Abstract
The MRP (multidrug resistance-associated protein) transmembrane transporter, which actively transports a wide variety of lipophilic substrates out of cancer cells, has been suggested to play a major role in cell detoxification via efflux of glutathione conjugates. Because bronchial epithelial cells are constantly exposed to environmental pollutants, MRP might be a particularly important defense mechanism against xenobiotics. This study was therefore designed to investigate MRP localization by immunohistochemistry in bronchial epithelial cells collected by scraping from surgical specimens. In parallel, MRP mRNA was detected by reverse transcriptase chain reaction (rt-PCR) in bronchial cell lysates. However, the pattern of protein expression differed markedly according to cell type. In ciliated epithelial cells, immunostaining was restricted to the basolateral surface, without any labeling at the apical surface, which is at variance with the localization of CFTR and MDR1 proteins, other members of the same family of transporters. In basal cells, MRP was present over the entire circumference of the plasma membrane. Basal cells were identified by their morphology and specifically after incubation with an anticytokeratin 17 monoclonal antibody. In conclusion, the different patterns of localization suggest specific roles for MRP in basal and ciliated cells.
T
Materials and Methods
Bronchial Epithelial Cells
Normal bronchial epithelial cells were collected by gentle scraping of the lumen of a normal main or lobar bronchus of a surgical specimen. These specimens were collected in 21 patients (15 men, six women) who underwent pulmonary resection for primary or secondary lung carcinoma. Thirteen were current smokers, five were ex-smokers who had quit smoking for more than 2 years before surgery, and three were non-smokers. The bronchial section was previously examined and considered to be histologically normal. The quality of the samples was controlled by phase-contrast microscopy showing more than 97% epithelial cells, predominantly basal cells and ciliated cells (viable as assessed by their ciliary movements). Cytocentrifuge preparations were made when the material was sufficient (n = 11) and were stored at -80C until immunocytochemistry (ICC).
Identification of basal cells was performed by ICC using a specific anti-human cytokeratin 17 MAb (Dako; Glostrup, Denmark) as previously reported (Troyanovsky et al. 1989). The ICC method was identical to that described below for MRP ICC.
T47D cells from a breast carcinoma cell line, purchased from the American Type Culture Collection (Rockville, MD), were used as positive controls for rt-PCR and ICC. We have previously checked, using rt-PCR and ICC, that T47D cells express MRP in the same way as MCF7 cells, another breast cancer cell line (Flens et al. 1994).
Evaluation of MRP mRNA Expression by rt-PCR
Preparation of cDNA. Bronchial epithelial cells were pelleted by centrifugation. Total RNA was isolated by acid guanidine thiocyanate-phenol-chloroform as previously described (Chomczynski and Sacchi 1987). The absence of RNA degradation was confirmed by agarose gel electrophoresis. The RNA concentration was determined by spectrophotometry. Total RNA was stored at -80C. cDNA was synthesized from 1–2 μg of total RNA in a 50-μl reaction volume containing rt buffer (Gibco-BRL Life Technologies; Les Ulis, France), 0.1 mM of each dNTP (Boehringer Mannheim France; Meylan, France), 0.2 mM DTT (Gibco-BRL), 200 ng of random hexanucleotide primers (Pharmacia Biotech; Saint-Quentin-en-Yvelines, France), and 100 U MMLV reverse transcriptase (Superscript II, Gibco-BRL). After incubation at 42C for 15 min, cDNA was stored at -20C.
PCR Amplification. For PCR, 5 μl of the cDNA mixture was amplified in a 25-μl reaction volume containing 2.5 mM MgCl2, 0.5 mM of each dNTP, 0.5 U Taq polymerase (ATGC Biotechnologie; Noisy-le-Grand, France), and 37.5 pmol of sense-strand and antisense-strand MRP primers. β2-Microglobulin (β2m) cDNA was co-amplified in the same reaction with 20 pmol of each β2m primer to control the quality of the cDNA and for quantification. Thermal cycling was performed on a Kontron thermoblock (Biometra-Trio-Thermoblock; Tampa, FL). After an initial denaturation at 95C for 5 min, each sample underwent 30 cycles of denaturation at 95C for 1 min, annealing at 55C for 1 min, and extension at 72C for 1 min. Final extension at 72C lasted 7 min. In a preliminary experiment using a range from 10 to 40 cycles for amplification, we confirmed that 30 cycles was still in the exponential phase for MRP as well as for β2m (results not shown). Negative control reactions were included by omitting RNA, reverse transcriptase, or cDNA template. PCR products were separated on a 2% agarose gel containing ethidium bromide (Bioprobes Systems; Montreuil sous Bois, France).
Primers. MRP primers (Oligo Express; Paris, France) were designed according to previously published sequences (Abbaszedegan et al. 1994): MRP upstream, 5'-GGA CCT GGA CTT CGT TCT CA-3’ (residues 4109–4128); MRP downstream, 5'-CGT CCA GAC TTC TTC ATC CG-3’ (residues 4380–4399), resulting in a 290-
Quantification of the PCR Products. Semiquantitative rt-PCR analysis was performed on photographs from ethidium bromide-stained agarose gels, scanned and analyzed with a computerized image analysis system (Biorad Imaging Densitometer GS670; Ivry-sur-Seine, France). For each of the samples, MRP mRNA content was expressed as the ratio of the densitometry value of MRP in relation to that of β2m.
Statistical Analysis. The number of samples with a positive MRP mRNA detection was compared in the two groups of current smokers on the one hand (n = 13), and ex- and non-smokers on the other hand (n = 8) using the Fisher's exact test. The results of MRP mRNA semiquantification obtained in each group were expressed as means and standard errors. The Mann-Whitney U-test was used to compare the mean ratio in the two groups. Statistical significance was set at p<0.05.
Evaluation of MRP Protein Localization by Immunocytochemistry
Anti-MRP Antibodies. Two anti-MRP MAbs were tested for immunodetection of MRP protein: MC-201 and MC-202 (Syrinx–Diagnostika; Frankfurt, Germany). Both are reported to recognize internal epitopes and not to crossreact with the human MDR1 and MDR3 P-glycoproteins (Flens et al. 1994). MC-201 is a rat IgG2a MAb and MC-202 is a murine IgG1 MAb.
Immunocytochemical Procedure. Conditions for immunodetection of MRP were evaluated using the T47D human breast cancer cell line. The two MAbs (MC-201 and MC-202) were used at various dilutions. The best immunostaining was obtained using MC-201 at a 1:40 dilution. MAb MC-201 was therefore used for MRP immunolocalization in bronchial epithelial cells.
Centrifuge preparations of bronchial epithelial cells collected by scraping were air-dried at room temperature for 1–2 hr and were fixed in acetone for 10 min at 4C. The slides were incubated for 20 min with normal rabbit serum (1:70; Vector, Burlingame, CA), followed by MAb MC-201 (1:40) incubation for 1 hr. MAb binding was detected using biotinylated rabbit anti-rat IgG (1:200) and avidin–biotin complex (1:50; Vector). Bound peroxidase was developed with diaminobenzidine tetrahydrochloride (Vector) and H2O2. The slides were counterstained with hemalum and mounted for examination.
Various negative controls were performed, omitting either the primary or the secondary antibody or substitution of the primary antibody by an isotype-matched rat nonimmune IgG. T47D cells were used as positive controls.
Results
Detection of MRP mRNA by rt-PCR in Bronchial Epithelial Cells
MRP mRNA was detected in 18 (86%) of the 21 samples tested. In a series of nine cases, two independent PCRs were performed in two different batches and their results were completely concordant. As illustrated in Figure 1, after reverse transcription and amplification with MRP-specific primers, a 290-
Immunolocalization of MRP Protein in Bronchial Epithelial Cells
Similar results were obtained in all of the 11 samples analyzed (from nine smokers and two ex-smokers).
Basal cells were clearly identified on the basis of their morphology and their intense labeling when incubated with the anti-CK 17 MAb (Figure 3A). After incubation with the anti-MRP MC-201 MAb, strong labeling was observed on the entire circumference of the plasma membrane (Figure 3B). No intracytoplasmic staining was observed.
Ciliated epithelial cells were identified by phase-contrast microscopy and by the absence of staining when incubated with the anti-CK17 MAb. After incubation with the anti-MRP MC-201 MAb (Figure 3D), the labeling pattern was different from that observed in basal cells. No immunostaining was detected at the apical region. In contrast, dense, continuous immunostaining was observed along the basolateral membrane of the cells. No intracytoplasmic staining was observed. Similar labeling was observed at the basolateral periphery of mucous cells.
Negative results were obtained when non-immune rat IgG was used instead of specific anti-MRP MAb (Figure 3C).
Discussion
Analysis of the distribution of ABC proteins in bronchial epithelial cells constantly exposed to environmental pollutants is particularly important to the understanding of the mechanisms of resistance to xenobiotics. Indeed, transporters such as MDR1 Pgp or MRP exert various roles, providing a major contribution to cell detoxification.
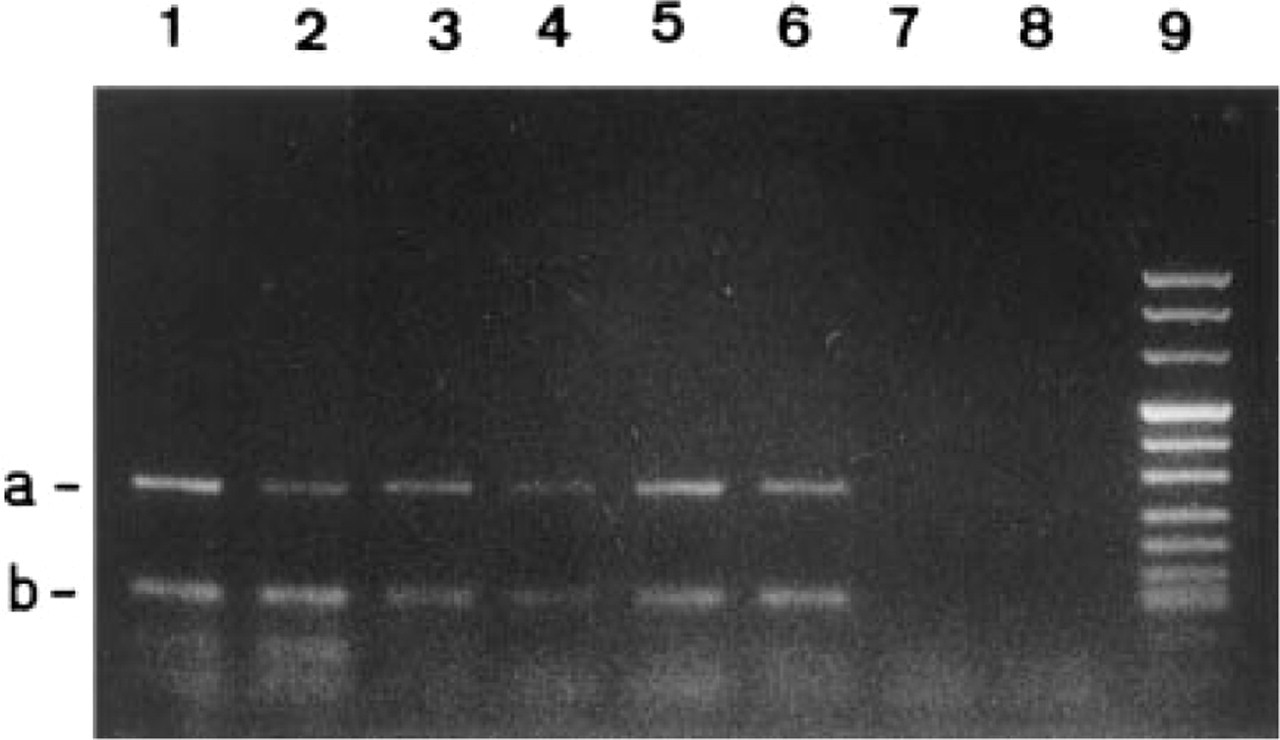
rt-PCR of MRP (
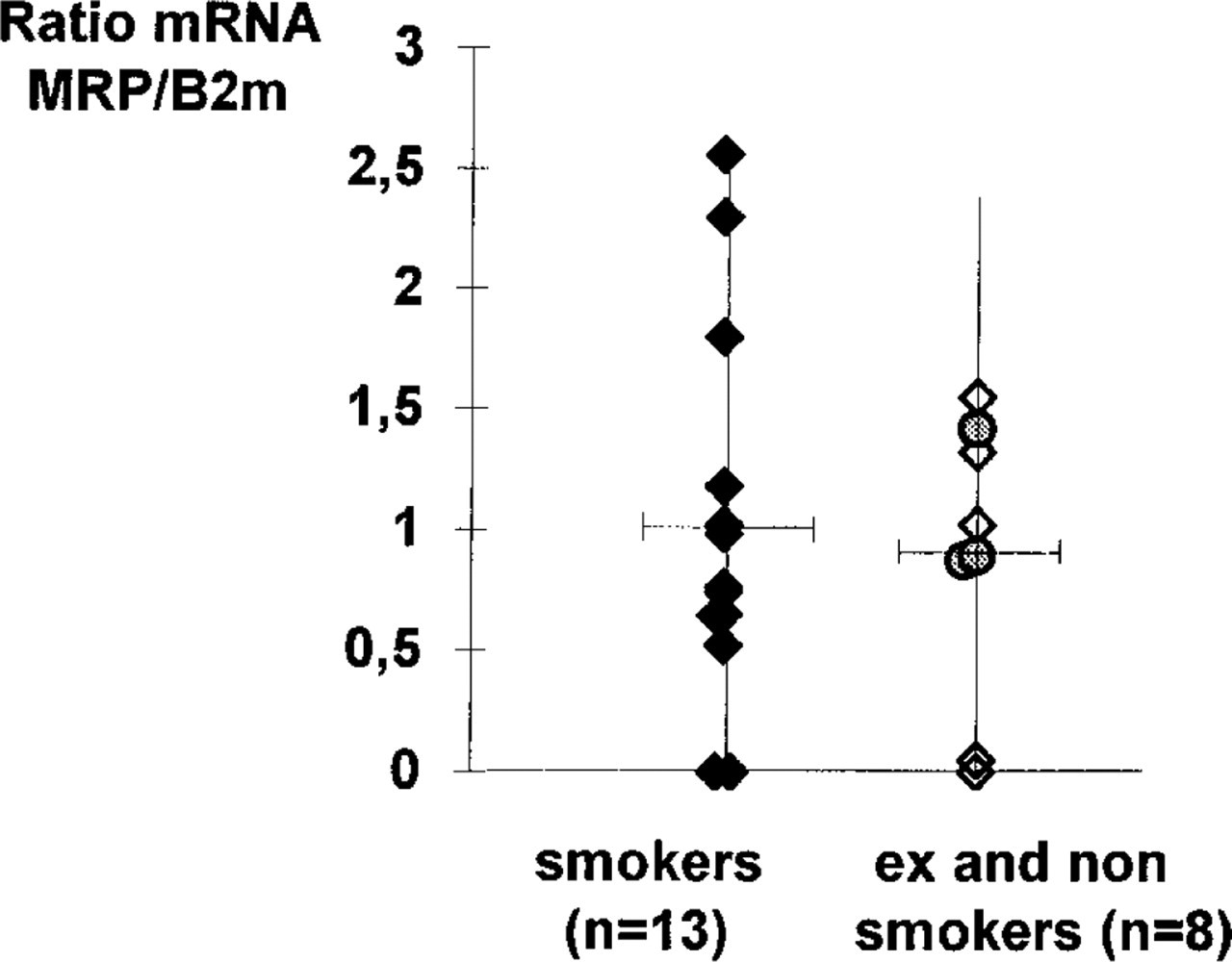
Semiquantitative rt-PCR analysis by densitometric evaluation of MRP mRNA compared to β2m mRNA (♦, smokers; ⋄, ex-smokers; ○, non-smokers). No statistically significant difference was observed (p<0.9, Mann-Whitney test)
In the present study, MRP mRNA was detected by rt-PCR in the majority of samples, in smokers as well as in non- or ex-smokers. Similar findings were previously reported in non-neoplastic bronchial epithelium, either by in situ hybridization (Thomas et al. 1994) or by RNase protection assay (Nooter et al. 1996). No statistically significant difference was observed when the levels of MRP expression were compared in the two groups of samples. This suggests an absent or limited induction of MRP gene expression by current smoking. We detected the MRP protein in all samples tested, independently of the patient's past or current smoking habits. However, the pattern of expression differed according to the bronchial cell subtype. In basal cells, identified by use of an anti-CK17 MAb, dense labeling was observed over the entire circumference of the plasma membrane after incubation with the anti-MRP MAb. This finding is in accordance with previous results (Nooter et al. 1996). In contrast, in ciliated cells we found that the MRP protein was distributed along the basolateral membrane but not at the apical region. Such a localization was also observed for mucous cells. In contrast, MRD1 Pgp (Lechapt–Zalcman et al. 1997) and CFTR (Crawford et al. 1991; Puchelle et al. 1993), proteins known to be restricted to the apical part of the bronchial ciliated cells, were never detected in basal cells.
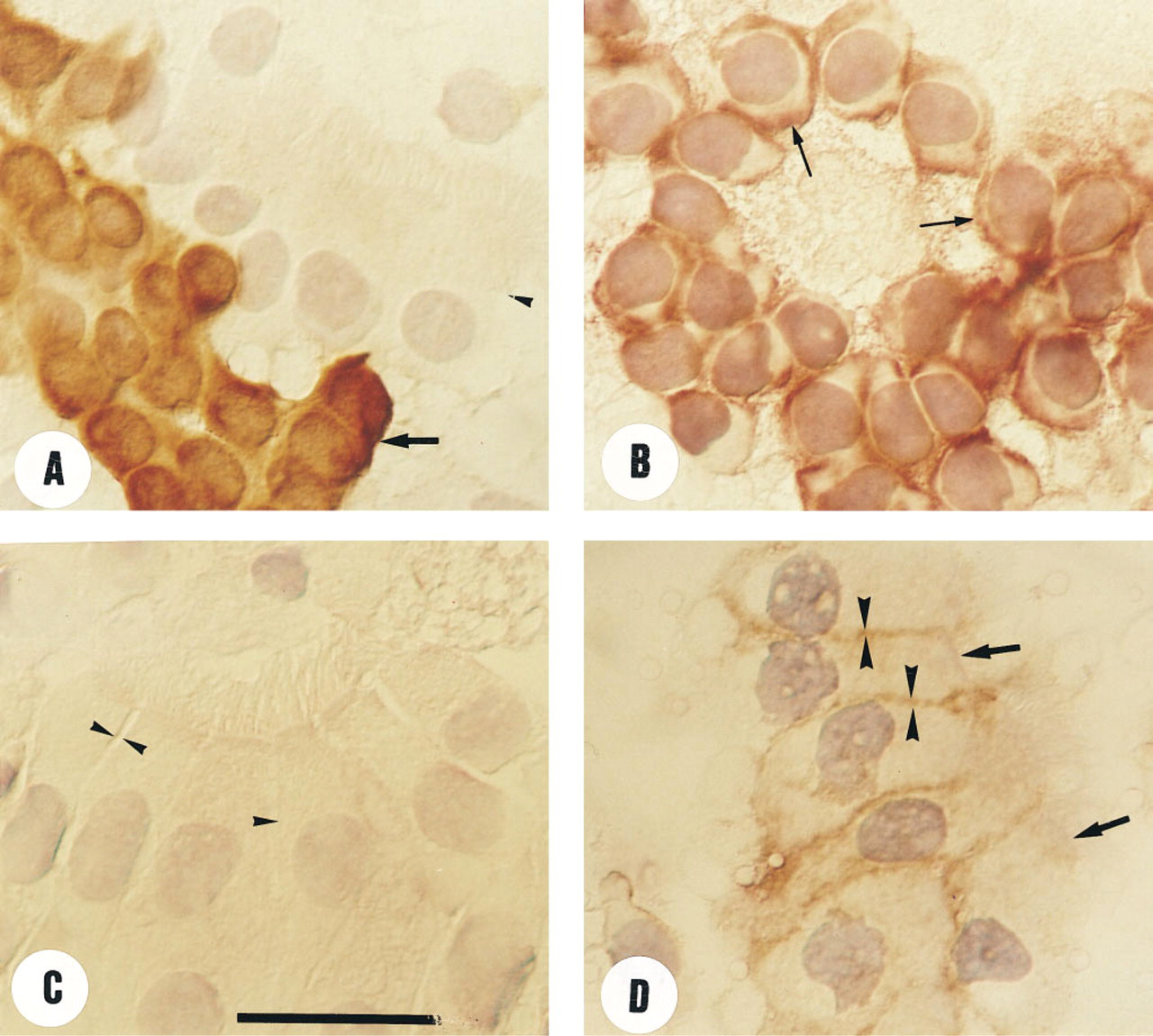
Immunocytochemical analysis of epithelial bronchial cells. (
This different pattern of distribution appears to be correlated with the existence of cell polarity. In basal cells with no tight junctions limiting apical and basolateral membrane domains, MRP was distributed over the entire circumference of the plasma membrane. In contrast, in polarized ciliated human bronchial cells MRP was restricted to the basolateral membrane. This pattern of expression has been previously reported in the polarized pig kidney epithelial cell line LLC-PK1 (Evers et al. 1996) but not in vivo, particularly in human epithelial cells. Specific signals for sorting of ABC transporters to either the apical or basolateral plasma membrane are still unknown.
The different distribution of MDR1 Pgp, MRP, and CFTR in bronchial epithelial cells suggests specific functions for each one. MRP has been characterized as an ATP-dependent pump for endogenous and exogenous glutathione, glucuronate and sulfate conjugates (Jedlitschky et al. 1994,1996; Zaman et al. 1995). In LLC-PK1, basolateral localization of MRP was in accordance with an increased transport of glutathione-S conjugates and daunorubicin to the basal side; in contrast, MDR1-transfected cells demonstrated a high level of daunorubicin transport towards the apical side (Evers et al. 1996). It has recently been demonstrated that MRP is an ATP-dependent export pump for the glutathione conjugate leukotriene C4 (LTC4) and structurally related conjugates (Jedlitschky et al. 1996). Because bronchial cells are known to produce different types of LTC, particularly LTC4 (Holtzman 1991), one of the functions of MRP in bronchial cells could be the transport of leukotriene-glutathione conjugates. In conclusion, MRP gene expression in human bronchial epithelial cells from smokers and from ex- and non-smokers and the patterns of protein distribution suggest specific functions as varied as epithelial cell defense against xenobiotics or efflux of endogenously synthesized products such as LTC4.
Footnotes
Acknowledgements
Supported by a grant from the Association Française de Lutte contre la Mucoviscidose 1994, France.
We thank M. Antoine, R. Lacave, S. Ricci, J. Rochemaure, and T. Urban for their constant support, V. Gerber for editorial assistance, and F. Stephan for statistics.
