001). These results provide further support for the notion that NO is an important regulator of mucociliary function in the airways. Our findings suggest that ciliated epithelia are the probable source of exhaled NO, which may participate in the process of regulating ciliary motility.
eNOS staining was present in the oviduct also. Histologically, the wall of the oviduct consists of a mucosa, a muscular layer, and an external serosa. The epithelia of the mucosa are composed of two kinds of cells, one of which contains cilia. The other cell type is devoid of cilia and is considered to be secretory. Functionally, the oviduct is the site of fertilization and early embryonic development. The role of the oviduct in reproduction is more than a passive conduit and involves active transport of ova and sperm. The infundibulum is involved in oocyte pickup; the ampulla is the site of sperm penetration and fertilization; the isthmus is responsible for the transport of embryos into the uterus. Coordinated muscular peristalsis and ciliary movement are required for successful reproduction. However, the egg reaches the site of fertilization within a normal time frame even after blocking of rapid muscle-induced egg movements with isoproterenol (Halbert et al. 1976). This suggests that the role of cilia is important in transporting the egg.
NADPH-diaphorase activity was identified in pig (Gawronska et al. 2000) and rat (Bryant et al. 1995) fallopian tubes at all stages of the estrous cycle. In the study by Gawronska and co-workers, eNOS rather than iNOS was detected by Western blotting analysis. In the study by Bryant and co-workers, all three NOS isoform antibodies recognized a protein band at approximately 125 kD by Western blotting analysis. Actually, from our experience, the same protein will migrate differently if the pH and salt concentration of the sample varies. Therefore, it remains to be confirmed by further study whether fallopian tube NOS is a novel isoform of NOS. Gawronska et al. (2000), Chatterjee et al. (1996), and Bryant et al. (1995) address the general phenomenon that eNOS is present in the entire epithelia without regard to the presence of cilia. However, we addressed the issue that eNOS was localized specifically in the ciliated epithelia of rat oviduct and not the nonciliated epithelia. Gawronska and co-workers also found iNOS in the oviduct by IHC. In contrast to eNOS, it was confined to the endothelium of lymphatics and some blood vessels. We detected only eNOS rather than iNOS or bNOS in the oviducts by Western blotting. This supports Gawronska's view that eNOS is the main isoform of NOS expressed in the oviduct.
Earlier studies have addressed the localization of NOS in spermatozoa. eNOS (Lewis et al. 1996; O'Bryan et al. 1998) and bNOS (Lewis et al. 1996) were localized in human spermatozoa from normozoospermic samples. In addition, in the presence of L-NAME, an NOS inhibitor, sperm motility was significantly reduced (Lewis et al. 1996). This suggests that endogenous NO is necessary for adequate sperm motility.
However, controversial results have been reported. Zini et al. (1996) found that eNOS was localized to the Leydig cells and Sertoli cells rather than in the “normal germ cells.” As they stated, the tissues used were from men with either early metastatic prostate cancer, reproductive tract obstruction, or a variable degree of impaired spermatogenesis. These findings may not be generalized to normal tissues. In our study we detected eNOS and bNOS in the testis by Western blotting analysis. IHC study revealed that eNOS was localized to the type B spermatogonia, spermatocytes, spermatids, and spermatozoa, in addition to Leydig cells and Sertoli cells. These results suggest a possible role of NO in sperm maturation and hormone secretion.
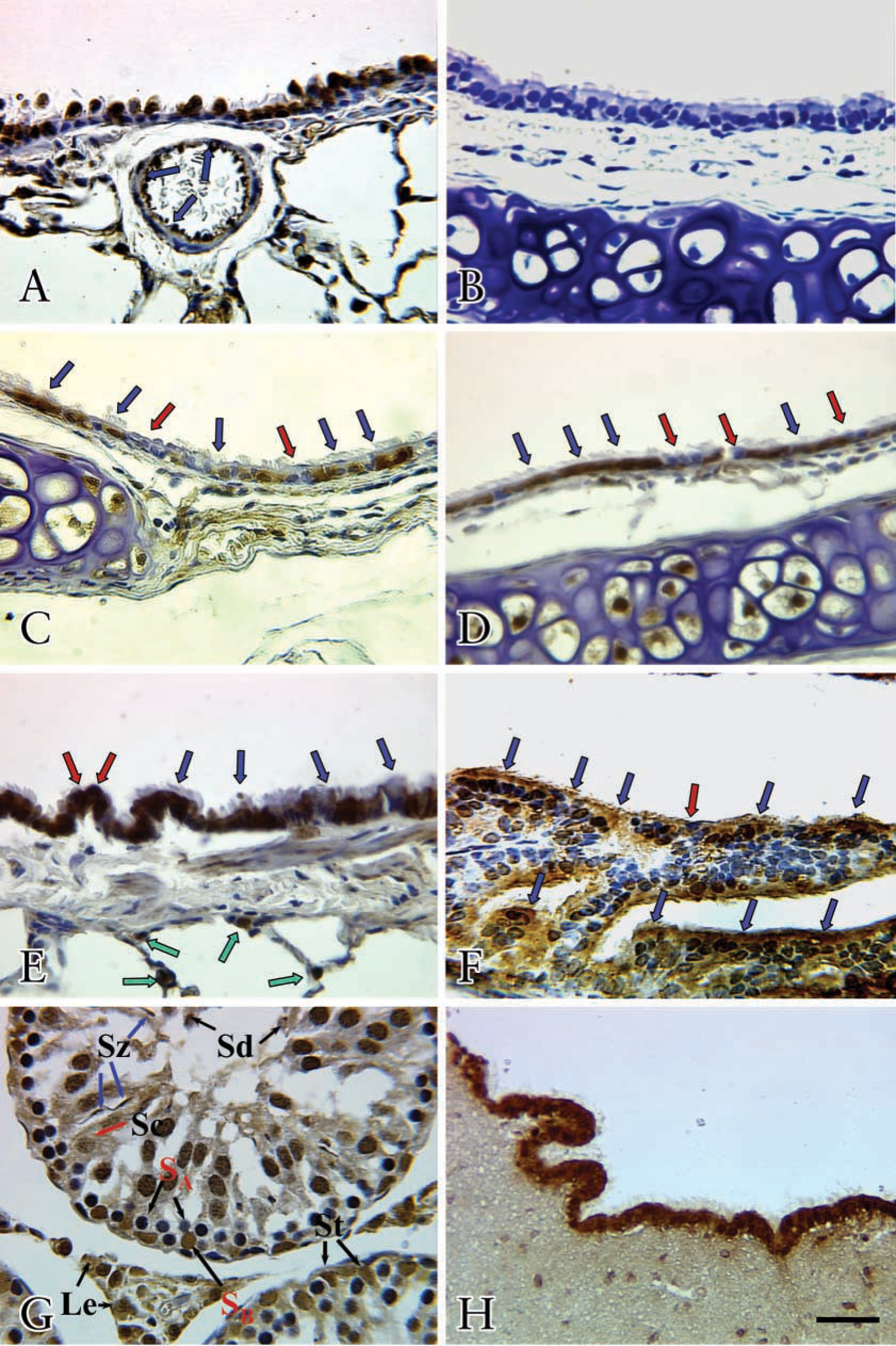
IHC localization of eNOS in ciliated epithelia. eNOS immunostaining was detected in the endothelia of the pulmonary vessel (
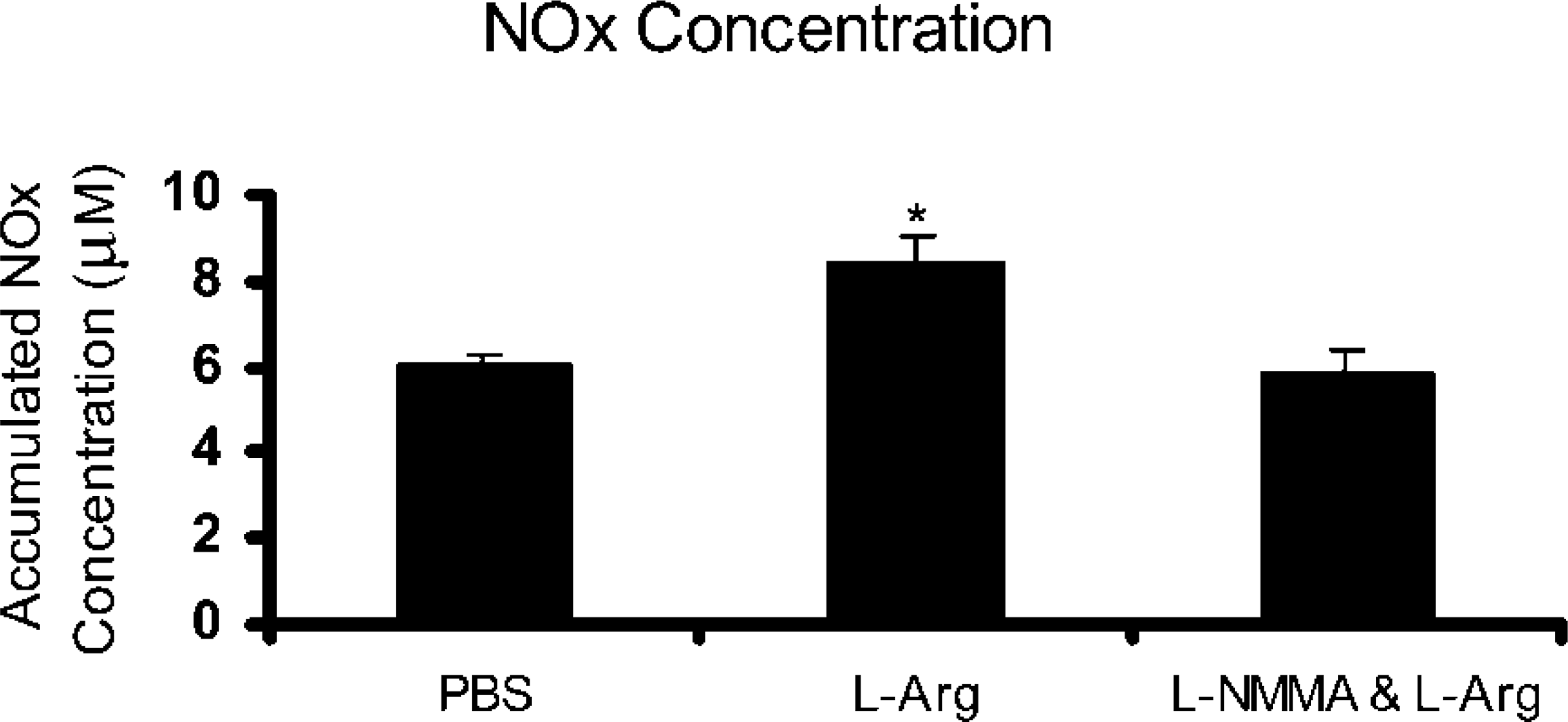
Effects of
We also found eNOS staining in the ependymal cells of ventricles. In vertebrate brain, ependymal cells line the ventricles and central canal of the spinal cord, forming a wall separating CSF and the neural tissues. There are also several reports concerning the distribution of NADPH diaphorase-positive epithelia in ependyma. However, the widespread distribution of NOS, as revealed by NADPH diaphorase histochemistry, makes it difficult to relate ependymal NO to any specific physiological function because NADPH-diaphorase detects all isoforms of NOS. Here we demonstrated that eNOS was localized in the ciliated epithelia of the ependyma. Our findings suggest that eNOS may be involved in ependymal ciliary functions.
sGC α1, sGC β1, PKG I-α, and PKG I-β were also found in the tissues of cerebrum, lung, trachea, testis, and oviduct. The sGC α1-subunit was dominant in the brain, testis, and oviduct. The sGC β1-subunit was dominant in the lung and trachea. This suggests that NO may activate different sGC subunits in different cells. As we reported in a previous study (Zhan et al. 1999a,b), PKG I-β might transduce the NO-cGMP signaling into biological responses in these ciliated epithelia. However, further study is needed to confirm this.
To confirm if the NOS was activated in the ciliated epithelia, we examined the effect of
In summary, eNOS is present in the ciliated epithelia of oviduct, trachea, bronchus, bronchiole, lung, ependymal cells, and testis. In addition,
Footnotes
Acknowledgements
Supported by NIH grants R01 HL 39706 and R01 GM 49111.
