Abstract
In this study, caveolin-1 (cav-1), an inhibitor of endothelial nitric oxide synthase (eNOS), was semi-quantified in diseased human and rabbit blood vessels. New Zealand White rabbits were fed, for 12 weeks, a high methionine diet (to induce intimal hyperplasia), 0.5% cholesterol diet, a normal diet, or the combination of both experimental diets. Excess segments of human internal mammary arteries (IMA) and radial arteries (RA) were obtained from patients undergoing coronary artery bypass surgery. eNOS and cav-1 were localized throughout both human and rabbit vessels. In rabbit arteries, eNOS was significantly increased in the endothelium overlying intimal thickening and atherosclerotic plaques compared with the adjacent endothelium overlying normal media. Interestingly, the endothelial cav-1:eNOS ratio increased 5-fold only in endothelium overlying plaques but decreased in endothelium overlying vessels with neo-intimal thickening. In human tissue, there was no difference between RA and IMA eNOS immunoreactivity in endothelium, intima, or media; however, RA endothelial, intimal, and medial cav-1 immunoreactivity increased 4-fold (
C
As cav-1 is involved in endothelial function, evidence is accumulating to suggest a regulatory role for cav-1 in both intimal hyperplasia (Hassan et al. 2004) and atherosclerosis (Frank and Lisanti 2004) formation. Both cholesterol (Feron et al. 1999) and homocysteine (Li et al. 2002) can increase cav-1 expression in cultured endothelial cells, and statins have been shown to not only increase eNOS mRNA expression (Hernandez-Perera et al. 1998) but also to decrease cav-1 mRNA and protein levels (Feron et al. 2001), suggesting that the normalization of endothelial function observed during statin therapy (Egashira et al. 1994; Treasure et al. 1995) or incubation of aortic rings in vitro with cerivastatin (Nakamura et al. 2002, 2003) might also be due to decreasing the abundance of the cav-1.
Frank and Lisanti (2004) have shown a marked decrease in atherosclerotic lesion area by ∼70% in ApoE−/− cav-1−/− double knockout mice, indicating that a lack of cav-1 inhibits atherogenesis. However, the same group has shown that neo-intima formation is exacerbated in the carotid arteries of cav-1−/− mice after surgical ligation (Hassan et al. 2004). In a recent review the authors suggest that endothelial cell cav-1 has a pro-atherogenic role, whereas smooth muscle cell cav-1 has the opposite role (Frank and Lisanti 2004).
To further identify a role for cav-1 in atherogenesis, we semi-quantified both eNOS and cav-1 immunohistochemistry in the endothelial cell layer, intima, and media of human internal mammary arteries (IMA) and radial arteries (RA). Furthermore, as we have previously reported in a rabbit model of dietary-induced intimal hyperplasia and atherosclerosis, we also semiquantified both eNOS and cav-1 immunohistochemistry in the endothelium overlying the normal vessel wall, intimal thickening, and atherosclerotic areas of rabbit thoracic aorta.
Materials and Methods
Human Tissue
Excess segments of IMA (
Rabbit Tissue
Thirty-two male New Zealand White rabbits, 3 months of age, were divided into four groups (
Immunohistochemistry
Immunohistochemistry was performed using the two-step Envision Kit (Dako; Carpinteria, CA) following the protocol provided by the manufacturer. Monoclonal antibodies to human eNOS and cav-1 were purchased from Transduction Laboratories (BD Biosciences; NSW, Australia). Monoclonal (Clone 1A4) α-smooth muscle cell actin, human macrophage marker (Ham 56), and rabbit macrophage marker (RAM-11) were purchased from DAKO. All primary antibodies (dilution 1:100) were incubated overnight. A monoclonal antibody raised against yeast glucose oxidase diluted 1:100 was used as a negative control. Sections were washed in 10 mM Tris HCl (pH 7.3) and incubated for 2 hr with the envision molecule, which has both mouse immunoglobulins and horseradish peroxidase attached to it. The slides were rinsed and both the red AEC chromogen (3-amino-9-athylcarbazole) and the brown DAB chromogen (diaminobenzidine) were used to identify binding sites. Slides were counterstained with heamatoxylin, blued in Scotts tap water, dehydrated, and mounted in DPX medium.
Semi-quantification of eNOS and Cav-1 Proteins
Sections were placed under a microscope (Olympus BX50, X40 lens; Olympus Optical Company, Shibuya-ku, Tokyo, Japan) and fitted with a Fujifilm HC2000 color digital camera (Fuji; Bertold Australia, Victoria, Australia) (James and Hauer-Jensen 1999; Rimsza et al. 1999; Dean et al. 2003). For human arteries, an unbiased section of the vessel wall was randomly selected. For rabbit tissue, three sections of aorta were randomly selected by an observer who was blinded to the groups: (a) an endothelial section overlying an area free of pathology, (b) an endothelial section overlying an area with intimal thickening, and (c) an endothelial section overlying an atherosclerotic plaque. The largest possible stretch of endothelium was sampled per aorta while the observer was blinded to the groups. The digital image was captured using the MCID Elite 6.0 software (Imaging Research Inc.; GE Healthcare Technologies, Waukesha, WI). The color intensity, saturation, and hue were adjusted to allow the imaging software to acquire either the brown or red immunostaining. The ribbon tool on the MCID Elite 6.0 software was used to obtain the largest possible area of endothelia that did not have any bound macrophages.
In each group, the color intensity value obtained with the MCID software (Imaging Research) was multiplied by the proportional staining area value, and each group was divided by the mean value obtained from the control group normal endothelial layer [described above as (a)]. Please note: this value also acts as the “internal control.” Thus, the endothelial layer overlying normal media taken from the control group is reported as 1 ± SEM, and all other values are reported as “fold increase over control.”
Cav-1:eNOS Ratio
To determine the cav-1:eNOS ratio in rabbit aortae, all endothelial cav-1 and eNOS quantitative results were “normalized” by dividing by the mean results obtained from the endothelium overlying normal vessel wall in the control group. In this way, the results obtained from the normal areas of the wall in the control group acted as the “internal control” and its represented value is 1 ± SEM. For example, the cav-1 data obtained from the endothelium overlying plaques were divided by the data obtained from the endothelium overlying the normal wall in the control group. To determine the ratio, normalized cav-1 data were then divided by the normalized eNOS data.
To determine the cav-1:eNOS ratio in human tissue, all RA endothelial, medial, and intimal cav-1 and eNOS quantitative results were “normalized” by dividing it by the mean results obtained from the IMA. In this way, the results obtained from the IMA acted as the “internal control” and its represented value is 1 ± SEM. For example, the RA cav-1 media data were divided by the mean data obtained from the IMA media. To determine the ratio, the normalized cav-1 data were then divided by the normalized eNOS data.
For both the human and rabbit study, immunoquantification of eNOS and cav-1 was performed on three separate serial sections, three separate times on three separate days, and all results showed similar findings. In the Results section, we report only the third assessment.
Statistical Analysis
Statistical analyses were carried out using Graphpad Prism version 3.0 (Graphpad Software; San Diego, CA). Experimental data were analyzed using a one-way ANOVA to determine whether there were significant differences among the four experimental groups, and Tukey's posthoc test was applied to determine where between the groups the statistical difference lay. All data are expressed as means ± standard error of the mean (SEM).
Comparisons between endothelium overlying normal media and endothelium overlying neo-intimal thickening [(a) and (b), see above] for all groups were also analyzed by oneway ANOVA to determine whether there were significant differences among the four experimental groups, and Tukey's posthoc test was applied to determine where between the groups the statistical difference lay. In all cases, statistical significance was accepted at a level of
Results
Human Tissue
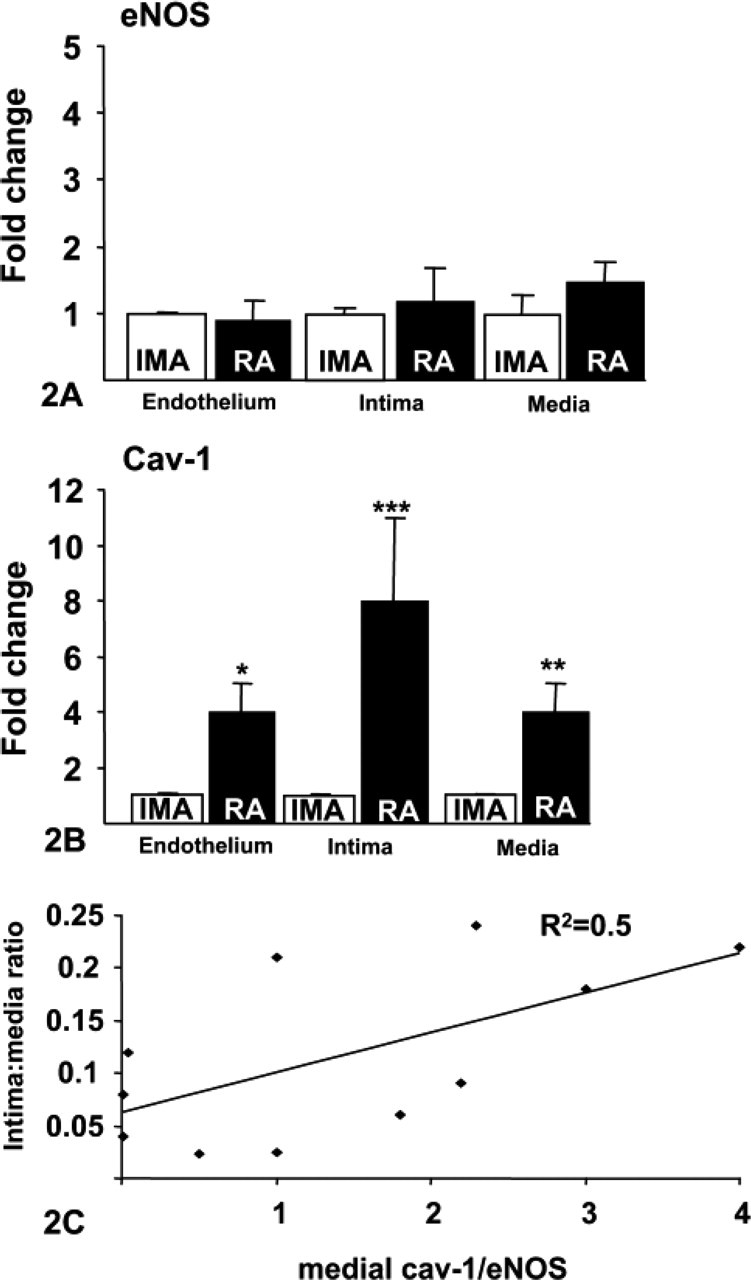
In the RA, eNOS and cav-1 were localized to the endothelium and media and to cells within the neointima (Figures 11-A1). There was no significant difference in endothelial or intimal eNOS protein between the IMA and RA; however, medial eNOS was increased 1.5-fold in the RA compared with the IMA, but this failed to reach significance (Figure 2). However, endothelial and medial cav-1 immunoreactivity increased 4-fold (
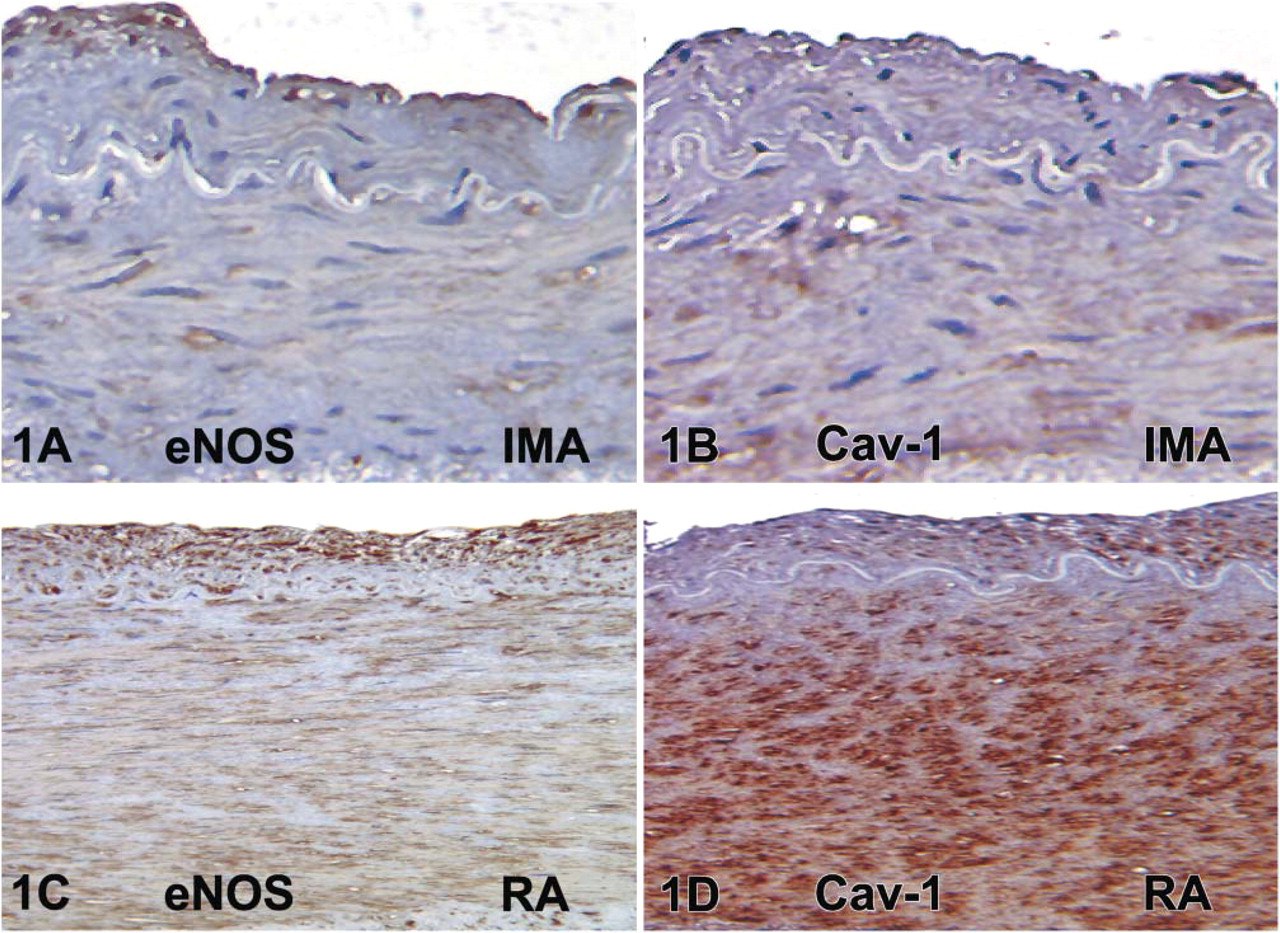
Photomicrographs of human internal mammary arteries (IMA) (
(
The increase in intimal cav-1 was due to an increase in the number of cav-1-positive cells. These cells were also positive for α-smooth muscle cell actin (Figure 3) but not macrophages (Figure 3B, Ham 56). In fact, the population of intimal cells within the RA intima was almost all α-smooth muscle cell actin positive, with only scarce macrophages present (Figure 3B, inset).
Rabbit Tissue
Cav-1 was identified in the endothelial layer overlying normal media and intimal thickening of the thoracic aorta of the control, methionine, cholesterol, and the combination group (Figures 4A, 4C, and 4E—only combination group shown). In addition, cav-1 was also identified within the atherosclerotic plaques of the thoracic aorta from the high dietary cholesterol and high dietary cholesterol plus methionine group (Figure 4E—only combination group shown).
Immunolocalization of eNOS in these experimental animals has been previously published (Zulli et al. 2003). In this study, we confirm our previous eNOS immunohistochemical results whereby eNOS immunoreactivity was observed in the combination group (Figures 4B, 4D, and 4F—only combination group shown). There were two types of cells within the plaques that stained for both eNOS and cav-1. These cells were specifically α-smooth muscle actin-positive cells (Figure 3C, possibly smooth muscle cells or myofibroblasts) and macrophages (Figure 3D, RAM-11 positive).
In the endothelial layer overlying thickened intima of the control group, eNOS immunodensity significantly increased 2.8-fold compared with the endothelial layer overlying disease-free areas (
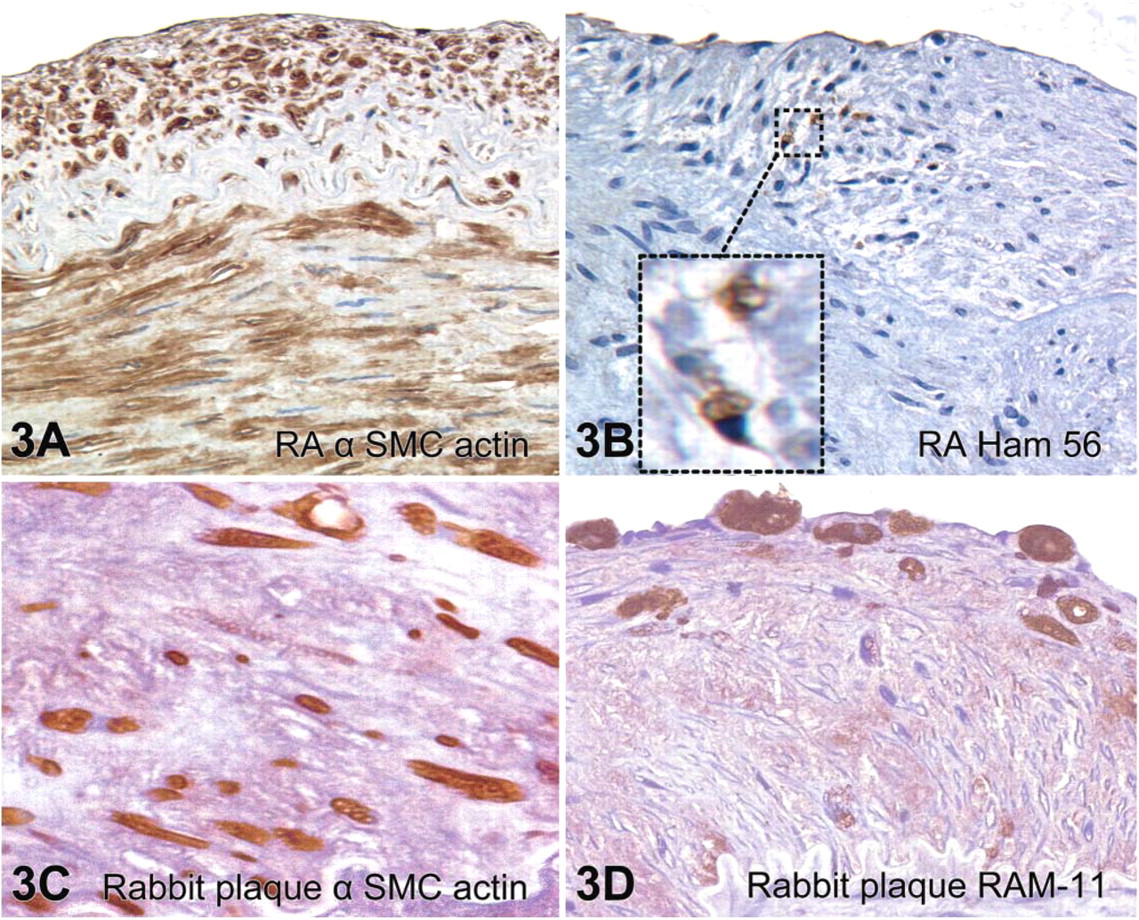
(
Interestingly, cav-1 immunodensity was markedly increased only in the endothelium overlying plaques in both the Chol group (75-fold increase,
Discussion
The major finding in this investigation is that cav-1 immunoreactivity is increased in both rabbit and human diseased blood vessels. We have also associated an increase in cav-1 compared with eNOS immunoreactivity with an increase in the intima:media ratio of human vessels and in the endothelial layer overlying atherosclerotic plaques in rabbit vessels.
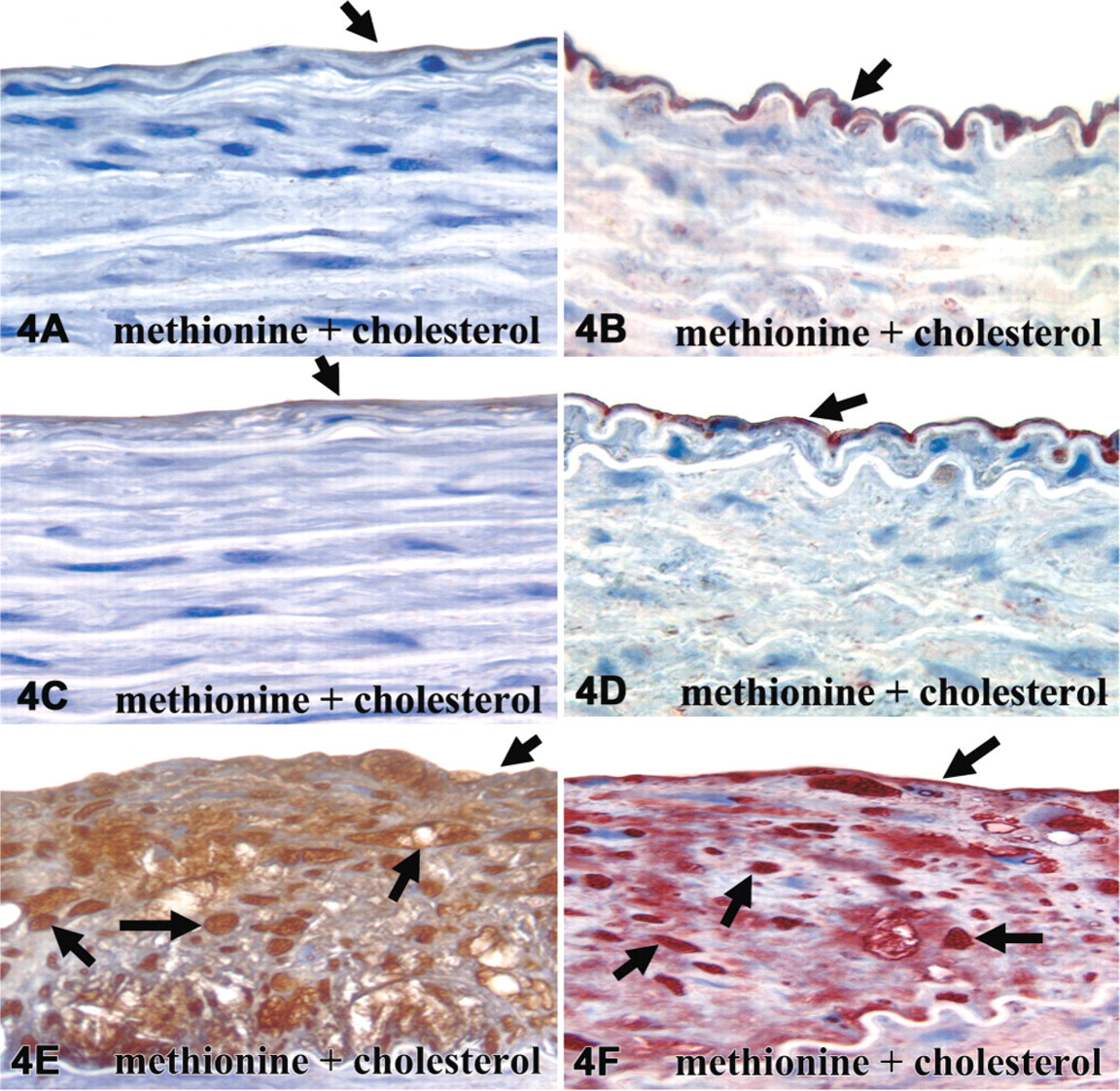
Photomicrographs of rabbit aorta cav-1 and eNOS binding in the endothelial layer overlying normal media (
An increase in endothelial cav-1 has been suggested to induce endothelial dysfunction. For example, recent studies have demonstrated that cav-1 regulates eNOS function in the endothelial cell. Evidence from cav-1 knockout mice showed that low concentrations of bradykinin (Drab et al. 2001; Razani et al. 2001) completely relaxed arteries in these mice, whereas aortic rings from wild-type mice showed only 20% relaxation. In addition, the incubation of mouse aortic rings with a peptide mimicking the eNOS binding site on cav-1 completely inhibits arterial relaxation induced by ACh (Bucci et al. 2000). Thus, these studies indicate that the association of eNOS with cav-1 may also be a novel marker for eNOS enzymatic activity. Indeed, we have shown by semi-quantitative techniques that cav-1 immunoreactivity is increased only in the endothelium overlying plaques compared with the endothelium overlying neo-intima or normal wall in our rabbit model. Although eNOS immunoreactivity was also significantly increased in these areas, the amount of cav-1 was 5-fold greater. Thus, we suggest that cav-1 decreases eNOS activity in the endothelium overlying plaques resulting in a possible local NO decrease and increase in localized cellular activity. Such a decrease in local NO can stimulate leukocyte recruitment (Eriksson 2004) and smooth muscle cell proliferation (Ignarro et al. 2001), which are key events in atherogenesis. This theory is supported by a recent investigation by Frank and Lisanti (2004). The authors show that atherosclerosis is reduced by ∼70% in cav-1/ApoE double-knockout mice fed a Western-type diet compared with the ApoE control. In addition, recent evidence suggests that one of the major mechanisms whereby statins improve endothelium-dependent relaxation is due to its effects on increasing NO bioavailability (Vaughan et al. 1996), which may not only be due to its effects on eNOS mRNA and enzyme (Hernandez-Perera et al. 1998) content but also via the downregulation of cav-1 protein and mRNA (Feron et al. 2001).
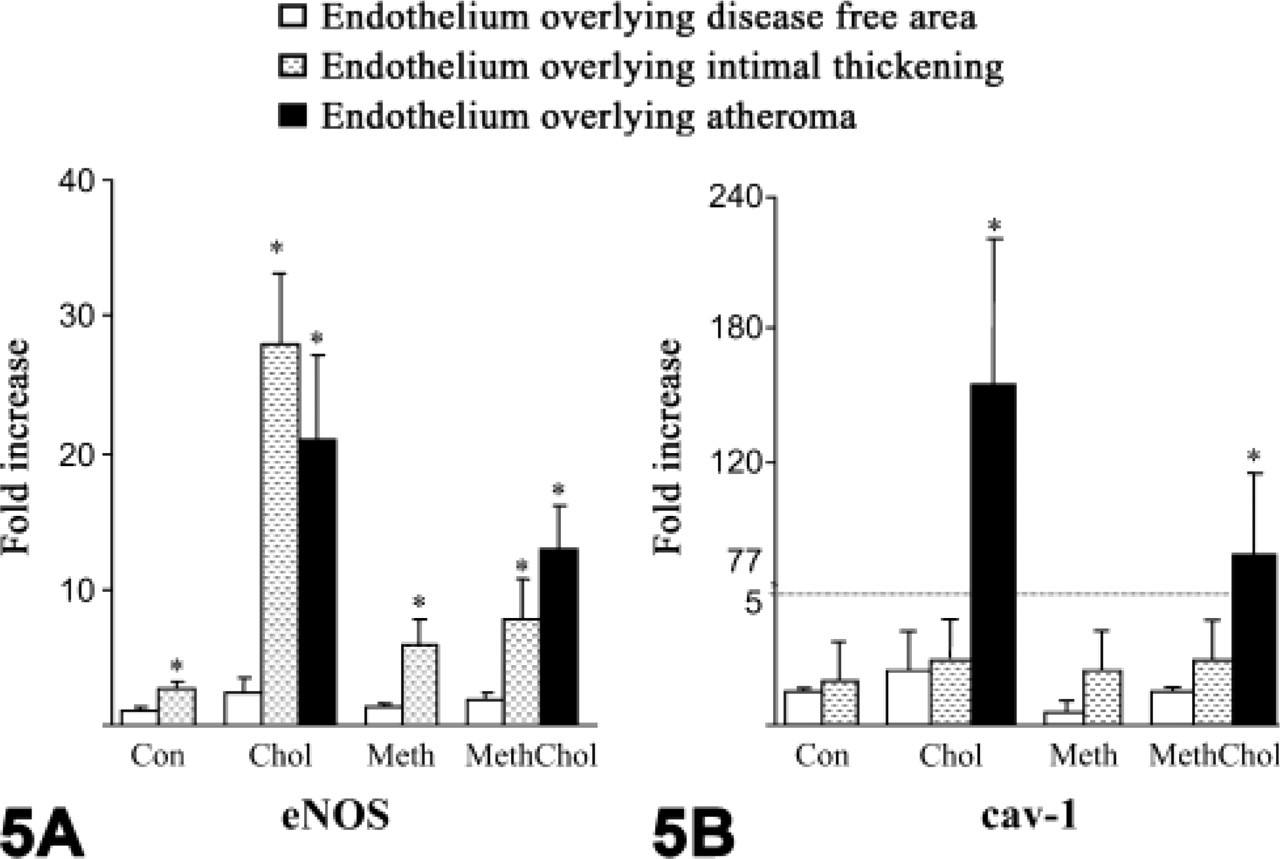
Graph showing the immunoquantitative results of both eNOS and cav-1 in rabbits. (
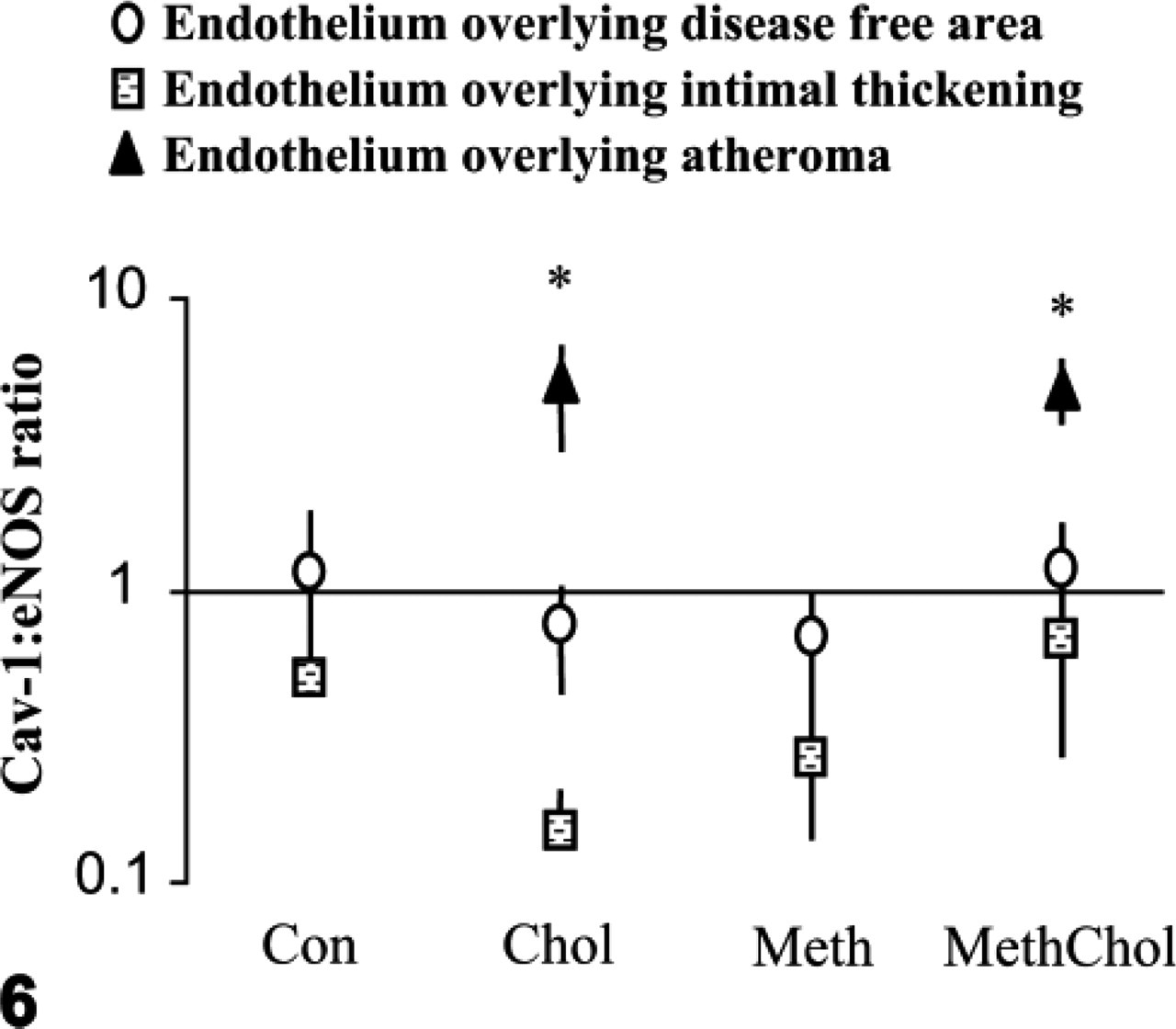
Graph showing the cav-1:eNOS ratio of the endothelial layers overlying normal media and neo-intima overlying plaques. Interestingly, in endothelia overlying normal media in all four groups, the cav-1:eNOS ratio remained close to 1:1, whereas in endothelia overlying neo-intimal thickening, the cav-1:eNOS ratio remained <1, suggestive of “active” eNOS. However, in endothelia overlying plaques, the cav-1:eNOS ratio was raised ∼5-fold (
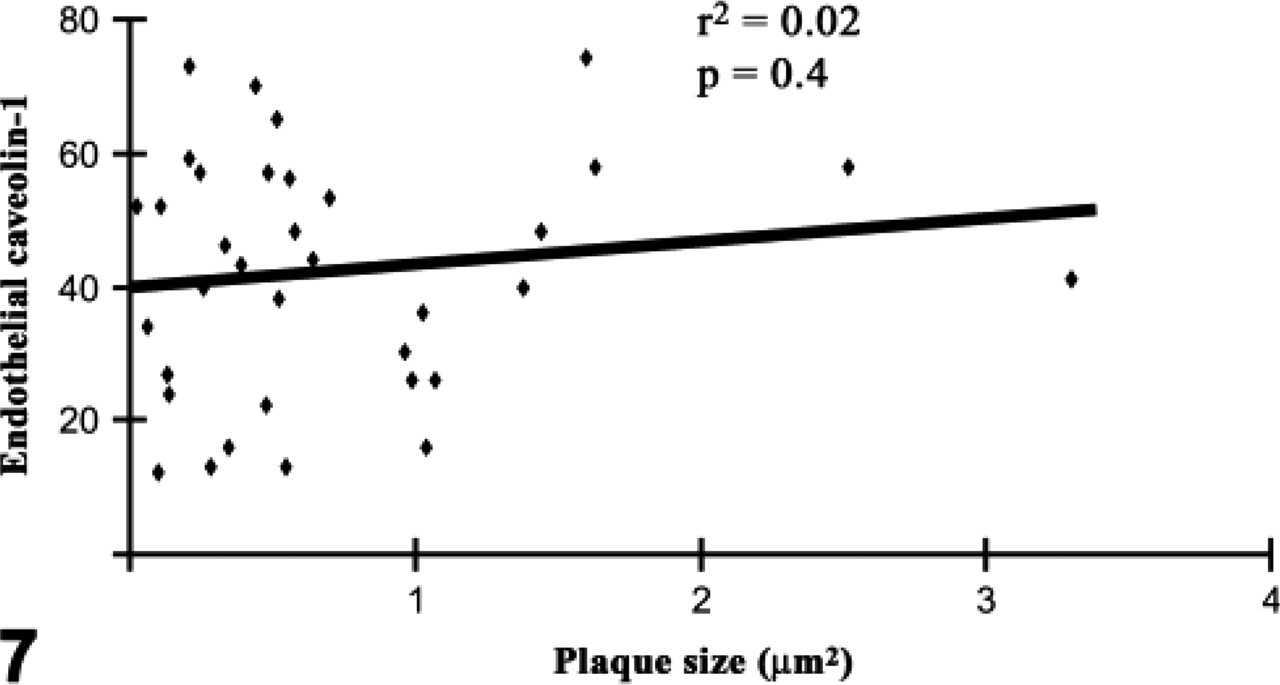
Graph showing the amount of endothelial cav-1 immunoreactivity vs plaque size. There was no correlation between the size of the plaque examined and the amount of endothelial cav-1 protein, suggesting that an increase in endothelial cav-1 is not necessary for plaque growth.
We have previously shown that endothelium-dependent relaxation was impaired in the abdominal aorta of these rabbits fed high dietary cholesterol and methionine and virtually abolished by the combination on these two diets, but eNOS protein was not compromised in any group (Zulli et al. 2003). Here we suggest that such eNOS enzymatic dysfunction in the group fed high dietary cholesterol plus methionine might not be due to an association of eNOS with cav-1 protein, because eNOS protein was increased in all endothelial layers including the layers overlying neo-intimal thickening and plaques. In contrast, cav-1 was increased only in the endothelial layers overlying plaques, thus increasing the cav-1:eNOS ratio ∼5-fold. Such results suggest a close association between cav-1 and atherogenesis.
Caveolae also play an important role in maintaining vascular tone via the detection of fluid shear stress generated by blood flowing over the endothelium. Thus, any change in local rheology of blood due to the presence of atherosclerosis would also affect the local shear stress present on the endothelial cells overlying intimal thickening and atherosclerotic plaques. In this case, an increase in shear stress would be expected in areas of intimal thickening and plaque formation, which in itself can increase cav-1 (Boyd et al. 2003) and eNOS synthesis (Davis et al. 2004). Therefore, in the rabbit model of atherosclerosis it is not clear at this stage whether the increase in cav-1 detected in endothelia overlying plaques is a cause or consequence of plaque development.
Here we report that the RA and IMA express similar endothelial, medial, and intimal eNOS protein immunoreactivity. These results are in contrast with Gaudino et al. (2003), who have shown a marked increase in medial eNOS immunoreactivity in the IMA compared with the RA and saphenous vein. This may be due to technical variations, as Gaudino and colleagues did not process vessels concomitantly or mount all vessels in one paraffin block to maintain immunohistochemical procedures exactly the same between all vessels. Furthermore, Cable et al. (1999) observed that the RA exhibits lower eNOS immunoreactivity compared with the IMA; however, the authors did not present their data and did not quantitate their results. Our study is the first to semi-quantitate eNOS immunoreactivity in these two vessels using concurrent tissue processing and immunohistochemical techniques.
However, in this study we identify a possible role for cav-1 in human diseased vessels. We observed a significant increase in endothelial, intimal, and medial cav-1 immunoreactivity in the diseased radial arteries compared with diseased areas of the internal thoracic artery. Here we propose that, in contrast to endothelial cav-1, medial cav-1 might be inhibiting eNOS activity. For example, although it has been shown that the genetic transfer of eNOS mRNA to the RA improved vascular function, even though eNOS was previously present in these vessels (Cable et al. 1999), we speculate that the excess eNOS may override the negative effects of increased cav-1 expression. In this case, it would be worthwhile to study the subcellular localization of cav-1 using transmission electron microscopy and gold-labeled immunohistochemistry to determine the amount of cav-1 present in non-caveolar locations.
The diseased human radial arteries showed a marked increase in medial cav-1 protein, a phenomenon not observed in our rabbit model. We suggest that this difference could be due to different blood vessel types, as the IMA also showed minimal cav-1 immunoreactivity, which is similar to the results observed in our animal model. Although it has been shown that a lack of cav-1 gene exacerbates neo-intimal thickening (Hassan et al. 2004) in mouse, we suggest that in the RA, the excess cav-1 in the media is inhibiting medial eNOS activity to inhibit NO release, thus promoting smooth muscle cell migration and proliferation into the media. This idea is reflected by the strong correlation observed between the medial cav-1:eNOS ratio and the intima:media ratio of these blood vessels.
In conclusion, we report that cav-1 immunoreactivity is increased within the atherosclerotic plaques of rabbits and within the media and intima of diseased human arteries, suggesting a close relationship between cav-1 and blood vessel disease.
