Abstract
Cysteine sulfinate decarboxylase (CSD) is the rate-limiting biosynthetic enzyme of the pathway that forms taurine, a putative osmolyte in the kidney, which was previously localized in various segments of the nephron. Although CSD is known to be expressed in whole kidney extracts, no information on CSD mRNA regional expression and histological localization is yet available. Western blotting and Northern blotting were performed in four dissected regions of the kidney using an antiserum against recombinant CSD and a [32P]-dCTP-labeled CSD cDNA probe, respectively. In situ hybridization was carried out using a [35S]-CTP-labeled CSD RNA probe. A single protein (53 kD) and a single mRNA (2.5 kb) were detected, both of which appeared to be most enriched in the outer stripe of the outer medulla. In situ hybridization of CSD mRNA showed strong labeling of the thick tubules in the outer stripe of the outer medulla and in cortical medullary rays that corresponded to the proximal straight tubules. The significance of this restricted expression of CSD is discussed in relationship to the data previously reported on the location of taurine and the location of the taurine transporter along the nephron.
C
The cellular localization of taurine in the kidney was investigated by immunocytochemistry. The most comprehensive study (Amiry-Moghaddam et al. 1994), which included quantification at the electron microscopic level, reported that taurine was most concentrated in the cortical and medullary collecting ducts, in the proximal straight tubules, and in the descending limbs of Henle's loops. Taurine was also found to a lesser extent in the proximal convoluted tubules (Amiry-Moghaddam et al. 1994). Taurine immunolabeling was similarly reported to be present in the collecting ducts by other investigators (Trachtman et al. 1993; Ma et al. 1994), who, however, did not report it to be present in the proximal straight tubules. No taurine labeling was found in the distal straight and convoluted tubules (Trachtman et al. 1993; Amiry-Moghaddam et al. 1994; Ma et al. 1994). Taurine can be accumulated in a cell via two mechanisms: transport from the extracellular space and biosynthesis within the cell.
Taurine is taken up through a high-affinity sodium-dependent transporter. A taurine transporter has been cloned from a rat brain cDNA library and characterized by expression in mammalian cells (Smith et al. 1992). In the rat kidney, the mRNA of this transporter was located in the S3 segment of the proximal straight tubules, in the loop of Henle, and in the glomerular epithelial cell layer (Matsell et al. 1997). In the S3 segment, the gene expression of the taurine transporter appeared to be downregulated by a high-taurine diet (Matsell et al. 1997). This result is consistent with earlier findings showing that taurine transport at the level of the brush border membrane in the proximal tubules was regulated by circulating taurine (Chesney et al. 1985) and maintained the taurine body pool by regulating its renal reabsorption (Chesney 1985).
Taurine shows a broader distribution in the kidney than that of its transporter. This discrepancy may reflect the contribution of local synthesis of taurine in some segments of the nephron. In contrast to the taurine transporter, the expression and regulation of which have been well studied (Matsell et al. 1997) as noted above, very little information is available concerning the biosynthesis of taurine in the kidney. CSD activity (Jacobsen et al. 1964; Jerkins and Steele 1991) and CSD mRNA (Kaisaki et al. 1995; Tappaz et al. 1998) were measured in whole kidney extracts. This expression of CSD indicates that taurine can be synthesized from cysteine through the cysteine sulfinate pathway in the kidney, because CSD is an enzyme of this pathway (Jacobsen and Smith 1968). However, no information is available about regional tissue distribution or cellular localization of CSD. We have investigated the distribution of CSD by immunoblotting and CSD mRNA by Northern blotting in the four main regions of rat kidney. We also report the histological localization of CSD mRNA along the nephron by in situ hybridization. Molecular cloning of the CSD cDNA that we achieved from a rat liver cDNA library (Reymond et al. 1996) provided the analytical tools used in this study.
Materials and Methods
Animals and Tissue Preparation
Male adult Sprague–Dawley rats (200–250 g body weight) were used (Iffa Credo; Lyon, France). Animals were maintained under controlled temperature, humidity, and light/dark cycle for an adaptation period of 2 days. Commercial food (Souffirat; Jenthon, France) and tapwater were supplied ad libitum. The animals were anesthetized with chloral hydrate (400 mg/kg body weight) and sacrificed by decapitation. Kidneys were rapidly removed. A sagittal section about 1 mm thick was cut with a razor blade and the different regions of the kidney [superficial cortex (called “cortex” in the text below), outer stripe of the outer medulla, inner stripe of the outer medulla and inner medulla] were dissected out under a microscope and immediately frozen in liquid nitrogen. For in situ hybridization the kidneys were rapidly removed and frozen in isopentane at −70C.
CSD Antiserum
A recombinant fusion protein glutathione-S-transferase–CSD (GST–CSD) was produced, isolated by affinity chromatography on glutathione-agarose beads, and cleaved by thrombin protease as previously reported (Tappaz et al. 1999). The recombinant CSD was separated from GST by preparative SDS-PAGE, recovered by electroelution, and used as an immunogen to produce a CSD antiserum in rabbit.
Nucleotide Probes
The DNA probe used in Northern blotting was the 1.9-kb CSD cDNA cloned previously from a rat liver cDNA library (Reymond et al. 1996). It was labeled using [32P]-dCTP (3000 Ci/mmol), random primers, and DNA polymerase (Appligene Oncor; Illkirch, France). The antisense and sense probes used for in situ hybridization were [35S]-CTP-labeled riboprobes synthesized by in vitro transcription of the 1.9-kb CSD cDNA previously cloned (Reymond et al. 1996). The plasmid was linearized with the appropriate restriction enzymes and the entire sequence of the CSD cDNA was transcribed using T7 or T3 RNA polymerase to synthesize antisense or sense riboprobes. In vitro transcription reactions were carried out as described by the manufacturer (Promega; Madison, WI) using 1 μg DNA and 30 μCi of [35S]-CTP (1250 Ci/mmol). After alcohol precipitation, the transcripts were treated for 20 min with NaOH 0.1 M to obtain fragments approximately 400 nucleotides in length.
Western Blotting
The tissues, whole kidney and dissected kidney regions, were homogenized in water (10% w/v) with a polytron (Poly-block; Strasbourg, France) and ultracentrifuged at 100,000 × g for 1 hr at 4C. The proteins of the supernatant were precipitated with 5% TCA for 90 min at 4C. The pellet was solubilized in 8 M urea. Proteins (20 μg/well) were separated by electrophoresis on a 10% acrylamide/SDS gel and electrophoretically transferred to a nitrocellulose membrane (Schleicher & Schuell; Dassel, Germany) in an electroblotting apparatus (BioRad Laboratories; Hercules, CA). The blots were incubated for 45 min in PBS containing 1% fat-free dry milk and 0.1% Triton X-100. They were further incubated overnight with rabbit CSD antiserum diluted 1:10,000 in the same solution. The blots were washed in PBS containing 0.1% Triton X-100 and then incubated with anti-rabbit secondary antibodies conjugated to peroxidase (Boehringer; Mannheim, Germany) diluted 1:1000 for 2 hr. Bands were revealed using a chemiluminescence kit according to the supplier's protocol (Pierce; Rockford, IL).
Northern Blotting
Total RNA was extracted from whole kidney or main kidney regions by the RNA B method (Bioprobe Systems; Montreuil-sous-bois, France). After alcohol precipitation, the pellet was air-dried and solubilized in RNase-free water. Twenty μg of total RNA was run on a 1.2% agarose/0.2 M formaldehyde gel and transferred to a nitrocellulose membrane (Schleicher & Schuell). Prehybridization was performed at 42C for 3 hr in a mixture made of 50% deionized formamide in 5 × SSPE (50 mM, pH 7.4 sodium phosphate buffer containing 0.75 M NaCl and 5 mM EDTA), 5 × Denhardt's solution, 0.05% SDS, and 100 mg/ml denatured salmon sperm DNA. Hybridization was carried out at 42C overnight in the same mixture to which the [32P]-dCTP-labeled random-primed cDNA probe was added (about 3.106 cpm/ml). The membrane was washed at 42C as follows: (a) once in 2 × SSPE containing 0.1% SDS for 15 min; (b) once in 0.5 × SSPE containing 0.1% SDS for 15 min; and (c) twice in 0.1 × SSPE containing 0.1% SDS for 20 min. The membrane was exposed to Reflection film (NEN Life Science Products; Boston, MA) for autoradiography with an intensifying screen at −80C overnight. The membranes were treated with hot water containing 0.1% SDS to remove the CSD cDNA probe. They were then hybridized with a [32P]-dCTP-labeled random-primed 1-kb cDNA probe of a housekeeping gene (GAPDH) (Clontech; Heidelberg, Germany).
In Situ Hybridization
Cryostat sections (16 μm) were collected on silanated RNase-free slides. They were dipped in acetone for 5 min at 4C and air-dried. Fixation was performed with 4% paraformaldehyde in 100 mM phosphate buffer (PB), pH 7.4, for 15 min at 4C. The sections were acetylated in 0.1 M triethanolamine-HCl buffer (pH 8) containing 0.25% acetic anhydride for 10 min at room temperature and then treated with 1 × SSC containing 50% formamide for 10 min at 55C. After alcohol dehydration and air-drying, each slide was hybridized overnight at 52C in 50 μl of hybrization solution made of 20 mM Tris-HCl, pH 6.8, containing 50% formamide, 10% dextran sulfate, 0.3 M NaCl, 1 mM Na2HPO4, 5 mM EDTA, 1 × Denhardt's solution, 0.01 M DTT, 0.5 mg/ml yeast tRNA, and the labeled probe (about 3.106cpm). The sections were rinsed in 1 × SSC containing 50% formamide for 30 min at 55C, 2 × SSC for 10 min at room temperature, and then incubated with 20 μg/ml RNase A in 0.01 M Tris-HCl, pH 7.5, containing 0.5 M NaCl and 5 mM EDTA for 20 min at 37C. They were washed twice in 1 × SSC containing 50% formamide for 1 hr at 55C, rinsed with 0.1 × SSC for 10 min at 55C, dehydrated in alcohol, and air-dried. Slides were initially exposed to X-ray films for 2–3 days to provide an indication of the intensity of the hybridization signal. They were then coated with Ilford Nuclear Research emulsion (Ilford, UK) diluted 1:1 with water. Sections were exposed for 2–3 weeks at 4C in sealed dark boxes. They were developed in Kodak D19, fixed in Ilford Hypam, and washed abundantly. They were counterstained with toluidine blue for histological identification.
Results
By Western blotting, the CSD antiserum labeled a single band of 53 kD in the soluble fraction of homogenates of each of the four main dissected regions of the rat kidney (Figure 1A). The molecular weight of this labeled protein subunit is identical to that of the recombinant CSD produced in bacteria. The labeling of this 53-kD band was most intense in the outer stripe. By Northern blotting, the CSD cDNA probe recognized a band of 2.5 kb in the total kidney, in the outer stripe, and in the inner stripe (Figure 1B). It was not detectable in the inner medulla and was barely visible in the cortex. In the latter extract, the signal appeared underestimated because of some degradation of the RNA revealed by the reduced level of GAPDH mRNA. The CSD mRNA was much more overexpressed in the outer stripe.
Histological localization of CSD and CSD mRNA was then performed. Although the antiserum we produced gave a reliable detection of a single protein on Western blots of kidney extracts, no satisfactory immunolabeling could be achieved on cryostat sections of paraformaldehyde-fixed tissue with this antiserum. Apparently this antiserum recognized the native form of the protein very poorly. This may be due to the fact that the antiserum was produced using as antigen the denatured form of the recombinant protein isolated after SDS-PAGE. Histological localization of the CSD mRNA along the nephron was then achieved through in situ hybridization. On film autoradiography of sections incubated with the antisense riboprobe, strong labeling was observed in the outer stripe, from which characteristic labeled indentations penetrated the deeper part of the cortex (Figure 2A). In the inner medulla no labeling was detectable, but a very faint darkening of the film could be distinguished over the inner stripe. No labeling was obtained when the tissue sections were incubated with the sense probe (Figure 2B). On emulsion-dipped tissue sections conterstained with toluidine blue, an intense signal was observed in the tubules located in the outer stripe, which abruptly stopped at the limit between the outer stripe and the inner stripe (Figures 3A and 3B). A strong labeling was present over the tubules of the cortical medullary rays. No labeling was detected in the inner medulla. The glomeruli were devoid of labeling. Faint but detectable labeling was visible over the structures of the inner medulla and the cortical labyrinths between the medullary rays. At higher magnification, the strong labeling in the outer stripe was localized over many slightly sinuous, thick-walled tubules, around 30 μm in diameter, whereas thinner tubules (<10 μm in diameter) were not labeled (Figures 3C and 3D).
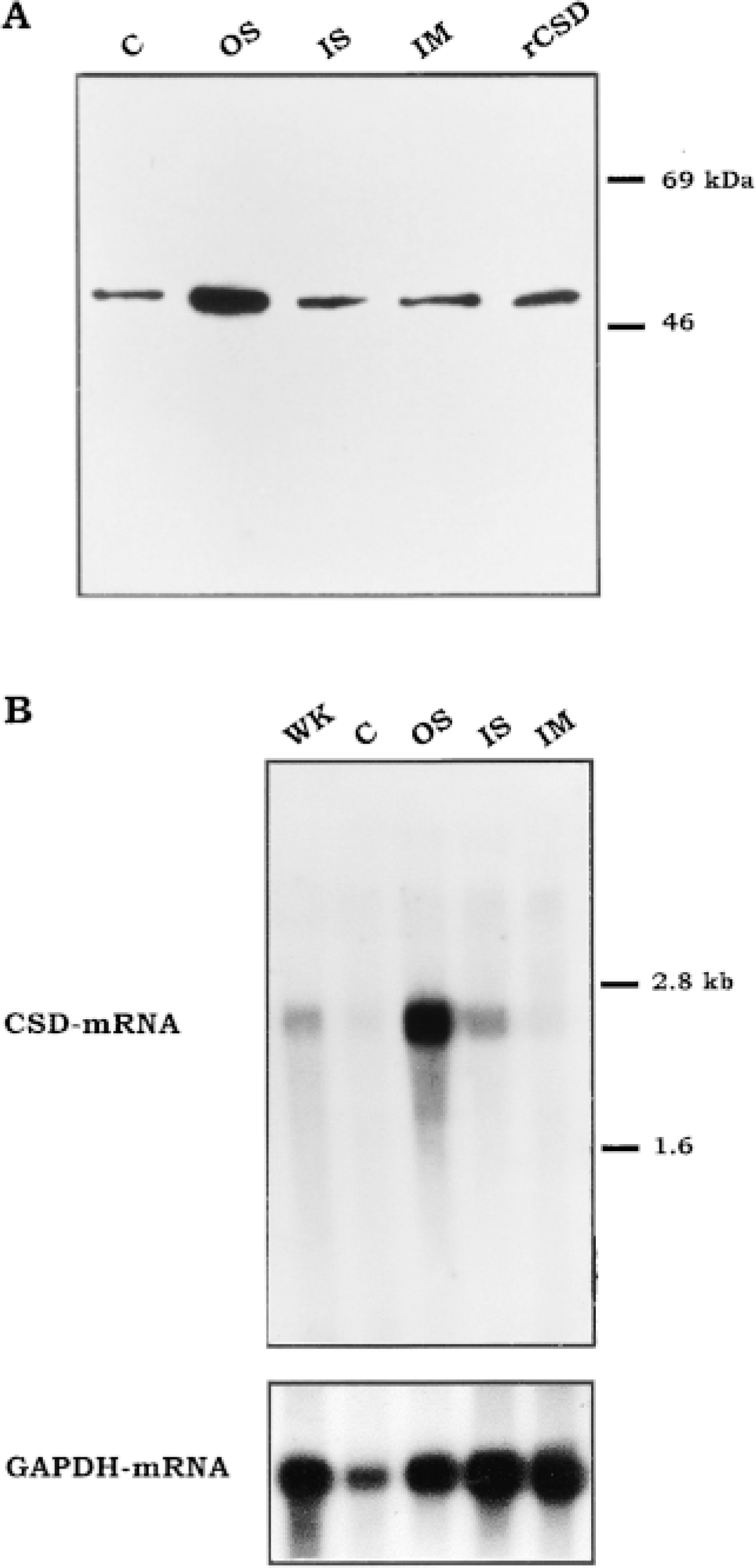
CSD and CSD mRNA distribution in different regions of the rat kidney. (
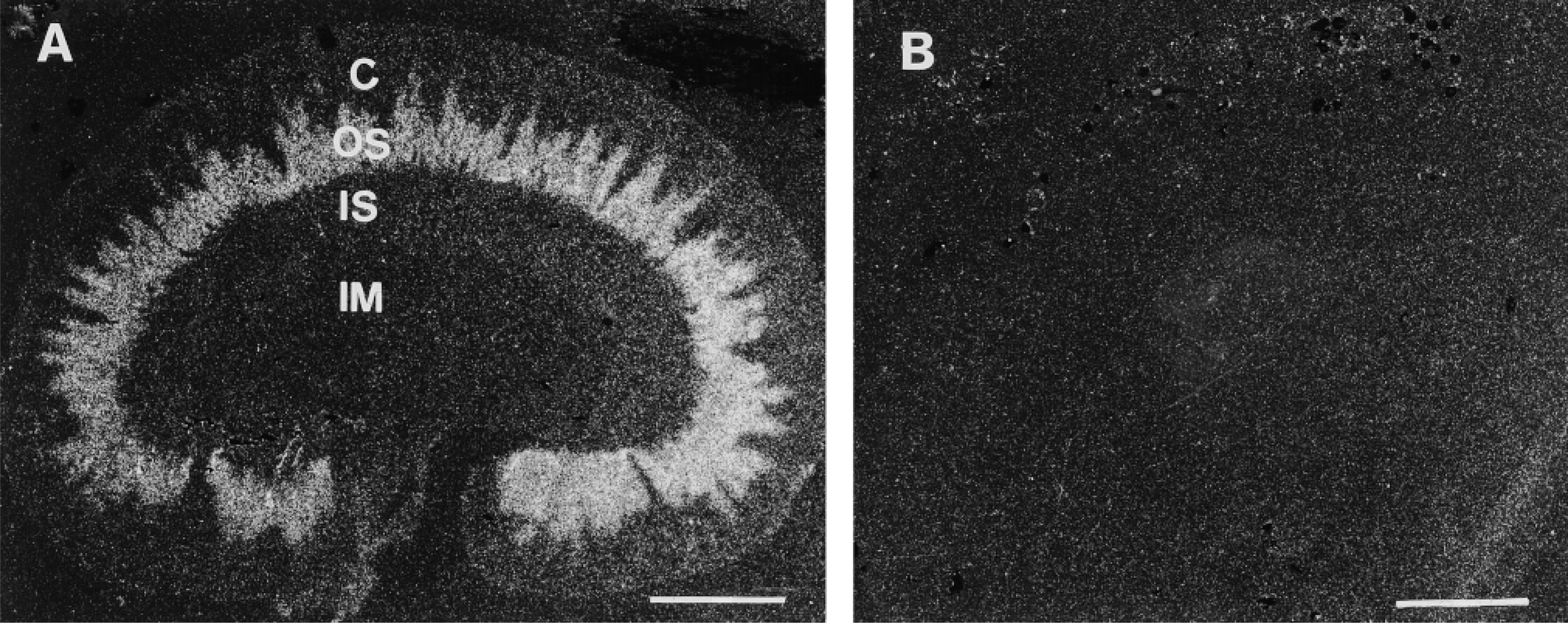
Localization of CSD mRNA in rat kidney by in situ hybridization. Film autoradiograph of sagittal cryostat sections. (
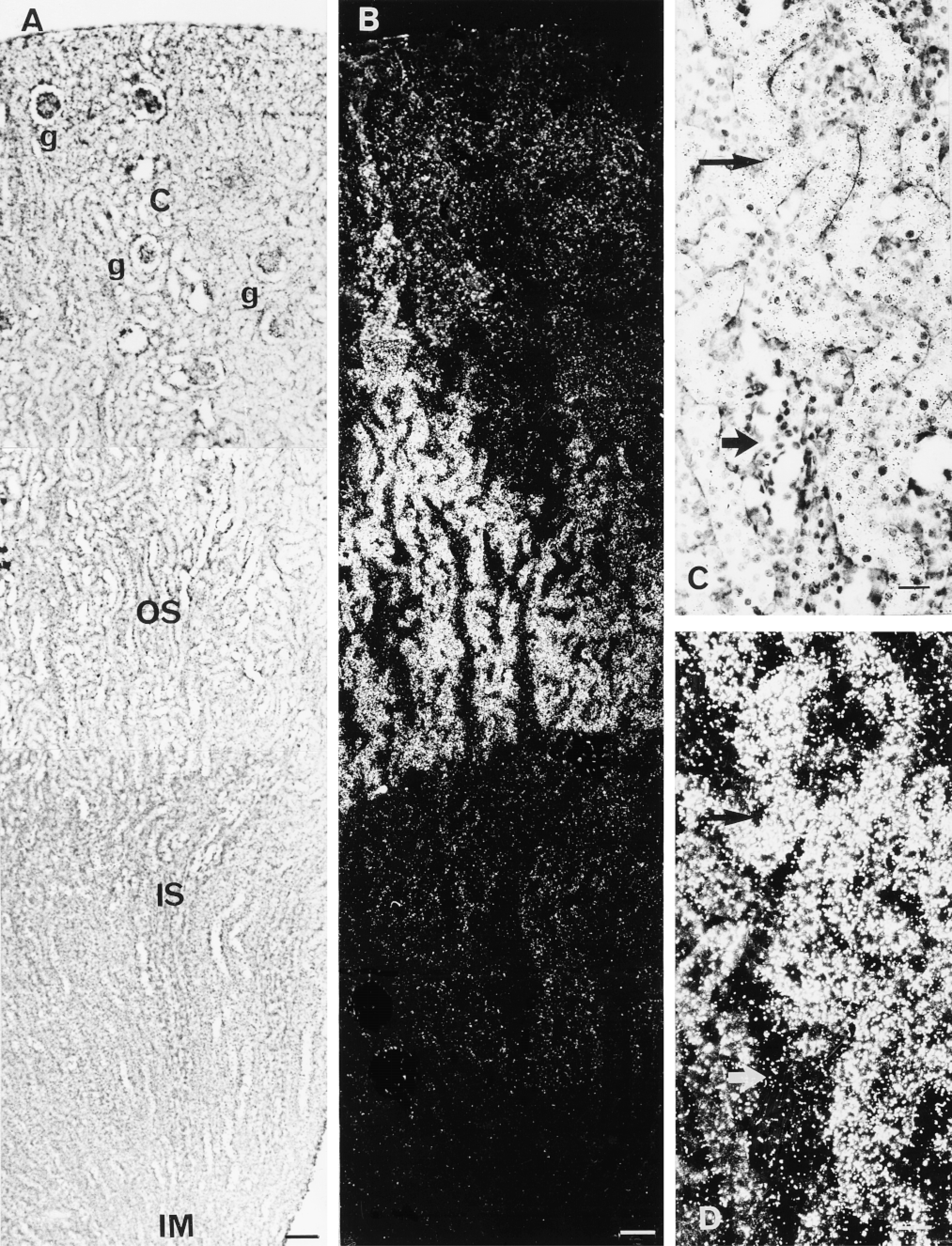
Localization of CSD mRNA in rat kidney by in situ hybridization. Emulsion autoradiographs of sagittal cryostat sections hybridized with an antisense 35S-labeled CSD RNA probe, dipped with a photographic emulsion and counterstained. (
Discussion
In this study, we provide new information on the regional distribution of CSD and CSD mRNA and on the histological localization of CSD mRNA in the kidney. By Western blotting only one protein band appeared labeled in soluble protein extracts from the dissected regions of the kidney. This protein has the same molecular weight (53 kD) as the recombinant CSD. By Northern blotting only one mRNA band appeared labeled with the CSD cDNA probe. This mRNA has the same size (2.5 kb) as that found in liver or brain RNA extracts (Kaisaki et al. 1995; Tappaz et al. 1998). These labeled bands therefore correspond to CSD protein and CSD mRNA, respectively. Both CSD protein and CSD mRNA appeared most enriched in the outer stripe. The relative enrichment of the mRNA was more pronounced than that of the protein.
In agreement with the regional distribution of the CSD mRNA, in situ hybridization with an antisense riboprobe showed very strong labeling in the tubules located in the outer stripe of the outer medulla and the cortical medullary rays, and only faint labeling in the inner stripe of the outer medulla and the cortical labyrinths. The lack of any labeling in the inner medulla indicates that the inner medullary collecting ducts, as well as the thin limbs of the long-looped nephrons, do not significantly express CSD. The strong labeling in the tubules of the outer stripe of the outer medulla might be located in some of the tubules present at this level, i.e., the proximal straight tubules, the distal straight tubules, and the collecting ducts. If the labeling were associated with the latter two, it would not be restricted to the outer stripe but should also extend into the inner stripe according to the anatomic organization of the nephrons (Kriz and Bankir 1988). In contrast, the proximal straight tubules are found in the outer stripe of the outer medulla but not in the inner stripe. Likewise, the strong labeling is restricted to the outer stripe. Therefore, we conclude that CSD is strongly expressed in proximal straight tubules. This conclusion is further supported by the fact that the labeled tubules show the conspicuous cytological features of proximal straight tubules at higher magnification: they are thick-walled, sinuous, and have a diameter of around 30 μm. Furthermore, strong labeling was observed over the tubules of the cortical medullary rays, which contain the proximal straight tubules of the short-looped nephrons. It must be stressed that the overall pattern of CSD mRNA labeling in the outer stripe of the outer medulla and cortical medullay rays is strikingly similar to the expression pattern of the mRNA of various proteins that were found primarily localized in the proximal straight tubules, such as ornithine decarboxylase (Blackshear et al. 1989), growth hormone receptor (Chin et al. 1992), iodothyronine 5′-deiodinase (Lee et al. 1993), glucose transporter GT5 (Chin et al. 1993), glutamate transporter (Shayakul et al. 1997), and renal chloride channel ClC5 (Luyckx et al. 1998). The very faint labeling in the inner stripe of the outer medulla and cortical labyrinths appears to be consistent with the low level of CSD mRNA detected by Northern blotting in these dissected renal regions and indicates a low expression of CSD outside the proximal straight tubules. We have not attempted to precisely identify the structures with which this quantitatively very minor labeling is associated.
Our results thus indicate that taurine formation through the sulfinate pathway in which CSD is held as the rate limiting step (De La Rosa and Stipanuk 1985) occurs primarily in the proximal straight tubules of the nephron. So far the biosynthesis of taurine by the kidney has been overlooked mostly on the basis that the average CSD activity in whole tissue extract was much lower in kidney than in liver (Jacobsen et al. 1964; Jerkins and Steele 1991). As a whole organ, the kidney was therefore rightly considered of minor relevance to the taurine body pool, which originates primarily from dietary intake and synthesis by the liver (Chesney 1985). The restriction of CSD expression to a limited number of cells in the nephron suggests that CSD content in these cells is indeed much higher than would be expected from the CSD assay in the whole organ. Moreover the selective ability of some cells to synthesize taurine suggests that taurine may be used in these cells for specific purposes.
The main conclusions of our investigation are that CSD expression occurs primarily in the part of the nephron, i.e., the proximal straight tubules, in which taurine is concentrated (Amiry-Moghaddam et al. 1994) and in which the taurine transporter is highly expressed (Matsell et al. 1997). These findings raise the possibility that both CSD and the taurine transporter might be expressed by the same cells. Double labeling is needed to establish this co-localization experimentally. According to this hypothesis, taurine might accumulate in the cells of the proximal tubules by local synthesis through the cysteine sulfinate pathway or by uptake from the lumen of the tubules through the taurine transporter. It was shown that taurine transporter mRNA expression was upregulated in the proximal straight tubules with dietary taurine restriction, whereas it appeared downregulated in animals supplemented with taurine (Matsell et al. 1997). Whether or not CSD expression may be affected by circulating taurine at this level remains to be studied. Thus far, the results that we have obtained in vitro in primary astrocyte cultures showed that taurine transporter and CSD expressions are differently sensitive to taurine in the culture medium. Whereas taurine transporter expression was downregulated by taurine, as previously reported in renal cell culture (Han et al. 1997), CSD expression did not appear to be affected (our unpublished results). One possible speculation is that the cells of the proximal tubules have the ability to accumulate taurine through their own biosynthesis capacity independently of regulation of expression of their taurine transporter. On the one hand, the cells of the proximal tubules regulate taurine reabsorption by a taurine transporter, the expression of which is regulated by circulating taurine (Chesney et al. 1985; Matsell et al. 1997), and thus maintain the taurine body pool (Chesney 1985). On the other hand, these cells can form taurine that they may use for their own purpose, e.g., as an osmolyte that could be accumulated through biosynthesis in response to an hyperosmotic environment. If this working hypothesis is correct, CSD expression in the proximal straight tubules is not expected to be affected by manipulation of dietary taurine intake, in contrast to the expression of the taurine transporter (Matsell et al. 1997).
Taurine was reported to be highly concentrated in the collecting ducts throughout the renal cortex and the renal medulla (Trachtman et al. 1993; Amiry-Moghaddam et al. 1994; Ma et al. 1994). This result appears quite puzzling because no significant expression of the taurine transporter (Matsell et al. 1997) or CSD, as reported in the present study, was found in the collecting ducts. These results, which are apparently not consistent with each other, may indeed reveal other mechanisms in taurine metabolism than those presently studied. There may exist other types of the taurine transporter than the one investigated thus far (Matsell et al. 1997). It must be recalled that the latter was localized using a probe corresponding to a taurine transporter cloned from a rat brain cDNA library (Smith et al. 1992). Taurine may also be synthesized through alternative pathways, such as the so-called cysteamine pathway, which does not involve decarboxylation of CSA by CSD (Jacobsen and Smith 1968; Griffith 1987). This pathway may be operative in kidney because cysteamine could be assayed in kidney extracts (Kuriyama et al. 1984). To our knowledge, the enzymes involved in this pathway have been neither isolated nor cloned to date. Consequently, no specific antibody or probe is available to test this hypothesis.
In conclusion, in this report we have provided evidence for a restricted localization of CSD mRNA in the cortical and medullary proximal straight tubules. Accordingly, a significant synthesis of taurine through the cysteine sulfinate pathway occurs only in this restricted portion of the nephron, where a taurine transporter was reported to be also highly expressed. Because taurine was reported to have a broader distribution, notably in the collecting ducts, where CSD or taurine transporter are not expressed, other synthesis pathways and/or transporters may be involved, which remain to be identified and precisely localized in the kidney.
Footnotes
Acknowledgements
Marc Bitoun is the recipient of a DRET fellowship DGA. We thank Didier Decimo (INSERM U 412 ENS, Lyon) for his help in setting up the in situ hybridization and Jean Louis Borach (INSERM U 94, Bron) for the photographic work.
