Abstract
Salivary carbonic anhydrase VI (CA VI) appears to contribute to taste function by protecting taste receptor cells (TRCs) from apoptosis. The serous von Ebner's glands locating in the posterior tongue deliver their saliva into the bottom of the trenches surrounding the TRC-rich circumvallate and foliate papillae. Because these glands deliver their saliva directly into the immediate vicinity of TRCs, we investigated whether CA VI is secreted by the von Ebner's glands, using immunochemical techniques. The immunohistochemical results showed that CA VI is present in the serous acinar cells, ductal cells, and ductal content of von Ebner's glands and in the demilune and ductal cells plus ductal content of rat lingual mucous glands. More importantly, CA VI was also detected in taste buds and in the taste pores. Western blotting of saliva collected from the orifices of human von Ebner's glands and CAs purified from rat von Ebner's glands confirmed that CA VI is expressed in these glands and secreted to the bottom of the trenches surrounding the circumvallate and foliate papillae. These findings are consistent with the hypothesis that locally secreted CA VI is implicated in the paracrine modulation of taste function and TRC apoptosis.
(
C
Von Ebner's glands are small serous glands lying among lingual muscle fibers in the posterior tongue. Saliva from von Ebner's glands contains both specific proteins and common proteins produced by other exocrine glands (Calvert et al. 1993; Li and Snyder 1995). Saliva from von Ebner's glands is delivered directly into the bottom of the trenches surrounding the circumvallate and foliate papillae. Most human taste buds locate on the lateral aspects of these papillae and open directly into these trenches (Moss–Salentijn and Klyvert 1980). Taste buds contain epithelial taste receptor cells (TRCs) that transmit taste stimuli to the nervous system (Wong et al. 1996). Taste reception is a complex process involving both extraepithelial and intraepithelial molecules plus transmembrane proteins (Kinnamon et al. 1988; Wong et al. 1996; Chandrashekar et al. 2000). The saliva from von Ebner's glands is known to modulate the microenvironment at taste reception sites in the trenches of the circumvallate and foliate papillae which, in turn, affects taste reception (Gurkan and Bradley 1988). The von Ebner's gland saliva is also an important source of growth factors for TRCs, because removal of submandibular and sublingual salivary glands damages all TRCs in the dorsum of the tongue but not those located in the circumvallate papillae (Morris–Wiman et al. 2000). Rat taste buds and von Ebner's glands have been shown to contain CA activity (Brown et al. 1984), which may contribute to taste function. This hypothesis was based on the previous observations that CA inhibitors alter the perception of carbonated water (Graber and Kelleher 1988; Simons et al. 1999). Of the CA isoenzymes, CA II has also been localized in the taste buds, whereas the isoenzyme(s) expressed in the von Ebner's glands has remained undefined (Daikoku et al. 1999). Interestingly, Henkin et al. (1999a) recently demonstrated that patients with taste loss have a lower salivary CA VI concentration and more apoptosis in TRCs than controls. Elevation of the salivary CA VI concentration in those patients using zinc treatment resulted in recovery of taste perception and TRC morphology (Henkin et al. 1999b). The present study was based on the hypothesis that CA VI is secreted, in addition to the large salivary glands, also by the von Ebner's glands, which are located at an optimal site for the delivery of CA VI into the immediate proximity of TRCs in the circumvallate and foliate papilla taste buds.
Materials and Methods
Antibodies and Chemicals
Peroxidase-conjugated goat anti-rabbit IgG, biotin-conjugated swine anti-rabbit IgG, and streptavidin-conjugated peroxidase were obtained from Sigma Chemicals (St Louis, MO). Freund's adjuvants were obtained from Gibco (Grand Island, NY). CM Bio-Gel A, p-aminomethyl benzene-sulfonamide, and all reagents for SDS-PAGE and transblotting were purchased from Bio-Rad Laboratories (Richmond, CA). Complete protein inhibitor tablets were purchased from Boehringer–Mannheim (Mannheim, Germany). Pilocarpin was a product of Santen (Napa, CA).
Tissue and Saliva Samples
Ten male Sprague–Dawley rats were sacrificed and their tongues, kidneys, and parotid glands were dissected out. Rat saliva was collected from anesthetized animals. Secretion of saliva was stimulated by injecting 6 mg/kg of pilocarpin IP. The rats were bled from the heart after collection of saliva. Human tongue and parotid gland specimens were obtained from the Department of Forensic Medicine, University of Oulu, and from patients undergoing surgery for lingual carcinoma at the Clinic of Otorhinolaryngology, Oulu University Hospital. Saliva from human von Ebner's glands was collected from healthy volunteers’ circumvallate papillae with a micropipette and from foliate papillae with nitrocellulose sheets (Spielman et al. 1993). Human whole saliva was collected into test tubes as described by Parkkila et al. (1990).
Production of Anti-CA VI Sera
Rat CA VI was purified from saliva using inhibitor-affinity chromatography. The saliva samples were centrifuged for 10 min at 13,000 × g at 4C and the supernatant was used for purification. An affinity chromatography matrix was prepared by coupling the CA inhibitor p-aminomethyl benzene-sulfonamide to CM Bio-Gel A according to Khalifah et al. (1977). The rest of the purification was performed as described by Murakami and Sly (1987). Purified rat salivary CA VI was mixed with SDS sample buffer and subjected to PAGE under non-reducing conditions using 4% loading and 10% stacking polyacrylamide gels (Laemmli 1970). The proteins were visualized using Coomassie Brilliant Blue staining. The 41-kD band of CA VI was cut off the gel and crushed. Coomassie Brilliant Blue stain was removed from the gel crush by extensive washing in a solution containing 40% methanol, 10% ethanol, and 50% distilled water. The gel crush was carefully mixed with Freund's adjuvant. Antigen (100 μg) was injected intradermally into a rabbit's back at 4-week intervals. The first two injections were given with complete Freund's adjuvant and the last two with incomplete Freund's adjuvant. Four weeks after the fourth injection, rabbit blood was collected through a heart puncture and the serum was isolated by centrifugation.
The production and characterization of rabbit anti-human CA VI serum have been described by Parkkila et al. (1990). The crossreactivity of this antibody has been tested in dot-blots, Western blotting, and time-resolved immunofluorometric assays, in which it has shown high isoenzyme specificity (Parkkila et al. 1990, 1993).
Immunohistochemistry and Western Blotting
Tissue samples for immunohistochemistry were fixed in Carnoy's fluid or 4% paraformaldehyde for 18 hr and embedded in paraffin. Thick sections of 5 μm were subjected to immunohistochemistry. Immunohistochemical staining was performed using the immunoperoxidase procedure as described by Kivelä et al. (2000).
Parotid glands, rat von Ebner's glands, and kidneys were homogenized in 0.1 M Tris-SO4, pH 8.7, containing Complete protease inhibitors dissolved and diluted according to the manufacturer's instructions. The homogenized parotid glands, rat von Ebner's glands, and saliva samples were then centrifuged for 10 min at 13,000 × g at 4C. The resulting supernatants were used for the experiments. CAs were purified from the homogenized von Ebner's glands using inhibitor-affinity chromatography as described in detail by Karhumaa et al. (2000). The homogenized kidney samples were first centrifuged at 1000 × g for 10 min, followed by centrifugation of the supernatants at 70,000 × g for 90 min. The membrane pellet was used for the experiments. Red blood cells were isolated from the whole blood by gentle centrifugation. The cell pellet was lysed with an equal volume of distilled water. The human and rat saliva supernatants, the purified CAs obtained from the rat von Ebner's glands, rat kidney membranes, rat red cell extracts, and the nitrocellulose sheet containing human foliate papilla saliva were dissolved in SDS sample buffer. The samples were then subjected to SDS-PAGE under reducing conditions using 4% loading and 10% stacking polyacrylamide gels (Laemmli 1970). The separated proteins were transferred to nitrocellulose sheets according to a standard method (Towbin et al. 1979). Polypeptide bands were immunostained using anti-CA VI serum and visualized using enhanced chemiluminescence (ECL; Amersham, Poole, UK) as described earlier in detail (Leinonen et al. 1999).
Results
Characterization of Anti-rat CA VI Serum
Rabbit antiserum raised against rat CA VI identified two major polypeptides of 41 and 37 kD in rat saliva, purified rat CA VI, and homogenized parotid gland plus a minor 56-kD polypeptide in rat whole saliva (Figure 1, Lanes 1–3, respectively). The 56-kD polypeptide band is probably attributable to protein complex formation because its intensity increased during storage of the sample (data not shown). The 37-kD band most probably represents a partially deglycosylated form of CA VI reported earlier in the human enzyme (Murakami and Sly 1987). The antiserum did not detect rat CA II or CA I from red cell extracts or CA IV from isolated kidney membranes (data not shown). Preimmune serum did not recognize any polypeptide bands in the control immunoblots (data not shown).
Immunohistochemistry
Immunohistochemical staining shows that CA VI is abundantly expressed in von Ebner's glands, which empty into the bottom of the trenches of the papillae (Figure 2A). An intense peroxidase reaction is seen in the serous acinar cells of both human (Figure 2B) and rat (Figure 2C) von Ebner's glands. In addition, CA VI was found in the demilune cells of rat lingual mucous glands (Figure 2D). Ductal content and cells in rat von Ebner's glands and mucous glands also stained for CA VI (Figures 2C and 2D). Interestingly, taste pores and some cells in rat taste buds stained specifically for CA VI (Figures 2E and 2F). Control sections stained with preimmune serum remained unstained (Figures 2G and 2H). Sections from rat parotid and submandibular glands stained for positive control with anti-CA VI serum yielded a specific reaction in serous acinar cells (data not shown).
Western Blotting
Immunoblotting experiments confirmed that the immunohistochemical stainings represent CA VI staining. Antiserum to human CA VI recognized a strong 40-kD polypeptide band from circumvallate and foliate papillae saliva (Figure 3, Lanes 1 and 2, respectively). A similar 40-kD band was also recognized in human whole saliva, purified human salivary CA VI, and homogenized human parotid gland (Figure 3, Lanes 3–5, respectively). In addition, a weak 56-kD polypeptide band was seen in the human von Ebner's gland saliva collected from the foliate papilla. Antiserum to rat CA VI recognized 41-kD and 37-kD polypeptide bands in the rat CA VI purified from von Ebner's glands (Figure 3, Lane 6). Preimmune serum did not recognize any polypeptide bands in the control immunoblots (data not shown).
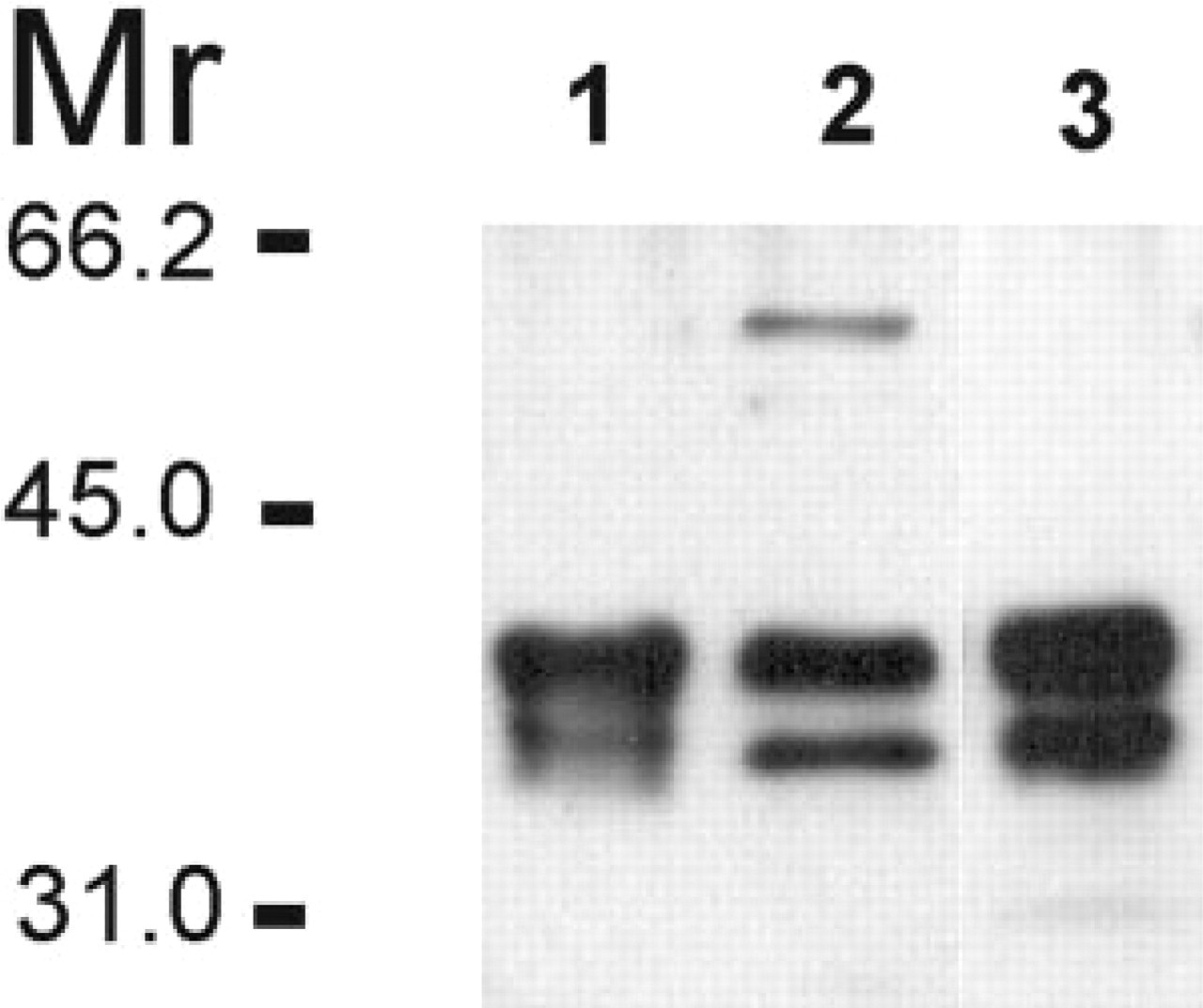
Immunoblots of rat samples. Lane 1, rat CA VI purified from saliva. Lane 2, rat whole saliva. Lane 3, rat parotid gland.
Discussion
Recently, Henkin et al. (1999a,b) proposed that salivary gustin, a protein identical to salivary CA VI (Thatcher et al. 1998), is capable of modulating taste functions. Some patients suffering from taste loss show low CA VI concentrations in whole saliva associated with increased apoptosis in TRCs in the circumvallate papilla taste buds (Henkin et al. 1999a). Elevation of the salivary CA VI concentrations in those patients using zinc treatment restored taste reception and TRC morphology (Henkin et al. 1999b). Previous studies from our laboratory have shown that CA VI is abundantly expressed in the large salivary glands (submandibular and parotid glands) (Parkkila et al. 1990), suggesting that CA VI present in whole saliva could be responsible for the modulation of taste functions. There are, however, serous salivary glands, called von Ebner's glands, located among lingual muscle fibers in the posterior tongue (Moss–Salentijn and Klyvert 1980). They deliver their saliva directly into the bottom of the trenches surrounding the circumvallate and foliate papillae. The walls of the trenches contain most of the human taste buds that open into these trenches (Moss–Salentijn and Klyvert 1980). Here, we demonstrate that von Ebner's glands abundantly express CA VI and deliver it directly into these trenches in the immediate proximity of the taste buds. Because whole saliva does not readily enter the trenches, it is feasible that this locally secreted CA VI is directly linked to the taste bud function. This view is also supported by the finding that removal of submandibular and sublingual glands damages the TRCs in the dorsum of the tongue but not those located in the circumvallate papillae (Morris–Wiman et al. 2000).
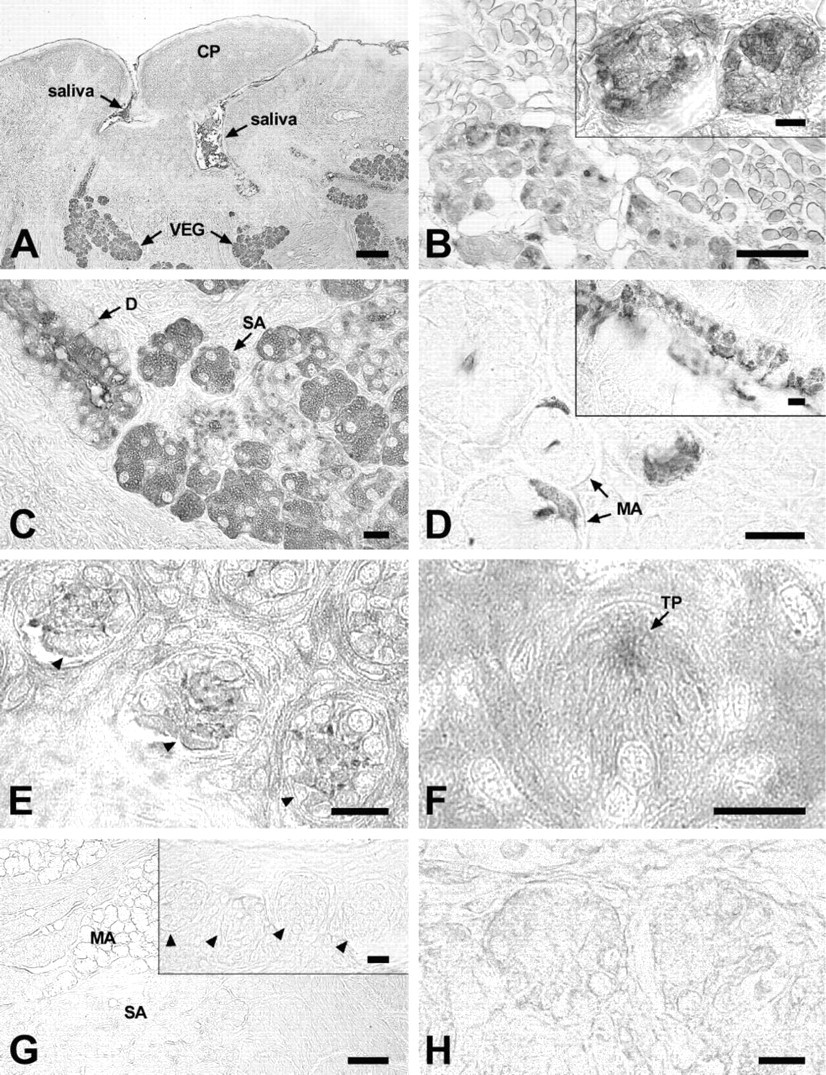
Immunohistochemical localization of CA VI in rat and human tongue sections. (
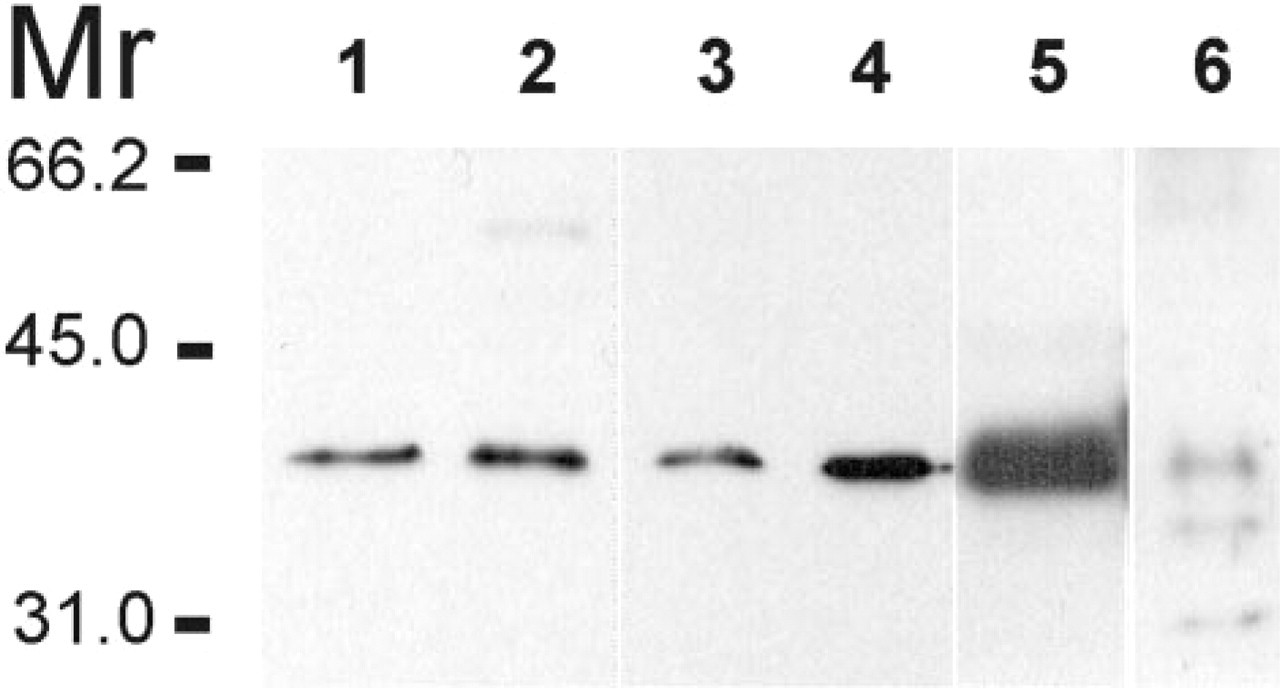
Immunoblots of human von Ebner's gland saliva collected from circumvallate and foliate papillae (Lanes 1 and 2, respectively), human CA VI purified from saliva (Lane 3), human whole saliva (Lane 4), human parotid gland (Lane 5), and rat CA VI purified from von Ebner's gland (Lane 6).
Although we provide no functional data on the linkage of CA VI with taste function here, at least three possible roles for CA VI can be proposed on the basis of previous studies. First, CA VI is an enzymatically active member of the α-CA gene family and, accordingly, the enzyme present in von Ebner's gland saliva may be linked to pH in the immediate proximity of the taste buds. Acid stimuli are known to inactivate the proton-gated potassium channels that result in depolarization of TRCs (Kinnamon and Roper 1987, 1988). CA VI secreted directly into the close proximity of the taste buds may catalyze the removal of excess protons and thereby speed up the recovery of the proton-gated potassium channels.
Second, CA VI present in von Ebner's gland saliva may also affect the apoptosis of TRCs in the taste buds of the circumvallate and foliate papillae (Henkin et al. 1999a,b) via pH regulation. It has been shown that extracellular pH affects intracellular pH in TRCs (Lyall et al. 1997). Furthermore, it is also known that long-standing exposure of epithelial cells to low pH leads to apoptosis (Hamilton et al. 1998). Accordingly, being a high-activity carbonic anhydrase isoenzyme (Feldstein and Silverman 1984), CA VI may protect TRCs in the taste buds of the circumvallate and foliate papillae from apoptosis by regulating the pH in TRCs.
Third, CA VI may function as a paracrine growth factor in TRCs. This hypothesis is supported by the present finding that CA VI was indeed located in the taste pores and in some cells within the taste buds. The CA VI-positive cells in the taste buds were not identified here. Nevertheless, they do not appear to represent CA II-producing cells (Type I) because these cells are more abundant than the CA VI-positive cells (Daikoku et al. 1999). It is known from previous studies that von Ebner's gland saliva is an important source of growth factors for TRCs, because the removal of submandibular and sublingual salivary glands damages TRCs only in the dorsum of the tongue, whereas those locating in the circumvallate papillae remain unaffected (Morris–Wiman et al. 2000). The putative growth factors present in von Ebner's gland saliva are undefined, but its CA VI is one good candidate because of its reducing effect on the apoptotic process in circumvallate papillae TRCs (Henkin et al. 1999,b) and its other growth-factor like properties (Henkin et al. 1988). Our results demonstrate that CA VI is secreted by the serous von Ebner's glands. The presence of CA VI in von Ebner's gland saliva, in the taste pores, and in some cells in the taste buds is consistent with the hypothesis that locally secreted CA VI plays a paracrine role in taste reception and TRC apoptosis.
Footnotes
Acknowledgements
Acknowledgments
Supported by grants from the Finnish Cultural Foundation and the Finnish Dental Association to Jukka Leinonen.
We gratefully thank Ms Lissu Hukkanen, Ms Pirkko Peronius, and Mr Eero Oja for technical assistance.
