Abstract
Recent studies suggest important functions for laminin-8 (Ln-8; α4β1γ1) in vascular and blood cell biology, but its distribution in human tissues has remained elusive. We have raised a monoclonal antibody (MAb) FC10, and by enzyme-linked immunoassay (EIA) and Western blotting techniques we show that it recognizes the human Ln α4-chain. Immunoreactivity for the Ln α4-chain was localized in tissues of mesodermal origin, such as basement membranes (BMs) of endothelia, adipocytes, and skeletal, smooth, and cardiac muscle cells. In addition, the Ln α4-chain was found in regions of some epithelial BMs, including epidermis, salivary glands, pancreas, esophageal and gastric glands, intestinal crypts, and some renal medullary tubules. Developmental differences in the distribution of Ln α4-chain were detected in skeletal muscle, walls of vessels, and intestinal crypts. Ln α4- and Ln α2-chains co-localized in BMs of fetal skeletal muscle cells and in some epithelial BMs, e.g., in gastric glands and acini of pancreas. Cultured human pulmonary artery endothelial (HPAE) cells produced Ln α4-chain as Mr 180,000 and 200,000 doublet and rapidly deposited it to the growth substratum. In cell-free extracellular matrices of human kidney and lung, Ln α4-chain was found as Mr 180,000 protein.
L
To date, five laminin α-, three β-, and three γ-chains have been described and are known to form at least 14 laminin isoforms (Colognato and Yurchenco 2000; Libby et al. 2000). Using molecular probes and monoclonal antibodies (MAbs), we have elucidated tissue distributions of most Ln α-chains. The Ln α5-chain appears to be the most abundantly expressed α-chain, whereas the Ln α1-chain is the most restricted (Miner et al. 1997; Ekblom et al. 1998; Falk et al. 1999; Virtanen et al. 2000). Ln α3-chain is found in most epithelial BMs, with the exception of some simple epithelia (Carter et al. 1991; Marinkovich et al. 1992; Lohi et al. 1997). In contrast to the aforementioned laminin chains, which mostly appear to be products of epithelial cells, the Ln α2-chain appears to be predominantly a product of cells of mesodermal origin. It is confined to mesoderm-derived tissues, such as skeletal muscle and peripheral nerves (Vuolteenaho et al. 1994; Perreault et al. 1998; Lefebvre et al. 1999).
The laminin α4-chain is a component of three known laminins, Ln-8 (α4β1γ1), Ln-9 (α4β2γ1), and the recently identified Ln-14 (α4β2γ3) (Colognato and Yurchenco 2000; Libby et al. 2000). It has been previously shown that in rodents it is widely expressed in various mesodermal tissues, including, e.g., skeletal muscle, smooth muscle, heart, and endothelium (Liu and Mayne 1996; Frieser et al. 1997; Iivanainen et al. 1997; Miner et al. 1997; Talts et al. 2000). In rodents, Ln α4-chain is produced at least by endothelial cells, adipocytes, and alveolar fibroblasts (Frieser et al. 1997; Niimi et al. 1997; Pierce et al. 1998). Although the distribution of this protein has remained largely unresolved in human tissues, recent studies suggest important functions for Ln-8 in the biology of human blood and endothelial cells (Geberhiwot et al. 1999,2000a,2001; Pedraza et al. 2000; Siler et al. 2000; Sixt et al. 2001; Spessotto et al. 2001; Thyboll et al. 2002).
The aim of the present study was to elucidate the distribution of the Ln α4-chain in developing and adult human tissues. For this purpose we raised and characterized MAb FC10 recognizing the human Ln α4-chain. Using immunohistochemistry (IHC), we show that the Ln α4-chain is found in all human tissues studied. With the exception of the glomerular BM, Ln α4-chain was found in all endothelial BMs in fetal and adult tissues. Our results also show that endothelial cells produce two variants of the protein, of which only one is detectable in tissues.
Materials and Methods
Monoclonal Antibodies to Human Laminin α4-chain
Female Balb/c mice were first immunized using Freund's complete adjuvant (Sigma; St Louis, MO) and native human Ln-8, purified from T98G glioblastoma cells as described (Fujiwara et al. 2001). Three further immunizations were made using Freund's incomplete adjuvant (Sigma) and a recombinant hybrid Ln-8, produced in a mammalian expression system and containing human Ln α4-chain (Kortesmaa et al. 2000). The last immunization was performed 3 days before hybridization. Hybridomas were generated according to standard methods (Köhler and Milstein 1975). Briefly, the spleen was minced and cells were collected in RPMI 1640 medium and mixed with P3X63Ag 8.653 mouse myeloma cells (American Type Culture Collection; Manassas, VA). The mixed cells were centrifuged, the cell pellet was resuspended in 1 ml of polyethyleneglycol (PEG 4000; Life Technologies, Grand Island, NY) prewarmed to 37C, and RPMI 1640 medium was added to the suspension. The cells were centrifuged, re-suspended in HAT selection medium (Biological Industries; Kibbutz Bait Maemek, Israel), and transferred to 96-well plates, into which macrophages had been plated the day before. The macrophages were collected from Balb/c mice by rinsing the peritoneal cavity with RPMI 1640 medium. The study plan was approved by the Animal Experimentation Committee of the University of Helsinki. For screening of the hybridomas, sections of human fetal and adult skeletal muscle and kidney were immunostained with the hybridoma supernatants. The selected hybridomas were cloned by manually picking single cells from the cultures. Further characterization was performed by using immunoprecipitation and EIA techniques.
Antibodies
Mouse monoclonal antibodies (MAbs) to laminins and integrins (Int) were as follows: 4C7 to Ln α5-chain (Engvall et al. 1986; Tiger et al. 1997); DG10 against Ln β1-chain (Virtanen et al. 1997); BC7 and 22 to Ln γ1-chain (Geberhiwot et al. 2000b; Määttä et al. 2001; 22 obtained from BD Transduction Laboratories, Lexington, KY); TS2/7 to Int α1-subunit (Hemler et al. 1984; kindly provided by Dr. M.E. Hemler, Boston, MA); 10G11 to Int α2-subunit (Giltay et al. 1989; kindly provided by Dr. J. van Mourik, Amsterdam, The Netherlands); J143 to Int α3-subunit (Fradet et al. 1984; kindly provided by Dr. L.J. Old, New York, NY); 102DF5 to Int β1-subunit (Ylänne and Virtanen 1989); and 3E1 to Int β4-subunit (Chemicon; Temecula, CA). In double immunolabeling experiments, rat MAbs to Ln α2-chain (4H8–2; Schuler and Sorokin 1995) and to Int α6-subunit (GoH3; Sonnenberg et al. 1987; Chemicon) were used. Affinity-purified rabbit antibody (Tiger et al. 1997; kindly provided by D. Gullberg, Uppsala, Sweden) was used to identify Ln α1-chain and tetramethylrhodamine isothiocyanate-coupled Ulex europaeus-I agglutinin (TRITC-UEA-I; Vector Laboratories, Burlingame, CA) was used to demonstrate endothelial cells (Holthöfer et al. 1982). Rabbit antiserum to EHS-laminin (Sigma) was used to visualize all BMs. Rabbit antibodies to recombinant human laminin α4 I/II domains were produced and immunoaffinity-purified as previously described (Iivanainen et al. 1997), and were used in Western blotting experiments.
Enzyme-linked Immunoassay (EIA)
The specificity of MAb FC10 was first assessed by reactivity against isolated recombinant hybrid Ln-8 containing human Ln α4- and γ1-chains and mouse Ln β1-chain (Kortesmaa et al. 2000), human recombinant Ln β1- (Pikkarainen et al. 1992) and Ln γ1-chains (kindly provided by Dr. Sirpa Salo, Univ. of Oulu, Finland; unpublished), as well as placental Ln-10/11 (Life Technologies). Briefly, 96-well plates (Maxi-Sorp; Nunc, Roskilde, Denmark) were coated overnight with either laminins (1 μg/ml) or chains (0.4 μg/ml). After blocking with human serum albumin (10 mg/ml; Sigma), undiluted FC10 supernatant was added and allowed to interact with the proteins for 1 hr at 4C. After three washes with 0.1% Tween-20 in PBS, bound antibodies were detected using goat antibodies to mouse Ig coupled to horseradish peroxidase (DAKO; Glostrup, Denmark) and the enzyme activity was measured using orthophenylenediamine (Sigma).
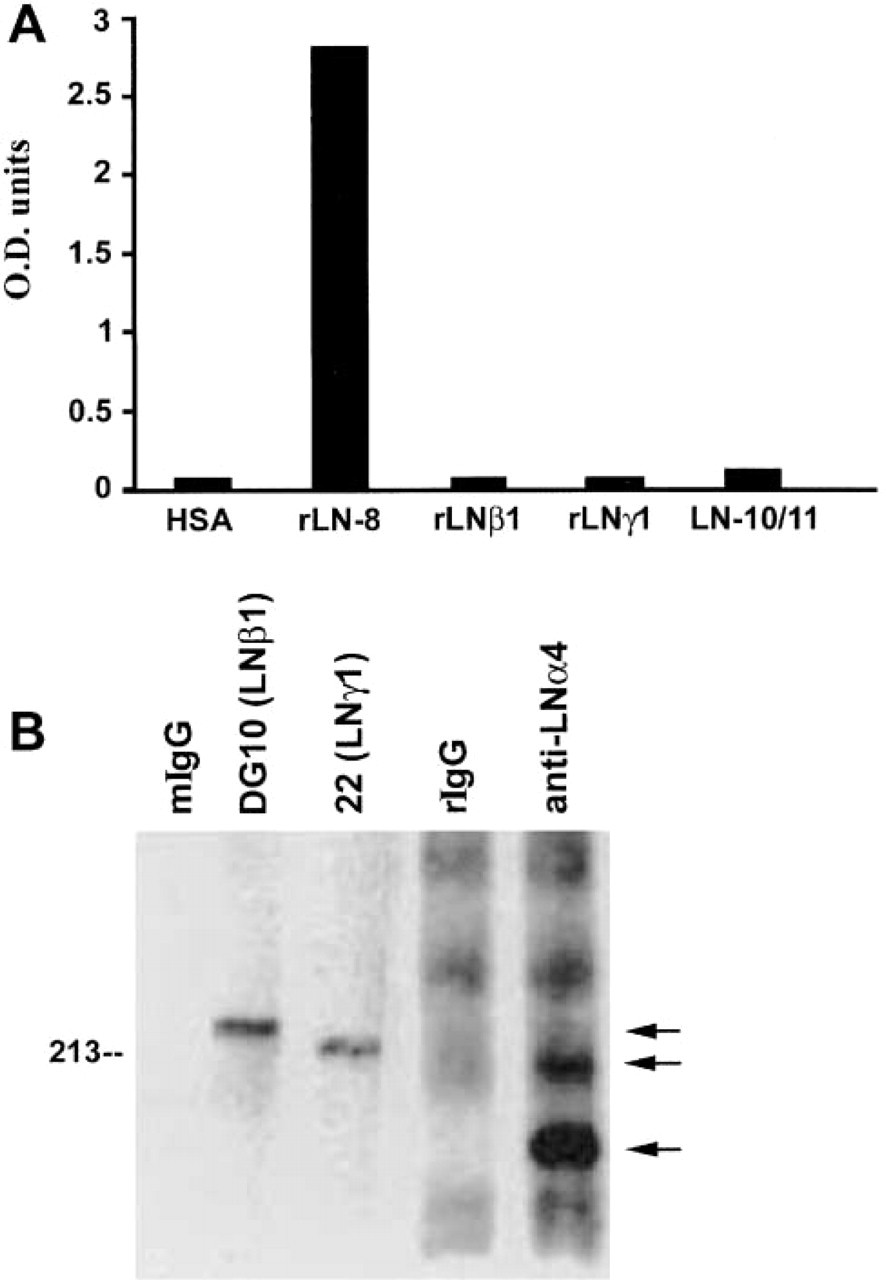
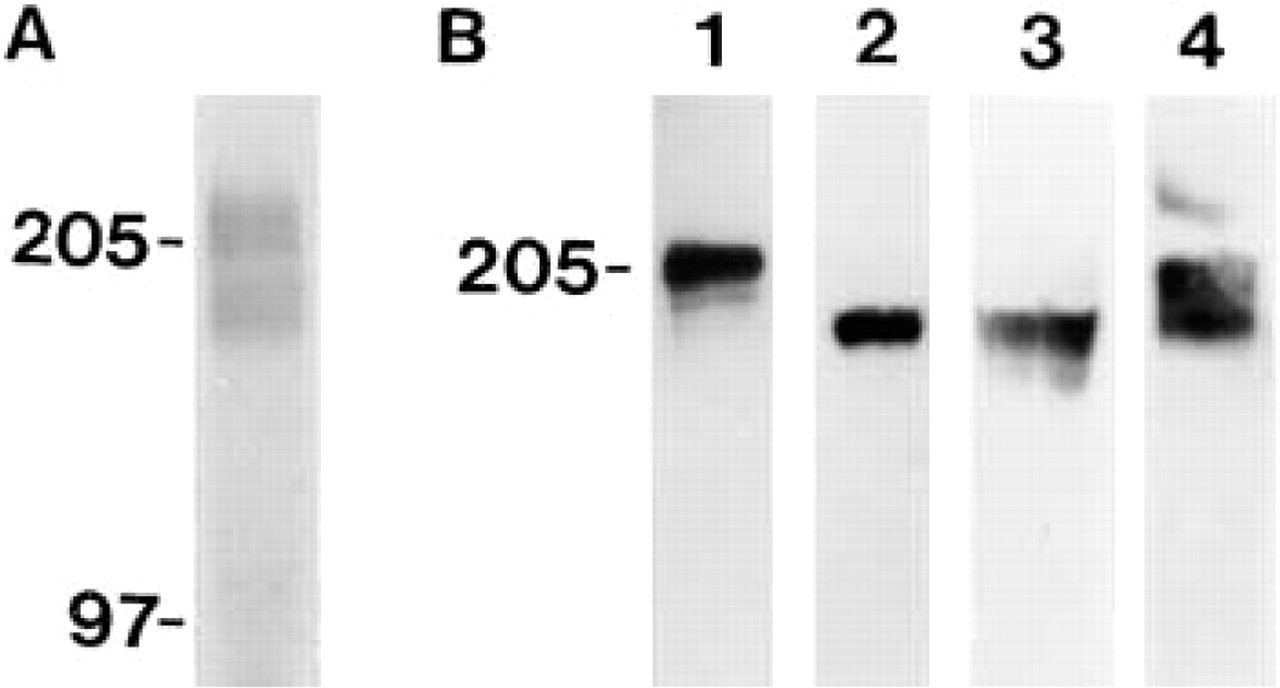
Recognition of laminin-8 (α4β1γ1) by MAb FC10. (
Cell Culture, Immunoprecipitation, and Western Blotting
Immunoprecipitation of laminin from platelet lysates was performed as previously described (Geberhiwot et al. 1999). The immunoprecipitates were analyzed by Western blotting with antibodies to laminin β1-, γ1-, and α4-chains. MAb FC10 was used as undiluted supernatant for immunoprecipitation. In Western blots, protein samples were run in SDS-PAGE using 6% polyacrylamide gels under reducing conditions, and filters were blocked with 0.1% Tween-20 and 5% dry milk in PBS. Peroxidase-linked anti-mouse and anti-rabbit immunoglobulins were used as secondary antibodies (DAKO) and bound antibodies were visualized using enhanced chemiluminescence (Amersham Biosciences; Uppsala, Sweden).
Human pulmonary artery endothelial (HPAE) cells (Promo-Cell Bioscience Alive; Heidelberg, Germany) were cultured in endothelial cell growth medium-2 (EGM-2; Clonetics, Walkersville, MD). For immunoprecipitation experiments, the culture medium of HPAE cells was first cleared by centrifugation and supplemented with Triton X-100 (0.5%) and normal mouse serum. The supernatant was then preabsorbed with GammaBind Plus Sepharose (Amersham Biosciences) and subsequently applied to GammaBind Plus Sepharose beads preincubated with MAbs FC10, DG10, and BC7. Finally, the bound proteins were eluted with Laemmli's sample buffer and subjected to SDS-PAGE in 6.5% gels under reducing conditions. For Western blotting the samples were transferred onto nitrocellulose filters, after which the filters were blocked using 5% dry milk in PBS. The sheets were subsequently exposed to rabbit antiserum to Ln α4-chain. Immunoreactive bands were visualized using either peroxidase-coupled goat anti-rabbit IgG (Vectastain Elite ABC kit; Vector Laboratories) with diaminobenzidine tetrahydrochloride as substrate (DAB; Sigma) or HRP-coupled rabbit immunoglobulins (DAKO) with SuperSignal West Pico Chemiluminescent substrate (Pierce; Rockford, IL).
For SDS-PAGE and Western blotting, adult human kidney and lung tissues were used to produce cell-free extracellular matrices. The tissues were exposed to 0.5% sodium deoxycholate (Sigma) in 10 mM Tris-HCl, 150 mM NaCl, and 1 mM PMSF (pH 8.0) at 0C three times for 10 min, followed by three washes in 2 mM Tris-HCl, 150 mM NaCl, and 1 mM PMSF (pH 8.0) at 0C (Hedman et al. 1979). Recombinant human Ln-8 (unpublished data) containing human Ln α4-, β1-, and γ1-chains was produced in a mammalian expression system as previously described for recombinant hybrid Ln-8 (Kortesmaa et al. 2000).
Distribution of Ln α4-chain in fetal and adult human tissues by immunohistochemistry a
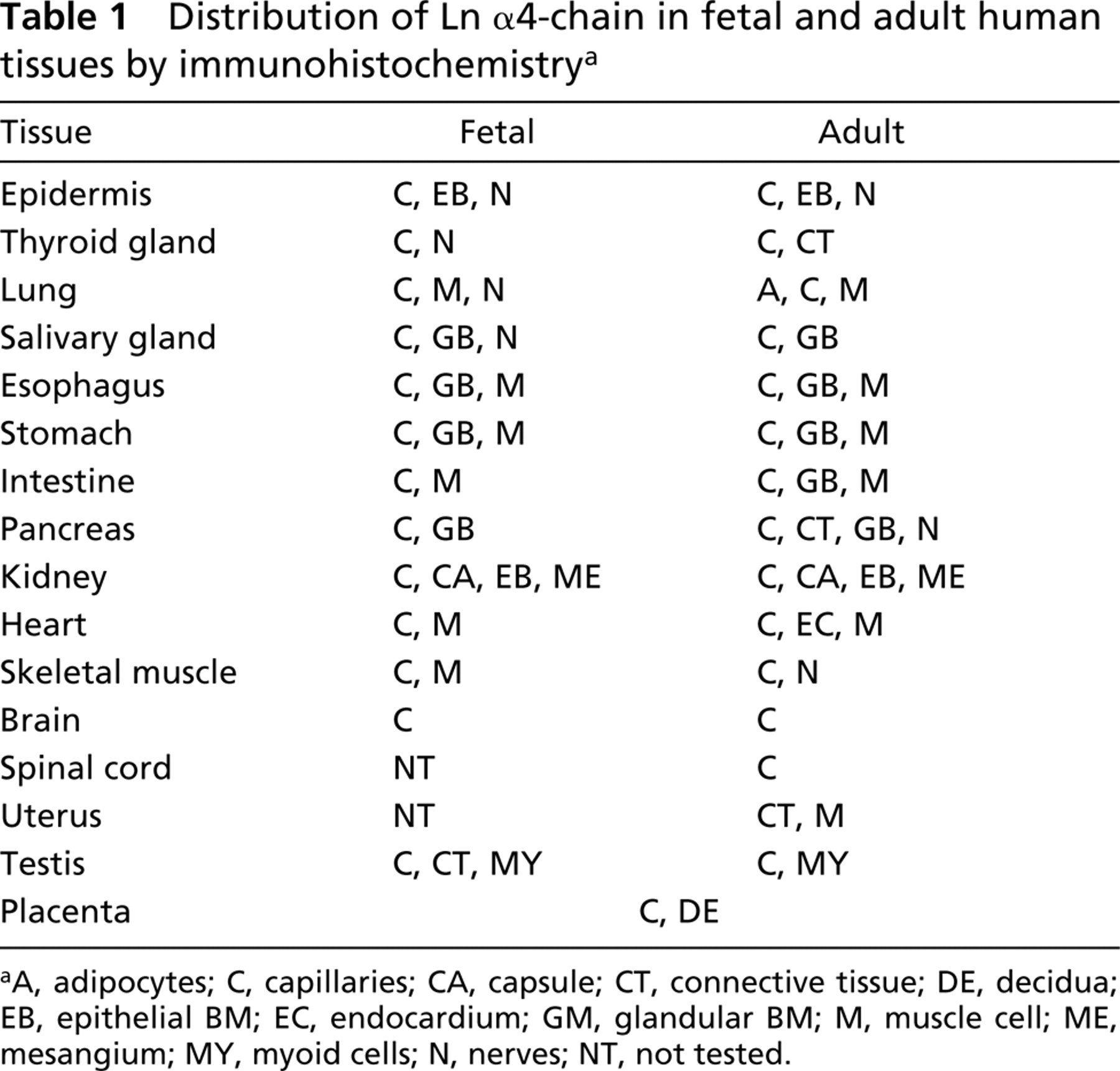
aA, adipocytes; C, capillaries; CA, capsule; CT, connective tissue; DE, decidua; EB, epithelial BM; EC, endocardium; GM, glandular BM; M, muscle cell; ME, mesangium; MY, myoid cells; N, nerves; NT, not tested.
Tissues
Specimens of normal human colon (n=2), endometrium (n=2), esophagus (n=4), kidney (n=8), lung (n=3), pancreas (n=9), salivary gland (n=1), skin (n=4), small intestine (n=4), stomach (n=6), testis (n=2), and thyroid gland (n=2) were obtained from surgical operations at Jorvi Hospital (Espoo, Finland) and from the second Department of Surgery (Helsinki University Central Hospital; Helsinki, Finland). Specimens of normal human brain (n=1) and spinal cord (n=1) were obtained at autopsy at the Department of Pathology, University of Oulu (Oulu, Finland). Biopsies of adult human skeletal muscle (n=3) were obtained after informed consent at the Department of Surgery, Umeå University Hospital (Umeå, Sweden). Specimens of adult heart (n=3) and vessels (n=6) were obtained in autopsy at the Department of Forensic Medicine (Umeå University Hospital) and specimens of fetal heart (n=3) and fetal human salivary gland (n=1) were from legal abortions performed at Umeå University Hospital. The Ethical Committee at the Umeå University approved collection of autopsy specimens and aborted material.
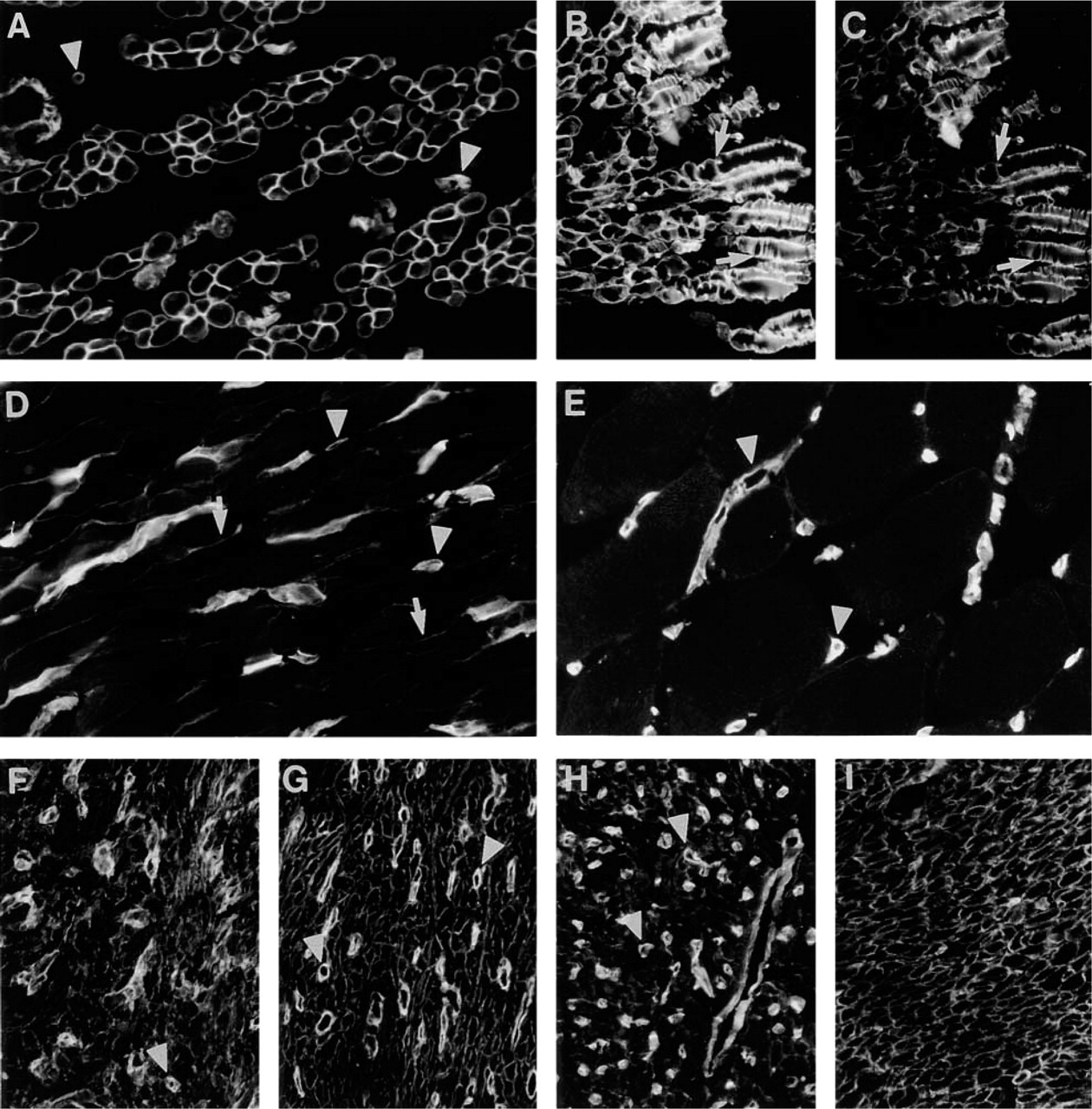
Immunoreactivity for MAb FC10 in skeletal (
Specimens (n=1–4) from 16- to 23-week fetal brain, esophagus, kidney, lung, pancreas, peripheral nerve, skin, skeletal muscle, small intestine, stomach, testis, and thyroid gland were obtained from fetuses legally aborted due to severe maternal complications or from spontaneous abortions resulting from rupture of fetal membranes at the Department of Obstetrics and Gynaecology (Helsinki University Central Hospital) or Jorvi Hospital. Specimens of 9- to 40-week placenta (n = 7) were obtained from Jorvi Hospital. The tissues were frozen in liquid nitrogen and stored at −80C until use. Histological evaluation of the specimens was done on sections stained with hematoxylin–eosin.
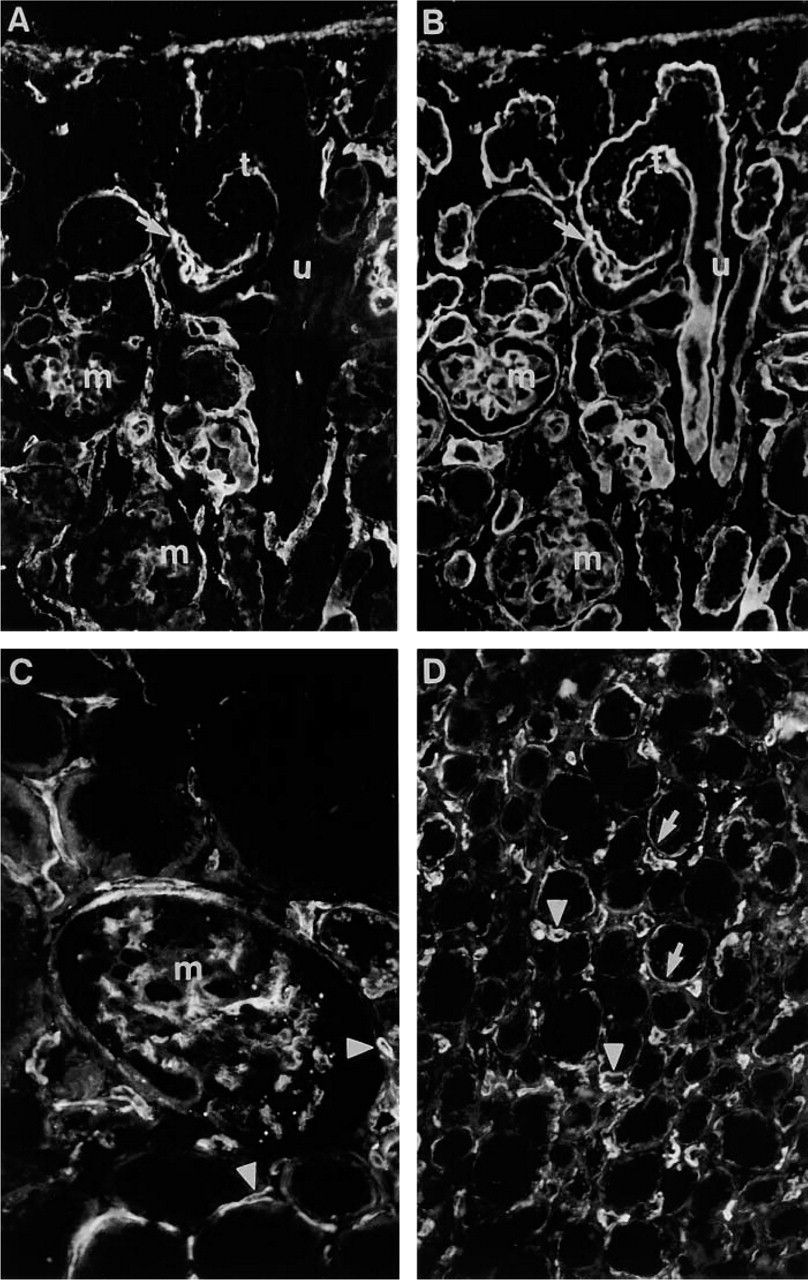
Immunoreactivity for Ln α4-chain in fetal (16-wk) and adult kidney. (
Immunohistochemistry
For indirect IHC, 6-μm tissue sections were fixed in acetone at −20C for 10 min. HPAE cells were grown on coverslips and fixed in methanol at −20C for 10 min. The specimens were first exposed to MAb FC10, followed by fluorescein isothiocyanate (FITC)-coupled goat anti-mouse IgG (Jackson Immunoresearch; West Grove, PA). For double labeling experiments the specimens were further exposed to rat MAb or polyclonal antiserum, followed by tetramethylrhodamine isothiocyanate (TRITC)-coupled goat anti-rat IgG or TRITC-coupled goat anti-rabbit IgG (both from Jackson Immunoresearch), respectively. The specimens were embedded in sodium veronal:glycerol buffer (1:1, pH 8.4) and examined with a Leica Aristoplan microscope equipped with appropriate filters. For light microscopy, the sections were exposed to MAbs for 30 min. Endogenous alkaline phosphatase was blocked with 1 M levamisole (Sigma). The bound antibodies were visualized with the alkaline phosphatase–anti-alkaline phosphatase method (APAAP mouse monoclonal; DAKO). The sections were counterstained with Mayer's hematoxylin (Merck; Darmstadt, Germany) and mounted in Eukitt (O. Kindler; Freiburg, Germany). For negative controls, the primary antibodies were omitted in both indirect immunofluorescence and APAAP.
Results
Specificity of the MAb to Ln α4-chain
Specificity of MAb FC10 was first determined by EIA (Figure 1A). The antibody strongly reacted with recombinant hybrid Ln-8 but not with Ln-10/11 (α5β1γ1/α5β2γ1) or with recombinant Ln β1- or Ln γ1-chains. MAbs to either the Ln β1- or the Ln γ1-chain recognized the laminin isoforms and the corresponding isolated chains, whereas MAb 4C7 to the Ln α5-chain reacted exclusively with Ln-10/11 (data not shown). In Western blots, MAb FC10 was unable to recognize platelet or recombinant hybrid Ln-8 under reducing or non-reducing conditions (data not shown). However, the antibody efficiently immunoprecipitated laminin-8 from platelet lysates because the characteristic laminin β1- (Mr 230,000), γ1- (Mr 220,000), and α4- (Mr 180,000 and 200,000) chains were detected in the immunoprecipitated material by Western blotting (Figure 1B). These bands were not observed when mouse IgG control was used for immunoprecipitation (data not shown).
Immunoreactivity for Ln α4-chain in Skeletal, Heart, and Visceral Smooth Muscle
All the IHC results of fetal and adult human tissues are summarized in Table 1.
In early fetal (16-week) skeletal muscle, immunoreactivity for Ln α4-chain was detected in BMs of myotubes (Figure 2A). Longitudinal sections of myotubes displayed a striated reaction pattern (Figure 2B) that closely co-localized with that of Ln α2-chain (Figure 2C). A similar striated immunoreactivity was also detected with MAbs to β1- and γ1-chains and with polyclonal antiserum to EHS laminin (results not shown). In 39-week skeletal muscle, only weak immunoreactivity for Ln α4-chain was detected in BMs of the muscle cells (Figure 2D). In adult skeletal muscle, immunoreactivity could be detected only in BMs of capillaries (Figure 2E).
In 14-week (Figure 2F) and 20-week (Figure 2G) fetal heart, immunoreactivity for Ln α4-chain was confined to BMs of myotubes. In adult myocardium (Figure 2H), distinct immunoreactivity was also found in the BMs of muscle cells, albeit weaker than in fetal heart. Some of the adult specimens included endocardium, and immunoreactivity was detected at the endothelial BM (not shown). In fetal and adult visceral smooth muscle from various sites, BMs of smooth muscle cells were immunoreactive, as shown in the external muscular layer of stomach (Figure 2I).
Immunoreactivity for Ln α4-chain in Developing and Adult Kidney
In fetal metanephros (16-week), immunoreactivity for Ln α4-chain was confined to BMs of developing glomerulus and evolving tubules (Figure 3A). BM of ureter bud (u in Figure 3A), identified by double labeling with rabbit antiserum to EHS laminin (Figure 3B), was not immunoreactive for Ln α4-chain. Before the comma-shaped body stage, BMs of developing epithelial aggregates showed faint and inconsistent immunoreactivity. At the comma-shaped body stage, however, prominent immunoreactivity was detected in the glomerular cleft containing the invading capillaries (results not shown). At the S-shaped body stage the glomerular cleft area and BM of the developing proximal tubule part were immunoreactive (Figure 3A). In the capillary loop-stage glomeruli, intraglomerular mesangium was clearly reactive for Ln α4-chain (Figure 3A), whereas no reactivity was found in glomerular BM, as shown by double labeling with rabbit antiserum to EHS laminin (Figure 3B). Bowman's capsule was inconsistently reactive (Figure 3A). In medulla of fetal kidney, weak immunoreactivity was detected in BMs of some tubules (not shown).
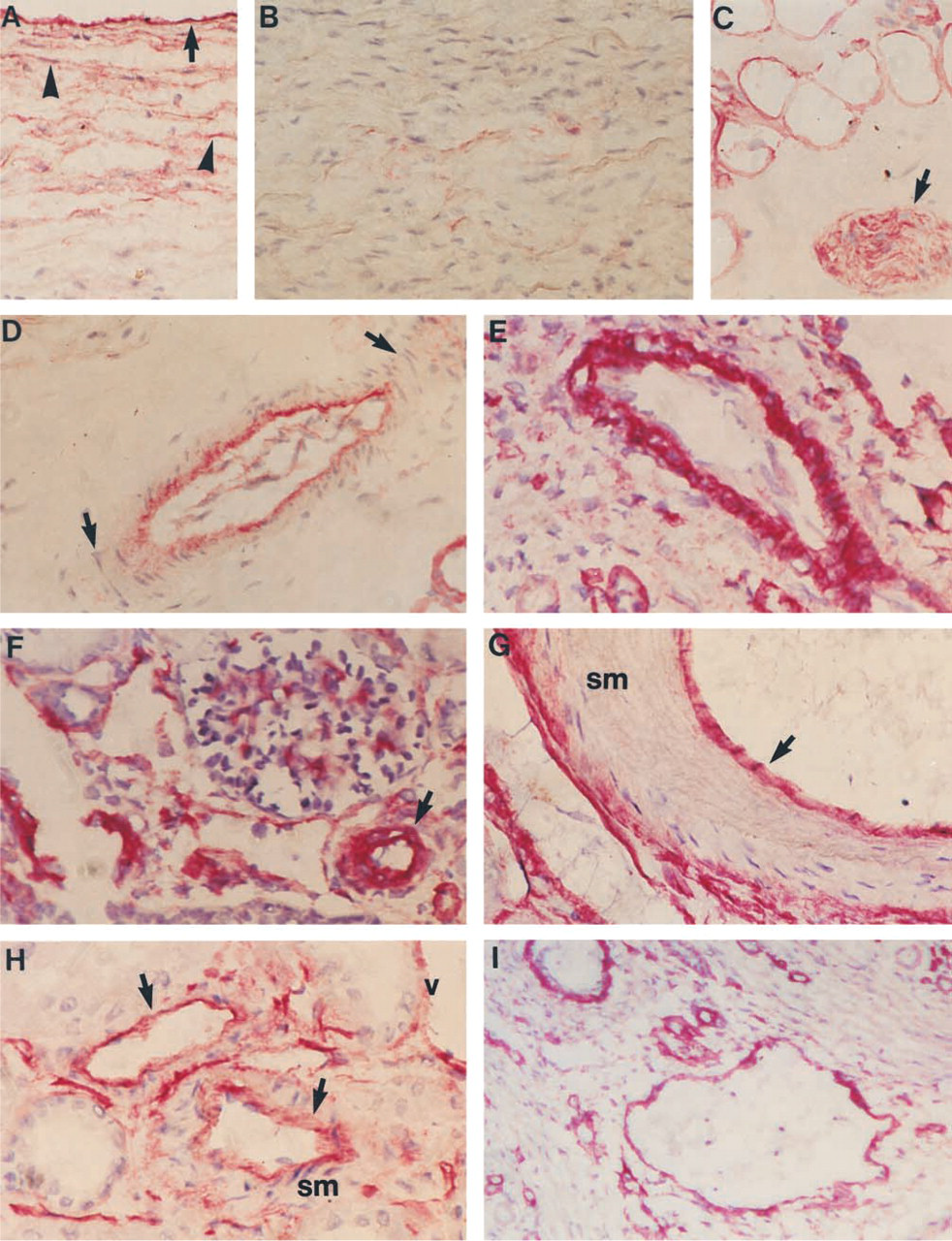
Immunoreactivity for Ln α4-chain in fetal and adult vasculature. (
In adult kidney the mesangium was immunoreactive for Ln α4-chain, whereas no reactivity was found in the glomerular BM (Figure 3C). Inconsistent immunoreactivity was found in BM of Bowman's capsule. In medulla of adult kidney, BMs of some tubules were immunoreactive (Figure 3D). The kidney capsule was reactive in both fetal and adult kidney (not shown).
Immunoreactivity for Ln α4-chain in Vasculature
Localization of Ln α4-chain in elastic arteries is illustrated in adult pulmonary trunk, whereas that of muscular arteries, arterioles and veins is illustrated in fetal and adult kidney. BMs of capillaries were consistently immunoreactive, and this is mentioned only in Table 1 and the figure legends. Comparable results were obtained in other tissues.
Endothelial BMs of fetal and adult blood vessels were consistently immunoreactive for Ln α4-chain (adult pulmonary trunk; Figure 4A). In adult pulmonary trunk, scattered fibrillar immunoreactivity was detected among the smooth muscle cells. This reactivity was strongest in the vicinity of endothelium (Figure 4A) and was lacking from BMs of smooth muscle cells of the media (Figure 4B). BMs of adipocytes and nerves were distinctly immunoreactive in the adventitial layer of pulmonary trunk (Figure 4C), as were the adipocytes and nerves in all other locations found. Endothelial BMs of vasa vasorum were reactive, but no reactivity was found in BMs of smooth muscle cells (Figure 4D). In arteries (Figure 4E) and arterioles (Figure 4F) of fetal kidney, strong immunoreactivity was found in the BMs of smooth muscle cells. In adult kidney, only inconsistent immunoreactivity was found in the smooth muscle layers of arteries (Figure 4G) and arterioles (Figure 4H). In adult (Figure 4H) and fetal (Figure 4I) veins, endothelial BMs were constantly immunoreactive.
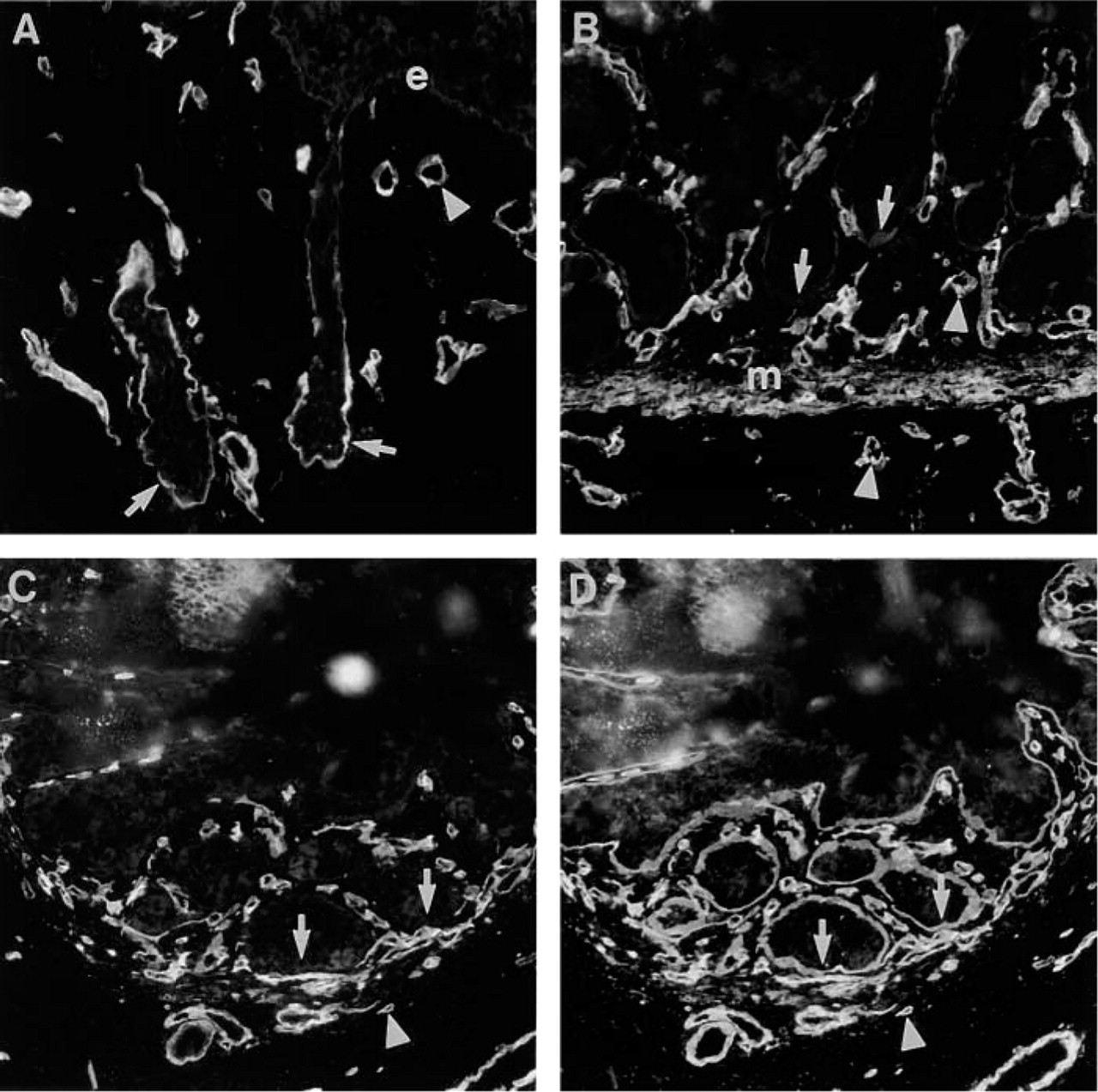
Distribution of Ln α4-chain in fetal gastrointestinal tract. (
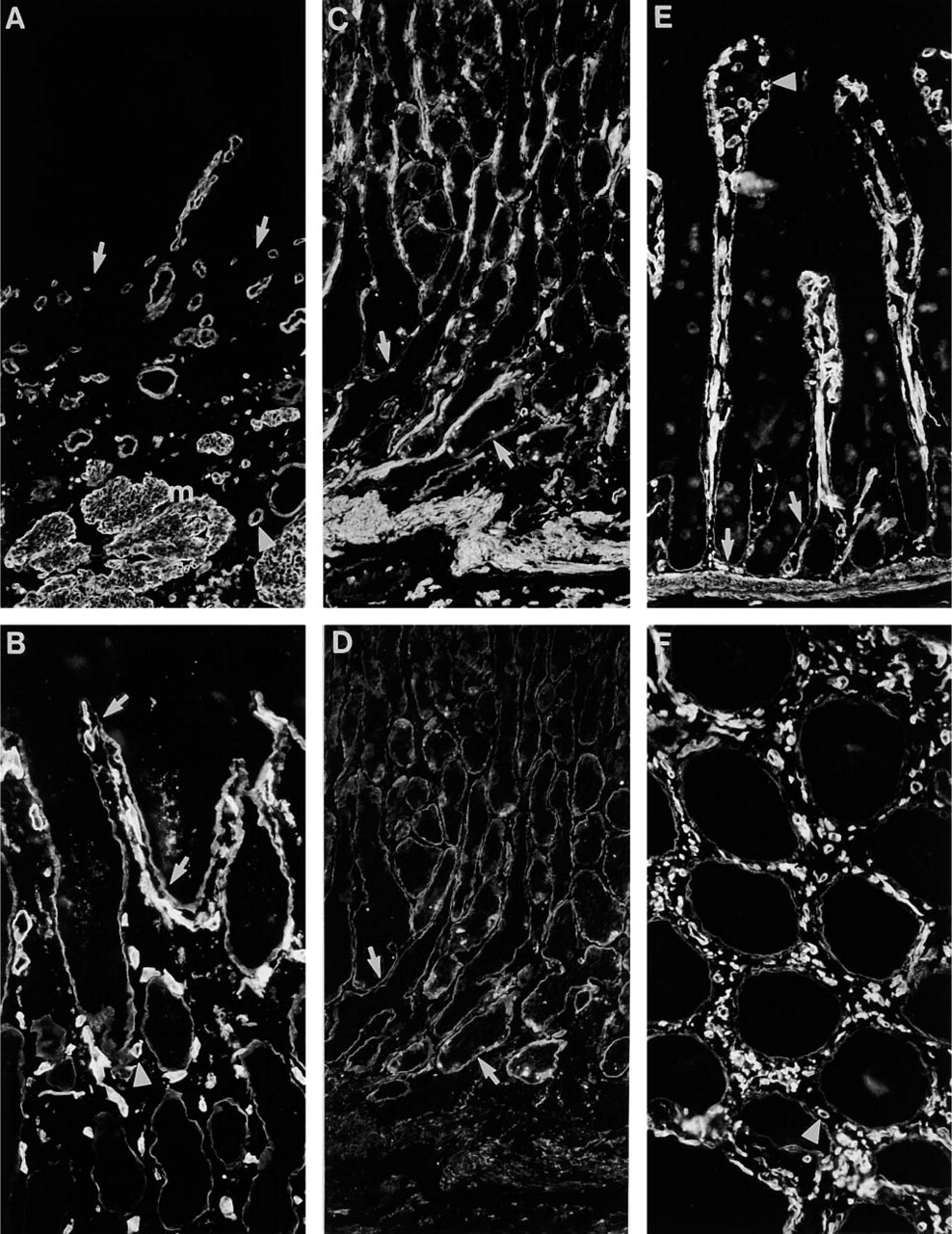
Distribution of Ln α4-chain in adult gastrointestinal tract. (
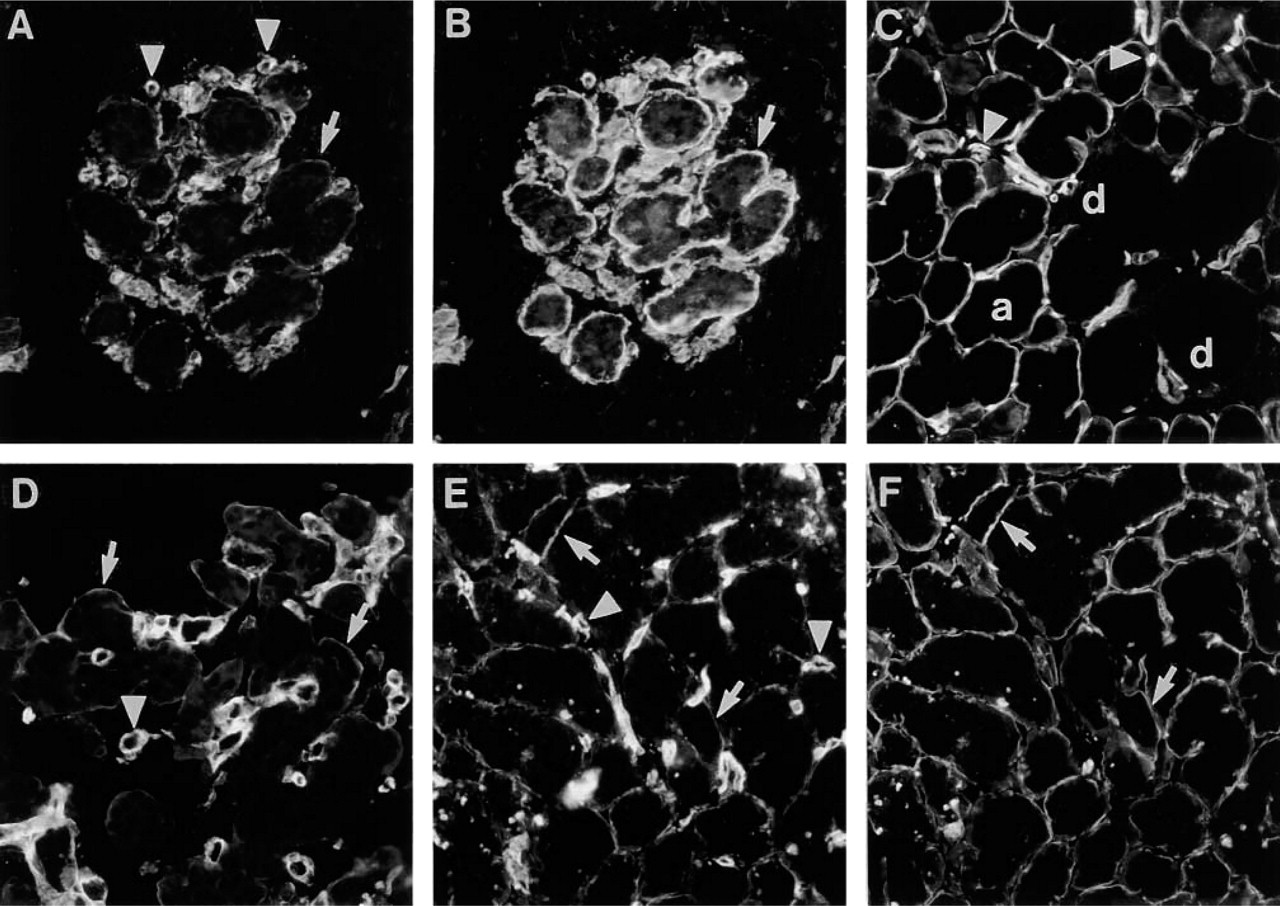
Distribution of Ln α4-chain in glands of gastrointestinal tract. (
Ln α4-chain in Fetal and Adult Alimentary Tract
In fetal esophagus (16-week), BM of surface epithelium was not immunoreactive for Ln α4-chain. Instead, immunoreactivity was found in BMs of developing esophageal submucosal glands (Figure 5A). In fetal stomach (16-week), faint immunoreactivity was found in BMs of developing gastric glands (Figure 5B). In fetal small intestine (19-week), neither BM of surface epithelium nor crypts were immunoreactive (Figure 5C; 5D shows BMs identified by double labeling with rabbit antiserum to EHS laminin). In all parts of fetal gastrointestinal tract, BMs of smooth muscle cells were immunoreactive.
Immunoreactivity for Ln α4-chain in adult esophagus was identical to that found in fetal tissues (Figure 6A). No reactivity was found in BM of surface epithelium, whereas BMs of submucosal glands (not shown) were immunoreactive. In the antrum of adult stomach, immunoreactivity was found in BMs of gastric glands (Figure 6B). In corpus of stomach, reactivity was found in the lower part of the gland (Figure 6C), in partial co-distribution with reactivity for Ln α2-chain (Figure 6D). In adult small intestine, immunoreactivity for Ln α4-chain was detected in BMs of crypts (Figure 6E), whereas BMs of Brunner's glands lacked reactivity (not shown). In colon, BMs of glands were reactive (Figure 6F). In all parts of adult gastrointestinal tract, BMs of smooth muscle cells showed prominent reactivity.
In the fetal salivary gland (22-week), immunoreactivity for Ln α4-chain was confined to the BM of acini (Figure 7A), as confirmed by double labeling with EHS laminin antiserum (Figure 7B). In adult salivary gland the immunoreactivity was also confined to BMs of acini, whereas BMs of ducts lacked immunoreactivity (Figure 7C). In fetal (Figure 7D; 19-week) and adult (Figure 7E) pancreas, faint immunoreactivity for Ln α4-chain was detected in the BM of acini, in colocalization with reactivity for Ln α2-chain (Figure 7F). In adult pancreas, connective tissue septa were immunoreactive (not shown).
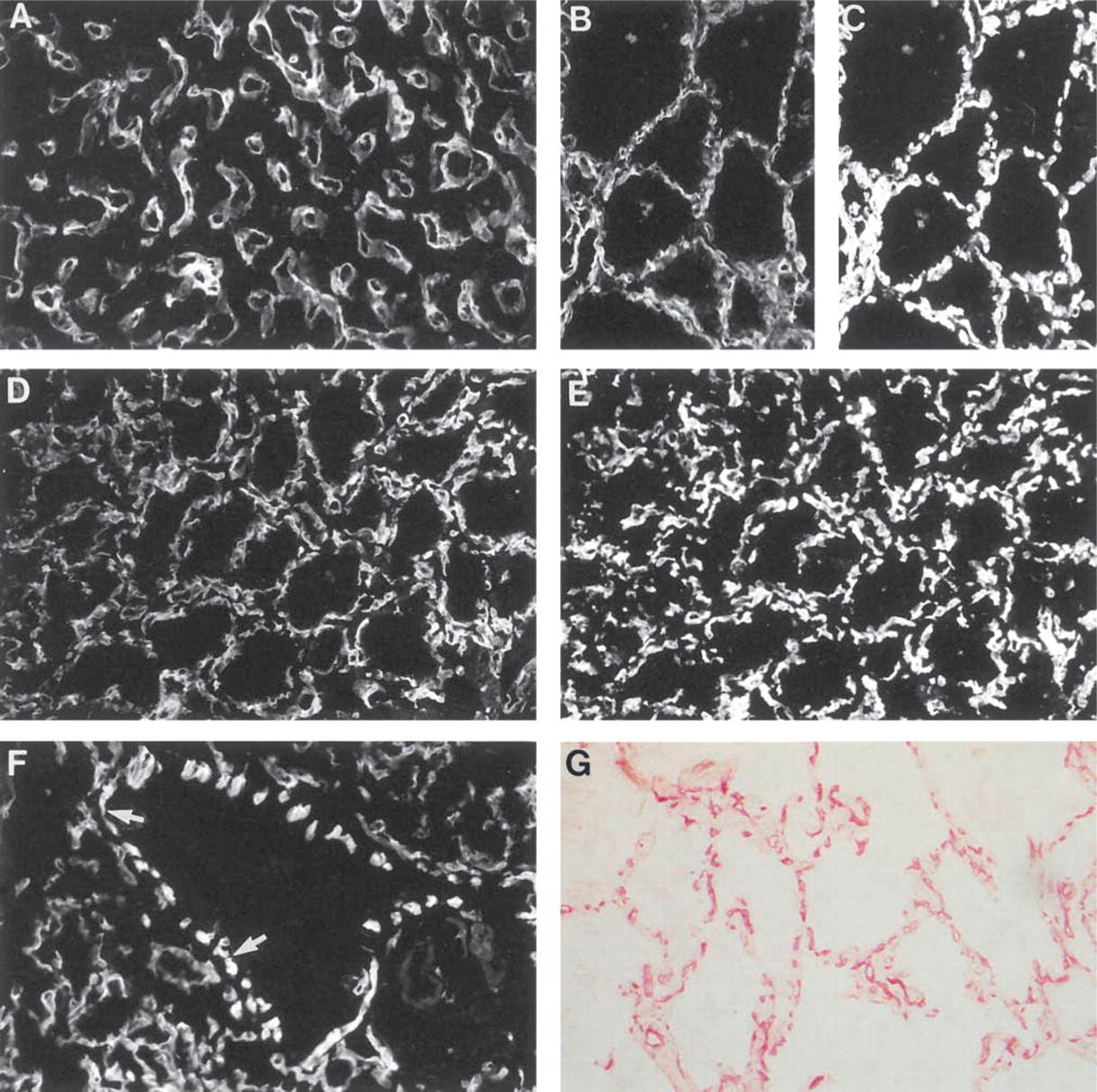
Immunoreactivity for Ln α4-chain in thyroid gland and lungs. (
Immunoreactivity for Ln α4-chain in Other Fetal and Adult Tissues
In fetal (16-week; Figure 8A) and adult (Figure 8B) thyroid gland, immunoreactivity for Ln α4-chain was mostly restricted to BMs of capillaries, as confirmed for adult tissue by double labeling with TRITC–UEA-I (Figure 8C). In the adult gland, some scattered immunoreactivity was found in the interstitium between the follicles and capillaries (Figure 8B). In fetal lung at the late pseudoglandular (19-week; not shown) and early canalicular stages (22-week; Figure 8D), strong immunoreactivity was detected in capillary BMs around budding bronchial tubules and around developing respiratory ducts, as confirmed by double labeling with TRITC–UEA-I (Figure 8E). BM of bronchial epithelium was not immunoreactive, whereas the BMs of smooth muscle cells in the bronchial wall were immunoreactive (Figure 8F). In adult lung the BMs of capillaries were clearly immunoreactive (Figure 8G). No reactivity was found in BM of bronchial epithelium, whereas BMs of smooth muscle cells were immunoreactive (not shown).
In fetal (16-week) testis, faint immunoreactivity for Ln α4-chain was found in the myoid cell layer around the seminiferous tubules (Figure 9A). Scattered immunoreactivity was also found in the interstitium. In adult testis, strong immunoreactivity was found in the myoid cell layer (Figure 9B). In fetal (16-week; Figure 9C) and adult (Figure 9D) skin, reactivity was found in the region of epithelial BM. No reactivity was detected in fetal or adult dermis, with the exception of BMs of nerves and capillaries.
In fetal brain (16-week), immunoreactivity was found in BMs of capillaries (not shown). In adult brain (Figure 10A) and spinal cord (Figure 10B), immunoreactivity was found in BMs of capillaries. In larger vessels, immunoreactivity for Ln α1-chain (red, Figure 10B) surrounded that for Ln α4-chain (green, Figure 10B). Smaller Ln α4-chain-reactive capillaries were not reactive for Ln α1-chain. In fetal ischiadic nerve (16-week; Figure 10C), the BMs of axons were reactive for Ln α4-chain. The reactivity co-localized with that for Ln α2-chain (Figure 10D). In addition, fragmentary immunoreactivity for Ln α4-chain was found in perineurium (Figure 10C) and epineurium (not shown). In adult peripheral nerve, BMs of axons and epineurium were strongly immunoreactive (nerve from pancreas, Figure 10E).
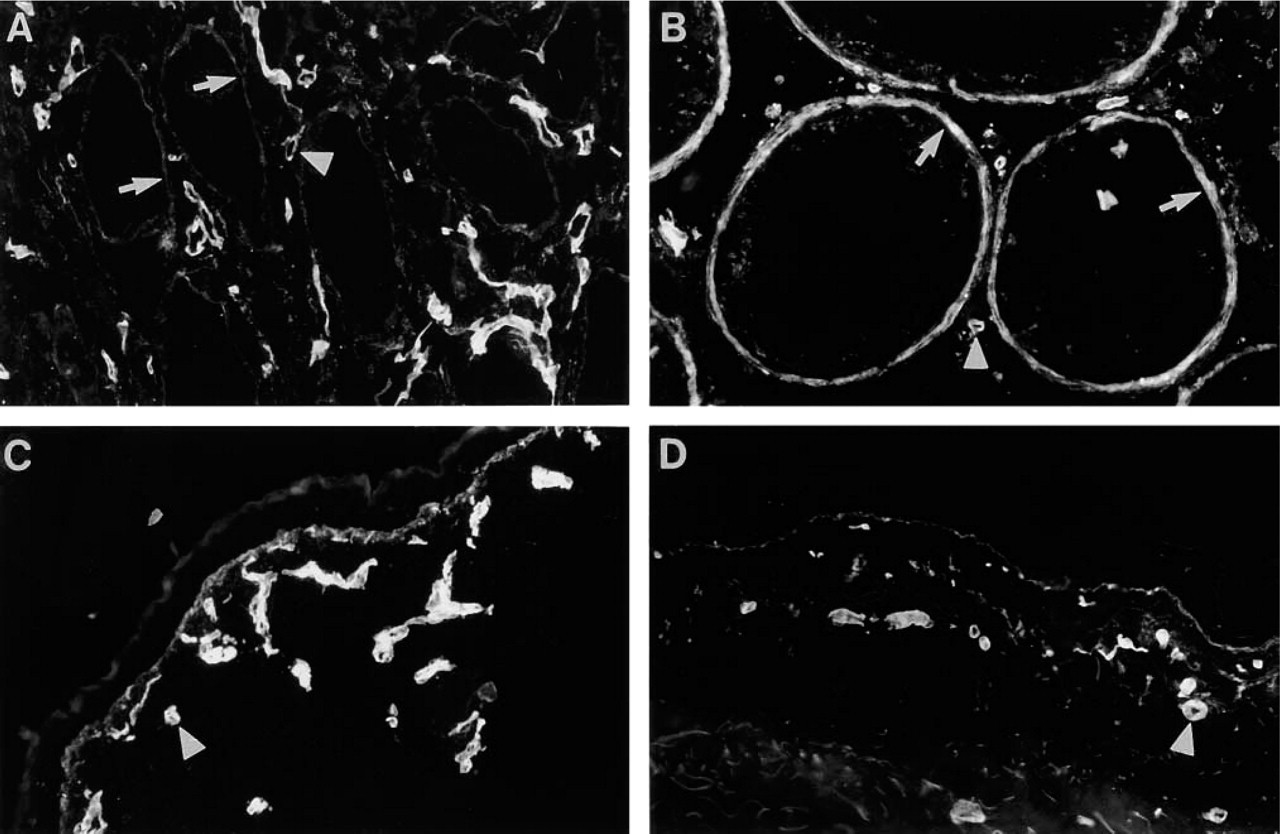
Distribution of Ln α4-chain in testis and skin. (
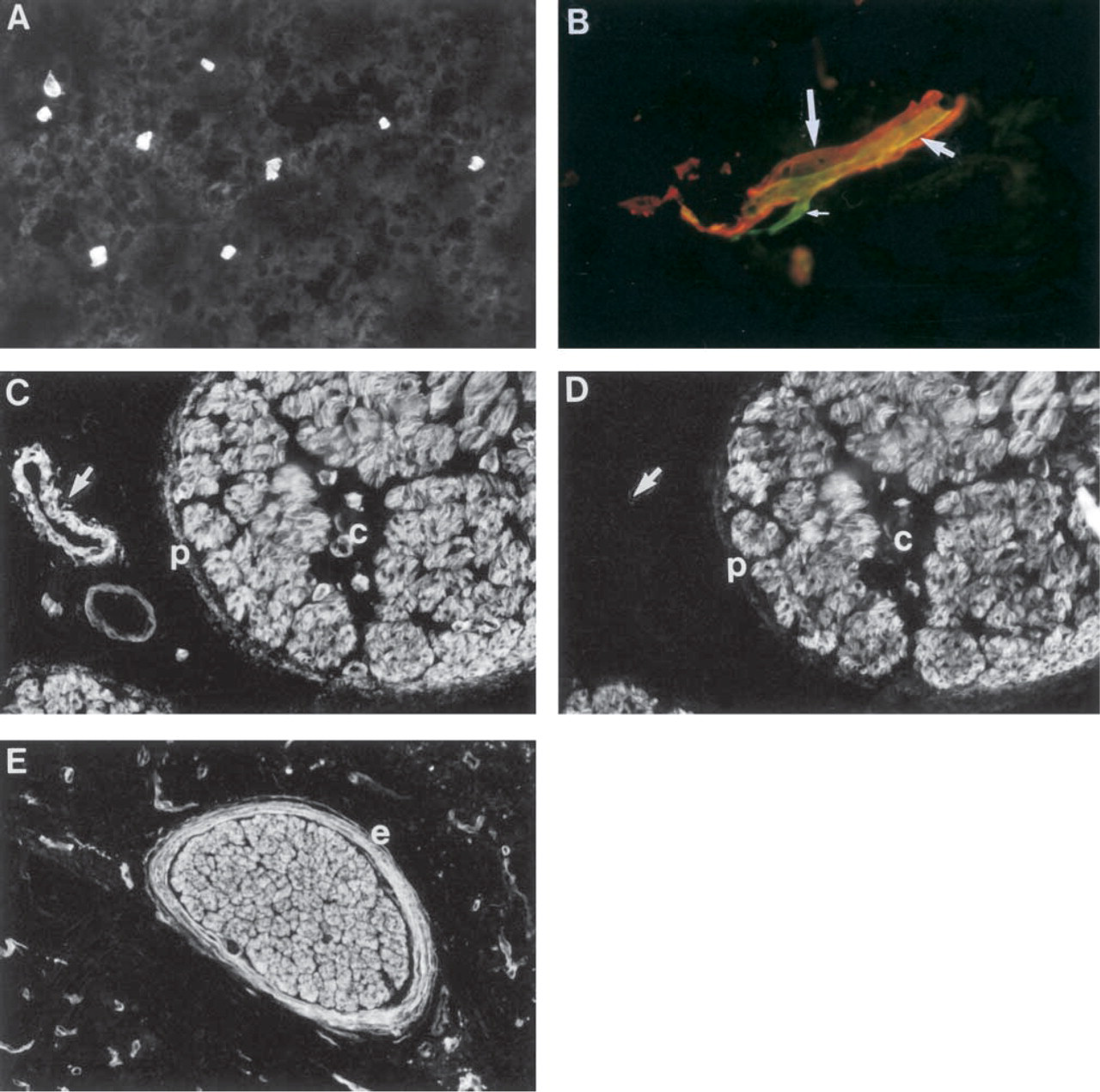
Distribution of Ln α4-chain in nervous system. (
In adult human endometrium, fibrillar immunoreactivity for Ln α4-chain was clearly found in the connective tissue stroma, whereas the epithelial BMs of endometrial glands from various stages of proliferation were not immunoreactive (Figure 11A). BMs of decidual cells showed immunoreaction (Figure 11B). In placental villi (9–40-week), immunoreactivity for Ln α4-chain was found in the BMs of capillaries (20-week; Figure 11C), while prominent reactivity for Ln α2-chain was also detected in the BM of trophoblast (Figure 11D).
Ln-8 Is Produced by Endothelial Cells and Is Found in Cell-free Extracellular Matrices
We utilized MAb FC10 to study the production of Ln α4-chain by endothelial cells. Culture medium of human pulmonary artery endothelial cells was precipitated with MAb FC10 and analyzed by Western blotting with affinity-purified rabbit antibodies to Ln α4-chain. We had to use an indirect immunoprecipitation with these cells because in radioactive immunoprecipitation of HPAE cells a broad Mr 200,000 band was unspecifically bound. In Western blotting, two immunoreactive bands of c. Mr 180,000 and 200,000 were detected (Figure 12A). The Ln α4-chain could similarly be detected by Western blotting when HPAEC medium was immunoprecipitated with MAbs to Ln β1-(not shown) and γ1-chains, suggesting that these cells produce Ln-8.
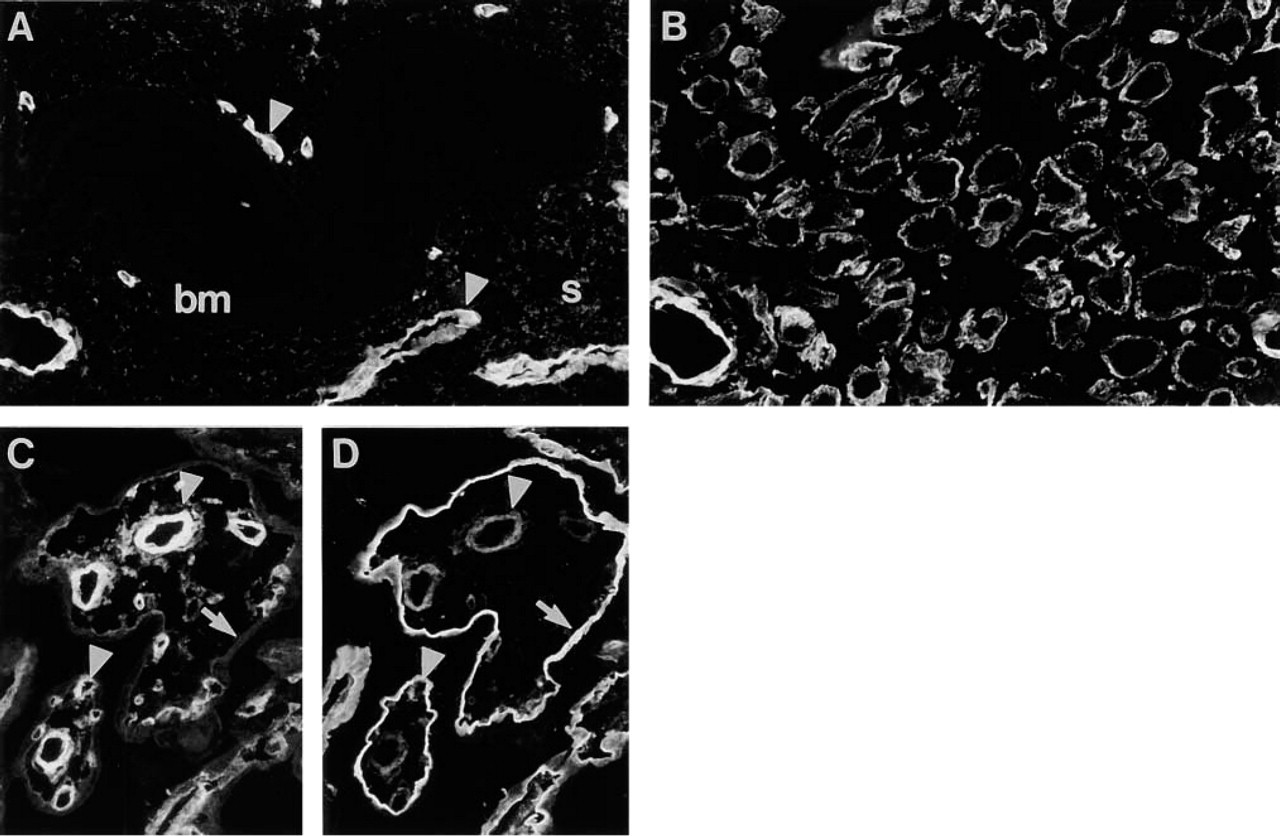
Distribution of Ln α4-chain in uterus and placenta. (
We then compared recombinant human Ln-8 and cell-free extracellular matrices of adult human kidney and lung by Western blotting with antibody to Ln α4-chain. For recombinant human Ln-8, a major band of c. Mr 200,000 was detected (Figure 12B, Lane 1). In cell-free extracellular matrices of kidney (Figure 12B, Lane 2) and lung (Figure 12B, Lane 3), a smaller band of c. Mr 180,000 was detected, which co-migrated with the lower band detected in the culture medium of HPAE cells (Figure 12B, Lane 4).
We also studied whether endothelial cells deposit Ln α4-chain into the extracellular matrix. Immunoreactivity for Ln α4-chain was found in the extracellular matrix of HPAE cells (Figure 13A). A cell surface-confined fluorescence was seen for Int α2-, Int α3-, Int α6-, and Int β1-subunits, but not for Int α1- or β4-subunits (not shown). In double immunolabeling, reactivity for Ln α4-chain was found to partially co-localize with that for Int α6-subunit (Figure 13B), suggesting that the deposition of Ln-8 and interactions of HPAE cells with Ln-8 are mediated by integrin α6β1.
Discussion
The primary purpose of the present study was to elucidate the distribution of Ln α4-chain in developing and adult human tissues by IHC. To achieve this, we raised MAb FC10 and showed that it reacts specifically with recombinant hybrid Ln-8 and with Ln-8 from human platelets. We were able to exclude reactivity with other Ln isoforms (Ln-10/11) and with isolated Ln β1- and Ln γ1-chains. Because other Ln α4-chain-containing laminin isoforms were not available, we could not confirm whether MAb FC10 reacts with all Ln α4-chain containing laminins or only with Ln-8 trimer. However, because this novel MAb also reacts with a recombinant Ln-8 containing mouse Ln β1-chain and human Ln α4- and Ln γ1-chains, we find the latter possibility unlikely and consider that the immunoreaction patterns reflect the tissue distribution of Ln α4-chain.
(
Genetic alterations of laminin molecules that are found in human skeletal muscle can cause disease (Gullberg et al. 1999). Gene-targeting work in mice and genetic diseases in human have generated valuable information of functions of Ln α2-, α5-, β2-, and γ1-chains in skeletal muscle (Gullberg et al. 1999). In addition, Ln α4-chain was found in Ln α2-chain-deficient muscle of congenital muscular dystrophy patients (Patton et al. 1999). This has raised the question of whether laminin Ln α4-chain could functionally substitute for the deficient Ln α2-chain (Gullberg et al. 1999). Previous IHC studies have detected Ln α4-chain in BMs of skeletal muscle cells in fetal mice, whereas it was confined to BMs of capillaries in adult mouse skeletal muscle (Iivanainen et al. 1997; Patton et al. 1999; Ringelmann et al. 1999; Talts et al. 2000). Ln α4-chain mRNA has also been found in human skeletal muscle (Iivanainen et al. 1995; Richards et al. 1996). Using a polyclonal antiserum against a recombinant protein fragment, Patton et al. (1999) showed Ln α4-chain immunoreactivity in BMs of capillaries in adult human skeletal muscle. Our results show that Ln α4-chain is found in BMs of myotubes in fetal human skeletal muscle but that it disappears before adulthood. As in mice, Ln α4-chain distribution appears to be developmentally regulated. We also found that in fetal skeletal muscle Ln α4-chain has a novel striated distribution that co-localizes with Ln α2-chain. Such striation probably reflects a laminin polymerization-induced receptor-cytoskeleton network that has been demonstrated in cell culture studies (Colognato et al. 1999; Lohikangas et al. 2001).
Ln α4-chain mRNA is expressed in cardiac tissue of fetal and adult mice (Liu and Mayne 1996; Frieser et al. 1997; Iivanainen et al. 1997; Miner et al. 1997; Talts et al. 2000) and in adult human heart (Iivanainen et al. 1995; Richards et al. 1996). The distribution of Ln α4-chain in mouse heart has been studied using polyclonal antisera, but the results have been contradictory (Iivanainen et al. 1997; Miner et al. 1997; Talts et al. 2000). In the present study we show that in human fetal heart Ln α4-chain is found in BMs of capillaries and in BMs of muscle cells. Although the intensity of immunoreactivity declines during development, BMs of adult cardiomyocytes retain immunoreactivity for Ln α4-chain. In this respect, the distribution of Ln α4-chain in humans differs in skeletal and cardiac muscle.
Glukhova et al. (1993) showed that developmental maturation of human arterial smooth muscle cells is accompanied by a switch in laminin β-chain content. In addition, they showed that whereas adult visceral smooth muscle contains β1- and β2-chains, the Ln β1-chain is absent from adult arterial smooth muscle. Ln α4-chain mRNA has been detected in both mouse and human smooth muscle cells (Iivanainen et al. 1995; Frieser et al. 1997). In the adult mouse, immunoreactivity for Ln α4-chain is found in BMs of vascular smooth muscle cells (Talts et al. 2000). Our results show that Ln α4-chain distribution is also different in visceral and vascular smooth muscle. BMs of human visceral smooth muscle cells are uniformly immunoreactive for Ln α4-chain, whereas BMs of vascular smooth muscle cells are immunoreactive only during the fetal period. Our results show that, in addition to smooth muscle, Ln α4-chain is found in other contractile tissues, such as glomerular mesangium and testicular myoid cell layer.
Studies on developing mouse kidney were among the first to suggest a role for Ln in tissue development (Ekblom 1981; Klein et al. 1988). At the gene expression level, several studies on mouse kidney (Richards et al. 1996; Iivanainen et al. 1997; Sorokin et al. 1997) have reported strong expression of Ln α4-chain mRNA during development but lack or a neglible level of expression at adult stage. Instead, Miner et al. (1997) found mRNA expression both in developing and adult tissues, but in adult tissues immunoreactivity for Ln α4-chain was found only in medullary capillaries. Although many of our results agree with their study, we did not detect immunoreactivity for Ln α4-chain in glomerular BMs of any stage. Instead, we found reactivity in the mesangial area of capillary loop stage glomeruli, as well as in mesangium of adult glomeruli. With the exception of glomerular capillaries, all vascular endothelial BMs were reactive in adult human kidney. Furthermore, our results show that in both developing and adult kidney BMs of medullary tubules contained Ln α4-chain. The presence of Ln α4-chain in adult human renal cortex was also confirmed by Western blotting, which revealed prominent polypeptide.
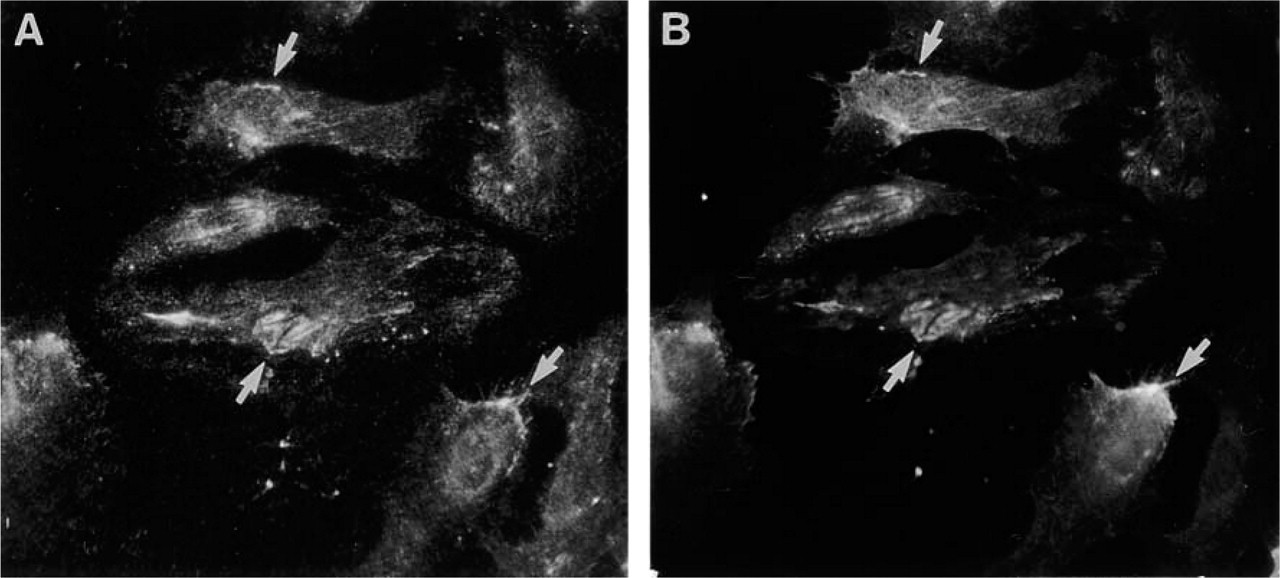
Double immunolabeling of HPAE cells with MAb FC10 (
Mouse and bovine endothelial cells express mRNA for Ln α4- and α5-chains (Tokida et al. 1990; Sorokin et al. 1994; Frieser et al. 1997). Using polyclonal antisera, Ln α4-chain has been identified in mouse endothelial BMs (Iivanainen et al. 1997; Miner et al. 1997; Talts et al. 2000; Sixt et al. 2001). Frieser et al. (1997) found that, in mouse vasculature Ln α4-chain, mRNA expression was restricted to a subset of endothelia. They showed that, in mouse hemangioma-derived endothelial cells, Ln α4-chain expression is influenced by cytokines and angiostatic steroids, and they suggested that Ln α4-chain expression is characteristic of cytokine-activated endothelia (Frieser et al. 1997; Sixt et al. 2001). Studies with polyclonal antisera have identified Ln α4-chain in BMs of capillaries in human bone marrow and brain (Gu et al. 1999; Siler et al. 2000; Ljubimova et al. 2001). Using MAbs, Spessotto et al. (2001) found Ln α4-chain in vascular structures of various human tissues but could not identify its location in blood vessel walls. On the basis of immunogold staining, it has been proposed that in mouse skeletal muscle Ln α4-chain would be more localized to interstitium of capillaries, whereas Ln α2-chain is found in BMs (Talts et al. 2000). Our results on fetal human muscle tissue, showing an exact co-localization of Ln α2- and Ln α4-chains, do not support this notion.
Our results show that immunoreactivity for Ln α4-chain is found in all endothelial BMs of developing and adult human tissues. This suggested to us an important role for Ln α4-chain in endothelial cell biology. In line with this proposal are recent findings on Ln α4–/– mice that show impaired microvessel maturation (Thyboll et al. 2002). Therefore, the second aim of our study was to elucidate the source of Ln α4-chain in endothelial BMs. We have previously shown that healthy human thrombocytes contain Ln-8 (Geberhiwot et al. 1999). In addition, fresh blood lymphocytes, monocytes, several human blood cell-derived neoplastic cell lines (Geberhiwot et al. 2000a,2001; Pedraza et al. 2000), and glioma cells (Fujiwara et al. 2001) produce Ln-8. By immunoprecipitation combined with Western blotting, we show that HPAE cells produce Ln α4-chain. Identical results were observed with human aortic endothelial cells (unpublished data). In agreement with previous studies (Geberhiwot et al. 1999,2000a,2001; Pedraza et al. 2000; Fujiwara et al. 2001) two bands of c. Mr 180,000 and 200,000 were found to be produced by HPAE cells, the higher Mr form co-migrating with the recombinant whole length Ln α4-chain. The two immunoreactive bands and variation in fragment sizes have been explained by the presence of alternative cleavage sites (Talts et al. 2000). In cell-free extracellular matrices of human kidney and lung, Ln-8 was found to migrate with the lower Mr variant, presumably a cleaved form. HPAE cells also deposited Ln α4-chain into a fibrillary extracellular matrix in co-localization with integrin α6-subunit, implying laminin polymerization-induced receptor–cytoskeleton assembly, as shown previously for Ln α1- (Virtanen et al. 2000) and Ln α2-chains (Colognato et al. 1999).
In summary, in the present study we show that Ln α4-chain is widely distributed in developing and adult human tissues. It is mainly found in BMs of mesoderm-derived tissues, such as adipose tissue and all forms of muscle. We show that this Ln chain is produced by human endothelial cells. With the exception of glomerular BM, all endothelial BMs are shown to be reactive for Ln α4-chain. In addition, Ln α4-chain is found in some epithelial BMs, but its origin in these BMs remains elusive.
Footnotes
Acknowledgements
MP was supported by Cancerfonden and the Karolinska Institutet.
We thank Ms Pipsa Kaipainen, Mr Hannu Kamppinen, Mr Reijo Karppinen, Ms Marja-Leena Piironen, Outi Rauanheimo, and Ms Hanna Wennäkoski for skillful technical assistance.
