Abstract
ZPK is a recently described protein serine/threonine kinase that has been originally identified from a human teratocarcinoma cell line by the polymerase chain reaction and whose function in signal transduction has not yet been elucidated. To investigate the potential role of this protein kinase in developmental processes, we have analyzed the spatial and temporal patterns of expression of the ZPK gene in mouse embryos of different gestational ages. Northern blot analysis revealed a single mRNA species of about 3.5 KB from Day 11 of gestation onwards. In situ hybridization studies demonstrated strong expression of ZPK mRNA in brain and in a variety of embryonic organs that rely on epithelio-mesenchymal interactions for their development, including skin, intestine, pancreas, and kidney. In these tissues, the ZPK mRNA was localized primarily in areas composed of specific types of differentiating cells, and this expression appeared to be upregulated at a time concomitant with the onset of terminal differentiation. Taken together, these observations raise the possibility that the ZPK gene product is involved in the establishment and/or maintenance of a fully cytodifferentiated state in a variety of cell lineages.
P
As a means of exploring the possible physiological functions of this unusual protein kinase, we recently isolated a full-length cDNA clone encoding the murine homologue of human ZPK and localized the sites of expression of the ZPK transcripts in a number of adult mouse tissues by in situ hybridization (Blouin et al. 1996). The results of these experiments have demonstrated that the murine ZPK gene exhibits a remarkable pattern of cell type-specific expression in most organs analyzed, particularly in the central nervous system, in the gastrointestinal tract, and in the testis. On the basis of these findings, we hypothesized that the ZPK gene product might be required for development, function, and maintenance of various organ systems. To explore this possibility, we have studied the expression and localization of ZPK transcripts in the developing mouse embryo by Northern blot analysis and in situ hybridization. Our data demonstrate striking spatial and temporal patterns of ZPK gene expression during the later stages of embryogenesis and provide evidence that this novel protein kinase may be involved in developmental processes related to cell differentiation.
Materials and Methods
Embryos
Mouse embryos were collected on Days 11.5, 13.5, 15.5, or 17.5 of gestation [embryonic days (E) 11.5, 13.5, 15.5, and 17.5] and were obtained from F1 crosses between CD-1 mice. Noon on the day of finding a vaginal plug was designated as Day 0.5. This procedure was approved and controlled by the Institutional Ethical Committee for Experimental Animals.
Probes
A 192-BP fragment of the mouse ZPK cDNA (nucleotides 837–1028) (Blouin et al. 1996) was cloned into pBluescript KS+ plasmid (Stratagene; La Jolla, CA). For Northern hybridization, this cDNA fragment was labeled to high specific activity with [α-32P]-dCTP (Amersham; Oakville, Ontario, Canada) by the random priming method of Feinberg and Vogelstein (1983). For in situ hybridization, sense and antisense riboprobes were generated from linearized pBluescript plasmid containing the 192-BP fragment using T3 (antisense) or T7 (sense) RNA polymerase and digoxigenin-11-UTP (Boehringer Mannheim; Laval, Quebec, Canada).
Northern Hybridization
A multiple-tissue Northern blot (Clontech Laboratories; Palo Alto, CA) containing poly(A+) RNA (≈2 μg/lane) purified from E7, E11, E15, and E17 mouse embryos was hybridized with a 32P-labeled mouse ZPK cDNA fragment and rehybridized with a human β-actin cDNA probe. Hybridization was carried out overnight at 42C in 5 X SSPE (1 X SSPE is 150 mM NaCl, 10 mM NaH2PO4, 1 mM EDTA, pH 7.0), 10 X Denhardt's solution (1 X Denhardt's is 0.02% Ficoll, 0.02% polyvinyl pyrolidone, 0.02% bovine serum albumin), 100 μg/ml salmon sperm DNA, 2% SDS, and 50% formamide. After hybridization the membrane was washed for 40 min in 2 X SSC (1 X SSC is 150 mM NaCl, 15 mM sodium citrate, pH 7.0), 0.05% SDS at room temperature (RT) and twice for 20 min in 0.1 X SSC, 0.1% SDS at 50C. The membrane was then exposed to Kodak BioMax film at −70C with an intensifying screen and the resulting autoradiograph was scanned with a laser densitometer (Bio-Rad Imaging Densitometer model GS-670; Bio-Rad Laboratories, Mississauga, Ontario, Canada).
In Situ RNA Hybridization
In situ hybridizations were performed essentially as described previously (Blouin et al. 1996). Briefly, embryos were fixed overnight in 4% paraformaldehyde/0.1 M sodium cacodylate, dehydrated with graded ethanol solutions, and embedded in paraffin. Paraffin sections were cut serially at 4-μm thickness, mounted on polyionic slides (Superfrost Plus; Fisher, Montreal, Quebec, Canada), and kept at 4C. Sections were then deparaffinized with xylene, permeabilized with 0.2 M HCl (10 min) and 0.3% Triton X-100 (15 min), treated with 20% acetic acid (5 min) to reduce nonspecific background, and digested with proteinase K (2 μg/ml, 30 min). After digestion, sections were washed in 0.2% glycine, fixed with 4% paraformaldehyde, and acetylated with triethanolamine. Hybridization was carried out overnight at 42C in 5 X SSC, 5 X Denhardt's solution, 50% formamide, 250 μg/ml yeast tRNA, and 4 mM EDTA with either antisense or sense control RNA probes at a concentration of 10 ng/μl. After hybridization, the slides were washed twice in 2 X SSC for 15 min at RT, incubated once for 10 min with RNAse A (1 μg/ml) at RT, rinsed twice in 2 X SSC at RT for 15 min, and finally washed twice in 1 X SSC at 37C for 15 min. Immunological detection of the hybridized probes was performed with alkaline phosphatase-conjugated anti-digoxigenin antibody and nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolyl-phosphate color substrates (Boehringer Mannheim). Finally, sections were counterstained with Mayer's hematoxylin solution (Sigma; Mississauga, Ontario, Canada) (Figures 3-8) and mounted with Crystal/Mount (Biomeda; Foster City, CA). Photographs were taken under brightfield illumination using a Zeiss photomicroscope and Kodak TMAX 100 print film.
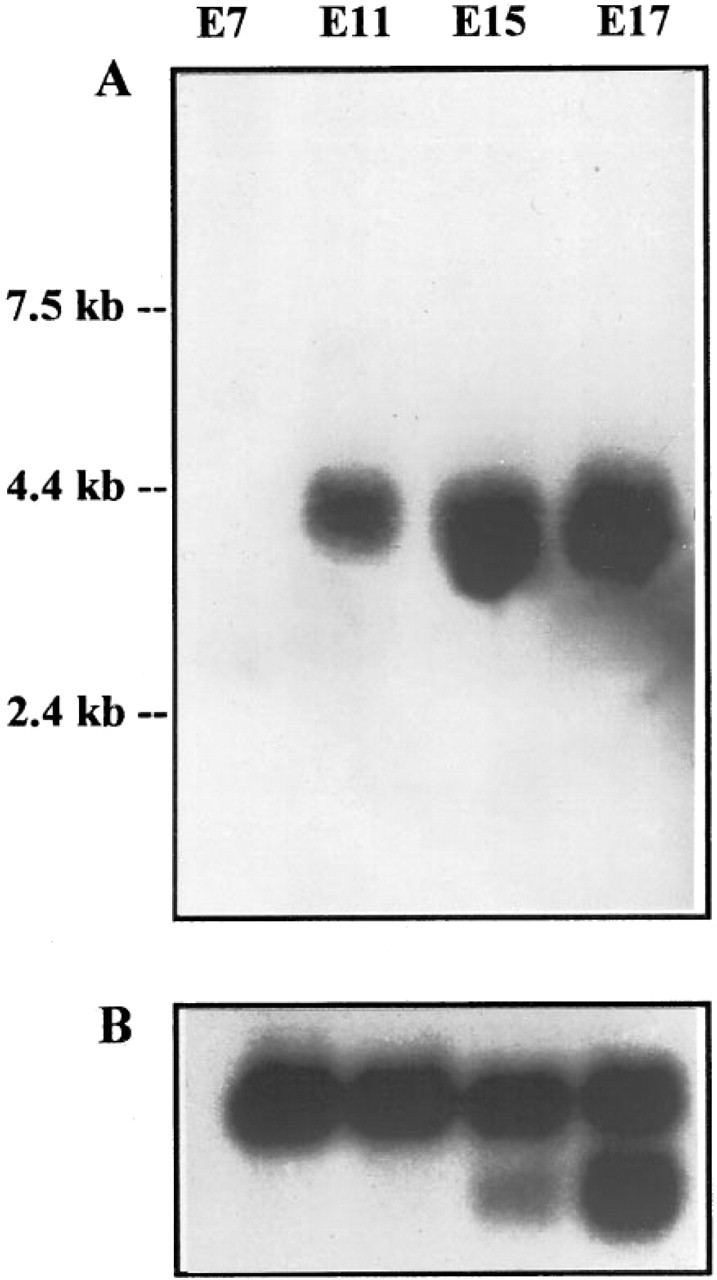
Northern blot analysis of ZPK gene expression in developing mouse embryos. A multiple-tissue Northern blot containing poly(A+) RNA (≈2 μg/lane) purified from E7, E11, E15, and E17 mouse embryos was hybridized to a 32P-labeled mouse ZPK cDNA probe (A). As a control, the same blot was rehybridized with a human β-actin cDNA probe (B).
Results
Expression of the ZPK Gene in Developing Mouse Embryos
Poly(A+) RNA isolated from mouse embryos of different gestational ages (E7-E17) was analyzed by Northern hybridization using as a probe a fragment of the mouse ZPK cDNA. As shown in Figure 1, a single mRNA transcript of about 3.5 KB was detected in mouse embryos from around E11 onwards. The abundance of this mRNA remained relatively constant during late fetal stages.
Tissue Distribution of ZPK mRNA in E17.5 Mouse Embryos
To determine the identity of the tissues in which the ZPK gene is expressed during mouse embryogenesis, sagittal sections of E17.5 mouse embryos were first examined by in situ hybridization with an antisense digoxigenin-labeled ZPK RNA probe. At this stage of development, widespread distribution of ZPK mRNA transcripts was detected in several tissues (Figure 2A). The highest levels of ZPK gene expression were found in neuronal structures, including brain and spinal ganglia, as well as in the developing epithelia of the skin, intestine, pancreas, and kidney (the latter is not shown on the micrograph). In skin, the ZPK messages exhibited a graded pattern of distribution, with the highest levels observed posteriorly and caudally. No expression was detected in the epithelium of the lung and liver. As a control of the specificity of the hybridizations, a ZPK sense probe was also hybridized on adjacent tissue sections. This probe always revealed absence of labeling (Figure 2B).
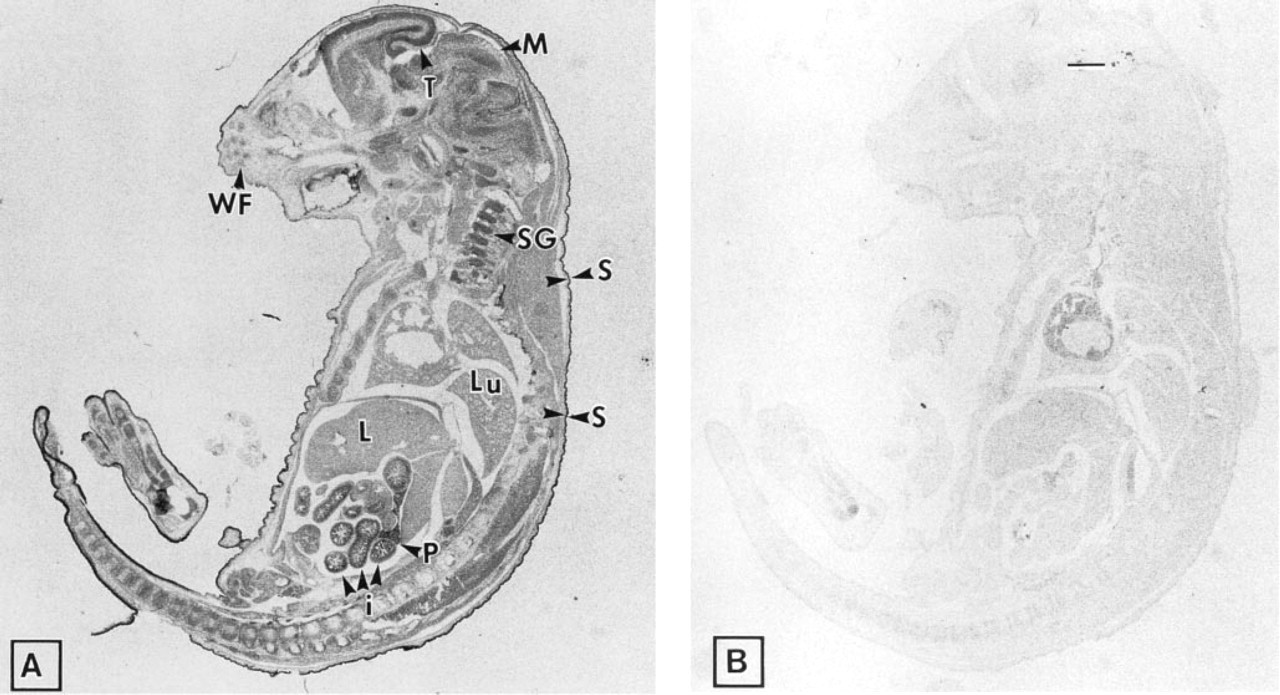
Expression of ZPK mRNA in an E17.5 mouse embryo. Adjacent parasagittal sections of an E17.5 mouse embryo were hybridized with either digoxigenin-labeled anti-sense
Localization of ZPK mRNA in Embryonic Tissues of Different Developmental Stages
To more precisely define the spatial and temporal localization of ZPK mRNA in the tissues that demonstrated highest expression, as seen in Figure 2, sections of mouse embryos from different stages of gestation (E11.5, E13.5, E15.5, and E17.5) were hybridized in situ with anti-sense and sense ZPK riboprobes.
Brain. Expression of ZPK mRNA was detected in several regions of the developing brain at all embryonic stages examined. However, the most regionalized signal for ZPK expression was found in the neuroepithelial layer of the telencephalic vesicle. At E11.5, ZPK mRNA expression appeared to be confined only to a small group of neuronal cells distributed throughout the telencephalon (Figures 3A and 3B). Subsequently (E17.5), the number of ZPK-expressing cells increased remarkably and became essentially localized in the marginal zone of the developing telencephalon which contains postmitotic differentiated neurons (Figures 3D and 3E). In the paravertebral ganglions of E15.5 and E17.5 embryos, a high level of ZPK mRNA expression was found in the ganglionic neurons (Figures 3G and 3H). In contrast, the peripheral nerves entering the spinal ganglia remained unlabeled. Negative control with the sense probe resulted in no signal in any of these regions (Figures 3C, 3F, and 3I).
Skin. No expression of ZPK mRNA was detected in the two- to three-layer thick, poorly differentiated epidermis of E13.5 embryos (Figure 4A). However, at E15.5 (Figure 4B) and E17.5 (Figures 4C and 4D), when epidermal differentiation occurs, expression of ZPK mRNA was detected in the stratum granulosum of the developing skin and in hair follicles. In contrast, no expression was observed in the stratum germinativum, which contains mitotically active cells, or in the mesenchymal cells of the dermis (Figures 4B-4D). Hybridization of adjacent tissue sections with the sense probe revealed no signal at all (Figure 4E).
Whisker Follicles. In the snout region of E15.5 (Figure 5A) and E17.5 (Figures 5B and 5C) mouse embryos, a strikingly heterogeneous pattern of ZPK mRNA distribution was observed in the whisker follicles. All the follicles examined showed a restricted pattern of ZPK mRNA expression in one part of the inner root sheath (Figures 5A-5C). No expression was observed in the outer root sheath cells or in follicle cortical cells. Control hybridization with the ZPK sense probe gave no signal (Figure 5D).
Intestine. At E13.5, no mRNA signal for ZPK was observed in the poorly differentiated epithelium of the gastrointestinal tract and in the surrounding mesenchymal tissue (Figure 6A). However, at E15.5, when the finger-like intestinal villi began to form, a heterogeneous distribution of ZPK mRNA was detected in the epithelium of the developing gut but not in the mesenchymal cells (Figure 6B). This specific pattern of expression became more conspicuous at E17.5 (Figure 6C), when the single columnar epithelium of the villi had already formed. Control hybridization with the ZPK sense probe gave no signal (Figure 6D).
Pancreas. In the developing pancreas, ZPK mRNA expression was first detected at around E15.5 in some acini (Figure 7A). During the following stages of development, this expression increased remarkably, and almost all the pancreatic acinar cells of E17.5 embryos exhibited a strong hybridization signal (Figure 7B). Control hybridization with the ZPK sense probe gave no signal (Figure 7C).
Kidney. At early developmental stages (E13.5), ZPK mRNA could not be detected in the metanephros (Figure 8A). In contrast, at E15.5 (Figure 8B) and E17.5 (Figure 8C), high levels of ZPK transcripts were present in the columnar epithelium that lined the convoluted tubules of the kidney. Endothelial cells and mesenchymal cells of the developing kidney showed no detectable levels of ZPK expression throughout all the stages examined (Figures 8A-8C). In the adrenal gland of E17.5 embryos (Figure 8C), ZPK mRNA expression was detected at high levels in the cortex, whereas cells of the medulla appeared mostly negative for this transcript (Figure 8C). Negative control with the ZPK sense probe gave no signal (Figure 8D).
Discussion
The ZPK gene encodes a non-receptor serine/threonine protein kinase originally identified in human after a search for protein kinases potentially involved in neuronal development (Reddy and Pleasure 1994). To date, no biological role for ZPK has been established. However, recent expression studies in adult mouse tissues have suggested a possible involvement in the function and maintenance of a variety of specialized cells (Blouin et al. 1996). To gain an initial insight into the role of ZPK during embryogenesis, we have investigated the expression patterns of the ZPK gene in mouse embryos by both Northern blot analysis and in situ hybridization.
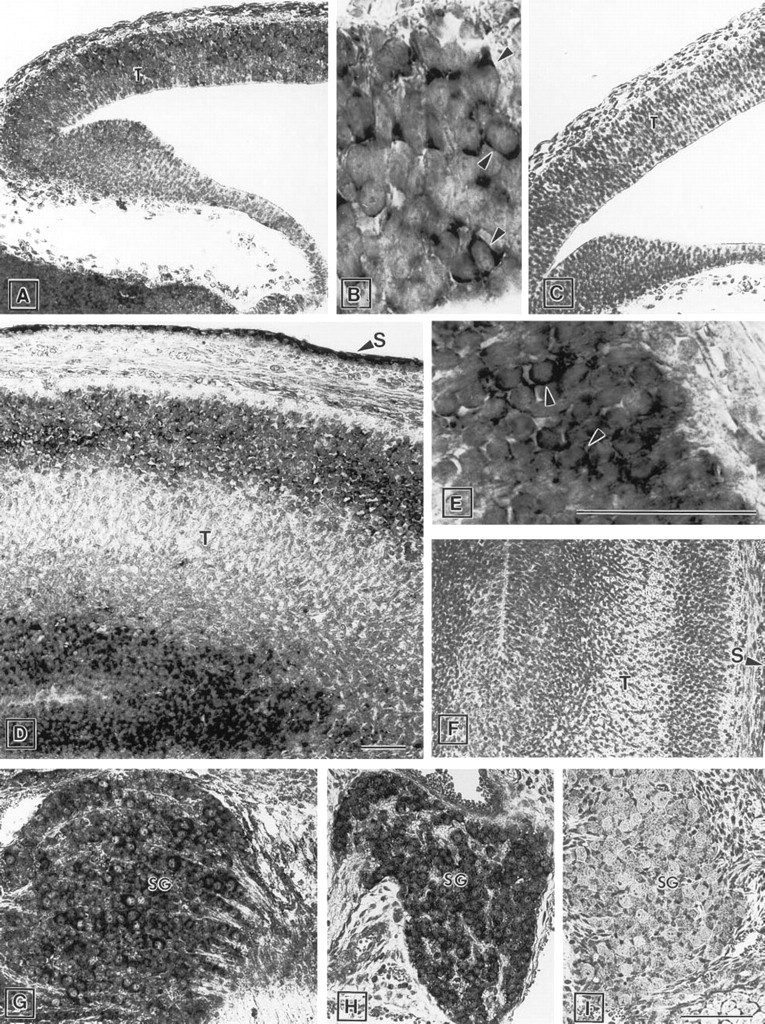
Expression of ZPK mRNA in the developing brain and spinal ganglion. Parasagittal sections of mouse embryos were hybridized with either anti-sense
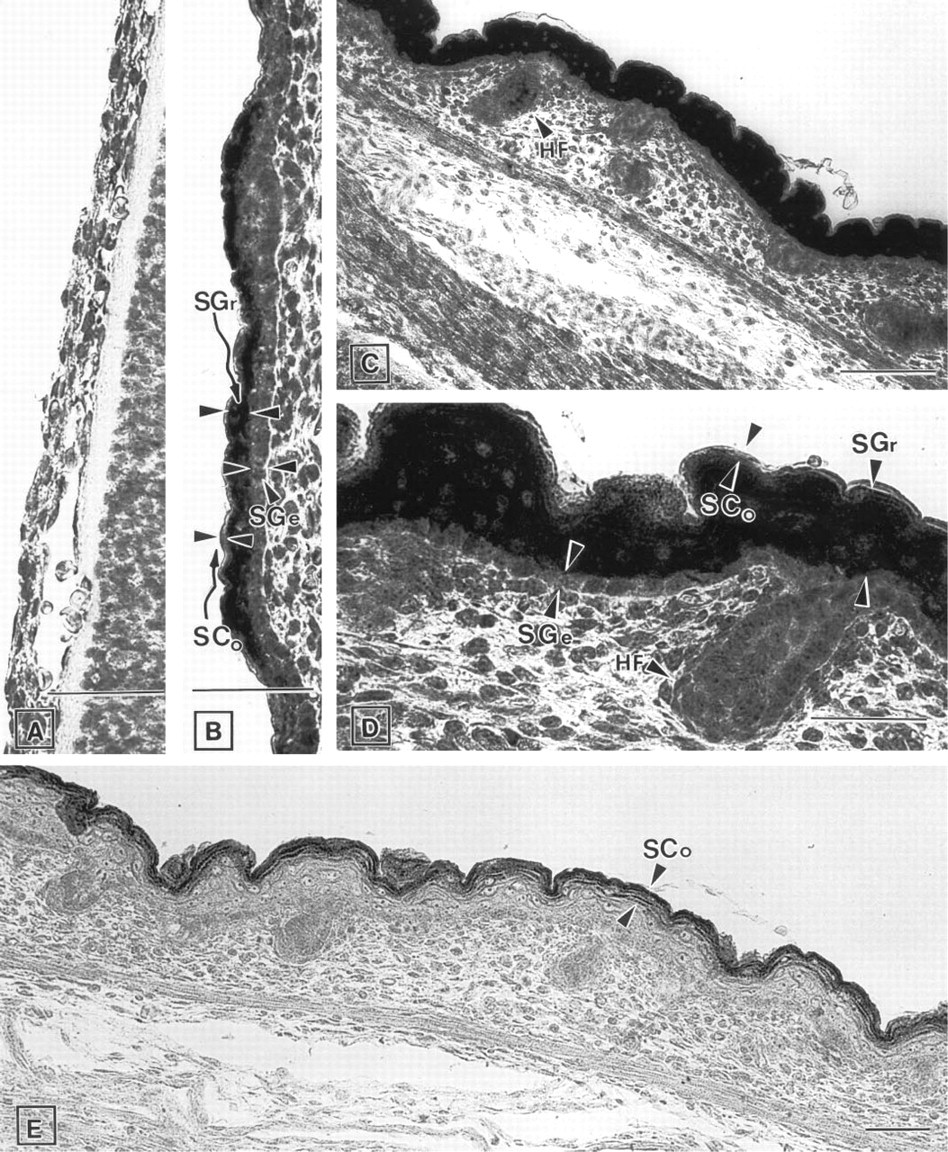
Expression of ZPK mRNA in the developing skin. Parasagittal section of mouse embryos were hybridized with either anti-sense
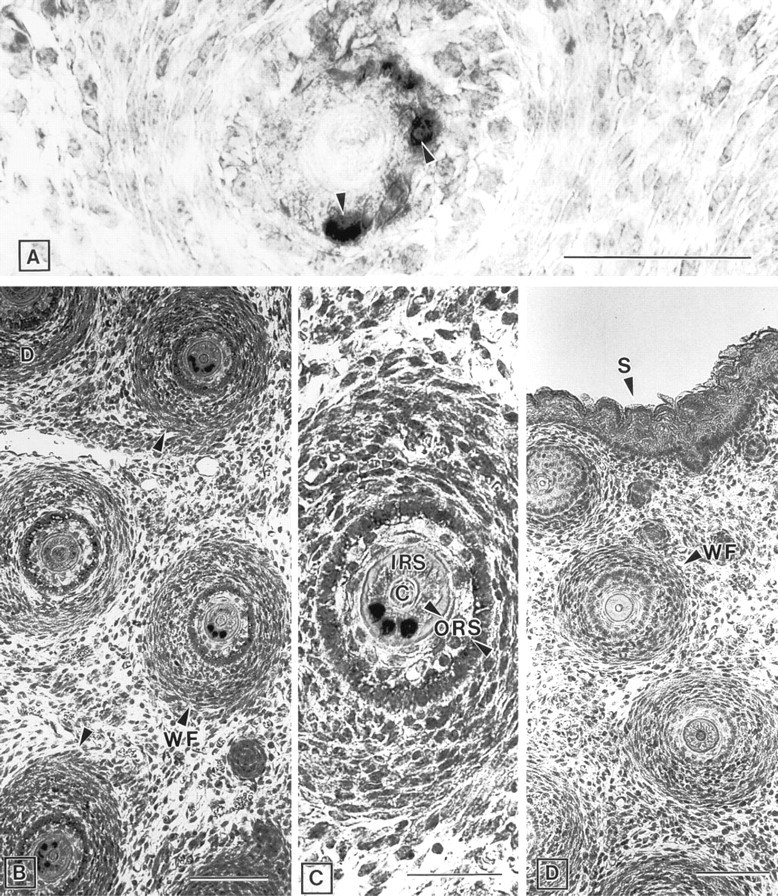
Expression of ZPK mRNA in the developing whisker follicles. Parasagittal section of mouse embryos were hybridized with either anti-sense
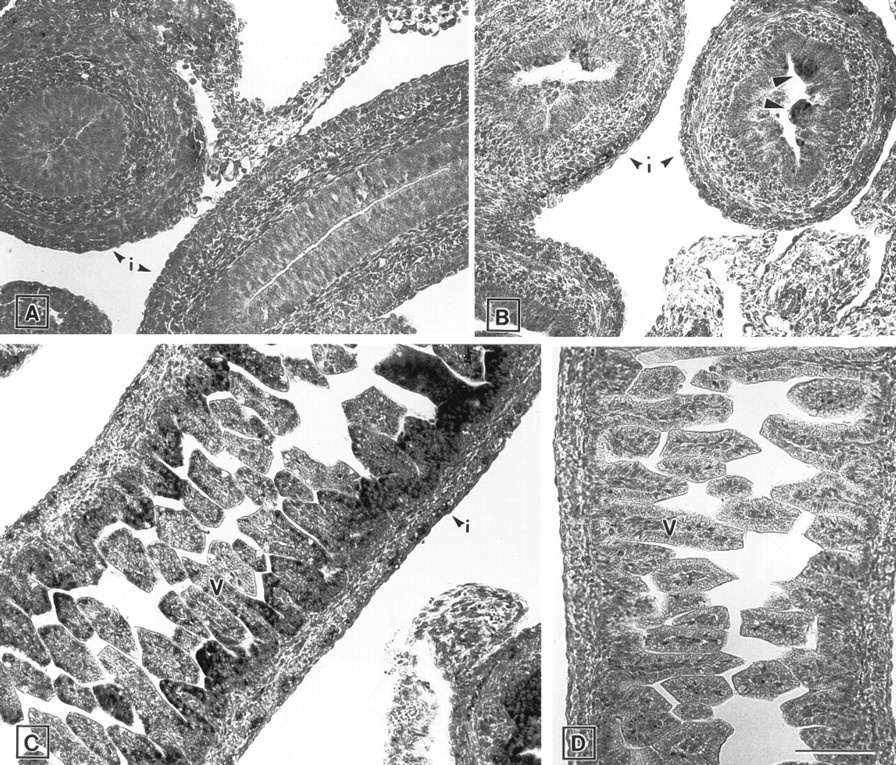
Expression of ZPK mRNA in the developing intestine of E13.5
Northern blot analysis using a ZPK cDNA probe revealed a single mRNA species from around Day 11 of gestation onwards. This result indicates that expression of ZPK transcripts is regulated in a temporal manner during mouse development, appearing when the embryo is undergoing organogenesis. The size of the detected mRNA was determined to be approximately 3.5 KB, which is in agreement with that observed in adult mouse tissues (Blouin et al. 1996).
In situ hybridization studies demonstrated that the ZPK gene exhibits remarkable patterns of cell type-and developmental stage-specific expression in a variety of organ systems in the mouse embryo. The highest levels of ZPK transcripts were found in neuronal tissues, including brain and spinal ganglia, as well as in the developing epithelia of organs that rely on inductive interactions from the surrounding mesenchyme for their development. These include skin, whisker follicles, intestine, pancreas, and kidney. By contrast, no expression of ZPK mRNA was detected in lung and liver, two other tissues known to develop by epithelio-mesenchymal interactions (Gilbert 1991). The absence of expression in these two tissues is apparently limited to the embryonic stages because ZPK mRNA was detected at very low levels in adult lung and liver by both Northern blot analysis (Reddy and Pleasure 1994) and in situ hybridization (Blouin et al. 1996). These data suggest a role for ZPK in the specialized functions of these two organs rather than in their development.
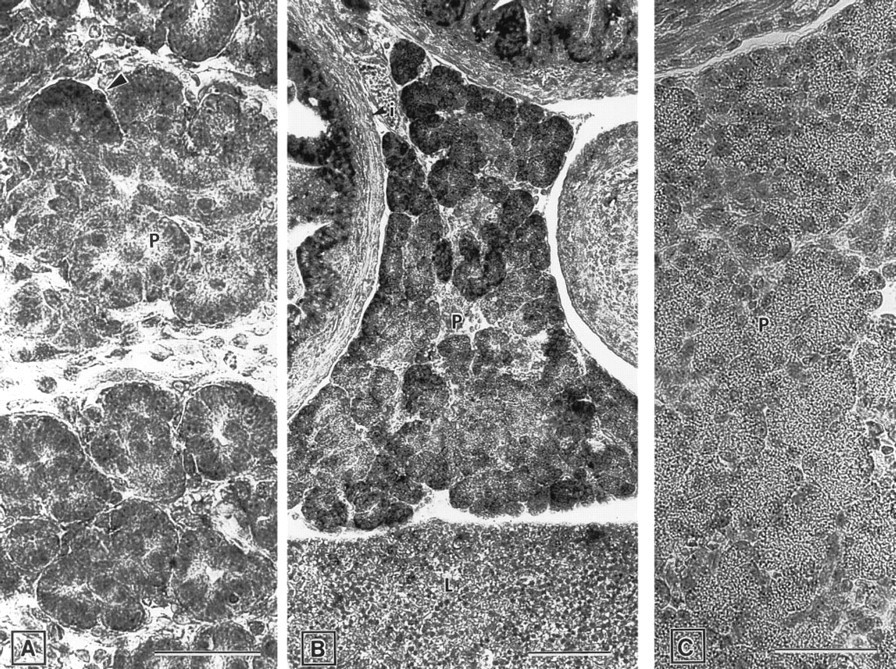
Expression of ZPK mRNA in the developing pancreas of E15.5
The pattern of expression observed in the developing central nervous system suggests a role for ZPK in neuronal differentiation. At the earliest stage examined (E11.5), ZPK mRNA was primarily detected in the developing telencephalon and was confined to a limited number of cells randomly distributed throughout the neuroepithelium. Subsequently, its expression became more regionalized within the telencephalon and almost all the ZPK-expressing cells were found in the marginal zone, which contains postmitotic neurons (Uylings et al. 1990). The preferential localization of ZPK transcripts to this region is in agreement with our previous report showing prominent expression of the ZPK gene in areas of the adult mouse brain composed of terminally differentiated neurons, such as the cerebral cortex, the hippocampus, and the Purkinje cell layer of the cerebellum (Blouin et al. 1996).
Expression of ZPK mRNA in tissues whose development requires inductive interactions was detected in specific cell types during their terminal differentiation (E15.5 onwards). For example, in the developing skin, expression of ZPK mRNA was clearly localized in the differentiating suprabasal cell layers of the epidermis, thus suggesting a role for ZPK in epidermal cell stratification and differentiation. Consistent with this, a similar expression pattern was also seen in the inner root sheath of the whisker follicles, which possess histological and histochemical characteristics comparable to those of keratinizing epidermal cells (Holbrooke et al. 1988). Furthermore, in the gut, expression of the ZPK gene was detected in the intestinal epithelia at a time concomitant with the morphological reorganization and differentiation of the intestine (Trier and Moxey 1979; Mathan et al. 1976). Finally, in the pancreas and kidney, the ZPK mRNA signal was detected in the acinar and tubular epithelial cells, respectively, during cytodifferentiation of these two cell populations (Saxén 1987; Han et al. 1986; Teitelman et al. 1981; Sanders and Rutter 1974). The preferential expression of ZPK mRNA within these differentiating cells supports the hypothesis that ZPK may somehow be involved in developmental processes related to cell differentiation. Such a function would be compatible with the finding of Reddy and Pleasure (1994), who originally identified ZPK in a human teratocarcinoma cell line induced to differentiate into postmitotic neurons by treatment with retinoic acid.
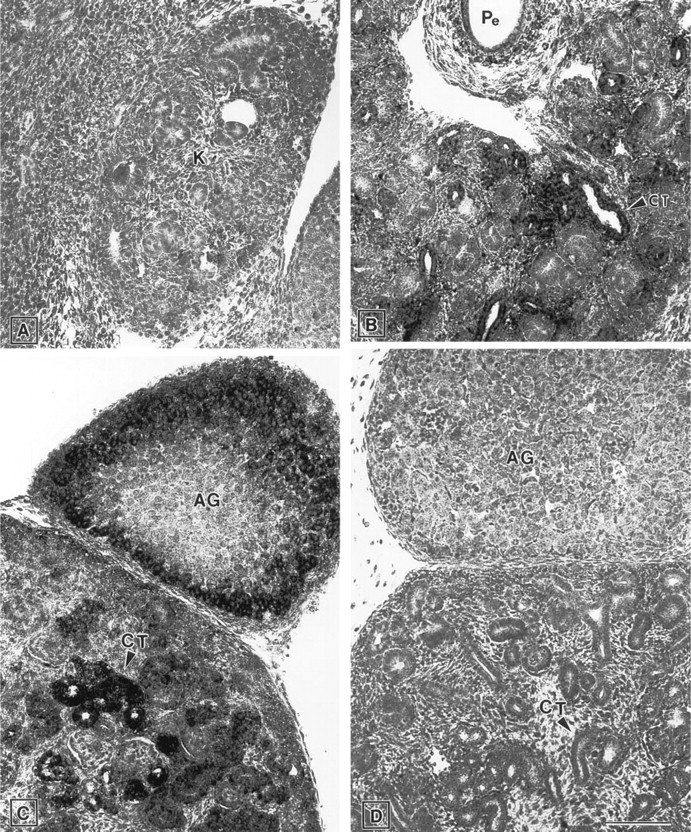
Expression of ZPK mRNA in the developing kidney of E13.5
An intriguing feature of ZPK gene expression in the developing epidermis is the variation of its spatial expression. In the skin of E17.5 mouse embryos, ZPK transcripts were found at high levels throughout the dorsal and caudal regions, whereas little or no hybridization signal was detected in the ventral and rostral regions. The significance of this dorsal/ventral and caudal/rostral sequence of regionalized expression is unknown, but may relate to the fact that the stratum granulosum, the cell layer in which ZPK is expressed, is discontinuous during development of the mouse embryonic skin (Sengel 1976). Another possibility would be that the graded distribution of ZPK mRNA reflects regional variation in the expression level of the ZPK gene. By analogy to the observations made in the fetal mouse skin, we also detected a heterogeneous distribution of ZPK transcripts in whisker follicles of mouse embryo. In this case, ZPK mRNA expression appeared to be restricted to a few cells of the inner root sheath. Because the inner root sheath is populated by three layers of keratinocytes that differentiate successively (Sengel 1976), our results raise the possibility that ZPK expression might be confined to one of these layers.
Interactions that occur between epithelial and mesenchymal cells during development of most organ systems in which ZPK is expressed in the mouse embryo are mediated by a variety of signaling molecules that act in concert to regulate various aspects of tissue development and maintenance (Hirai et al. 1992; Hay 1990; Mercola and Stiles 1988; Grobstein 1967). To date, little is known about the nature and identity of these molecules, although recent reports have provided evidence that growth factors expressed in the vertebrate embryo may mediate growth, motility, differentiation, and morphogenesis (Birchmeier et al. 1995; Jessel and Melton 1992). Most of the growth factors characterized to date share the common feature of binding to and activating cell surface receptors that possess intrinsic tyrosine-specific protein kinase activity (van der Geer and Hunter 1994). The binding of growth factors to receptor tyrosine kinases initiates a cascade of phosphorylation events that leads to the subsequent activation of several serine/threonine protein kinases whose function is to transmit signals from the receptors to the nucleus (Bishop 1991; Cantley et al. 1991; Hunter 1991). Because the ZPK gene encodes a non-receptor type of serine/threonine protein kinase, it is tempting to speculate that this novel protein kinase could potentially transduce signals in response to the activation of specific receptor tyrosine kinases. Examples of receptor tyrosine kinases with exclusive or prevalent expression on epithelial cells include the c-met receptor, which mediates mitogenic, motogenenic, and morphogenic signals in a broad spectrum of epithelial and endothelial cells in culture (Weidner et al 1993; Gherardi 1991), the epidermal growth factor receptor, which appears to play a key role in epithelial development of several organs (Miettinen et al. 1995; Threadgill et al. 1995), and the c-ret receptor, which is required in organogenesis of the kidney and normal development of the enteric nervous system (Schuchardt et al. 1994). It will be very interesting to determine in future work whether the intrinsic kinase activity of ZPK can be regulated by members of the growth factor receptor family in response to ligand binding.
In summary, our results show that the murine ZPK gene exhibits spatial and temporal patterns of mRNA expression in the central nervous system as well as in the epithelia of the skin, intestine, pancreas, and kidney during mouse development. In all these tissues, the expression of ZPK mRNA increases with development and correlates with areas undergoing terminal cell differentiation. On the basis of these observations, it appears likely that ZPK may fulfill specific signaling functions required in either the induction or the maintenance of the differentiated state for a wide variety of cell populations. Alternatively, it is also conceivable that ZPK could contribute to the differentiation process by exerting a negative effect on cell growth. Studies addressing this question are now in progress.
Footnotes
Acknowledgements
We thank Dr A. Beaudoin and Mr W. Home for critical comments on the manuscript, and N. Daigle and P. Bergeron for support during the course of this work. We are also grateful to Dr E. Calvo for help with the densitometer.
Supported by grants from the Natural Science and Engineering Research Council of Canada (RB) and by the “Fonds pour la Formation de Chercheurs et l'Aide à la Recherche” of the Province of Quebec.
