Abstract
This study was designed to determine the cellular and ultrastructural distribution of the gonadotropin-releasing hormone (GnRH) and the relative expression of its mRNA in the oviduct of rats during different time points (days 7, 9, 16, and 20) of pregnancy. Immunofluorescent localization and confocal microscopic techniques were used to determine the cellular distribution of GnRH in the oviduct. Immunogold electron microscopy indicated its localization at the ultrastructural level, and real-time PCR was used to study the expression pattern of GnRH mRNA in the oviduct during pregnancy. In general, GnRH was localized within the epithelial cells lining the oviductal lumen at each selected time point. A strong correlation between the fluorescence intensity of GnRH-immunoreactive cells and the relative expression of GnRH mRNA was noted on days 7 and 16, followed by a plateau by day 20. At the ultrastructural level, uniform labeling of colloidal gold particles was observed in secretory vesicles and lamella of the luminal epithelium as well as the lumen of the oviduct. Collectively, these results demonstrate for the first time that the oviductal epithelium synthesizes and secretes the decapeptide GnRH during pregnancy in rats, which may have a possible role in postimplantation embryonic development and the maintenance of pregnancy.
T
A functional form of the hypothalamic decapeptide, gonadotropin-releasing hormone (GnRH), which is considered a key neuromodulator of mammalian reproduction (Conn and Crowley 1991), has been reported to be expressed in non-neuronal reproductive tissues such as the oviduct, ovary, placenta, testis, and uterine endometrium (Ramakrishnappa et al. 2005) in several mammalian species where it performs local functions. Reports in human and other mammals exist regarding the presence of GnRH receptors in oocytes and spermatozoa (Dekel et al. 1988; Minaretzis et al. 1995; Morales et al. 1999). Increases in the ability of human sperm to bind to the zona pellucida of oocytes when incubated in a medium containing GnRH (Morales 1998), induction of oocyte cleavage by GnRH (Funston and Seidel 1995), stimulation of early embryonic development by a GnRH agonist (Tesarik et al. 2004), and a dose-dependent suppression of in vitro fertilization by a GnRH antagonist (Morales et al. 1999) support the idea that GnRH has functional significance in regulating processes such as fertilization and early embryonic development, leading to induction of pregnancy in mammals. Expression of immunoreactive GnRH in the human maternal reproductive tract during the luteal phase of the menstrual cycle has been studied (Casan et al. 2000), providing evidence of its role as a molecular autocrine/paracrine regulator of processes such as fertilization, early embryonic development, and implantation. The role of GnRH in embryonic/endometrial dialogue during early implantation of human embryos has been reported by Raga et al. (1998). Immunohistochemical localization of GnRH in preimplantation mouse embryos confirms the local role of this decapeptide in endometrial receptivity and, hence, implantation processes (Raga et al. 1999) in mammals.
Description of the sense and antisense pairs of primers specific for GnRH and 18S mRNA amplification used in real-time polymerase chain reaction

There is a paucity of information regarding the role of oviductal GnRH during cyclicity in rodents as well as on postimplantation processes and/or maintenance of pregnancy in mammals. To focus on the role of oviductal GnRH during postimplantation embryonic development and, as such, maintenance of pregnancy in rodents, the present study was designed to determine the expression patterns of immunoreactive GnRH and its mRNA in the rat oviduct during different time points of pregnancy. Immunogold labeling of GnRH was done to understand the intracellular distribution of this protein in the luminal epithelium of the oviduct and its secretion into the lumen. Each of the chosen time points (days 7, 9,16, and 20) of pregnancy corresponds to changes in the serum profile of the key hormone progesterone critical for induction and maintenance of pregnancy in rodents in relation to changes in the circulating levels of luteinizing hormone (LH) and prolactin, which are interrelated throughout gestation (Morishige et al. 1973).
Materials and Methods
Animals
All experiments were performed in compliance with the animal care regulations of Morehouse School of Medicine following protocols approved by the Institutional Animal Care and use Committee using accepted standards of NIH guidelines for humane care and use of experimental animals. Timed-pregnant Sprague Dawley rats (∼8 weeks old) were obtained from Charles River Laboratories (Wilmington, MA) and housed in the Institutional Animal Care Facility at a temperature of 23-25C under a photoperiodic schedule of daily 14 hr light:10 hr darkness. Purina rat chow and tap water were accessible ad libitum. Considering the day of insemination (identified by the appearance of the sperm plug) as day 1 of pregnancy, animals (
Tissue Collection
Embryonic implantation within the uterus was checked for each animal during each of the considered time points to ensure induction of pregnancy prior to tissue collection. Paired oviducts along with the hypothalamus and liver of each animal (
Immunofluorescent Localization of GnRH and Confocal Microscopy
A portion of the paraformaldehyde-fixed oviducts from each animal were cryoprotected in 30% sucrose solution overnight and cut into 25-μm sections using a cryostat set at −15C. Free-floating sections were collected in 0.02 M KPBS (pH 7.2). For immunostaining, sections were pretreated with 0.2% sodium borohydride for 20 min to reduce autofluorescence, preincubated in a blocking solution containing 10% normal horse serum, 0.25% BSA, 0.1% gelatin, and 0.1% Triton X-100 in 0.02 M KPBS for 1 hr at room temperature, followed by overnight incubation at 4C with mouse monoclonal anti-GnRH antibody (100% immunoreactivity with the fully processed bioactive form of LH-releasing hormone, sequence: pGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2; cat. #MAB5456, Chemicon International, Temecula, CA) diluted 1:500 in KPBS solution containing 1% normal horse serum and 0.1% Triton X-100. Negative controls included sections in which the primary antibody was omitted or the anti-GnRH primary antibody was preabsorbed with a synthetic GnRH peptide (Peninsula Laboratories; San Carlos, CA). Sections of each group were then washed sequentially in 0.02 M KPBS and incubated in donkey anti-mouse Alexa Fluor 594 fluorescent secondary antibody (1:1000 dilution; Molecular Probes, Carlsbad, CA) for 1 hr at room temperature. After multiple washes, the stained sections were mounted on glass slides with Vectashield (Vector Laboratories; Burlingame, CA) mounting medium and observed using an Olympus laser-scanning confocal microscope system (IX71; Olympus, Tokyo, Japan) to observe the localization and distribution of GnRH-immunopositive cells in the oviduct collected from rats during different days of pregnancy. Immunostaining of sagittal sections of pregnant rat brains (prior to oviductal study) exhibited clear immunolocalization of GnRH neurons in the hypothalamic region, indicating specificity of the anti-GnRH antibody used throughout the present study.
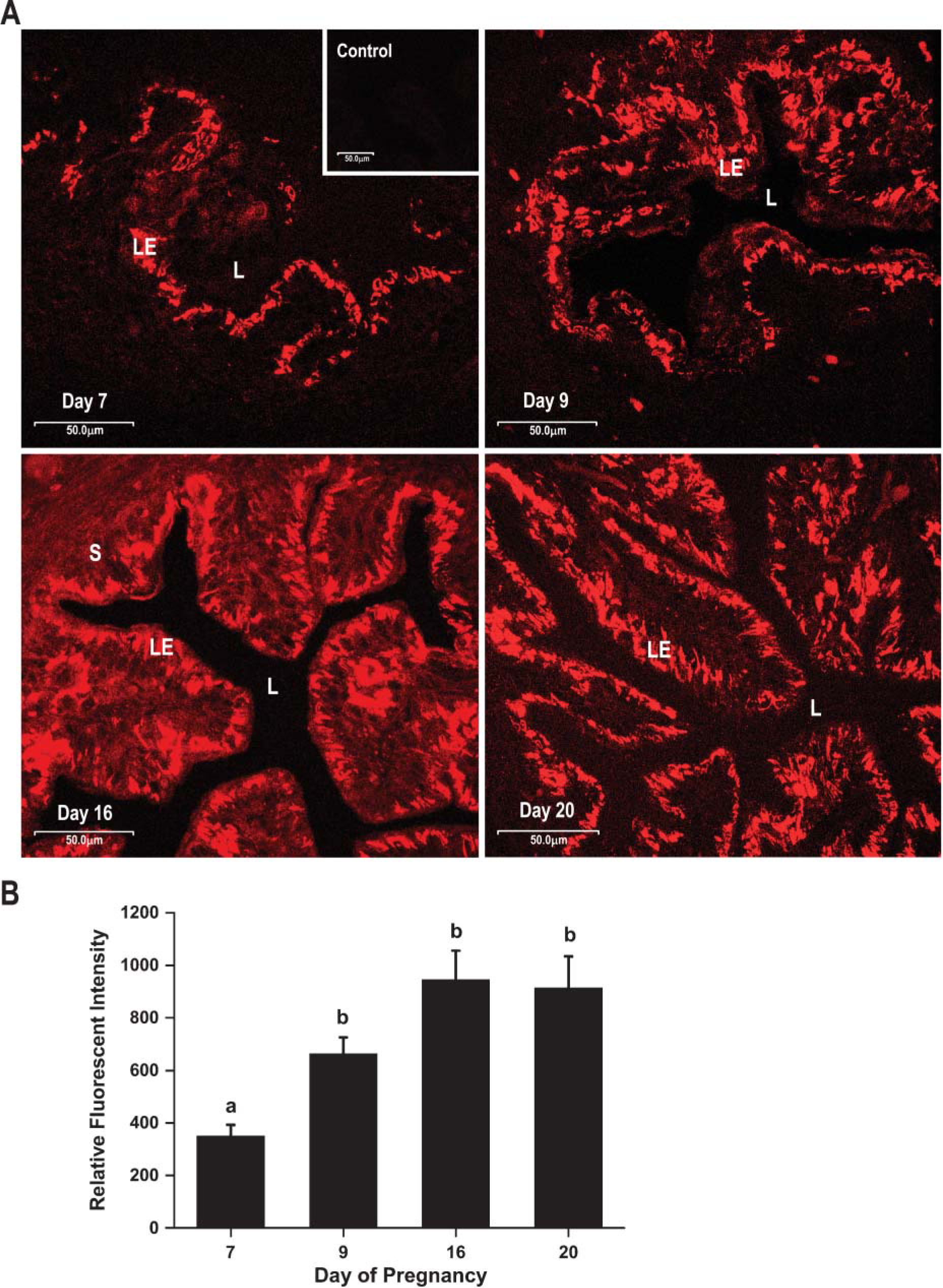
(
Measurement of Relative Fluorescent Intensity of the GnRH-immunopositive Cells
Measurement of the fluorescent intensity of GnRH-immunopositive cells was done in five different non-overlapping fields from three to four sections taken from each animal, collected during the selected time points. Fluorescent intensity values of different cells in a line, in a particular field of an image taken from a particular section, were recorded using the Olympus Fluoview Fv300 (version 4.3), Image Analysis (Line Profile) software. Average fluorescent intensity (AFI) of all the cells located in the five such selected fields in a particular section was calculated. Multiple peak fluorescent intensities of the GnRH-immunopositive cells of a particular section, where the values are higher than the AFI, were normalized by subtracting the AFI value of that specific section. Normalized peak fluorescent intensity was represented as relative fluorescent intensity (RFI) in the present study. The mean RFI values during different days of pregnancy were plotted graphically.
Isolation of Total RNA
Frozen oviductal tissues were weighed and homogenized in 1 ml of RNeasy lysis buffer (Qiagen; Valencia, CA) containing 10 μl of β-mercaptoethanol. Homogenate was centrifuged at 3200 x g for 10 min at room temperature. The supernatant was loaded on an RNeasy midi column (Qiagen), and the entire process of RNA isolation and purification was carried out according to detailed manufacturer's instructions provided in the kit. RNA samples were eluted in 25-μl nuclease-free water (provided in the kit) and quantified in an Eppendorf AG 2231 Biophotometer (Hamburg, Germany). RNA purity was assessed with the same instrument using the 260:280 nM ratios, which were in the range of 1.8 and 2.1. RNA integrity was assessed by examining the 28S and 18S rRNA bands of representative samples loaded on an Agilent 2100 Bioanalyzer using RNA 6000 Nano Assay kit (Agilent Technologies; Santa Clara, CA).
Reverse Transcription
Two μg of RNA samples from each tissue type (oviduct, hypothalamus, and liver) were converted to cDNA by reverse transcription at 48C for 37 min using murine leukemia virus reverse transcriptase enzyme and random hexamers (Taqman Reverse Transcription Reagent kit; Applied Biosystems, Foster City, CA) according to manufacturer's instructions. cDNA samples were then stored at −20C for real-time PCR studies.
Real-time PCR
Sequence of
A temperature curve was done to determine the annealing temperature of the specific primers for GnRH followed by the melt curve to determine the absence of primer dimer formation and any nonspecific fluorescent signals. The final annealing temperature of the sense and antisense GnRH and 18S rRNA primers were determined to be 54C [temperature at which the threshold cycle (CT) value was minimum and fluorescence value was maximum].
Quantitative PCR assay was performed with the help of a MyiQ iCycler (Bio-Rad Laboratories; Hercules, CA) real-time PCR detection system using 750 ng of cDNA for each reaction (as determined from the standard curve of rat GnRH using various dilutions of oviductal cDNA). Each gene expression was assessed in a separate PCR reaction with each reaction mixture containing 2 μl of sense and antisense primers (0.35 nM/μl concentration) of GnRH and 18S rRNA, respectively, 10 μl of 2X quantiTech SYBERGreen PCR Master Mix (containing HotStarTaq DNA polymerase enzyme, quantiTech SYBERGreen PCR buffer, dNTP mix including dUTP, fluroscent dyes like SYBERGreen 1 and ROX, and 5 mM MgCl2) obtained from Qiagen and DNase RNase-free water to make a total volume of 20 μl. Samples were amplified with a precycling hold of 95C for 15 min, followed by 50 cycles of denaturation at 94C for 15 sec, annealing at 54C for 30 sec, and extension at 72C for 30 sec. The reaction was terminated at 72C for 5 min and cooled to 4C. Real-time PCR amplification efficiencies were indicated by the linearity of the logarithmic algorithm and determined by Pearson's correlation coefficient and slope of the linear curve.
Quantification of results was expressed in terms of CT value. The rat hypothalamus and liver tissue in which the GnRH gene expresses the maximum and minimum, respectively, were used as positive and negative controls. The comparative CT method (Qiagen) was used to compare the relative expression of GnRH mRNA during different days of pregnancy, using day 7 as 1, the calibrator. Briefly, CT values were averaged from each duplicate and differences between the mean CT values of the GnRH and 18S rRNA (housekeeping gene) were calculated as ΔCTsample = CT GnRH–CT 18S rRNA for normalization. Finally, sample calibrator ratios, expressed as N-fold differences of GnRH expression on different days relative to day 7, were determined as 2−(ΔCT sample-ΔCT calibrator) (Livak and Schmittgen 2001; Zhoa et al. 2004).
Immunogold Labeling for GnRH
As immunofluorescent staining exhibited maximum localization of GnRH in the rat oviduct collected during day 16 of pregnancy, we used oviductal tissues of this specific day for immunogold labeling of this protein. Oviducts fixed overnight in 4% paraformaldehyde were washed in PBS to remove excess fixative, dehydrated in graded alcohol, infiltrated in LR white resin, and polymerized in a vacuum oven at 45C for 48 hr. Sixty-nm ultrathin sections were cut and placed on formvar-coated nickel grids for postembedding immunogold labeling for GnRH. Potential nonspecific labeling was blocked by incubating the sections in PBS containing 5% normal goat serum, 5% bovine serum albumin, and 0.1% cold-water fish gelatin at room temperature for 1 hr. Sections were then incubated overnight at 4C with mouse monoclonal anti-GnRH primary antibody that recognizes the mature, fully processed bioactive form of the antigen with an NH2 group at position 10 of the decapeptide (Chemicon International) at a dilution of 1:500 in PBS buffer and incubated in 10-nm colloidal gold-conjugated goat anti-mouse IgG secondary antibody (Aurion; Wageningen, The Netherlands) at a 1:20 dilution for 90 min at room temperature. Sections were then subsequently washed in PBS, further fixed in glutaraldehyde, counterstained in uranyl acetate and lead acetate, and examined under a transmission electron microscope (1200EX; Jeol, Tokyo, Japan). To assess specificity of the immunolabeling, an experiment with negative control was carried out in which sections of oviduct were labeled with colloidal gold-conjugated secondary antibody without prior labeling with the primary antibody for GnRH.
Statistical Analysis
All data are presented as mean ± SEM. Data for the relative expression of the GnRH mRNA and RFI of GnRH-immunopositive cells in the oviduct were analyzed by one-way ANOVA for significant difference between the chosen time points of pregnancy. Pairwise comparison of the RFI of GnRH-immunopositive cells between different days was conducted using Student's-Newman-Keuls multiple comparison test. Dunnett's multiple comparison test was used for the pairwise comparison in the expression pattern of GnRH mRNA during the considered time points relative to day 7. Comparisons were considered statistically significant when
Results
Localization of Immunoreactive GnRH
Our results present data on rats with confirmed pregnancies at each selected time point between postimplantation to preparturition period. Localized GnRH immunoreactivity in the luminal epithelium within the oviductal mucosa of rats was observed from day 7 through day 20 of pregnancy. The number of GnRH-immunoreactive cells during day 7 of pregnancy was much less when compared with that of days 9, 16, and 20 (Figure 1). We were unable to detect immunolabeling for GnRH in the stromal or muscularis layers of the oviduct during days 7, 9, and 20. Weak staining of the stromal layer was observed on day 16 (Figure 1). Localized immunoreactivity for GnRH was absent in the luminal epithelium, stroma, or muscularis layer in the negative control/preabsorbed control sections of the oviduct (Figure 1).
RFI
A significant increment in the RFI of the GnRH-immunopositive cells was observed (Figure 1) between days 7 and 9 (
Expression of GnRH mRNA
The relative expression of GnRH mRNA was highest in the rat hypothalamus with no expression in the rat liver (day 16: hypothalamus, 1; liver, 0; oviduct, 0.15 ± 0.01). 18S mRNA expression (used as internal standard control) was equal across samples throughout the experiment (
Collectively, both the transcription of GnRH mRNA and translation of the protein reached their peak by day 16 of pregnancy and attained a plateau thereafter by day 20. However, the increase in GnRH mRNA expression between days 7 and 9 was not significant, whereas the increase in RFI of the GnRH-immunoreactive cells between both days was statistically significant.
Immunoelectron Microscopic Studies
Negative control sections (without primary antibody) lacked specific localization of gold particles (Figure 3) in any of the subcellular organelles of the luminal epithelium or the lumen of the oviduct. Colloidal gold particles appeared to be clustered only in the putative secretory vesicles (Figures 4B, 4D, and 4F) and lamella (Figure 4) of the luminal epithelial cells in the immunolabeled sections of the oviduct. Immunogold localization of GnRH was also noted in the oviductal lumen (Figure 4F) but not in the stromal or muscularis layer of the immunostained sections.
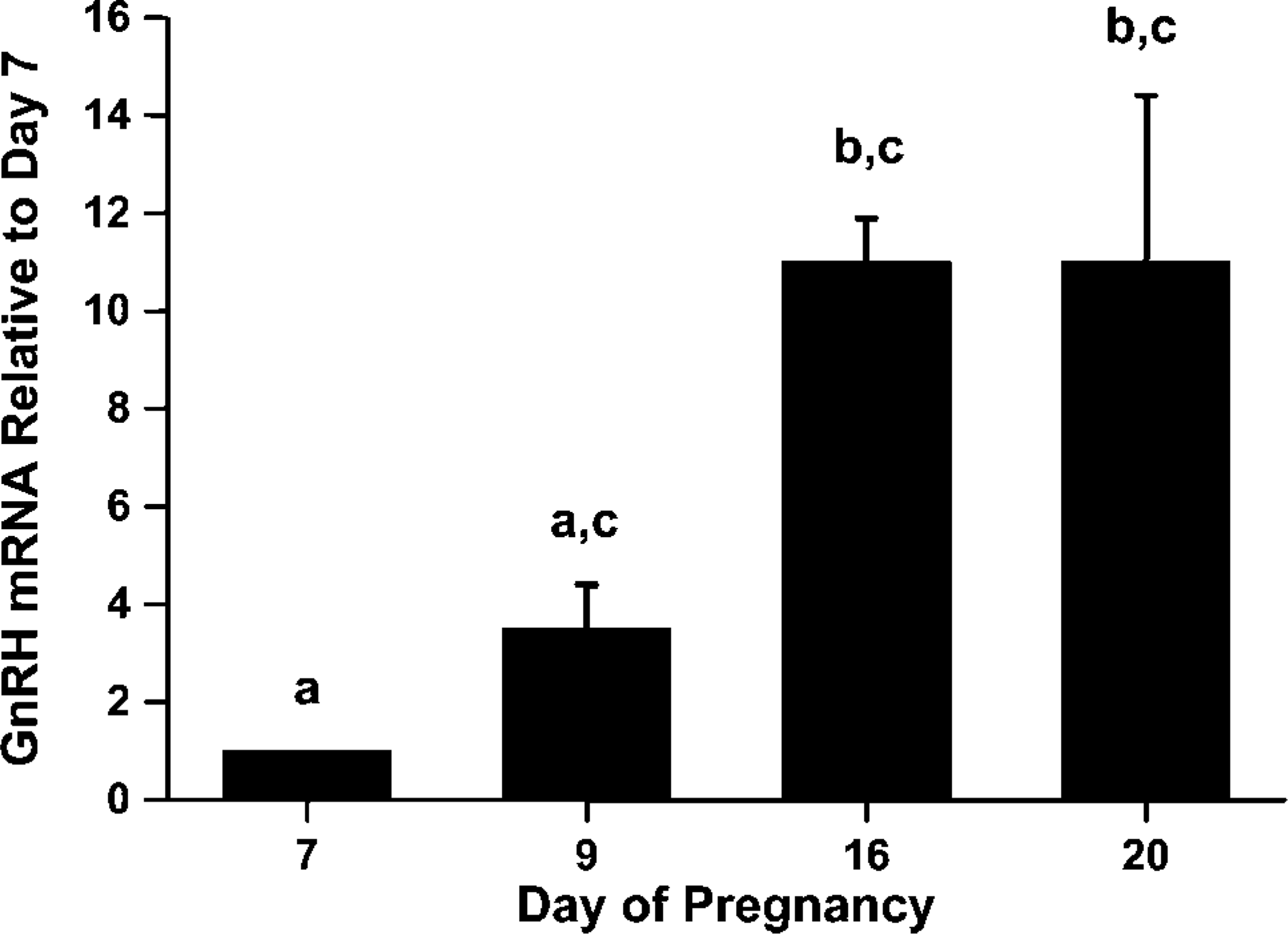
Comparison between the relative expressions of GnRH mRNA (mean ± SEM) in the rat oviduct during the chosen time points of pregnancy. Group means with different letters differ significantly (
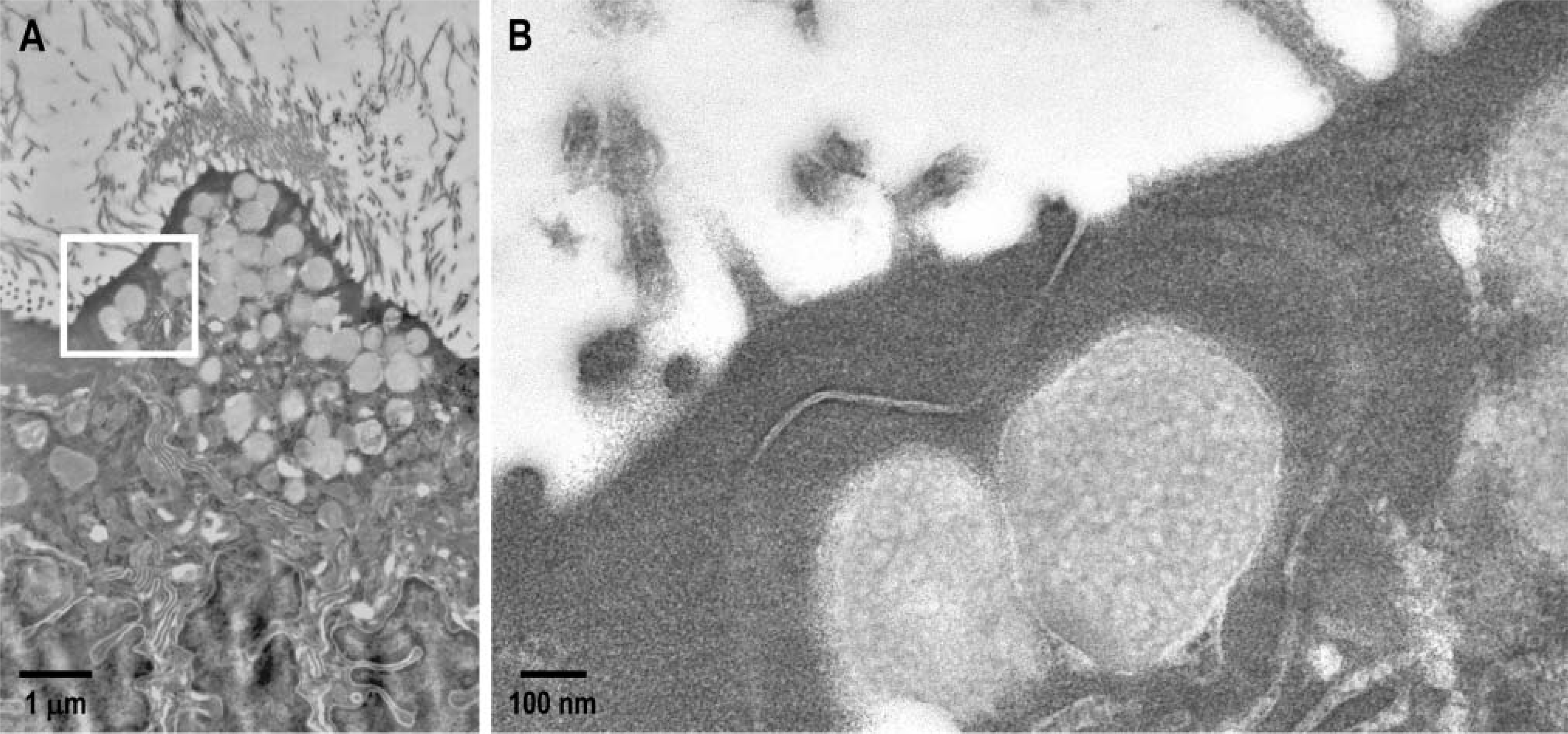
Electron micrographs showing absence of colloidal gold labeling in the subcellular organelles of the negative control section of pregnant rat oviduct. Enlarged boxed area
Discussion
The present study demonstrates, for the first time, the relative expression of GnRH mRNA and its immunolocalization in the luminal epithelium of rat oviduct during pregnancy, corresponding to a period between postimplantation to preparturition. The presence of both GnRH message as well as its translated product (protein) in the oviduct of pregnant rats during each of the time points tested demonstrates its local endogenous synthesis in the reproductive tract. Increases in GnRH mRNA and protein expression from day 7 through day 16, followed by a plateau thereafter by day 20, indicate the functional importance of this decapeptide in the maintenance of pregnancy. Labeling of GnRH in the secretory vesicles of the oviductal epithelium as well as in the lumen at the ultrastructural level is indicative of the possible release of this protein into the lumen of the oviduct.
It is well documented that the mammalian oviduct undergoes hormone-induced cyclic changes during the preovulatory period through synthesis and secretion of specific proteins, which provide a microenvironment promoting processes like fertilization, implantation, and embryonic development (Boatman 1997; Buhi et al. 1997). Earlier studies in humans have reported expression of GnRH and its mRNA in the fallopian tube epithelium but not in the stromal or muscularis layer during the luteal phase of the menstrual cycle (Casan et al. 2000). In our present study, considering the absence of immunogold labeling in the stromal layer of the oviduct on day 16 at ultrastructural level, we can assume that the level of antigen in this particular layer of the oviduct (as evident from the weak staining by immunofluorescent localization) during day 16 is negligible in comparison to its expression in the luminal epithelium. Expression of GnRH has been detected in porcine oviduct (Li et al. 1993) as well as in the oviduct of protochordates (Terakado 2001) and cephalopods like the octopus (Di Cristo et al. 2002; Ukena et al. 2004). The hypothesis that the oviductal GnRH in mammals is likely to enhance embryonic development and implantation through a paracrine action (Casan et al. 2000) is supported by the fact that preimplantation human/mouse embryos cultured in vitro exhibit a delay in development in comparison to their in vivo counterparts (Tazuki and Guidice 1996). However, this delay improves when cultured in a medium containing GnRH (Funston and Seidel 1995) or cocultured with fallopian tube epithelium (Bongso et al. 1992; Yeung et al. 1996). Increased expression of GnRH and its mRNA in the blastocyst stage, in comparison to the morula stage in cultured human and mouse embryos, as well as the expression of GnRH receptors in all embryonic developmental stages (Casan et al. 1999; Raga et al. 1999), strengthens the argument for a role of GnRH in embryonic development in mammals. In light of these observations, the presence of immunoreactive GnRH in the rat oviduct during the postimplantation period of pregnancy may suggest a function of this protein of non-neuronal origin on postimplantation embryonic development and, thus, maintenance of pregnancy in rats.
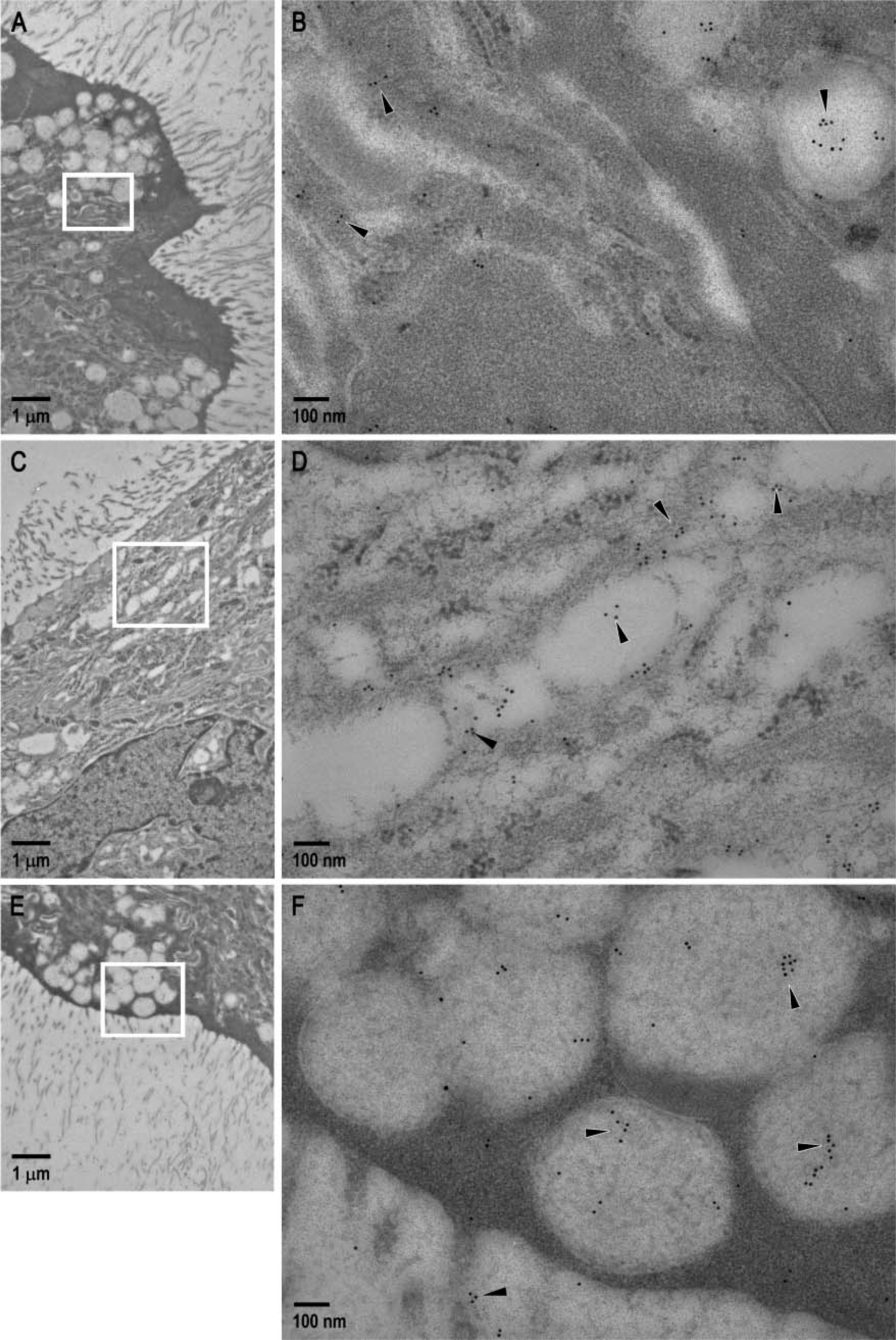
Representative colloidal gold immunoelectron micrographs of rat oviduct during pregnancy. Enlarged boxed areas in
In the present study, immunolabeling of GnRH in the putative secretory vesicles and lamella of the luminal epithelium at the ultrastructural level indicates that the epithelial cells of the rat oviduct synthesize GnRH during the postimplantation period of pregnancy. The appearance of colloidal gold particles in the lumen of the oviduct highlights the secretion of this decapeptide from the oviductal epithelium into the lumen. Involvement of oviductal epithelium of the isthmus in the secretion of an oviduct-specific high molecular mass glycoprotein during the estrous cycle in rats has been reported (Abe 1996). Reports regarding the synthesis and release of proteins/glycoproteins by oviductal epithelium in other mammals like mice (Zhang and Martin-DeLeon 2003), pigs (Buhi et al. 1993,1997), cows (Gerena and Killan 1990), and baboons (Boice et al. 1990) also exist. These proteins/glycoproteins secreted by the oviductal epithelium were found to associate with either zona pellucida of the oviductal ova or the early embryos (Kan et al. 1993), implicating their paracrine role in fertilization and early embryonic development. Involvement of the oviductal secretory proteins with the hyaluronic acid turnover in the myometrium of the uterus in mice indicates its paracrine control of uterine functions (Zhang and Martin-DeLeon 2003). Expression of the demilune cell and parotid protein mRNA in murine oviductal epithelium, which produces a 150-amino acid protein with a signal peptide (suggestive of its secretary function) during pregnancy, was found to influence embryo-maternal dialogue (Lee et al. 2006). Based on the reports of secretory activity of the oviductal epithelium, it can therefore be assumed that luminal epithelium of the oviduct may synthesize and secrete GnRH during the postimplantation period of pregnancy which, like the oviductal glycoproteins, may act either locally or at a differential site within the reproductive system, playing a direct/indirect role in the induction of embryonic growth and, thus, maintenance of pregnancy in rats. Substantiation of this statement requires further in-depth investigation.
A high level of serum progesterone is an absolute necessity for fetal survival and maintenance of pregnancy in mammals. In pregnant rats, following implantation, serum progesterone concentration starts increasing from day 7, attaining its peak by day 16 followed by a plateau, and thereafter decreasing by day 19 (Morishige et al. 1973). With the aim of establishing possible correlations between oviductal GnRH and serum progesterone levels on postimplantation processes, we chose days 7, 9, 16, and 20 of pregnancy as specific time points for this study. Surprisingly, both the expression of the oviductal GnRH and serum concentrations of progesterone during pregnancy in rats were low on day 7. However, these increased from day 7 through day 9, attaining peak values by day 16. This indicates a positive correlation between serum progesterone levels and oviductal GnRH expression up to mid-pregnancy (day 16) in rats. The relationship between endogenous peripheral GnRH and serum progesterone levels during pregnancy in mammals remains unknown. The possibility of the existence of cross talk between endogenous non-hypothalamic GnRH and progesterone secretion by the corpus luteum within the ovary of rats during pregnancy remains unexplored. However, administration of exogenous GnRH and its agonists on cultured granulosa cells of mammals like rat, monkey, and human have been reported to stimulate progesterone production (Wang and Leung 1988; Liu et al. 1991; Bussenot et al. 1993; Hori et al. 1998). Augmentation of progesterone synthesis and enhancement of pregnancy rates by the administration of a GnRH agonist (deslorelin) in lactating dairy cows during timed insemination protocols through its effects on corpus luteum functions and follicular dynamics have been established (Ambrose et al. 1998; Rajamahendran et al. 1998). Suppression of progesterone release from human placental cells following administration of GnRH antagonists by mid-gestation and its partial restoration by the addition of GnRH (Siler-Khodr et al. 1983) is indicative of the possible stimulatory role of GnRH on placental progesterone secretion during pregnancy in human. Together these reports suggest that exogenous GnRH/GnRH agonists may have a role in sustaining high levels of progesterone in mammals. Thus, it can be hypothesized that high expression of oviductal GnRH during day 16 of pregnancy may induce a positive influence on progesterone production in rats.
Expression pattern of GnRH in the oviduct by day 20 of pregnancy remains relatively high, although the concentration of serum progesterone in pregnant rats has been reported to decline sharply by day 19 (Morishige et al. 1973). This is supportive of the fact that high concentrations of oviductal GnRH toward the tail-end of pregnancy may play a role in suppressing serum progesterone levels, a necessity for parturition and, thus, termination of pregnancy. This concept is consistent with the working hypothesis of our laboratory that high concentrations of non-neuronal GnRH toward the end of pregnancy in rats are essential for luteolysis as well as the suppression of progesterone synthesis and release by the corpus luteum. Suppression of progesterone surges in vivo during late luteal phase of monkeys by long-term infusion of GnRH agonist is indicative of the inhibitory role of sustained doses of GnRH on progesterone production in mammals (Fraser and Sandow 1985). Exogenous GnRH or its agonist directly suppresses luteal steroidogenesis in mammals in general (Clayton et al. 1979; Sridaran et al. 1999; Rabiee et al. 2001; Yang et al. 2003) and follicular steroidogenesis in primates in particular (Wickings et al. 1990).
Collectively, oviductal GnRH may have a stimulatory/inhibitory role to play in progesterone production through luteotropic/luteolytic effects (as reported in human by Vaananen et al. 1997) depending upon the stage of pregnancy in rats. The exact mechanism governing the pathways by which oviductal GnRH may either promote or suppress progesterone secretion from the ovary during pregnancy in rats requires further investigation.
In conclusion, using combined real-time PCR and immunolocalization approaches, we provide evidence for the synthesis and secretion of GnRH by the oviductal epithelium of pregnant rats during a period between postimplantation and preparturition. Our present data demonstrate a correlation between expression of immunoreactive GnRH in the luminal epithelium of the oviduct and hormonal milieu of serum progesterone (as demonstrated by Morishige et al. 1973) in pregnant rats. It may thus be tempting to hypothesize the existence of either a paracrine/autocrine/endocrine mechanism, through which this decapeptide of oviductal origin may influence progesterone production and, thus, pregnancy in rat. The etiology of expression of this protein in the oviduct during pregnancy and its exact sites of action in the reproductive system (through study of the expression pattern of its receptors) along with its exact functional significance in pregnancy in rats is currently under investigation in our laboratory. Existence of any local control mechanism for the synthesis and secretion of sex steroids by reproductive tissues in rodents, similar to that of gonadotropin inhibitory hormone reported in the mammalian brain (Kreigsfeld et al. 2006), will also be a focus of future studies in our laboratory.
Footnotes
Acknowledgements
This study was supported by NIH Grants (HD-41749 to RS), RR-03034 and, in part, by NS-38194 and NS-38963 (to JW).
Our sincere thanks to the staff of the animal care facility of Morehouse School of Medicine for care of the experimental animals, Mr. L. Brako and Dr. H. Yi for assistance, Dr. X. Yao for suggestions during preparation of electron micrographs, Dr. W. Kirlin for statistical analysis, Mr. S. Pitts for assistance in confocal microscopy, and Mr. P. Abramson for technical support.
