Abstract
Psoriasis is a complex chronic inflammatory skin disease characterized by epidermal thickening on the basis of increased keratinocyte proliferation and insufficient apoptosis. Laminins are important components of the basement membrane (BM) and impact on epidermal keratinocyte growth/apoptosis. Although several laminins are involved in the pathogenesis of psoriasis, it is still controversial about the expression patterns of laminin isoforms and which laminins are important in the development of psoriasis. Because laminin-511 and -332 are key BM components in human skin, and laminin-511 stimulates human hair follicle growth, we asked whether the BM zone in psoriasis shows any laminin-related abnormalities. This showed that the BM expression of laminin-511 and -332 was significantly increased within the skin lesion of psoriasis. Immunofluorescence microscopy revealed that laminin-511, -332, and collagen type IV proteins were also significantly increased in psoriasis-like skin lesions of Imiquimod-treated mice. Transmission electron microscopy showed a few gaps of lamina densa, and its thickness was significantly increased. Finally, laminin-511 treatment significantly stimulated the proliferation and inhibited apoptosis of HaCaT cells, while laminin-α5 chain gene knockdown decreased proliferation and induced apoptosis. These phenomenological observations raise the question of whether laminin-511-controlled keratinocyte growth/death may be a previously overlooked player in the pathogenesis of psoriatic epidermal lesions.
Introduction
Psoriasis is a chronic inflammatory skin disease that is characterized by hyperproliferation of keratinocytes (KC), along with insufficient KC apoptosis1,2 and hyperkeratosis. 3
As a treatment, topical therapies including glucocorticosteroids, vitamin D derivatives, and calcineurin inhibitors, a systemic therapy by cyclosporin, methotrexate, or retinoic acid derivatives, and phototherapies are used. Although several effective antipsoriatic drugs including biologicals have been developed, severe psoriasis remains a major therapeutic challenge, and novel treatment strategies that go beyond the mainstream intervention principles of current biologicals4–6 are most postulated.
Here, we explore the working hypothesis that the basement membrane (BM) zone, which divides epidermis and dermis, deserves greater attention in psoriasis pathobiology and possibly psoriasis management than it has been considered so far.
The BM is composed of extracellular matrix (ECM) proteins, including various types of collagen, elastin, fibronectin, laminin, fibrin, perlecan, entactin, and nidogen.7–11 These ECM proteins interact with different cell types, particularly epidermal cells, fibroblasts, and inflammatory cells 12 to regulate cell proliferation, cell differentiation, cell motility, and apoptosis.13,14
However, the role of ECM proteins in the development of psoriatic epidermal lesions remains to be fully investigated.15–17 Notably, however, McFadden et al. have proposed a laminin-dependent mechanism by which self-limiting guttate psoriasis caused by streptococcal pharyngitis develops to chronic psoriasis. Briefly, streptokinase, which is produced by streptococci, can initiate lysis and disruption of laminin.18–20 This may then promote KC instability and proliferative tendency via inducing the interaction of fibronectin with α5β1 integrin.18–20 To support this hypothesis, other studies have also reported that the disruption of laminin can be detected in psoriasis skin. Mondello et al. reported that the expression of laminin-1 (composed of α1, β1, and γ1 chains) was discontinuous and fragmented in uninvolved psoriatic skin. 21 Vaccaro et al. reported that laminin α1 chain expression was discontinuous both in involved and uninvolved psoriatic skin, while the expression of fibronectin was overexpressed in the papillary dermis. 22 Toti et al. found that laminin α2 chain expression was weak and irregular in psoriatic skin lesion. 23 However, in contrast to the observations by others,21,22 they also reported that laminin α1, β1, and γ1 chains expression were normal. 23 Fleischmajer et al. reported that the expression of laminin α2, α5, β1, and γ1 chains were reduced in the involved area of psoriasis. 15
Because the psoriatic epidermal lesion and active hair growth (anagen development) share a number of intriguing, yet underappreciated, common features, 24 it is interesting to note that laminins play an important role in hair follicle (HF) biology.25–29 We have previously shown that laminin-332, which is composed of α3, β3, and γ2 chains, and laminin-511 composed of α5, β1, and γ1 chains represent key laminins in human skin. 9 These laminins play important roles as regulators of KC migration in wound healing and tumorigenesis and modulate skin appendage development. 9 During the hair cycle, laminin-332 and its binding partner, α6β4 integrin, are not expressed along the BM of the HFs during the anagen and then expressed from midcatagen to telogen.28,29 In line with this, the number of hemidesmosomes is decreased along the BM of the lower HF at late anagen. 28 In contrast, laminin-511 and its binding partner α3β1 integrin are stably expressed along the BM of the whole HF during the hair cycle.28,29 Finally, we have shown by using the normal human HF organ culture system that laminin-511 stimulated human HF growth, while laminin-332 inhibited it.28,30 Thus, laminin-511 operates largely as a hair growth stimulator, while laminin-332 appears to work as a brake on HF growth by increasing the number of hemidesmosomes.9,28–30
Therefore, we hypothesized that similar differential effects of laminins on epidermal KC proliferation may also play a role during the development of the psoriatic epidermal lesion,2,31 and have asked in the current study whether laminin-332 and -511 expression is abnormal in the BM zone of psoriatic skin lesions, and whether laminin-511 has an impact on KC proliferation and apoptosis.
Materials and Methods
Antibodies
In mice samples, a rabbit polyclonal antiserum termed J18 generated against all three subunits of laminin-332, a rabbit polyclonal antibody against mouse recombinant laminin-α5, a rat monoclonal antibody against mouse laminin-β1 (LT3, ab44941; Abcam; Cambridge, UK), a rat monoclonal antibody against mouse laminin-γ1 (SPM277, ab17792; Abcam), a rat monoclonal antibody against mouse β1 integrin (MB1.2, MAB1997; Merk, Millipore; Darmstadt, Germany), a rat monoclonal antibody against mouse β4 integrin (BD Biosciences; Franklin Lakes, NJ), a rabbit polyclonal antibody against mouse fibronectin (ab2413; Abcam), and a rabbit polyclonal antibody against type IV collagen (ab6586; Abcam) were used.
In human samples, a rabbit polyclonal antibody against human laminin-5 (ab14509; Abcam) and a mouse monoclonal antibody against human laminin-α5 (4B12, MABT39; Merk, Millipore), and a rat monoclonal antibody against human laminin-γ1 (ab17792; Abcam) were used.
For HaCaT cells, a mouse monoclonal antibody against human laminin-α5 (Merk, Millipore), a mouse monoclonal antibody against Ki-67 (MIB-1, M7240; Dako; Santa Clara, CA), and a rabbit polyclonal antibody against cleaved caspase-3 (D175; Cell Signaling Technology; Danvers, CO) were used.
Goat anti-rabbit IgG Alexa Fluor 594, goat anti-rat IgG Alexa Fluor 568, goat anti-rabbit IgG Alexa Fluor 488, and goat anti-mouse IgG Alexa Fluor 488 were obtained from Molecular Probes (Eugene, OR). FITC-conjugated goat anti-rabbit IgG was obtained from Zymed Laboratories Inc. (South San Francisco, CA). FITC-conjugated goat anti-mouse IgG was obtained from CHEMICON (Tokyo, Japan).
Mice and Treatment
We used 8-week-old C57BL/6 mice to induce psoriasiform skin lesions by topical Imiquimod application as previously described.32–34 Mice were purchased from Japan SLC Inc., and provided with food and water ad libitum.
All experiments were approved by the animal ethics committee.
Mice at 8 weeks of age received a daily topical dose of 62.5 mg of commercially available Imiquimod (IMQ) cream (5%; Mochida Pharmaceutical Co., Ltd.; Tokyo, Japan) on the shaved back for 5 consecutive days, translating to a daily dose of 3.125 mg of the active compound. This dose was empirically determined to cause most optimal and reproducible skin inflammation in mice (data not shown).
We obtained back skin specimens of the mice that received IMQ cream just after the termination of drug application (on day 6, after 5 consecutive days of treatment).
Control mice were treated similarly with a control vehicle cream (Vaseline Lanette cream; Kenei Pharmaceutical Co., Ltd.; Osaka, Japan).
Human Samples
Human normal skin samples were obtained from peripheral skin after the benign skin tumorectomy. The number of normal human skin samples is 5 (aged 64–81 years; average, 72.6 years). The parts of the resected benign tumors are abdomen (two subjects), lower limb (two subjects), and head (one subject).
Human skin samples of psoriasis were obtained from Kochi University and Nagoya City University. The number of human skin samples of psoriasis is 5 (aged 34–63 years; average, 50.4 years). We evaluated the stage of psoriatic lesion of human skin samples by H&E histochemistry. All of them showed “absence of spongiosis,” “parakeratosis with neutrophils collections,” “decreased granular zone within the epidermis,” and “psoriasiform hyperplasia with rete ridges.” Therefore, we considered skin samples that we obtained for this study as the “fully developed” stage.
Human tissue collection and handling were performed according to Declaration of Helsinki guidelines, with institutional research ethics committee approval (Osaka City University, Kochi University, and Nagoya City University), and written informed consent was obtained from the patients.
Histopathology and Immunohistochemistry (IHC)
For histochemical examination, skin specimens of mice were fixed in 4% paraformaldehyde in PBS, dehydrated, and embedded in paraffin. Sections of 4-µm thickness were cut and stained with H&E.
For IHC, fresh skin specimens of mice were embedded in physiological saline and frozen in liquid nitrogen. Samples from the back were immersed in TissueTek (Sakura Finetek; Japan), snap-frozen in liquid nitrogen, and stored at −80C until use. Cryosections of 4 µm of snap-frozen skin were cut using a cryostat.
For laminin-332 and β4 integrin staining, tissue sections were treated with 1% Triton X-100 in PBS for 10 min and then fixed in 2% formaldehyde in PBS for 20 min at room temperature. For laminin-α5, -β1, -γ1, and fibronectin staining, tissue sections were treated with 1% Triton X-100 in PBS for 10 min and fixed in acetone at 4C for 2 min. For β1 integrin and Ki67 staining, tissue sections were fixed in 3.7% formaldehyde in PBS for 5 min and treated with 1% Triton X-100 in PBS for 10 min. Sections were washed in PBS, and incubated overnight at 4C with primary Abs against laminin-α5, -β1, -γ1, -332, β4 integrin, β1 integrin, fibronectin, and Ki-67. Secondary antibodies in PBS at room temperature were then added for 45 min. The following secondary antibodies were used: goat-anti rabbit IgG Alexa Fluor 594 for laminin-α5 and fibronectin and Ki-67, and goat-anti rat IgG Alexa Fluor 568 for laminin-β1, γ1, and β1 integrin, and FITC-conjugated goat-anti rabbit IgG for laminin-332 and β4 integrin. Sections were washed in PBS, mounted, and examined by fluorescence microscope (BZ-8000; KEYENCE; Osaka, Japan).
For IHC, fresh-skin specimens of human and mouse back skin were immersed in TissueTek (Sakura Finetek; Japan), snap-frozen in liquid nitrogen, and stored at −80C until use. Cryosections of 5 µm of snap-frozen skin were cut using a cryostat. We used 5-µm cryosections for human skin samples, while 4-µm cryosections were used for mouse skin samples. For laminin-332, α5, and -γ1 staining, tissue sections were treated with 1% Triton X-100 in PBS for 10 min and fixed in acetone at 4C for 2 min. Sections were washed in PBS, and incubated overnight at 4C with primary Abs against laminin-332, α5, and -γ1. The following secondary antibodies were used: goat-anti rabbit IgG Alexa Fluor 488 for laminin-332, goat anti-mouse IgG Alexa Fluor 488 for laminin-α5, FITC-conjugated goat anti-rabbit IgG for laminin-γ1. Sections were washed in PBS, and treated with 4′,6-diamidino-2-phenylindole (DAPI) for 1 min. They were washed in PBS, mounted, and examined by fluorescence microscope. Normal human or murine skin samples were used as positive controls for laminin-α5, -β1, -γ1, -332, β4 integrin, β1 integrin, fibronectin, Ki-67, and cleaved caspase-3 immunofluorescence. Immunofluorescence without primary antibodies was used as negative controls.
Transmission Electron Microscopy (TEM)
Skin specimens were fixed in 0.1 M cacodylate buffer containing 2.5% glutaraldehyde and 2% paraformaldehyde and were then immersed in 1% osmium tetroxide in the same buffer. The tissue was dehydrated in a gradient series of ethanol, immersed in propylene oxide, and embedded in plastic resin. Thin and thick sections were generated on a Leica Ultracut UCT (Leica; Wetzlar, Germany). Thin sections were stained with uranyl acetate and lead citrate and observed with an electron microscope (H-7500; HITACHI; Tokyo, Japan). To evaluate the number of hemidesmosomes, we counted the number of hemidesmosomes and measured the length of lamina densa per each visual field. Then, we calculated the density of hemidesmosomes (the number of hemidesmosomes per 1 nm of lamina densa). For evaluating the thickness of lamina densa, we measured its thickness at 5 points per one image by using ImageJ software v1.49 (National Institutes of Health). At least 5 images per 1 mouse were used for the evaluation.
HaCaT Cells Culture and IHC
We performed HaCaT cells culture with vehicle and laminin-511 (at 0.1 μg/ml and 1 μg/ml, 24 hr) to evaluate their proliferation/apoptosis. In addition, we performed laminin-α5 gene knockdown in HaCaT cells using siRNA. Transfection duration was 6 hr and then cultured with normal supplemented medium for 15 hr. Laminin-511 was obtained from BioLamina (Sundbyberg, Sweden), and laminin-α5 siRNA was obtained from Santa Cruz Biotechnology (Dallas, TX).
For laminin-α5, Ki-67, and cleaved caspase-3 staining by immunohistochemical examination, tissue sections were treated with 4% paraformaldehyde in PBS for 5 min. Sections were washed in PBS, and incubated overnight at 4C with primary Abs against laminin-α5, Ki-67, and cleaved caspase-3. Secondary antibodies in PBS at room temperature were then added for 45 min. The following secondary antibodies were used: FITC-conjugated goat anti-mouse IgG for laminin-α5 and Ki-67, FITC-conjugated goat anti-rabbit IgG for cleaved caspase-3. Sections were washed in PBS, and treated with DAPI for 1 min. They were washed in PBS, mounted, and examined by fluorescence microscope.
HaCaT cells were cultured in DMEM supplemented with Penicillin streptomycin and FBS.
Measuring Fluorescence Intensity by Using NIH ImageJ
We used NIH ImageJ software for measuring fluorescence intensity of laminin-α5, β1, γ1, and laminin-332, β4 and β1 integrins, and fibronectin. Mean fluorescence intensity value within the basal membrane or dermis (for fibronectin) was measured per each image. At least 10 images were evaluated per each immunofluorescence study.
Statistical Analysis
Data were analyzed by using either the Mann-Whitney U test for unpaired samples or Dunn’s multiple comparison test by using Prism 6.0 software (GraphPad Prism; GraphPad Software; San Diego, Calif). Regarded as significant were p values of less than 0.05. All data in the figures are expressed as mean ± standard error of the mean (SEM).
Results
The Expression of Laminin-332 and -511 Is Significantly Upregulated in Psoriatic Lesions
First, we investigated the expression of main laminins within human skin, namely, laminin-332 (composed of α3, β3, and γ2 chains) and -511 (composed of α5, β1, and γ1 chains)9,11 of lesional area of psoriasis vulgaris. Immunofluorescence microscopy using antibodies against laminin-332, laminin-α5, and γ1 chains showed that the expression of all laminin components was increased in psoriatic lesions compared with healthy control skin (Fig. 1A).
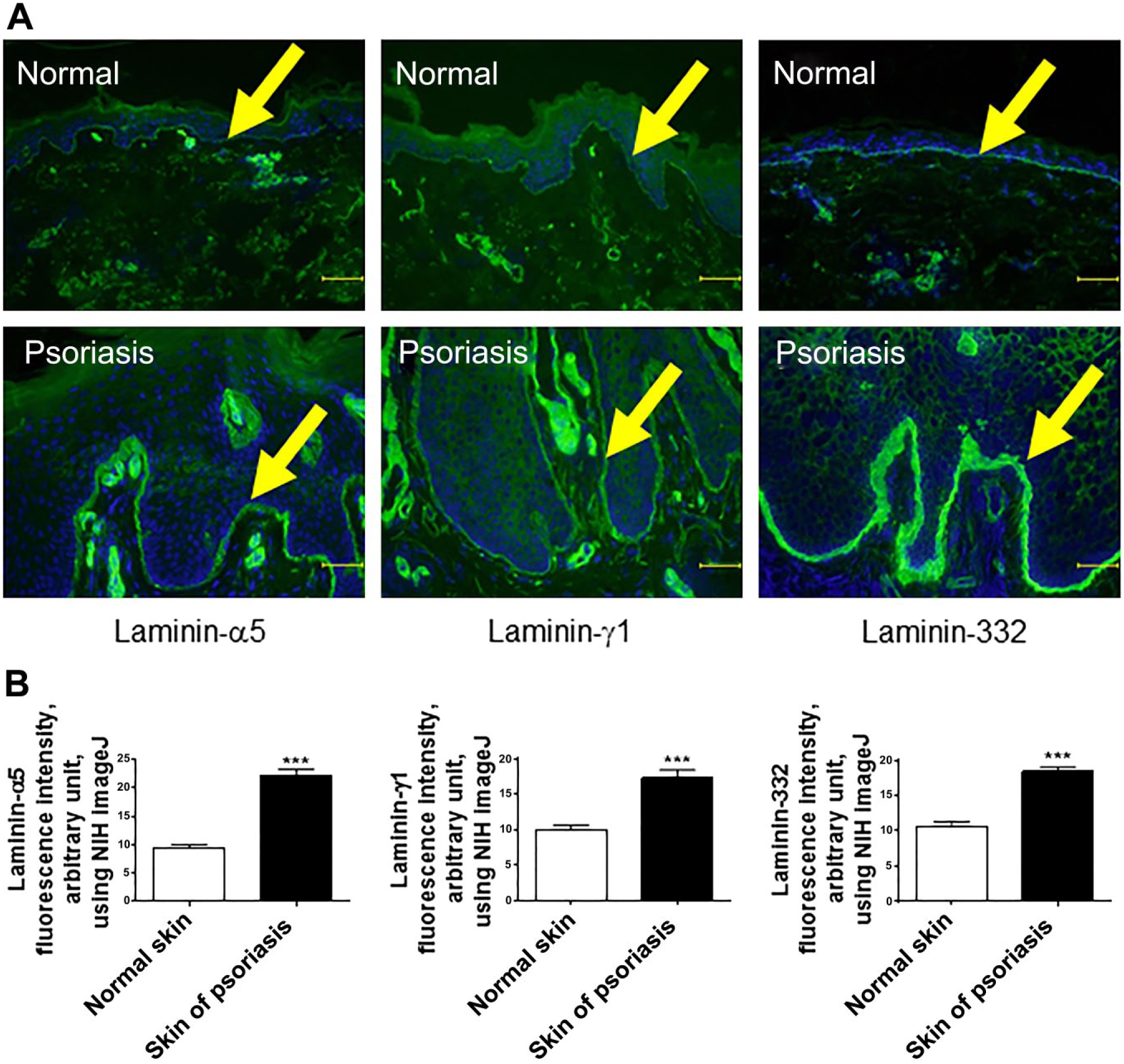
The expression of both laminin-511 and -332 is significantly increased in psoriasis in human. (A) Laminin-α5, γ1, and laminin-332 immunofluorescence of skin samples from both healthy individuals and patients with psoriasis vulgaris. The arrow indicates BM. (B) The expression of laminin-α5, γ1 chains, and laminin-332 of the BM was significantly increased in skin lesions of psoriasis compared with healthy individuals. Immunofluorescence intensity was measured by NIH ImageJ. (Mean ± SEM, n=5, ***p<0.001. Scale bar = 50 μm.) Abbreviations: BM, basement membrane; NIH, National Institutes of Health; SEM = standard error of the mean.
When the immunofluorescence intensity of laminin-332 and the components of laminin-511 within the skin specimens from the patients of psoriasis were quantitated by using NIH ImageJ, the expression of both laminin-332 and the components of laminin-511 of the BM were significantly increased in psoriatic lesions (Fig. 1B). In stark contrast to the previous findings,15,21 our quantitative data obtained from immunofluorescence study shows that main laminins along the BM within the skin, namely, laminin-332 and -511, were highly expressed in psoriatic lesional skin.
The Expression of Laminin-332 and -511 Is Also Upregulated Within the Lesional Skin of Psoriasis in Mice
Next, we wished to clarify whether similar laminin-expression abnormalities can also be seen in experimentally induced psoriatic skin lesions in a widely used mouse model of psoriasis-like dermatitis.32–34 As expected, C57BL/6 mice that had received IMQ cream on their shaved back skin for 5 consecutive days developed psoriasis-like skin lesions, 34 along with epidermal thickening, parakeratosis with neutrophils, and absence of a granular layer (Fig. 2A). Because the major laminin-binding integrins consist of β1 or β4 integrins,9,11,35 and fibronectin was reported to be involved in the development of psoriasis,18,36–38 we examined these lesions by IHC using antibodies against the main BM components, including laminin-α5, β1, γ1, laminin-332, β1 integrin, β4 integrin, and fibronectin. Similar to the findings obtained from human skin samples, we found that, compared with vehicle-treated skin, the expression of laminin-α5, β1, and γ1 chains of psoriasis-like skin lesions was significantly increased along the BM (Fig. 2B–D). β1 integrin expression was also significantly upregulated along the BM as well as on the basolateral surface of basal layer epidermal KCs (Fig. 3A). In addition, laminin-332 and β4 integrin was also significantly increased along the BM in psoriasis-like skin (Fig. 3B and C).
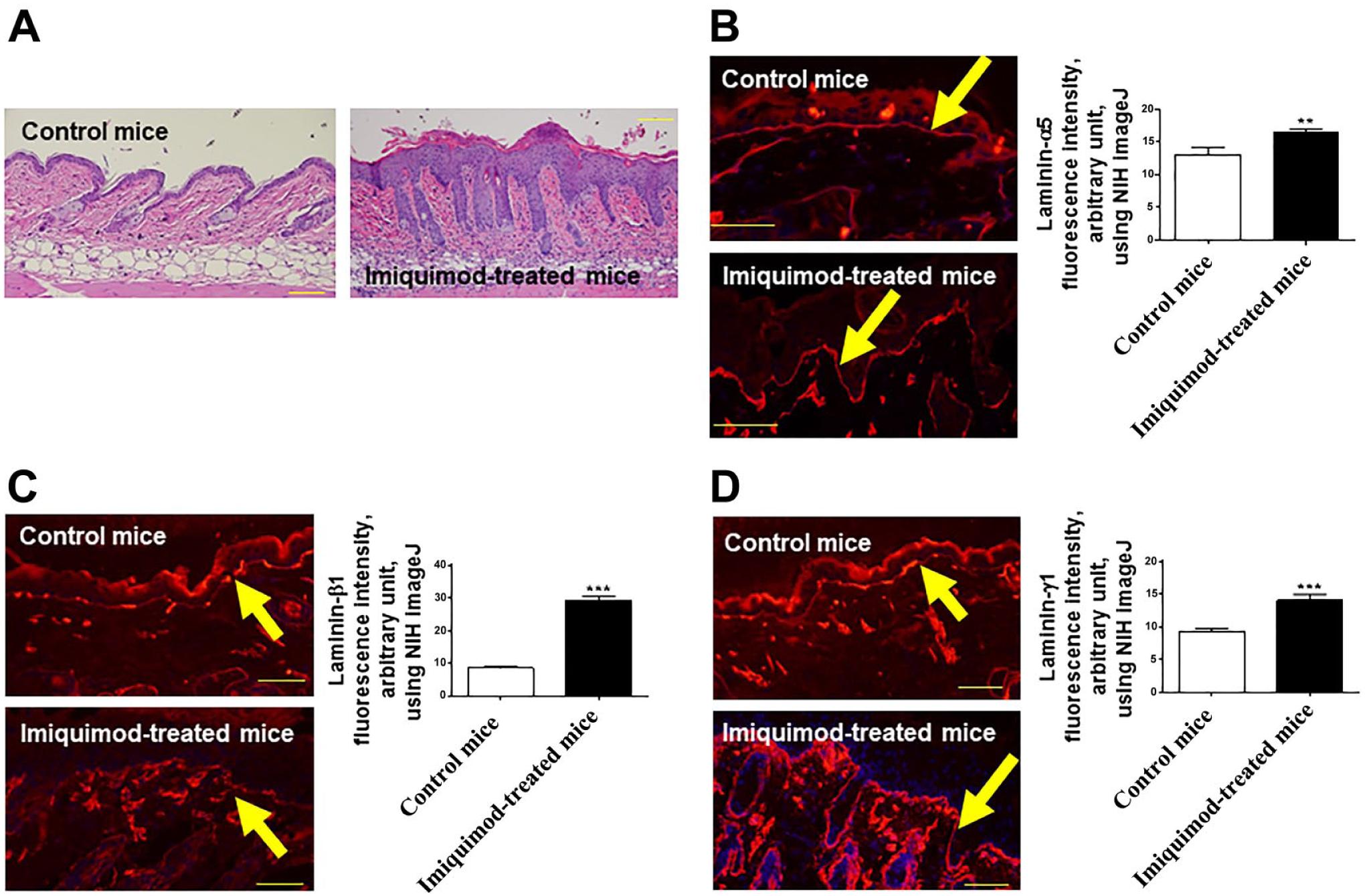
Laminin-511 expression is upregulated in psoriatic skin lesion in mice. (A) C57BL/6 mice, which received IMQ cream on the shaved back for 5 consecutive days, showed psoriasis-like skin. This model mouse showed epidermal thickening, parakeratosis with neutrophils, and absence of a granular layer. (H&E, original magnification ×100.) Immunofluorescence study of both normal and psoriatic skin samples. Compared with vehicle treated skin, the expression of laminin-α5 (B), β1 (C), and γ1 chains (D) of psoriasis-like skin lesions was significantly increased along the BM. The arrow indicates BM. (Mean ± SEM, n=5, **p<0.01. ***p<0.001.) (Scale bar A = 100 μm; scale bars B–D = 50 μm.) Abbreviations: IMQ, Imiquimod; BM, basement membrane; NIH, National Institutes of Health; SEM = standard error of the mean.
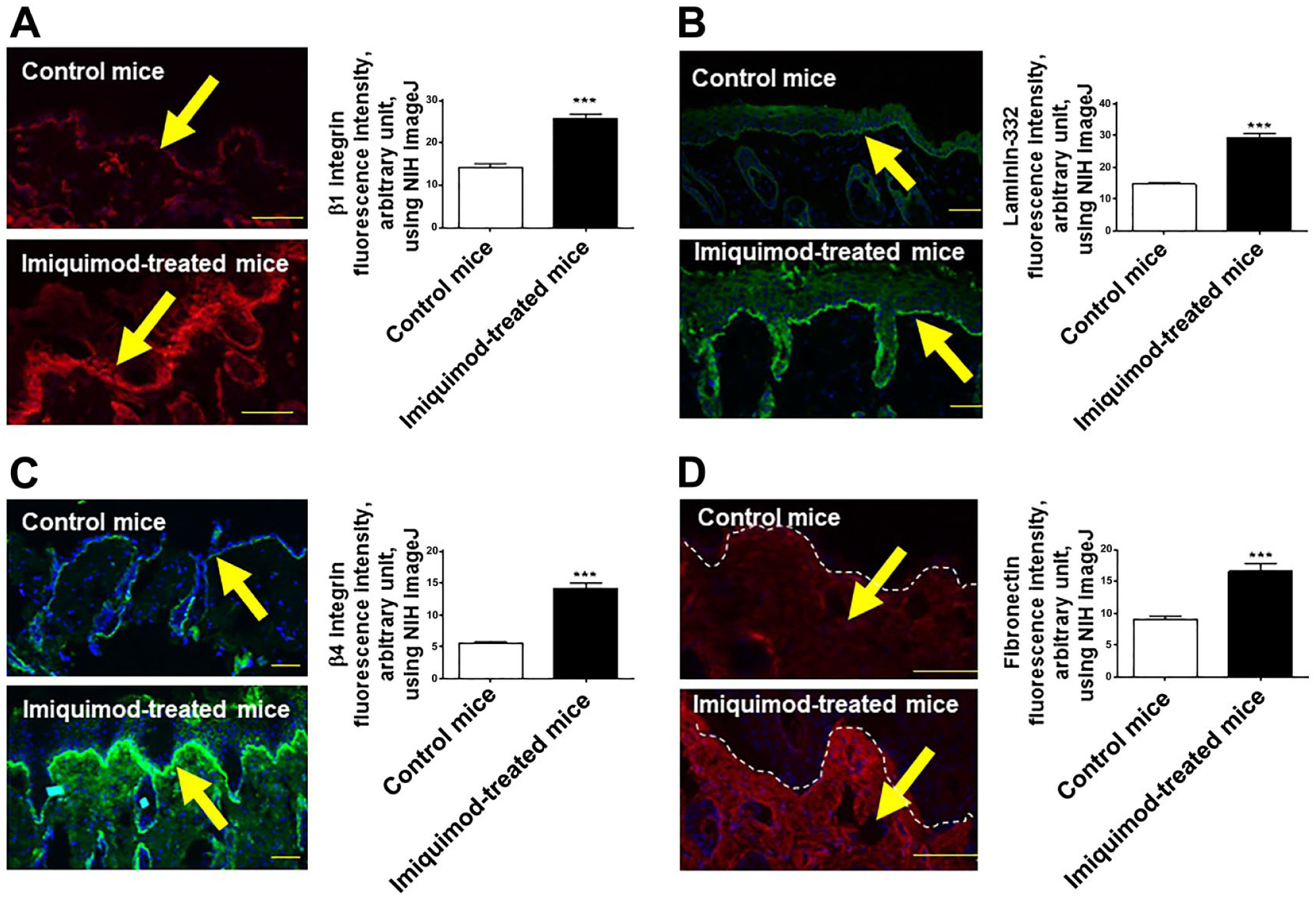
The expression of other BM components is also upregulated in psoriasis model mice. Immunofluorescence study of normal and psoriatic skin sections in mice. Compared with vehicle treated skin, β1 integrin expression was also significantly upregulated along the BM as well as basolateral surface of basal keratinocytes (A). The expression of laminin-332 and β4 integrin was also significantly increased along the BM in psoriasis-like skin (B, C). Fibronectin was significantly upregulated within the dermis of these mice (D). The arrow indicates BM, while yellow arrows in (D) indicate fibronectin positive immunoreactivity. (Mean ± SEM, n=5, ***p<0.001. Scale bars = 50 μm.) Abbreviations: BM, basement membrane; NIH, National Institutes of Health; SEM = standard error of the mean.
Furthermore, as previously reported in human, 22 fibronectin was also significantly upregulated within the dermis of these mice (Fig. 3D), and the positive immunoreactivity seemed to be detected predominantly in the papillary dermis. In line with the observations obtained by human psoriatic skin samples (Fig. 1A and B), the expression of both laminin-332 and -511 as well as their binding partners, integrin β1 and β4, were significantly increased in psoriatic lesions of the mouse model of psoriasis-like dermatitis. McFadden et al. have previously proposed a laminin-dependent mechanism by which self-limiting guttate psoriasis caused by streptococcal pharyngitis develops to chronic psoriasis. Streptokinase, which is produced by streptococci, can initiate lysis and disruption of laminin. Next, to investigate whether the disruption of laminin-511 and -332 is induced during the early period of psoriasis, we evaluated the expression of laminin-α5 and γ1 chains as well as laminin-332 of the IMQ-treated mice after the drug application for 1 or 2 consecutive days. As a result, the disruption of laminin-α5 and γ1 chains as well as laminin-332 was not detected (Fig. 4).
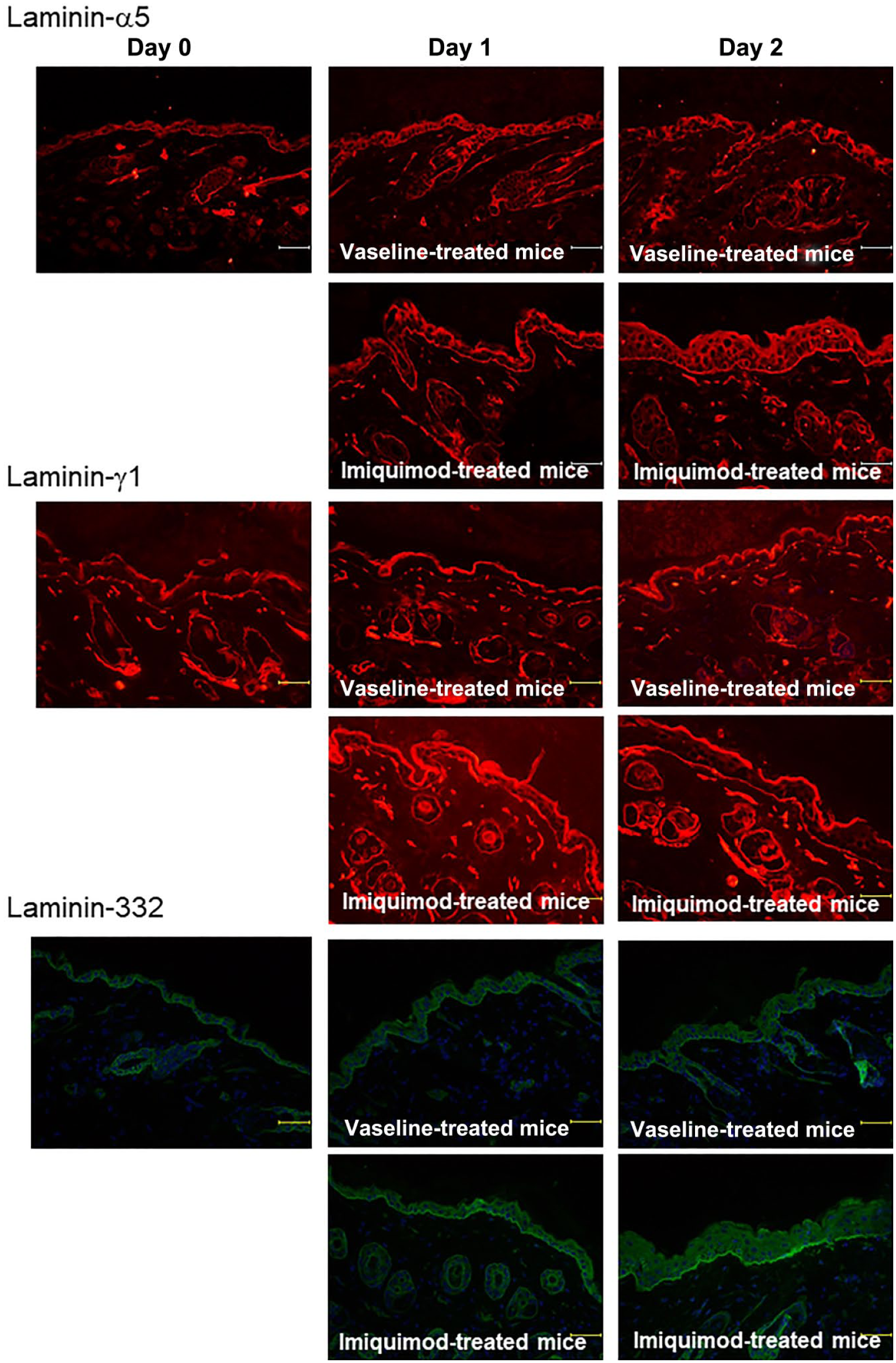
The disruption of laminin-α5, γ1 chains, and laminin-332 is not detected during the early phase of psoriasis. The disruption of laminin-α5 and γ1 chains as well as laminin-332 of the Imiquimod-treated mice after the drug application for 1 or 2 consecutive days was not detected. (Scale bars = 50 μm.)
Ultrastructural Evaluation by TEM
Fleischmajer et al. reported that the lamina densa showed gaps, splitting, or reduplication in involved skin of psoriasis in human. 15 In addition, both laminin-332 and β4 integrin are crucial components of hemidesmosomes.9,11,12,28,39 We next investigated the ultrastructure of BM of the mouse model of psoriasis-like dermatitis.
TEM showed that the number of hemidesmosomes was not affected in psoriasis-like skin lesions compared with vehicle control (Fig. 5A and B). However, the thickness of lamina densa was significantly increased in psoriatic lesions (Fig. 5A and B). In addition, we could detect a few gaps of lamina densa in psoriasis-like skin lesions (Fig. 5C). Interestingly, there are abundant reticular, string-like structures attached to lamina densa of psoriatic lesions (Fig. 5A). When we carefully investigated fibroblasts, numerous string-like structures attached to dermal fibroblast were detected in psoriasis model mouse samples (Fig. 5D). Taken together with fibronectin immunofluorescence study (Fig. 3D), production of fibronectin seems to be increased by the fibroblasts of psoriasis.
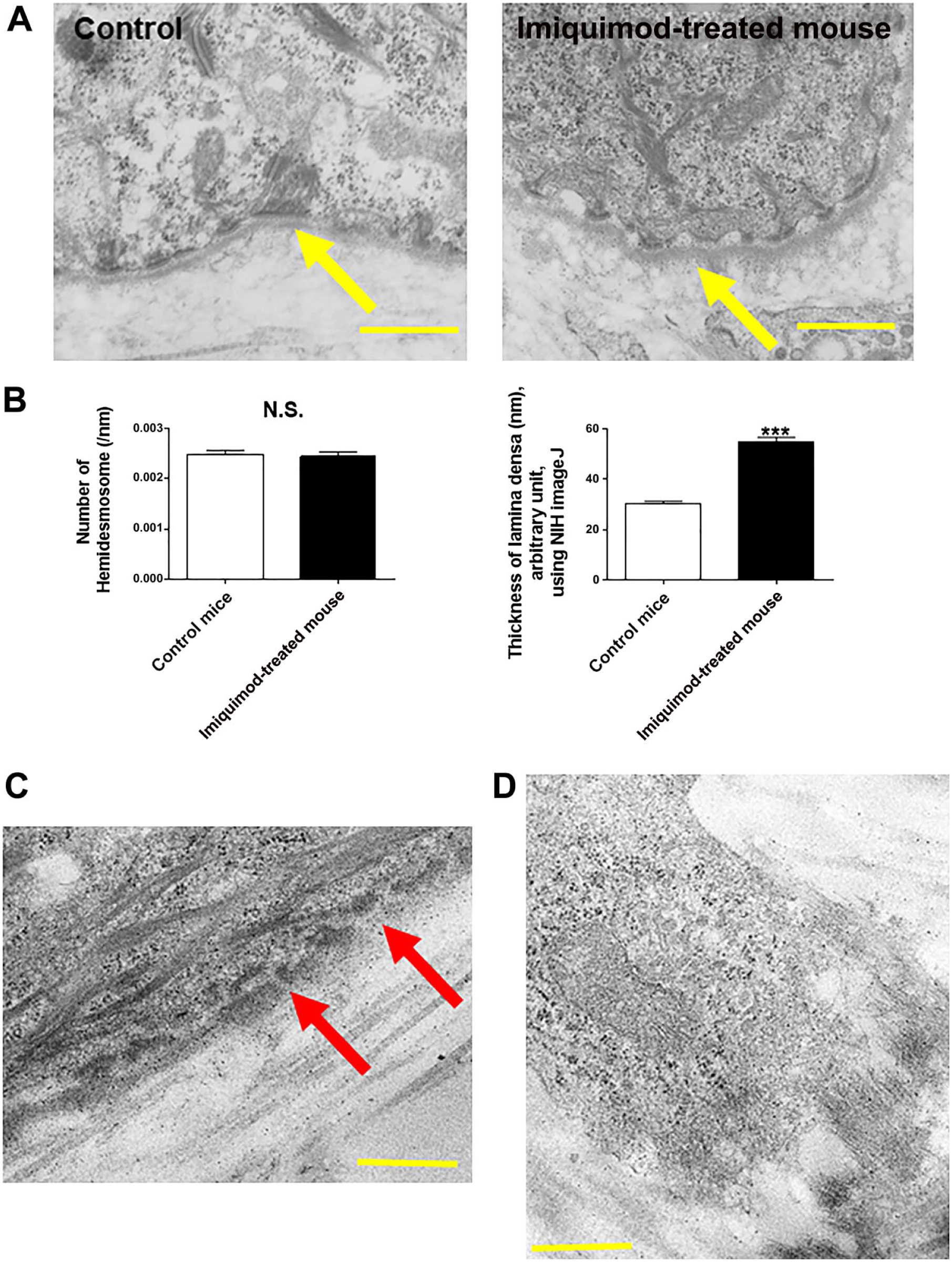
The number of hemidesmosomes is not changed in psoriasis model mice. (A) By using TEM, the number of hemidesmosomes was not affected in psoriasis-like skin lesions compared with vehicle control. The arrow indicates lamina densa. (B) The thickness of lamina densa was significantly increased in mouse model of psoriasis. (Mean ± SEM, n=5, ***p<0.001.) (C) A few gaps of lamina densa in psoriasis-like skin lesions were seen. (D) Numerous string-like structures attached to dermal fibroblast were detected within the lesional skin samples of psoriasis model mouse. (Scale bars = 500 nm.) Abbreviations: TEM, transmission electron microscopy; N.S., not significant; NIH, National Institutes of Health; SEM = standard error of the mean.
Type IV Collagen Expression Is Significantly Upregulated Within the Lesional Skin of Psoriasis in Mice
Since the main component of lamina densa is considered to be type IV collagen,40,41 we next investigated the expression of type IV collagen of psoriasis model mouse. As a result, type IV collagen expression within the BM was significantly increased in lesional skin of psoriasis compared with the control mice (Fig. 6A and B). Interestingly, there were a few gaps of type IV collagen within the BM (Fig. 6A).
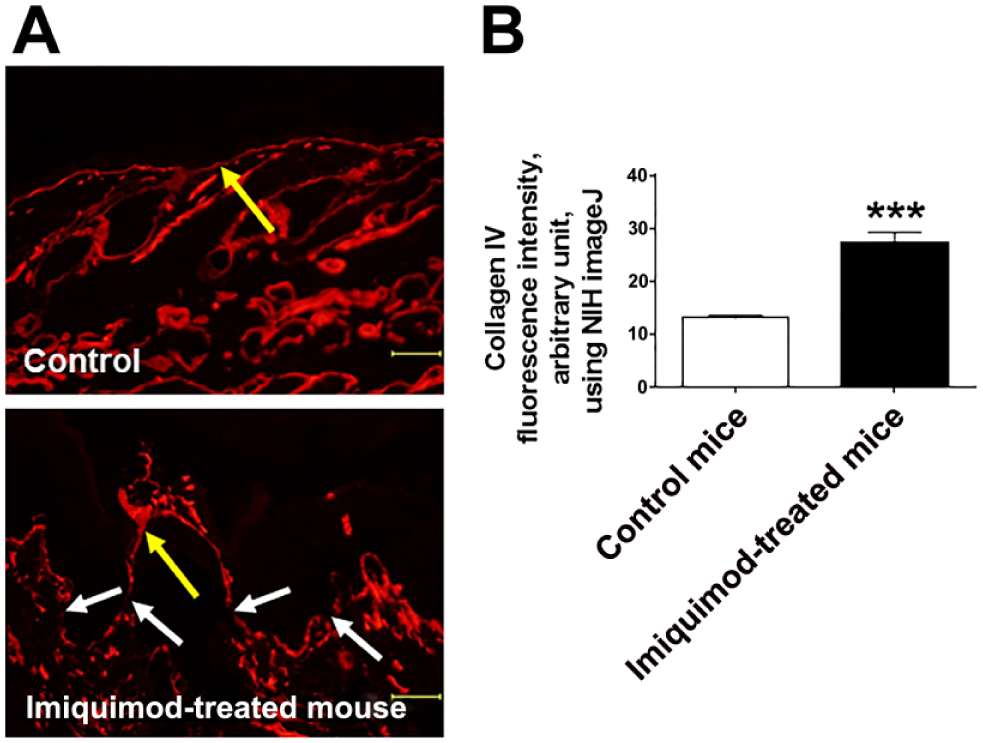
Type IV collagen expression is upregulated in psoriatic skin lesion in mice. (A, B) Immunofluorescence study of both vehicle-treated control and psoriatic skin samples. Compared with the vehicle-treated skin, the expression of type IV collagen of psoriasis-like skin lesions was significantly increased along the BM. There were a few gaps of type IV collagen within the BM (the white arrow). The yellow arrow indicates BM. (Mean ± SEM, n=5, ***p<0.001. Scale bars = 50 μm.) Abbreviations: BM, basement membrane; N.S., not significant; NIH, National Institutes of Health; SEM = standard error of the mean.
Laminin-511 Inhibits Apoptosis of HaCaT Cells
Human placental laminin-511/521 is reported to increase the human KCs number. 42 To investigate the effect of laminin-511 but not laminin-521 on KC proliferation/apoptosis, we next performed HaCaT cells culture with various concentrations of recombinant laminin-511. HaCaT cells are immortalized human KCs, share several characteristics with psoriatic KCs, and are often used as a cellular model of psoriasis.43–50
As a result, laminin-511 at 1 μg/ml significantly increased HaCaT cells proliferation compared with the control evaluated by Ki-67 immunofluorescence (Fig. 7A). This suggests that laminin-511 plays a role as a KC growth stimulator, which is similar to our previous findings by human HF organ culture. 28 Furthermore, laminin-511 treatment at 0.1 μg/ml and 10 μg/ml significantly decreased HaCaT cells apoptosis (assessed by cleaved caspase-3 immunofluorescence; Fig. 7B). As HaCaT cells are reported to constitutively express laminin-α5, β1, and γ1 chains, 51 this suggests that laminin-511 may be important for KC survival and in the pathobiology of psoriasis.
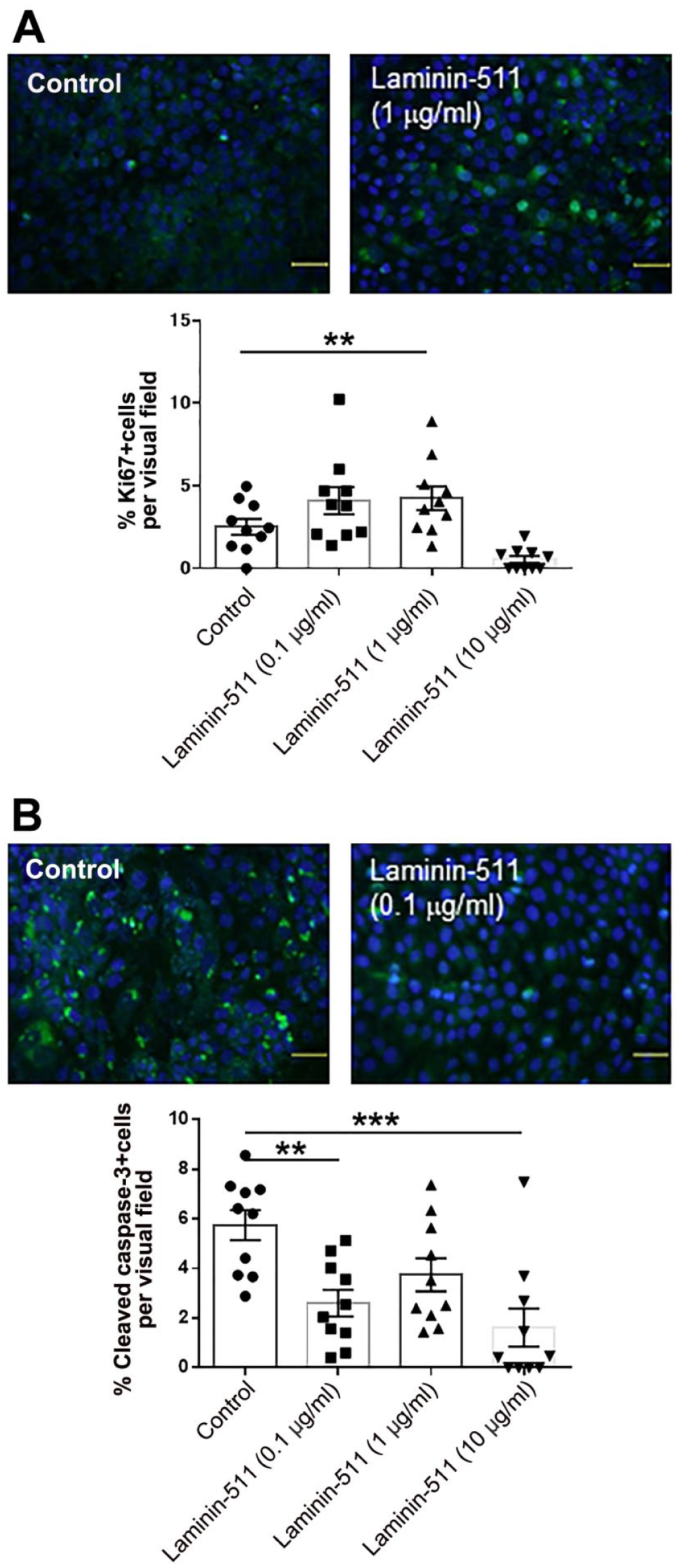
Laminin-511 stimulates keratinocytes proliferation and inhibits their apoptosis. (A) (Upper images) Representative images of Ki-67 immunofluorescence. Green indicates Ki-67 positive immunoreactivity. (Lower graph) Laminin-511 at 1 μg/ml significantly increased HaCaT cells proliferation compared with the control evaluated by Ki-67 immunofluorescence. (B) (Upper images) Representative images of cleaved caspase-3 immunofluorescence. Green indicates cleaved caspase-3 positive immunoreactivity. (Lower graph) Laminin-511 at 0.1 μg/ml and 10 μg/ml significantly decreased apoptosis assessed by cleaved caspase-3 immunofluorescence. (**p<0.01. ***p<0.001. One-way ANOVA, Bonferroni’s Comparison Test.)
Laminin-α5 Chain Gene Knockdown in HaCaT Cells Decreases Proliferation and Induces Apoptosis
As shown above, laminin-511 treatment did not only stimulate KCs proliferation but also inhibited their apoptosis (Fig. 7A and B). This suggested that laminin-511 plays important roles in maintaining KCs homeostasis.
Therefore, to next investigate the effect of laminin-511 blockade on epidermal KCs, we performed laminin-α5 gene knockdown in HaCaT cells using siRNA. Successful laminin-α5 gene knockdown was demonstrated by immunofluorescence microscopy, which showed significantly decreased laminin-α5 protein expression to control cells treated with scrambled siRNA (SCR; Fig. 8A).
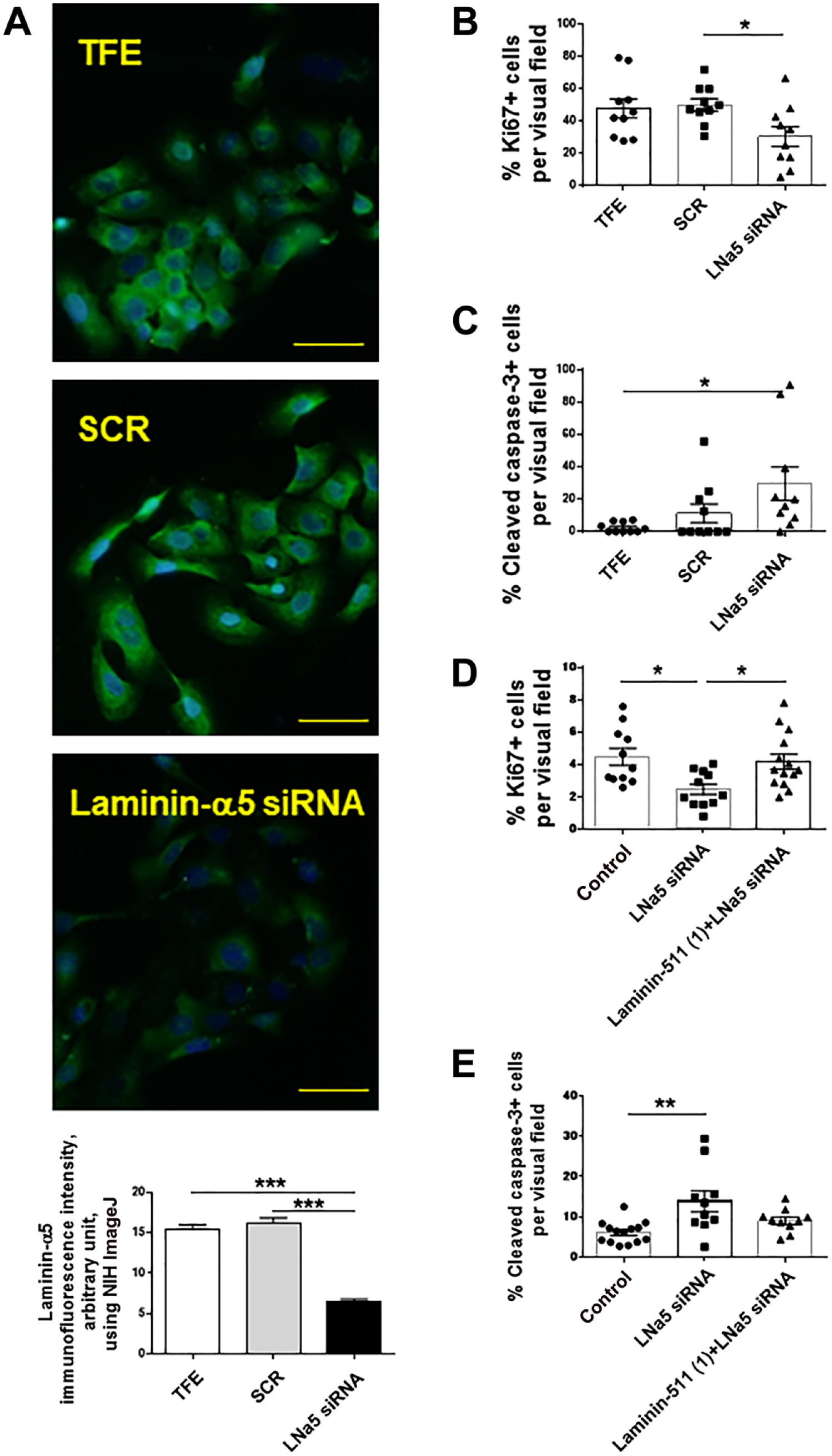
The gene knockdown of laminin-α5 chain of laminin-511 decreases keratinocytes proliferation and increases their apoptosis. (A) Successful laminin-α5 gene knockdown was assessed by laminin-α5 immunofluorescence. (B, C) Laminin-α5 gene knockdown significantly decreased HaCaT cell proliferation (evaluated by Ki-67 immunofluorescence), whereas it increased apoptosis (evaluated by cleaved caspase-3 immunofluorescence). (D, E) The decreased proliferation of HaCaT cells by laminin-α5 gene knockdown was recovered by laminin-511 treatment (at 1 μg/ml), whereas the significantly increased apoptosis by laminin-α5 gene knockdown was partially diminished by the co-administration of laminin-511. (*p<0.05. **p<0.01. ***p<0.001. One-way ANOVA, Bonferroni’s Comparison Test. Scale bars = 50 μm.) Abbreviations: TFE, transfection reagent; NIH, National Institutes of Health; SCR, scrambled siRNA.
Importantly, laminin-α5 gene knockdown in HaCaT cells significantly decreased their proliferation as measured by Ki-67 immunofluorescence (Fig. 8B), whereas it significantly increased HaCaT cell apoptosis (Fig. 8C).
Furthermore, additional HaCaT cell cultures showed that the decreased proliferation of HaCaT cells by laminin-α5 gene knockdown could be recovered by co-administration of laminin-511 (Fig. 8D). In addition, significantly increased apoptosis by laminin-α5 gene knockdown was partially diminished by co-administration of laminin-511 (Fig. 8E).
Taken together with the above findings in human and murine psoriatic skin, these functional cell culture experiments in a human KC cell line that shares some features with psoriatic epidermal KCs are consistent with the concept that laminin-511 may be important for maintaining KC hyperproliferation and reduced apoptosis in psoriatic epidermal lesions.
Discussion
In this study, we showed that (1) the expression of laminin-511 and -332 was significantly upregulated along the BM within the lesional skin of psoriasis vulgaris in human; (2) the expression of BM components, including laminin-α5, β1, γ1, laminin-332, β1 integrin and β4 integrin, as well as fibronectin, within the lesional skin of mouse model of psoriasis were also significantly increased along the BM by immunofluorescence study; (3) although the number of hemidesmosomes was not affected in psoriasis-like skin lesions compared with vehicle control, the thickness of lamina densa was significantly increased, and there were a few gaps detected in the lamina densa; (4) laminin-511 significantly increases surrogate “psoriatic” KCs, 50 HaCaT cells proliferation, and inhibits their apoptosis; (5) laminin-α5 chain gene knockdown in HaCaT cells decreases their proliferation and induces apoptosis; (6) laminin-511 treatment diminished the effect of laminin-α5 chain gene knockdown on HaCaT cells proliferation and apoptosis.
McFadden et al. hypothesized that basal KCs can interact with fibronectin via α5β1 integrin due to the disruption of laminins, and, therefore, the proliferation of these KCs may be stimulated.18,20–23,52 This seems to contradict our results on the expression of laminin-332 and -511. However, they focused on laminins composed of laminin α1 or α2 chains.18,20–23,52 In addition, in contrast to the concepts by McFadden et al.,18,20–23,52 Toti et al. reported that the expression of laminin-111 (composed of α1, β1, and γ1 chains) was normal, with intense and continuous reaction in human psoriatic skin. 23
Although both laminin α1 and α2 chains are expressed in skin,53,54 main laminins within the skin are laminin-332 and -511. 9 Our results showed that the expression of laminin-511 was significantly increased in lesional skin in mouse model of psoriasis (Fig. 2B–D) and of psoriasis patients (Fig. 1A and B). We also considered the possibility that the disruption of laminin-511 and -332 could be induced during the early phase of psoriasis. However, we could not find any obvious results that indicate the disruption of these laminins (day 0 to 2 after IMQ application; Fig. 4). It is still possible that the disruption of these laminins is induced during the very early phase after the IMQ application (e.g., 4 hr). This needs to be further investigated. In contrast to our findings, Fleischmajer et al. reported that the expression of laminin including laminin-α5, β1, and γ1 chains was reduced in psoriatic skin. 15 It is very difficult to explain the discrepancy of laminin-511 expression pattern in skin lesion of psoriasis. However, Fleischmajer et al. included and evaluated the patients treated with ultraviolet B (47%), psolaren plus ultraviolet A (11%). 15 It is possible that the treatment altered these laminin expressions. For instance, the reduced laminin expression (α2 chain) in psoriatic skin was reported to be recovered after the treatment by cyclosporine. 23 Furthermore, Rouabhia et al. reported that the ultraviolet (UV) irradiation on engineered human skin showed a complete absence of laminin deposition. 55 There is a possibility that the treatment for psoriasis, including UV irradiation, affects laminin expression.
Fleischmajer et al. also reported that there are gaps along the lamina densa of psoriatic skin detected by TEM. 15 Similar phenomena has been reported by others.56–58 Interestingly, Madalene et al. reported that basal KC herniation through the gap of lamina densa seems to correlate with disease activity. 58 Although we could not find obvious basal KC herniation, there are several gaps of lamina densa detected in psoriasis model mouse (Fig. 5C). Lamina densa is mainly composed of laminins and collagen type IV.8,41 However, we could not detect any disruption of laminin-511 and -332. Taken together with the results of collagen type IV immunofluorescence study (Fig. 6A), the gaps along the lamina densa seem to be related to the disruption of collagen type IV. We also found that the thickness of lamina densa was significantly increased in psoriatic lesional skin in mice (Fig. 5A and B). This seems to be related to the increased expression of collagen type IV along the BM (Fig. 6A and B). It is possible that collagen type IV expression might be stimulated to compensate the disruption of its expression along the BM (Fig. 6A). Interestingly, laminin-332 is reported to accelerate the formation of lamina densa. 59 In addition, β3 chain short arm of laminin-332 is reported to induce laminin-511 matrix assembly. 60 The upregulated laminin-332 or -511 expression in psoriatic skin (Figs. 2B–D and 3B) could be related to the thickened lamina densa (Fig. 5A and B). Because the number of hemidesmosomes was not affected in psoriatic lesional skin in mice (Fig. 5A and B), the thickened lamina densa might be not only induced by upregulated collagen type IV, but also by the upregulated laminin-511 expression. It is possible that the upregulation of laminin-511 is induced by the disruption of collagen type IV to compensate the gaps of lamina densa. Further investigation on whether the upregulated laminin-511 expression is related to increased expression of β3 chain of laminin-332 or disruption of collagen type IV would be needed as a future study.
Interestingly, psoriasis shares many features, including antimicrobial peptides expression and several KC markers expression, with wound healing. 61 Importantly, laminin-511/521 is shown to stimulate KC migration in an in vitro wound healing assay, 42 and this suggests that laminin-511/521 may promote wound healing. 62 It is also known that both fibronectin and laminin-332 are upregulated during wounding. 63 This “psoriasis-wound healing” concept supports our findings of upregulated laminin-332 and -511 expression in psoriatic skin both in human and mice (Figs. 1A and B, 2B–D and 3B).
Our HaCaT cell culture showed that laminin-511 blockade by laminin-α5 chain gene knockdown decreased the proliferation of normal HaCaT cells (Fig. 8B) as well as stimulated HaCaT cells by laminin-511 treatment (Fig. 8D). Taken together with our immunofluorescence results of laminin-511 (Fig. 2B–D), agents that may inhibit the effect of laminin-511 could be useful for the treatment for psoriasis.
For instance, laminin-511 has epidermal growth factor (EGF)-like repeats within the α5 chain. 26 The EGF-like repeats of laminin and tenascin-C are reported to directly trigger epidermal growth factor receptor (EGFR) signaling. 64 There is an interesting case report on EGFR and psoriasis. A patient with advanced colon cancer and concomitant long-standing psoriasis vulgaris who received anti-EGFR antibody monotherapy as a third-line treatment for metastatic disease showed dramatical improvement of psoriatic skin lesions 1 week after the initiation of treatment. 65 It is possible that anti-EGFR antibody could not only block the binding of endogenous EGF to its receptor but also reduce the effect of EGF-like repeats of laminin-511. This may suggest that the blockade of laminin-511 can be considered as an optional antipsoriasis therapy.
In conclusion, the phenomenological observations reported here raise the question whether laminin-511-controlled KC growth/death may be a previously overlooked player in the pathogenesis of psoriatic epidermal lesions.
Footnotes
Acknowledgements
The excellent technical support of Ms Ayano Yonamine is gratefully appreciated. We thank Dr Jeffrey Miner for providing an antibody against murine laminin α3 chain. We also thank Prof Jonathan C.R. Jones for J18.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AN, KS, MY, and YM performed all of the experiments. AN, KS, DT, and RP assisted in experimental design, data interpretation, and manuscript writing. SS and AM provided human skin samples and assisted in manuscript writing. All authors have read and approved the manuscript as submitted.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the ethics committee of Osaka City University Graduate School of Medicine, Osaka, Japan (2016-3490).
