Abstract
The fetus must transport considerable and increasing amounts of calcium across the placental trophoblast epithelium to support growth and development and bone formation. Active calcium transport across epithelia has been shown to correlate with calbindin D9k or 28k content. This study examined the distribution of calbindin D9k (9CBP) protein and mRNA during pregnancy in the bovine placenta to determine its possible role in calcium transport in this system. The immunocytochemical results show 9CBP in an increasing percentage of interplacentomal uninucleate trophoblast cells until, at term, all show a level at least eight times that of any other placental cell. There is a similar, although smaller, rise in their 9CBP mRNA content. The mature interplacentomal binucleate cell (∼5% of the total) contains no 9CBP at any stage of pregnancy. In interplacentomal uterine epithelium, 9CBP protein and mRNA decrease to zero in late pregnancy but the glands maintain constant low levels throughout. In the placentome trophoblast, uninucleate cells show insignificant amounts but binucleate cells (15–20% of the total trophoblast cells) contain considerable levels of both 9CBP protein and mRNA, as do all the uninucleate uterine epithelial cells. The placentomal binucleate cells show peak values at mid-pregnancy; the placentomal uterine epithelium shows only small changes in levels in the second half of pregnancy. Increase in fetal calcium demand in the second half of pregnancy therefore correlates with a major increase in 9CBP only in the interplacentomal trophoblast, as we have also shown in the sheep and goat, indicating an important role for this region in active calcium transport by the ruminant placenta. The 9CBP is distributed uniformly in the cytosol and nucleoplasm, supporting a role in facilitated diffusion of calcium through the cell rather than a vesicular shuttle system.
I
We have also shown that in sheep these interplacentomal trophoblast areas are capable of active uptake (Jones et al. 1997) and transport (Abbas et al. 1993) of calcium. These results support the hypothesis that a 9CBP-mediated process is the basis for active calcium transport in the ruminant placenta and is restricted to the interplacentomal trophoblast because there is very little 9CBP in the sheep placentome. This pattern of trophoblast 9CBP distribution, is common to all of the other ruminants we have checked thus far (goat, Wooding et al. 1996a; and several deer and antelope genera, unpublished results). However, the cow, although showing a similar high interplacentomal 9CBP level, also demonstrated a unique placentomal pattern of high 9CBP content in the trophoblast binucleate cells and in the maternal uterine epithelium, which confirmed the localization by Reiswig et al. (1995). This study quantitated the distribution of 9CBP protein and mRNA throughout pregnancy in the cow to establish how far the cow fell outside the normal ruminant pattern and how far this compromised the hypothesis of a localized area of the ruminant placenta set aside for active calcium transport.
Materials and Methods
Tissue Preparation
Placentas of 23 Friesian cows [three nonpregnant, two 55, three 100, four 150, one 155, three 219, one 248, two 270, four 280 days post conception (dpc)] were used. Animals were sacrificed with a captive bolt pistol. The uterus was removed immediately and perfused first via the uterine and subsequently the fetal umbilical arteries with 3% (w/v) paraformaldehyde in 0.1 M cacodylate buffer, pH 7.2, plus 5% (w/v) sucrose, with or without 1% glutaraldehyde. Perfused dye was used to select the best dually perfused areas of placentome and interplacentomal samples as detailed previously (Wooding et al. 1996a). Frozen samples of unfixed placentomal and interplacentomal regions were prepared by plunging tissue pieces into isopentane cooled to −60C in liquid nitrogen and were stored either in liquid nitrogen or in a freezer at −70C. Eight- to 12-μm sections of frozen tissue were fixed briefly (5 min) in 4% (w/v) paraformaldehyde and stored in 95% ethanol (v/v) at 4C until required (Sirinathsinghji et al. 1990). For structural studies, low-temperature embedding in Lowicryl K4M at −20C was carried out according to the schedule in Wooding et al. (1996a). Semithin Lowicryl K4M (LK4M) sections were picked up on poly-L-lysinecoated coverslip squares and dried at 60C for 30 min. Ultrathin Lowicryl K4M sections were picked up on naked 100 (or 300) mesh or celloidin-covered 100-mesh or slot grids.
Immunocytochemistry
Protocols for immunocytochemical localization by silver intensification for light microscopy and 15-nM gold colloid for electron microscopy were as detailed in Wooding (1980) and Wooding et al. (1996a). The frequency of gold particles per square micrometer was determined by counting in the electron microscope using a superimposed wire circle (Wooding et al. 1996a). Quantitation for light microscopic immunocytochemistry was carried out on a Leitz MPV-3 microscope (see below). No labeling was seen on controls, which included omission of the primary antibody and absorption against pure 9CBP or bPL.
Antibodies
The antibodies have been characterized previously and were raised against calbindin D9k (Bruns et al. 1978,1988; Reiswig et al. 1995) and bovine placental lactogen (Murthy et al. 1982).
In Situ Hybridization
Oligonucleotide probes (45-mer) were synthesized on a Bio-search 8600 DNA synthesizer. The probe sequences derived from the bovine 9CBP cDNA sequence 5′CAGCTCCTCC-TTGGACAGTTGGTTTGGATCACCTTCTTTGGCTGC-3′, complementary to nucleotides 67–111 in EMBL Accession Number M18344 (Kumar et al. 1989). Sense 45-base oligonucleotides, exactly complementary to the anti-sense probe, were used as controls. Probes were 3′-end-labeled with [α35S]-dATP and 3 × 105 dpm of the probe used per slide (usually three to six sections per slide). Hybridization was performed at 42C as previously reported in detail (Sirinathsinghji et al. 1990; Morgan et al. 1997). In situ hybridization specificity was checked by omitting the anti-sense probe, by using the sense probe, by RNase digestion, and by adding excess unlabeled anti-sense probe. Quantitation was carried out on a Leitz MPV-3 microscope photometer using eosin-stained sections, as detailed in Wooding et al. (1996a).
Double-labeling Procedure
The immunoperoxidase staining method of Brahic and Ozden (1992) was used to detect bPL in binucleate cells, followed by in situ hybridization as detailed above on the same section to demonstrate 9CBP mRNA in the same binucleate cell. Appropriate controls were used for both procedures.
Northern Blot Analysis
Total cellular RNA was extracted by the method of Chomczynsky and Sacchi (1987) from 250 dpc placentomal and interplacentomal tissues with manually separated fetal and maternal components and was hybridized to the radioactive 9CBP probe as detailed in Wooding et al. (1996a).
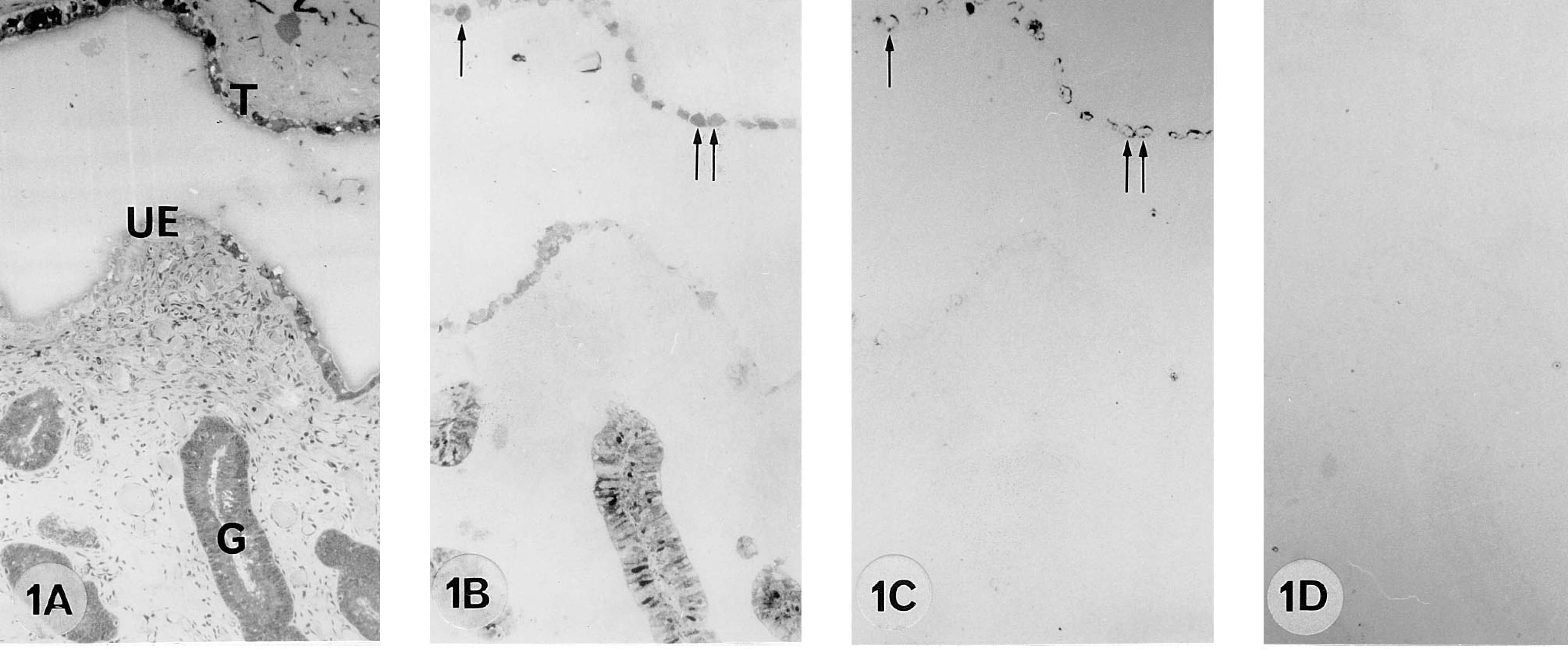
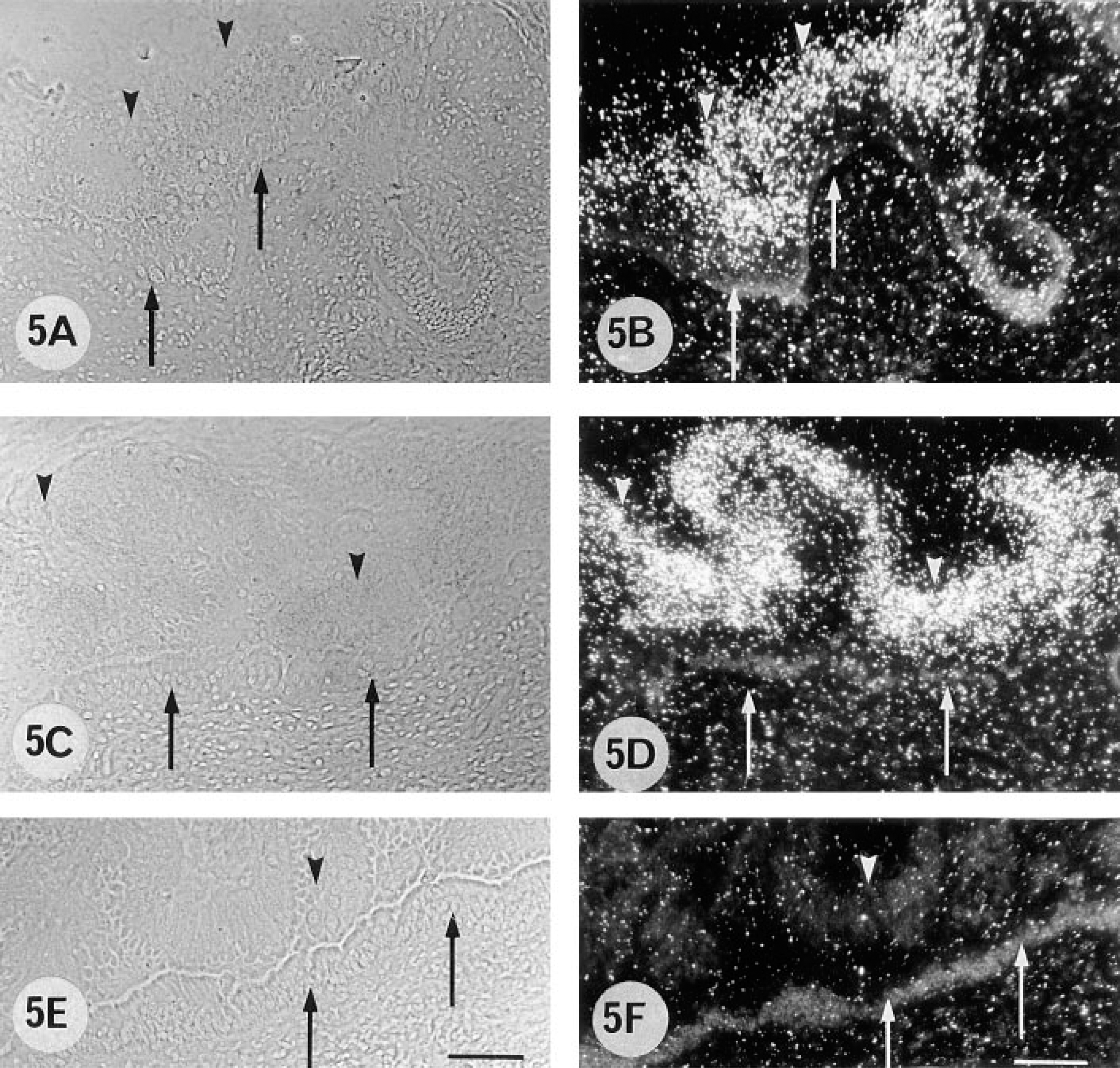
A 55 dpc pregnant cow. 9CBP localization (B) in the trophoblast binucleate cells (arrows), in uterine epithelium and in glands. Note bPL localization in binucleate cells (C, arrows).
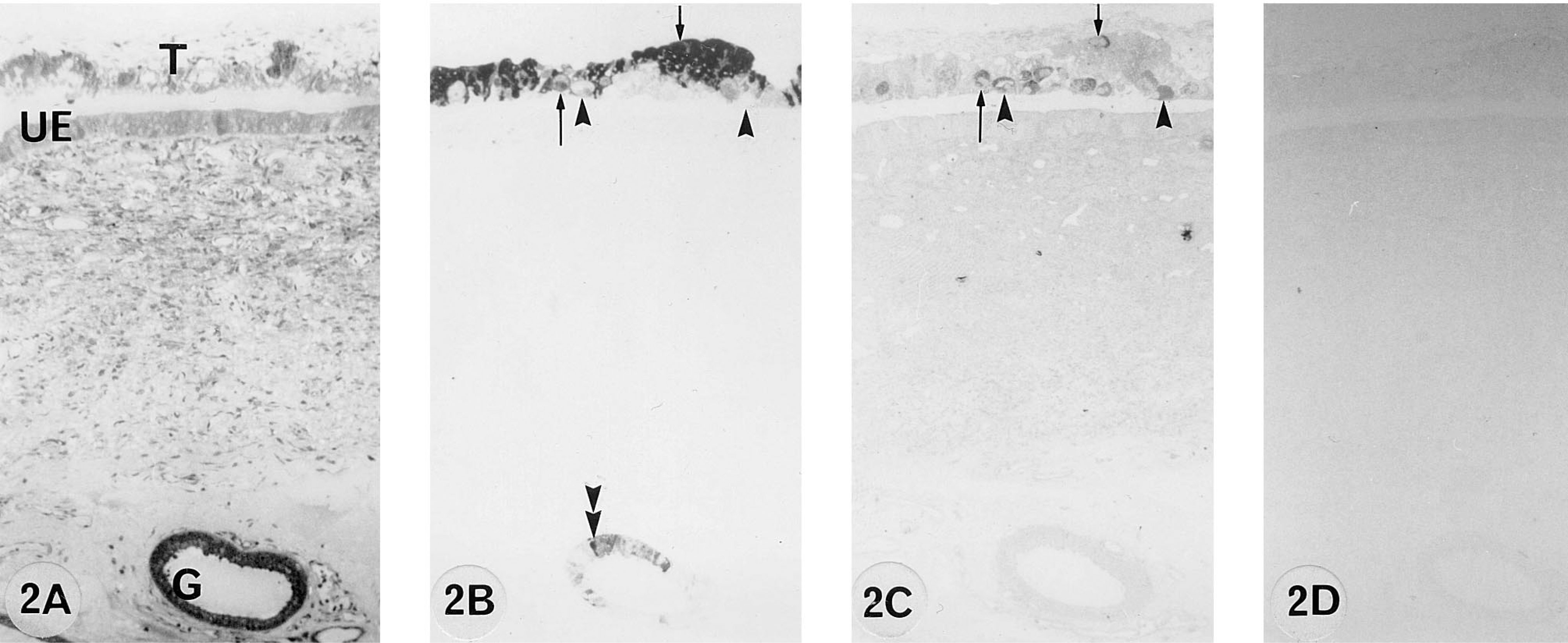
A 155 dpc pregnant cow. 9CBP is localized (B) predominantly in the trophoblast uninucleate cells and some of the binucleate cells (arrows). Other binucleate cells (arrowheads) and uterine epithelium are 9CBP-immunonegative. 9CBP immunoreactivity is also localized within individual glandular cells (double arrowheads). All binucleate cells are identified by bPL content (C).
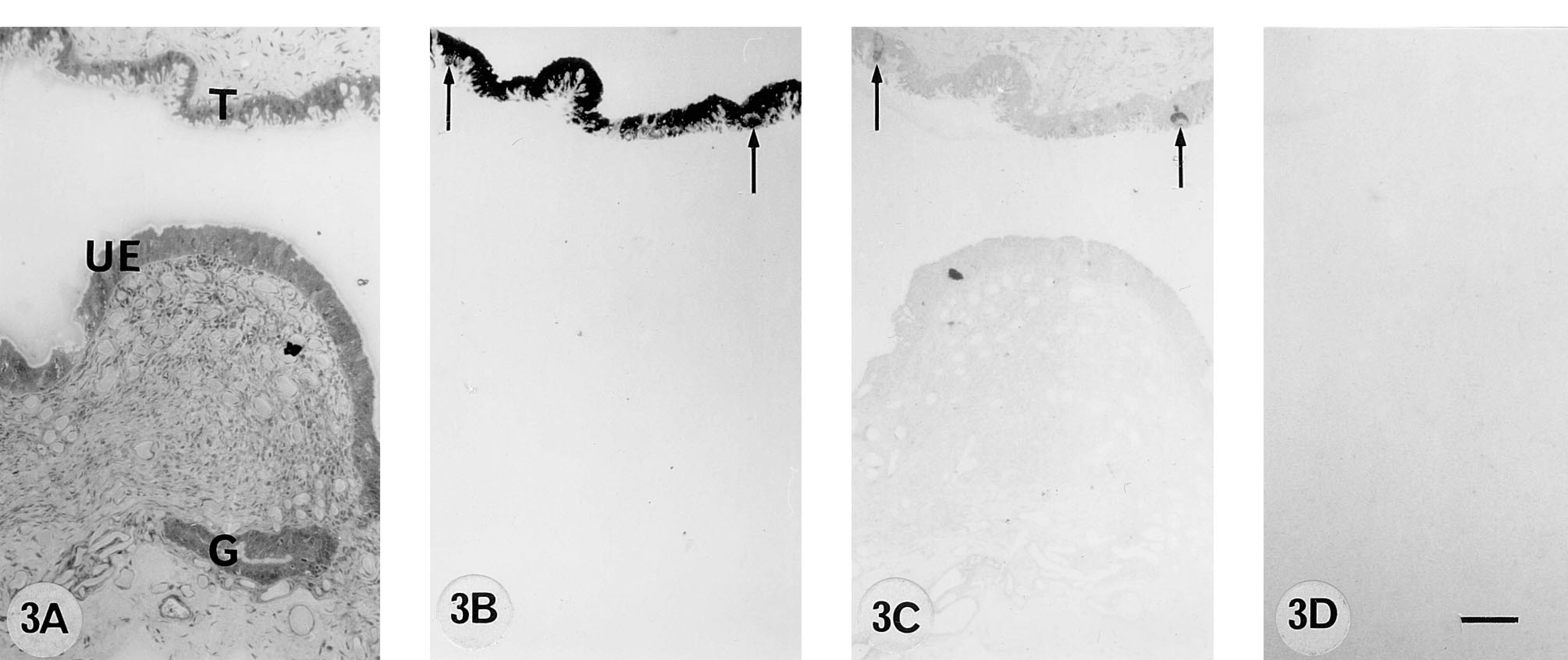
A 270 dpc pregnant cow. 9CBP localization (B) in the trophoblast uninucleate cells and binucleate cells (arrows). Uterine epithelium and glands on this section are 9CBP-immunonegative.
Results
Interplacentomal Immunocytochemistry
From 50 to 100 days post coitum there is a high level of 9CBP only in individual interplacentomal trophoblast cells, which can be identified as the characteristic ruminant binucleate cells on the basis of their structure and bovine placental lactogen content (Figure 1). Remarkably, the 9CBP is lost from these cells once they mature and start to migrate through the apical tight junctions of the trophoblast epithelium. This pattern of initial 9CBP expression, which is lost after the production of their characteristic granules, is found throughout pregnancy in these binucleate cells (Figure 4). Very few uninucleate trophoblast cells show label initially, only those over the gland mouths, but between 100 and 150 dpc the expression of 9CBP spreads rapidly to all uninucleate cells (Figure 2), and this high level is maintained to term (Figure 3), when it is about eight to 10 times higher than in any other region of the placenta at that stage.
The nonpregnant maternal gland epithelium has a high level of 9CBP expression in about 50% of the cells, and this is maintained to 55 dpc (Figure 1) but then, as the glands increase in size as the uterus grows to accommodate the expansion of the conceptus, the number of 9CBP-positive cells decreases. From 100 dpc to term the glands show a checkerboard pattern of 9CBP expression, with about 30% of the cells strongly positive and the remainder negative (Figures 6A and 6C).
In the nonpregnant columnar uterine luminal epithelium, up to 10% of the cells contain 9CBP in apparently random distribution. This decreases to very low levels by 150 dpc (Figure 2B) and is unchanged for the rest of pregnancy (Figure 3B).
Interplacentomal In Situ Hybridization
The amount of 9CBP mRNA, quantitated microspectrophotometrically using a 35S-labeled 45-mer oligonucleotide probe, showed an increase during pregnancy only in the fetal trophoblast, with a constant low level on the maternal side (Figures 5 and Figures 11A). The level in the trophoblast increased to 150 dpc and then remained at this high level for the rest of pregnancy. The glands showed a consistent low level, although patchy (Figures 6B and 6D), throughout pregnancy, and the uterine epithelium even less.
Interplacentomal Double Labeling
Co-localization of bovine placental lactogen and 9CBP mRNA was carried out by initially marking the bovine placental lactogen cells using immunoperoxidase techniques and subsequently the 9CBP and mRNA-replete cells with autoradiographic probe detection. This confirmed at the light microscope level the presence of 9CBP mRNA in both uninucleate and the premigratory binucleate cells and its loss from binucleate cells on migration (results not shown).
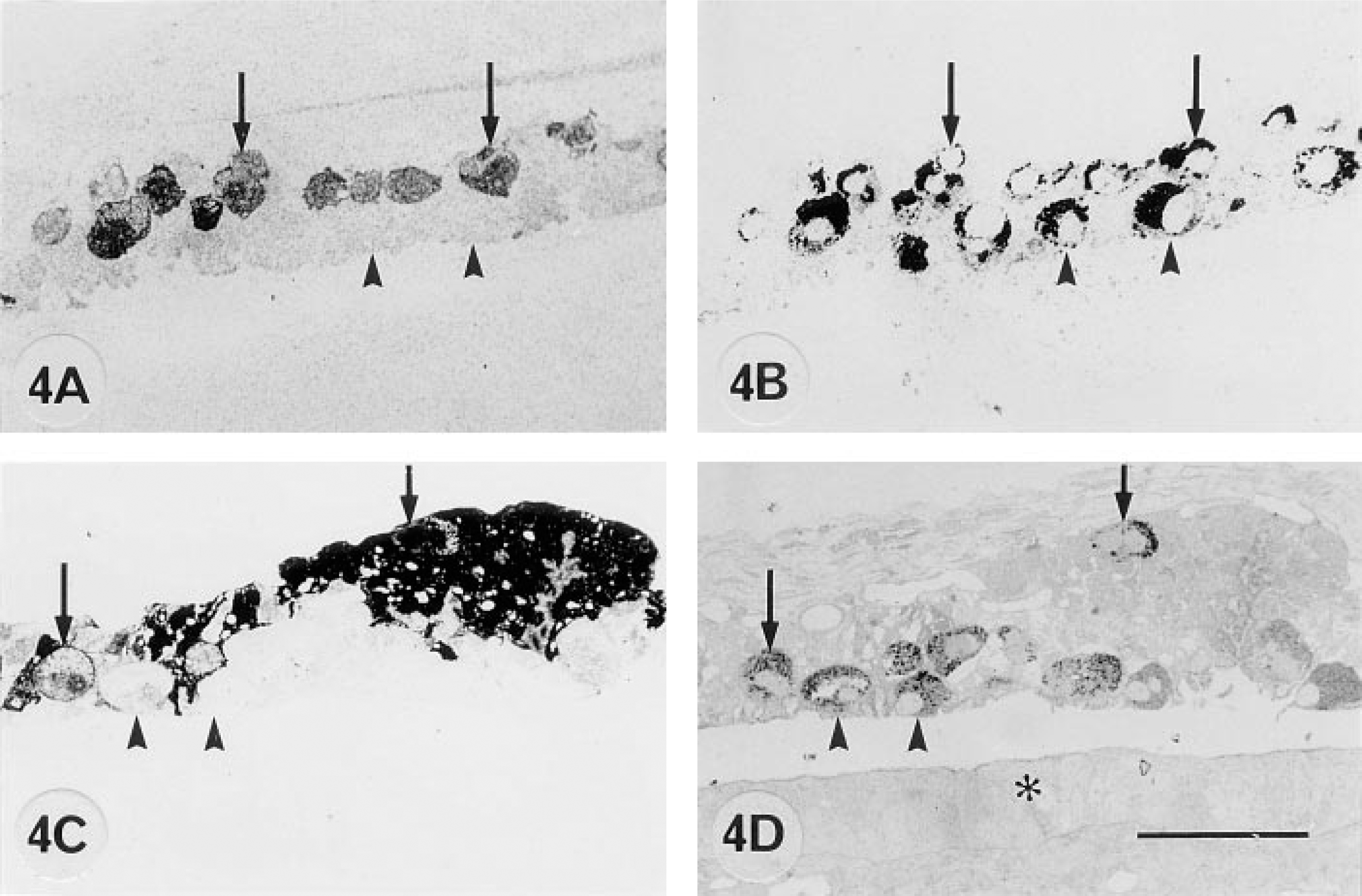
Differences in 9CBP localization in trophoblast binucleate cells of interplacentomal region of bovine placenta at 100 dpc (
Electron Microscopic Immunocytochemistry
Postembedding immunocytochemical analysis and quantitation of colloidal gold distribution on Lowicryl K4M sections showed that 9CBP label, when present, was found throughout nucleoplasm and cytoplasm but absent from all cell organelles—mitochondria, endoplasmic reticulum, lysosomes, Golgi cisternae, and cytoplasmic vesicles (Figure 7). Quantitation showed that label was evenly distributed throughout the cell in gland epithelium (7 ± 1 gold particles per μm2 ± SEM,
Phase-contrast (A,C,E) and darkfield (B,D,F) micrographs of in situ hybridization on paraformaldehyde-fixed cryosections of the interplacentomal region of bovine placenta at 55 dpc (A,B) and 250 dpc (C,D) with anti-sense [α-35S]-dATP-labeled 9CBP oligonucleotide probe. Control section (E,F) was probed with sense 9CBP oligonucleotide probe. Expression of 9CBP mRNA is high in interplacentomal trophoblast (arrowheads). Uterine epithelium (arrows) shows no evidence of 9CBP mRNA. Results from quantitation of the mRNA signal on such slides are shown in Figure 11A. Eosin stain. Bars = 100 μm.
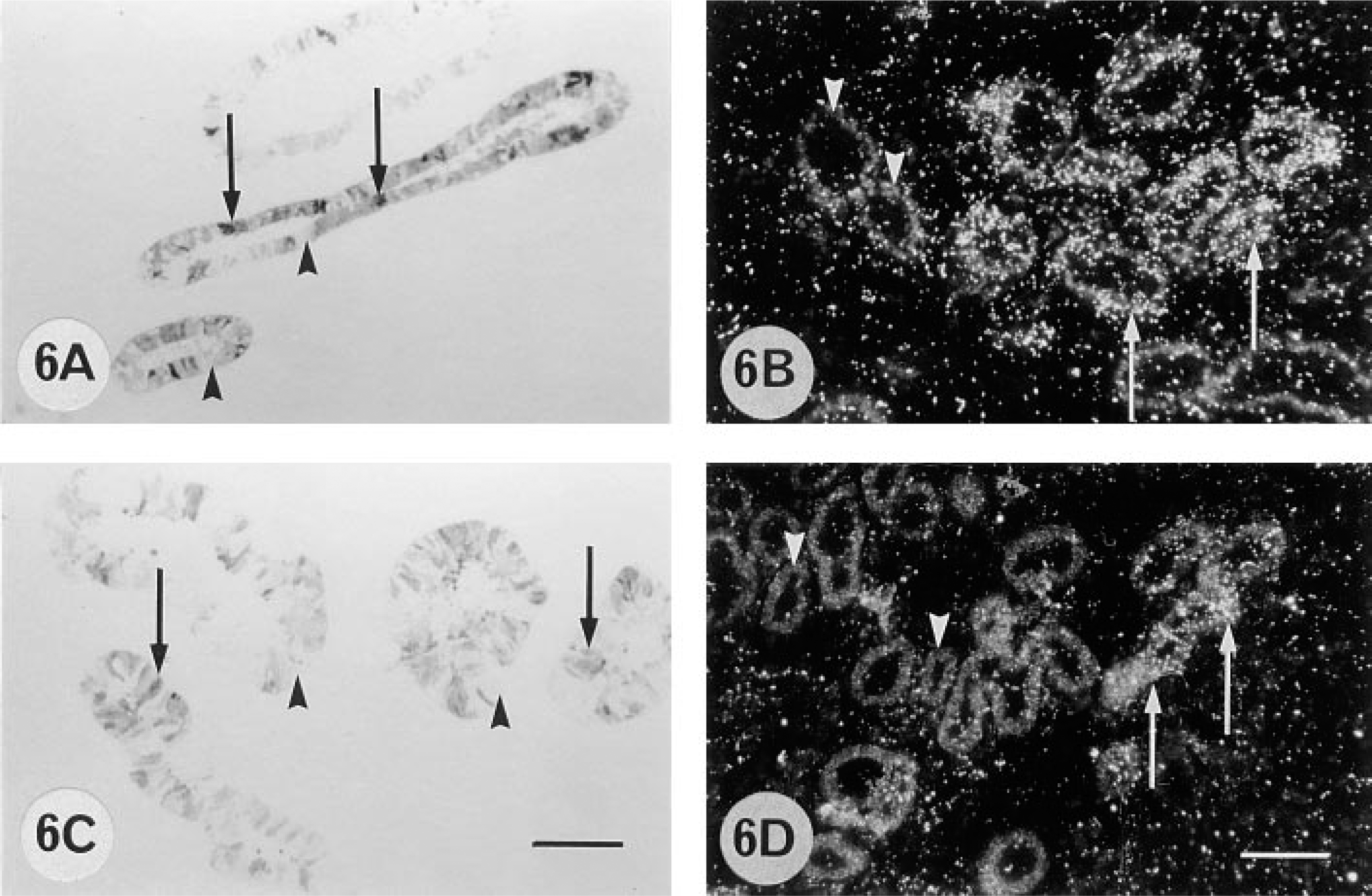
Brightfield micrographs of Lowicryl K4M sections from 155 dpc (A) and 270 dpc (C) pregnant cows and darkfield micrographs of cryosections of 155 dpc (B) and 250 dpc (D) pregnant cows, showing immunohistochemical localization of 9CBP (A,C) and in situ hybridization with 9CBP [α-35S]-dATP-labeled oligonucleotide probe (B,D) in glandular epithelium. 9CBP immunoreactivity and 9CBP mRNA are localized within individual glandular cells (arrows); other glandular cells are 9CBP-immuno-negative and mRNA-negative (arrowheads). Bars =100 μm.
The initially nonpolarized interplacentomal binucleate cells showed about half (18 ± 2 gold particles per μm2) the average cytoplasmic label of the uninucleate cells (and never any label over their many characteristic granules), but this decreased to insignificant levels (less than 1 ± 1 gold particle per μm2) in migrating binucleate cells (Figure 4), defined by participation in the apical trophoblast tight junction.
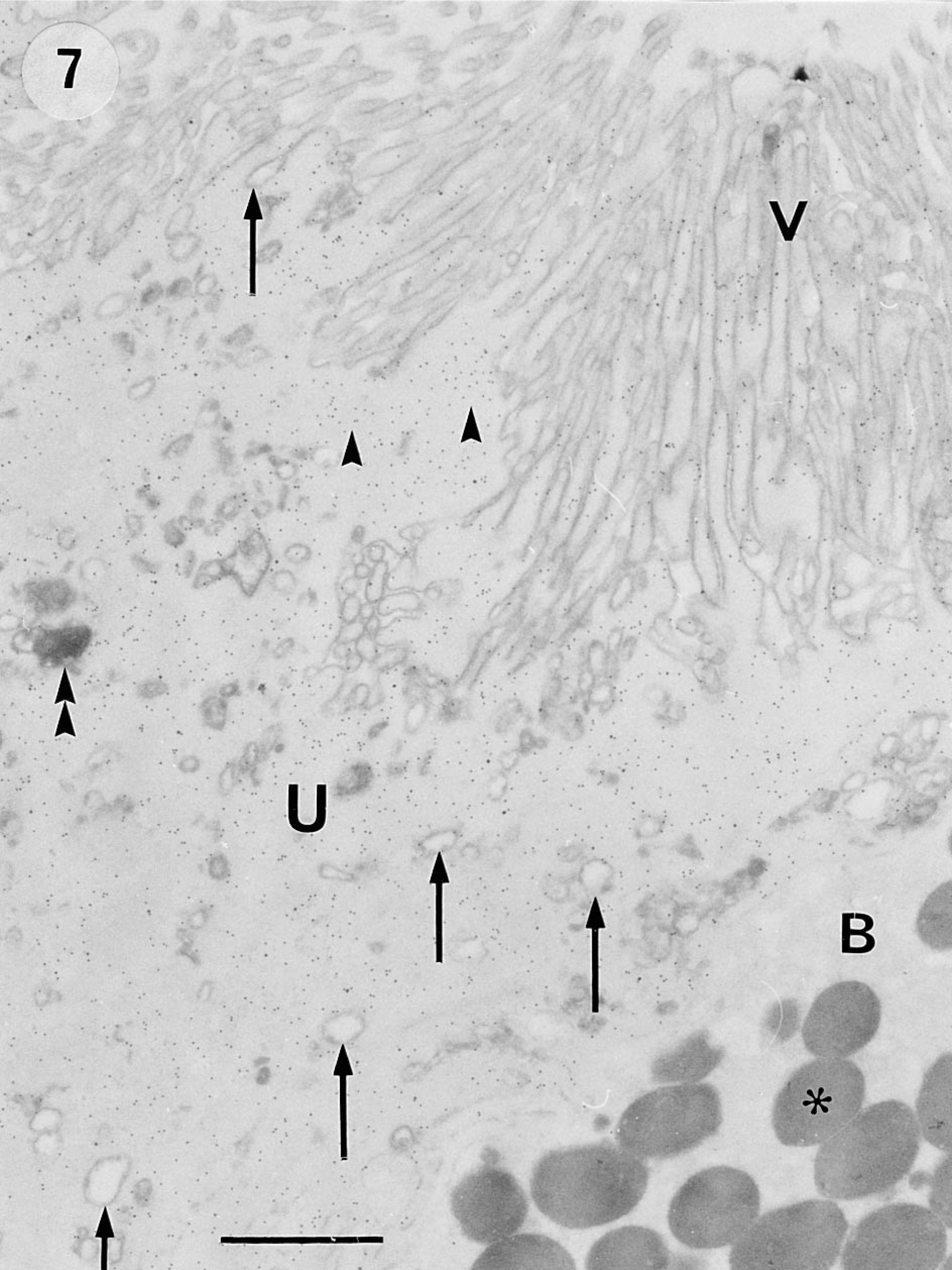
Electron micrograph of immunocytochemistry on ultrathin Lowicryl K4M section from interplacentomal region of bovine placenta at 270 dpc, showing the even distribution of label in the entire trophoblast uninucleate cell (U) and the absence of label within apical vesicles (arrows). The region immediately below the microvilli (V) is very heavily labeled (arrowheads). In the bottom right-hand corner, part of a migrating binucleate cell (B) shows no label over its cytoplasm or characteristic granules (asterisk). The microvilli, the apical vesicles, and phagolysomes (double arrowheads) of the uninucleate cell and the granules of the binucleate cell are the only organelles clearly shown on the phosphotungstic acid-stained sections. Fixation with 3% paraformaldehyde-1% glutaraldehyde; Lowicryl K4M embedding; phosphotungstic acid staining. Bar = 1 μm.
Placentomal Immunocytochemistry
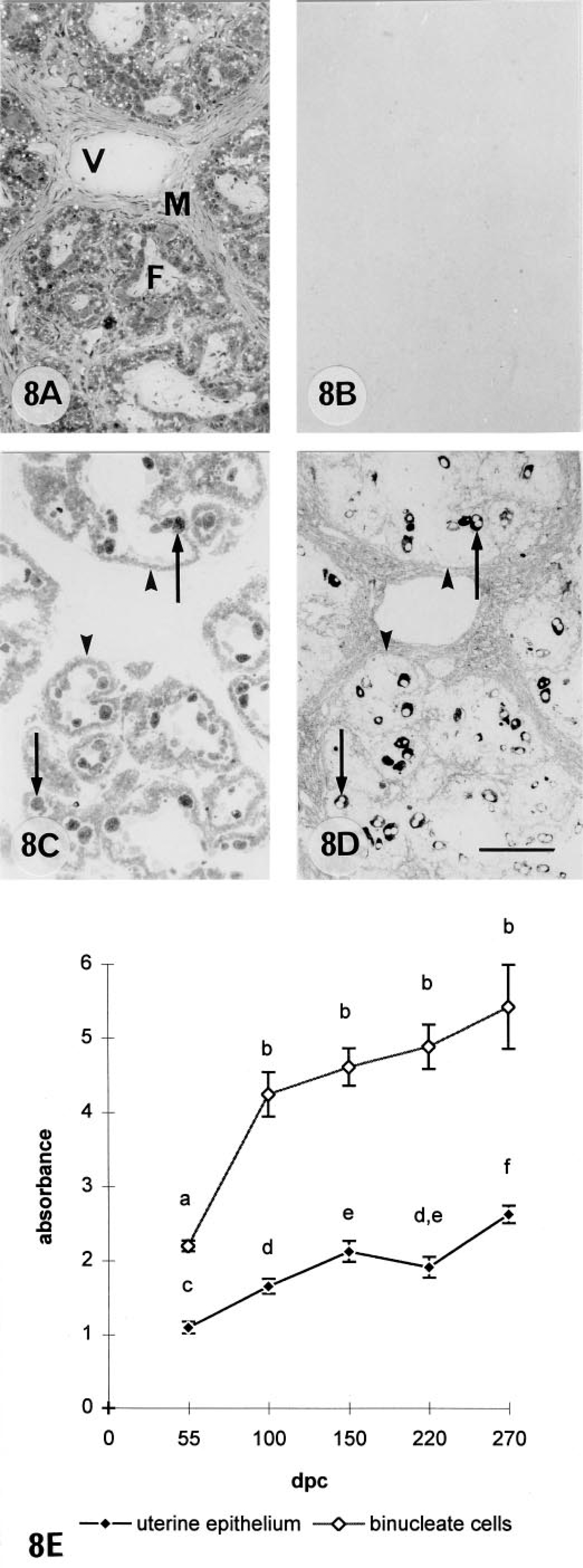
The level of 9CBP expression in the placentomes was much less than and shows far less change than the interplacentomal level throughout pregnancy. The trophoblast uninucleate cells showed only an insignificant background leel, whereas the binucleate cells (15–20% of the trophoblast placentomal cells and defined immunologically; see below) expressed considerable amounts (8 ± 1 gold particles per μm2) from their earliest appearance (Figure 8). Unlike the interplacentomal binucleate cells, they continued to express the 9CBP protein throughout their complete life cycle. The cuboidal uterine epthelial cells expressed levels of 9CBP throughout pregnancy, which are on average at about half (3 ± 1 gold particles per μm2) the level found in the binucleate cell and which showed much smaller increases in the second half of pregnancy than during placental initiation. Unequivocal identification of the 9CBP-positive cells in the placentome (Figure 8C) as binucleate can be achieved by using immunodetection of a binucleate cell-specific antibody, bovine placental lactogen, on a section serial (Figure 8D) to the 9CBP-stained example. Such sections clearly show an identical pattern of cells, with 85–95% correlation between the two distributions. The large size of the binucleate cell, 25–30 μm, makes the comparison very straightforward and the restriction of the bovine placental lactogen to the cytoplasmic granules elegantly outlines the two nuclei at certain levels, further unequivocally identifying this population. Favorable sections also illustrate the formation of trinucleate cells in the uterine epithelium.
Placentomal In Situ Hybridization
Placentomal binucleate cells showed the highest levels of 9CBP mRNA found anywhere in the placenta at between 100 and 150 days of pregnancy (Figure 9). The levels then decreased to equal those throughout the interplacentomal trophoblast towards term (Figure 11B). There was a negligible amount in the uninucleate trophoblast cells, but a constant low level of 9CBP mRNA was measurable in the uterine epithelial cells throughout pregnancy. The heavily labeled cells in the placentome were identified as binucleate cells on structural grounds by histological criteria, by serial section immunocomparison, and by double labeling as detailed above, using a combination of immunocytochemical and in situ hybridization protocols.
Northern Blot Analysis
The bovine 9CBP oligonucleotide probe hybridized to a 500-base transcript in bovine placentomal and interplacentomal mRNA blots. This corresponded to the results of Kumar et al. (1989) for bovine 9-kD calcium binding protein. At approximately equivalent RNA loadings there was little difference in mRNA band densities between the fetal part of the placentome and the interplacentome. The maternal part of the placentomal tissue contained much less and the surface interplacentomal uterine epithelium produced no detectable hybridization (Figure 10).
Discussion
This study demonstrates for the first time that, in the cow, it is only in the areas of flat interplacentomal trophoblast that there is a significant increase in the amount of 9CBP protein and mRNA during pregnancy. This is consistent with a limited area of trophoblast providing the basis for an increasing level of active calcium transport across the placenta to support growth and development of the fetal long bones. In other systems for bulk calcium transport across cells, e.g., in gut enterocytes and kidney tubules, active transport dependent on 9 or 28CBP concentrated in a limited area has been demonstrated to be a vital, controllable complement to the much greater passive transport of calcium taking place over a much wider area (Bronner 1991; Kumar 1995).
We have already shown in the sheep the same potential for active calcium transport: a high concentration of 9CBP restricted to the interplacentomal trophoblast as well as demonstrating the capacity of this trophoblast for calcium uptake and transport in Ussing chambers (Abbas et al. 1993) and trophoblast disc experiments (Jones et al. 1997). In vivo in ruminants (as in all vertebrates), because the fetal serum calcium concentration is normally higher than that in the mother's blood (Pitkin 1985; Care 1991), the calcium transport process must by definition be an active process, and we have shown by in vivo placental perfusion in the sheep the involvement of parathyroid hormone-related protein in the control of the overall placental calcium transport (Care 1991; Care et al. 1990,1996).
Light micrographs (
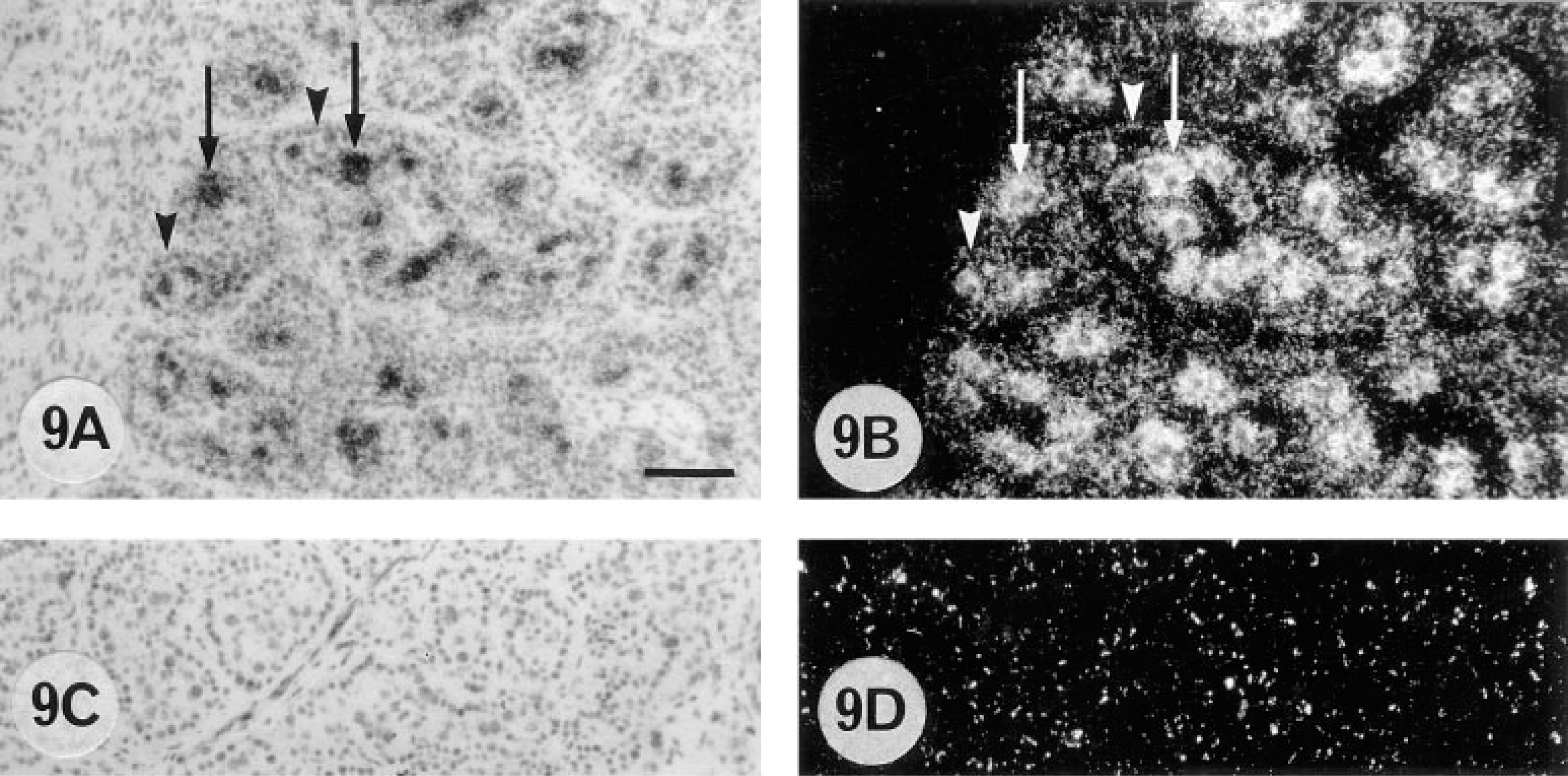
Cellular localization of 9CBP mRNa in the placentomal region of bovine placenta (150 dpc) by in situ hybridization with anti-sense [α-35S]-dATP-labeled 9CBP oligonucleotide probe (
Although the interplacentomal trophoblast 9CBP protein expression correlates well with peak fetal demand in both sheep and cow, the absence of any change in the low level in the cow uterine glands throughout pregnancy is very different from the considerable increases seen in sheep glands (Morgan et al. 1997). However, because control of the net transplacental calcium flux lies primarily with the fetus, it is the trophoblast level and control of the 9CBP protein that are more important, and in the cow the transport of calcium from the maternal blood to the interplacentomal uterine lumen could be mainly passive, driven by trophoblast demand.
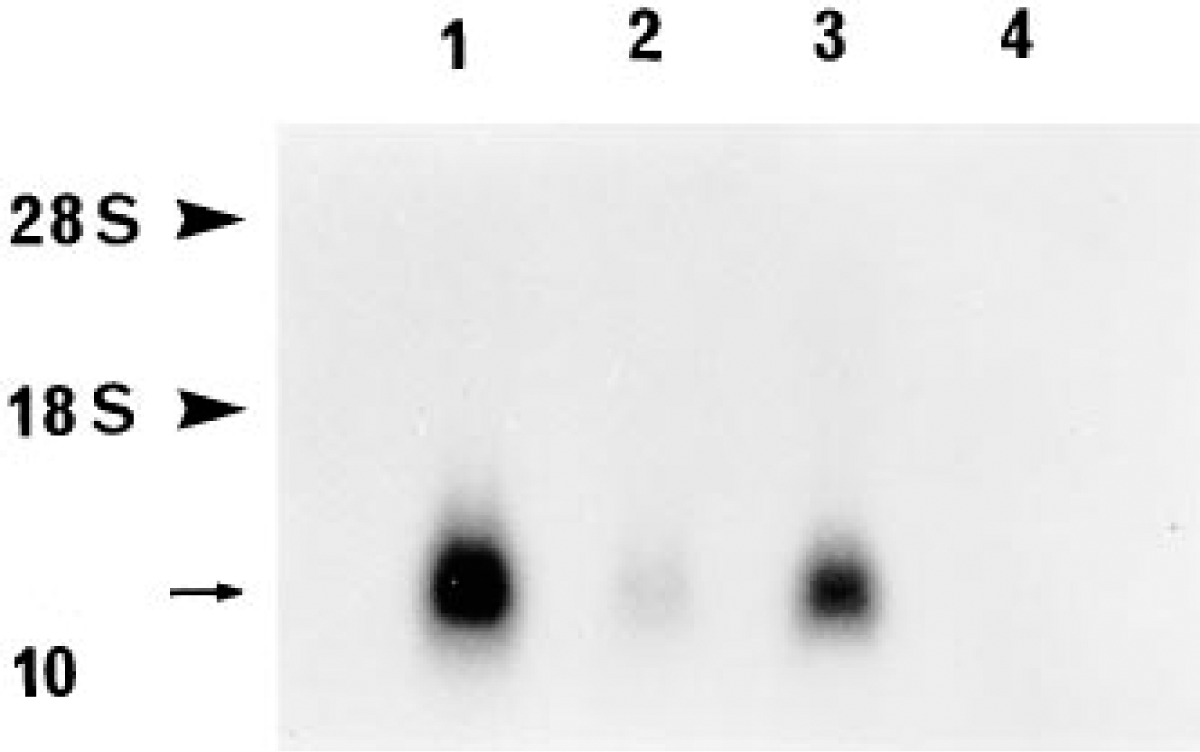
Northern blot hybridization of total RNA obtained from different regions of bovine placenta with the 9CBP oligonucleotide probe, demonstrating specificity of hybridization and difference in 9CBP mRNA content. An arrow indicates the position of 9CBP mRNA bands (-0.5 kb) in the fetal part of the placentome (Lane 1), in the fetal part of the interplacentome (Lane 3), in the maternal part of the placentomal (Lane 2) samples, and negative reaction in the maternal interplacentomal region (Lane 4). Note the difference in hybridization signal in the samples from different regions of the placenta. Arrowheads indicate the position of the ribosomal RNA bands obtained by comparison with ethidium bromide-stained RNA gel.
If the active calcium transport in the interplacentomal trophoblast is mediated by cytoplasmic 9CBP, our quantitative electron microscopic localization results, with cow as with the sheep, indicate a process of facilitated diffusion. We found no evidence for any 9CBP localization in membrane-bound structures capable of a vesicular shuttle, as suggested by Nemere in chick enterocytes (Nemere et al. 1991; Nemere 1992).
The asymmetric (higher apical than basal) cytoplasmic distribution of CBP that we find in the cow trophoblast cells has also been reported in chicken enterocytes for 28CBP (Thorens et al. 1982; Nemere et al. 1991) and correlates with the initial localization of 47Ca uptake by ion microscopy in this system (Fullmer et al. 1996). It could represent a transient store or a device to enhance the rate of facilitated diffusion.
The placentomal localization of 9CBP protein and mRNA to binucleate cells and uterine epithelium is unique among ruminants thus far examined. There is no correlation with calcium transport requirements because changes through pregnancy are minimal, the binucleate cells are migrating (Wooding 1992; Wooding et al. 1992; Wooding and Flint 1994) in the wrong direction for calcium transport, and the low levels of 9CBP in uterine epithelial cells could only move insignificant amounts of calcium halfway.
We found no evidence for a significant proportion of mature binucleate cells not expressing 9CBP (Reiswig et al. 1995). All binucleate cells identified by bPL immunocytochemistry showed 9CBP on serial sections. This is the first example of high concentrations of 9CBP in a cell quite incapable of calcium transport in a specific direction. By analogy with the suggested non-calcium-transporting function of 28CBP in brain and other tissues (Christakos et al. 1989), the binucleate cell 9CBP may be involved in an extension of the normal system of calcium homeostasis necessary for the complex differentiation processes in the binucleate cell. Why this should be unique to the cow binucleate cell remains unknown, but binucleate cells do show protein content variation during development (Morgan et al. 1989) and in different areas of the placenta (Wooding et al. 1996b).
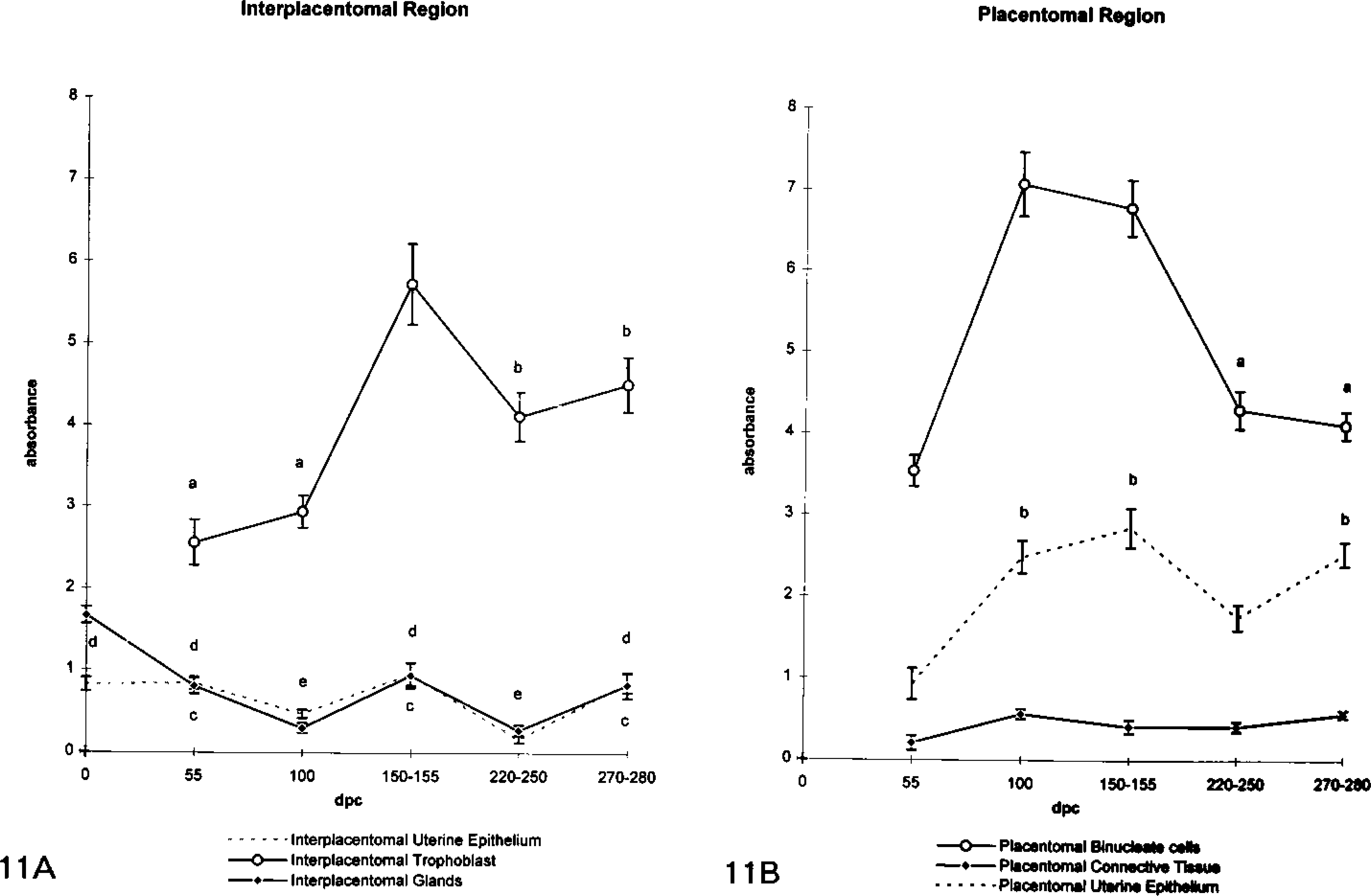
Quantitation of 9CBP oligonucleotide probe hybridization signal on interplacentomal (
In summary, 9CBP protein and mRNA are found in the cow only in particular populations of placental cells, with a clear increase in the second half of pregnancy only in interplacentomal trophoblast uninucleate cells. Intracellular distribution of the 9CBP protein indicates a cytoplasmic, non-membrane-bound molecule. This evidence supports the hypothesis that 9CBP-dependent active calcium transport across the placental barrier in the cow occurs by facilitated diffusion and is restricted to interplacentomal trophoblast cells, as demonstrated in other ruminants. The function of 9CBP in the placentomal binucleate cells and uterine epithelium is unique to the cow and is unlikely to be involved in calcium transport.
Footnotes
Acknowledgements
We are very grateful to Drs Bruns, Inpanbutr, and Friesen for generous gifts of antibodies and to Dr Simon Lennard for help with the Northern blotting. L. Nikitenko would like to make a personal acknowledgement of the unstinting advice and encouragement from Dr A.L. Glazyrin during his training in Irkutsk and was supported in England by a postgraduate scholarship from the Russian Presidents fund awarded by the Russian Federation State Committee for Higher Education.
