Abstract
Neuropsin is a novel serine protease whose mRNA is expressed in the mouse central nervous system. We examined the expression of neuropsin mRNA during embryonic development using Northern and in situ hybridization in non-neural tissues. The pregnant uterus showed strong expression of neuropsin mRNA, whereas the nonpregnant uterus did not express this mRNA. Expression was first detected in the primary decidual zone at 5.5 days post coitum and was maximized at 10 days post coitum, decreasing remarkably thereafter. During mouse organogenesis, neuropsin expression was observed in the developing heart, lung, thymus, pituitary, choroid plexus, and epithelial linings of the skin, oral cavity, tongue, esophagus, and forestomach. In adult mouse organs, neuropsin mRNA was expressed in epithelial tissues covered by keratinocytes with moderate density, whereas low expression was observed in lung, thymus, and spleen. Neuropsin mRNA expression in developing organs and adult keratinocytes suggests that neuropsin is associated with extracellular matrix modifications and cell migrations.
Keywords
N
During implantation and fetal development in pregnant mice, many proteases are also expressed in tro-phoblasts and decidual cells (Strickland et al. 1976; Strickland and Richards 1992; Schatz et al. 1994; Hurskainen et al. 1996). These proteases are metallo-proteases and serine proteases, which are believed to work in a proteolytic cascade to degrade the extracellular matrix of the maternal endometrium, resulting in trophoblast invasion of the uterine wall. The enzymes in this cascade are mainly synthesized in invading tro-phoblasts, and maternal decidual cells are believed to function as “protectors” by secreting protease inhibitors such as TIMP-3, PAI-1, and α2-macroglobulin (Strickland and Richards 1992; Cross et al. 1994; Harvey et al. 1995). However, decidual cells express a proteolytic activity that can degrade laminin and entactin, both of which are components of the basement membrane (Wewer et al. 1986). Although proteases expressed in trophoblasts have been extensively studied, relatively few studies have been performed on the proteases expressed in decidual cells (Schatz et al. 1994; Lefebvre et al. 1995; Alexander et al. 1996).
The results of the present study clearly demonstrated that large populations of cells express neuropsin in the embryonic stages and in the pregnant uterus. The abundant expression of neuropsin mRNA in the various organs of embryos and pregnant mice suggests that neuropsin is a multifunctional protein participating in implantation and embryogenesis as well as neural plasticity.
Materials and Methods
All of the general reagents were purchased from Wako Chemicals (Osaka, Japan) unless otherwise specified.
Preparation of Tissues
The procedure was approved by the Institutional Ethical Committee for Experimental Animals. ddY mice (SLC; Hamamatsu, Japan) were housed under a 12-hr light/dark cycle with food and water available ad libitum. To obtain timed pregnancies, 8-week-old female mice were caged with adult males until the appearance of a vaginal plug. For Northern blot analysis, tissues were dissected from 8-week-old males and females and 16.5-day post coitum (d.p.c.) embryos under deep ether anesthesia, immediately frozen in liquid nitrogen, and then processed for total RNA extraction. For in situ hybridization, embryos or uteri containing embryos were dissected from pregnant females under deep ether anesthesia on the indicated d.p.c. The dissected embryos or uteri were immediately frozen in dry ice and then embedded in Tissue-Tek OCT compound (Miles; Elkhart, IN). Once frozen, the tissues were stored at -80C.
Northern Blot Analysis
Total RNA was isolated from frozen tissues by a single-step extraction method (Chomczynski and Sacchi 1987). Samples of 25 μg of total RNA were electrophoresed on 1% agar-ose-formaldehyde gels, and vacuum transferred to nylon membranes (Gene Screen plus, NEN, Boston, MA). The membranes were hybridized for 18 hr at 42C with 32P-labeled cDNA probe in 50% formamide, 5 X SSPE (1 X SSPE is 150 mM NaCl, 10 mM NaH2PO4, 1 mM EDTA, pH 7.0), 5 X Denhardt's solution (1 X Denhardt's is 0.02% Ficoll, 0.02% polyvinyl pyrolidone, 0.02% bovine serum albumin), 10% dextran sulfate sodium salt, 1% SDS, and 0.1 mg/ml denatured salmon sperm DNA. The 32P-labeled probe was prepared by random priming using a PCR-amplified DNA fragment corresponding to nucleotides 487–690 of neuropsin cDNA (Chen et al. 1995) as a template. The membranes were then washed at high stringency (0.3 M NaCl, 20 mM NaH2PO4, 2 mM EDTA, 2% SDS at 68C) and exposed to Kodak XAR films. The blots were stripped and rehybridized with a mouse glyceraldehyde 3-phosphate dehydrogenase (GAPDH) probe to confirm integrity, equal loading, and blotting of RNA.
In Situ Hybridization
In situ hybridization was performed as described previously (Chen et al. 1995). Briefly, 20-μm frozen sagittal sections of mouse embryos and cross-sections of uteri on indicated d.p.c. were cut on a cryostat, thaw-mounted on glass slides, and stored at -80C until use. The sections were fixed with 4% formaldehyde in 0.1 M phosphate buffer (pH 7.4), treated with 10 mg/ml protease K, and acetylated with acetic anhydride in 0.1 M triethanolamine. After dehydration through an ascending alcohol series, the sections were hybridized with 35S-labeled cRNA probes in 50% formamide, 0.3 M NaCl, 20 mM Tris-HCl, pH 8.0, 5 mM EDTA, 10% dextran sulfate sodium salt, 1 X Denhardt's solution, 0.2% sarcosyl, 250 μg/ml tRNA, 400 μg/ml salmon testis DNA, and 20 mM dithiothreitol at 55C overnight. The sections were washed at 65C in 50% formamide, 2 X SSC (1 X SSC is 0.15 M NaCl, 0.015 M sodium citrate), 5 mM dithiothreitol. Subsequently, the sections were treated with RNase A (1 μg/ml) then washed again at high stringency. After dehydration through an ascending alcohol series, the sections were immersed in Kodak NTB2 emulsion and exposed for 4 weeks. After development in D-19 developer (Kodak; Rochester, NY), the sections were counterstained with hematoxy-lin/eosin and examined under a light microscope.
Results
Northern Blot Analysis of Neuropsin mRNA Expression
Northern blot analysis demonstrated that neuropsin mRNA was expressed in a variety of tissues in adult mice. As shown in Figure 1A, neuropsin mRNA was detected in the hippocampus, stomach, skin, thymus, lung, spleen, and pregnant uterus of adult mice. A single band of approximately 1.3
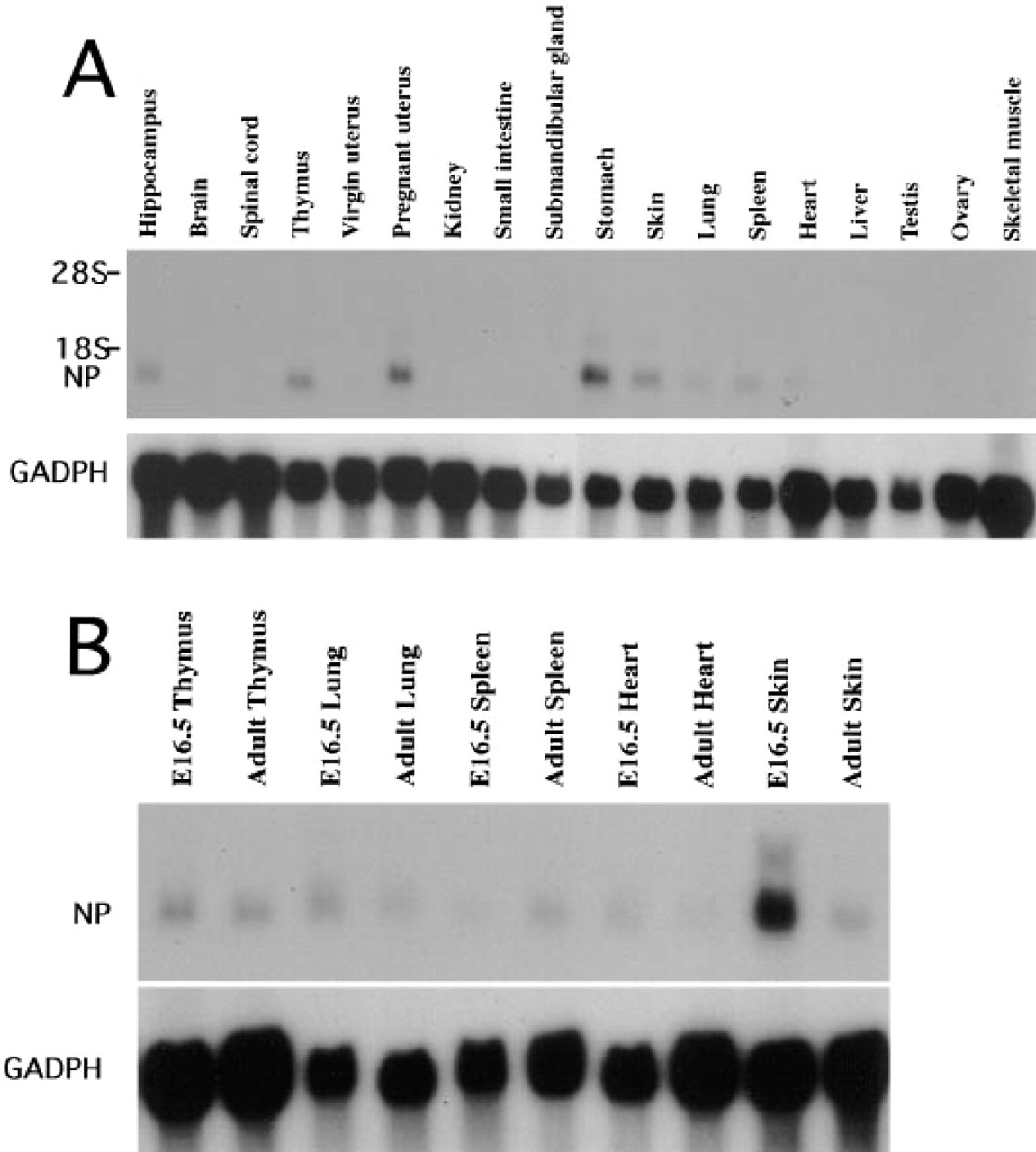
Northern blot analysis of neuropsin mRNA expression. (
In Situ Hybridization Analysis of Neuropsin mRNA
We next analyzed topographical expression of neuropsin mRNA in various organs by in situ hybridization. In agreement with our previous report, cells in certain regions of the central nervous system expressed neuropsin mRNA during development (Suzuki et al. 1995). Moreover, neuropsin-expressing cells were localized in various tissues, including the skin and the pregnant uterus, in accordance with the results of Northern blot analysis. The developmental expression of neuropsin mRNA in the pregnant uterus, visceral organs, and surface epithelia will be described separately.
Temporospatial Expression of Neuropsin mRNA in the Pregnant Uterus
In mice, blastocyst implantation begins at 4.5 d.p.c. At this stage, no positive hybridization signals above background were observed in either the implanting blastocyst or the uterus (data not shown). However, at 5.5 d.p.c., hybridization signals were observed in the primary decidual zone (Figures 2A and 2B). The most obvious signals appeared from 6.5 d.p.c. to 10 d.p.c. (Figures 2C–2H). During these stages, the decidua is subdivided into antimesometrial decidua, mesometrial decidua, and the glycogen-rich region that is situated between the antimesometrial and mesometrial decidua (Abrahamsohn and Zorn 1993). From 6.5 d.p.c. to 8.5 d.p.c., neuropsin mRNA was strongly expressed in both antimesometrial and glycogen-rich regions surrounding the embryo (Figures 2C–2F). However, from 8.5 d.p.c. to 10 d.p.c., expression of neuropsin mRNA was reduced and was dispersed in the antimesometrial region (Figures 2E–2H). In contrast, neuropsin mRNA expression in the glycogen-rich region of decidua maximized at 10 d.p.c. and then decreased rapidly, and at 11.5 d.p.c. its expression could only be seen in a layer of decidual cells facing the labyrinth of the placenta (Figures 2I and 2J). From 11.5 d.p.c. to 18.5 d.p.c., neuropsin mRNA expression remained the same as that at 11.5 d.p.c. Close examination at high-power magnification confirmed that the hybridization signals were not located on the giant trophoblast cells but on the decidual cells at 6.5 d.p.c. (Figure 3A) and 10 d.p.c. (Figure 3B). The control section hybridized with a sense probe did not show any significant signals (Figure 3C).
Neuropsin mRNA Expression in the Developing Heart, Lung, Pituitary, and Thymus
At 9.5 d.p.c., the developing heart showed low levels of neuropsin mRNA expression (data not shown). Neuropsin-positive cells were located in the trabeculae of the ventricle, whereas the atrium showed little or no expression of neuropsin. The expression in the trabeculae of the ventricle increased until 12.5 d.p.c. (Figures 4A–4C). At this stage, neuropsin mRNA was also obvious in pituitary, thymus, and lung (Figure 4A). Epithelial cells of the choroid plexus expressed neuropsin mRNA from this stage, as reported previously (Figure 4A) (Suzuki et al. 1995). Then the expression of neuropsin mRNA in the heart and lungs decreased at 14.5 d.p.c., although that in pituitary and thymus remained unchanged and continued after birth (data not shown). At 16.5 d.p.c. neuropsin mRNA was detectable in the thymus but was hardly detectable in the heart or lungs (Figure 4D). In the thymus, neuropsin mRNA expression was restricted to certain areas that coincided with the appearance of the precursors of the thymic medullary core (Kaufman 1992). At 18.5 d.p.c., neuropsin mRNA expression was undetectable by in situ hybridization (data not shown).
Neuropsin Expression in Skin and Surface Epithelia
Skin and other surface epithelia with keratinocytes expressed neuropsin mRNA (Figures 4 and 5). A rather late onset of neuropsin mRNA expression was first detected by in situ hybridization at 12.5 d.p.c. in the facial area (Figure 4A, arrowheads), whereas skin in other areas expressed neuropsin only faintly (Figure 5A). As shown in Figure 4D, neuropsin mRNA expression extended throughout the skin of the entire body at 16.5 d.p.c. Figure 5 shows the expression of neuropsin mRNA in the mid-back skin at high-power magnification at various developmental stages. At 12.5 d.p.c., only faint hybridization signals were seen in this area (Figure 5A). Thereafter, neuropsin gene expression gradually increased (Figures 5B and 5C), peaking at 18.5 d.p.c. (Figure 5D). Postnatal expression of the mRNA gradually decreased after the completion of epidermal morphogenesis and the expression became much weaker but still remained detectable in adults (Figure 5E). When the stratified squamous structure of the epidermis of the skin was well developed and the suprabasal layer could be clearly discriminated from the basal layer, neuropsin mRNA expression localized in the suprabasal cells, i.e., granule cells and spinous cells, but not in the basal cells (Figures 5C–5E). Close examination of counterstained sections indicated that neuropsin mRNA-expressing cells were a subpopulation of the suprabasal keratinocytes that migrate toward the surface while undergoing differentiation (Figure 5C, arrowheads). Neuropsin mRNA was not evenly expressed among different regions of the skin. Prominent expression was seen in the regions where the skin folded and thickened, e.g., in the regions of the snout, oral orifice, and developing limbs (Figures 4D and 6A). In the developing limbs, neuropsin mRNA was strongly expressed, especially in the regions between the digits (Figures 6A and 6B). The hybridization signals were also seen in mucosal epithelium with keratinocytes, such as the tongue (Figure 7F), esophagus (Figure 7G), and the forestomach from 12.5 d.p.c. embryonic to adult stages (Figures 7A–7D). The expression, however, unlike that in the skin, became stronger in adult animals (Figures 7A and 7B). The hybridization signals were also localized on the supra-basal cell layers close to the basal cell layer coinciding with those in the epidermis of the skin. It is noteworthy that cells in the basal cell layer that were in the mitotic cycle lacked neuropsin gene expression. Therefore, neuropsin was expressed in postmitotic migrating keratinocytes both in the skin and in the stratified squamous mucosal epithelia.
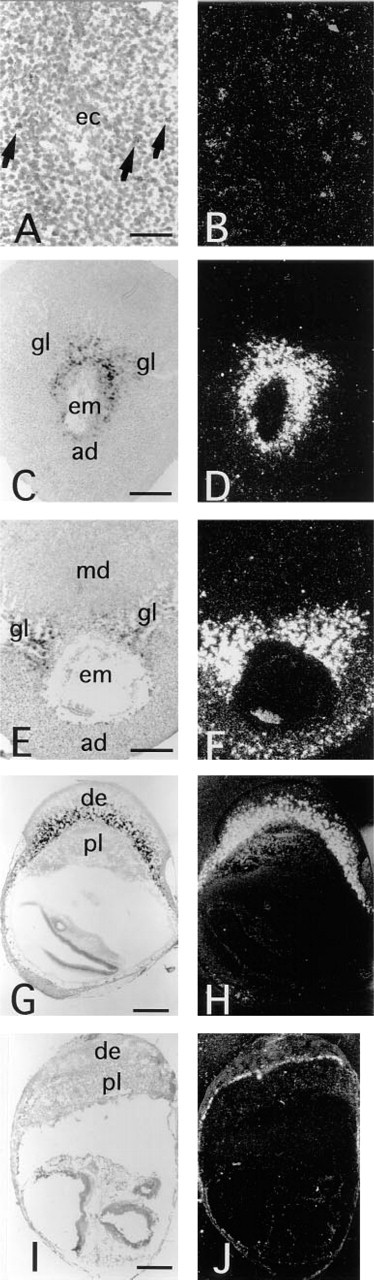
In situ hybridization of neuropsin mRNA in the pregnant uterus. Transverse sections were hybridized with anti-sense [35S]-RNA probes specific for mouse neuropsin. (
Discussion
In this study, we demonstrated widespread expression of neuropsin mRNA during organogenesis in mice and also in adult animals. Neuropsin mRNA was intensely expressed in the primary decidual cells, epithelial cells of choroid plexus, and keratinocytes in non-neural areas. In pregnant mice, decidual cells have mainly been believed to be associated with expression of certain protease inhibitors, whereas trophoblast cells were associated with the expression of most of the proteases involved in embryonic implantation and placentation (Strickland and Richards 1992). In contrast to this assumption, neuropsin mRNA was markedly expressed from 5.5 to 10 d.p.c. in the decidual cells, at which time placentation is most prominent. This dynamically variable stage coincides with the occurrence of functional and morphological changes in decidual cells (Schlafke et al. 1985; Yang et al. 1995). Therefore, neuropsin may be involved in maternal reactions to implantation. Proteolysis of extracellular matrix and/or adhesion molecules is important for implantation (Alexander et al. 1996). One of the possible targets of neuropsin is fibronectin which can be degraded with this protease (Shimizu et al., submitted for publication). Furthermore, in our preliminary experiment neuropsin anti-sense phosphorothioate oligonucleotides were delivered to the implantation site through a minipump from 5.5 d.p.c to 12.5 d.p.c., and the embryos were obviously smaller than in controls and the placenta showed some deficits (unpublished observation). Therefore, neuropsin might act on placentation and implantation, although further investigations are necessary to confirm this hypothesis.
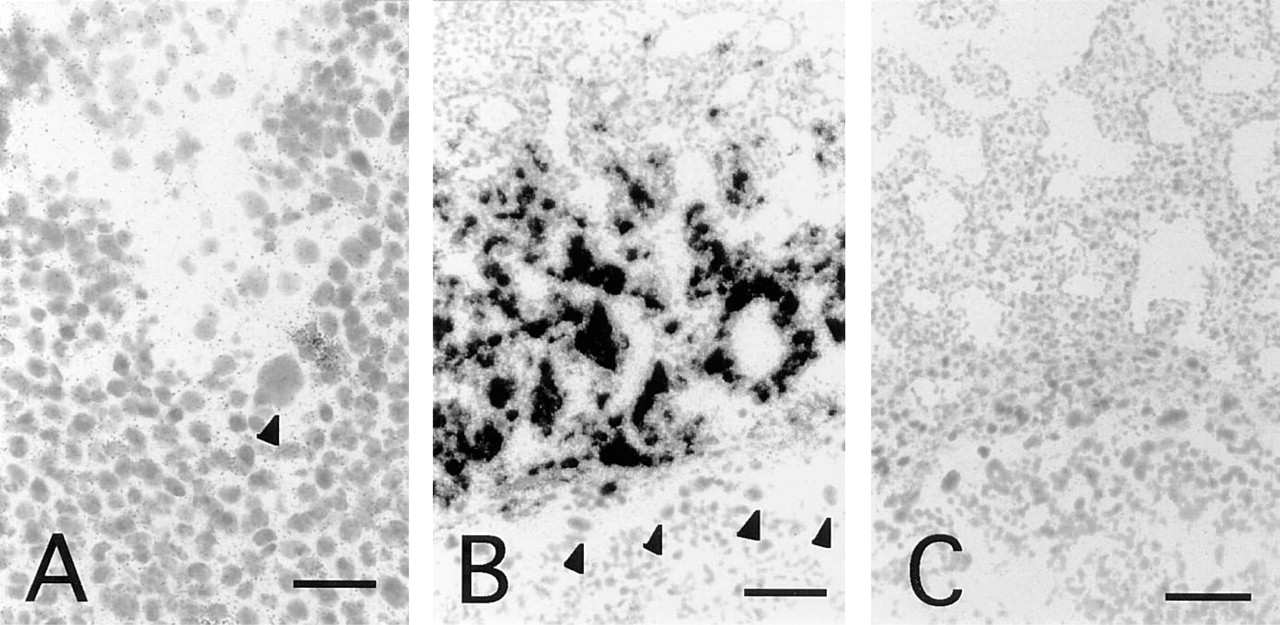
High-power magnification of decidual cells expressing neuropsin mRNA. (
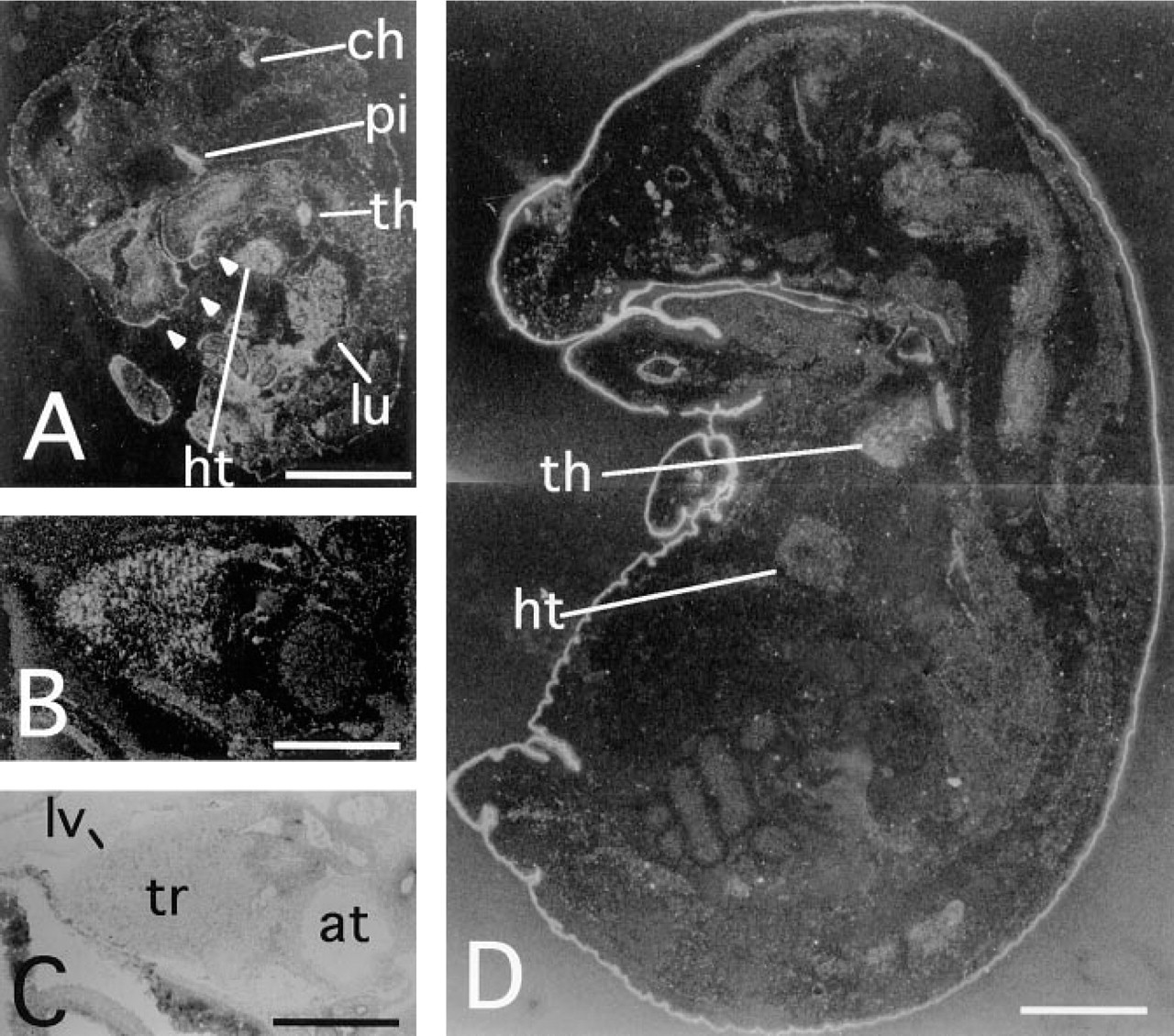
Neuropsin mRNA expression during mouse organogenesis. Darkfield micrographs of sagittal sections of (
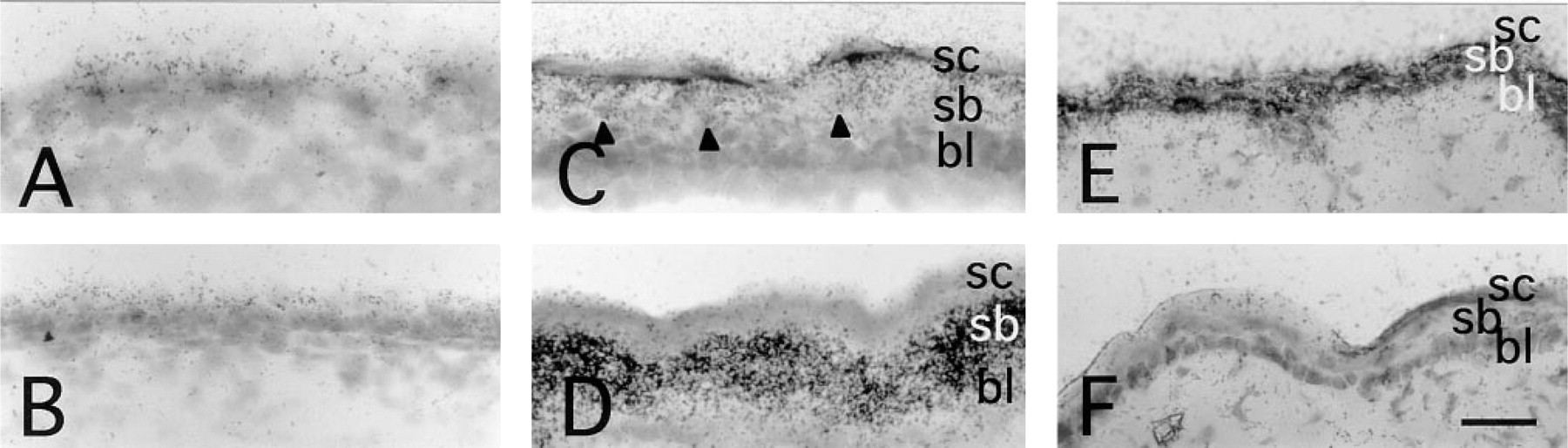
Developmental changes of neuropsin mRNA expression in the skin. Mid-back skin sections that were hybridized with 35S-labeled neuropsin anti-sense probe (
The development of the heart is accompanied by morphological changes by 14.5 d.p.c., when all of the components of the heart are readily recognizable (Kaufman 1992). The positive signals in the heart were first visible at 9.5 d.p.c, very strong at 12.5 d.p.c., and markedly decreased by 14.5 d.p.c. Neuropsin gene expression in the heart was restricted in the trabeculae of the ventricles. The trabeculation of the ventricles takes place most actively from 9 d.p.c. to 14.5 d.p.c. Other proteases, collagen and urokinase, were reported to be expressed in trabeculae at a similar stage and were believed to regulate migration of the cells (McGuire and Orkin 1992; Nakagawa et al. 1992). That the neuropsin gene expression also coincided with the active trabeculation might suggest a relationship between neuropsin and the migration of the cells forming the trabeculae. Neuropsin also exhibited transient expression in lung and thymus during a critical time window during which morphological formation occurs. Neuropsin expression was not detectable in these organs by in situ hybridization analysis or by Northern blot analysis in adults. Therefore, we hypothesize that neuropsin plays a role in organogenesis in heart, lung, and thymus rather than functioning in the mature organs.
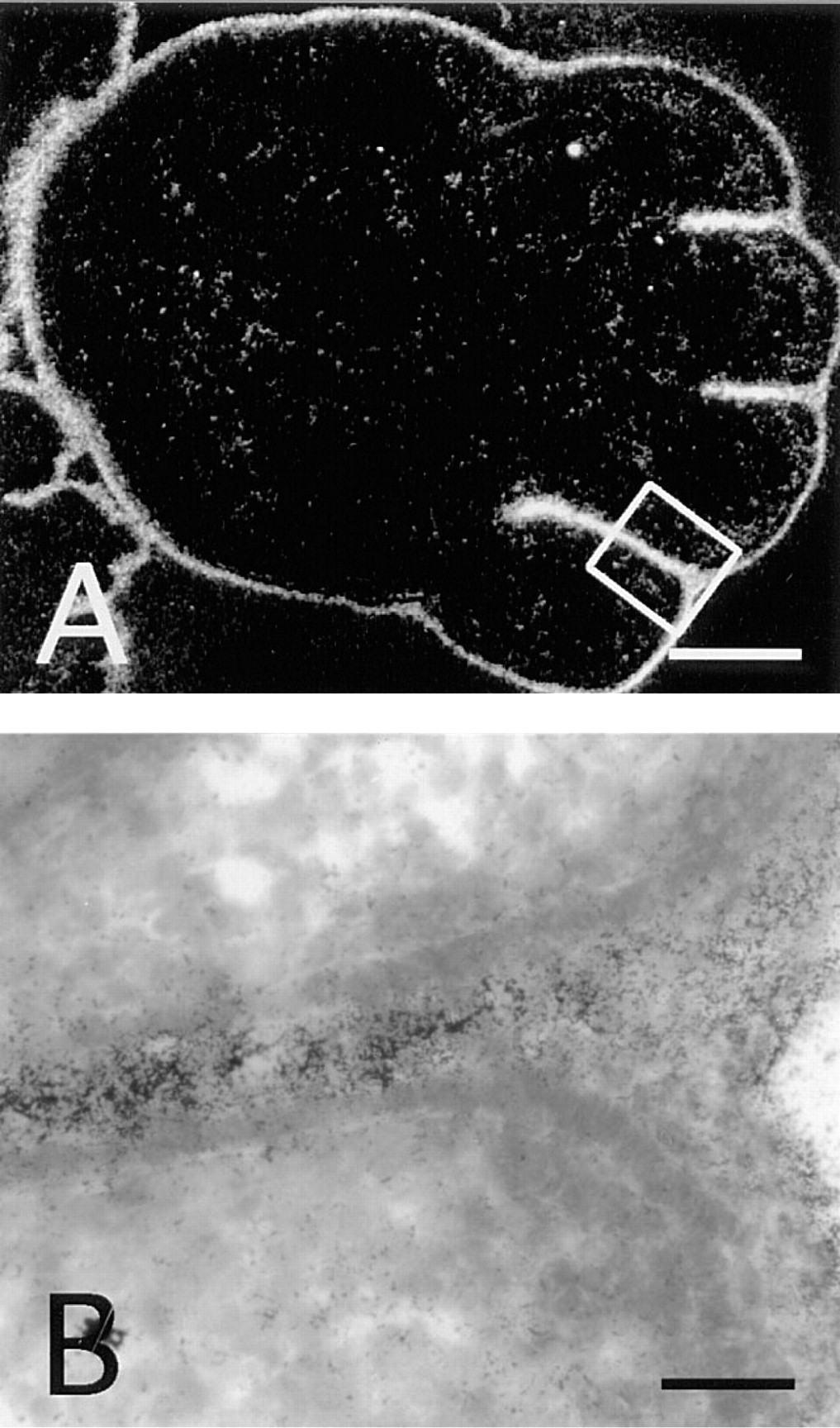
Neuropsin mRNA expression in the 15.5 d.p.c. forelimb skin. (
The developmental expression of neuropsin mRNA in the surface epithelia was classified into two patterns. In the epidermis of the skin that exhibits keratinization, neuropsin expression increased from 12.5 d.p.c. to 18.5 d.p.c. as the epidermis developed. Then, as the epidermis became thinner in the adult, neuropsin mRNA expression decreased. By contrast, in the mucosal epithelia of the oral cavity, esophagus, and fore-stomach, where physiological parakeratinization occurs in the mouse, neuropsin was expressed more strongly in adult animals. However, no neuropsin mRNA was detected in nonstratified epithelium in adult. It is very interesting that neuropsin was markedly expressed in keratinocytes wherever they were localized. The formation of multiple cell layers of the epithelium requires tremendous organization of cell–cell and cell–extracellular matrix adhesion. Adhesive structures must be disrupted in a regulated manner as the keratinocytes migrate vertically towards the outermost epithelial layer. Urokinase plasminogen activator was shown to be localized on the basal cell layer in human skin and is no longer expressed as the cells migrate towards the outer layer (Chen et al. 1993). The outermost layer of the skin has been shown to express another serine protease, stratum corneum chymotryptic enzyme, which is probably involved in desquamation (Hansson et al. 1994). The results of the present study suggest a novel serine protease localized in a suprabasal layer, which might be involved in keratinization and keratinocyte migration.
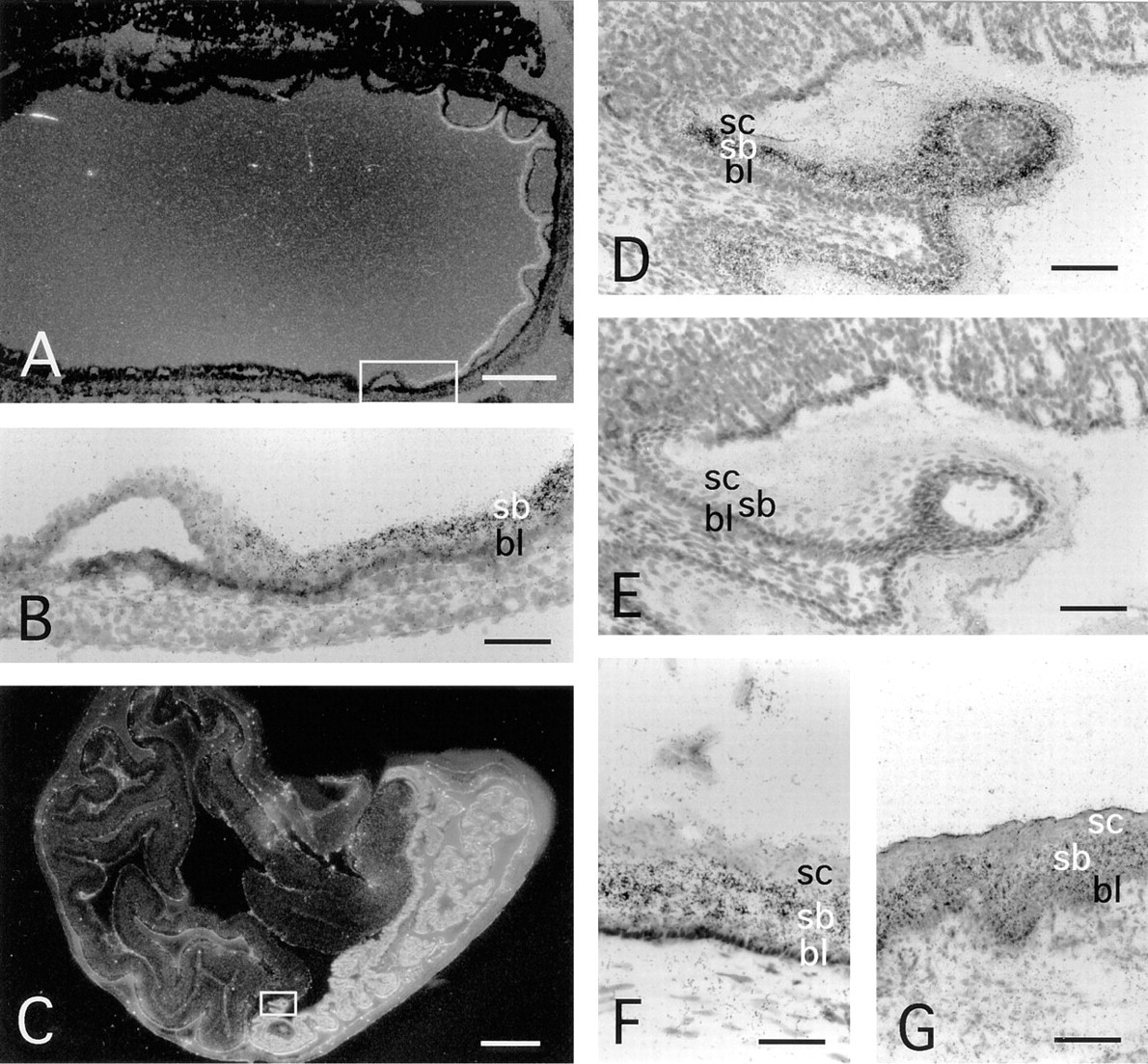
Neuropsin mRNA expression in the epithelia of the upper digestive tract. Sections of 16.5 d.p.c. embryonic stomach (
In conclusion, our results show that neuropsin mRNA was expressed in a wide range of tissues involved in a series of physiological processes. The common features of these processes are tissue remodeling and cell migration, based on extracellular proteolytic activity. The endogenous target substrates for the novel serine protease neuropsin are not clear. Subsequent biochemical and genetic studies will clarify the substrates and functions of neuropsin in vivo.
Footnotes
Acknowledgements
Supported by a Grant-in Aid for Scientific Research on Priority areas from the Ministry of Education, Science and Culture of Japan.
We thank Dr Ishida–Yamamoto of Asahikawa Medical College for suggestions regarding the manuscript.
