Abstract
Melatonin is synthesized in the pineal gland and retina during the night. Retinal melatonin is believed to be involved in local cellular modulation and in regulation of light-induced entrainment of circadian rhythms. The present study provides the first immunohistochemical evidence for the localization of melatonin 1a-receptor (MT1) in human retina of aged subjects. Ganglion, amacrine, and photoreceptor cells expressed MT1. In addition, MT1 immunoreactivity was localized to cell processes in the inner plexiform layer and to central vessels of the retina, as well as to retinal vessels but not to ciliary or choroidal vessels. These results support a variety of cellular and vascular effects of melatonin in the human retina. Preliminary evidence from patients with Alzheimer's disease (AD) revealed increased MT1 immunoreactivity in ganglion and amacrine cells, as well as in vessels. In AD cases photoreceptor cells were degenerated and showed low MT1 expression.
Keywords
T
In mammals, the specific actions of melatonin are mediated by two different subtypes of G-protein-coupled receptors, the melatonin 1a (MT1) and melatonin 1b (MT2) receptors (Brydon et al. 1999; Dubocovich et al. 2000). Specific polyclonal antibodies are available against MT1 (Brydon et al. 1999). Its expression has been studied in rodents using immunocytochemistry and in situ hybridization (Fujieda et al. 1999, 2000). However, corresponding studies in humans remain to be done. In recent studies we were able to show localization of MT1 in humans in cerebrovascular tissue emphasizing melatonin's vasoactive properties (Savaskan et al. 2001) and in hippocampal neurons (Savaskan et al. in press).
The aim of this study was to provide immunohistochemical evidence, the first we are aware of, for the distribution of MT1 in human retina and in the ocular vascular system. In addition, eyes of two AD patients were included in the study as a preliminary investigation of possible alterations in the disease process.
Materials and Methods
Human Tissue
Paraffin-embedded human eyes fixed in 4% paraformaldehyde were obtained from seven subjects without neurological or psychiatric disease (six women and one man, mean age 83.1 ± 9.8 years and two male AD cases, 83 and 84 years old. The mean postmortem delay was 21 hr and 31 min ± 9 hr and 10 min) for controls and 16 hr and 15 min for AD cases (Table 1). The diagnosis of AD was made with clinical evaluation and confirmed by postmortem neuropathological examination. The cause of death in most cases was heart failure or pneumonia, and pancreatitis for one control case (Table 1). The sample collection was approved by the Ethics Committee criteria and according to the Helsinki Declaration of 1975. Tissue samples were cut in the sagittal plane and 4-μm-thick consecutive sections were made with a microtome.
Immunohistochemistry
The sections were mounted on coated glass slides and deparaffinized. Endogenous peroxidase activity was blocked by bathing the sections in 80% methanol, 0.6% H2O2 for 20 min at room temperature (RT). After several washes in PBS and preincubation in PBS with 3% blocking serum for 30 min the sections were reacted with primary antibody overnight at RT. The affinity-purified polyclonal antibody used to specifically detect MT1 was developed against a peptide (peptide 536) corresponding to a sequence found in the C-terminus of the receptor and the antibody recognition of native MT1 has been ascertained (Brydon et al. 1999). The non-commercial antibody was kindly provided by L. Brydon and R. Jockers (Institut Cochin de Génétique Moleculaire; Paris, France). In addition, the antibody has been applied successfully in previous immunohistochemical experiments (Savaskan et al. 2001; Savaskan et al. in press). The optimal concentration of the primary antibody was experimentally determined to be 1:100. In a second step, after several washes with PBS the sections were incubated with biotinylated secondary antibody (Vector Laboratories; Burlingame, CA). The optimal dilution of the secondary antibody was 1:100. After several washes, a pre-formed ABC reagent (Vectastain; Vector Laboratories) was conjugated to the free biotin of the secondary antibody. The antigen was visualized using the peroxidase substrate 3-amino-9-ethylcarbazole (ACE). The samples were then counterstained with Mayer's hemalum. The sections were coverslipped with Kaiser's glycerol gelatin. To test the specificity of the antibody, control sections were stained simultaneously following the same procedure, with the exception that the primary antibody was omitted. All sections were assessed for localization and intensity of specific immunoreactivity on a semiquantitative scale of +/+++ by two blinded observers (Table 2).
Data of control (C) and Alzheimer's disease (AD) casesa
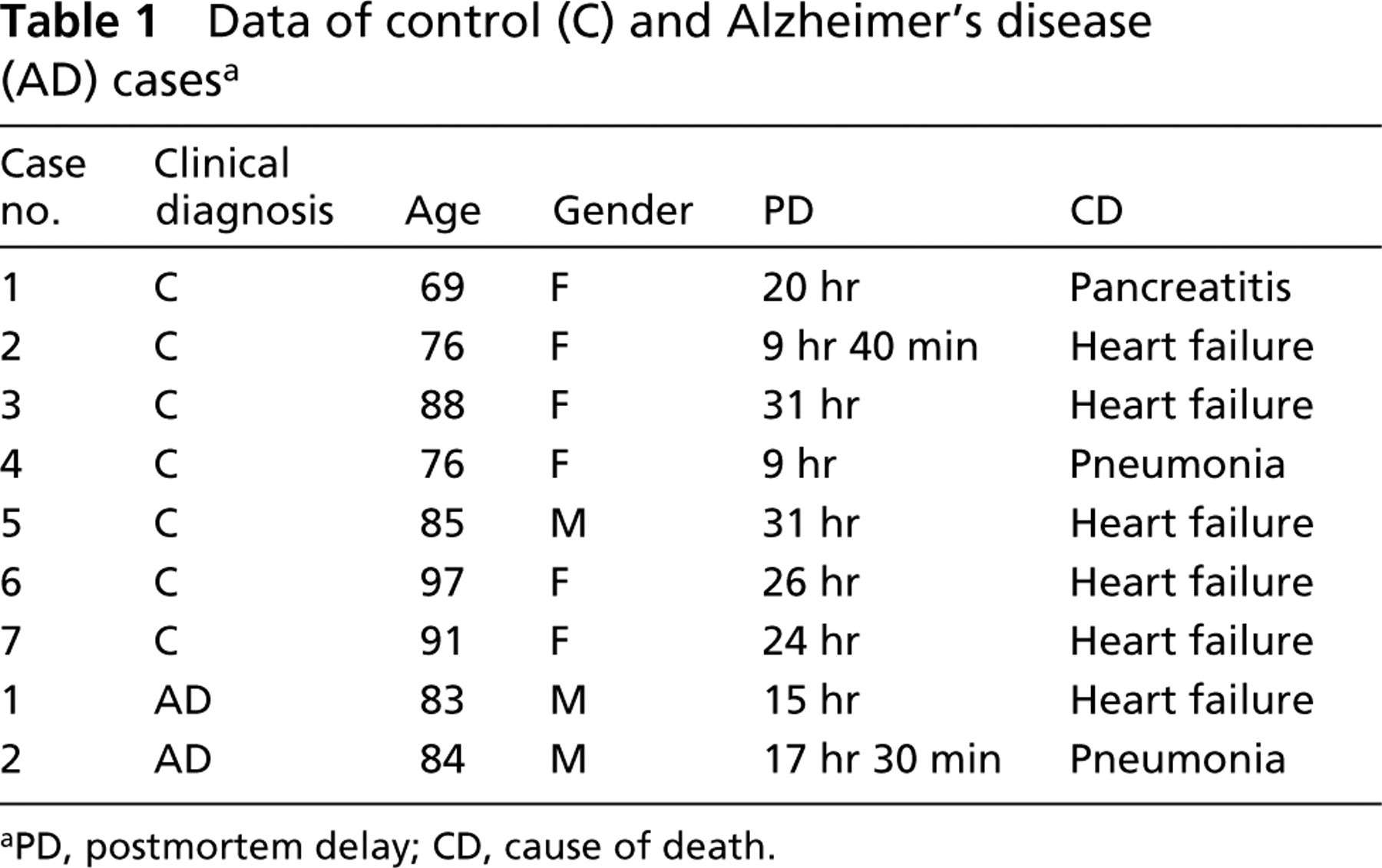
PD, postmortem delay; CD, cause of death.
Results
The cytoarchitectural classification regarding cells, different layers, and vascularization in this section follows detailed descriptions by Spencer (1996).
Cellular Structures
Different cells within the retina displayed specific MT1 immunoreactivity (Figures 1A-1D), whereas adjacent control sections omitting the primary antibody revealed no immune reaction (Figure 1E and 1F). The majority of ganglion cells were immunoreactive for MT1 in their somata and dendritic processes, both in controls and in AD cases (Figure 1A and 1C). Staining was homogeneously distributed in the perinuclear area. In five control cases (Table 1), single MT1-immunoreactive amacrine cells were located in the inner nuclear layer immediately adjacent to the inner plexiform layer. Only a minor subset of amacrine cells were stained. In all cases there was a slight granular MT1 labeling in the inner plexiform layer located on cell processes. The outer plexiform layer and the horizontal cells were not immunolabeled. Photoreceptor cells revealed distinct MT1 immunoreactivity in their inner segments (Figure 1B). Cell somata and outer segments were excluded. All photoreceptor cells were stained so that MT1 extended as an immunoreactive band throughout the photoreceptor cell layer.
Distribution and intensity of MT1 immunoreactivity in human retina in controls (C) and Alzheimer's disease patients (AD)a,b
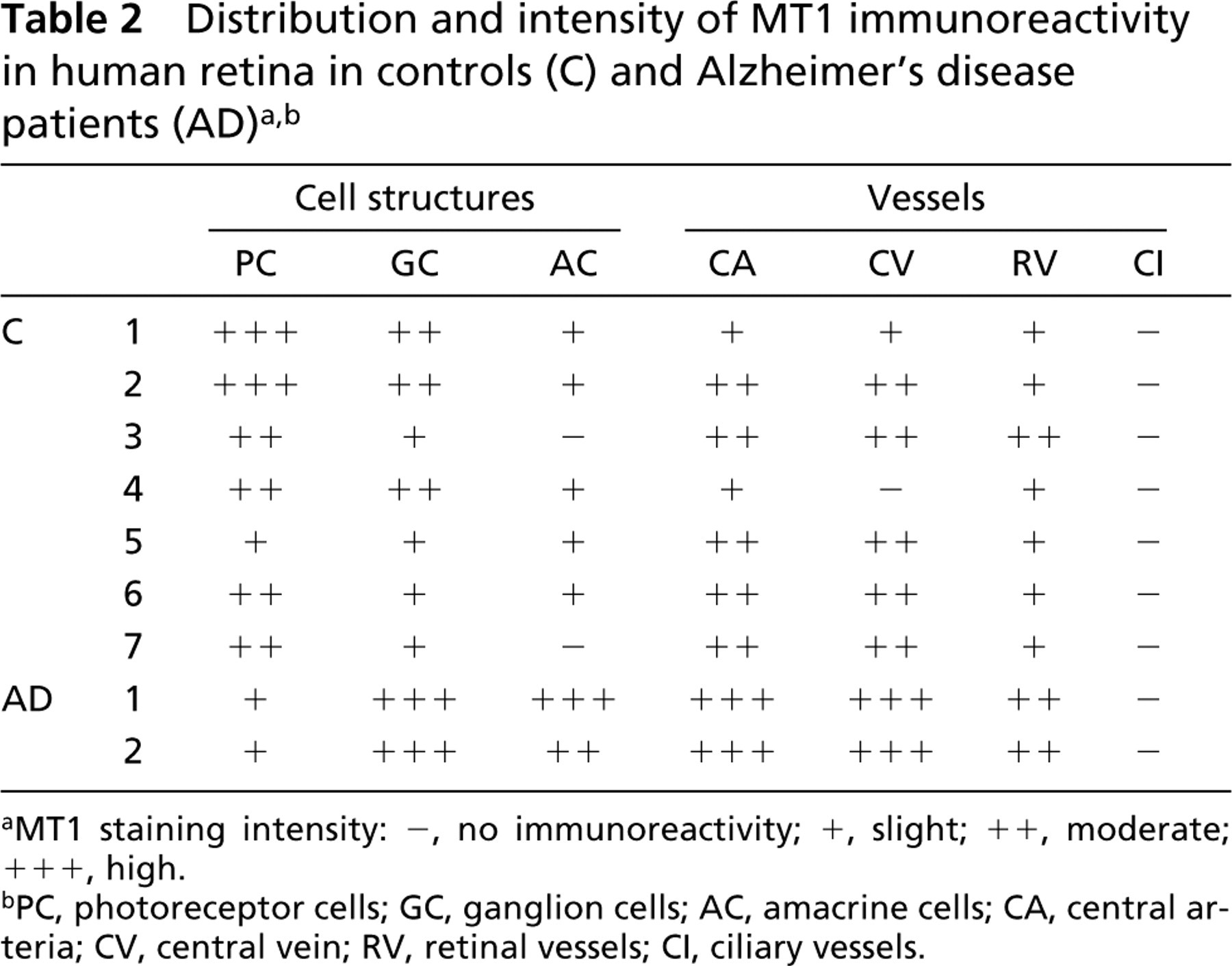
MT1 staining intensity: -, no immunoreactivity; +, slight; ++, moderate; +++, high.
PC, photoreceptor cells; GC, ganglion cells; AC, amacrine cells; CA, central arteria; CV, central vein; RV, retinal vessels; CI, ciliary vessels.
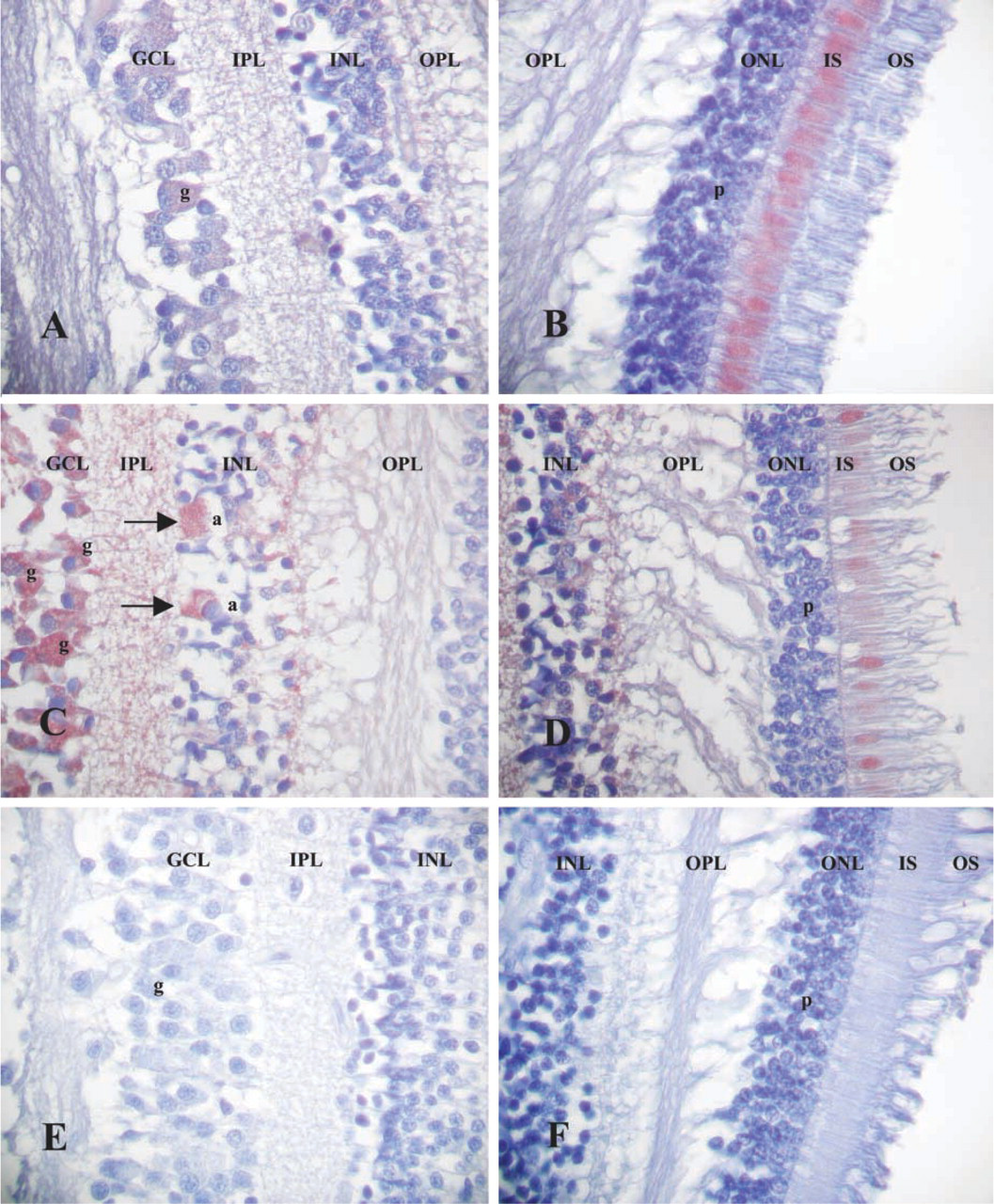
Sagittal sections through the human retina. GCL, ganglion cell layer; IPL, inner plexiform layer; INL, inner nucleiform layer; OPL, outer plexiform layer; ONL, outer nucleiform layer consisting of cell somata of photoreceptor cells; IS, inner segments; OS, outer segments of photoreceptor cells; g, ganglion cells; a, amacrine cells; p, photoreceptor cells. (
The two AD cases showed the following differences in the distribution of MT1 immunoreactivity. First, the staining intensity and the numbers of MT1-immunoreactive cells were distinctly increased in ganglion cells and inner plexiform cell layers (Figure 1C). Second, both AD cases revealed well-stained amacrine cells, and the number of these MT1-immunoreactive cells was twice that in controls (Figure 1C). There was also increased staining intensity within the inner plexiform layer. A decreased MT1 staining in the highly degenerated photoreceptor cell layer could be observed (Figure 1D). Therefore, in AD cases only single cells were immunoreactive for MT1 so that there was no continuous band of immunoreactivity throughout the entire photoreceptor cell layer as in controls (Figures 1B and D).
Ocular Vessels
In all control eyes a distinctly positive MT1 immunoreactivity in the adventitia of central retinal arteries and veins could be observed (Figure 2A and 2B). The same staining pattern was found even in the small retinal vessels located in the ganglion cell layer and inner plexiform layer (Figure 2E). Both ciliary and choroidal vessels were devoid of MT1 immunoreactivity (Figure 2F). In AD, increased MT1 staining was detected in central arteries and veins and in retinal vessels, whereas ciliary and choroidal vessels were not stained (Figure 2D and 2E).
Discussion
The present study has demonstrated the localization of MT1 in cellular structures of the retina and in ocular vessels of aged humans, and has provided preliminary evidence for alterations in retinal MT1 expresssion in AD patients. After previous demonstration of melatonin, its synthesizing enzymes (Bubenik et al. 1974; Vivien-Roels et al. 1981; Wiechmann and Hollyfield 1989; Tosini 2000), and MT1 receptors in vertebrate retina (Fujieda et al. 1999,2000), this is the first demonstration of MT1 distribution in human retina.
Several types of retinal cells displayed MT1 immunoreactivity. The localization of MT1 to ganglion and amacrine cells is in accordance with previous reports in rodent retina (Fujieda et al. 1999,2000), where these cells are believed to be major sites of melatonin action. Because MT1-positive amacrine cells have been found to be dopaminergic and GABAergic by double-immunolabeling experiments, a direct regulatory action of melatonin on dopamine release by enhancing GABA activity has been postulated (Fujieda et al. 2000). Also in agreement with rodent studies, the strongly positive MT1 staining in the inner plexiform layer has been suggested to correspond to melatonin binding sites on dendritic processes of amacrine and ganglion cells (Fujieda et al. 1999,2000). In our series, no MT1 expression was detected in the outer plexiform layer or in the horizontal cells, similar to that in ganglion and amacrine cells. This contrasts to the strongly MT1-positive staining in the outer plexiform layer and horizontal cells in rat retina (Fujieda et al. 1999), which may reflect either interspecies differences or tissue-specific features.
Another contrast to rodents (Fujieda et al. 1999,2000) was the distinct MT1 staining in the inner segments of photoreceptor cells in elderly humans. Melatonin activates disc shedding in rodent photoreceptor cells (White and Fisher 1989). Melatonin has been located in the inner segments of photoreceptor cells and reaches a peak level in outer segments at the end of the night, which may be related to melatonin's protective action counteracting light's toxic effects (Hsu and Molday 1994; Marchiafava and Longoni 1999). Therefore, melatonin may be essential for photoreceptor cells. Our results indicate that, at least in aged humans, external melatonin sources may also play an important role in the maintenance of photoreceptor cells.
Although only two AD cases were investigated, the MT1 alterations were notable. The increase of MT1 immunoreactivity in ganglion and amacrine cells may indicate receptor upregulation, possibly as a regulative response to declining nocturnal melatonin levels in elderly subjects and in AD (Ferrari et al. 2000), as we have already found in hippocampal neurons of AD patients (Savaskan et al. in press). Previous data localizing MT1 to both dendrites and axons of ganglion cells raised the possibility that retinal melatonin may act on dendritic processes, whereas pineal melatonin may stimulate ganglion cell terminals in their target areas (Fujieda et al. 2000). Therefore, pineal melatonin's feedback to the SCN has been assumed to occur both directly and indirectly via ganglion cells. Ganglion cells may be influenced by both pineal and retinal melatonin (Fujieda et al. 2000). This emphasizes a role for melatonin in ganglion cells in modifying retinal sensitivity and thus contributing to entrainment of rhythms driven by the SCN (Fujieda et al. 2000). Mice completely lacking photoreceptor cells showed unattenuated phase-shifting responses to light, suggesting that novel ocular photoreceptors, possibly in ganglion and amacrine cells, may be active in circadian regulation (Freedman et al. 1999; Lucas et al. 1999). MT1 upregulation in retinal cells in AD may be related to altered photic input to the SCN, which itself is diminished in function in AD (Swaab et al. 1985), thus additionally contributing to the enhanced incidence of circadian rhythm disturbances in these subjects.
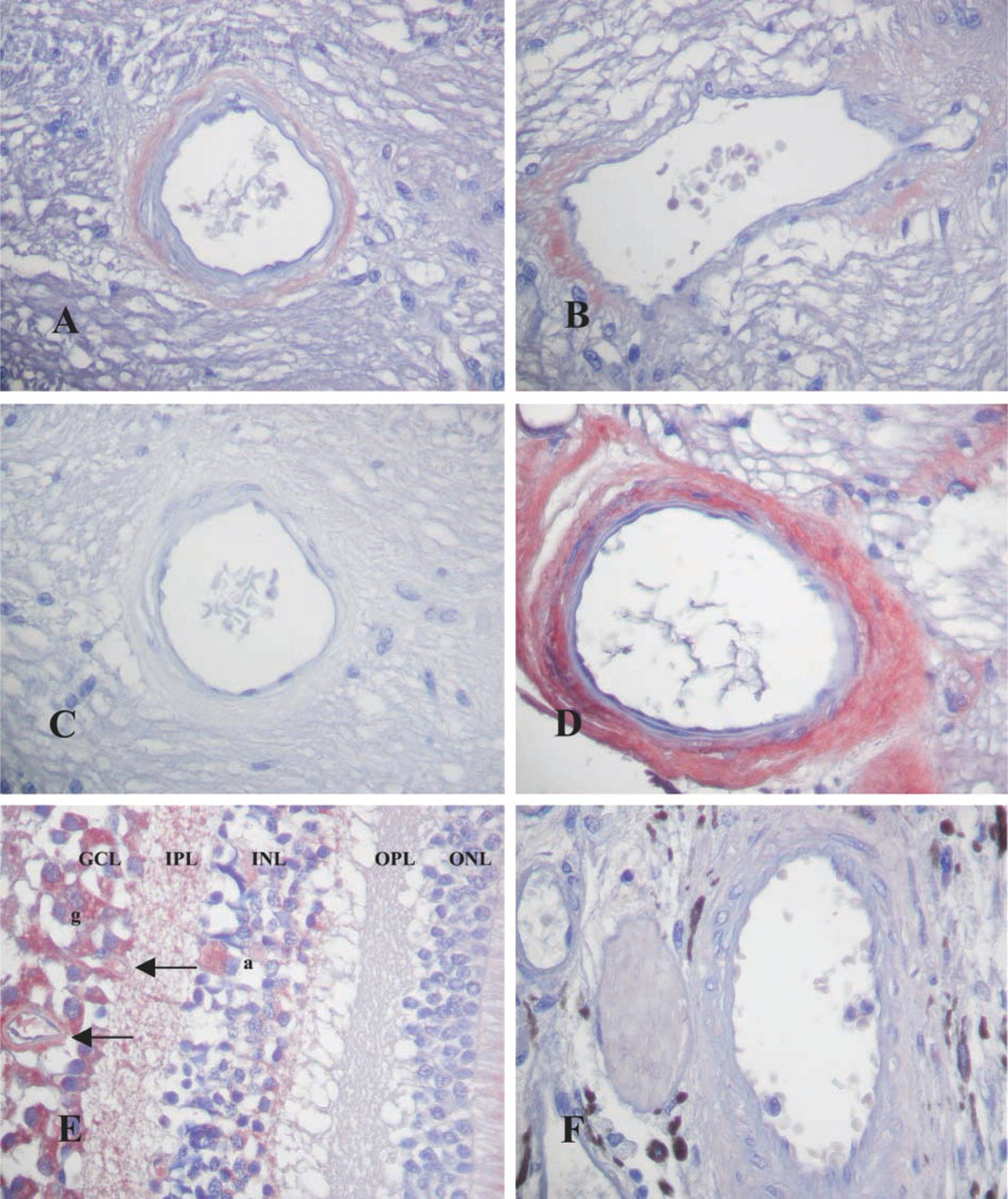
The vessels of the retina. (
Free radicals are generated in mitochondria owing to their high respiratory activity (Demontis et al. 1997) and by light absorption (Remé et al. 1996). Melatonin, as a highly effective antioxidant scavenging free radicals, may therefore be essential for photoreceptor cells that are prone to generation of reactive oxygen species (Marchiafava and Longoni 1999). On the other hand, there is contrary evidence about the protective role of melatonin within the retina. Systemic administration of melatonin increases retinal light damage and the melatonin receptor competitive antagonist luzindole protects photoreceptor cells from light damage (Sugawara et al. 1998). Melatonin's modulatory action on dopaminergic transmission has been postulated to regulate susceptibility to light damage (Iuvone and Gan 1995; Sugawara et al. 1998). Therefore, the decrease in MT1 immunoreactivity in photoreceptor cells in AD may be related to the marked degenerative changes found in these cells.
In addition to cellular structures, central and retinal vessels were also immunoreactive for MT1 in their adventitia. Melatonin is a potent vasoactive substance regulating cerebrovascular responsiveness and blood flow in brain (Régriny et al. 1998). Melatonin administration induces cerebral vasoconstriction, resulting in improved dilatory capacity (Régriny et al. 1998). The vascularization of the retina arises from two different sources, both originating from the arteria ophthalmica. Whereas the arteria centralis retinae supplies the inner layers, including ganglion cell and inner plexiform layers, ciliary and choroidal vessels are responsible for blood circulation in outer layers (Spencer 1996). The localization of MT1 was distinctly restricted to the vascular network of the retinal vessel system. MT1 is localized to the vascular adventitia and not to the smooth muscle layer (Savaskan et al. 2001). The adventitial localization of MT1 suggests an indirect action of melatonin on smooth muscle of the vasculature. The upregulation of vascular MT1 expression in AD retina parallels our recent findings in cerebrovascular MT1 immunoreactivity (Savaskan et al. 2001). Cerebral hypoperfusion plays a decisive role in initiating neurodegeneration in AD (De la Torre and Stefano 2000). Therefore, the observed alterations in vascular MT1 expression in other brain regions in AD (Savaskan et al. 2001) may be accompanied by corresponding changes in retina.
Taken together, the results provide evidence that melatonin may be involved in different cellular and vascular processes in the human retina via MT1. The evaluation of a larger number of cases will be necessary to confirm the striking alterations in AD. Understanding the effects of melatonin in retina as well as brain may be important to combat neurodegeneration and circadian disturbances in AD.
Footnotes
Acknowledgements
Supported by grants from the CNRS, the Université Paris VII, and the Association pour la Recherche sur le Cancer (ARC 5513) to LB and RJ.
We thank Prof Ch. E. Remé (University of Zürich, Switzerland) for her invaluable advice.
