Abstract
The pars tuberalis (PT) of the pituitary represents an important target site for the time-pacing pineal hormone melatonin because it expresses a large number of mt1 receptors. Functional studies suggest that the PT mediates the seasonal effects of melatonin on prolactin (PRL) secretion. The aim of this study was the characterization of the pheno-type of melatonin-responsive cells. Furthermore, we determined whether RORβ, a retinoid orphan receptor present in the PT, was co-expressed in the same cells. We combined nonradioactive in situ hybridization (ISH) with hapten-labeled riboprobes for detection of the receptors and immunocytochemistry (ICC) for detection of αGSU (α-glycoprotein subunit), βTSH, βFSH, βLH, GH, PRL, and ACTH. Expression of mt1 mRNA was found in small round cells, co-localized with αGSU and βTSH. However, not all βTSH-containing cells expressed mt1 mRNA. The distribution of mt1- and RORβ-positive cells appeared to overlap, although more cells were labeled for RORβ than for mt1. Gonadotrophs, as well as other pars distalis cell types, were never labeled for mt1 melatonin receptor. Therefore, this study identifies the “specific” cells of the PT as the mt1 melatonin receptor-expressing cells.
Keywords
T
High-affinity receptors for melatonin have been described using 2-iodo-melatonin as a ligand (Vakkuri et al. 1984). Two high-affinity receptors for melatonin have thus far been cloned in rodents: the mt1 (Mel1a) and the MT2 (Mel1b) receptor subtypes (Reppert et al. 1994,1995; Roca et al. 1996) which belong to the seven-transmembrane domain, G-protein-coupled receptor family (Dubocovich et al. 1999). However, the mt1 receptor appears to be the highly dominant subtype in the PT because targeted disruption of its gene in mice leads to undetectable levels of 2-iodomelatonin binding (Liu et al. 1997), whereas cells transfected with the MT2 receptor gene bind this ligand (Dubocovich et al. 1997). Furthermore, the MT2 receptor subtype is naturally nonfunctional in the golden hamster, which is a highly photoperiodic species (Weaver et al. 1996). Therefore, the mt1 receptor subtype appears sufficient to drive the melatonin-induced photoperiodic response. Although many studies have been performed on the circadian and seasonal transcriptional regulation of mt1 receptors in the PT (Gauer et al. 1993; Guerrero et al. 2000; Schuster et al. 2001), the exact cell type expressing this receptor has not yet been identified. Therefore, one aim of this study was the identification of this cell type in the rat by combining the nonradioactive in situ hybridization (ISH) for the mt1 mRNA with immunocytochemistry (ICC) for the different pituitary hormones.
Retinoid orphan receptors (RORα, -β, and -γ) are transcription factors that belong to the nuclear receptor superfamily and act as monomers at a single halfsite (Carlberg and Wiesenberg 1995; Mangelsdorf and Evans 1995). RORα is expressed mostly in peripheral tissues and in the cerebellum (Nakagawa et al. 1997; Dussault et al. 1998), while RORγ is present in skeletal muscle (Hirose et al. 1994). Interestingly, RORβ is expressed only in the central nervous system and in the PT of the pituitary gland. Its expression is particularly strong in components of the mammalian timing system such as the retina, the pineal gland, the SCN, and the PT (Schaeren-Wiemers et al. 1997; Park et al. 1997; Andre et al. 1998), where it displays daily variations (Baler et al 1996; Schaeren-Wiemers et al. 1997; personal observation). Melatonin was initially suggested as the endogenous ligand for these receptors (Becker-Andre et al. 1994; Wiesenberg et al. 1995), but the evidence supporting this hypothesis has since been questioned (Greiner et al. 1996; Hazlerigg et al. 1996a; Schaeren-Wiemers et al. 1997; Bogan et al. 1998). Thus far, the cell type expressing this receptor in the PT has not been identified. Therefore, in addition to the characterization of the cells expressing the mt1 mRNA, this study aimed to investigate whether mt1 and RORβ mRNAs could be co-expressed in the same cells in both the PT and PD.
Materials and Methods
Animals
Adult male and female Wistar rats were housed in a 12:12 light:dark cycle with food and water ad libitum. The animals were sacrificed during the light phase by transcardial perfusion under deep isoflurane anesthesia. Heparin (500 IU, Héparine Choay; Sanofi, Gentilly, France) was injected into the heart just before a PBS wash containing 0.02% diethylpyrocarbonate (DEPC; Sigma, St Quentin Fallavier, France), followed by the fixative (4% paraformaldehyde, 75 mM lysine, 10 mM sodium metaperiodate in 100 mM phosphate buffer, pH 7.4).
The brains were immediately dissected with the pituitary gland attached, blocked into 1-cm-thick slices, and postfixed in the fixative for 2 hr at 4C. They were then rinsed in 50% ethanol and embedded in polyethylene glycol as described by Klosen et al. (1993). Four to 12-μm-thick coronal and sagittal serial sections were cut and were either collected on silane-coated slides or processed as floating sections.
All procedures used in animal experimentation complied with the French National law implementing the European Communities Council Directive 86/609/EEC. All efforts were made to minimize the number of animals used.
In Situ Hybridization
Hapten-labeled (digoxigenin, fluorescein, or biotin) riboprobes with alkaline phosphatase detection were used. Three independent mt1 cDNAs were used: a partial 468-bp rat mt1 cDNA (Reppert et al. 1994), a 1021–bp rat mt1 cDNA (Poirel et al. 2001), and a full-length mouse mt1 cDNA (Roca et al. 1996). RORβ was detected using riboprobes transcribed from a 512-bp partial rat RORβ cDNA (Becker-Andre et al. 1993). For single ISH, the probes were labeled with digoxigenin. For double ISH, melatonin receptor probes were labeled with digoxigenin and RORβ probes were labeled with either fluorescein or biotin. Fluorescein- or biotin-labeled RORβ probes were first validated in single ISH for both sense and antisense probes.
Sense and antisense riboprobes were transcribed in the presence of labeled nucleotides according to standard procedures. The length of the probes was confirmed by formaldehyde-MOPS agarose gel electrophoresis and Northern blotting. Non-hydrolyzed full-length probes were used to ensure maximal specificity.
Polyethylene glycol sections were postfixed for 10 min at room temperature (RT) in 4% phosphate-buffered formaldehyde, rinsed in PBS, and digested for 30 min at 37C with 1–8 μg/ml proteinase K (Roche; Meylan, France) in PBS. Proteinase K digestion was stopped with 2% phosphate-buffered formaldehyde for 5 min on ice. After three rinses in PBS for 10 min each, the sections were acetylated twice for 10 min in 100 mM triethanolamine, 0.25% acetic anhydride. After two 10-min rinses in PBS, the sections were equilibrated in 5 X SSC, 0.05% Tween-20 before hybridization.
Hybridization was performed for 40 hr at 54C with 200 ng/ml labeled sense or antisense probes in 50% formamide, 5 X SSC, 5 X Denhardt's solution, and 500 μg/ml salmon sperm DNA. After hybridization, the probe was washed off in 5 X SSC at RT for 10 min. Stringency rinses were performed six times for 10 min in 0.1 X SSC at 72C. In some experiments, the sections were also treated with RNase (20 μg/ml in 400 mM NaCl, 10 mM Tris-HCl, pH 7.5, 2.5 mM EDTA for 30 min at 37C) either before or after the stringency rinses.
Hapten-labeled bound probes were detected with alkaline phosphatase-labeled sheep anti-digoxigenin antibodies, sheep anti-fluorescein antibodies, or streptavidin (Roche). Alkaline phosphatase activity was detected with bromo-chloro-indolyl phosphate (BCIP) and nitroblue tetrazolium (NBT) in the presence of 5% polyvinyl alcohol (70,000–100,000 MW; Sigma) (De Block and Debrouwer 1993).
For double ISH experiments, both probes were hybridized simultaneously and detected sequentially using BCIP/NBT as the first chromogen. After this first detection, the first anti-hapten antibodies were eluted by incubating twice for 15 min in 100 mM glycine-HCl buffer, pH 2.2, containing 0.1% Tween-20 (Philippe and Pollock 1998). On control sections, the elution was confirmed by the absence of detectable phosphatase activity.
The second probe was detected with peroxidaselabeled streptavidin (Roche), amplified with biotinylated tyramide (TSA; NEN, Boston, MA), and visualized with alkaline phosphatase-labeled streptavidin and Fast Red/naphthol-AS-MX phosphate (Sigma) as chromogen in the presence of 4% polyvinyl alcohol. The Fast Red/naphthol-AS-MX phosphate detection procedure is 10- to 100-fold less sensitive than the NBT/BCIP procedure, thus requiring a previous tyramide amplification. Without this amplification no signal could be obtained with Fast Red for either melatonin receptor probes or RORβ probes.
Immunocytochemistry
ICC was performed using antibodies (obtained from the NIDDK; Dr. F. Parlow) directed against the following pituitary hormones: rabbit anti-rat α-glyco-protein-subunit (αGSU), rabbit anti-rat β-thyroid-stimulating hormone (βTSH), rabbit anti-rat β-follicle-stimulating hormone (βFSH), rabbit anti-rat β-luteinizing hormone (βLH), guinea pig anti-rat growth hormone (GH), rabbit anti-rat prolactin (PRL), and rabbit anti-human adrenocorticotropin (ACTH). For further characterization, we also used a rabbit antibody to the transcription factor Pit-1 (Santa Cruz Technology; Santa Cruz, CA).
The primary antibodies were incubated overnight and detected using biotinylated secondary antibodies and streptavidin-peroxidase conjuate (Roche). Peroxidase activity was detected using amino-ethyl-carbazole (AEC; Sigma) as chromogen.
For combined ISH and ICC, the slides were first hybridized. After visualization of phosphatase activity with NBT/BCIP, the slides were rinsed and selected for double labeling. The slides were rinsed in 95% ethanol for 5 min to elute some background label and to shift the color of the in situ label to blue. The slides were then immunolabeled using the standard protocol described above, with AEC as the chromogen. The absence of interactions between the ISH and the ICC was verified by omission of the primary antibody in the ICC detection. Further tests included ICC on sections that were mock-hybridized to check for antigen inactivation or decloaking by the hybridization procedure.
For the combined ISH and double ICC, the first immunolabel was performed using nickel-diaminobenzidine as chromogen. The antibodies of the first immunolabel were then eluted with 100 mM glycine buffer (pH 2.2) containing 50 mM β-mercaptoethanol to reduce the disulfide links between the light and the heavy chains of the immunoglobulins. After PBS rinses, the second antibody was incubated overnight and detected using the standard protocol described above, with AEC as the third chromogen. The elution of the first sequence of antibodies was verified on control sections by omission of the primary antibody in the second ICC detection sequence.
Results
mt1 Melatonin Receptor Expression
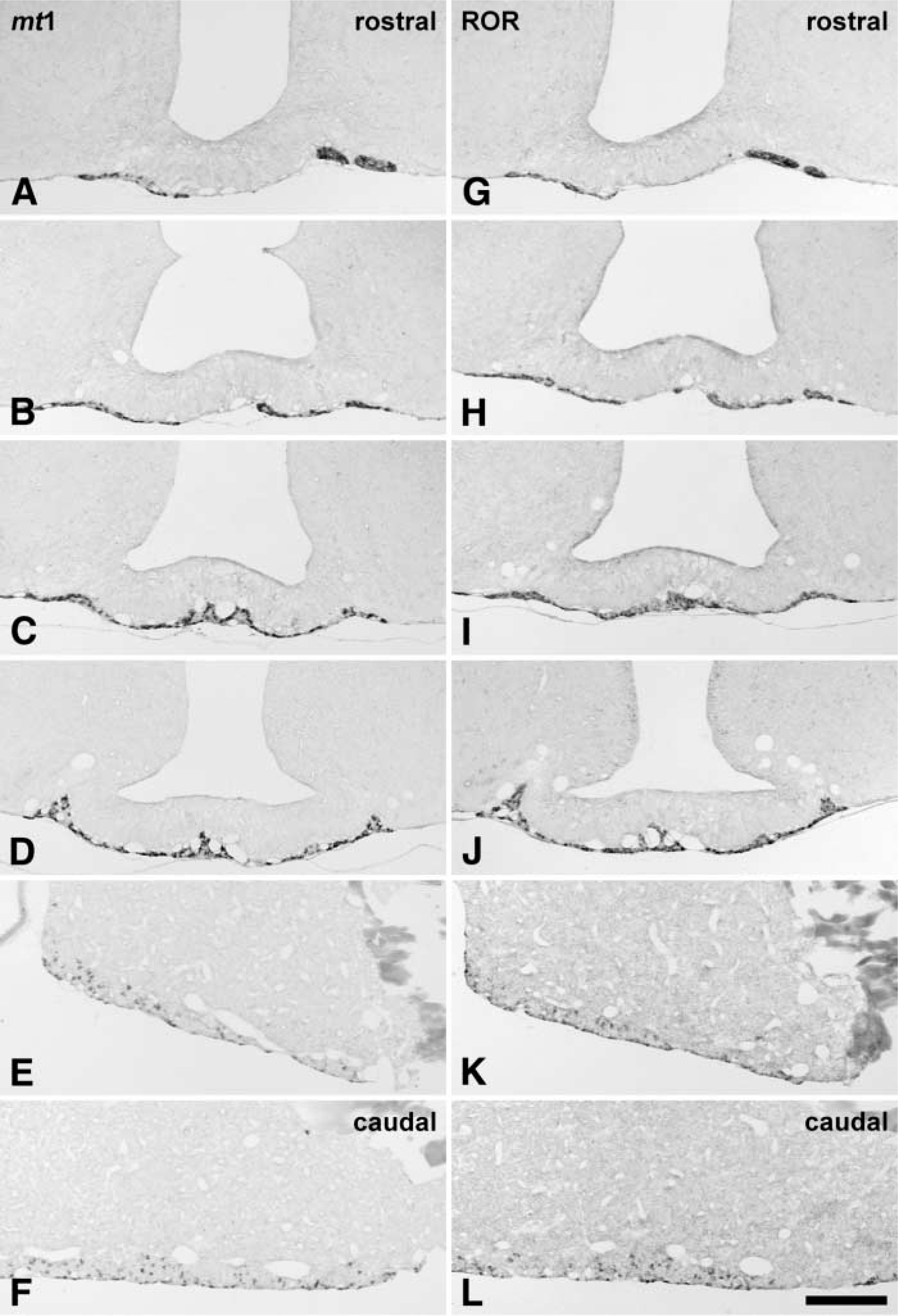
We detected mt1 melatonin receptor expression in the PT of the adenohypophysis, in accordance with previous studies using radioactive ISH (Figures 1A-1D and 2A). All three mt1 probes provided the same labeling pattern, although optimal results were obtained with the 1021-bp rat probe and the full-length mouse probe. The specificity of the ISH was confirmed by the absence of label with sense probes (Figure 2C). Furthermore, the in situ label was obtained at high stringencies (0.1 X SSC, 72C) and resisted RNase digestion.
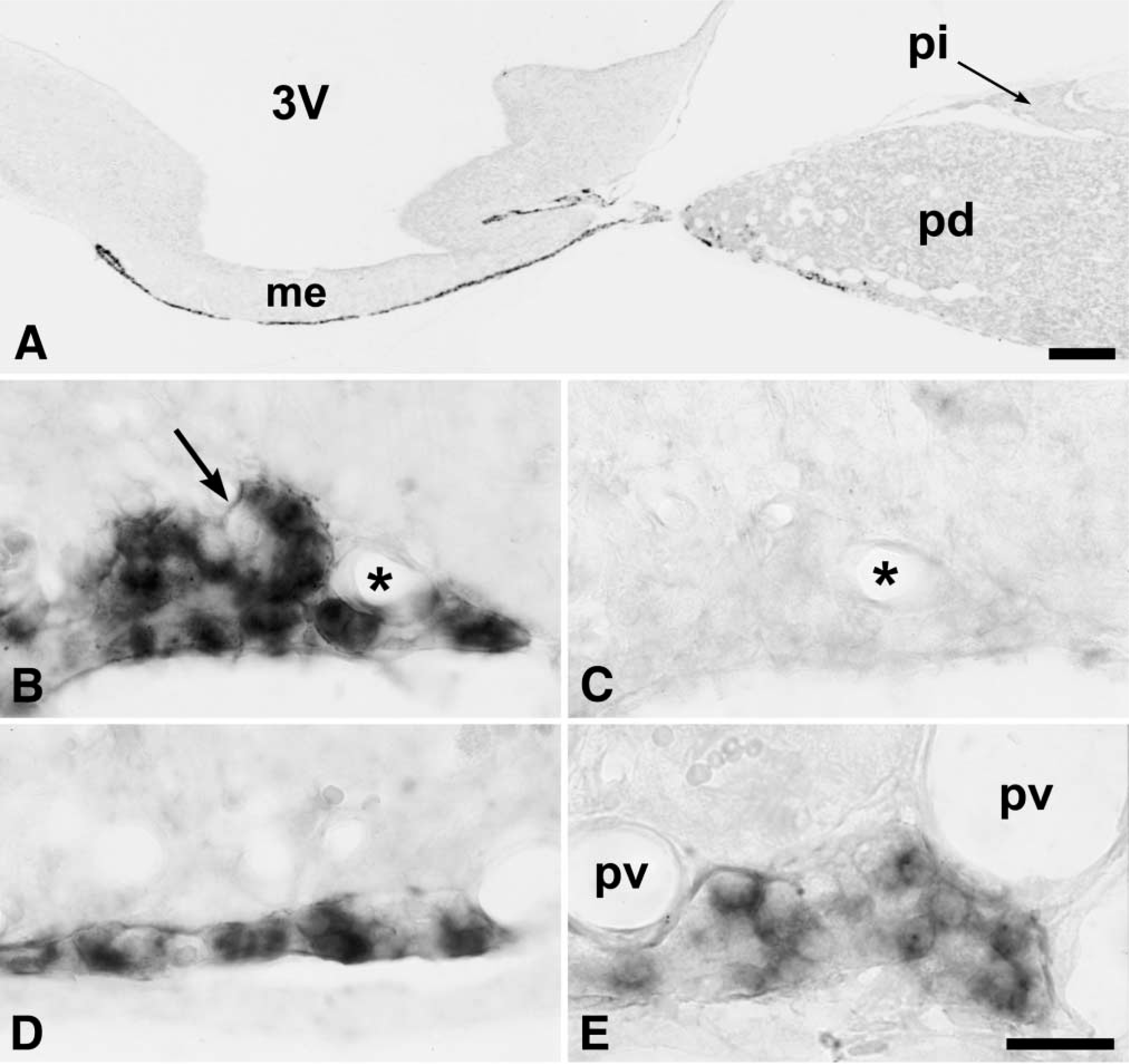
The labeled cells were small and ovoid, with the nucleus usually located in one end of the cell (Figures 2B, 2D, and 2E). Labeling was restricted to the cytoplasm, although some cells also displayed labeling of a small point-like structure in the nucleus, probably the nucleolus (Figure 2E). Not all cells of the PT were labeled. Although the proportion of labeled cells was very high, especially in the rostral part of the PT, a few nonlabeled ovoid cells could be detected (Figure 2B). We did not observe labeling of small stellate cells, suggesting that only endocrine cells contained the mt1 melatonin receptor. No mt1 melatonin receptor hybridization could be detected in the walls of the blood vessels (Figures 2B and 2E).
Rostrocaudal series of sections through the pars tuberalis (
The mt1 melatonin receptor was also expressed in the rostral part of the PD along the ventral surface (Figures 1E and 1F). These scattered cells appeared identical to those of the PT and were sometimes grouped into small clusters. On sagittal sections, mt1 melatonin receptor-expressing cells could be detected from the most rostral part of the PT to the ventral part of the PD (Figure 2A). However, the density of labeled cells appeared to decrease from the rostral to the caudal part, especially in the PD (Figure 2E). No difference was detected between animals sacrificed early or late in the light phase.
(
Co-expression with Pituitary Hormones
All pituitary hormone antibodies used in the study provided specific labeling of their corresponding cell populations in the PD. Only the anti-GH antibody appeared to crossreact slightly with pro-opiomelanocortin (POMC), as could be detected by a slight labeling of the pars intermedia, POMC-containing neurons and nerve fibers in the hypothalmus, and some slightly labeled corticotrophs in the PD (results not shown). No crossreactivity was observed for any of the other antibodies. Immunolabeling of mock-hybridized sections revealed no antigen inactivation nor decloaking for the antibodies tested. Only a reduction in the background label could be detected. Furthermore, no interference between the hybridization and the immunolabeling procedures could be detected.
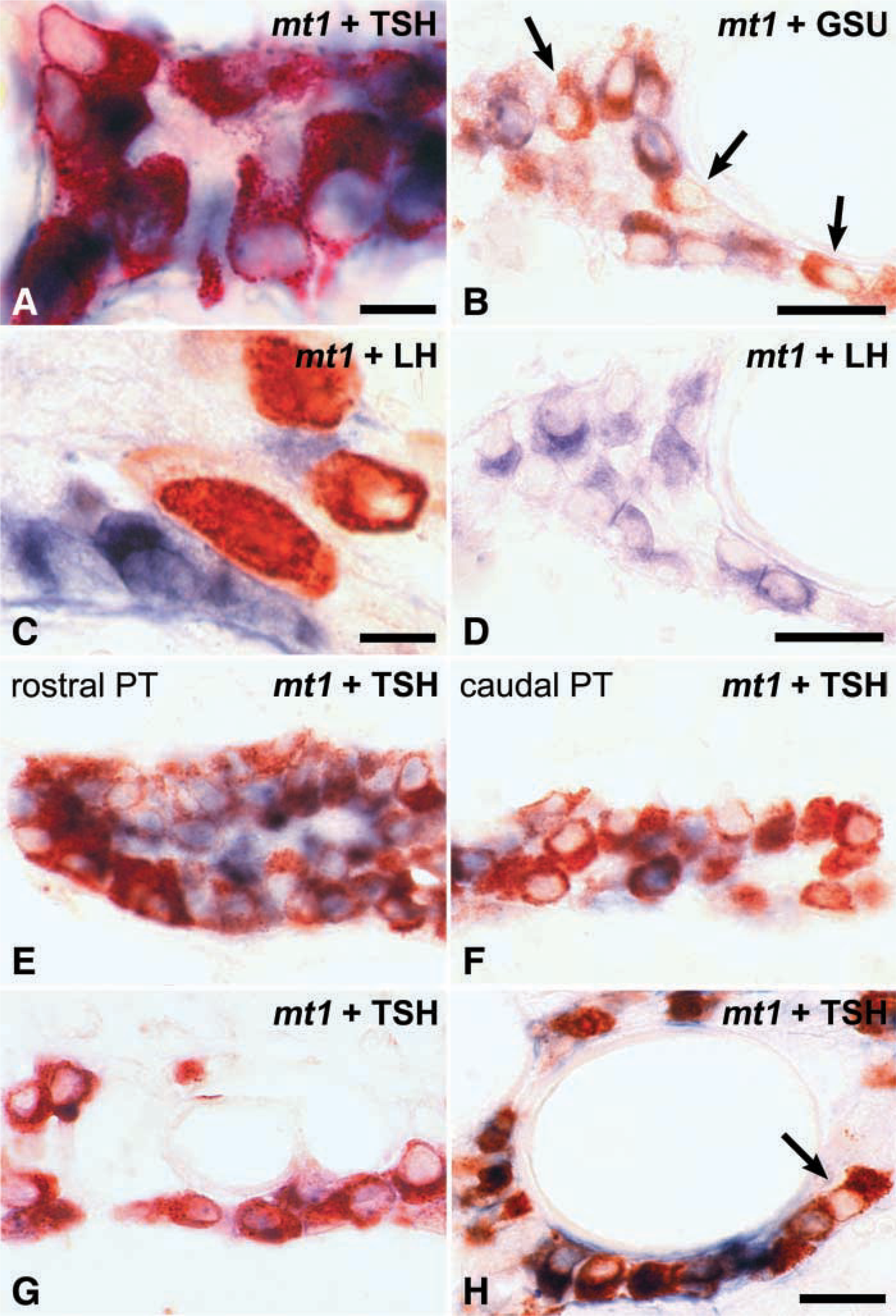
The pituitary hormones detected in the PT were αGSU, βTSH, βFSH, and βLH, in accordance with previous studies. No labeled cells were observed with antibodies to GH, PRL, or POMC. In αGSU-labeled PT sections, gonadotrophs were usually easily distinguished from TSH-expressing cells by their larger size. A large number of PT cells were immunolabeled and almost all of them were βTSH/αGSU-positive cells, with the FSH/LH-positive gonadotrophs representing only a very small fraction of the PT cells. Furthermore, these gonadotrophs were irregularly distributed throughout the PT, especially from one rat to another.
Co-expression of mt1 melatonin receptor was observed only with βTSH and αGSU (Figures 3A and 3B). The mt1 mRNA was never detected in βFSH-, or βLH-positive cells (Figures 3C and 3D). Not all βTSH-positive cells in the PT showed co-expression with mt1 melatonin receptor (Figures 3B and 3E-3G). The proportion of double-labeled cells decreased towards the caudal end of the PT, while in the rostral part of the PT almost all cells appeared double labeled. In the pituitary stalk, both βTSH single-labeled and mt1/βTSH double-labeled cells were often located around portal vessels (Figures 3G and 3H).
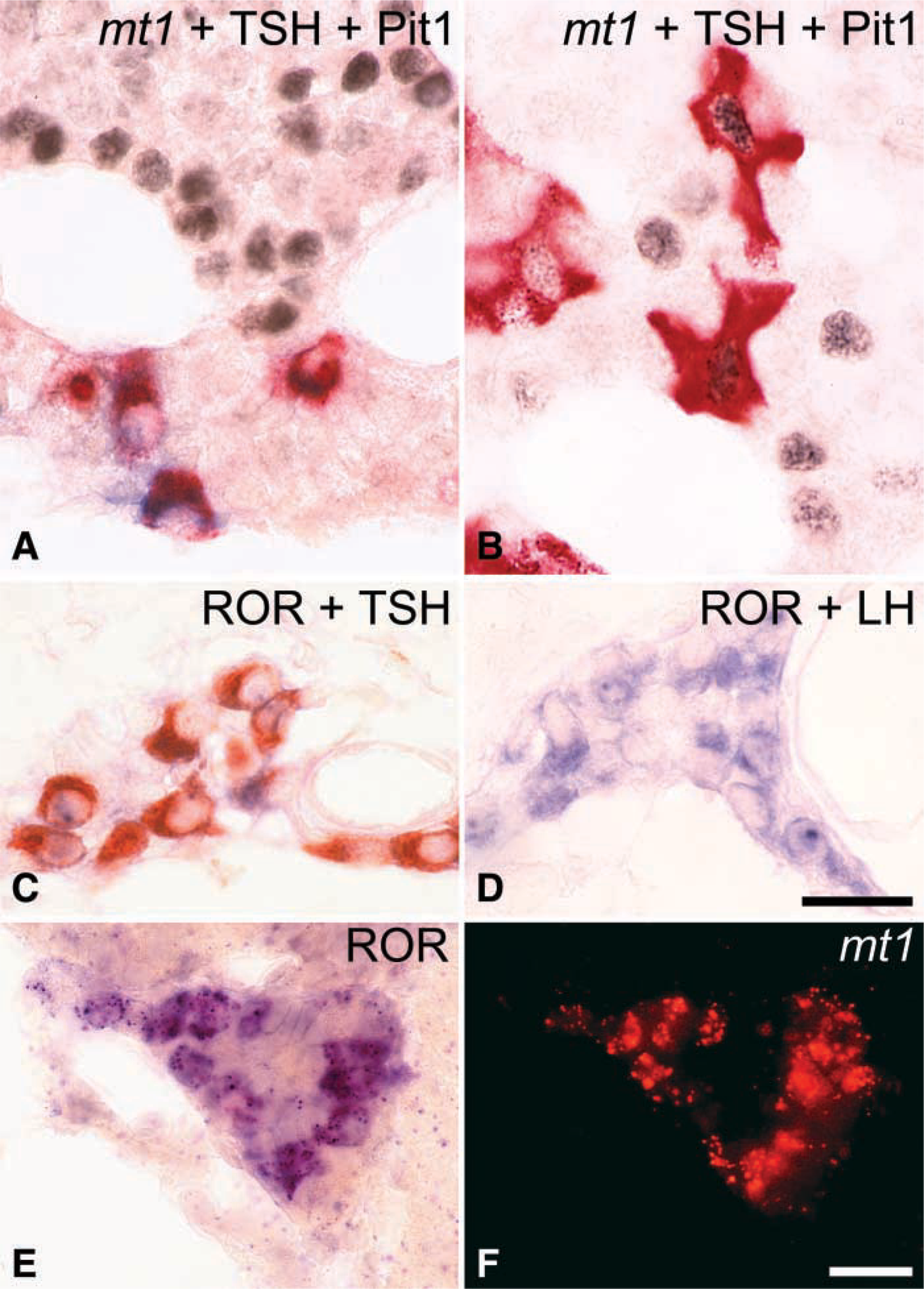
In the PD, the same pattern of co-expression was observed. The mt1 melatonin receptor was present only in a subpopulation of αGSU- and βTSH-positive cells (Figures 4A and 4B). The morphology of the βTSH-positive cells in the ventral part and the rest of the PD differed. Whereas in the ventral PD these cells were small ovoid cells identical to those of the PT, in the rest of the PD the βTSH-positive cells were large, with an angular shape. Furthermore, the small ovoid mt1-positive cells did not express the transcription factor Pit-1, which was present in the large angular thyrotrophs. A third population consisted of some small ovoid TSH-positive cells, which were devoid of mt1 melatonin receptor labeling and which were located among the mt1/βTSH double-labeled cells in the ventral PD. Pit-1 expression was completely absent from this ventral area of the PD (Figure 4A).
RORβ Expression in the PT and PD
A robust and specific signal for RORβ was obtained in the cortex, the suprachiasmatic nucleus, and the thalamus, as previously described by Schaeren-Wiemers et al. (1997) and Park et al. (1997). As for the mt1 melatonin receptor, the specificity of the hybridization signal was confirmed by the absence of labeling by sense probes, the high stringency of the hybridization procedure, and the resistance to post-hybridization RNase digestion. Almost identical results were obtained with all three hapten labels, except that signal-to-noise ratios were optimal for digoxigenin-labeled probes.
In the pituitary, the RORβ expression pattern was identical to that reported by previous studies using radioactive ISH (Park et al. 1997; Schaeren-Wiemers et al. 1997; Andre et al. 1998) (Figures 1G-1J). RORβ expression was strongest in the PT. The labeled cells were small ovoid cells, with some nonlabeled cells scattered among them (Figures 4C and 4D). The density of the RORβ cells was highest in the rostral PT and decreased slightly towards the caudal part.
In the PD, some very slight RORβ expression was detectable throughout the gland. Furthermore, slight labeling was seen in the pars intermedia. However, in the ventral part of the PD, scattered small ovoid cells showed strong hybridization with the RORβ probe. Therefore, the distribution of strong RORβ expression in the rat PT and PD was identical to that observed for the mt1 melatonin receptor.
The only pituitary hormones present in RORβ-positive cells were βTSH and αGSU (Figure 4C). As with the mt1 receptor, βLH and βFSH were absent from the RORβ-expressing cells (Figure 4D).
Co-expression of mt1 melatonin receptor and RORβ in the same cells of the PT and the PD was confirmed by hybridization of adjacent 4-μm sections (not shown) and by simultaneous double ISH (Figures 4D and 4F). We were unable to ascertain whether all cells co-expressed both the mt1 melatonin receptor and the RORβ receptor. The tyramide-amplified Fast Red detection procedure remained less sensitive than the direct NBT/BCIP, thus raising the possibility that the Fast Red procedure was unable to detect some cells that would be positive by the NBT/BCIP procedure. Nonetheless, the majority of the cells displayed labeling for both receptors.
Discussion
We have observed mt1 melatonin receptor expression in a subpopulation of endocrine cells in the PT of the rat. The distribution and morphology of these cells, as well as their content of αGSU and βTSH, identify them as the “specific” cells of the PT (Gross 1983; Stoeckel and Porte 1984; Rudolf et al. 1993; Sakai et al. 1999). However not all the “specific” cells of the PT express the receptor. No mt1 receptor expression was observed in gonadotrophs or follicular cells of the PT.
In the rat, although “specific” cells from the PT express the same hormonal subunits as the classical PD thyrotrophs, both cell types present many morphological and functional differences. Large and angular in the PD, they are small and ovoid in the PT (Figures 4A and 4B; Rudolf et al. 1993; Sakai et al. 1992,1999). Moreover, the transcription factor Pit-1 implicated in βTSH expression in the PD thyrotrophs is absent in the specific cells of the PT (Figures 4A and 4B; Lin et al. 1994; Bockmann et al. 1997; Sakai et al. 1999). Bockmann et al. (1997) have established that PT cells possess neither TRH receptors nor thyroid hormone receptors. Therefore, TSH synthesis and release in these cells are not subject to the classical controls by TRH and T4. Furthermore, the mt1 melatonin receptor mRNA is highly expressed in a large number of PT “specific” cells located in the PT but also in the rostroventral part of the PD, known as the zonula tuberalis in the sheep (Skinner and Robinson 1995), whereas it is never present in the classical thyrotrophs from the PD. Therefore, although they express the same hormones, TSH-immunoreactive cells in the PT and in the PD are not physiologically regulated in the same way. Wittkowski et al. (1988) have shown that the TSH content of PT-specific cells is strongly reduced in Djungarian hamsters subjected to short days compared to long days. Because this effect is prevented by pinealectomy and can be mimicked by injections of melatonin, it appears that melatonin is a major regulator of TSH expression in the PT (Böckers et al. 1995; Bockmann et al. 1996). Our results suggest that these effects could be mediated through the mt1 receptor.
Co-localization of mt1 melatonin receptor (blue label) with pituitary hormones (red label). In the PT, mt1 melatonin receptor is co-localized with βTSH (
(
The best-established effect of melatonin in mammalian PT cells is inhibition of forskolin-induced cAMP synthesis (Morgan et al. 1995; Vanecek and Watanabe 1998). However, this inhibitory effect requires a previous stimulatory input because melatonin alone has no effect on basal cAMP levels. Recent work by von Gall et al. (2002) suggests that this stimulatory input might be adenosine through the adenosine A2b receptor. The nocturnal downregulation of cAMP levels by melatonin sensitizes the A2b receptor.
Melatonin also downregulates the phosphorylation of CREB in ovine PT cells (McNulty et al. 1994). The phosphorylation of CREB in ovine PT cells is regulated through both cAMP-dependent and cAMP-independent mechanisms (McNulty et al. 1996). The same authors have described in vitro a serum-derived factor that provides a cAMP-independent stimulation of CREB phosphorylation, which is sensitive to melatonin. The presence of CRE-like elements in the promoter of both the αGSU and βTSH genes (Kim et al. 1993; Hashimoto et al. 2000) suggests that melatonin may downregulate their expression by the reduction of phosphoCREB levels during the night. However, no data are available on the nycthemeral regulation of either αGSU or βTSH.
In the hamster PT, Messager et al. (1999,2000,2001) have shown that the photoperiod also regulates the amplitude of expression of Per1, an early-response gene also present in the SCN and part of the circadian clock molecular mechanism (Bae et al. 2001; Reppert and Weaver 2001; Zheng et al. 2001). Interestingly, E-Box binding motifs for bHLH transcription factors such as CLOCK and BMAL1, two other clock-related genes, have been found upstream of the αGSU gene (Jackson et al. 1995; Wood et al. 1999). Because several clock-related genes are expressed in the PT of rodents (Messager et al. 1999; and personal observation), it could be hypothesized that the PT contains a slave molecular oscillator that could be reset and synchronized to the environment lighting conditions directly by melatonin action. In such a case, clock genes would constitute a relay for melatonin in mediating output effects, such as acting on thyrotropins.
Our study also demonstrates that mt1 and RORβ receptors are co-expressed in the same cells. Unpublished results obtained in our laboratory show that the daily variations in RORβ mRNA expression in the rat PT depend on the presence of endogenous melatonin. Furthermore, melatonin has been shown to regulate the transcriptional activity of RORα in MCF-7 breast cancer cells (Dai et al. 2001), which are known to express the mt1 melatonin receptor (Ram et al. 2000). Therefore, it is tempting to speculate that RORβ mRNA expression is dependent on melatonin through mt1 melatonin receptors. Melatonin might thus control the transcriptional activity of RORβ receptors, which might then control other downstream genes. However, thus far no response element for RORα or -β has been described or functionally evaluated in αGSU or βTSH genes.
Our results clearly establish the TSH-containing cells as the major cellular target of endocrine regulation by melatonin in the PT. However, the specific cells of the PT constitute only a small fraction of the thryotrophs of the adenohypophysis. Therefore, the photoperiodic downregulation by melatonin of βTSH expression, and possibly secretion, would affect only marginally the total pituitary output of βTSH. Furthermore, no clear effect of either αGSU or βTSH on the PD secretory activity has been demonstrated in the adult. Therefore, the identity of the endocrine-specific product of the PT, called tuberalin (Stoeckel et al. 1994; Morgan et al. 1996), regulated by melatonin receptors in the TSH-containing cells and responsible for the photoperiodic regulation of PRL secretion by the lactotrophs of the PD, remains to be identified.
Footnotes
Acknowledgements
Supported in part by the Institut de Recherche International Servier.
We wish to thank Dr F. Parlow and the National Hormone and Pituitary Program of the NIDDK for the gift of the anti-hormone antibodies, and Drs S. Reppert and I. Wiesenberg for the gift of the mouse mt1 and rat RORβ cDNAs, respectively. We thank Dr J.P. Loeffler for help with Pit-1 detection. The expert assistance of D. Bonn and A. Senser in animal husbandry is gratefully acknowledged.
