Abstract
After crown formation, the enamel organ reorganizes into Hertwig's epithelial root sheath (HERS). Although it is generally accepted that HERS plays an inductive role during root formation, it also has been suggested that it may contribute enamel-related proteins to cementum matrix. By analogy to the enamel-free area (EFA) in rat molars, in which epithelial cells express not only enamel proteins but also “typical” mesenchymal matrix constituents, it has been proposed that HERS cells may also have the potential to produce cementum proteins. To test this hypothesis, we examined the nature of the first matrix layer deposited along the cervical portion of root dentin and the characteristics of the associated cells. Rat molars were processed for postembedding colloidal gold immunolabeling with antibodies to amelogenin (AMEL), ameloblastin (AMBN), bone sialoprotein (BSP), and osteopontin (OPN). To minimize the possibility of false-negative results, several antibodies to AMEL were used. The labelings were compared with those obtained at the EFA. Initial cementum matrix was consistently observed at a time when epithelial cells from HERS covered most of the forming root surface. Cells with mesenchymal characteristics were rarely seen in proximity to the matrix. Both the EFA matrix and initial cementum exhibited collagen fibrils and were intensely immunoreactive for BSP and OPN. AMEL and AMBN were immunodetected at the EFA but not over the initial cementum proper. These two proteins were, however, present at the cervical-most portion of the root where enamel matrix extends for a short distance between dentin and cementum. These data suggest that epithelial cells along the root surface are likely responsible for the deposition of the initial cementum matrix and therefore, like the cells at the EFA, may be capable of producing mesenchymal proteins.
Keywords
T
A knowledge of the nature and the origin of the first deposited matrix constituents along root dentin is of importance to elucidate the function of HERS in cementogenesis. In some animals, this matrix is believed to be formed in the presence of epithelial cells (Paynter and Pudy 1958; Slavkin 1976; Owens 1978,1980; Lindskog and Hammarström 1982; Glazman et al. 1986; Slavkin et al. 1988,1989; reviewed in Bosshardt and Schroeder 1996). Consequently, enamel-related proteins have been proposed as possible candidates (Slavkin 1976; Lindskog and Hammarström 1982; Glazman et al. 1986; Slavkin et al. 1988,1989; Luo et al. 1991). Because HERS cells do not appear to express mRNA for amelogenin (AMEL), the major enamel protein (Luo et al. 1991), interest has recently shifted to amelin/ameloblastin (AMBN), a novel enamel protein (Fong et al. 1996; Krebsbach et al. 1996). Although amelin mRNA is found in some cells scattered along the root (Fong et al. 1996), the presence and distribution of this protein in the extracellular matrices along the root are unknown.
Other studies have shown that, at a time coincident with the initiation of cementogenesis, the root surface stains for bone sialoprotein (BSP) and osteopontin (OPN) (Somerman et al. 1990,1992; Bronckers et al. 1994; MacNeil et al. 1994), two typical noncollagenous proteins of cementum and other collagen-based mineralized tissues (reviewed in Butler and Ritchie 1995; McKee et al. 1996; Bosshardt and Selvig 1997). It has been demonstrated that the root-lining cells express mRNA for these two noncollagenous proteins (MacNeil et al. 1996; Sommer et al. 1996; D'Errico et al. 1997). However, it could not be determined whether these cells were derived from the dental follicle or from HERS. The problem of cell origin and identification does not exist at the enamel-free area (EFA), where a matrix layer consisting of both enamel and cementum proteins forms in the exclusive presence of epithelial cells (Bosshardt and Nanci 1997). Therefore, it has been proposed that the IEE at the EFA and along the root may share a similar developmental program and that in some species epithelial cells may also be responsible for the deposition of typical cementum proteins along the root (Bosshardt and Nanci 1997). To test this hypothesis, we have examined the nature of matrix constituents along the cervical root and the characteristics of the cells associated with them. Their presence at the EFA in rat molars was used to further support their epithelial origin.
Materials and Methods
Tissue Preparation
Wistar rats (Charles River; St.-Constant, QC, Canada) at 1, 3, 5, 11, and 14 days after birth were used in this study. A minimum of three rats per interval were analyzed. The animals were anesthetized with chloral hydrate (0.4 mg/g body weight) and perfused for 30 sec through the left ventricle with lactated Ringer's solution (Abbott Laboratories; Montréal, QC, Canada), followed by fixative consisting of 1% glutaraldehyde in 0.08 M sodium cacodylate buffer containing 0.05% calcium chloride (pH 7.3). The lower and upper jaws were dissected and immersed in the same fixative for an additional 12 hr at 4C. Most specimens were decalcified at 4C in 4.13% disodium ethylenediaminetetraacetic acid (EDTA) for 14 days (Warshawsky and Moore 1967). In the case of 1–5-day-old rats, some tissue specimens were not decalcified. The hemimandibles were cut into segments containing the first, second, or third molars. These segments were further subdivided sagittally into two or three thin slices. After extensive washing in 0.1 M sodium cacodylate buffer containing 5% sucrose, pH 7.3, some samples were postfixed with potassium ferrocyanide-reduced osmium tetroxide (Neiss 1984) and processed for embedding in Taab 812 epoxy resin (Marivac; Halifax, NS, Canada). Unosmicated samples were embedded in LR White resin (Mecalab; Montreal, QC, Canada). Semithin survey sections (1 μm thick) were cut with glass knives on a Reichert Ultracut E microtome, stained with toluidine blue, and observed by light microscopy. Thin (80–100-nm) sections at selected block areas were cut with a diamond knife, contrasted with uranyl acetate and lead citrate, and examined in a JEOL JEM 2000FX-II transmission electron microscope operated at an accelerating voltage of 80 kV.
Immunocytochemistry
The high-resolution protein A–gold technique (reviewed in Bendayan 1995) was used for immunocytochemical localization of enamel- and cementum-related extracellular matrix proteins. All incubations were performed at room temperature. Thin sections were mounted on formvar- and carbon-coated nickel grids. Sections from LR White-embedded tissues were floated for 10 min on a drop of 0.01 M PBS, pH 7.3, containing 1% ovalbumin (Sigma Chemical; St Louis, MO) to saturate nonspecific binding sites. The grid-mounted sections were then transferred and incubated for 1 hr on a drop of one of the following antibodies: (a) rabbit anti-rat bone sialoprotein (LF-87) diluted 1:10 with PBS (courtesy of Dr. L.W. Fisher, National Institutes of Health, Bethesda, MD) (Midura et al. 1990; Fisher et al. 1995); (b) rabbit anti-mouse AMEL diluted 1:20 with PBS (courtesy of Dr. H.C. Slavkin, Center for Craniofacial Molecular Biology, University of Southern California, Los Angeles, CA) (Slavkin et al. 1982); or (c) rabbit anti-rat AMBN diluted 1:20 with PBS (courtesy of Dr. P.H. Krebsbach, National Institutes of Health, Bethesda, MD) (Lee et al. 1996).
Sections of osmicated tissues embedded in Epon were first floated on a drop of saturated solution of sodium metaperiodate for 15 min and washed with distilled water. The epoxy sections were then incubated for 3 hr on a drop of egg yolk chicken anti-rat AMEL antibody diluted 1:150 with PBS (Chen et al. 1995), or sheep anti-porcine Bio-Gel peak F and C affinity-purified AMEL antibodies diluted 1:10 and 1:100, respectively, with PBS (courtesy of Dr. H. Limeback, Faculty of Dentistry, University of Toronto, ON, Canada) (Limeback and Simic 1990). For immunolocalizations of OPN, LR White sections were similarly incubated with a chicken anti-rat OPN antibody diluted 1:150 with PBS (Nanci et al. 1996). Thereafter, all sections were washed with PBS, floated again on PBS-1% ovalbumin for 10 min, and incubated for 1 hr on a drop of the corresponding polyclonal rabbit anti-chicken IgG (Cappel; Scarborough, ON, Canada) or anti-sheep IgG (Cappel) secondary antibody.
After incubations with primary or secondary antibody, the grids were rinsed with PBS, floated on PBS-1% ovalbumin for 10 min, and incubated with protein A-gold complexes prepared using gold particles of approximately 8 or 12 nm in diameter (Frens 1973). After rinsing with PBS and distilled water, the sections were grid-stained with uranyl acetate and lead citrate and examined in the transmission electron microscope. For comparison purposes, some consecutive sections were incubated with the various antibodies. As controls, sections were incubated with protein A–gold alone, nonimmune serum, or unrelated antibodies. The differential labelings obtained with the various antibodies also served as internal controls.
Results
Antibodies to AMEL and AMBN labeled the enamel matrix, and those against BSP and OPN the cementum proper and bone. The labeling pattern obtained with the various anti-AMEL antibodies used did not show any significant differences. As revealed in adjacent sections, gold particle labeling for BSP and OPN occupied the same matrix regions. Control incubations resulted in few, randomly distributed gold particles over the tissue section.
Enamel-free Area
The EFA matrix was lined throughout crown development by inner enamel epithelial (IEE) cells, as indicated by the presence of tonofilaments and (hemi)des-mosomes (Figures 1A, 1B, 2A, and 2B). Occasionally, irregular globular masses of organic matrix (Figures 1B and 2A) containing barely recognizable collagen fibrils (Figure 2A) extended between the epithelial cells in 14-day-old rats. In younger animals (Days 1–5), on the other hand, only uncalcified collagen fibrils were visible among the epithelial cells (Figure 2B). The EFA matrix was immunoreactive for AMEL (Figure 1A), AMBN (Figures 1B and 2B), BSP (not shown), and OPN (Figure 2A).
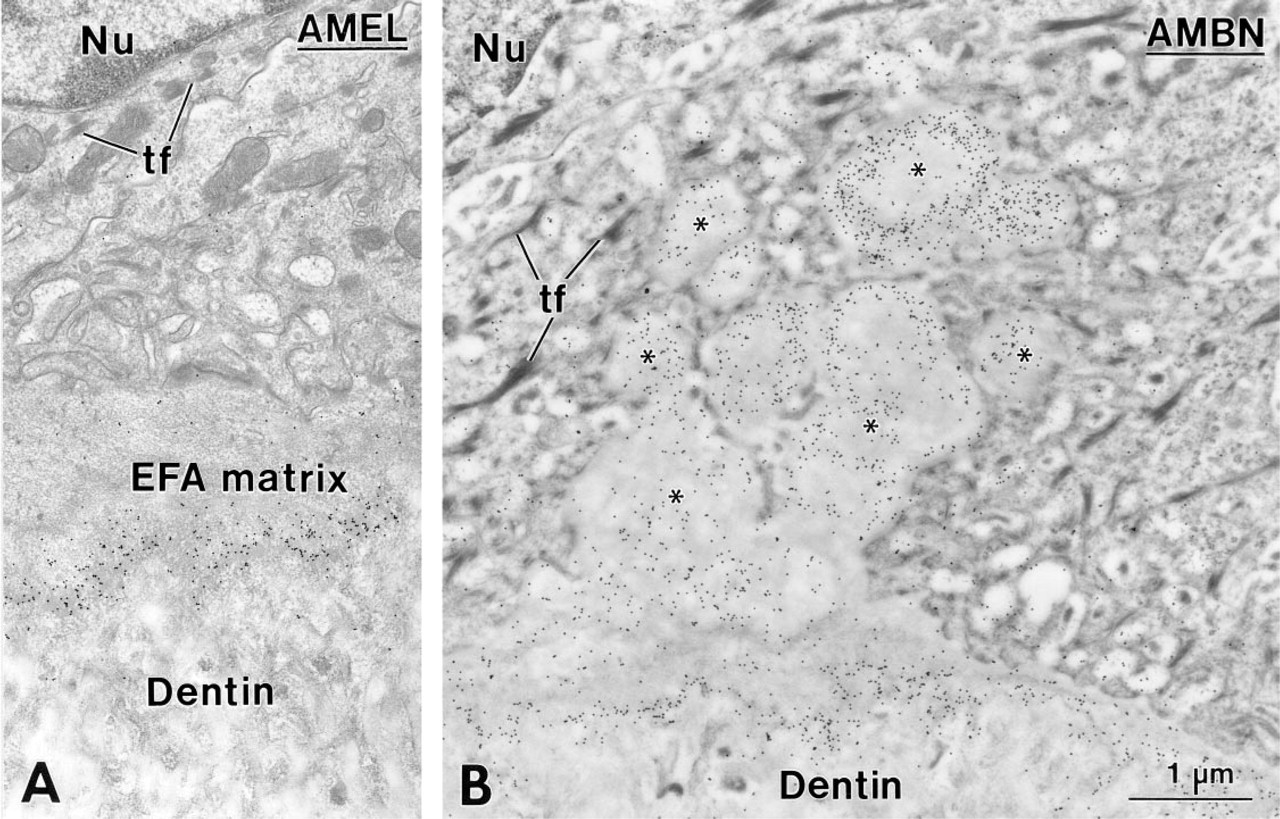
Enamel-free area (EFA) of the mesial cusp tip in a first mandibular molar from a 14-day-old rat. The EFA matrix is interposed between dentin and the inner enamel epithelial cells (
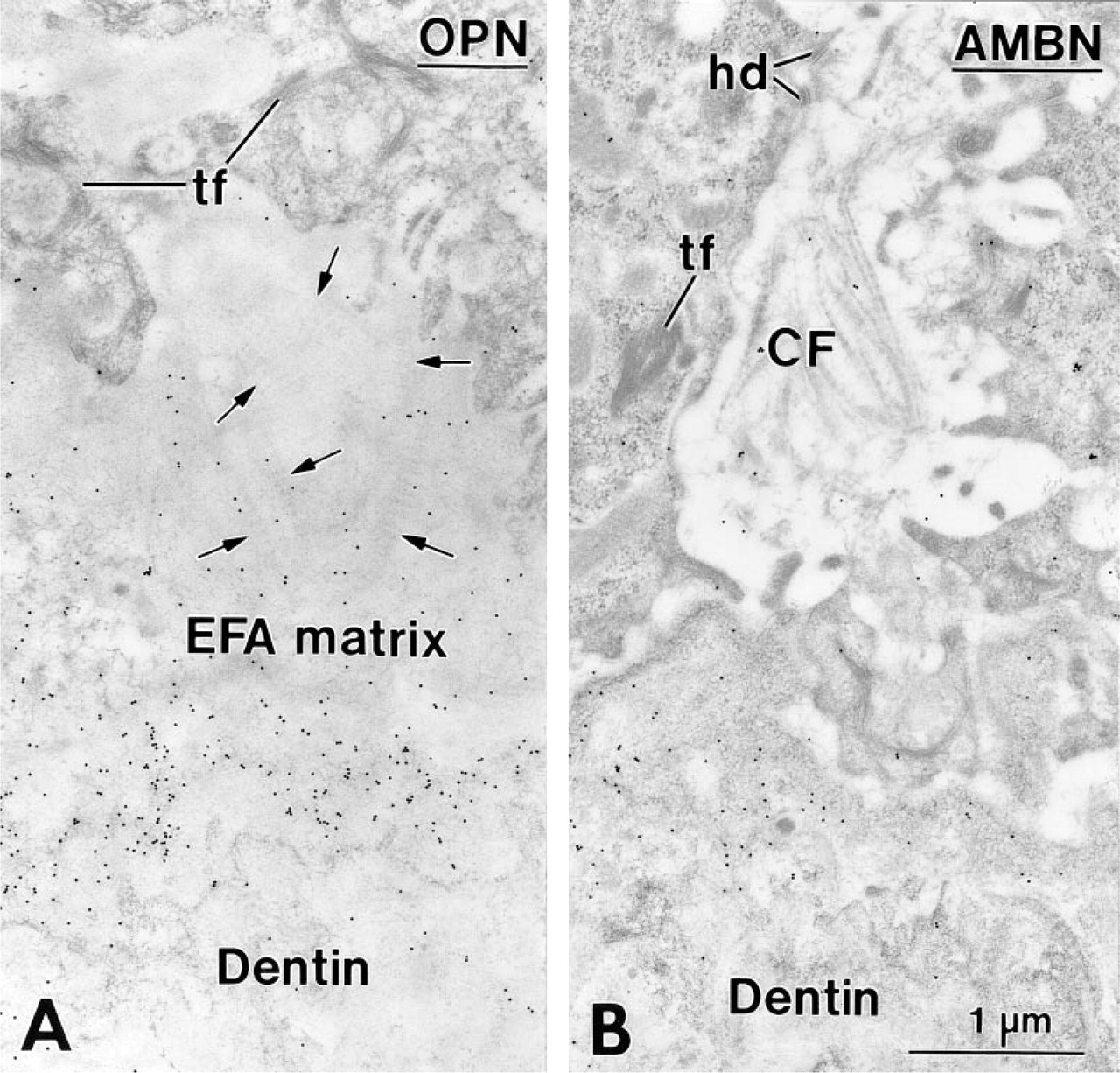
Regions of the enamel-free area (EFA) in a 14- (
Root Surface
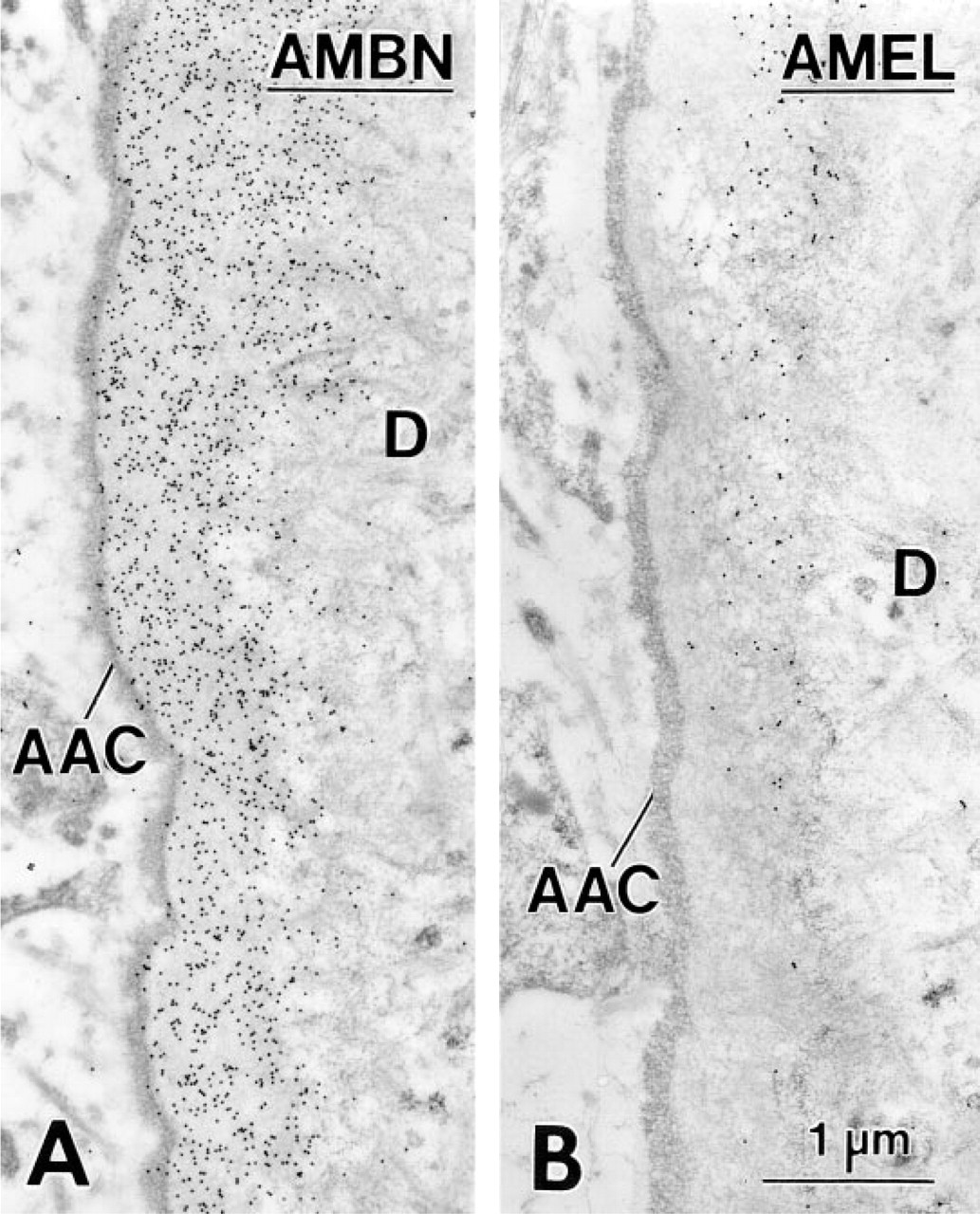
In 14-day-old animals, HERS covered the apical end of the forming root, and its fragmentation started slightly apical to the level where mineralization of the predentin matrix initiated (not shown). The remaining portion of the root was lined by epithelial cells. Between these epithelial cells and the root dentin, a thin layer of collagen-deficient cementum gradually formed, as revealed by a steady increase in thickness in the cervical direction. In the region of the cemento–enamel junction, enamel matrix extended for a short distance between root dentin and cementum. This matrix labeled for AMBN (Figures 3 and 4A) and AMEL (Figure 4B), but the labeling for AMBN extended slightly more apically. Beyond this narrow region near the cemento–enamel junction, immunoreactivity for AMEL (Figure 5A) and AMBN (Figure 5B) was no longer detectable along the root despite the consistent presence of epithelial cells (Figures 5A–5C). The initially deposited layer of cementum consisted mainly of an electron-dense, granular organic matrix that labeled for BSP (not shown) and OPN (Figure 5C). Collagen fibrils were scarce in cementum but abundant adjacent to it (i.e., in precementum), where they formed bundles (as seen in cross-sectional profiles) in bay-shaped concavities formed by the epithelial cells (Figures 5A–5C).
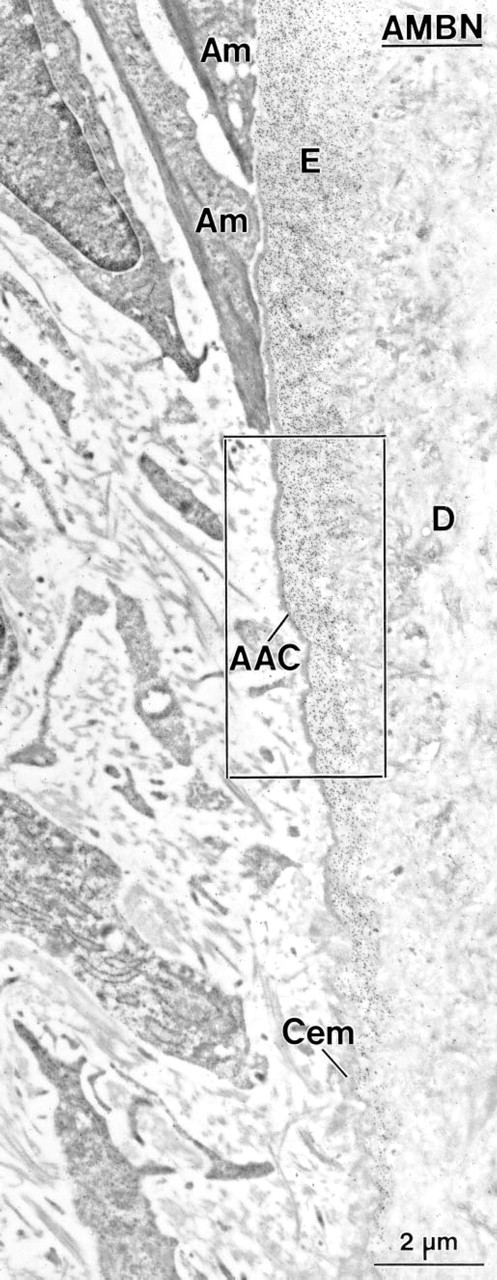
Incubation with anti-ameloblastin (AMBN) antibody showing the region of the cemento–enamel junction in a first mandibular molar of a 14-day-old rat. The cervical-most ameloblasts (Am) have a slender shape and abut on enamel or acellular afibrillar cementum (AAC). The enamel matrix (E) is immunoreactive for AMBN and extends from the crown over the root, where it tapers off. Cem, root cementum; D, dentin.
Consecutive sections from the boxed area in Figure 3 showing the immunolabeling for ameloblastin (AMBN) (
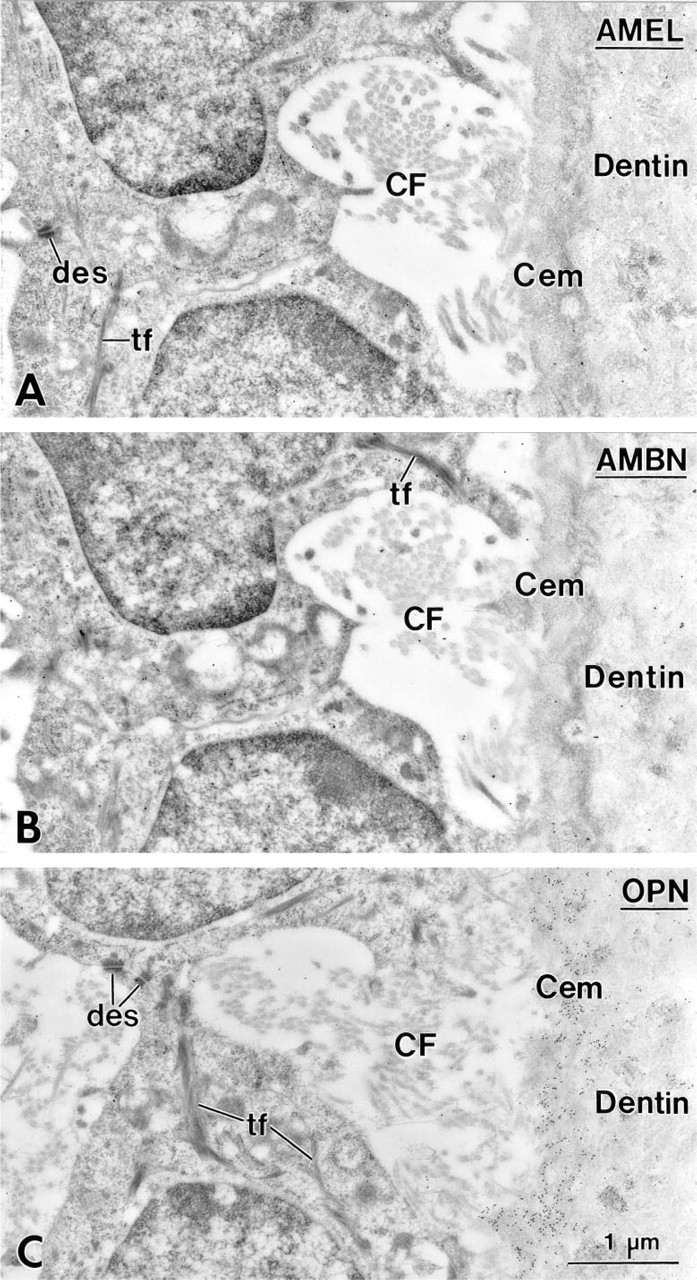
Cervical portion of the root in a first mandibular molar of a 14-day-old rat showing the first layer of cementum (Cem) deposited on mantle dentin. Adjacent sections incubated with antibodies against amelogenin (AMEL; rabbit anti-mouse antibody) (
Discussion
Previous studies in rodent molars have demonstrated that root-lining cells express mRNA for BSP and OPN (MacNeil et al. 1996; Sommer et al. 1996; D'Errico et al. 1997), and that these two noncollagenous matrix proteins accumulate on the root surface (Somerman et al. 1990,1992; Bronckers et al. 1994; MacNeil et al. 1994). According to the classical theory of cementogenesis, ectomesenchymal cells migrate from the dental follicle towards the root surface, where they insinuate between HERS cells and begin to deposit cementum matrix constituents (Cho and Garant 1988,1996). However, the uniform signal for BSP and OPN in cells along the root and the gradual increase in cementum thickness in the cervical direction cannot result from an occasional infiltration by ectomesenchymal cells. BSP, OPN, and collagen fibrils first appear at a time when the forming root surface is still lined by epithelial cells. In addition, mRNA for BSP has been detected in the enamel organ at the cervical-most portion of the crown (see Figure 5 in Sommer et al. 1996), where a layer of acellular afibrillar cementum covers enamel (Bosshardt and Nanci 1997). These data strongly suggest that epithelial cells may contribute to initial cementum deposition.
HERS cells have been proposed to secrete enamel-related proteins before dental follicle-derived cementoblasts form cementum proper (Slavkin 1976; Owens 1980; Lindskog and Hammarström 1982; Glazman et al. 1986; Slavkin et al. 1988,1989; reviewed in Bosshardt and Schroeder 1996). The present results confirm that the first matrix layer deposited against root dentin indeed forms in the presence of epithelial cells. These cells are associated with typical mesenchymal matrix proteins (see above), but NO enamel proteins were immunodetected along the root proper at early stages. These findings are in agreement with the lack of AMEL mRNA signal in root-lining cells (Luo et al. 1991) and with the pattern of expression of mRNA for amelin (Fong et al. 1996). Signal for amelin is found only in cells scattered over the middle and apical third of the root, where some epithelial cells become trapped in cellular cementum, but not in cells along the cervical third (Fong et al. 1996). It has been hypothesized that enamel proteins may be involved in cell signaling events during crown and root development (reviewed in Slavkin 1976; Lindskog and Hammarström 1982; Glazman et al. 1986; Slavkin et al. 1988; Nanci et al. 1989; Luo et al. 1991; Nanci and Smith 1992; Sawada and Nanci 1995; Fong et al. 1996; Zeichner–David et al. 1997). Amelogenins (reviewed in Nanci and Smith 1992) and tuftelin (Zeichner–David et al. 1997) have been unequivocally immunodetected during the early stages of tooth formation when histodifferentiation takes place, but there is no such evidence for the presence of AMEL and AMBN along the cervical root. Although one cannot rule out the possibility that they are present in very low amounts and therefore are below the detectability threshold, the lack of labeling for enamel proteins is not consistent with a signaling role similar to that proposed for crown formation. It is also possible that under pathological conditions, or in situations that deviate from the norm, enamel proteins may be expressed in tissues surrounding the root.
The matrix at the EFA in rat molars also comprises cementum-related proteins (Bosshardt and Nanci 1997). This site is sealed off by epithelial cells and no infiltration by mesenchymal cells has ever been reported (Sakakura et al. 1989; Diab and Zaki 1991; Nakamura et al. 1991; Inai et al. 1992; Bosshardt and Nanci 1997). Although AMEL, BSP, and OPN are known constituents of the EFA matrix (Nakamura et al. 1991; Inai et al. 1992; Bosshardt and Nanci 1997), we show here that it also comprises amelin/AMBN and banded collagen. Unlike on the root, collagen fibrils are scarce and not easily identifiable at the EFA. This may be due to a low expression of collagen and the poor visualization of their crossbanding in mineralized matrix. Indeed, collagen fibrils in the EFA matrix appear to be restricted to sites at which bulky masses protrude between the epithelial cells, suggesting that the deposition of collagen may actually lead to their formation. These data indicate that cells that express enamel proteins can also produce BSP, OPN, and fibrillar collagen, and lend support to the notion that epithelial cells may be involved in the deposition of “typical” mesenchymal proteins at the early stages of cementum formation.
The participation of epithelial cells in cementum formation in rat molars must be transient, because only cells with mesenchymal characteristics are present along forming cementum at more advanced stages of root development. It is conceivable that, after the deposition of the initial cementum layer by HERS cells, dental follicle-derived cementoblasts take over the subsequent formation of cementum. On the other hand, it is also possible that not all epithelial cells withdraw from the root surface and that these cells undergo epithelial–mesenchymal transformation to give rise to cementoblasts (Thomas and Kollar 1988; Bosshardt 1993; MacNeil and Thomas 1993; Bosshardt and Schroeder 1996; Bosshardt and Nanci 1997). Epithelial–mesenchymal transformation is a fundamental process in embryonic development and is well documented for palate formation (reviewed in Hay 1991, 1995). In general, transforming epithelial cells lose epithelial characteristics (e.g., cytokeratin, CAMs like E-cadherin, desmosomes, apical–basal polarity) and acquire mesenchymal ones (e.g., vimentin, fibroblast-like appearance), and then synthesize and secrete “typical” mesenchymal proteins (reviewed in Hay 1995). The expression of fibrillar collagen, BSP, or OPN has been particularly well documented in transforming epithelial cells in vitro or during neoplasia (reviewed in Hay 1991,1995; discussed in Bosshardt and Nanci 1997) but occurs also in normal and pathological conditions by epithelial cells that do not undergo a morphologically identifiable transformation (Hayashi et al. 1988; discussed in Bosshardt and Nanci 1997). In the rat molar tooth, the production of typical cementum proteins may start before the epithelial cells switch to a mesenchymal phenotype, and therefore epithelial cells are still associated with the first matrix deposited on dentin. Epithelial–mesenchymal transformation could also explain the apparent disappearance of the majority of HERS cells (reviewed in Bosshardt and Selvig 1997). Because there is no evidence for cell death in HERS during early root formation, the inconsistency in cell number between HERS and the epithelial cell rests of Malassez could reflect a phenotypic conversion.
Footnotes
Acknowledgements
Supported by the Medical Research Council of Canada through an operating grant (AN) and a fellowship (DDB).
We are grateful to Ms Sylvia Zalzal for preparing the protein A–gold complexes used for immunolabeling.
