Abstract
The distribution of the amino acid taurine in the female reproductive organs has not been previously analyzed in detail. The aim of this study was to determine taurine localization in the rat ovary, oviduct, and uterus by immunohistochemical methods. Taurine was localized in the ovarian surface epithelium. The granulosa cells and oocytes of primordial follicles were immunonegative. In primary and antral follicles, taurine was found mainly in theca cells and oocytes, whereas the zona pellucida, antrum, and most granulosa cells were unstained. However, taurine immunoreactivity in theca cells and oocytes decreased during follicular atresia. During corpora lutea development, the number of immunopositive theca lutein cells increased as these cells invaded the granulosa-derived region. Therefore, most luteal cells from the mature corpora lutea were stained. In the regressing corpora lutea, however, taurine staining in luteal cells decreased. In the fimbriae, infundibulum, and uterotubal junction, taurine was localized in most epithelial cells. In the ampullar and isthmic segments, taurine was found in the cilia of most ciliated cells and in the apical cytoplasm of some non-ciliated cells. In the uterus, most epithelial cells were immunopositive during diestrus and metestrus, whereas most of them were immunonegative during estrus and proestrus. Moreover, taurine immunoreactivity in the oviduct and uterus decreased with pregnancy. (
Taurine (2-aminoethanesulfonic acid) is a free ω-amino acid. Because of its zwitterionic nature at physiological pH, it has an extremely low capacity to diffuse across cell membranes (Huxtable 1992). However, taurine is an abundant intracellular amino acid. Its presence in a cell type is determined by its biosynthesis from cysteine and/or by the activity of specific membrane transporters (reviewed in Huxtable 1989, 1992; Sturman 1993). Taurine biosynthesis involves the formation of hypotaurine through the participation of cysteine sulfinate decarboxylase (EC 4.1.1.29). The cellular localization of taurine has been studied in a wide variety of tissues, including the central nervous system (Ottersen et al. 1985; Magnusson et al. 1988; Lake and Verdone–Smith 1989; Lee et al. 1992; Nagelhus et al. 1993), liver (Ding et al. 1993), kidney (Trachtman et al. 1993), muscle (Quesada et al. 1993; Lobo et al. 2000a), pancreas (Bustamante et al. 1998), and male reproductive system (Lobo et al. 2000b). Despite the increasing data reported, the functional roles of this sulfur-containing amino acid are still poorly understood. Taurine is not involved in protein synthesis or in any metabolic pathway, except in conjugation to bile salts in the liver. However, several functions have been demonstrated for taurine, such as osmoregulation, membrane stabilization, detoxification, anti-oxidation, modulation of ion flux, and as an inhibitory neurotransmitter or neuromodulator (Solís et al. 1988; Huxtable 1989, 1992; Martín del Río et al. 1990; Galarreta et al. 1996).
The roles of taurine in the reproductive system are multiple and complex. Sperm cells contain high amounts of taurine (Hernvann et al. 1986; Velazquez et al. 1986; Holmes et al. 1992). It may function as an antioxidant in preventing sperm lipid peroxidation, as a capacitating agent, and as a sperm motility factor (Meizel et al. 1980; Alvarez and Storey 1983; Meizel 1985; Fraser 1986; Boatman et al. 1990). Taurine might also modify sperm phospholipid methyltransferase activity, and inhibits sperm plasma membrane Na+, K+-ATPase (Mrsny and Meizel 1985; LLanos and Ronco 1994). Moreover, taurine is the predominant amino acid in genital secretions, including seminal, uterine, and oviduct fluids (Casslen 1987; Dumoulin et al. 1992; Guerin et al. 1995). It has been demonstrated that the ovary contains the mRNA of a taurine transporter (Jhiang et al. 1993), and the rat oviduct contains up to 10 μmol taurine/g tissue (Orensanz et al. 1986). Taurine is also present in high concentrations in the rat and human uterus, and its concentration decreases with pregnancy (Phoenix and Wray 1994; Turner et al. 1994). Despite all these data, the roles of taurine in the female reproductive system are largely unknown and there are no previous studies about its localization in these organs. The aim of this study was to determine the distribution of taurine in the rat ovary, oviduct, and uterus to provide some clues about its possible roles in these organs.
Materials and Methods
Adult female Sprague–Dawley rats (250 g) were used for this study. Rats were maintained on a 14L:10D cycle (lights-on at 0500 hr) with food and water provided ad libitum. Vaginal smears were taken to determine estrous cycle phase. A group of virgin female rats were mated and the first day of gestation was determined by the detection of a vaginal plug. Animals in all phases of the estrous cycle and on Days 9 and 18 of pregnancy were anesthetized with a mixture of ketamine, lidocaine, and atropine and perfused through the ascending aorta with 100 ml sodium phosphate buffer (0.12 M, pH 7.4), followed by 450 ml of one of four fixatives: 4% paraformaldehyde with 0.5%, 1%, or 1.5% glutaraldehyde in 0.12 M phosphate buffer, and 4% paraformaldehyde in the same buffer (control specimens). Tissue samples were removed from the rats and stored for 6–24 hr in the same fixative used during perfusion, washed in buffer, dehydrated, and embedded in paraffin. Streptavidin-peroxidase immunostaining was performed as previously described (Lobo et al. 2000a, b). Briefly, serial sections (5 μm thick) were mounted on silanized slides, deparaffinized, hydrated, and incubated for 20 min in 0.3% H2O2, followed by 0.03% H2O2 in methanol. After washing in Tris-buffered saline (TBS), pH 7.5, sections were incubated for 10 min in a 0.1 M sodium borohydride solution in TBS, pH 7.5. Sections were then washed and incubated in 3% normal goat serum with 0.05% Triton X-100 in TBS, pH 7.5, at room temperature (RT) for 30 min, followed by overnight incubation at 4C with the rabbit anti-taurine antibody (Chemicon International; Temecula, CA) diluted from 1:500 to 1:1500 in TBS. Afterwards the sections were washed twice in TBS and incubated with the secondary antibody, biotinylated goat antirabbit IgG (Zymed Labs; San Francisco, CA), for 1 hr at RT. After TBS washes, they were incubated with the streptavidin–peroxidase complex (Zymed) for 10–30 min and washed in TBS, followed by Tris-HCl buffer, pH 7.6. Then sections were incubated with a solution that contained 3′-diaminobenzidine tetrahydrochloride (DAB) as chromogen (Sigma; St Louis, MO): 10 μl of 8% aqueous nickel nitrate added to 1 ml of 0.05% DAB–0.02% H2O2 in Tris-HCl, pH 7.6, with all components mixed just before use. This method results in a dark black color of immunostained antigens. Finally, the sections were dehydrated in ethanol and mounted in Eukitt (O. Kindler; Freiburg, Germany) without being counter-stained. Negative controls were performed by (a) omitting the primary antibody, (b) using nonimmune serum instead of the primary antibody, (c) incubating with an inappropriate secondary antibody (goat anti-mouse) after the incubation with the anti-taurine antibody, and (d) immunolabeling on 4% paraformaldehyde-fixed samples. The last control should not give a positive immunoreaction because the antitaurine antibody used here was raised against taurine conjugated by glutaraldehyde to poly-
Results
The specific localization of taurine in the ovary (Figures 1 and 2), oviduct (Figure 3), and uterus (Figure 4) of the rat was studied. Similar results were obtained with the use of the different glutaraldehyde-containing fixatives (not shown). No immunostaining was observed in the negative controls (Figures 1k and 3a). In the cerebellar sections (positive controls), many Purkinje cells were stained, whereas glial cells were unstained (Figure 2i).
Most cells of the ovarian surface epithelium (germinal epithelium) were intensely immunolabeled (Figures 1a and 1g). In this cell type, taurine was localized in the cytoplasm, whereas the nucleus was weakly stained (Figure 1j). However, the collagen fibers and most cells of the tunica albuginea were unstained (Figure 1j). In follicle cells, the pattern of taurine staining varied with the different stages of follicle development. The single layer of granulosa cells and the immature oocytes of primordial follicles were immunonegative (Figure 1b). In contrast, when follicle development has begun and primary follicles contain two or three layers of granulosa cells, the oocytes became immunostained, whereas the granulosa cells remained unstained (Figure 1c). In preantral follicles showing a morphologically distinct theca layer, taurine was found in theca cells and in the oocytes, whereas the zona pellucida and the granulosa cells were poorly stained or unstained (Figure 1d). In antral follicles (Figures 1e and 1f) taurine was detected in the endothelial cells of the capillaries of the theca layer and in most cells of the theca interna. Moreover, the oocyte and some granulosa cells adjacent to the oocyte and the follicular antrum were also stained. However, granulosa cells were less intensely immunolabeled than theca cells and the oocyte. In these antral follicles, the zona pellucida, the follicular antrum, and most of the granulosa cells were not stained (Figures 1e and 1f). When the oocytes were immunolabeled, taurine appeared mainly distributed in the cytoplasm whereas the nucleus was usually weakly stained (Figures 1c and 1e). However, taurine was localized in the nucleus and cytoplasm of theca cells. In contrast to maturing follicles (Figures 1b–1f), taurine immunoreactivity progressively disappeared from the oocyte and theca cells during follicular atresia (Figures 1g–1i). Atretic follicles were identified by the presence of pyknotic cells in the granulosa layer, cell debris in the follicular antrum, the shrinking oocyte, accumulation of lipid droplets in theca and granulosa cells, and the final appearance of the collapsed follicle with a hypertrophied theca layer. In early atretic follicles (Figure 1g), identified by the presence in serial sections of a few pyknotic granulosa cells, taurine was detected in some cytoplasmic areas of the oocyte and in some theca cells. However, in the late stages of atresia taurine almost disappeared from most follicle cells, including the oocyte (Figures 1h and 1i).
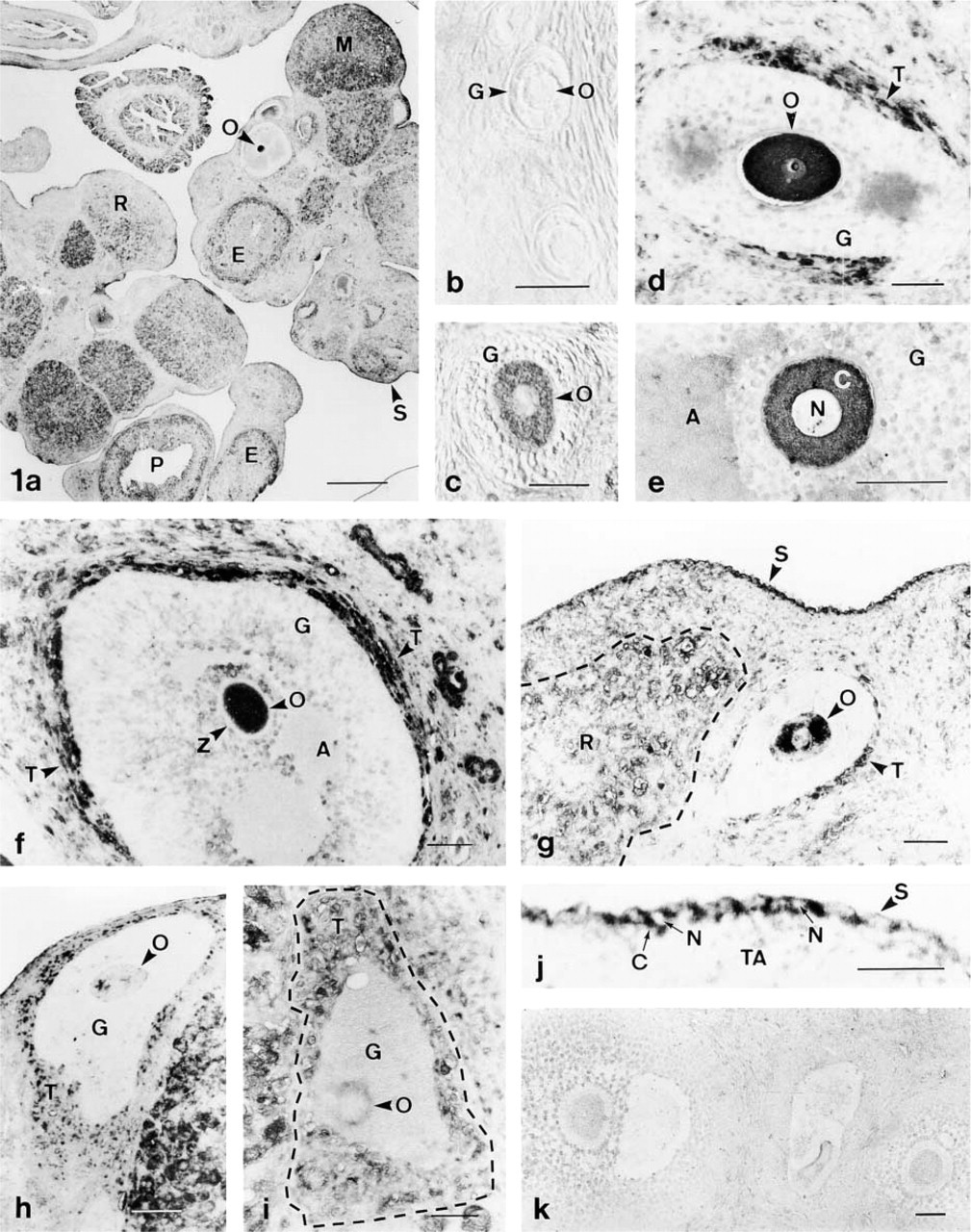
Taurine localization in the rat ovary. (
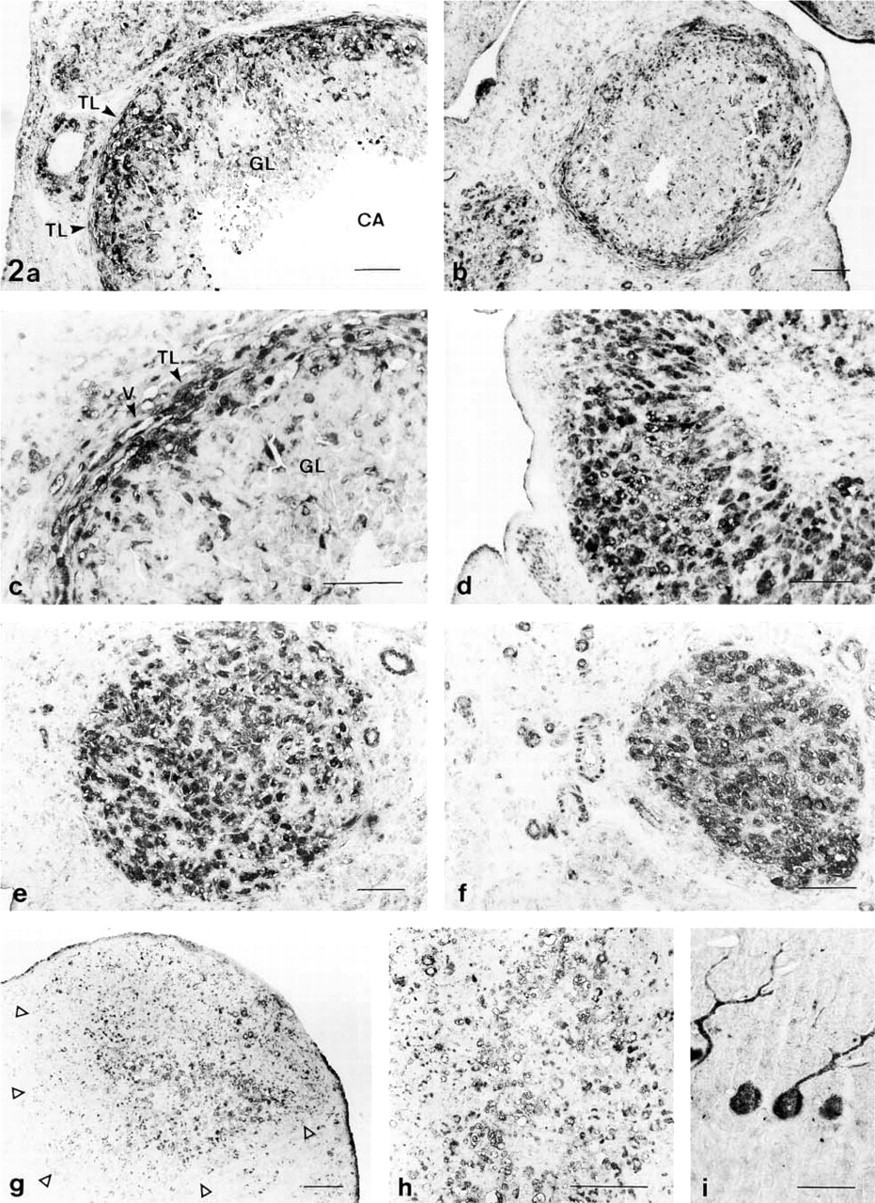
(
Taurine immunoreactivity varied in relation to the different stages of corpora lutea formation and regression (Figures 1a and 2). In the post-ovulatory period, taurine was detected in most theca lutein cells and in some cells scattered in the granulosa-derived region, whereas most granulosa lutein cells were unstained (Figure 2a). Moreover, the endothelial cells of the luteal vessels (mainly large-diameter capillaries) were also stained. A similar labeling pattern was observed in the early luteal phase (Figures 2b and 2c). During the luteinization process, when theca-derived cells invade the granulosa-derived region, an increase in the number of immunopositive luteal cells was observed. Finally, most cells from the mature corpora lutea were stained (Figures 2d–2f). However, in the regressing corpora lutea, taurine immunoreactivity decreased in luteal cells (Figures 2g and 2h). Positive immunostaining for taurine was also found in some polyhedral interstitial endocrine cells of the cortical stroma. In the cortex and medulla, some vascular endothelial cells and fibroblasts were immunolabeled, whereas collagen and elastic fibers were unstained.
The oviduct comprises four morphologically distinct segments: preampulla (which includes both the fimbriae and infundibulum), ampulla, isthmus, and uterotubal junction. The distribution of taurine in these regions was studied in the oviduct of nonpregnant (Figures 3a–3n) and pregnant rats (Figures 3o and 3p). In all oviducts examined, taurine was mainly found in the luminal epithelium which contained two main cell types, ciliated and nonciliated cells. The ciliated cells were identified by the presence of kinocilia on the apical cell surface (Figures 3f and 3g). The nonciliated cells presented microvilli extending into the lumen, and the apical surface of these cells was either at the same level as the ciliated surface or protruded into the oviduct lumen (Figures 3g and 3j). In nonpregnant rats, taurine immunoreactivity was higher in the uterotubal junction (Figure 3b) and preampulla (Figures 3k–3n) than in the isthmus (Figures 3c–3g) and ampulla (Figures 3h–3j). In the ampullar and isthmic segments, taurine was found in the apical surface of the epithelium (Figures 3c–3j). In ciliated cells, it was mainly found in the cilia, whereas the rest of the cell was usually unstained (Figures 3d–3f). Some scattered nonciliated cells were stained in these segments, taurine being found mainly in the apical cell part (Figures 3g and 3j). In the fimbriae (Figures 3k–3m), infundibulum (Figures 3k and 3n), and uterotubal junction (Figure 3b), both main cell types of the oviductal epithelium were stained. In nonciliated cells taurine was mainly found in the apical cytoplasm and in the nucleus (Figures 3b and 3l). Ciliated cells showed a variable degree of taurine immunostaining in these segments. In some ciliated cells taurine was mainly restricted to the ciliated surface (Figure 3l), whereas in other cells it was distributed all along the cell, including the ciliated surface (Figure 3m). In the oviduct of pregnant rats, taurine was found only in the fimbrial epithelium, whereas all other segments were weakly stained or unstained (Figures 3o and 3p). In all oviducts examined, only a few scattered cells of the oviductal stroma contained taurine, and the smooth muscle cells showed a variable degree of immunostaining, being weak in some cells and strong in other smooth muscle cells.
No cyclic changes of taurine immunoreactivity were found in the ovary and oviduct. However, dramatic cyclic variations were observed in the epithelial cells of the uterus. Most cells of the luminal and glandular epithelia were immunopositive for taurine during diestrus (Figures 4a–4c) and metestrus (Figure 4h), whereas most epithelial cells were immunonegative during proestrus (Figure 4d) and estrus (Figures 4e–4g). Moreover, many stromal cells (fibroblasts, vascular endothelial cells, lymphocytes, and macrophages) were stained. No cyclic changes in the stromal staining were observed (Figures 4a–4h). The smooth muscle cells of the uterus were usually weakly stained or unstained (Figure 4a). However, moderate staining was found in some muscle cells during estrus (Figure 4i). In the perimetrium, some vascular endothelial cells and fibroblasts were stained (Figure 4i). In addition, most cells in the epithelium, stroma, and myometrium of the gravid uterus were unstained (Figures 3p and 4j).
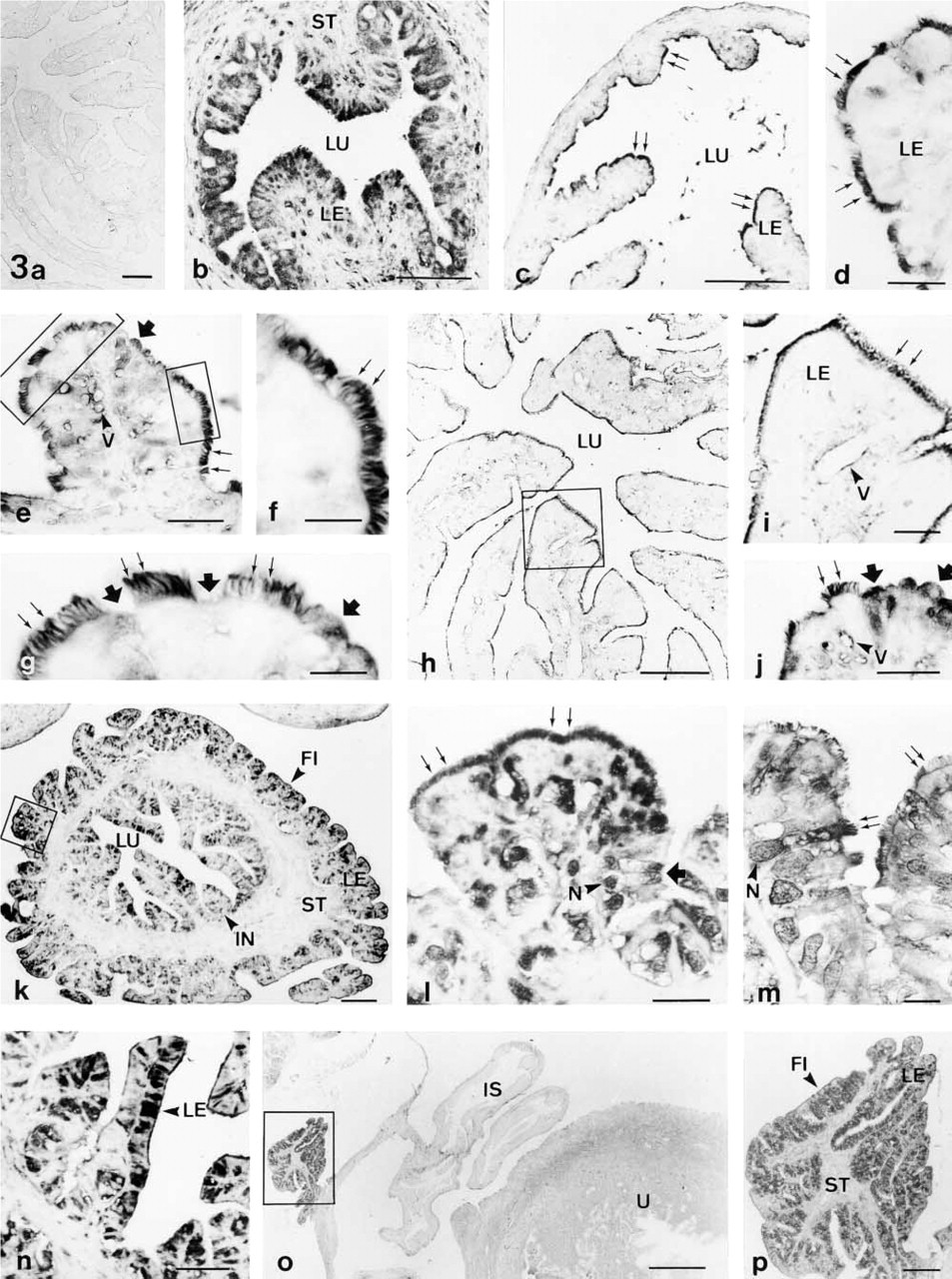
(
Discussion
The distribution of taurine in several cell types is highly dynamic and involves variations in the cellular taurine concentration under modifications of the cellular environment or functional state (Huxtable 1992; Nagelhus et al. 1993; Quesada et al. 1993; Lobo et al. 2000a). Moreover, changes in the cellular taurine content occur mainly in stress states, such as osmotic changes, anoxia, prolonged illumination of photo-receptors, or congestive heart failure (reviewed in Huxtable 1992). This study shows that taurine is not evenly distributed in the female reproductive organs but is localized in some specific cell types. We also show that the immunoreactivity of some cells depends on their physiological state. In this sense, taurine immunoreactivity varies in the oocyte and theca cells during follicular development and atresia, in luteal cells during corpora lutea formation and regression, in the oviduct and uterine epithelia comparing pregnant and nonpregnant rats, and in the uterine epithelium during the estrous cycle. Moreover, the decrease in taurine immunoreactivity observed in the oviduct and uterine epithelia during gestation is consistent with the decreased levels in taurine concentration reported in the uterus and in other tissues with pregnancy (Phoenix and Wray 1994; Turner et al. 1994).
It has been demonstrated by RT-PCR that the rat ovary contains the mRNA of a taurine transporter, which has been cloned from human thyroid cells (Jhiang et al. 1993). Other than this study, only few data have been reported dealing with taurine in the ovary. Therefore, the physiological significance of the results presented here remains to be elucidated. However, it is interesting to note that the immature oocytes of primordial follicles are unstained but that when oocytes initiate the maturation process they become immunoreactive for taurine. Similarly, it has been demonstrated that, in the testis, germ cells at all spermatogenic stages are immunonegative for taurine (Lobo et al. 2000b). However, sperm cells during their maturation process along the epididymis take up taurine from the epididymal fluid, and taurine has been implicated in some important roles in sperm cells (Holmes et al. 1992; Huxtable 1992). Therefore, the presence of taurine in the rat oocytes, once follicles enter the growing pool, emphasizes the proposed role of taurine in gamete maturation. Moreover, we demonstrate here that follicular cells and the oocyte lose their taurine immunoreactivity during the process of follicular atresia. Because no catabolic pathway for taurine has been reported, the loss of taurine immunoreactivity in atretic follicles probably corresponds to an efflux of taurine from these cells during follicular atresia. This taurine efflux may represent a new integral event of the atretic process and might be similar to the taurine release that occurs in Jurkat lymphocytes during apoptosis (Lang et al. 1999).
In the ovary, theca cells are intensely stained for taurine, whereas granulosa cells are weakly stained or unstained. The primary role of theca cells is to synthesize and to provide an aromatizable androgen substrate (i.e., testosterone, androstenedione) for granulosa cell estrogen biosynthesis. In the testis, taurine is mainly localized in Leydig cells (Lobo et al. 2000b), which are androgen-producing cells. Therefore, in both the testis and the ovary, the cells that use the steroidogenic pathway for the conversion of cholesterol to androgens contain taurine. This suggests the existence of a poorly known relationship between the physiology of steroidogenic cells and taurine. Because the steroidogenic pathway involves the production of free radicals and lipid peroxidation (Peltola et al. 1996), we propose that the presence of taurine in these cell types could be related to its antioxidant capacity.
The oviduct epithelium consists of two kinds of cell, ciliated and nonciliated (secretory) cells. Ciliated cells play important roles in the transport of gametes, whereas nonciliated cells synthesize and release specific secretory products to the oviduct fluid (reviewed in Abe 1996). Taurine was found in both cell types. In ciliated cells, taurine was mainly found in the cilia. Moreover, GABA (an ω-amino acid that shows a tight relationship with taurine) is present in the rat oviduct at high concentrations, 5 μmol GABA/g tissue (Martín del Río 1981). Immunohistochemical studies show a GABA distribution very similar to that reported here for taurine. GABA is localized in the oviduct epithelium, particularly at the ciliated surface (Apud et al. 1984; Erdö 1986; Erdö et al. 1986). Therefore, taurine could have a similar functional role to that previously proposed for GABA in the oviduct epithelium (Erdö 1986; Erdö et al. 1986); both molecules may be involved in ciliary motility and, accordingly, in the transport of gametes.
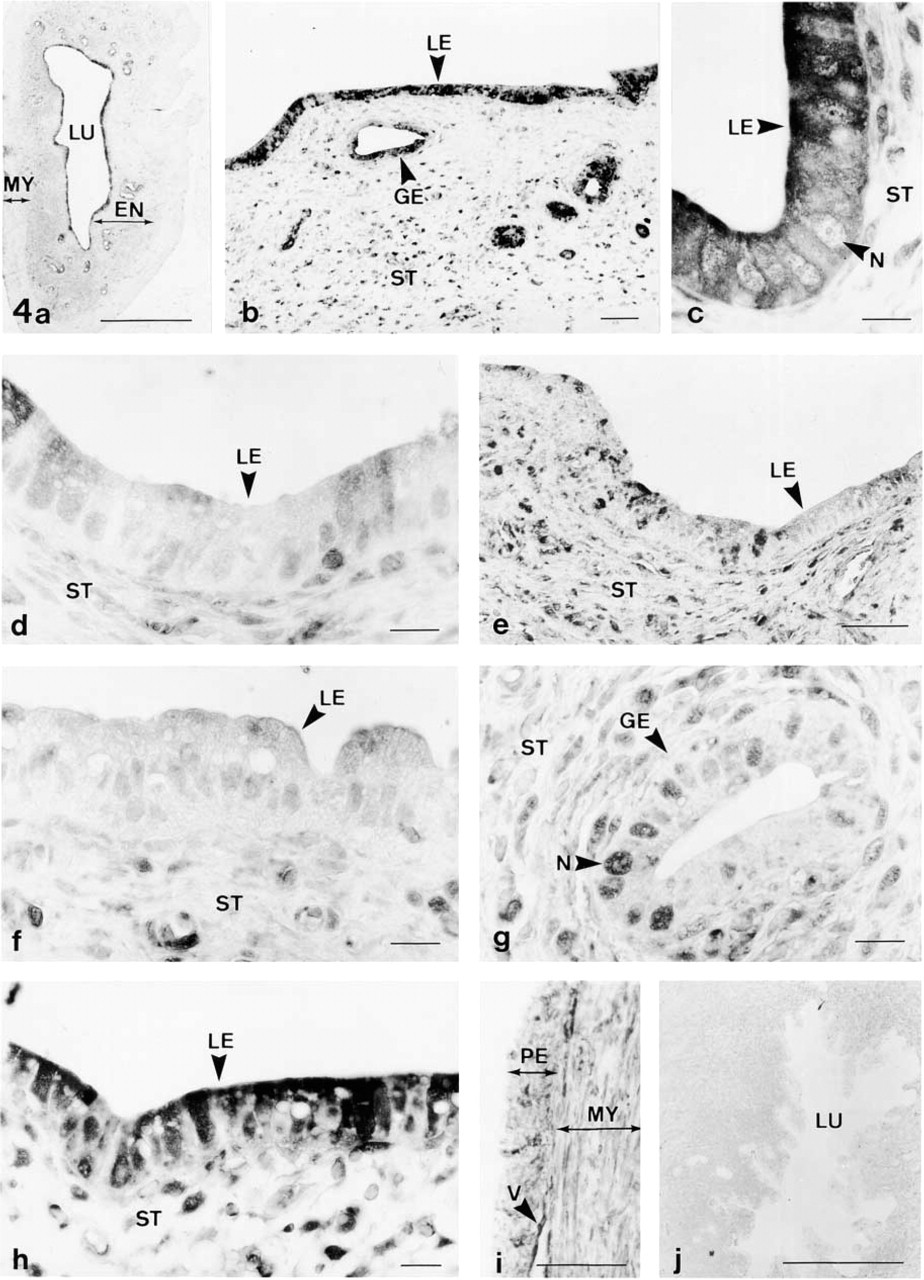
Immunolocalization of taurine in the uterus of nonpregnant (
Increasing research interest has recently addressed the identification and characterization of the secretory products released by the oviduct epithelium. They have special relevance in relation to the physiological events occurring in the oviduct, such as gamete transport, capacitation, fertilization, and early embryo development (reviewed in Abe 1996). In this sense, it has been reported that taurine is synthesized by oviduct cells via the cysteine sulfinate decarboxylase pathway, and it is secreted into the oviduct fluid (Guerin and Menezo 1995; Guerin et al. 1995). In accord with this notion, it has been shown that taurine comprises about 59% of the total free amino acid content in the mouse oviduct fluid, whereas it represents 17% in mouse serum (Dumoulin et al. 1992). Furthermore, taurine/hypotaurine enhances preimplantation embryonic development after in vitro fertilization (Barnett and Bavister 1992; Dumoulin et al. 1992). In relation to this functional role of taurine, a critical influence of the oviduct within the first hours of embryo development has been suggested (Barnett and Bavister 1992). Therefore, the localization of taurine in nonciliated cells could be related to secretion of this amino acid into the lumen, with a possible functional role in early embryonic development. Moreover, it has been shown that taurine is the major free amino acid of the human uterine fluid; its concentration is up to 400-fold that of plasma during the luteal phase (Casslen 1987). These taurine levels probably result from endometrial secretion, and therefore the localization of taurine in the uterine epithelium could be related with this secretory function.
No cyclic changes in the proportions of ciliated and nonciliated cells have been found in the oviduct of the golden hamster and rat. However, in other species, such as the primates and the cow, cyclic variations in the relative number of both cell types have been reported (Abe 1996). Therefore, although we have not found changes in the pattern of taurine localization in the rat oviduct during the estrous cycle, perhaps in other species it could be appreciated. Moreover, the changes found for taurine immunoreactivity in the uterine epithelium during the estrous cycle and the changes in taurine localization observed in pregnant rats compared with nonpregnant rats appear to indicate the existence of some hormonal mechanisms that regulate the taurine levels in the rat reproductive organs. According to this notion, it has been shown that the hypotaurine content of some tissues (prostate and seminal vesicle) is regulated by androgen (Kochakian 1973, 1975) and that the activity of the rate-limiting enzyme for taurine synthesis (cysteine sulfinate decarboxylase) is under inhibitory regulation by estrogen (Kaisaki et al. 1995). We found in the uterine epithelium the lowest expression level of taurine in the proestrous and estrous phases, which correspond with high estrogen levels in the plasma, while the highest levels of taurine were observed in the metestrous and diestrous phases, which correspond with low estrogen levels in plasma. Moreover, studies about the dynamics of the two types of estrogen receptors (α and β) in the female reproductive tract demonstrated that estrogen receptor-α is the predominant subtype in uterus and that its expression changes during the estrous cycle (Wang et al. 1999, 2000). These data suggest that the taurine content of the uterine epithelium could be regulated by cycling estrogen levels via binding to specific receptors in target cells, which could lead to a modulation of the activity of cysteine sulfinate decarboxylase.
This study is the first to demonstrate the distribution of taurine in the female reproductive organs. Although the physiological significance of these results remains to be determined, a growing body of evidence indicates that taurine may have important roles on the reproductive system. Future studies on the taurinergic mechanisms in these organs may lead to a better understanding of the physiology of the reproductive system.
Footnotes
Acknowledgment
We thank Dr Carlos Correa of the Experimental Surgery Service at the Hospital Ramón y Cajal, who carried out examinations of the animals and vaginal smears.
