Abstract
We examined immunocytochemical localization of the prohormone convertases, PC1 and PC2, in the thyroid gland and respiratory tract of the adult mouse using the indirect enzyme- and immunogold-labeled antibody methods for light and electron microscopy, respectively. In the thyroid gland, PC1- and/or PC2-immunoreactive cells were cuboidal, scattered in the follicular epithelium and in the interfollicular spaces. When serial sections were immunostained with anti-calcitonin, anti-PC1, anti-calcitonin-gene-related-peptide (CGRP), and anti-PC2 sera, respectively, localization of both PC1 and PC2 was restricted to the calcitonin/CGRP-producing parafollicular cells. In the respiratory tract, only PC1 immunoreactivity was observed in the basal granulated neuroendocrine cells, which were scattered in the tracheal epithelium. Consecutive sections immunostained with anti-PC1 and anti-CGRP sera showed that a subpopulation of these PC1-immunoreactive cells contained CGRP. Double immunogold electron microscopy of the thyroid parafollicular cells revealed that calcitonin- and/or CGRP-immunopositive secretory granules were also labeled with both PC1 and PC2. These findings suggest that procalcitonin is proteolytically cleaved by PC2 alone or by PC2 together with PC1, and that the proCGRP is cleaved by PC1.
Keywords
The prohormone convertases PC1 (also called PC3) and PC2 are responsible for the proteolytic cleavage of a large precursor molecule at paired basic sites to yield bioactive peptides in endocrine cells and neurons (Seidah and Chrétien 1992). Immunocytochemical (ICC) and in situ hybridization (ISH) studies have shown that these convertases are expressed in pancreatic islets (Malide et al. 1995; Itoh et al. 1996; Tanaka et al. 1996; Rawdon and Larsson 2000), in the pituitary gland (Braks et al. 1992; Marcinkiewicz et al. 1993; Kurabuchi and Tanaka 1997; Uehara et al. 2001), and in the brain (Birch et al. 1994). However, there is little information about in situ expression of these convertases in other endocrine cells.
Calcitonin is a peptide hormone that regulates the balance of serum calcium, which is secreted primarily from parafollicular cells (C-cells) in the thyroid gland (Copp 1994). Several ICC studies have demonstrated that C-cells are located not only in the thyroid but also in the parathyroid glands in some mammalian species (Kameda 1971). Moreover, several immunohistochemical (IHC) studies have detected calcitonin-gene related peptide (CGRP), derived as a splicing variant from a gene that also encodes calcitonin, in the C-cells of some laboratory rodents, including the mouse (Zabel et al. 1988; Arias et al. 1989; Wimalawansa 1991; Thomas et al. 1994; Sawicki and Zabel 1999). Although the antiserum against CGRP does not always react against all of the C-cells, the expression of CGRP in C-cells is confirmed by a study using ISH techniques (van Lieshout et al. 1995). On the other hand, IHC studies have revealed that basal granulated cells in respiratory tracts produce CGRP (Uddman et al. 1985; Lauweryns and Van Ranst 1987; Keith and Ekman 1988, 1990). In human (Tsutsumi 1989; Stahlman and Gray 1993) and rat (Shimosegawa and Said 1991), it has been reported that calcitonin is expressed in some CGRP-positive basal granulated cells of the respiratory epithelium. These basal granulated cells are considered to be neuroendocrine cells because they contain protein gene product 9.5, which is a general neuroendocrine marker (Sorokin et al. 1993).
It has not yet been fully confirmed whether or not PC1 and/or PC2 are involved in the limited proteolysis of the precursors of calcitonin and CGRP. As far as we know, there has been only one IHC study, showing the presence of PC1 and PC2 in the normal human C-cells and medullary thyroid carcinomas (Tomita 2000). ICC examination of PC1 and PC2 will provide important evidence for the substrate-specificity of these convertases in the processing of the calcitonin and CGRP precursors. In this study we examined the light and electron ICC localization of PC1 and PC2 in the mouse thyroid gland and respiratory tract, and then found a correlation between the presence of these convertases and the presence of calcitonin and CGRP.
Materials and Methods
Animals
Normal adult male ICR mice, weighing about 40 g at 11 weeks of age, were housed under controlled environmental conditions (22C; L:D 12 hr:12 hr) and were allowed food and water ad libitum. Under sodium pentobarbital anesthesia, the trachea, with attached thyroid glands and parathyroid glands, was quickly removed from five mice and processed for light and electron microscopic ICC. All animal experiments were in compliance with the NIH Guide for the Care and Use of Laboratory Animals.
Antibodies
The antibodies used in this study were prepared in rabbits and characterized as described previously: anti-PC1 serum (ST-28) and anti-PC2 serum (ST-29) against synthetic peptides covering amino acids 442–459 and amino acids 613–629 of the respective mouse proteins (Tanaka et al. 1996), anti-salmon calcitonin serum (a gift from Prof. K. Wakabayashi, Gunma University), and anti-synthetic rat CGRP (1–37) serum (Cambridge Research Biochemical; Cheshire, UK).
Light Microscopic ICC
The organs were fixed by immersion in Bouin-Hollande sublimate for 2 days. After dehydration and embedding in Paraplast, serial 5-μm-thick transverse sections of the trachea were cut, including both lobes of the thyroid gland and parathyroid glands, and mounted on gelatin-coated slides. The deparaffinized sections were treated with iodine alcohol and sodium pyrosulfite to eliminate the sublimate and then incubated in a solution of 0.3% H2O2 in methanol for 30 min to inhibit endogenous peroxidase activity. After rinsing with distilled water (DW) followed by PBS (0.01 M sodium phosphate buffer and 0.14 M NaCl, pH 7.5), the sections were immunostained by the streptavidin-biotin method. To amplify the signals for PC1 and PC2, the sections were treated with Gomori's oxidation mixture according to the procedure of Kurabuchi and Tanaka (1997). The sections were incubated sequentially at RT with the following reagents in accordance with the manufacturer's instructions provided in the Histofine Kit (Nichirei; Tokyo, Japan): 10% normal goat serum (NGS) for 1 hr, rabbit anti-PC1 (1:10,000) or PC2 (1:8000) serum for 3 hr, biotinylated goat anti-rabbit IgG for 10 min, and finally peroxidase-conjugated streptavidin for 5 min. Peroxidase activity was detected using 15 mg 3,3′-diaminobenzidine tetrahydrochloride (Dojin; Kumamoto, Japan) and 0.01% H2O2 in 100 ml of 0.05 M Tris-HCl buffer, pH 7.6. The sections were lightly counterstained with Mayer's hematoxylin, dehydrated with ethanol, and then mounted in Entellan (Merk; Gobbstown, NJ).
Adjacent serial sections were immunostained to identify parafollicular cells using the same method with rabbit anti-salmon calcitonin antiserum (1:10,000) and anti-CGRP serum (1:2000), respectively.
The specificity of the anti-PC1 and anti-PC2 sera was also tested using an ICC preabsorption test. Diluted antisera were preabsorbed with their corresponding peptides at a final concentration of 0.01–1 mg/ml at 4C for 16 hr before ICC studies.
Electron Microscopic ICC
One mm3 pieces of the thyroid glands were fixed with a mixture of 0.5% glutaraldehyde and 4% paraformaldehyde in 0.05 M cacodylate buffer, pH 7.4, for 2 hr at 4C and then dehydrated with a graded ethanol series and embedded in LR White (London Resin; Basingstoke, UK). Pale gold ultrathin sections were cut with an ultramicrotome (LKB 2088 Ultrotome V) and mounted on gold grids. Immunolabeling was performed by the two-face double-labeling method (Bendayan 1982). One side of the sections was processed to label for PC1 or PC2; the other side was labeled for calcitonin or CGRP. One side of the sections was incubated with 10% NGS for 1 hr, transferred into a droplet of the first antiserum, and incubated overnight in refrigerator at 4C. After rinsing in PBS, the sections were incubated with biotinylated goat anti-rabbit IgG (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA) for 1 hr. Finally, they were incubated with streptavidin conjugated with 5-, 10-, 15-, or 20-nm gold particles (1:60; Zymed Lab, San Francisco, CA) for 2 hr and then rinsed with PBS and DW. The same procedure was used to treat the other side of the sections with another first antiserum and streptavidin conjugated with the different sizes of gold particles. The dilution of the antiserum was the same as mentioned above. The immunolabeled sections were stained with saturated uranyl acetate in DW. After the sections were washed and dried, they were observed with a JEM 1200 EX electron microscope at an accelerating potential of 80 kV.
Results
PC1- or PC2-immunopositive cells were observed in the thyroid glands (Figure 1) and respiratory tract (Figure 2). In the absorption test, the immunopositive materials obtained with both antisera were completely eliminated when 0.01 mg/ml of the respective antigen peptide was used as an adsorbent (data not shown).
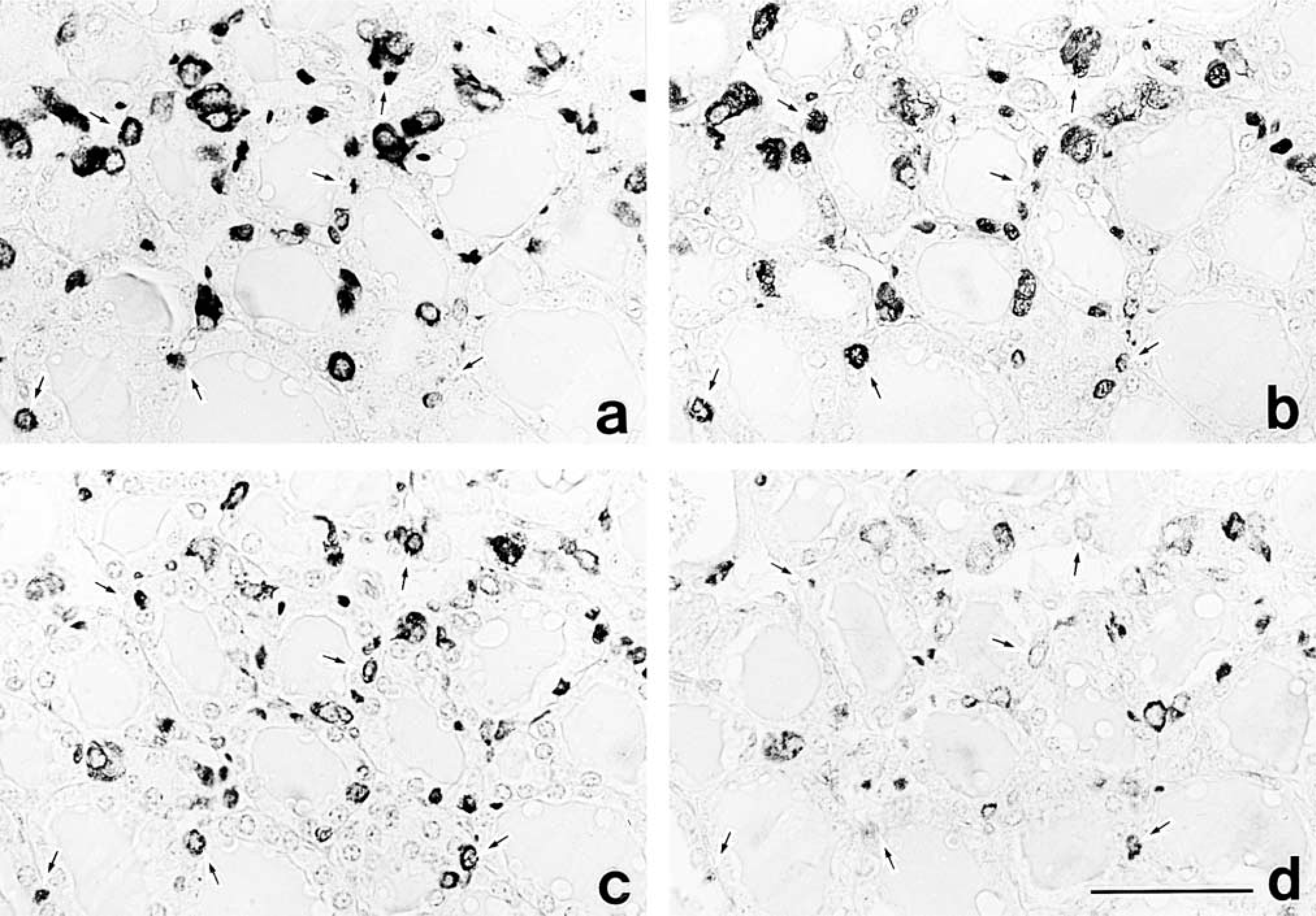
Light micrographs showing immunolabeling for calcitonin (
In the thyroid glands, PC1- or PC2-positive cells were round, scattered in the follicular epithelium and interfollicular space. PC1 immunoreactivity varied from cell to cell, suggesting a difference in PC1 content among the cells (Figure 1b). PC2 immunoreactivity was very faint (Figure 1d). When four serial sections of thyroid glands were immunostained with anti-calcitonin (Figure 1a), anti-PC1 (Figure 1b), anti-CGRP (Figure 1c), and anti-PC2 (Figure 1d), both PC1 and PC2 were localized in most of the C-cells containing both calcitonin and CGRP. However, the CGRP immunoreactivity was moderate or faint, whereas the calcitonin immunoreactivity was always strong.
In the respiratory tract, many PC1-positive cells were scattered in the tracheal epithelium (Figure 2a) and in the epithelium covering the ducts of the tracheal glands (Figure 2b), and a few CGRP-positive cells were seen located in the tracheal epithelium, but no PC2- or calcitonin-positive cells were observed (Figure 2d). When consecutive sections were immunostained with anti-PC1 and anti-CGRP, respectively, only a small population of PC1-positive cells corresponded with the CGRP-positive cells (Figures 2a and 2c).
Although several segments of CGRP-immunopositive nerve terminals were seen around thyroid follicles and in the subepithelial connective tissue of the trachea, they were not stained with either PC1 or PC2 antiserum (data not shown). On the other hand, several histological studies have shown that C-cells are localized in the parathyroid gland of some mammals (Welsch and Pearse 1969; Kameda 1971). However, no calcitonin- and CGRP-positive cells were observed in the parathyroid gland of the ICR mice used in this study.
The electron microscopic observations were performed to extend the light microscopic results. Immunoelectron microscopy of the thyroid C-cells showed many electron-dense secretory granules packed in the cytoplasm (Figure 3a). In double labelings, both PC1 and PC2 were co-localized in the same granules, although the PC2-labels had a remarkably low density (Figure 3c). The double labelings for PC1 and calcitonin or for PC1 and CGRP showed that PC1 coexisted with calcitonin and CGRP in most of the secretory granules (Figures 3a, 3b, and 3d), but fewer secretory granules were labeled for CGRP than for calcitonin (Figure 3d). No or few labels were seen in other organelles.
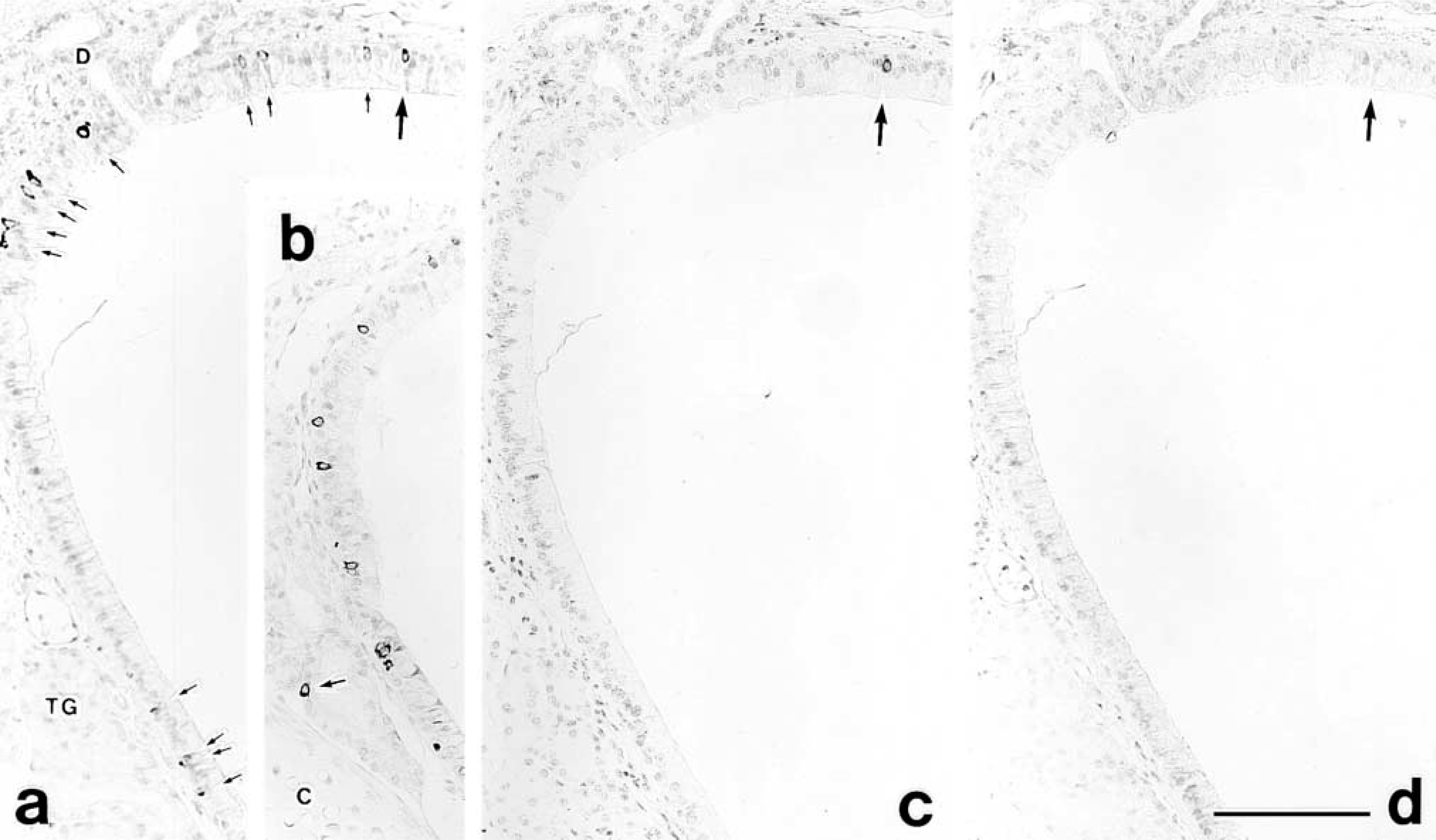
Light micrographs showing immunolabeling for PC1 (
Discussion
The present study, using specific polyclonal antisera raised against PC1 and PC2, clearly demonstrates that C-cells in the thyroid gland and basal granulated cells in the respiratory tract express these convertases. Both antibodies against calcitonin and CGRP marked almost all of the C-cells in the mouse thyroid, even though CGRP immunoreactivity is not always detected in all thyroid C-cells in several animals, including the mouse (Zabel et al. 1988; Arias et al. 1989; Sawicki and Zabel 1999; Thomas et al. 1994). The IHC co-localization of the convertases and calcitonin has also been reported in human medullary thyroid carcinomas, which contained elevated levels of these enzymes compared to normal thyroid C-cells (Tomita 2000). In the present study, on the other hand, PC1-immunopositive cells were detected in the tracheal epithelium and the ducts of the tracheal glands. Examination by the sequential staining method using adjacent sections revealed that a subpopulation of PC1-positive cells in the tracheal epithelium also produces CGRP. However, we could not detect any PC2-positive cells in the respiratory tract. The previous studies indicated that calcitonin and CGRP are co-expressed in thyroid C-cells and in some neurodendocrine cells of adult rat lung (Shimosegawa and Said 1991) and fetal and neonatal human lung (Tsutsumi 1989; Stahlman and Gray 1993). In this study, however, we found that some neuroendocrine cells express CGRP, but not calcitonin, in the adult mouse tracheal epithelium. Therefore, the expression of calcitonin in the tracheal epithelium may be different from animal to animal. It is known that alternative processing of RNA transcripts from the calcitonin gene results in the production of distinct mRNAs encoding calcitonin or CGRP (Amara et al. 1982). Simultaneously, the sequence analysis of their mRNAs revealed that procalcitonin is cleaved at the Lys-Arg and Gly-Lys-Lys-Arg sites to produce calcitonin, CCP, and 83-amino-acid N'-terminal peptide, whereas proCGRP is cleaved at two cleavage sites, Lys-Arg and Gly-Arg-Arg-Arg, to generate CGRP (Amara et al. 1980, 1982). The present ICC findings demonstrate co-localization of PC1 and PC2 with calcitonin/CGRP in thyroid C-cells and co-localization of PC1 with CGRP, suggesting that procalcitonin could be processed either by PC2 alone or by PC2 in concert with PC1, whereas proCGRP is cleaved by PC1. Our finding indicating that PC1 processes CGRP is supported by a biochemical study using site-directed mutants (Rosenblatt and Dickerson 1997). Because there are no biochemical studies on the specificity of PC1 and PC2 in the cleavage of calcitonin, our ICC observation provides further argument for the substrate specificity of PC1 and PC2 in the proteolytic cleavage of procalcitonin. In this connection, it is of interest that very high levels of serum procalcitonin are present in patients with severe systemic inflammation and sepsis, and this is therefore used as a clinical marker for systemic infection responses (Assicot et al. 1993; Dandona et al. 1994; Russwurm et al. 1999). Although the cellular sources of the high levels of serum procalcitonin have not been fully identified, various cells, including blood mononuclear cells, are presumed to be sources of the procalcitonin (Russwurm et al. 2001). Therefore, it is important to identify the precise cellular sites at which procalcitonin is synthesized during inflammatory conditions. The location may be found by determining which cells possess prohormone convertases, because procalcitonin should be processed to calcitonin in cells expressing PC1 and/or PC2.
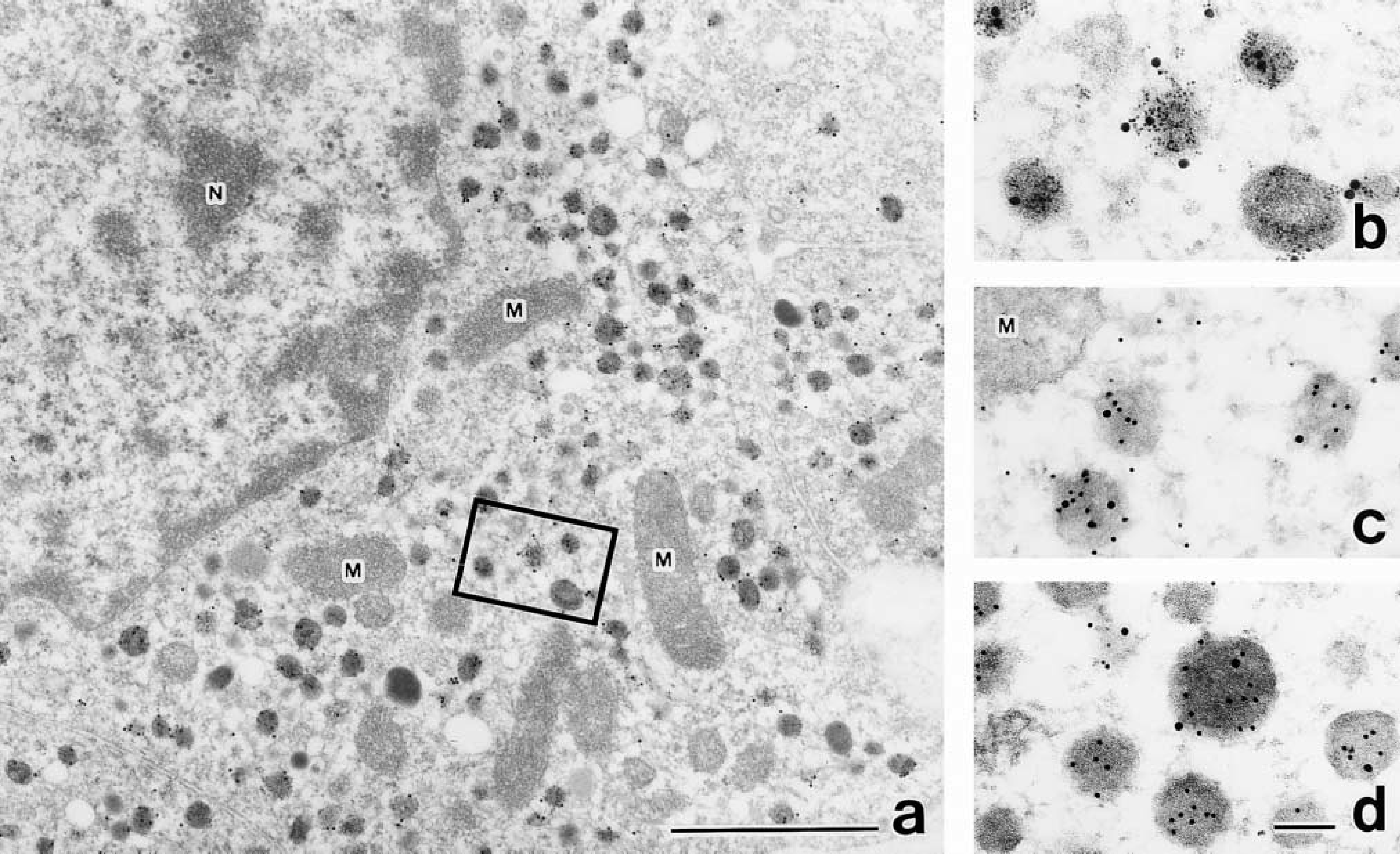
Immunoelectron micrographs showing parafollicular cells labeled with immunogold particles of different sizes (in parentheses): PC1 (20 nm) and calcitonin (5 nm) (
It is well known that the granin family members chromogranin A (CgA) and secretogranin II (SgII) play various important roles in the regulatory mechanism of secretion in many endocrine cells, although their exact function has not yet been elucidated (Huttner et al. 1991; Winkler and Fischer-Colbrie 1992). Several studies using gene transfer techniques suggest that PC1 and PC2 may be involved in the processing of the granin family (Arden et al. 1994; Dittie and Tooze 1995; Hoflehner et al. 1995; Eskeland et al. 1996; Laslop et al. 1998). This view is supported by our previous ICC study demonstrating co-localization of PC1, PC2, and CgA in large lucent secretory granules and co-localization of PC2 and SgII in small dense secretory granules of rat pituitary gonadotrophs (Uehara et al. 2001). It is well established that thyroid C-cells express CgA (Hawkins et al. 1989; Sawicki and Zabel 1999). Therefore, these findings suggest that PC1 and PC2 may participate in the processing of both CgA and calcitonin/CGRP in the thyroid C-cells.
It is known that somatostatin is also produced in thyroid C-cells (Van Noorden et al. 1977; Kameda et al. 1982), but its expression varies from species to species. Kameda et al. (1982) reported that some C-cells contain somatostatin in the mouse. A somatostatin-generating enzyme whose amino acid sequence displays a high homology to mammalian PC2 has been purified from anglerfish islets (Mackin et al. 1991). Using coexpression techniques involving different prohormone convertases and rat prosomatostatin, it has been shown that PC2 is a candidate endoprotease responsible for the production of somatostatin-14 (Brakch et al. 1995). Our previous ICC study detected PC2 in the pancreatic somatostatin-D cells (Tanaka et al. 1996). Taken together, these data suggest that PC2 in thyroid C-cells might be involved in the processing of prosomatostatin as well as procalcitonin. Other studies have shown that rat thyroid C-cells contain C-terminal gastrin/CCK-like immunoreactivity (Arias et al. 1989), but as far as we know there is no report of detection in mouse C-cells by anti-CCK. Therefore, gastrin/CCK can be excluded as a substrate of PC1 or PC2 in the C-cells.
There are several reports showing that neuroendocrine cells in the tracheal epithelium and lung have immunoreactivity for bombesin, calcitonin, somatostatin, and Leu-enkephalin, as well as for CGRP (Wharton et al. 1978; Cutz et al. 1981; Dayer et al. 1985). It is conceivable that PC1 is involved in the proteolytic cleavage of the precursors of these peptides because CGRP-positive cells are a subpopulation of the PC1-positive cells.
In the present study we were unable to stain the CGRP-immunopositive nerve fibers by either PC1 or PC2 antiserum; the reason for this is unclear.
In the present immunoelectron microscopic study, a small population of secretory granules in the C-cells were labeled with the antiserum against CGRP, unlike the heavy labeling for calcitonin, although most of the C-cells displayed similar immunoreactivity for both the hormones by light microscopy. This low labeling density for CGRP was also consistent with a previous report in the bat thyroid C-cells (Nunez et al. 1988). Double immunolabeling of prohormone convertases and calcitonin or CGRP revealed their co-localization in the secretory granules of thyroid C-cells. Our previous studies indicated that one or both of these convertases localize in the secretory granules of glucagon A-cells, insulin B-cells, somatostatin D-cells, and pancreatic polypeptide-producing cells (Tanaka et al. 1996; Itoh et al. 1996), in pituitary POMC cells (Kurabuchi and Tanaka 1997), and in gonadotrophs (Uehara et al. 2001). Because PC1 and PC2 are considered to be involved in the cleavage of paired basic amino acids of proproteins in the regulated pathway (Seidah and Chrétien 1992), the co-localization of these convertases with mature hormones in the secretory granules supports the view that the proteolytic precursors predominantly occur during maturation of secretory granules (Orci et al. 1985, 1987; Schnabel et al. 1989; Tanaka et al. 1997). Considering these data, it is possible that procalcitonin is processed in the secretory granules of C-cells either by PC2 alone or by PC2 together with PC1, whereas proCGRP is cleaved by PC1.
Footnotes
Acknowledgements
Supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan (SK and ST).
