Abstract
Interleukin-33 (IL-33) is an IL-1 family cytokine with pleiotropic effects on diverse cell types. Dysregulated IL-33 signaling has been implicated in pregnancy-related disorders, including preeclampsia and recurrent pregnancy loss, and in ovarian function in women undergoing controlled ovarian stimulation for in vitro fertilization. To date, expression of IL-33 and its receptor subunit, ST2, in the female reproductive tract remains poorly characterized. We identify IL-33-expressing oocytes surrounded by ST2-expressing granulosa cells at all stages of follicular development, in addition to IL-33+ and ST2+ non-endothelial cells in the ovarian stroma and theca layer in ovaries from adult mice. These expression patterns are similar in estrus- and diestrus-stage adults and in pubescent mice, suggesting a role for IL-33 signaling in ovarian function throughout development and in the estrous cycle. In the uterus, we find expression of IL-33 and ST2 in glandular and luminal epithelia during estrus and at the initiation of pregnancy. Uterine IL-33 expression was modulated by the estrous cycle and was reduced in pubescent females. Last, superovulation increases transcripts for IL-33 and the soluble form of ST2 (sST2) in ovaries, and for IL-33 in uteri. Collectively, our findings lay the foundation for studies identifying cell type-specific requirements for IL-33/ST2 signaling in the establishment and maintenance of mouse pregnancy.
Introduction
The interleukin-1 (IL-1) family member, IL-33, is a potent cytokine with pleiotropic effects on diverse cells in the body. 1 Originally described as an endothelial cell-derived factor, IL-33 can be produced by a broad array of cell types, including epithelial cells, fibroblasts, muscle cells, neurons, and immune cells. IL-33 is unusual in its role as both a nuclear factor with poorly understood roles in transcriptional regulation and as a secreted cytokine. 2 IL-33 protein typically remains localized in the nucleus until appropriate extracellular cues, for example, inflammation caused by infection or tissue damage, trigger its release into the extracellular milieu. IL-33 release can occur via active secretion processes or as a result of cell death, depending on the cell type and setting. Once released, IL-33 acts as a cytokine on target cells that express the heterodimeric IL-33 receptor (IL-33R), comprised of ST2 and the IL-1 receptor accessory protein (IL-1Rap). In some settings, ST2 can be secreted; this soluble form of ST2 (sST2), which lacks the transmembrane and intracellular regions of ST2, is thought to act as a decoy receptor to block or modulate IL-33 signaling. 1 Recent studies suggest that IL-33 signaling mediates both physiological and pathological reproductive processes.3–6
In both mice and humans, ovulation after ovarian stimulation with exogenous gonadotropins has been associated with changes in IL-33 and/or ST2 expression.4,6 In mice, transiently elevated ovarian expression of both IL-33 and ST2 was observed 6–9 hr after administration of human chorionic gonadotropin (hCG), suggesting that IL-33 signaling may modulate ovulation-related inflammation. 4 Human ovarian production of sST2, the decoy receptor, but not IL-33, was increased by controlled ovarian stimulation (COS) used for in vitro fertilization (IVF). 6 It is notable that higher levels of sST2 were detected in follicular fluid surrounding oocytes that produced better quality embryos, suggesting an association with oocyte quality. 6 Based on ex vivo experiments, luteinized human granulosa cells are the proposed source of sST26; the ovarian cell type(s) that produces ST2 in mice has not been reported. Although limited in scope, these data across species identify possible roles for IL-33 signaling in ovarian function following ovarian stimulation, which is used to enhance chances of pregnancy.
Recent studies also highlight a role for IL-33 signaling in pregnancy outcomes after natural cycle conception. Dysregulated IL-33 signaling has been implicated in several pregnancy-related disorders in humans, such as recurrent pregnancy loss (RPL) and preeclampsia, and in neonatal mortality in mice. Specific polymorphisms in the
Notwithstanding these suggestive associations between IL-33 signaling and pregnancy outcomes, expression patterns of IL-33 and ST2 are not well characterized in the female reproductive tract. The objective of this study was to identify which cell types produce and could respond to IL-33 in the mouse ovary and uterus and to determine the impact of superovulation (SO) on sST2 production. Herein, we define the expression of IL-33 and ST2 in ovaries and uteri of pubescent female mice, mature female mice at specific stages of the estrous cycle, and at the initiation of pregnancy. Our data provide a framework for genetic studies that will identify cell type-specific requirements for IL-33 signaling in the establishment and maintenance of mouse pregnancy.
Materials and Methods
Mice
The Rutgers University Institutional Animal Care and Use Committee approved animal studies. Pubescent C57BL/6 female mice (28 days old) and adult C57BL/6 virgin female mice (8–12 weeks of age) were obtained from the Jackson Laboratory. Ovaries and uteri were collected from pubescent female mice and adult female mice in the estrus and diestrus stages of the estrous cycle. For studies involving mature mice, estrous cycles were monitored by analysis of vaginal cytology recovered from daily vaginal swabs, as previously described. 10
For studies involving pregnancy in natural cycles (NCs), estrus-stage adult female mice were bred with C57BL/6 males of proven fertility. Noon on the day a mating plug was observed was designated embryonic day (E) 0.5. Ovaries and uteri were harvested and blood was collected in early pregnancy at E0.5 and E2.5.
For studies comparing pregnancies conceived in SO cycles with those in NCs, estrus-stage adult females were selected. To induce SO, mature female mice were injected intraperitoneally (i.p.) with 5 IU of pregnant mare serum gonadotropin (P237; Leinco Technologies, St. Louis, MO) in PBS and then with 5 IU of hCG (CG10-1VL; Sigma Aldrich, Milwaukee, WI) 46 hr later. Two hours after hCG administration, SO females and estrus-stage females (NC controls) were mated with C57BL/6 males of proven fertility. Ovaries and uteri were harvested and blood was collected at E0.5.
For immunostaining, ovaries and 3–5 mm pieces of uteri from non-pregnant female mice and pregnant females at E0.5 and E2.5 were embedded in Tissue-Tek O.C.T. Compound (Sakura Fine Technical, Torrance, CA), snap-frozen on dry ice, and stored at −80°C. For quantitative RT-PCR (qRT-PCR), ovaries and 3–5 mm pieces of uteri were snap-frozen on dry ice and stored at −80C. Each assay was performed on four to eight different female mice at each stage of analysis.
Immunostaining Controls
Immunofluorescence (IF) staining of tissue sections from mouse spleen 11 and human placentas5,12 was performed to establish external positive controls for primary antibodies used to detect IL-33 and ST2 proteins, respectively. Primary antibodies were omitted to detect nonspecific binding of the secondary antibodies (Supplementary Fig. 1A to G). Purified goat immunoglobulin G (IgG) and purified rabbit IgG were used as isotype-specific negative controls (Supplementary Fig. 1H to K).
Immunostaining and Histology
A full thickness human placental biopsy was collected from healthy, pregnant women with uncomplicated full-term pregnancies (n=3) undergoing cesarean delivery. The institutional review board at Columbia University Medical Center (IRB-AAAN4357) approved the study protocol, and all participants gave their signed informed consent. 7 μm frozen sections were generated from C57BL/6 adult mouse spleen and 10 μm frozen sections were generated from the placental biopsies.
To detect IL-33 and ST2 expression in mouse uteri and ovaries, frozen sections at 7 μm were generated for each piece of uterus. Each ovary was sectioned at 5 μm. For histology, sections were stained with hematoxylin and eosin (H&E). To identify cell types expressing IL-33 and ST2, specific staining for IL-33 and ST2 was performed at least three times on five different sections, spaced at 100 μm intervals along the tissue. Sections were stained as previously described. 13
Sections were incubated with isotype-specific controls at the same final concentration as the primary antibody. The following antibodies were used: goat IgG isotype control (02-6202, Thermo Fisher Scientific, Kalamazoo, MI) and rabbit IgG isotype control (02-6102, Thermo Fisher Scientific).
For IF, primary antibodies included goat anti-mouse IL-33 (1:50, AF3626; R&D Systems, Minneapolis, MN), rabbit anti-mouse ST2 (1:200, PA5-23316; ThermoFisher Scientific, Rockford, IL), rabbit anti-mouse ST2 (1:200, PRS3363; Sigma Aldrich, St. Louis, MO), rat anti-mouse CD31 (1:350, 553370; BD Biosciences, San Jose, CA), rabbit anti-mouse pan-cytokeratin (1:1000, Z0622, Dako, Carpinteria, CA), mouse anti-human CD31 (1:200, MS-353-S0, ThermoFisher Scientific), rat anti-mouse E-cadherin (1:200, M108, Takara Bio Inc, Mountain View, CA), and rabbit monoclonal anti-FOXA2 (1:200, ab108422, Abcam, Cambridge, MA). The following secondary antibodies (ThermoFisher Scientific) were used: donkey anti-goat IgG Alexa-Fluor 594 (A11058), donkey anti-goat IgG Alexa-Fluor 488 (A32814), donkey anti-rat IgG Alexa-Fluor 594 (A21209), donkey anti-rat IgG Alexa-Fluor 488 (A21208), donkey anti-rabbit IgG Alexa-Fluor 594 (A21207), donkey anti-rabbit IgG Alexa-Fluor 488 (A21206). Stained sections were covered with Vectashield containing 4′,6-diamidino-2-phenylindole (DAPI; H-1200, Vector, Burlingame, CA) for nuclear visualization.
For immunohistochemistry (IHC) staining, primary antibodies were rat anti-mouse CD31 and goat anti-mouse IL-33 and secondary antibodies were biotinylated rabbit anti-rat IgG (1:200, BA-4001; Vector) and biotinylated rabbit anti-goat IgG (1:400, BA-5000; Vector). The avidin/biotin blocking kit (SP-2001; Vector), the Vectastain ABC kit, and DAB substrate kit (SK-4100; Vector) were used. Sections were counterstained with methylene green and mounted with Permount (SP15-100, Fisher Scientific, Fairlawn, NJ).
Imaging
Images of H&E, IF, and IHC samples were taken with the Keyence BZ-X700 All-in-One fluorescence microscope (Keyence, Osaka, Japan). Standard filters were used to image DAPI, Alexa Fluor 488, and Alexa Fluor 594. Images were taken using the 10×, 20×, and 40× objectives. Images were captured using Keyence BZ-X Viewer version 01.03 software.
Evaluation of IL-33 Expression in Estrus-stage and Diestrus-stage Uteri
To quantitatively assess IL-33 expression, three uterine cross sections, equally spaced along each 3 mm piece of estrus- and diestrus-stage uterus were stained and imaged with a 40× objective. To compare the percentage of IL-33+ luminal cells in estrus and diestrus, the ratio of IL-33+ cells to the total number of DAPI+ cells in 0.23 mm2 lumen was determined for each uterine cross section. The average of three uterine cross sections yielded the percentage of IL-33+ luminal cells for that animal. To calculate the percentage of IL-33+ cells in the glandular epithelium, the total number of IL-33+ cells in 100–150 glandular cells was determined for each uterine cross section. The average of three uterine cross sections yielded the percentage of IL-33+ luminal cells for that animal. ImageJ software (http://rsb.info.nih.gov/ij/) was used to quantify positive cells.
qRT-PCR Analysis
Ovaries and uteri were homogenized using the Kontes Duall tissue grinder to produce a uniform homogenate (885450-0020; DWK Life Science, Millville, NJ). Total ribonucleic acid (RNA) was extracted from ovaries and uteri using the Qiagen RNeasy plus mini kit (74134; Qiagen, Frederick, MD). Complementary deoxyribonucleic acid (cDNA) was synthesized using the qScript cDNA synthesis kit (95047-100, Quantabio, Beverly, MA) and gene expression was determined by qRT-PCR using the QuantiNova SYBR Green PCR Kit (208056, Qiagen). The following primer sequences were used: 18s forward: CCGGGCTTCTATTTTGTTGGT and reverse: TAGCGGCGCAATACGAATG;
Serum Progesterone Immunoassay
Levels of progesterone were measured in serum samples from pregnant female mice at E0.5 and E2.5. Serum progesterone levels were measured using an Electrochemiluminescence immunoassay (Cobas e411 Analyzer, Roche Diagnostics, Indianapolis, IN). The detectable progesterone range was 0.03–60 ng/ml. Intra- and interassay coefficients of variation did not exceed 10%. Each assay was performed on sera from four to seven different female mice.
Enzyme-linked Immunosorbent Assay (ELISA) for Circulating ST2
Levels of circulating ST2 were quantified in serum samples from pregnant female mice at E0.5 and E2.5. Serum ST2 levels were quantified using a ST2 (IL-33R) mouse ELISA Kit (ThermoFisher Scientific). The detectable ST2 range was 80–10,000 pg/ml. Each assay was performed on sera from four to five different female mice.
Statistics
Non-parametric statistical analyses were performed with Prism v8.0 (GraphPad Software, La Jolla, CA, USA). We analyzed these data to identify outliers. Medians were compared using the Mann–Whitney
Results
Expression of IL-33 and ST2 in Ovaries and Uteri of Sexually Immature Mice
Before the onset of puberty, which occurs between 24 and 30 days of age in mice, ovaries contain follicles at all stages of development and uteri have an architecture that is similar to that of the adult uterus; the glands are sinuous and coiled, but lack extensive branching.16,17 To determine the expression of IL-33 and its receptor subunit, ST2, before sexual maturity, we stained ovaries and uteri from 28-day-old mice (Fig. 1, Table 1). Stages of ovarian follicle development are readily detected by H&E staining (Fig. 1A, secondary and antral follicles). Endothelial cell marker, CD31, identifies ovarian vasculature (Fig. 1B). Immunostaining revealed IL-33 expression in oocytes of primordial (Fig. 1D), secondary (Fig. 1C and F, asterisks), and antral ovarian follicles (Fig. 1C, arrow). ST2 expression was evident in granulosa cells surrounding each of the IL-33-expressing oocytes, as well as on numerous non-endothelial (CD31–) cells in the ovarian stroma (Fig. 1E and F). In the pubescent uterus, we observed IL-33 expression in a few luminal epithelial cells (Fig. 1G). In contrast, we found that all glandular epithelial cells express ST2 (Fig. 1H). We found both IL-33 and ST2 in a small subset of cells in the uterine myometrium. Taken together, these data suggest that IL-33 signaling within ovarian follicles and in uteri of pubescent female mice may contribute to ovarian and uterine function before sexual maturity.
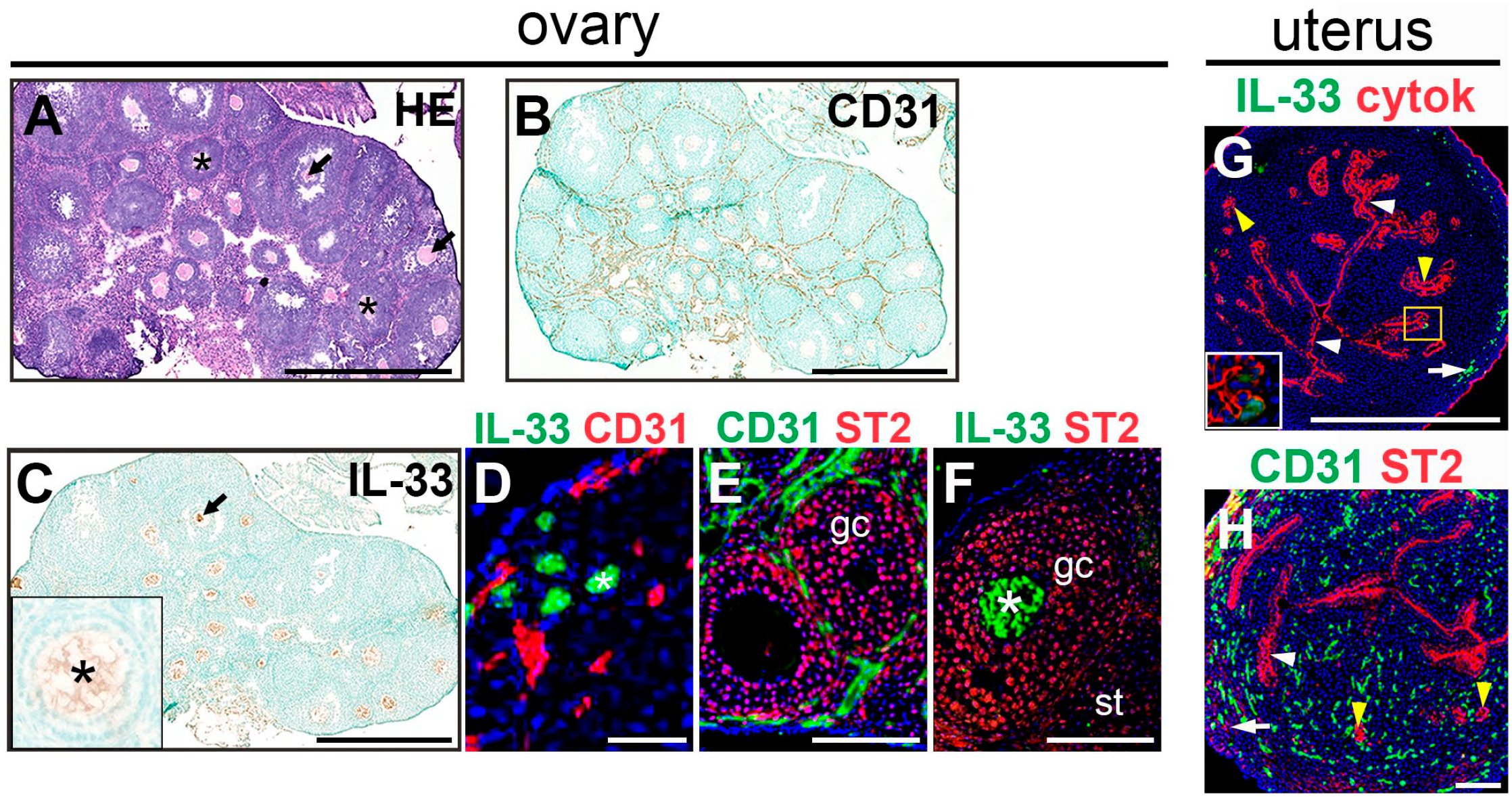
Expression of interleukin-33 (IL-33) and its receptor subunit, ST2, in pubescent ovaries and uteri. (A) Representative image of an H&E stained ovary. Asterisks identify oocytes in secondary follicles. Arrows identify oocytes in antral follicles. (B–F) Representative images of immunostained ovaries. (B, D, E) CD31 expression identifies endothelial cells associated with vasculature in the ovary. (C, D, F) IL-33 is expressed in oocytes of primordial (asterisk in D), secondary (asterisk in inset of C; asterisk in F) and antral follicles (C, arrow). (E, F) ST2 is expressed in granulosa cells (gc) of follicles and in the stroma (st). (G, H) Representative images of immunostained uteri. (G) Pan-cytokeratin (cytok) expression identifies glandular (yellow arrowheads) and luminal (white arrowheads) epithelia. IL-33 is detected in the myometrium (white arrow). A few IL-33+ cells are detected in the luminal epithelium (yellow box, inset). (H) CD31 expression identifies endothelial cells associated with vasculature in the uterine stroma and myometrium. ST2 is expressed in glandular (yellow arrowheads) and luminal (white arrowheads) epithelia. ST2 is detected in the myometrium (white arrow). Abbreviation: HE, hematoxylin & eosin; Bars = 500 µm (A–C, G), 50 µm (D), and 100 µm (E, F, H).
Summary of IL-33 and ST2 Expression in Pubescent and Sexually Mature Ovaries and Uteri.
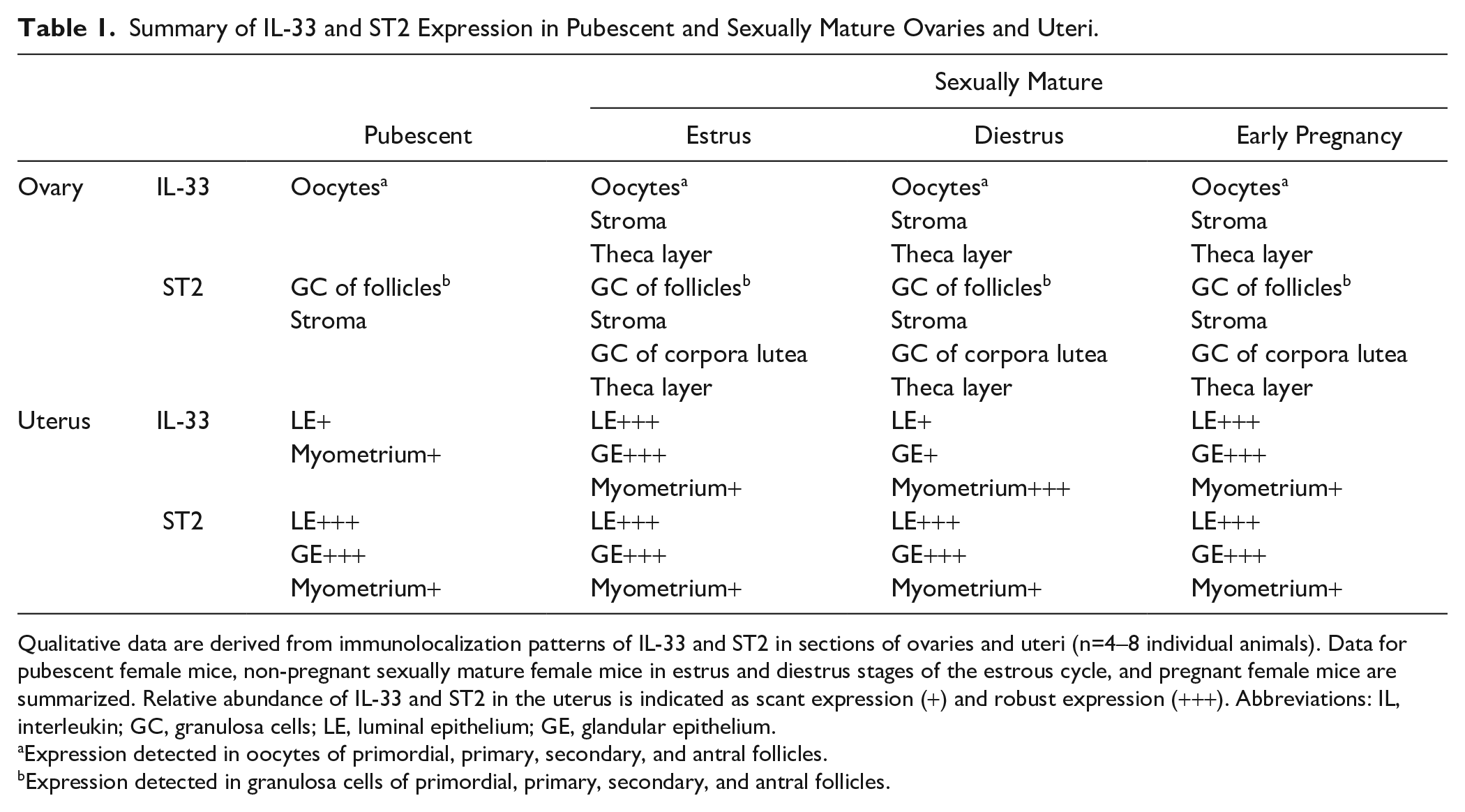
Qualitative data are derived from immunolocalization patterns of IL-33 and ST2 in sections of ovaries and uteri (n=4–8 individual animals). Data for pubescent female mice, non-pregnant sexually mature female mice in estrus and diestrus stages of the estrous cycle, and pregnant female mice are summarized. Relative abundance of IL-33 and ST2 in the uterus is indicated as scant expression (+) and robust expression (+++). Abbreviations: IL, interleukin; GC, granulosa cells; LE, luminal epithelium; GE, glandular epithelium.
Expression detected in oocytes of primordial, primary, secondary, and antral follicles.
Expression detected in granulosa cells of primordial, primary, secondary, and antral follicles.
Cell Type-specific Expression of IL-33 and ST2 Is Similar in Estrus-stage and Diestrus-stage Ovaries
The estrous cycle in mice is divided into four stages: proestrus, estrus, metestrus, and diestrus. Circulating levels of ovarian-derived estradiol and progesterone fluctuate during the estrous cycle. Estradiol levels peak just before ovulation, which occurs in the transition from proestrus to estrus, and progesterone levels peak in diestrus.16,18 As the estrous cycle in mice is a short reproductive cycle (4–5 days), all stages of follicular development and corpora lutea are present in estrus, metestrus, and diestrus, although diestrus contains corpora lutea that remain from the previous ovulation. H&E staining and CD31 expression identify ovarian follicles and corpora lutea in ovaries from cycling, reproductive-age mice (Fig. 2A and B). We assessed IL-33 and ST2 expression in estrus and diestrus stages (Table 1).
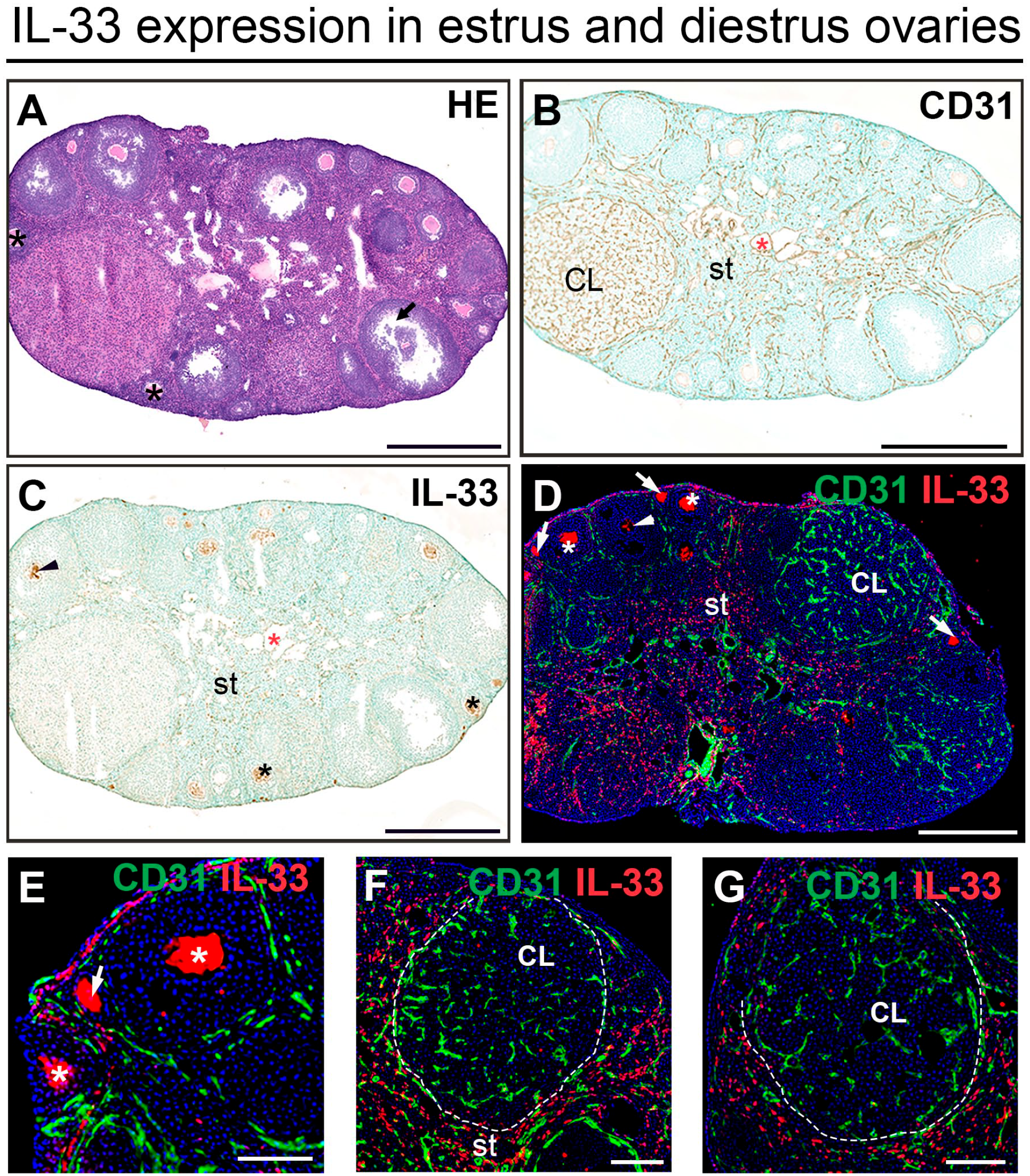
Expression of IL-33 in estrus- and diestrus-stage ovaries. (A) Representative image of an H&E stained diestrus-stage ovary. Black asterisks identify secondary follicles. Arrows identify antral follicles. (B–G) Representative images of diestrus (B, C) and estrus (D–G) immunostained ovaries. (B) CD31 expression identifies endothelial cells associated with vasculature (red asterisk) in the ovarian stroma (st), in the theca layer around ovarian follicles and in corpora lutea (CL) in the diestrus stage. (C) In diestrus, IL-33 is expressed in oocytes (arrowheads and black asterisks), in the ovarian stroma (st) and around the vasculature (red asterisk) in the stroma. (D–G) Double staining IF showing localization of IL-33 and CD31 in estrus. IL-33 is expressed in oocytes of primary (arrows), secondary (asterisks), and antral (arrowheads) follicles. IL-33 is expressed in the ovarian stroma (st). IL-33 is associated with the theca layer (white lines) around follicles and corpora lutea. Abbreviations: IL, interleukin; IF, immunofluorescence; HE, hematoxylin & eosin; Bars = 500 µm (A–D) and 100 µm (E–G).
Immunostaining revealed IL-33 expression in oocytes of primary (arrows in Fig. 2D and E), secondary (asterisks in Fig. 2C and D), and antral follicles (arrowheads in Fig. 2C and D) and ST2 in granulosa cells surrounding the IL-33+ oocytes (Fig. 3A and B). ST2 expression was maintained in luteinized granulosa cells in the corpora lutea (Fig. 3C). Both IL-33 (Fig. 2C, E, and F) and ST2 (Fig. 3A and B) were expressed in the ovarian stroma. Notably, we did not find estrous cycle-dependent differences in IL-33 and ST2 expression in follicles and corpora lutea in ovaries from mice in estrus and diestrus. Altogether, these findings suggest that a role for IL-33 signaling in oocyte-to-granulosa cell crosstalk in follicles, and in the function of luteinized granulosa cells post-ovulation warrants further investigation.
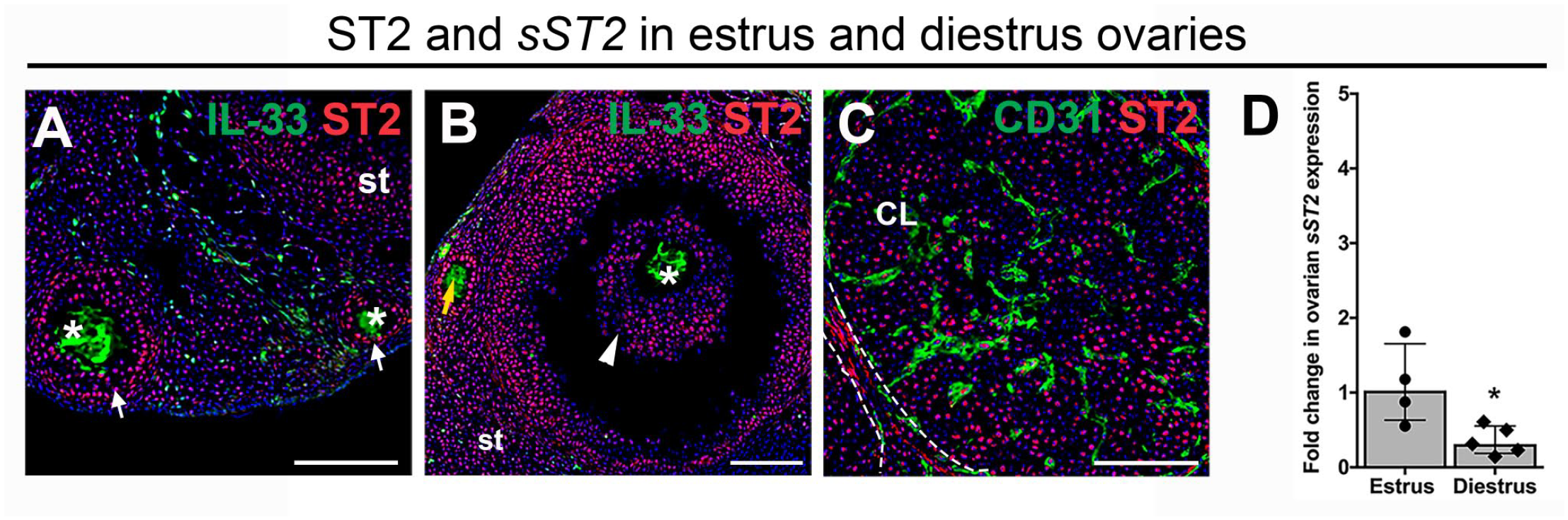
Expression of ST2 and
We observed similar expression of IL-33 and ST2 in the ovarian stroma and follicles of sexually immature pubescent and reproductive-age mice (Figs. 2 and 3). Whereas no IL-33+ or ST2+ cells were apparent in the theca layer in pubescent ovaries (Fig. 1C, E, and F), numerous IL-33+ cells and some ST2+ cells were detected in the vascularized theca layer surrounding follicles and corpora lutea in reproductive-age ovaries (Figs. 2F, G, and 3C). Although endothelial cells are known to produce IL-33 in other tissues, we did not observe any cells that coexpressed IL-33 and the vascular marker, CD31, in either pubescent or estrus-stage ovaries, indicating that endothelial cells may not be direct participants in ovarian IL-33 signaling. Together, our findings of similar expression of IL-33 and ST2 in the stroma and follicles, and differing expression of IL-33 and ST2 in the theca layer, of pubescent compared with reproductive-age mice suggest that IL-33 signaling may contribute to ovarian function throughout sexual development, with distinct roles acquired in sexual maturity.
In addition to existing as a membrane-associated receptor, ST2 can also be produced as a truncated, secreted protein (sST2) capable of binding and neutralizing IL-33 in the extracellular milieu. Ovarian stimulation with exogenous gonadotropins has been shown to increase ovarian production of sST2,4,6 raising the possibility that reproductive hormones may regulate physiological sST2 levels in the ovaries. We therefore determined whether sST2 expression in the ovary changes throughout the estrous cycle. Using qRT-PCR, with primers specific for the truncated sST2 form, 19 we found decreased expression of transcripts for sST2 in ovaries from diestrus-stage compared with estrus-stage mice (Fig. 3D). These observations suggest that differences in reproductive hormones at distinct stages of the estrous cycle may regulate sST2 levels and serve as a mechanism to modulate local IL-33 signaling in the ovary.
Uterine Expression of IL-33 and ST2 Is Modulated by the Estrous Cycle
We next sought to define the expression of IL-33 and ST2 in the mature, reproductive-age uterus, as well as determine whether uterine expression of IL-33 or ST2 is modulated by the estrous cycle. IF experiments revealed IL-33 expression in pan-cytokeratin-positive luminal and glandular epithelial cells in the estrus-stage uterus (Fig. 4B and G). Moreover, ST2-expression was also abundant in the estrus luminal and glandular epithelium, consistent with a potential role for autocrine IL-33 signaling in the function of these cells (Fig. 4E and G). Notably, only minor populations of IL-33+ or ST2+ cells were detected in the myometrium in estrus (Fig. 4B and E). Comparison of uteri from mice in estrus and diestrus revealed striking estrous cycle-dependent differences in IL-33. IL-33 was markedly reduced in the luminal epithelia (Fig. 4C and G), and to a lesser extent the glandular epithelia (Fig. 4C and H) during diestrus in contrast to its abundant expression in these regions during estrus. To quantitatively assess IL-33 expression, we calculated the percentage of IL-33+ cells in the lumen and glands of estrus- and diestrus-stage uteri. The percentage of IL-33+ cells was significantly lower in diestrus-stage lumens compared with estrus-stage lumens (Fig. 4I). We also observed a marked increase in IL-33-expressing cells in the myometrium of the uterus during diestrus, as compared with estrus (Fig. 4B and C). In contrast, we did not observe estrous cycle-dependent differences in ST2 expression (Fig. 4E–H). Expression of ST2 in both luminal and glandular epithelia was readily apparent in estrus and diestrus uteri, despite the collapsed, atrophic architecture of endometrial glands in diestrus.
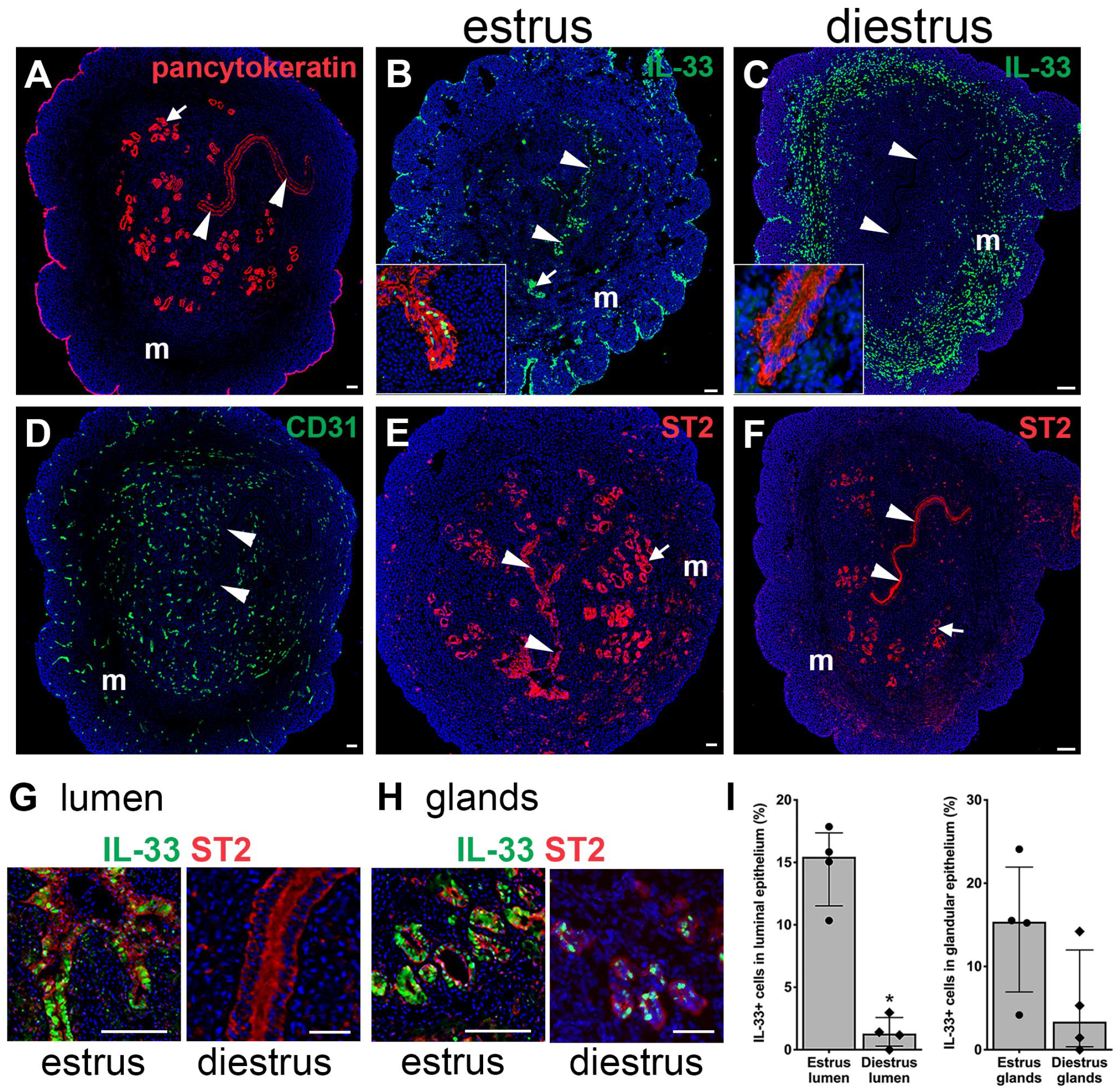
Estrous-stage alters IL-33, but not ST2 expression in uteri. (A–H) Representative images of immunostained uteri. A diestrus-stage uterine section showing pan-cytokeratin expression (A) in glandular (arrow) and luminal (arrowheads) epithelia and CD31 (A, D) expression in endothelial cells. In estrus, IL-33 (B) and ST2 (E) are expressed in luminal (white arrowheads) and glandular (white arrows) epithelia. The inset in B shows expression of IL-33 (green) and pan-cytokeratin (red) in an estrus-stage lumen. In diestrus, IL-33 (C) is not expressed in luminal epithelia (white arrowheads) but is increased in the myometrium (m). The inset in C is a diestrus lumen showing pan-cytokeratin (red) and lack of expression of IL-33 (green). In diestrus, ST2 (F) is expressed in luminal (white arrowheads) and glandular (white arrows) epithelia. Magnified images of lumen (G) and glands (H) highlight IL-33 and ST2 expression. To calculate the percentage of IL-33+ luminal cells, the ratio of IL-33+ cells to the total number of DAPI+ cells in 0.23 mm2 lumen was determined. To calculate the percentage of IL-33+ cells in the glandular epithelium, the total number of IL-33+ cells in 100–150 glandular cells was determined. (I) Compared with estrus, IL-33 expression during diestrus was lower in luminal but not glandular epithelium. Data are shown for each individual animal analyzed (n=4 animals) with median and interquartile range. Abbreviations: IL, interleukin; DAPI, 4′,6-diamidino-2-phenylindole. *
Comparison of uteri from sexually immature and reproductive-age females revealed similar expression patterns of ST2 by luminal and glandular epithelial cells. In contrast, IL-33 was notably absent in glandular epithelial cells in immature mice, similar to findings in diestrus-stage sexually mature mice and distinct from the IL-33 expression by uterine epithelia in estrus-stage mature mice (Table 1).
Expression of IL-33 and ST2 in Early Pregnancy Resembles Estrus-stage Uteri
Given the correlation between altered IL-33 signaling and human pregnancy complications associated with abnormal implantation and placentation, such as preeclampsia and RPL,5,7,9,20 we sought to determine IL-33 and ST2 expression in pregnancy, with a focus on pre-implantation stages (Table 1). Similar to findings in estrus-stage non-pregnant mice, ovaries from mice at E0.5 and E2.5 contained IL-33-expressing oocytes, surrounded by ST2-expressing granulosa cells, in primordial and secondary follicles (Fig. 5A and C, data not shown for E2.5). ST2+ and IL-33+ cells were also abundant in the stroma and theca layer surrounding the follicles and corpora lutea, with many IL-33+ cells adjacent to CD31+ endothelial cells (Fig. 5A and B, data not shown for E2.5). At E0.5 and E2.5, IL-33 and ST2 expression patterns in the uterus were similar to those observed in estrus-stage, non-pregnant uteri (Fig. 5D–G). Specifically, IL-33 and ST2 expression was maintained in the luminal and glandular epithelium, and IL-33+ cells were scattered throughout the myometrium. In the lumen, co-staining with epithelial markers revealed that all E-cadherin-positive luminal epithelial cells express ST2, but not all pan-cytokeratin-positive luminal epithelial cells express IL-33. In the glands, IL-33 was expressed by a subset of FoxA2+ glandular epithelial cells, but all E-cadherin-positive glandular epithelial cells expressed ST2. IL-33 and ST2 expression in the pre-implantation uterus is similar to that in the post-ovulatory, estrus-stage uterus. These data suggest that IL-33/ST2 crosstalk may be important in preparing the uterus for embryo implantation.
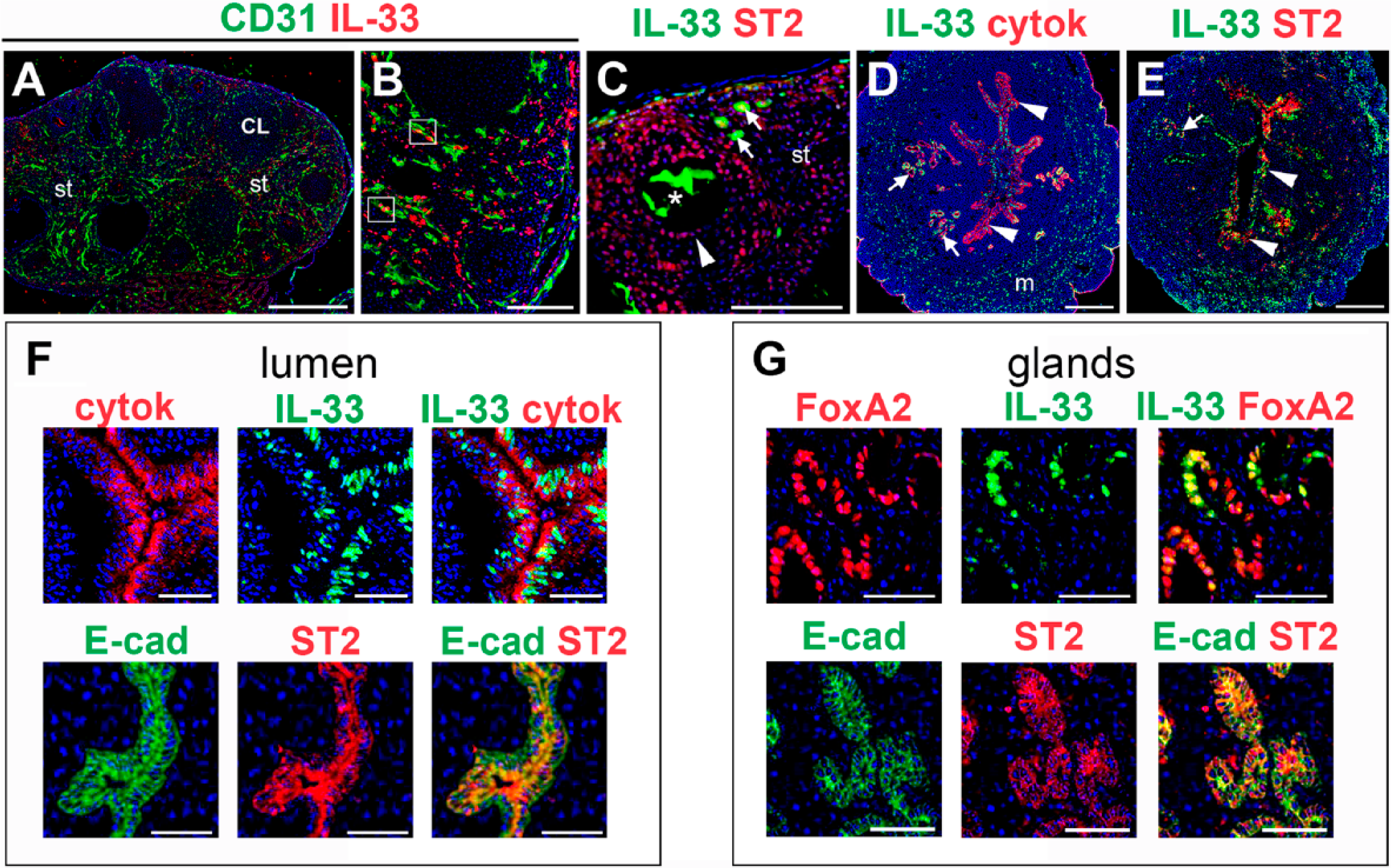
Expression of IL-33 and ST2 in ovaries and uteri in early pregnancy. (A–C) Representative images of immunostained ovaries. (A–C) At E0.5, IL-33 is expressed in the stroma (st) and in the theca layer surrounding follicles and corpora lutea. Cells expressing IL-33 are adjacent to endothelial cells expressing CD31 (white boxes in B). (C) IL-33 is expressed in oocytes of primordial follicles (arrows) and in oocytes (asterisk) of secondary follicles (arrowhead). ST2 is expressed in granulosa cells and in the stroma (st). (D–G) Representative images of immunostained uteri in early pregnancy. Pan-cytokeratin (cytok) expression identifies glandular (arrows) and luminal (white arrowheads) epithelia. (D) At E0.5, IL-33 is detected in the myometrium (m) and in the glandular (arrows) and luminal (arrowheads) epithelium. (E) At E0.5, ST2 is expressed in the luminal (arrowheads) and glandular (arrows) epithelia. (F) At E2.5, co-staining of uteri with epithelial markers, pan-cytokeratin (cytok), and E-cadherin (E-cad) shows ST2 expression in all luminal epithelium and IL-33 in a subset of those cells. (G) At E2.5, co-staining of the uteri with glandular marker FoxA2 and E-cadherin shows ST2 expression in all glandular epithelial cells and IL-33 in a subset of those cells. Abbreviation: IL, interleukin; Bars = 500 µm (A), 100 µm (B, C) and 50 µm (D–G).
SO Perturbs Local lL-33 and ST2 Expression
Ovarian stimulation with exogenous gonadotropins is used to increase the number of oocytes available to establish pregnancy. However, the exogenous gonadotropins and/or products secreted by hyperstimulated ovaries have been shown to negatively affect endometrial function and are linked to decreased implantation rates in mice21–23 and humans.24–26 Given that COS increased ovarian production of sST2 in humans 6 and ovarian expression of IL-33 and its full-length, membrane-associated receptor, ST2, peaked 6–9 hr after hCG injection in SO cycles in mice, 4 we sought to investigate the impact of SO on the expression of local and circulating IL-33 and ST2. For these studies, we first characterized IL-33 and sST2 expression in early pregnancy at pre-implantation time points, E0.5 and E2.5, and then determined how SO altered IL-33 and sST2 expression.
To assess changes in IL-33 and sST2 in the context of known pregnancy-related factors, we also determined expression levels for ovarian steroidogenic acute regulatory protein (StAR) and vascular endothelial growth factor A (VEGFa), uterine PR, and serum progesterone. Whereas ovarian expression of transcripts for StAR and VEGFa were similar in NCs at E0.5 and E2.5 (Fig. 6A), uterine expression of transcripts for PR and serum progesterone levels were significantly higher at E2.5 as compared with E0.5 (Figs. 6B and 7A). In NCs, we found that ovarian expression of
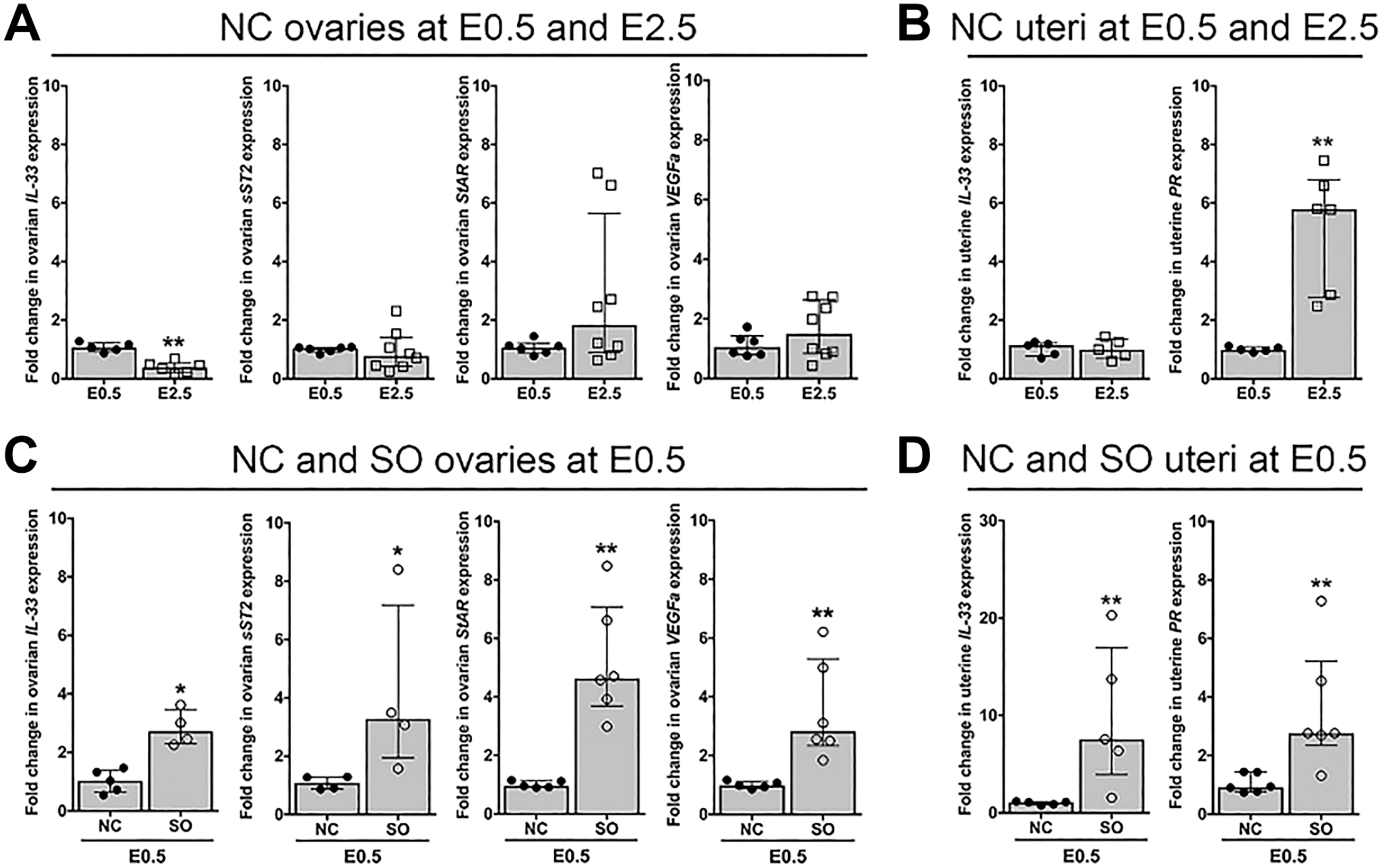
Superovulation increases local
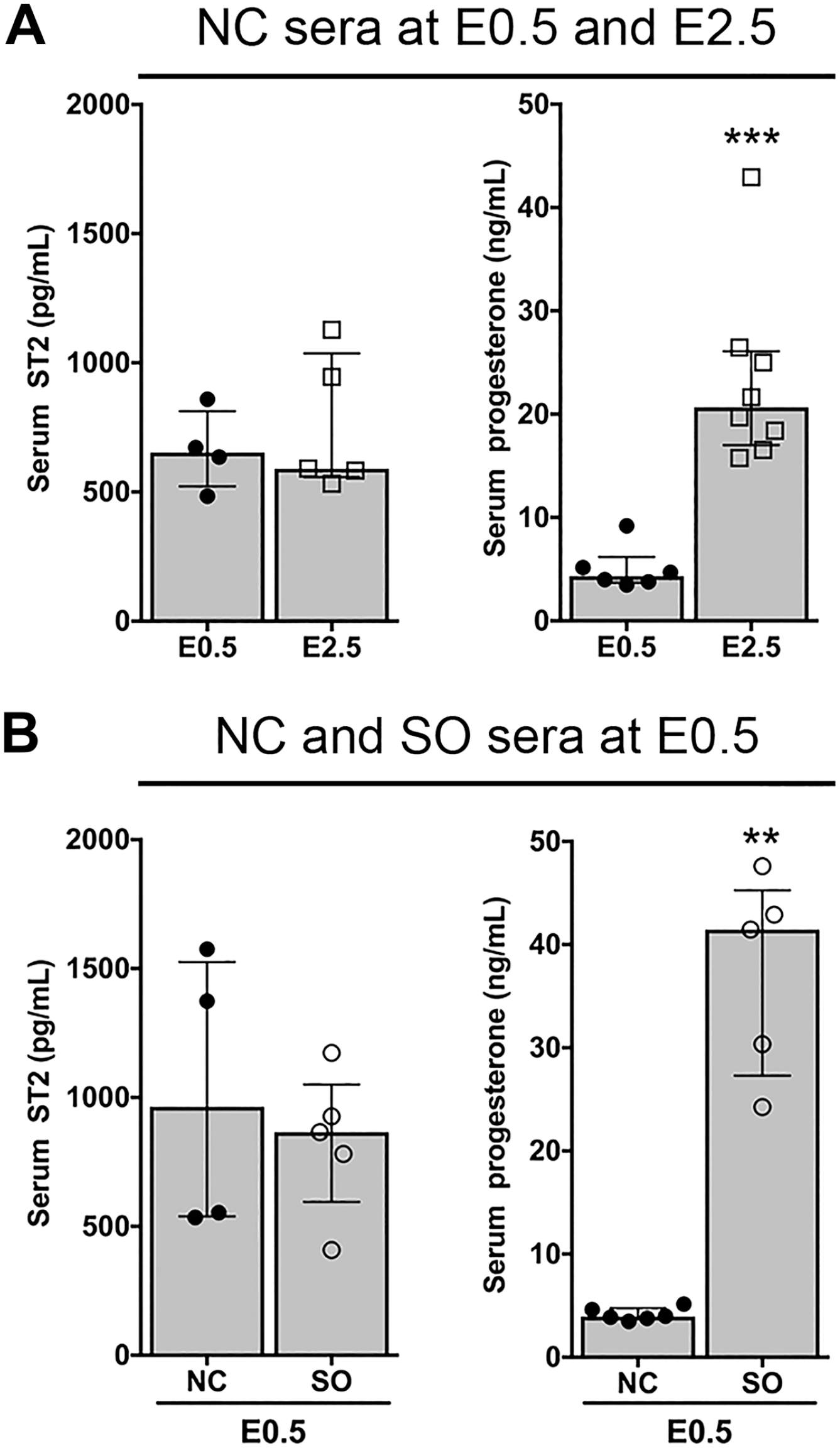
Superovulation does not alter circulating IL-33 and sST2 levels. Circulating levels of ST2 and progesterone were determined by ELISA and immunoassay, respectively. (A) Serum levels of ST2 were similar at E0.5 and E2.5. Serum progesterone levels were higher at E2.5 as compared with E0.5. (B) Serum levels of ST2 were similar in NCs and SO cycles at E0.5. Serum progesterone levels were higher in SO cycles as compared with NCs. Data are shown for each individual animal analyzed (n=4–7 animals) with median and interquartile range. Abbreviations: IL, interleukin; sST2, soluble form of ST2; ELISA, enzyme-linked immunosorbent assay; NC, natural cycle; SO, superovulation. **
Impact of Superovulation on IL-33/sST2.
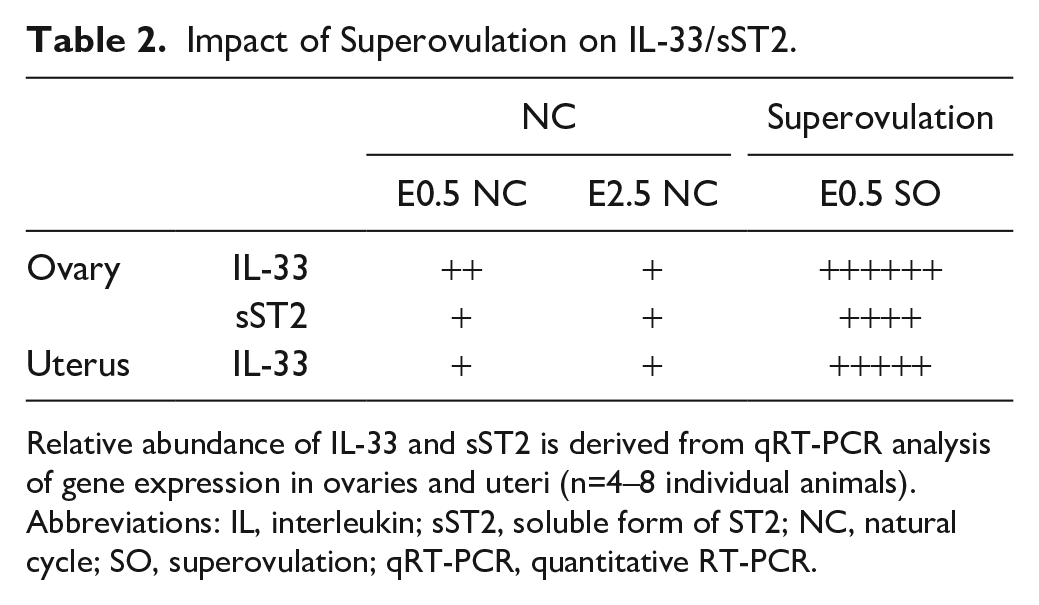
Relative abundance of IL-33 and sST2 is derived from qRT-PCR analysis of gene expression in ovaries and uteri (n=4–8 individual animals). Abbreviations: IL, interleukin; sST2, soluble form of ST2; NC, natural cycle; SO, superovulation; qRT-PCR, quantitative RT-PCR.
Discussion
IL-33 signaling is implicated in ovarian and uterine functions that support the initiation and maintenance of pregnancy. In this report, we identify IL-33- and ST2-expressing cells in the ovaries and uteri of sexually immature mice and of adult mice in distinct stages of the estrous cycle and in early pregnancy (Table 1). We find that oocytes express IL-33 and granulosa cells express ST2 in all stages of follicular development, throughout the estrous cycle, and even before sexual maturity. In adult estrus-stage females, uterine glandular and luminal epithelial cells express both IL-33 and ST2; this expression is maintained in early pregnancy, but IL-33 is reduced in diestrus stage and in sexually immature mice. Notably, our studies reveal that SO alters ovarian expression of IL-33 and sST2, the decoy receptor for IL-33 (Table 2). By identifying which cell types produce IL-33 and ST2 in the mouse ovary and uterus and assessing changes induced by SO, our findings contribute to and extend analyses that implicate IL-33/ST2 signaling in the establishment and maintenance of pregnancy.
Follicular development is a complex and highly organized process in which growth and maturation of the oocyte is tightly synchronized with the proliferation and differentiation of surrounding somatic cells. In primordial and primary follicles, folliculogenesis is largely gonadotropin-independent and driven by extensive bidirectional crosstalk between the oocyte and the granulosa cells that surround it. 28 Oocyte-granulosa crosstalk continues throughout the subsequent gonadotropin-dependent stages of follicle maturation, when the granulosa cells also engage in crosstalk with theca cells that surround the maturing follicle. The intercellular dialogue, oocyte-to-granulosa and granulosa-to-theca, is mediated by secreted growth factors (e.g., GDF9, BMP15) and cytokines (e.g., IL-7) that support oocyte growth and maturation, as well as drive somatic cell proliferation and differentiation.29–31 Our finding of IL-33 expression in oocytes and ST2 expression in granulosa cells raises the intriguing possibility that IL-33 signaling plays a role in cellular crosstalk underlying folliculogenesis. IL-33 has been shown to drive the proliferation and differentiation of immune cells and epithelial cells; whether oocyte-derived IL-33 has a similar effect on granulosa cells is an important question for future studies.
Recent studies suggest that IL-33 signaling in the uterus mediates endometrial receptivity and key events at the maternal–fetal interface, such as implantation and placenta formation, in both human and mouse pregnancies.5,7,8,20 In humans, expression of IL-33 in stromal and epithelial cells in secretory phase endometrium
20
and both IL-33 and ST2 in the placenta, localized to syncytiotrophoblasts by immunostaining,
5
identifies IL-33 and ST2 in key cell types that contribute to the maternal–fetal interface. As a framework for investigating the role of IL-33 signaling at the maternal–fetal interface, we determined the cellular sources of IL-33 and ST2 in the mouse uterus. In this study, we show that both IL-33 and ST2 are expressed in glandular and luminal epithelial cells. ST2 is constitutively expressed, with expression in pubescent and reproductive-age, cycling mice and in pre-implantation stages of early pregnancy. In contrast, IL-33 expression is sex hormone- and estrous cycle-dependent. We detected very little uterine expression of IL-33 in sexually immature mice. Whereas, in reproductive-age, cycling mice, IL-33 is abundantly expressed in estrus-stage uterine epithelium. Sex hormone- and estrous cycle-dependent expression has been reported for other IL-1 family members, IL-1α and IL-1β.32,33 Uteri from ovariectomized mice contained very low amounts of
Genes that are more highly expressed in the estrus stage and maintained in the pre-implantation uterus, such as IL-1 and IL-33, may be involved in providing a local uterine microenvironment that supports successful, timely embryo implantation. Peri-implantation administration of the IL-1 receptor antagonist prevented embryo implantation, 34 demonstrating a role for IL-1 signaling at the maternal–fetal interface in establishing pregnancy. Similarly, exposure of pre-implantation-stage mouse uteri to decidualized human endometrial stromal cells lacking IL-33 resulted in a 6-fold reduction in embryo implantation rates and increased hemorrhage at the maternal–fetal interface in the remaining implantation sites. 20 Furthermore, >50% of litters from female mice lacking ST2 had non-viable pups, 3 suggesting a requirement for IL-33 signaling in placenta function later in gestation. Thus, studies that investigate early pregnancy events in IL-33 null mice and mice with loss of IL-33 in uterine epithelia will further define the requirement for IL-1 family members in early pregnancy events.
Collectively, both human and animal studies suggest that SO leads to histological changes in the endometrium that affect endometrial receptivity and embryo implantation and/or have the adverse ripple effect of abnormal placenta formation and function.23,35,36 SO is associated with altered ovarian hormone levels that change gene expression profiles in the luminal and glandular epithelium. Each of these endometrial epithelial cell types has unique, non-redundant roles in promoting stromal cell decidualization, endometrial receptivity, and embryo implantation.37–40 Aberrant ovarian expression of
Taken together, our data suggest a role for IL-33 signaling in both physiological and pathological reproductive processes. We propose that proper spatiotemporal regulation of IL-33/ST2 function in the female reproductive tract supports normal reproductive function, and that dysregulated uterine IL-33 signaling (such as that seen in SO cycles) may result in poor pregnancy outcomes. This study forms the basis for developing cell type–specific loss-of-function and gain-of-function models that will modulate IL-33 and ST2 activity to understand the requirements for IL-33 signaling in reproductive processes.
Supplemental Material
2019-00169R1_Production_Supplemental_Figure_1_online_supp – Supplemental material for Dynamic Expression of Interleukin-33 and ST2 in the Mouse Reproductive Tract Is Influenced by Superovulation
Supplemental material, 2019-00169R1_Production_Supplemental_Figure_1_online_supp for Dynamic Expression of Interleukin-33 and ST2 in the Mouse Reproductive Tract Is Influenced by Superovulation by Salma Begum, Barry E. Perlman, Nuriban Valero-Pacheco, Valerie O’Besso, Tracy Wu, Sara S. Morelli, Aimee M. Beaulieu and Nataki C. Douglas in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We acknowledge the technical assistance provided by Dr. Hojoon Lee. We thank Marc Brillantes for technical assistance and insightful discussions.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: SB, BEP, SSM, AMB, and NCD designed the study and drafted the manuscript. SB, BEP, NVP, VO, and TW performed the experiments. SB, BEP, NVP, SSM, AMB, and NCD analyzed the data. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under Award Number R01 AI148695, awarded to A.M.B. and N.C.D. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
