Abstract
PAPPA2 is a metalloproteinase which cleaves insulin-like growth factor binding protein (IGFBP)-3 and IGFBP-5, and its role in pregnancy and postnatal growth is primarily studied. Using exclusion mapping, we reported a subcongenic (26-P) rat where a 0.71-Mbp region containing the pregnancy-associated plasma protein a2 (Pappa2) allele of salt-insensitive Brown Norway (BN) was introgressed into Dahl saltsensitive (SS) genetic background, resulting in the reduction of salt sensitivity. Pappa2 was differentially expressed in the adult kidney of 26-P and SS rats. Here, the expression and cellular localization of Pappa2 in embryonic and postnatal kidneys of 26-P and SS rats were examined. Pappa2 mRNA expression was 5-fold higher in the embryonic kidney (day 20.5) of the 26-P rat compared with the SS rat. Pappa2 mRNA expression progressively increased with the development of kidney, reaching a peak at postnatal day 5 before trending downward in subsequent stages of development in both strains. At all tested time points, Pappa2 remained higher in the 26-P compared with the SS rat kidney. Immunohistochemistry studies localized PAPPA2 in the ureteric bud (UB) and distal part of S-shaped body. PAPPA2 was colocalized with IGFBP-5 in the UB and Na+/K+/2Cl− cotransporter–stained tubules, respectively. Future studies are needed to determine the role of Pappa2 in kidney development and mechanistic pathways involved in this process.
Introduction
The Dahl salt-sensitive (SS) rat model of hypertension has been a critical tool for identification of genes of hypertension, 1 providing unforeseen insights into determinant pathways of blood pressure (BP) regulation. SS rats subjected to a high-salt diet exhibit a rise in BP 2 associated with increased oxidative stress in the brain, vasculature, and kidneys,3–8 culminating in a malignant form of hypertension and renal failure.3,6,8–12 Mimicking the human diseases, 13 SS rats are insulin resistant, 14 hyperlipidemic,15,16 and exhibit proteinuria with rapid development of end-stage renal disease.17,18 SS rats exhibit 15% reduction in nephron number compared with salt-resistant rats, 19 which may contribute to hypertension, and kidney injury has yet to be examined.
Nephrogenesis is a reciprocal interaction between metanephric mesenchyme (MM) and ureteric bud (UB). In mice and rat at embryonic day 11.5 and 13.5, respectively, MM secretes glial cell line–derived neurotrophic factor (GDNF), which drives the tyrosine kinase receptor–expressing UB to outgrow and invaginate into the MM. The MM which condenses to the tip of UB called cap mesenchyme (CM) which migrates under the armpit of UB and forms pretubular aggregates (PTA). After a series of morphogenic events such as renal vesicle (RV), comma-shaped body (CSB), and S-shaped body (SSB), PTA eventually gives rise to a nascent nephron that remains fused with the UB which later differentiating into collecting duct. Meanwhile, the UB continues to branch and induce new pretubular aggregates at the UB tips. 20 Fine tuning of these processes is essential for the determination of nephron number.
By exclusion mapping of congenic rat strains, a subcongenic 26-P rat was identified in which a 0.71-Mbp narrow region of the salt-insensitive BN rat was introgressed into the genetic background of SS rats, resulting in the reduction of BP salt sensitivity and kidney injury. 21 Basically, 26-P and SS are genetically identical strain except for the difference in the narrow region containing three genes, including the pregnancy-associated plasma protein a2 (Pappa2). 21 We have identified the first specific genetic variant in rat (Pappa2) that would help to identify a single gene involved in kidney development, which could in part explain a contributing factor to BP saltsensitivity in SS rats.
PAPPA2 is a metalloproteinase of metzincin superfamily which cleaves insulin-like growth factor binding protein (IGFBP)-3 and IGFBP-5 to release IGF-1 (insulin-like growth factor-1) and is a homolog of PAPPA1 with which it shares only 45% amino acid sequence identity. 22 Both are found as circulating proteins produced by the embryo and placenta during pregnancy 23 and used by some as a diagnostic test for adverse pregnancy.23–26 PAPPA1 plays a role in the regulation of bone matrix mineralization, skin healing, aging, and angiogenesis. 27 Growing evidence suggests that PAPPA2 is important for the growth and postnatal development of animals and humans. Ablation of Pappa2 in mice resulted in the retardation of postnatal growth with normal size at birth. 28 Slight increases in the weight of various organs including kidney relative to body weight were observed in the knockout animal compared with the wild-type littermate 28 . However, femur and body length were reduced in these mice. 28 In humans, Pappa2 mutations are associated with postnatal growth retardation and short stature, a condition found treatable with recombinant human IGF-129,30 and with plasma transfusions from individuals with normal serum levels of PAPPA2. 31 Studies in mice found that Pappa2 has limited tissue expression compared with Pappa1, 28 being expressed in abundance in placenta and skeletal and less in kidney. 28 In humans, single-cell transcriptome analysis confirmed the expression of Pappa2 in fetal kidney development. 32 Our study on adult rats found that Pappa1 and Pappa2 were both expressed in the renal cortex but that they were not coexpressed in the same regions. 21 Also, it was Pappa2 but not Pappa1 that was found to be differentially expressed in the renal cortex of 26-P and SS strains. 21
Moreover, recently Pappa2 is identified as a candidate gene associated with human hypertension by genome-wide association study, 33 and by exclusion mapping we have identified Pappa2 to be strongly associated with BP salt sensitivity in SS rats. 21 Kidneys are critical for the salt-sensitive hypertension of SS rats, with limited information related to renal developmental defects being known. Azar et al. 19 reported that kidneys of SS rats have 15% fewer nephron than age-matched salt-resistant rats. The loss of nephron has an inverse relationship with renal hemodynamics, BP, and renal function in human and rodent animal models,34–36 which is discussed later.
Despite the critical evidence of Pappa2 in the growth and postnatal development of organs, the localized expression and role of Pappa2 in kidney development have not been determined. This study aimed to determine the differential and temporal expression and localization of Pappa2 in the developing kidneys of 26-P and SS strains.
Methods
Experimental Animals and Tissue Collection
Male and female Dahl SS (SS/JrHsdMcwi; https://rgd.mcw.edu/rgdweb/report/strain/main.html?id=61499) and subcongenic 26-P (SS.BN- [D13Hmgc1048-D13Hmgc1050/Mcwi; https://rgd.mcw.edu/rgdweb/report/strain/main.html?id=12792213]) rats were obtained as weanlings from colonies maintained at the Medical College of Wisconsin. Embryonic (E15.5, E17.5, E20.5) and postnatal (P0, P5, P10, P15, P20) kidneys were collected from SS and 26-P rats. Time-pregnant females of SS and 26-P rats were anesthetized at E15.5, E17.5, and E20.5 to obtain embryonic kidneys. One kidney was snap-frozen for RNA analysis and the other fixed in 10% formalin for immunohistochemical analyses. Similarly, for postnatal stages such as P0, P5, P10, P15, and P20 of 26-P and SS rats, one kidney was snap-frozen for RNA analysis and Western blot and another kidney was fixed in 10% formalin for immunohistochemical study of P20 of 26-P and SS rats. All the animals were provided water and food ad libitum. All protocols were approved by the Medical College of Wisconsin Institutional Animal Care and Use Committee.
Quantitative Polymerase Chain Reaction
Total RNA was extracted from embryonic and postnatal kidney tissue using Trizol reagent. The quality and quantity of each sample were determined by spectrophotometry (Nanodrop). Two µg of total RNA was reverse-transcribed by random hexamer primers into cDNA (Thermo Scientific RevertAid First Strand cDNA synthesis kit) and real-time polymerase chain reaction (RT-PCR) analysis performed using 8 to 10 ng of total RNA using SYBR Green Chemistry on a Quant Studio 6 Flex (Applied Biosystems, Foster City, CA). Pappa1 (Rn-Pappa-2-SG) and Pappa2 (Rn-Pappa2-1-1-SG) primers were obtained from Qiagen (Hilden, Germany). The gene expression was normalized to housekeeping gene control glyceraldehyde 3-phosphate dehydrogenase (Gapdh).
Immunohistochemistry
As described previously, 37 10% formalin–fixed embryonic and postnatal kidneys were embedded in paraffin. Sections were cut at a thickness of 5 μm. Slides were deparaffinized in xylene washes and rehydrated with graded series of ethanol. Antigen retrieval was performed with 1X Trilogy (Cell Marque; Rocklin, CA) at a subboiling temperature for 10 min and sections incubated in PBS with 3% H2O2 for 10 min to eliminate endogenous peroxidase. Slides were washed for 5 min in PBST (PBS + 1% Tween 20) and blocked with 3% bovine serum albumin in PBST (blocking buffer) for 2 hr at 4C. Goat anti-PAPPA2 primary antibody (R&D Systems; Minneapolis, MN), 1:1000) was diluted in blocking buffer and incubated at 4C overnight. Slides were washed three times for 30 min in PBST and incubated in donkey anti-goat horseradish peroxidase (HRP) secondary antibody (1:1000 in blocking buffer) for 1 hr at room temperature (RT). Tyramide amplification was done as per the manufacturer’s instructions (Perkin Elmer; Waltham, MA). Slides were washed three times for 10 min in PBST and blocked with 3% bovine serum albumin for 2 hr at 4C. Rabbit anti-JAGGED1 (Santa Cruz; Dallas, TX), 1:500) primary antibody or Dolichos Biflorus Agglutinin (Vectashield, 1:1000; Burlingame, CA) or anti-IGFBP-5 (Santa Cruz, 1:1000) was diluted in blocking buffer and incubated at 4C overnight. Slides were washed three times for 30 min in PBST and incubated in goat Alexa-555 coupled secondary antibody (Life Technologies; Carlsbad, CA), 1:1000 in blocking buffer) for 1 hr at RT. Slides were washed three times for 30 min in PBST and mounted with Vectashield containing 4′, 6-diamidino-2-phenyldole (DAPI). Once processed and prepared for imaging, kidney slides were viewed and images captured with Nikon Ni-E Motorized Upright microscope equipped with DS-QiMc camera, NIS-Elements software, and Nikon ECLIPSE 80i microscope equipped with a QIClick CCD camera (Q imaging) and MetaVue research imaging system (Molecular Devices; San Jose, CA).
For no primary antibody control, the tissues were incubated with blocking buffer, without the primary antibody included. Similarly, for absorption control, the antigen recombinant human PAPPA2 (R&D Systems) to anti-PAPPA2 was made at a working dilution of 10:1 (molar ratio) and was preincubated overnight at 4C.
Western Blot
Western blot was performed as described previously. 38 Briefly, blotting experiments were performed with 30 μg of protein obtained from a renal tissue homogenate. Proteins were separated on a 10% to 20% SDS-PAGE gel (Bio-Rad; Hercules, CA), transferred to a polyvinylidene fluoride membrane (PVDF), blocked for 1 hr at room temperature, and incubated overnight with primary antibodies (PAPPA1 and PAPPA2) at 4C. Bound primary antibodies were detected with an HRP-labeled secondary antibody and visualized by chemiluminescence. A ChemiDox XRS+ imaging system (Bio-Rad) was used for chemiluminescence detection of bands, and Image Laboratory software, version 5.1 was used for quantification by densitometry. PAPPA1 and PAPPA2 expression levels were normalized with the internal control GAPDH.
Statistical Methods
Data are presented as mean values ± standard error. Unpaired Student’s t-test was used to compare between the strains, with p<0.05 considered significant.
Results
Embryonic and Postnatal Expression of Pappa2 in the Developing Kidney of Salt-insensitive 26-P and Salt-sensitive SS Rat Strains
The mRNA expression of Pappa2 in embryonic (E15.5, E17.5, E20.5) and postnatal (P0, P5, P10, P15, P20) kidneys was determined by quantitative RT-PCR (qRT-PCR). As shown in Fig. 1A, Pappa2 mRNA expression was 5-fold higher at E20.5 in 26-P compared with SS embryonic kidneys. Following E20.5, mRNA expression progressively increased, with the age of kidney reaching a peak at P5 in both strains. At this stage, 26-P rats exhibited 3-fold higher levels of Pappa2 compared with SS rats and at P15 were 9-fold higher. These high expression levels subsided by P20, but 26-P rats continued to remain at significantly higher levels than SS rats. Pappa2 expression was below detection level at E15.5 in both strains (not shown). The protein expression of Pappa2 was assessed in postnatal kidneys by Western blot. As shown in Fig. 1B and C, a clear correlation between mRNA message and protein expression of PAPPA2 was observed in postnatal kidneys of 26-P and SS rats. It is evident that Pappa2 is closely associated with both the embryonic and postnatal development of both the SS and 26-P rat kidneys, albeit expressed in greater abundance in the 26-P rat.
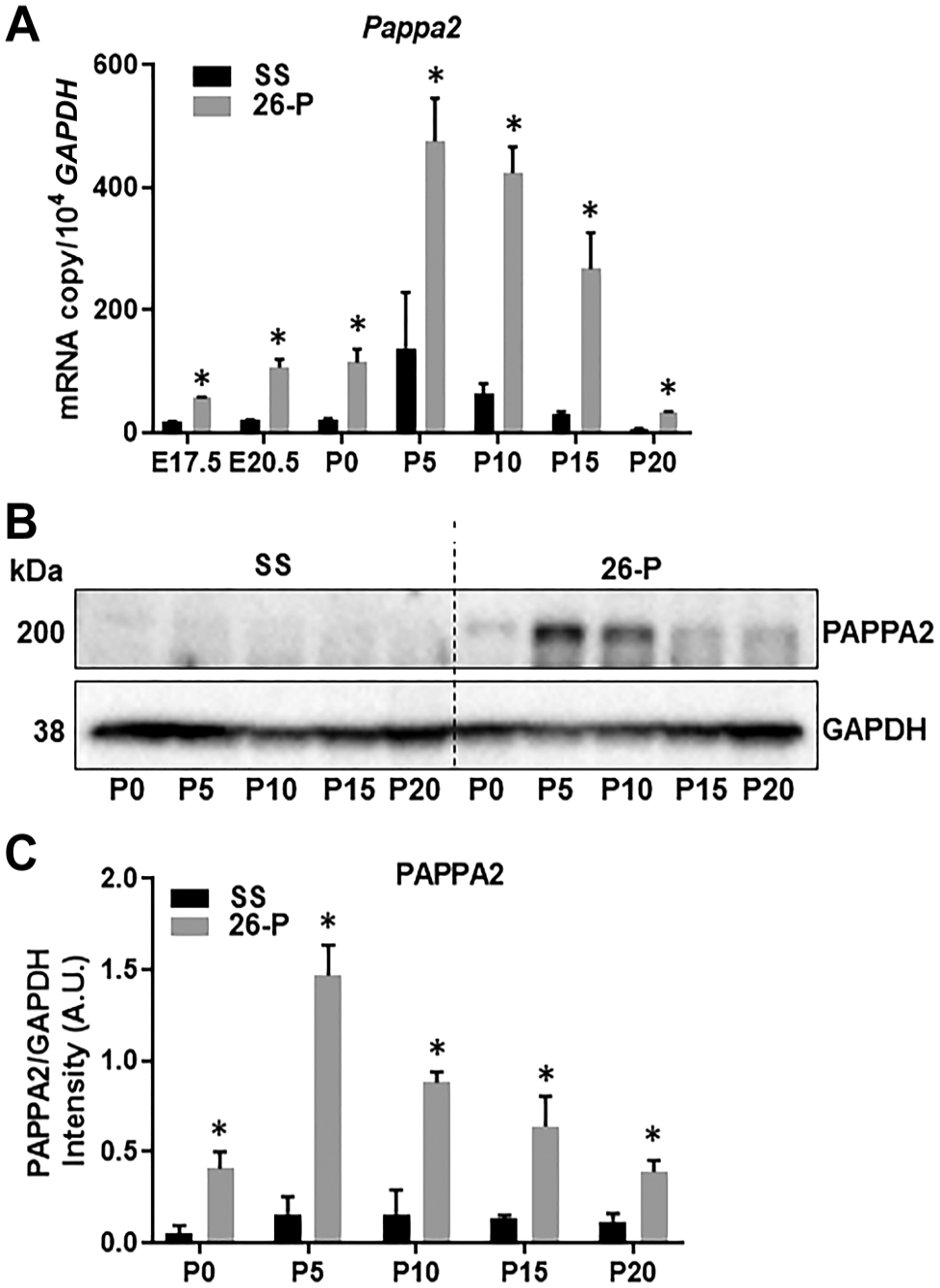
mRNA and protein were extracted from homogenates of embryonic and postnatal kidneys of 26-P (n=3) and SS (n=3) rats to determine the mRNA expression and protein level by quantitative polymerase chain reaction and Western blot analysis, respectively. (A) Pappa2 expression in the developing kidneys. mRNAs from various kidney developmental stages were used for Pappa2 expression analysis. (B) Representative immunoblots for kidneys are shown. *Significant difference between strains at the given kidney stage. (C) Western blot of PAPPA2 was normalized with GAPDH and data are represented in intensity (A.U.) for each stage. Abbreviations: GAPDH, glyceraldehyde 3-phosphate dehydrogenase; SS, salt-sensitive.
Expression of Pappa1 in the Developing Kidney of 26-P and SS Rats
PAPPA1 is another metalloproteinase in the same metzincin superfamily and expressed in abundance in kidney. 28 To determine whether Pappa1 was also differentially expressed in these rats, mRNA expression levels of embryonic and postnatal kidneys were compared by qRT-PCR between 26-P and SS rats. As shown in Fig. 2A, the expression of Pappa1 at various kidney developmental stages did not differ significantly in these two rat strains. This was further confirmed at the protein level by Western blot analysis (Fig. 2B and C). These data suggest that unlike Pappa2, Pappa1 is not differentially expressed during embryonic and postnatal kidney development in SS and 26-P rat kidneys.
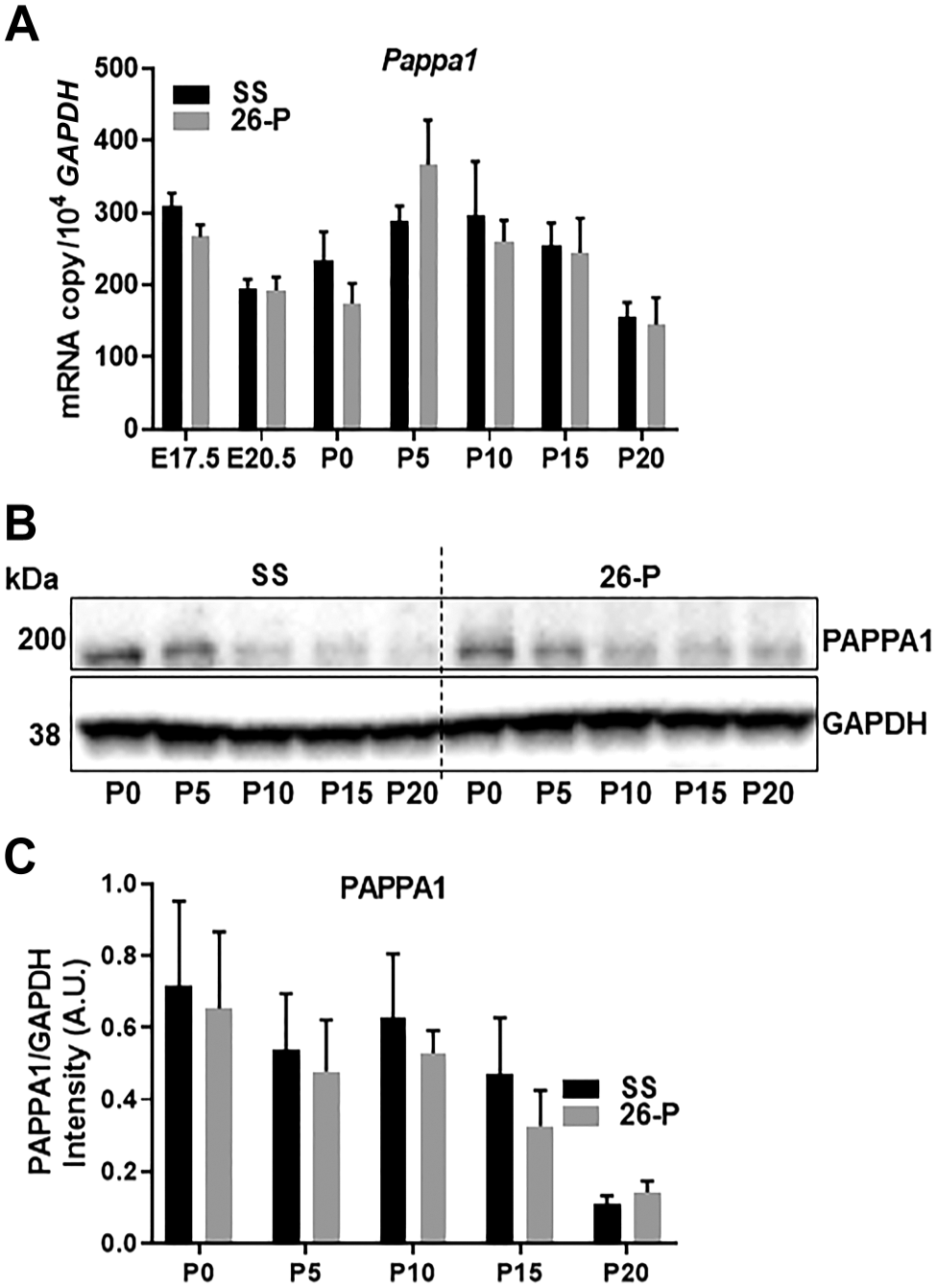
mRNA expression and protein of Pappa1 in the kidney homogenates from embryonic and postnatal kidneys of 26-P (n=3) and SS (n=3) strains. mRNA expression and protein level were normalized to Gapdh. (A) Pappa1 expression in the developing kidneys of 26-P and SS rats did not differ. (B) Representative immunoblots of PAPPA1 for kidneys are shown. (C) Western blot of PAPPA1 data is represented in intensity (A.U.) for each stage. *Significant difference between strains at the given kidney stage. Abbreviations: GAPDH, glyceraldehyde 3-phosphate dehydrogenase; SS, salt-sensitive.
Localization of PAPPA2 in the Developing Kidney of SS and 26-P
Immunohistochemistry (IHC) was used to describe the localization of PAPPA2 in developing kidneys beginning from E17.5 through P20 of 26-P and SS strains. Consistent with mRNA and protein expression studies, as shown in Fig. 3, at E17.5 PAPPA2 was only detected in the 26-P rat which increased at E20.5. Expression of Pappa2 at E17.5 and E20.5 of SS kidney was very weak. A unique pattern of the expression of Pappa2 was observed as development advanced to P10 when it was found to be confined largely to the cortical region. This was even more evident at P20 of 26-P when weak expression could be observed in the SS rat kidney. Together, it indicates that Pappa2 is important for embryonic as well as postnatal kidney development.
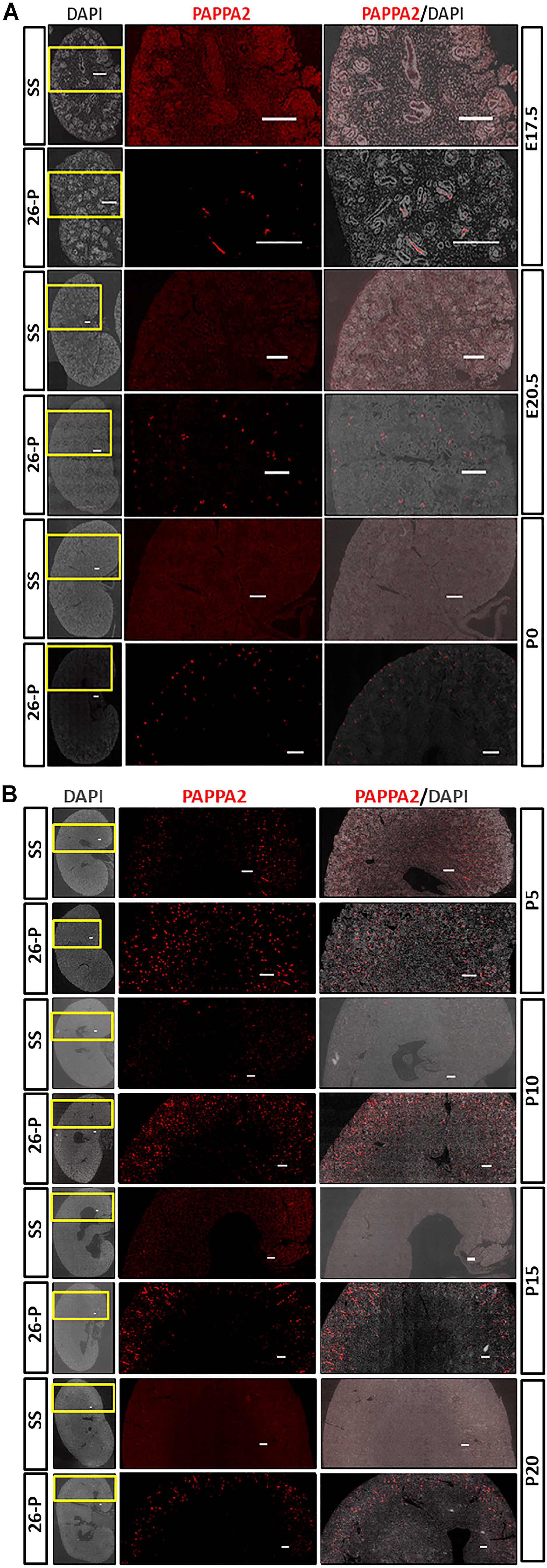
Immunohistochemical analysis of PAPPA2 in the various kidney developmental stages of 26-P and SS rats. DAPI was used to counterstain the nuclei. Representative whole kidney images and corresponding enlargements of yellow boxed areas are shown for each developmental stage of kidney. Scale bar = 250 µm. As shown in the figure, PAPPA2 expression was below detection level in E17.5, E20.5, and P0 kidneys of SS rat. Immunoreactivity was detected in the 26-P strain at the same-aged kidneys. The 26-P strain exhibited a unique pattern from P0 to P20. At P0, PAPPA2 was expressed throughout the kidney tissue, but as it developmentally grew it was mainly confined to the cortical region of the kidney. Abbreviation: SS, salt-sensitive; DAPI, 4′, 6-diamidino-2-phenyldole; PAPPA2, Pregnancy-associated plasma protein A2.
PAPPA2 Localized in UB and SSB and Colocalized With NKCC2-stained Tubules
At E20.5, PAPPA2 was localized in the peripheral nephrogenic zone of 26-P rat kidneys (Fig. 4A), a feature not observed in SS rat kidneys (Fig. 4B). As found on closer examination, expression of PAPPA2 was observed in the UB at E20.5 of 26-P rat. Intermediate nephron progenitors such as renal vesicle, comma-shaped body, and S-shaped body were determined by labeling with JAGGED1 protein (Fig. 4C and D). As shown in Fig. 4E and F, PAPPA2 was localized in the distal part of SSB. A clear staining of PAPPA2 was apparent in the nephron tubular structure at E20.5 and P5 of 26-P kidneys, and PAPPA2 was colocalized with Na+/K+/2Cl− cotransporter (NKCC2)-positive stained structures (Fig. 4G and H). PAPPA2 was secreted and detected in the lumen of nephron tubules (Fig. 4H). Together, these observations suggest a role for Pappa2 in the regulation of kidney development and sodium homeostasis.
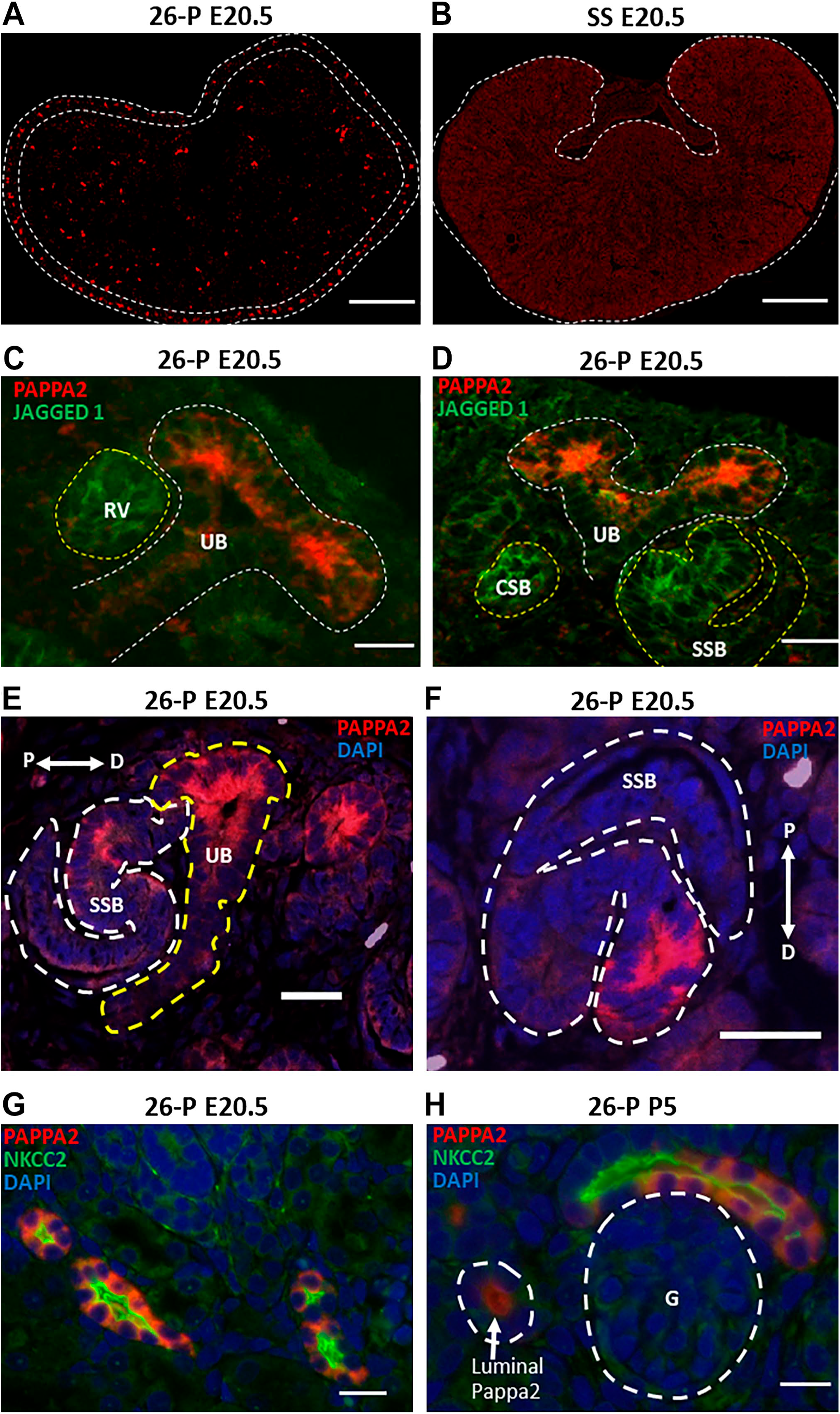
PAPPA2 protein localization at E20.5 of 26-P and SS is shown in (A) and (B). Nephrogenic zone is outlined by white dashed line in (A) and (B). Scale bar = 500 µm. As shown in (C) and (D), PAPPA2 was localized in the UB, which is outlined by white dashed line at E20.5 of 26-P rats. JAGGED 1 costaining was performed to illustrate the nephron progenitors such as RV, CSB, and SSB. These intermediates structures are outlined by yellow dashed lines. Scale bar = 20 µm. (E) and (F) showed the localization of PAPPA2 in SSB outlined by white dashed line and in UB outlined by yellow dashed line. SSB was found to be fused with UB. Patterning of SSB is shown by double-headed arrow with proximal (P) and distal (D) orientation. Scale bar = 20 µm. (G) and (H) showed the colocalization of PAPPA2 and NKCC2-positive tubules at E20.5 and P5 of 26-P rat kidneys. In the same image, luminal PAPPA2 was also detected, which is outlined by white lines. Glomeruli (G) are shown here in white dashed line. Scale bar = 20 µm. Abbreviations: SS, salt-sensitive; UB, ureteric bud; RV, renal vesicle; CSB, comma-shaped body; SSB, S-shaped body; NKCC2, Na+/K+/2Cl− cotransporter.
PAPPA2 Colocalized With Its Substrate IGFBP-5 in UB
Figure 5 shows a representative photomicrograph of the immunohistochemical localization of PAPPA2 and its substrate IGFBP-5, showing that these two proteins are colocalized in the UB of 26-P rats at E20.5, indicating that they are positioned to interact with each other in UB.
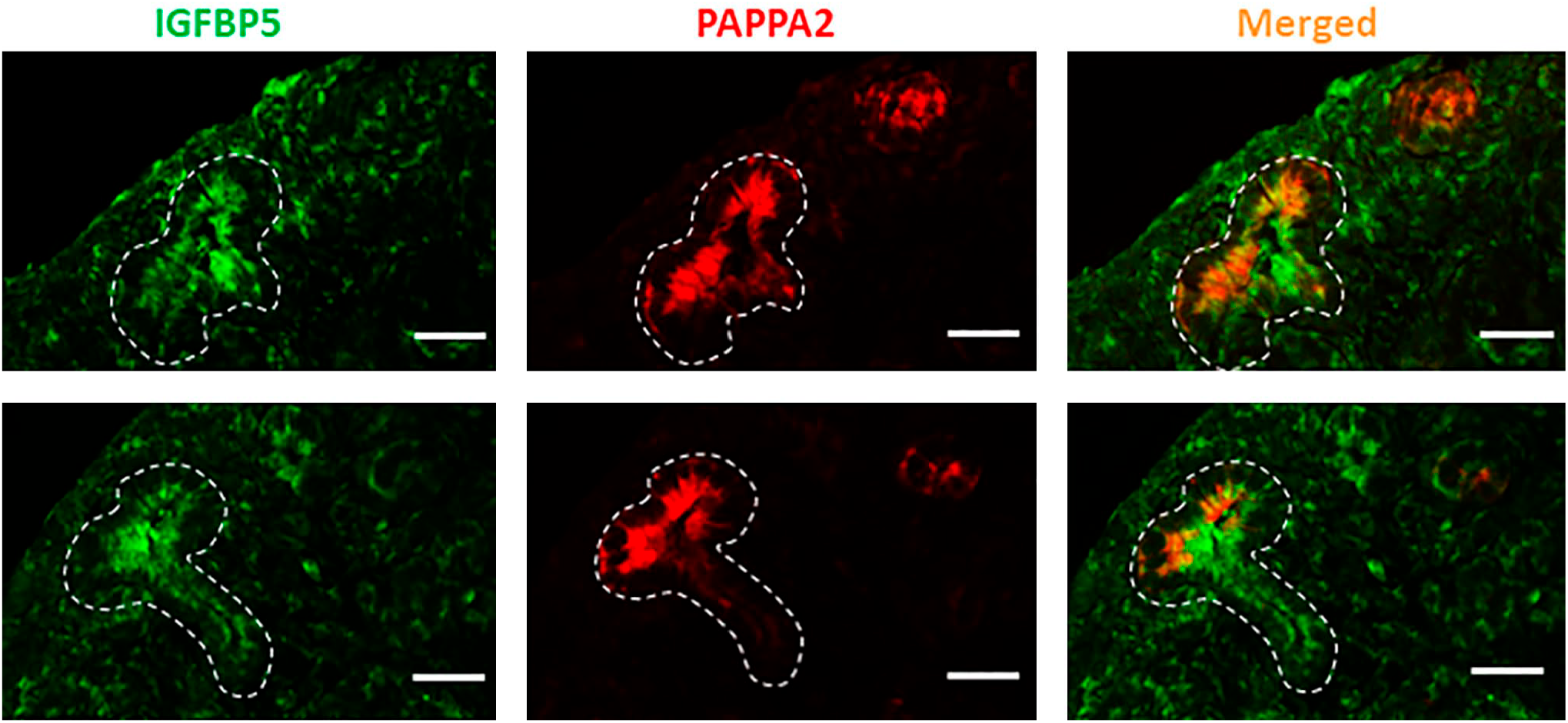
Colocalization of PAPPA2 and IGFBP-5 was performed at E20.5 kidney of 26-P rat. As shown in the merged panel, PAPPA2 and IGFBP-5 were colocalized in the ureteric bud (UB), which is outlined by white dashed lines in all images. Scale bar = 20 µm. Abbreviation: IGFBP-5, insulin-like growth factor binding protein-5.
Validation of PAPPA2 Antibody in Embryonic Kidney
We have previously reported the specificity of anti-PAPPA2 antibody in the kidney of 7-week-old 26-P rat. 21 Here, we examined the specificity of the same antibody in the embryonic kidney at E20.5 of the 26-P strain. For absorption control, the antigen recombinant PAPPA2 was obtained and incubated with anti-PAPPA2 at working dilution of 10:1 (molar ratio) at 4C overnight. In another control study, tissues were incubated without primary antibody with blocking buffer followed by secondary antibody and image was taken. As shown in Fig. 6A and B, both negative controls did not show any staining, whereas tissue incubated with anti-PAPPA2 showed immunoreactivity (Fig. 6C). These control studies show that anti-PAPPA2 antibody has specificity to PAPPA2 protein in both embryonic and adult kidneys. 21
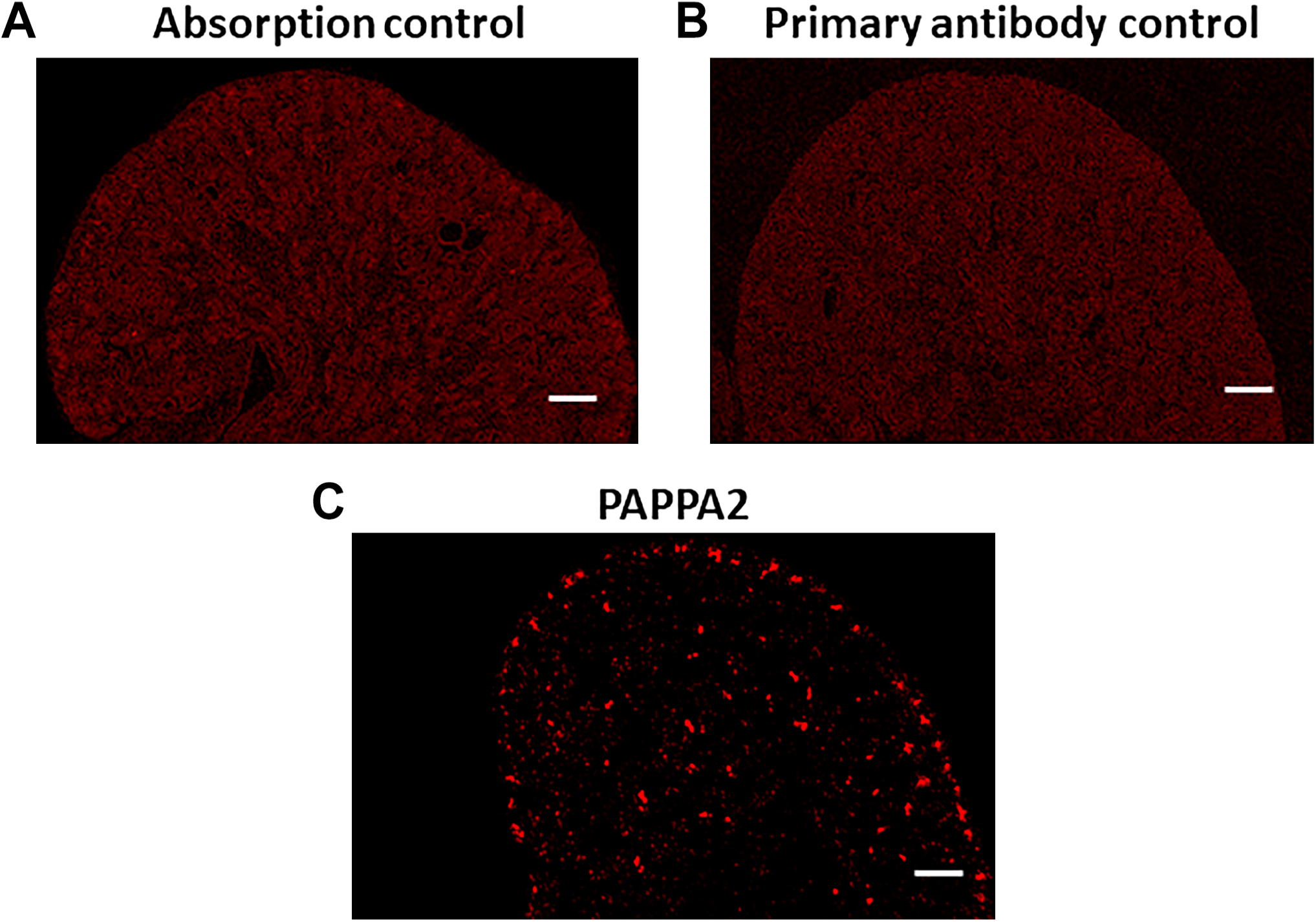
As shown in (A) and (B), immunoreactivity of PAPPA2 at E20.5 of 26-P rat kidney was not detected in either antigen absorption or primary antibody control. (C) serves as a positive control in the experiment. Scale bar = 500 µm.
Discussion
This study documents the differential expression of Pappa2 in embryonic and postnatal kidneys, showing that the allele variants of Pappa2 have profound effects on these patterns. These expression patterns were unique for Pappa2 because Pappa1 did not change in any consistent manner, nor were differences observed between kidneys of SS and 26-P rats throughout development. Distinct patterns of expression of Pappa2 were also observed as PAPPA2 colocalized with its substrate IGFBP-5 in the UB and localized in the distal part of SSB in the embryonic kidney of 26-P rat. PAPPA2 was found to be colocalized with NKCC2-stained tubules in embryonic and in postnatal kidneys. 21 Analysis of the temporal changes in gene expression of Pappa2 in kidney revealed a continuous and progressive increase from E17.5 to P5 and then a gradual reduction from P10 to P20, suggesting that it functions in both embryonic and postnatal kidneys. The most obvious explanation for this pattern is active nephrogenesis during the embryonic and postnatal period in rat kidney, a process known to require IGF for cell proliferation. In this context, a similar expression profile of IGF-2 has been observed in the developing mouse kidney. 39 These studies pave the way for a novel hypothesis regarding the role of Pappa2 as a local facilitator of IGF-1 in kidney development or hypertension.
To identify specific sites of PAPPA2 expression, IHC was performed in various embryonic and postnatal kidneys of 26-P and SS strains. In the embryonic kidney, PAPPA2 was found to be expressed in the nephrogenic zone at E20.5 of 26-P rats, but below detection level was seen in SS kidneys at E20.5. The nephrogenic zone is the active site where nephrogenesis begins with reciprocal interactions between MM and the UB. The primary UB outgrowth and branching are initiated by GDNF, which is secreted by MM, and the Ret receptor, expressed in the UB cells. GDNF and its canonical receptor Ret signaling are important in both processes and are activated by many pathways such as ERK/MAP kinase and IGF-1/PI3-kinase. 40 IGFBP-5, the substrate of PAPPA2, is expressed in the kidney41,42 and as found in this study is colocalized with PAPPA2 in the UB. Given that PAPPA2 contributes to the proteolysis of IGFBP-5, colocalization in the UB likely leads to the breakdown of IGFBP-5 in the epithelial duct of UB, freeing IGF-1 which might contribute to play a role in prenatal and postnatal kidney development of rat. Further studies are warranted to determine the role of Pappa2 in UB branching morphogenesis and thereby nephron number.
The link between low nephron number and the risk of hypertension and kidney disease has been shown in humans and experimental animals.34–36 Brenner et al.
43
first identified an inverse association between nephron number and human hypertension. There has been great interest in the hypothesis proposed by this group that reductions in nephron numbers lead to glomerular hyperfiltration, enlargement, and injury.
44
Many human studies have since found association of nephron numbers and hypertension, but given the methodological limitation related to non-invasive glomerular counting, studies have not consistently been supportive of this hypothesis.45,46 However, there remains considerable interest in the Brenner hypothesis, with many human studies finding evidence of reduced nephron number associated with hypertension and renal failure.44,47–50 Reduced nephron number is of particular interest as being related to premature birth with underdeveloped kidneys because there is considerable evidence of increased risk of cardiovascular and renal disease later in life in this population as recently reviewed by Wang and Garrett.
46
Recently, single-cell transcriptome analysis of fetal membrane of preterm babies suggested a candidate biomarker of premature birth.
51
Animal studies have provided greater insight into the relationship of nephron number, hypertension, and renal disease, given direct and higher fidelity methods of nephron counting. Mouse models have found that reduced nephron numbers (~20–30%) associated with premature birth52,53 or genetically modified mice with similar reductions of nephron numbers53,54 exhibit significant increases in systolic BP by 1 year of age. Similar inverse correlations have been reported in rat models in which it has also become clear that genetic background importantly modifies the effects of nephron number on cardiovascular and renal outcomes in humans and rodents.
46
For example, the lower nephron endowment and renal injury present in Munich Wistar Frömter (MWF) rats can be prevented when a QTL on chromosome 6 of the spontaneous hypertensive rat (SHR) is transferred into the MWF rat.
55
The rodent studies have found that even if reduced nephron number is associated with minimum changes of BP (such as uninephrectomy), exaggerated chronic BP responses are seen if a “second hit” is superimposed. This has been observed with a variety of stressors superimposed upon animals with reduced nephron mass such as salt loading and administration of angiotensin II or
The SS rat is a commonly used genetic model to discover gene which would lead to hypothesis-driven renal physiology to unravel the molecular mechanism underlying SS hypertension. SS rats develop BP salt sensitivity, which results in kidney injury including glomerulosclerosis and tubulointerstitial and vascular hypertrophy. 19 SS rats showed 15% fewer glomeruli in the kidney compared with salt-resistant rats. 19 Using an approach similar to ours, Williams et al. 64 identified a narrow congenic strain where 375 kb of SHR was introgressed in SS genetic background, which increases nephron number and protects kidney injury independent of BP. Together, these observations clearly support that less nephron number is an inherent problem in SS rats and probably could be one of the important causative factors in the regulation of BP salt sensitivity and kidney injury.
We have reported earlier that the allelic variants of Pappa2 in subcongenic 26-P strain influence the regulation of Pappa2 and probably the capacity of sodium handling in the adult kidney. 21 The effect of strain difference on gene expression has been previously demonstrated in various organs from mouse, rat, and other species.65–67 However, evidence for such variation in embryonic and postnatal kidney development is limited. 68 The 26-P strain is of special interest because it is salt-insensitive compared with the SS strain which is genetically identical except for allelic difference in three genes including Pappa2 which more effectively excretes high dietary salt intake. 21
Using single-cell transcriptome analysis of human fetal kidney, Hochane et al. 32 identified Pappa2 in the medial/distal portion of SSB, which accounts for the precursor cells of loop of Henle and distal tubule. Our present study reconfirmed the presence of PAPPA2 in the distal part of SSB and was colocalized with NKCC2-positive tubules in embryonic and postnatal kidneys of the 26-P rat. In contrast, immunoreactivity in the SS strain was very weak and PAPPA2 was below detection level even at birth in SS compared with 26-P. At P5, however, PAPPA2 was detected in the NKCC2 of SS strain (data not shown) and thereafter diminished and was undetectable in the adult kidney. 21
Previous work with mice found that Pappa2 plays a role in postnatal growth, including tail length, and skeletal growth. 28 In rat, nephrogenesis continues until P8 to P10, and it is likely that Pappa2 contributes to the formation of a mature nephron which requires segmentation and patterning. 40 Conditional/tissue-specific gene knockout models are not yet feasible in rats, and at this time, quantification of channels, transporters, and cotransporters represents the only useful strategy to assess the function of Pappa2 in nephron segmentation.
In conclusion, we have found that Pappa2 but not Pappa1 gene is differentially expressed in the embryonic and postnatal kidneys of salt-insensitive and salt-resistant strains. The localization of PAPPA2 was found to be in the UB, distal part of SSB, and NKCC2-stained tubules in the developing kidney of the subcongenic salt-insensitive rat. Further studies are needed to unravel the function of Pappa2 in the nephrogenesis and nephron number and its functional consequence on BP.
Footnotes
Acknowledgements
We thank Alison Zappa from Geurts’ lab at Medical College of Wisconsin for helping us obtain time-pregnant female rats.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
VK and AWC jointly conceived and were responsible for coordinating all aspects of this work. VK and CY conducted experiments and performed analysis. VK performed Western blot and immunohistochemical analysis. CY performed RNA experiments. VK was responsible for the first draft and revision of the manuscript, which was modified by working with AWC prior to being sent to all other authors. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institute of Health grant P50 DK096373 via UV PCEN P&F Grant (to V.K.) and AHW-REP award (to A.W.C.).
