Abstract
Keywords
The submandibular gland, an important secretory organ in vertebrates, consists of a terminal portion and a ductal system. In the terminal tubular region of the submandibular gland in the neonatal rat, three different types of cells, terminal tubule cells, proacinar cells, and acinar cells, can be identified on the basis of their histological and ultrastructural features. However, the adult gland contains only acinar cells (Pink-staff 1980). The terminal tubule and proacinar cells disappear during postnatal development, whereas the acinar cells undergo rapid proliferation (Alvares and Sesso 1975; Srivastava 1977; Klein 1982; Hayashi and Sasa 1989). It has been suggested that the proacinar cells, which disappear early in the life cycle, are actually the committed stem cells for the acinar cells (Srinivasan and Chang 1975; Yamashina and Mizuhira 1976). Conflicting views are held concerning the fate of terminal tubule cells, with Gresik (1975) and Leeson and Jacoby (1959) claiming that they are converted to typical intercalated duct cells, whereas other authors describe their direct conversion to acinar cells (Chang 1974; Hand et al. 1996; Mirels et al. 1998).
In recent years, apoptosis (programmed cell death) has been implicated as the mechanism responsible for reducing cell numbers during the regression of parenchymal hyperplasia that occurs in many organs during development or cell differentiation (Tomei and Cope 1991). Walker et al. (1992) provided evidence in the pancreas, an exocrine gland, that the differentiation of stromal fibroblasts into contractile myofibroblasts may be induced by endothelial cell apoptosis, which probably accounts for the associated reduction in the capillary bed. We report here that, in addition to an auto-phagocytosis-mediated process, apoptosis may also be responsible for reducing the number of terminal tubule cells in the male rat submandibular gland.
Male Wistar rats were used at various stages between 7 and 35 days after birth. Morphological aspects of the postnatal development of the submandibular gland were studied by light and electron microscopy, whereas potential apoptotic cells were identified by the TUNEL assay by a conventional immunohistochemical method using an ApopTag Plus in situ apoptosis detection kit (ONCOR; Gaithersburg, MD) according to the manufacturer's instructions.
In agreement with a previous study (Pinkstaff 1980), three different cell types, terminal tubule cells, proacinar cells, and acinar cells, were seen in the terminal portion of the submandibular gland within the first 3 weeks after birth (data not shown). However, in the secretory region, only two types of exocrine cells could be detected: acinar cells, which contained secretory granules weakly stained by toluidine blue, and terminal tubule cells, which contained many strongly stained granules in the apical part of the cytoplasm (Figure 1Aa). When the terminal tubule cells were examined by electron microscopy, they were seen to contain a large amount of rough endoplasmic reticulum, located principally in the basal cytoplasm, and a large spherical or oval nucleus (Figure 1Ab). At later postnatal stages, vacuoles were seen in the supranuclear region of the terminal tubule cells, their number increasing to a maximum by postnatal Day 28 (Figures 1Ac-1Ae). The vacuoles, which often contained debris, were fused to secretory granules that were apparently being digested (arrows in Figure 1Ae), the products of which were then discharged into the lumen. This was followed by loss of the greater part of the cytoplasm (arrows in Figure 1Af) and the subsequent transformation into typical intercalated duct cells (Figure 1Ag). These findings agree with previously reported morphological aspects of submandibular gland development based on light and electron microscopic observations in the postnatal rat (Pinkstaff 1980), and suggest that the terminal tubule cells may be replaced by intercalated duct cells via an autophagocytotic process.
TUNEL analysis revealed that, during Days 21-30 of postnatal development, TUNEL-positive cells were found only in the terminal tubular region (Figures 1Bb-1Be). They were most abundant (about 14% of the total terminal tubule cells) on Day 23 (Figures 1Bc and 1C), after which their numbers decreased progressively (Figures 1Bd, 1Be and 1C), with no TUNEL-positive cells being detected by postnatal Day 35 (Figures 1Bf and 1C). Similar experiments, carried out using a very recently published modified in situ TUNEL technique for the improved detection of apoptotic cells (Cuello-Carrión and Ciocca 1999), showed a similar time course of the postnatal appearance of TUNEL-positive cells in the terminal tubular region (not shown). Figure 1Ah shows nuclear fragments with characteristic chromatin condensation, which are typical features of apoptosis and indicate apoptotic death of terminal tubule cells, suggesting that apoptosis may also be responsible for reducing the numbers of terminal tubule cells.
Several studies have suggested that the original in situ TUNEL assay for apoptosis does not discriminate among apoptosis, necrosis, and autolytic cell death (Grasl-Kraupp et al. 1995; Hand et al. 1996). However, the modified TUNEL assay, which was improved to detect apoptotic cells, gave the same result for the time course of appearance and disappearance of TUNEL-positive cells. In addition, in the present study, neither necrotic nor autolytic structures were ever seen in the terminal tubular region of the submandibular gland by either light or electron microscopy, as also reported by Ito (1990). Finally, if autophagic vacuoles induced autolytic changes in the terminal tubular region, all of the terminal tubule cells would be expected to be TUNEL-positive, as autophagic vacuoles were seen in all the cells in the terminal tubular region after 23 days of age. However, this was not the case, as the maximal number of TUNEL-positive cells (on postnatal Day 23) was only about 14% of the total terminal tubule cells. Taken together, these results strongly suggest that the TUNEL-positive cells are apoptotic cells.
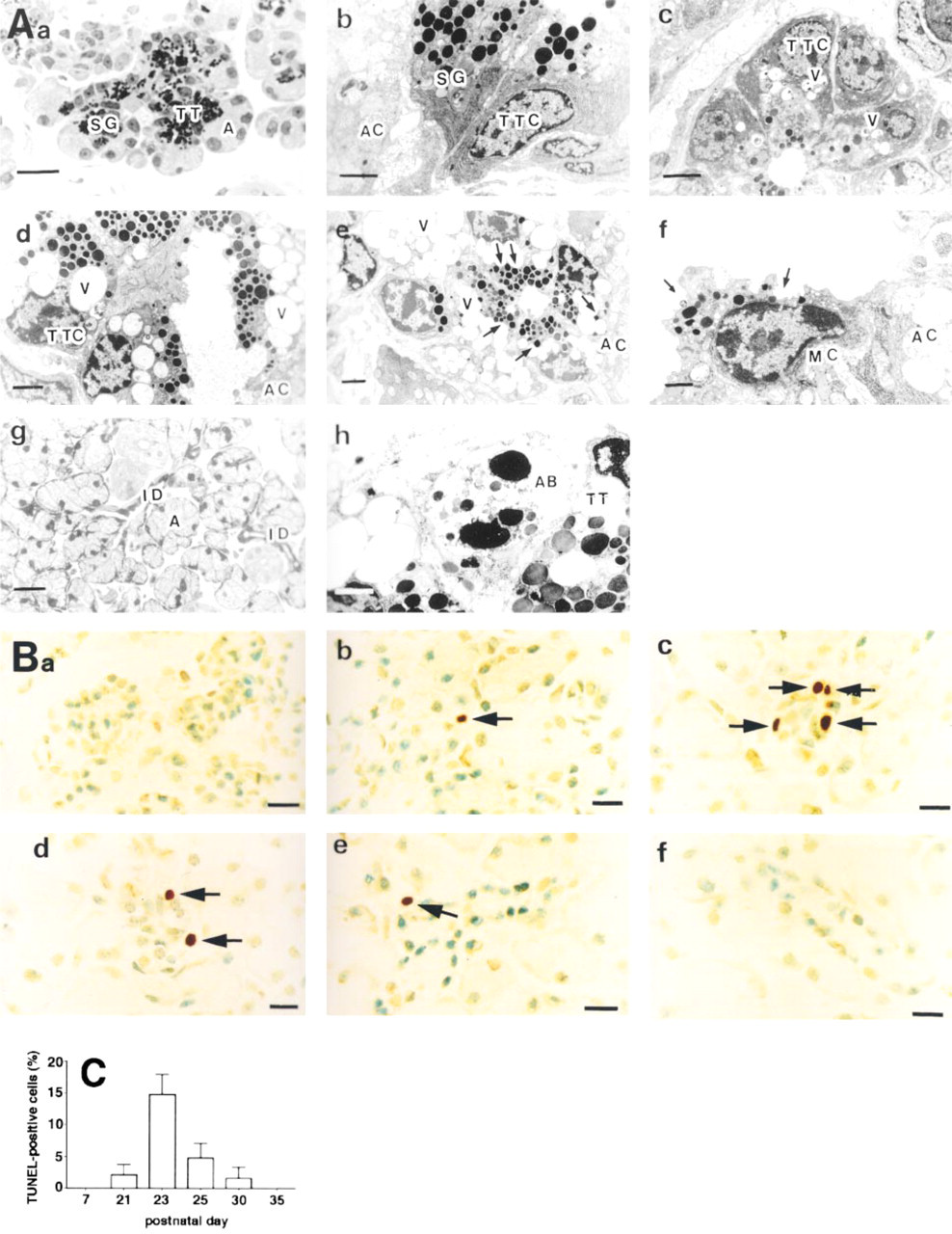
Micrographs of the submandibular gland. (
Footnotes
Acknowledgements
Acknowledgments
Supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Science and Culture of Japan (08835009; 07906320).
