Abstract
During the maturation stage of amelogenesis, the loss of matrix proteins combined with an accentuated but regulated influx of calcium and phosphate ions into the enamel layer results in the “hardest” tissue of the body. The aim of the present investigation was to examine the effects of chronic hypocalcemia on the maturation of enamel. Twenty-one-day old male Wistar rats were given a calcium-free diet and deionized water for 28 days, while control animals received a normal chow. The rats were perfused with aldehyde and the mandibular incisors were processed for histochemical and ultrastructural analyses and for postembedding colloidal gold immunolabeling with antibodies to amelogenin, ameloblastin, and albumin. The maturation stage enamel organ in hypocalcemic rats exhibited areas with an apparent increase in cell number and the presence of cyst-like structures. In both cases the cells expressed signals for ameloblastin and amelogenin. The content of the cysts was periodic acid–Schiff- and periodic acid–silver nitrate–methanamine-positive and immunolabeled for amelogenin, ameloblastin, and albumin. Masses of a similar material were also found at the enamel surface in depressions of the amelo-blast layer. In addition, there were accumulations of glycoproteinaceous matrix at the interface between ameloblasts and enamel. In decalcified specimens, the superficial portion of the enamel matrix sometimes exhibited the presence of tubular crystal “ghosts.” The basal lamina, normally separating ameloblasts and enamel during the maturation stage, was missing in some areas. Enamel crystals extended within membrane invaginations at the apical surface of ameloblasts in these areas. Immunolabeling for amelogenin, ameloblastin, and albumin over enamel was variable and showed a heterogeneous distribution. In contrast, enamel in control rats exhibited a homogeneous labeling for amelogenin, a concentration of ameloblastin at the surface, and weak reactivity for albumin. These results suggest that diet-induced chronic hypocalcemia interferes with both cellular and extracellular events during enamel maturation. (J Histochem Cytochem 48:1043–1057, 2000)
CALCIUM regulates many cellular activities, such as cell communication, signal transduction, activation of enzymes, and polymerization of cytoskeletal proteins (Poyner 1994). During the maturation stage of amelogenesis, the epitheliam-derived cells responsible for the formation of enamel undergo a major reorganization and cyclically alternate the morphology of their apical extremity (Takano and Ozawa 1980; Josephsen and Fejerskov 1977; Smith et al. 1987; Smith 1998). These changes create a physiological environment that permits the bulk loss of matrix proteins and growth in width and thickness of enamel crystals (reviewed in Smith 1998). Unlike other calcified tissues, mature enamel contains only trace amounts of organic matrix, making it the hardest tissue of the body (reviewed in Nanci and Smith 2000). Although for a short period of time during ameloblast modulation when apical junctional complexes are dissociated the enamel organ is leaky (Smith 1998), it generally seals off the forming enamel layer and limits the passage into maturing enamel of interstitial fluid molecules, such as albumin (Chen et al. 1995; Nanci et al. 1996). Indeed, the accumulation of albumin in enamel is believed to be indicative of defective maturation and to result in enamel hypoplasia (Robinson et al. 1992).
Despite the importance of calcium in amelogenesis, there have been relatively few studies on the influence of dietary calcium on the formation of enamel (Lozupone and Favia 1989; Limeback et al. 1992; Bonucci et al. 1994). Complete absence of calcium in the diet results in the formation of hypomineralized enamel (Bonucci et al. 1994). Furthermore, thyro-parathy-roidectomy, which produces severe hypocalcemia, affects the morphology of ameloblasts as well as the organization and degree of mineralization of enamel (Acevedo et al. 1996).
The aim of the present investigation was to examine the effect of chronic hypocalcemia on the integrity of the enamel organ and the distribution of enamel proteins during the early maturation stage, using histochemical staining for glycoconjugates, in situ hybridization, and postembedding colloidal gold immunocytochemistry.
Materials and Methods
Animals and Diet
Twenty-six male Wistar rats weighing approximately 45 g (Charles River; St- Constant, QC, Canada) were used for the experiment. They were housed two per cage, maintained on a 12-hr light/12-hr dark cycle, and given free access to food and deionized water. The animals were divided into a treated (n = 18) and a control (n = 8) group. The treated group was fed a calcium-free diet (Altromin DP1031; Rieper, Vandoies, Italy) and the control group a normocalcic chow (Altromin DP1000; Rieper) for 28 days. Both the normal and calcium-free food formulations contained 1000 IU of vitamin D/kg. Each pair of control and treated animals was given equal daily quantities of food and water. Food consumption was measured every 2 days and water every 4 days. The data were averaged and expressed as the weekly food and water consumption per rat. The difference among groups was assessed by the bidirectional t-test adjusted for multiple comparison. One-way analysis of variance (ANOVA) was performed to determine significant differences. The experimental protocol was approved by the Comité de Déontologie de l'Expérimentation sur les Animaux of the Université de Montréal.
Blood Sampling
The rats were anesthetized with chloral hydrate (0.4 mg/g body weight; Sigma Chemical, St Louis, MO) and approximately 3 ml of blood was drawn from the jugular vein. The serum levels of (a) phosphorus, calcium, and alkaline phosphatase were measured as an indicator of calcification, (b) creatinine of renal status, (c) albumin of intestinal protein absorption, and (d) aspartate aminotransferase and alanine transferase liver activity in relation to alkaline phosphatase levels. These parameters were measured using routine hospital biochemical assays [Pavillon St-Luc, Centre Hospitallier de l'Université de Montréal (CHUM), Montreal, QC, Canada].
Morphological Analyses
After blood was drawn, the anesthetized rats were perfused through the left ventricle first with lactated Ringer's solution (Abbott; Montreal, QC, Canada) for approximately 30 sec and then with fixative for 20 min. The fixative solution consisted of either 4% paraformaldehyde + 0.1% glutaraldehyde (PG) in 0.08 M sodium cacodylate buffer, pH 7.3, or 1% glutaraldehyde (G) in the same buffer. The hemimandibles were removed and immersed in the corresponding fixative for 3 hr (PG) or overnight (G) at 4C. The right hemimandibles were left calcified, whereas the left ones were decalcified in 4.13% disodium EDTA, pH 7.2, for 21–28 days at 4C (Warshawsky and Moore 1967). Segments of enamel organ and enamel from the early to mid-maturation stage were prepared using a molar reference line (Smith and Nanci 1989) and processed for embedding in Taab 812 epoxy resin (Marivac; Halifax, NS, Canada) or LR White acrylic resin (Mecalab; Pointe-aux-Trembles, QC, Canada). For Epon embedding, specimens were postfixed in potassium ferrocyanidereduced osmium tetroxide (Neiss 1984), dehydrated in graded ethanols, cleared in propylene oxide, infiltrated with resin, and polymerized at 60C for 48 hr. For LR White embedding, the specimens were dehydrated in graded ethanols, infiltrated at 4C, and polymerized at 48C for 48 hr (Nanci et al. 1989). Decalcified hemimandibles fixed with PG were embedded in paraffin and sections processed for in situ hybridization (see below). For light microscopy, 1-μm-thick sections of plastic-embedded tissues were stained with either toluidine blue or azure II-methylene blue. Periodic acid-Schiff (PAS) and periodic acid-silver nitrate-methenamine (PASNM) (Marinozzi 1961) were used to detect glycoproteins. Some sections from calcified specimens were also stained for calcium phosphate by the von Kossa method. Ultrathin sections of selected areas from both calcified and decalcified specimens were cut using a diamond knife, mounted on Formvar/carbon-coated nickel grids, and processed for postembedding colloidal gold immunolabeling as described below, or stained with uranyl acetate and lead citrate for ultrastructural observation. Sections were examined in a JEOL JEM-1200 EX-II transmission electron microscope operated at 60 kV.
Immunocytochemistry
The postembedding protein A-gold method was used for immunolabeling (reviewed in Bendayan 1995). Briefly, sections of Epon-embedded osmicated tooth samples were first treated with sodium metaperiodate (Bendayan and Zollinger 1983) and then incubated with (a) a chicken egg yolk antibody against a 24-kD amelogenin (diluted 1:200; Chen et al. 1995) for 3 hr, (b) a secondary rabbit anti-chicken IgG antibody (diluted 1:2000; Sigma) for 1 hr, and (c) protein A-gold complex for 30 min. The complex was prepared as described by Bendayan (1995) using colloidal gold particles of approximately 8-nm diameter (Frens 1973). Some LR White tissue sections were also similarly incubated for amelogenin detection but without pretreatment with sodium metaperiodate. For immunodetection of albumin, sections of LR White-embedded teeth were incubated with rabbit anti-rat albumin (diluted 1:70; Cappel, Scarborough, ON, Canada) followed by protein A-gold for 30 min. Immunolabeling for ameloblastin was carried out with an antibody recognizing the intact molecule, as recently described by Nanci et al. (1998). After immunolabeling, the grids were extensively washed with PBS followed by distilled water and stained with uranyl acetate and lead citrate for observation in the transmission electron microscope. All incubations were carried out at room temperature. Controls consisted of incubations with the secondary antibody and protein A-gold or with protein A-gold alone.
Preparation of mRNA Probes
For mRNA probes, an ameloblastin DNA insert (1900 bp) (Matsuki et al. 1995) was placed in Bluescript SK vector (Stratagene; La Jolla, CA) and linearized using the restriction enzymes BamH1 and Xhol (Amersham Canada; Oakville, ON, Canada) for antisense and sense probe production, respectively. The abbreviated cDNA for the 26-kD mouse amelogenin (753 bp) (Snead et al. 1983) was inserted into the pGEM-7Zf vector (Promega; Madison, WI) and linearized with the enzymes Xba1 and pst1 (Amersham). Ameloblastin antisense and sense cRNA hybridization probes were transcribed in vitro with T7 and T3 RNA polymerase, respectively, in the presence of digoxigenin-UTP using an RNA labeling kit (Boehringer Mannheim Canada; Laval, QC, Canada). SP6 and T7 polymerases (Boehringer Mannheim) were used for production of amelogenin antisense and sense cRNA, respectively.
In Situ Hybridization
Sections of the entire hemimandible were cut at 5–10-μm thickness and mounted on aminoalkylsilane-coated glass slides (Sigma). The paraffin was removed with xylene and the sections were rinsed in PBS and treated at 37C with proteinase K (20 μg/ml; Boehringer Mannheim) for 30 min in a buffer consisting of 100 mM Tris-HCl and 50 mM EDTA, pH 8.0. After digestion, they were rinsed in 0.2% glycine, fixed with 4% paraformaldehyde in PBS for 5 min, and immersed for 10 min in 20 mM triethanolamine containing 0.5 ml concentrated acetic anhydride. The slides were then rinsed with PBS and treated with a prehybridization solution consisting of 2 × SSC (300 mM NaCl + 30 mM sodium citrate) containing 50% deionized formamide, for 60 min at 50C. Hybridization was carried out by incubating the slides overnight at 50C in a humidified chamber with 50% formamide, 2 × SSC, 1 × Denhardt's solution, 10% dextran sulfate, 500 μg/ml herring sperm DNA, and 250 μg/ml yeast tRNA containing ~0.5 ng/μl of the antisense or sense probe. After hybridization, they were washed several times with 4 × SSC. Nonhybridized transcripts were digested for 30 min at 37C with 20 μg/ml RNAse A (Boehringer Mannheim) in 500 mM NaCl, 10 mM Tris-HCl, 1 mM EDTA, pH 8.0. Digested sections were washed with decreasing concentrations of SSC (4 ×, 2 ×, 1 × and 0.1 ×), for 30 min each at 4C. The hybridized probe was then detected by incubating with a sheep anti-digoxigenin antibody conjugated to alkaline phosphatase (Boehringer Mannheim) for 2 hr at 4C. Phosphatase activity was revealed with 450 μg/ml nitroblue tetrazolium and 175 μg/ml of 5-bromo-4-chloro-3-indolyl phosphate in 100 mM Tris-HCl, pH 9.5, containing 100 mM NaCl and 50 mM MgCl2.
Results
Measurement of Metabolic Parameters
Measurements of the various metabolic parameters analyzed are summarized in Figure 1. During the last 2 weeks of the experiment, consumption of food by animals receiving the calcium-deficient diet declined to almost half that of controls. Body weight in treated animals paralleled their pattern of food intake and, at Day 28, they weighed on the average 49% less than control rats. There was no correlation between food and water intake.
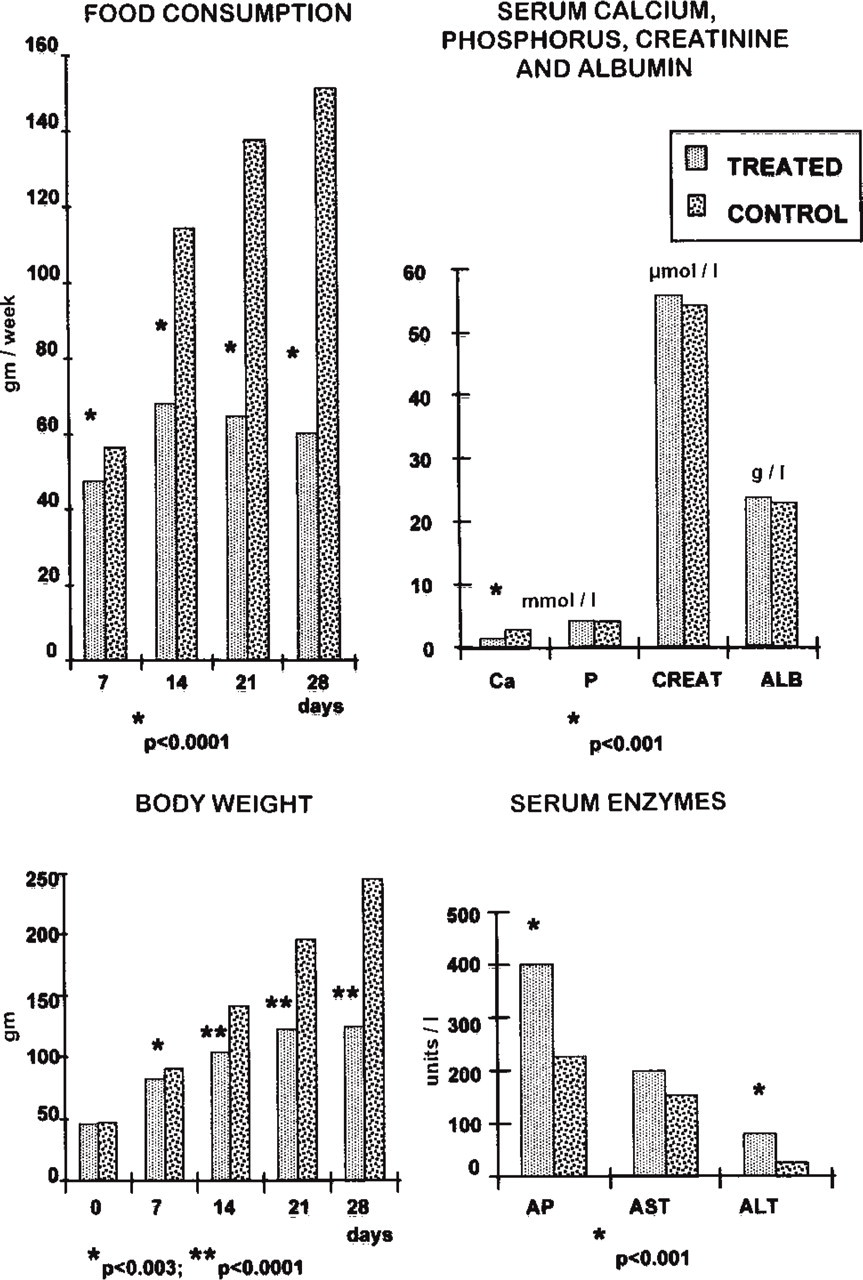
Graphs illustrating the body weight, food consumption, and serum levels of various indicators of metabolic activity over the 28-day duration of the experiment in treated and control rats. ALB, albumin; ALT, alanine transferase; Ca, calcium; CREAT, creatinine; AP, alkaline phosphatase; AST, aspartate aminotransferase; P, phosphorous.
Absence of calcium in the diet resulted in a significant reduction in serum calcium. Serum phosphorus, creatinine, albumin, and aspartate aminotransferase showed no statistical difference between the two experimental groups. Serum alkaline phosphatase and alanine transferase increased in animals receiving the calcium-deficient diet.
During the third week of the experiment, two treated rats died as a consequence of bone fractures and general debility. The surviving animals showed no distinctive physical features aside from their lesser body weight and some degree of pili erection. Behavior was not systematically assessed. However, rats were more sensitive to general animal room handling. In addition, towards the end of the diet period, the rats in the treated group were generally less active. There were no deaths in the control group.
Light Microscopy
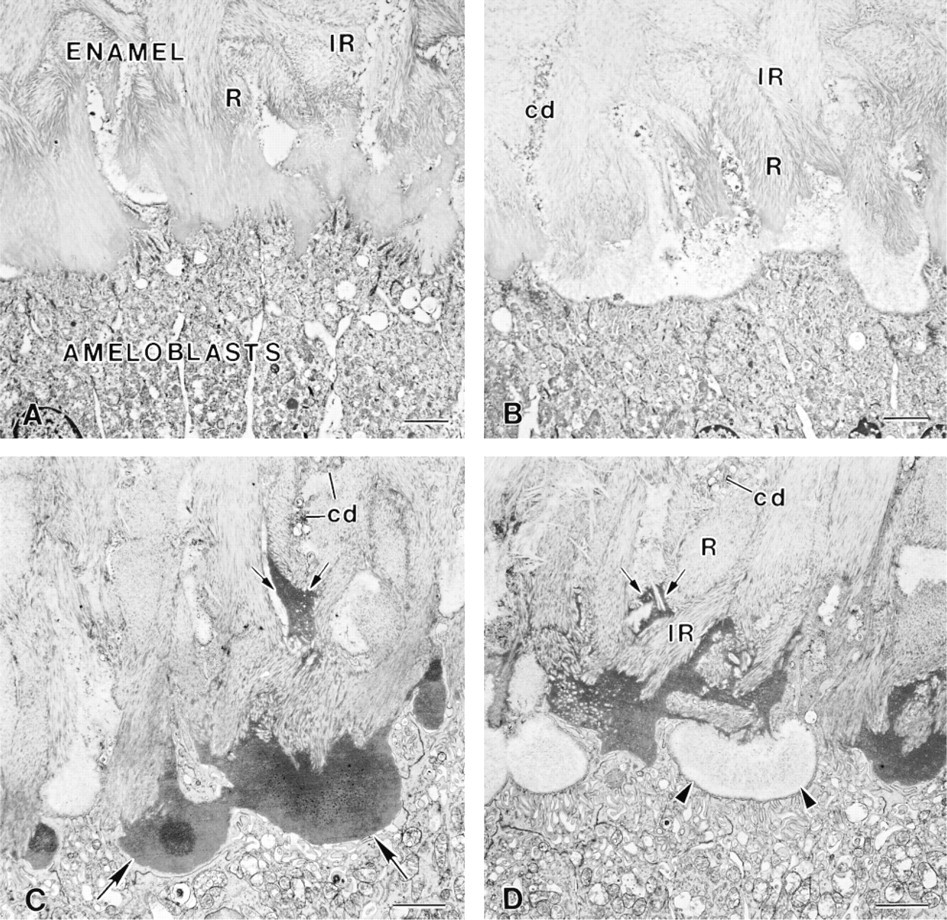
Total absence of calcium in food and water for 28 days produced significant changes in the organization of both the enamel organ and the enamel (Figures 2A-2D). Maturing enamel was hypoplasic and its surface irregular compared to that of control rats (compare Figures 2A and 2E). Alterations of enamel were most prominent in the outer portion, where there were accumulations of organic material (Figures 2B-2D). Although smooth- and ruffle-ended ameloblasts were distinguishable, the enamel organ appeared disorganized and there were regions showing accumulations of cells (Figures 2A-2C). In some regions, ameloblasts were clearly shorter and occasionally appeared cuboidal.
The enamel organ of hypocalcemic rats frequently showed the presence of cyst-like structures that varied in size and shape. Some of them contained an extracellular matrix that stained uniformly with toluidine blue, methylene blue–azure II and PASNM (Figures 2A-2C). In others, only the periphery of the matrix reacted to the stains (Figures 2A-2C). The central unstained portion was von Kossa-positive (not shown). Masses of matrix with similar texture and staining properties were also found at the enamel surface in areas where the ameloblast layer was invaginated (Figures 2A and 2B).
In control animals, PASNM staining clearly revealed the basal lamina present at the interface between the ameloblasts and enamel (Figure 2E). In hypocalcemic rats, this basal lamina was obscured by accumulations of PASNM-, PAS-, and methylene blue–azure II-positive material near the ameloblast surface (Figures 2A-2D).
In Situ Hybridization
In hypocalcemic rats, amelogenin and ameloblastin mRNAs were detected in ameloblasts, cells constituting the cyst-like structures, and groups of cells accumulating in some areas of the enamel organ (Figure 3). Papillary layer cells proper showed no signal. In control animals, only ameloblasts expressed amelogenin and ameloblastin signals. Odontoblasts and bone cells in both groups of animals were not reactive. Control incubations with sense probes resulted in no significant staining.
Electron Microscopy
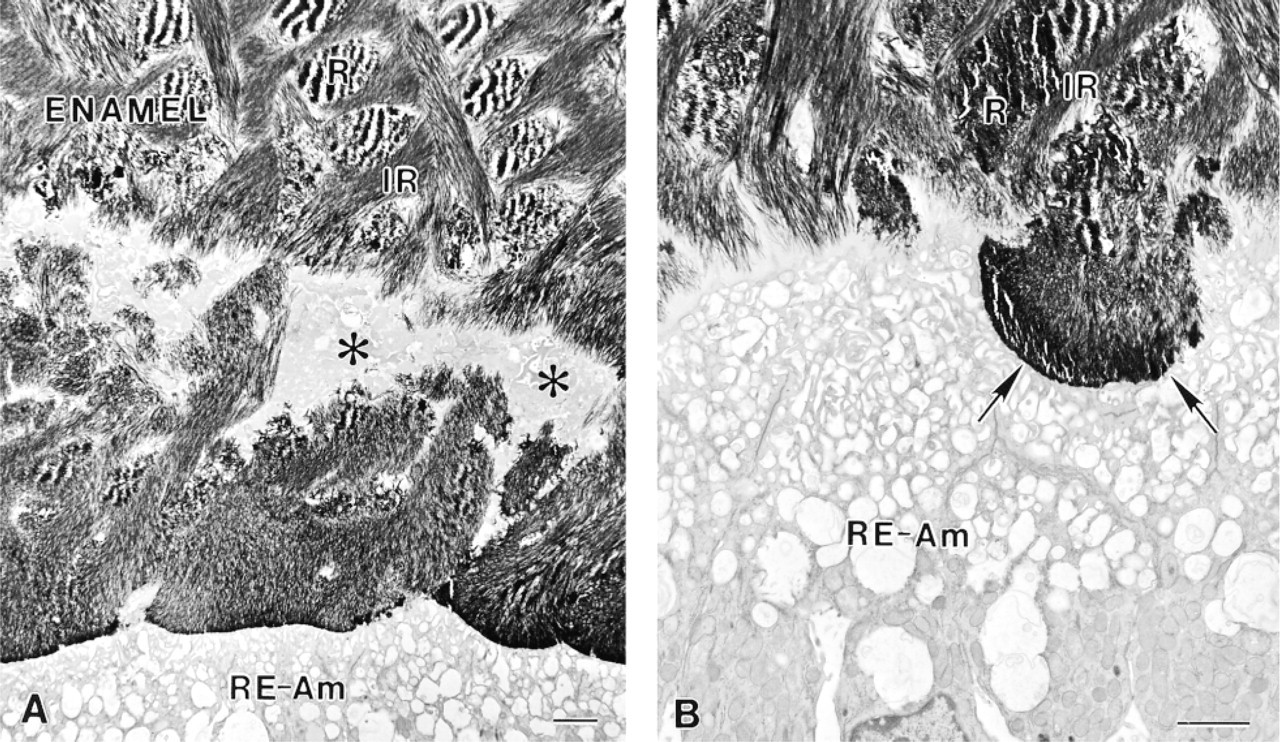
Except in areas where there were cyst-like structures in the enamel organ, ameloblasts in hypocalcemic rats showed a normal intracellular organization. They contained well-developed protein synthetic organelles, endosomal/lysosomal elements, and occasionally secretory granules (Figure 4). However, the enamel layer was hypoplasic (Figure 5). Rod and inter-rod enamel were generally distinguishable in the deeper portions of the enamel layer but not near the surface (Figures 5 and 6). At this site, crystal packing was disorganized, there were accumulations of organic material among the crystals, and cell processes and remnants of Tome's processes were frequently encountered (Figures 5 and 7). Small vesicular profiles were present between crystals and around ameloblast processes (Figure 8A). The enamel did not always extend up to the ameloblasts; a layer poor in organic matrix or patches of amorphous material were sometimes found at the ameloblast surface (Figures 5, 7D and 9). In areas where there was no basal lamina at the interface between the ameloblasts and the enamel, enamel crystals or tubular crystal “ghosts” (in decalcified specimens; see Leblond and Warshawsky 1979; Bonucci 1984) extended into the apical membrane infoldings of ruffle-ended ameloblasts or shallow invaginations at the apex of smooth-ended ameloblasts (Figure 8).
The cyst-like structures in the enamel organ were generally formed by cells with ameloblast characteristics, often showing a ruffled border (Figure 10) and less frequently smooth ones (Figure 11B). Processes extended from the cells into the matrix they englobed; in some cases, a basal lamina was present between the cells and the matrix (Figures 10A and 10C). Tubular “crystal ghosts” were sometimes identifiable in the matrix, whereas in other cases it appeared finely granular or homogeneous (Figures 10 and 11B). In decalcified teeth, plate-like “ghosts” like those seen in decalcified bone (Bonucci 1984) were present in the electron-lucent core of some cyst-like structures (Figure 10B). Similarly to the matrix of cyst-like structures, masses of material exhibiting a variety of appearances were found in surface invaginations of the ameloblast layer (Figures 5C, 7C, and 9C). Other masses of organic matrix, with a finely granular or homogeneous texture, were also present along the lateral surfaces of ameloblasts and among cells of the papillary layer (Figure 12).
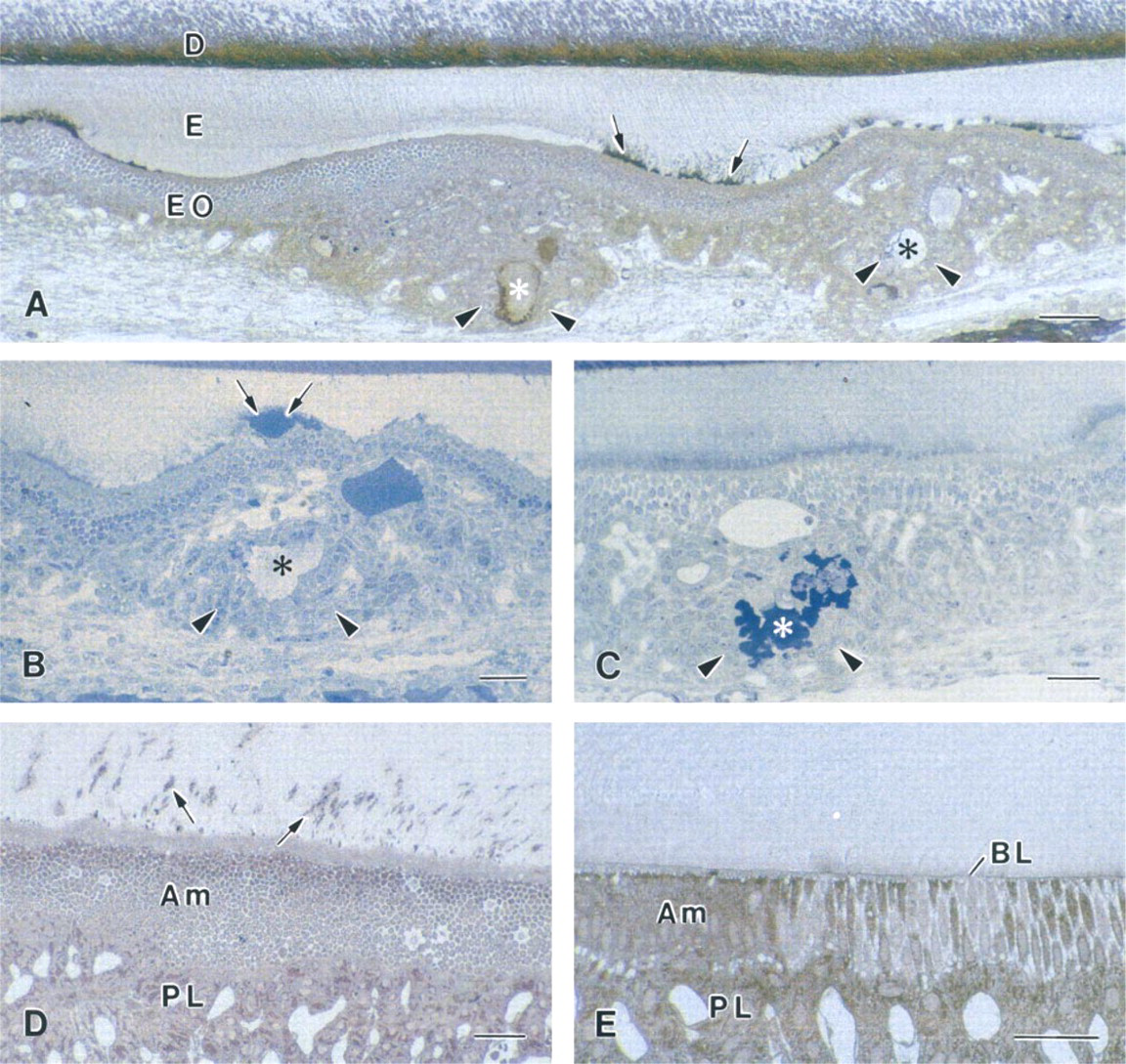
Histochemical preparations of the enamel organ (EO) and enamel (E) from calcium-deficient (
None of the above altered cell and matrix characteristics was observed in control animals (data not shown).
Immunolocalization of Ameloblastin, Amelogenin, and Albumin
Early maturation stage ameloblasts from control rats showed labeling for ameloblastin and amelogenin over organelles involved in protein synthesis and secretion, and over endosomal/lysosomal elements. Immunore-activity for amelogenin was found throughout the enamel layer. There was a concentration of intact ameloblastin near the cell surface and labeling for albumin was weak and mostly restricted to the superficial portion of the enamel layer. These immunolabeling results in the control group are similar to what has been extensively documented for normal animals (Nanci et al. 1987, 1996, 1998).
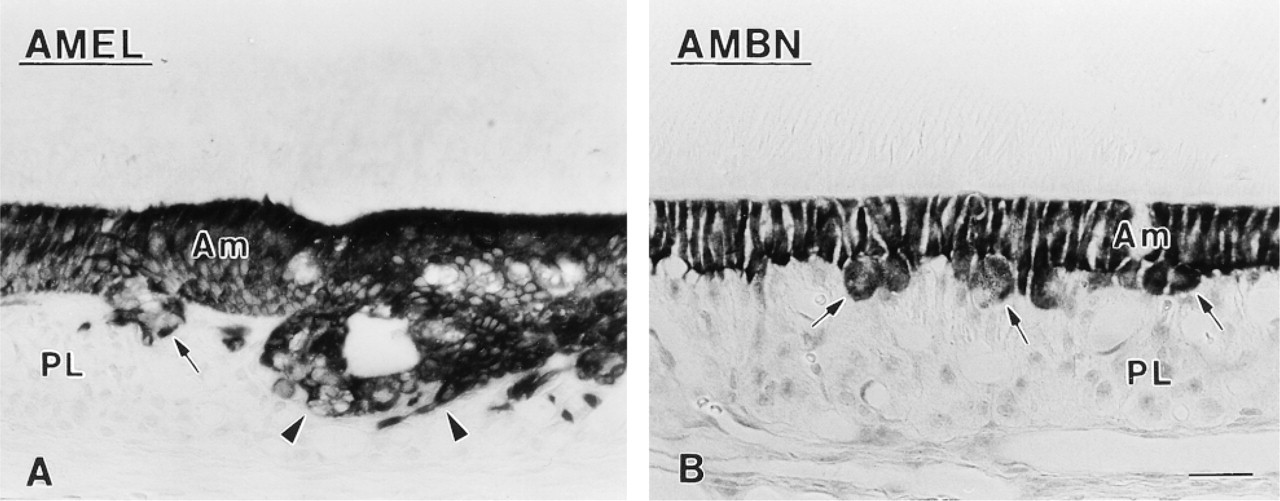
In situ hybridization reactions with antisense probes to (
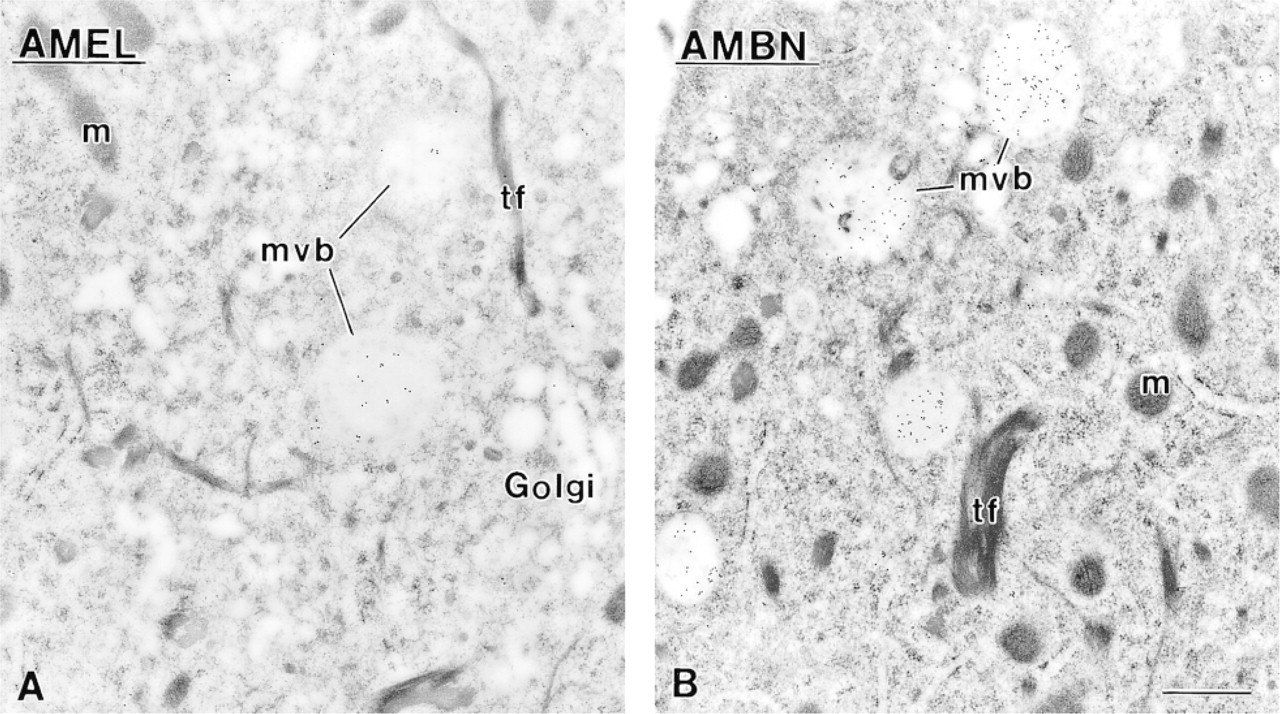
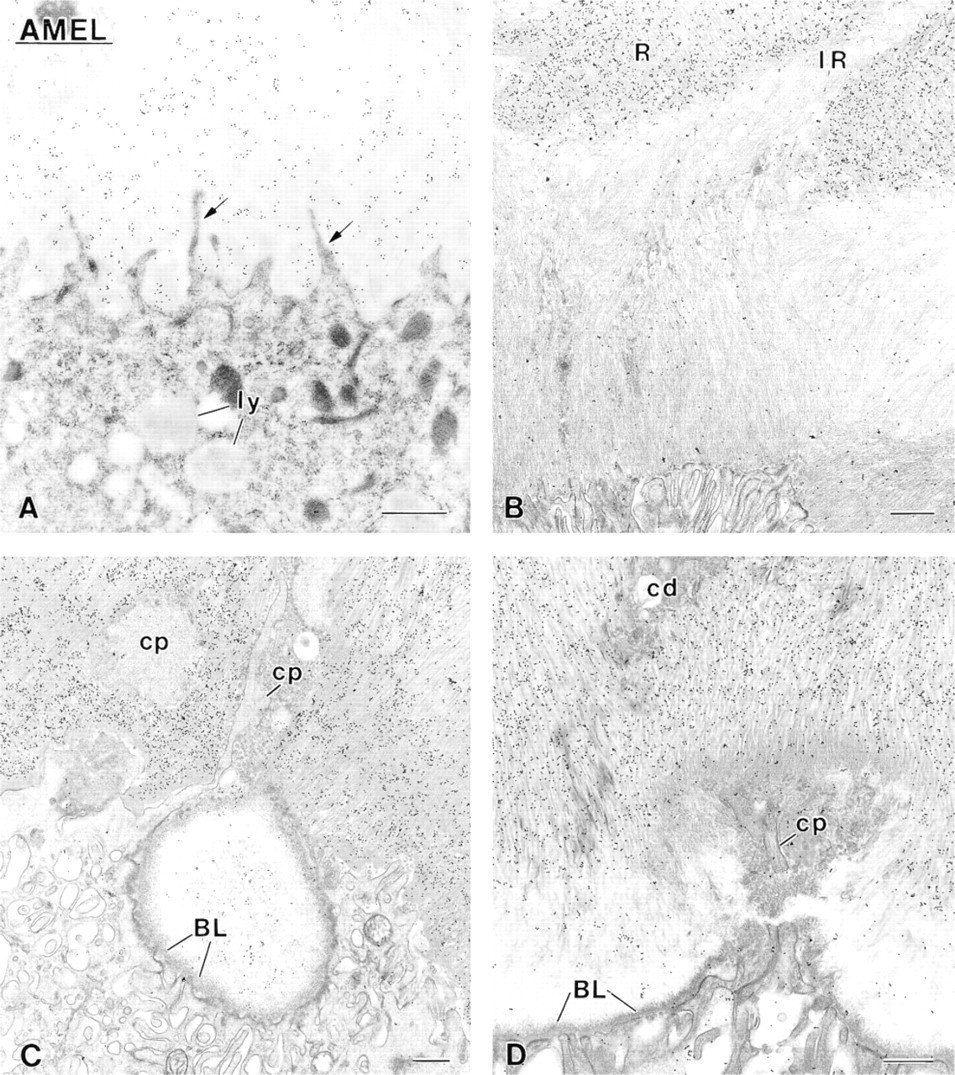
In ameloblasts of hypocalcemic rats, the organelles involved in protein synthesis and secretion as well as multivesicular bodies were consistently labeled for ameloblastin (Figure 4B). The intracellular reactivity for amelogenin was variable and was lower than in controls, particularly for multivesicular bodies, whose frequency and density of labeling were both reduced (Figure 4A). Labeling for ameloblastin and amelogenin was heterogeneously distributed throughout the maturing enamel layer (Figures 7 and 9). There were regions of intense immunoreactivity alternating with weakly-labeled ones (Figures 7 and 9). Immunolabeling for albumin was clearly more intense than in control rats and also exhibited a heterogeneous distribution (Figure 11A). The accumulations of organic matrix in the superficial portion of the enamel layer were variably immunoreactive (Figures 9A-9C), whereas the content of the cyst-like structures labeled for all three proteins (Figures 10B, 10C, and 11B). Large patches of organic matrix were sometimes found along the basolateral surface of ameloblasts and among cells of the papillary layer. These were intensely immunoreactivity for amelogenin but showed very little or no labeling for ameloblastin (Figure 12).
Immunocytochemical preparations illustrating the presence of (
(
(
Control incubations resulted in few gold particles, randomly distributed throughout the entire tissue section (data not shown).
Discussion
This report shows that diet-induced hypocalcemia directly or indirectly results in major alterations of both the enamel organ and the enamel. The results are consistent with previous studies on experimentally induced chronic hypocalcemia suggesting that low calcium levels, rather than phosphate levels, cause enamel hypoplasia (Nikiforuk and Fraser 1979; Limeback et al. 1992; Acevedo et al. 1996). In the same experimental model as in the present study, it was previously found that diet-induced hypocalcemia results in hypocalcified enamel (Bonucci et al. 1994). This abnormal mineralization is, at least in part, due to abnormal levels and distributions of indigenous matrix proteins and the accumulation in enamel of the serum protein albumin.
The organic matrix of enamel comprises two classes of proteins, amelogenins and nonamelogenins. Amelogenins, the dominant component of developing enamel, undergo bulk enzymatic breakdown and removal during enamel formation to accommodate the growth in width and thickness of enamel crystals (reviewed in Deutsch et al. 1995; Nanci and Smith 1992, 2000; Robinson et al. 1995; Smith 1998). The heterogeneous but intense immunolabeling for amelogenin indicates that this process is affected by calcium deficiency. Ameloblastin, a non-amelogenin, normally accumulates near the cell surface and undergoes rapid extracellular processing into lower molecular weight forms (Nanci et al. 1998). Because the antibody we have used recognizes the intact molecule (discussed in Nanci et al. 1998), the presence of areas of intense immunolabeling for ameloblastin throughout the enamel layer demonstrates that processing of this non-amelogenin was also hampered. Therefore, protein degradation and removal do not take place normally in incisors of hypocalcemic rats. It is likely that calcium deprivation affects the secretion, activation, and/or activity of the various enzymes believed to be active during enamel formation (DenBesten et al. 1989; Bartlett et al. 1996; Robinson et al. 1997; Smith 1998).
Ameloblasts do not synthesize and secrete albumin (Couwenhoven et al. 1989; Yuan et al. 1996), and this exogenous plasma protein has limited access to the enamel layer in normal rats (McKee et al. 1986; Robinson et al. 1994; Chen et al. 1995; Nanci et al. 1996). The intense immunolabeling for albumin in maturing enamel of hypocalcemic rats suggests that the integrity and permeability of the enamel organ are affected by low calcium levels, a conclusion consistent with the observed alterations in its organization. Together with the abnormal removal of enamel proteins, the accumulation of albumin is likely responsible for the observed structural defects in enamel. Indeed, albumin has been associated with the presence of hypoplastic lesions in enamel (Robinson et al. 1992). The enamel organization appeared less affected in areas where the basal lamina separating ameloblasts and enamel was still present, suggesting that it may play an important role in the maturation process. This interfacial structure is rich in glycoconjugates and, like the glomerular basement membrane, may have filtering functions, selectively controlling what goes in and out of the enamel layer during enamel maturation (Nanci et al. 1993). Therefore, its partial absence may contribute to the abnormal influx of albumin into the enamel layer.
Immunocytochemical preparations with anti-amelogenin (AMEL) antibody, illustrating the variable pattern and density of labeling for this matrix protein over enamel in incisors from hypocalcemic rats. (
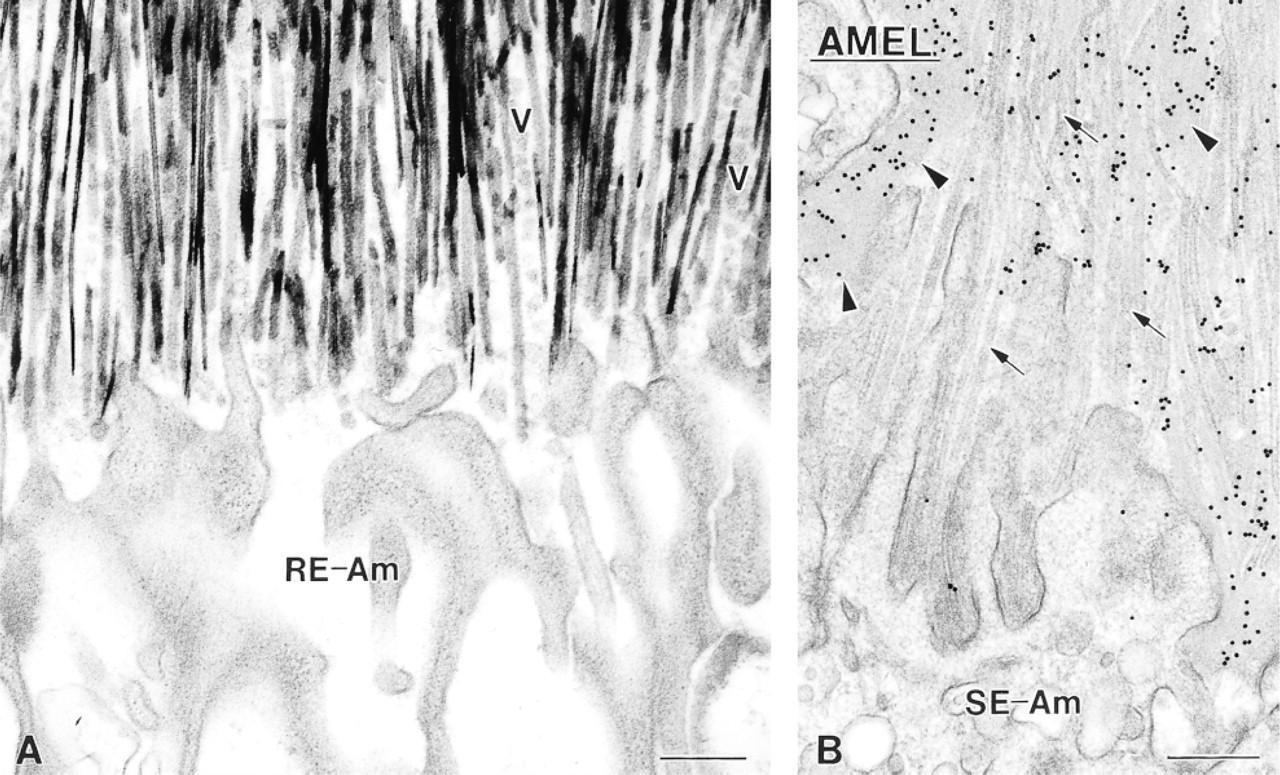
Electron micrographs from hypocalcemic rats illustrating at high magnification the interface between enamel and ameloblasts in areas where there is no basal lamina. (
The detection of mRNA signal for amelogenin and ameloblastin in the cells forming cyst-like structures and clusters in the enamel organ of calcium-deficient rats strongly suggests that they are of ameloblastic origin. Indeed, the matrix within the cysts comprises both amelogenin and ameloblastin and mineralizes. One possible mechanism for their formation is an interference with the cell cycle program of ameloblasts. Calcium is known to regulate the cell cycle (Poyner 1994) and to act as a triggering mechanism for apoptosis (reviewed in McConkey and Orrenius 1997). In the rat incisor, as many as 25% of ameloblasts die during postsecretory transition and another 25% are gradually lost during the remainder of the maturation stage (Smith and Warshawsky 1977; Smith and Nanci 1995). The mechanism for the reduction of the ameloblast population is still not clear, although a number of studies suggest that it is largely dependent on apoptosis (Smid et al. 1992; Dodds et al. 1993; Joseph et al. 1994; Nishikawa and Sasaki 1995, 1996). The dramatic drop in serum calcium level during chronic hypocalcemia may affect its availability to ameloblasts and may inhibit their programmed death. To accommodate the excess number of cells, the ameloblast layer could buckle into itself, resulting in an apparent accumulation of cells and the formation of cyst-like structures in the enamel organ. Consistent with the capacity of early maturation stage ameloblasts to produce enamel proteins (Nanci et al. 1998), the cells forming the cyst-like structures release secretory products, such as ameloblastin and amelogenin, which favor ectopic mineral deposition in the space they delimit. Cystlike structures have also been observed in cases of fluoride intoxication (Mornstad and Hammarstrom 1978; Lange Nordlund et al. 1986; Simmelink and Lange 1986) and tetracycline administration (Westergaard and Nylen 1975; Kallenbach 1980; Westergaard 1980).
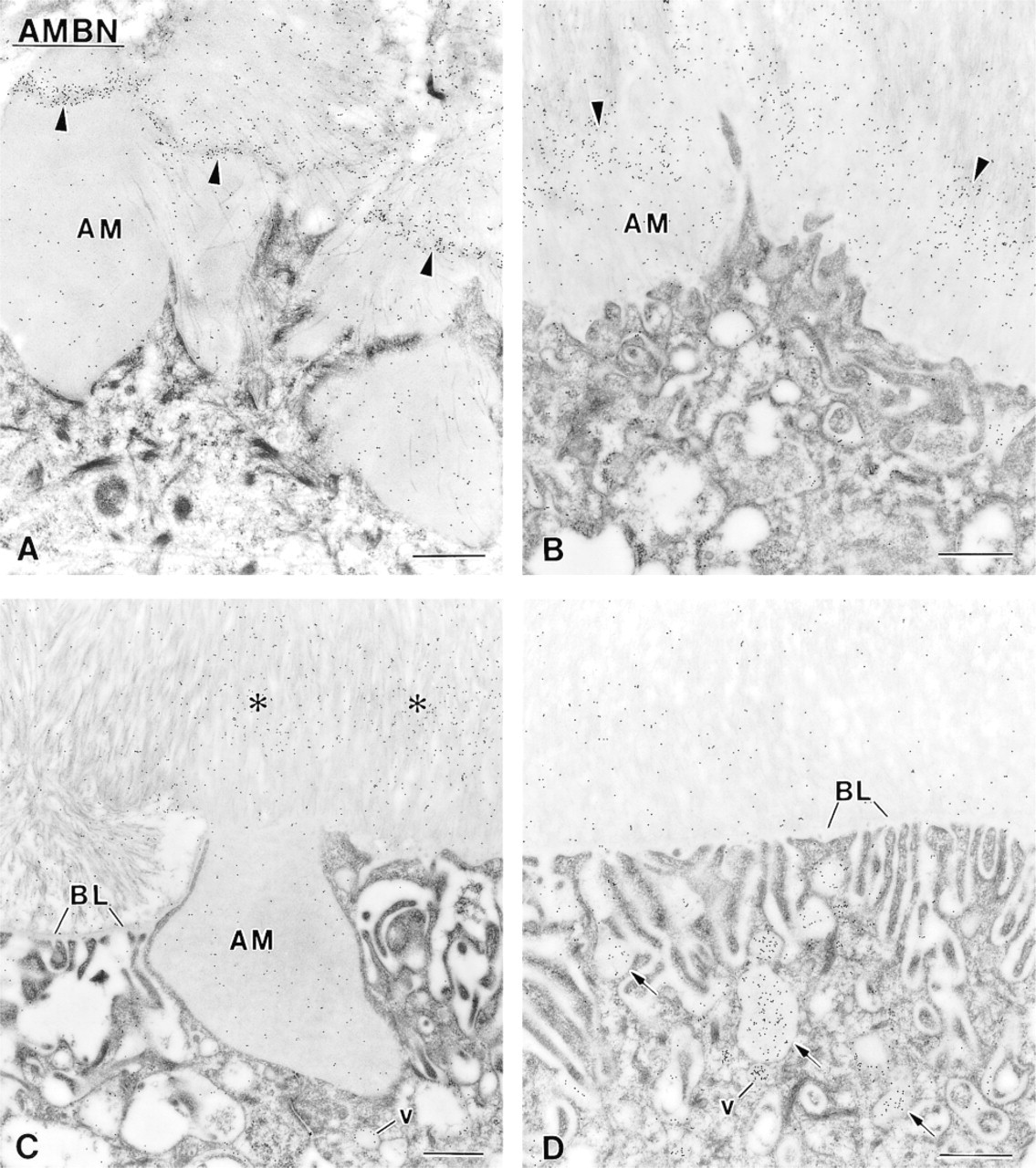
Immunocytochemical preparations with anti-ameloblastin (AMBN) antibody illustrating the labeling observed over the superficial enamel. This portion of the layer shows a variable appearance and pattern of labeling. (
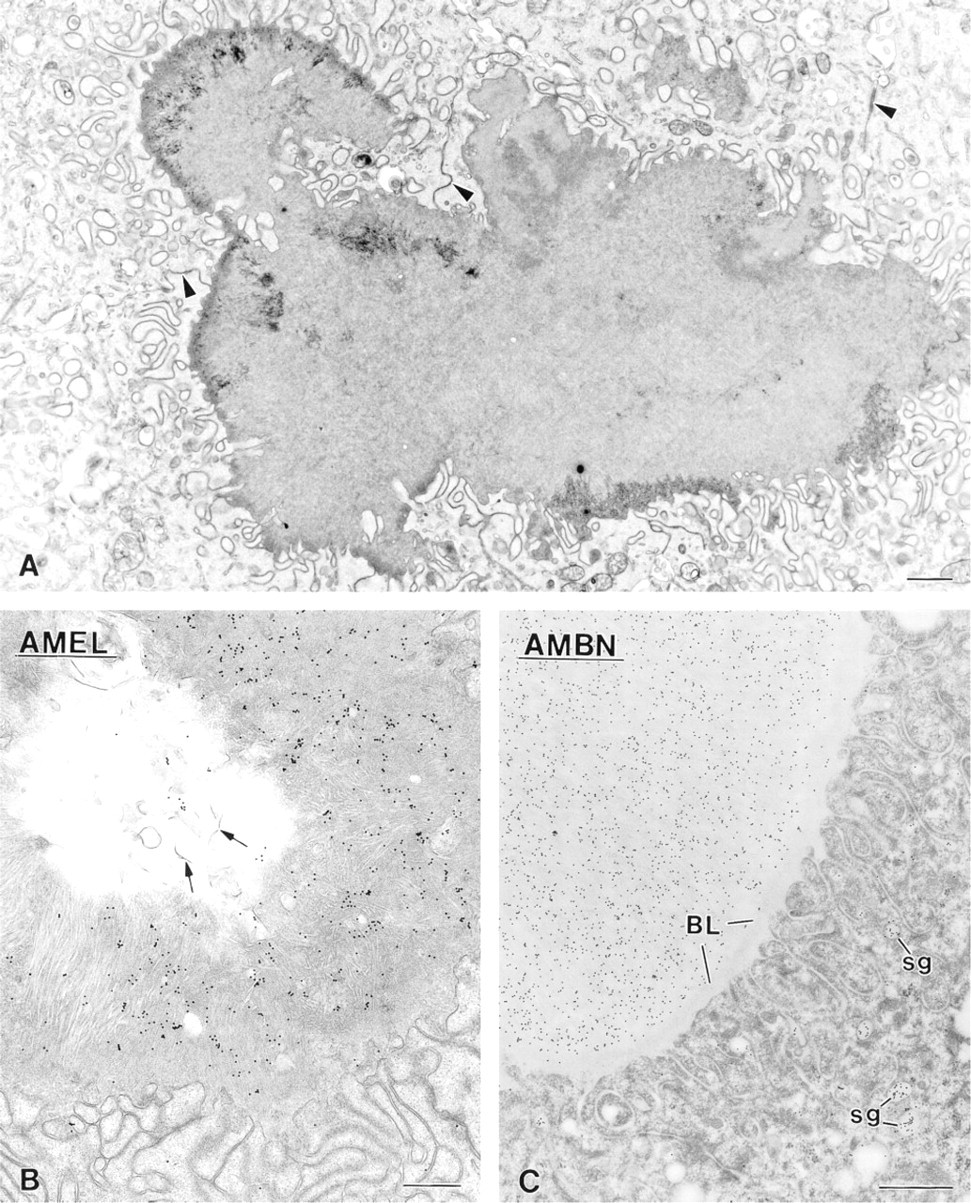
(
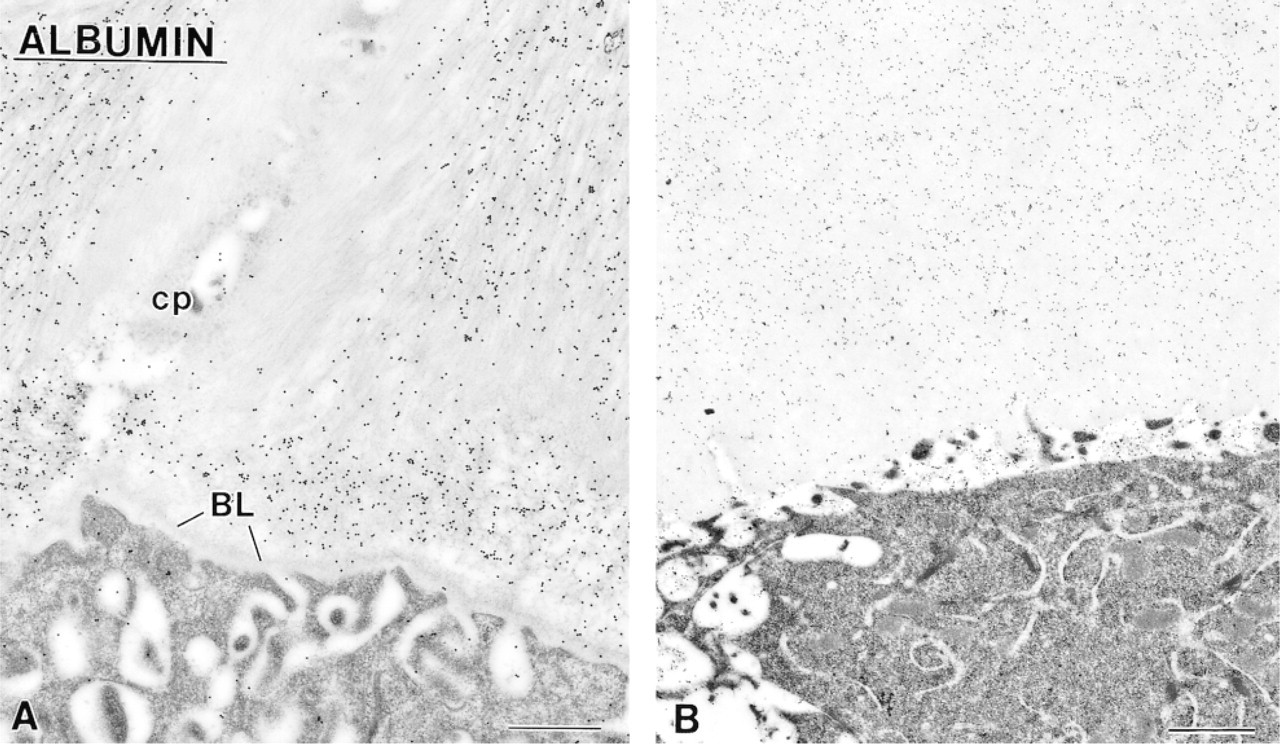
Immunocytochemical preparations for albumin. There is intense labeling for this serum protein over both (
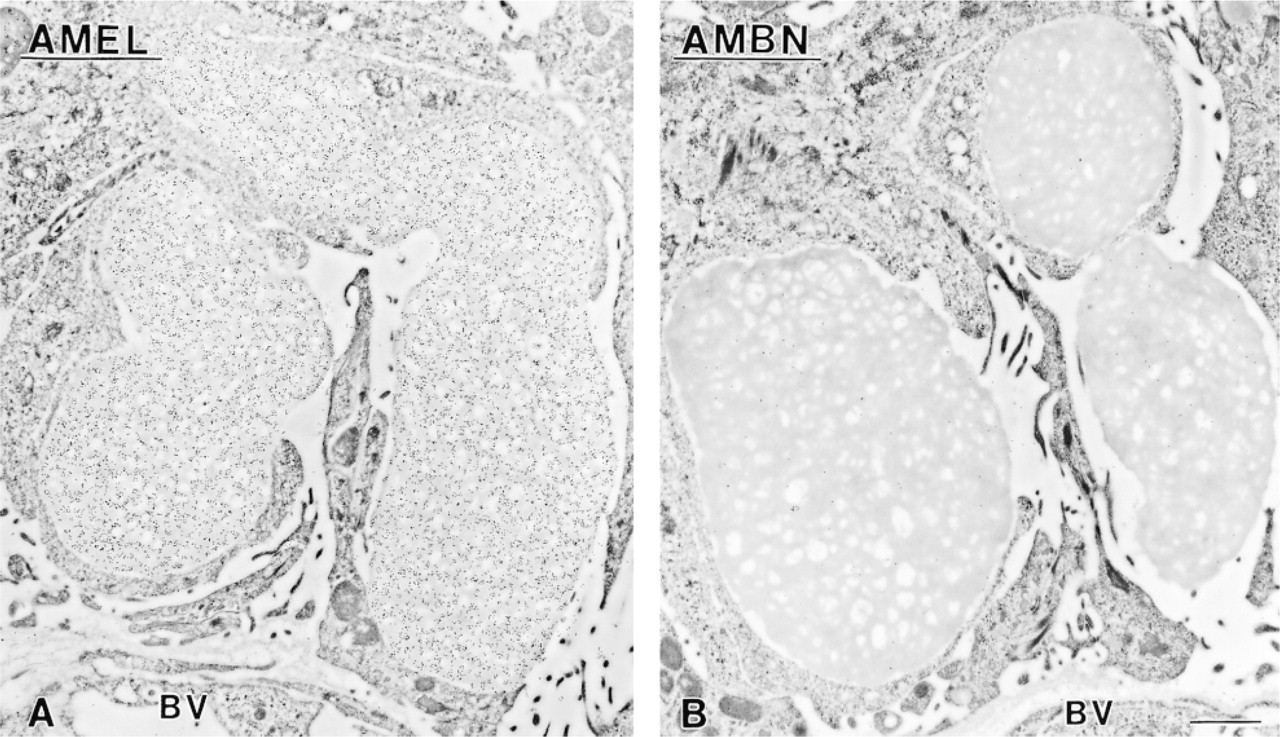
Patches of organic matrix found along the basolateral surface of ameloblasts and among cells of the papillary layer are (
Among the various enamel proteins, only nonamelogenins are believed to show an important degree of glycosylation (reviewed in Smith and Nanci 1996). The intense staining for glycoproteins at the enamel surface may therefore represent the accumulation of nonamelogenins, most likely breakdown products of molecules such as ameloblastin. Their accumulation would not be detected by the anti-ameloblastin antibody used because it recognizes only the intact molecule (discussed in Nanci et al. 1998). One interesting observation is the inverse correlation between the degree of histochemical staining for glycoproteins and von Kossa staining for mineral in the cyst-like structures. Clearly, as calcification progresses glycoproteinaceous components are lost and/or their reacting groups are masked or altered by mineralization. Such a finding is consistent with the transient nature of nonamelogenins and their proposed role in promoting/stabilizing crystal elongation, a temporally restricted event (Nanci et al. 1998; Nanci and Smith 2000).
In early maturation stage ameloblasts from normal rat incisors, elements of the endosomal/lysosomal system, such as multivesicular bodies, are, in general, intensely immunoreactive for amelogenin and ameloblastin (see Nanci et al. 1987, 1998). In hypocalcemic rats, multivesicular bodies show ameloblastin immunoreactivity but these endosomal/lysosomal structures are inconsistently and weakly labeled for amelogenin. The possibility that hypocalcemia could selectively affect just one of the two classes of enamel proteins is very intriguing. In attempting to determine the significance of this observation, one must first identify the origin of the immunoreactive material in multivesicular bodies. It may derive either from the intracellular rerouting of secretory products before their secretion and/or their endocytosis along the apical and basolateral surfaces of ameloblasts (discussed in Nanci and Smith 1992). It is likely that both synthesis and endocytosis would be implicated.
In conclusion, chronic hypocalcemia affects the physiological events that regulate enamel maturation. The enamel organ normally provides a “sealed” and controlled microenvironment for these events. Its disruption therefore represents a major cause for the impairment of mineralization. Protein processing and removal are slowed and the enamel layer is actually flooded by exogenous proteins such as albumin, thereby limiting crystal growth. This situation differs from what happens in bone during chronic hypocalcemia, in which calcium availability may have an important and direct effect on mineral deposition (see Mocetti et al. 2000).
Footnotes
Acknowledgments
Supported by grants from the Medical Research Council of Canada and the Ministry of University and Scientific and Technological Research (MURST) of Italy. P. Mocetti was the recipient of a fellowship from the Québec Ministry of Education.
We are grateful to Dr R. Lepage [Pavillon St-Luc, Centre Hospitallier de l'Université de Montréal (CHUM) Montreal, QC] for carrying out the biochemical assays of blood samples, to Dr Paul H. Krebsbach (School of Dentistry, University of Michigan, Ann Arbor, MI) for providing the anti-ameloblastin antibody, to Micheline Fortin for diligent technical and photographic assistance, and to Sylvia Zalzal for assistance with the supervison of the experimentation.
