Abstract
This study was designed to determine whether apoptosis and proliferation of myoepithelial cells occur in atrophic rat submandibular glands. The excretory duct of the right submandibular gland was doubly ligated with metal clips. The atrophic right submandibular glands removed after 1–28 days of duct ligation were investigated using immunohistochemical double staining for actin as a marker for myoepithelial cells and proliferating cell nuclear antigen (PCNA) as a marker for proliferating cells, double staining for actin immunohistochemistry, nick end-labeling (TUNEL) as a marker for apoptotic cells, and transmission electron microscopy (TEM). A few PCNA- and no TUNEL-positive myoepithelial cells were found in the control submandibular glands taken from animals with no operation. In the experimental glands, PCNA-positive myoepithelial cells were common 2 and 3 days after duct ligation and then decreased in number. TUNEL-positive myoepithelial cells appeared at 2 days and were observed most frequently at 5 days. Apoptotic myoepithelial cells were also identified by TEM. These observations suggest that both apoptosis and proliferation of myoepithelial cells occur, especially in the early phase of atrophy, in the rat submandibular gland.
M
Myoepithelial cells have been speculated to play an important role in the histogenesis of some salivary gland tumors, such as pleomorphic adenoma, myoepithelioma, adenoid cystic carcinoma, and certain other tumors (Batsakis et al. 1983; Dardick and Burford-Mason 1993; Redman 1994). However, details of the role they may play are not clearly established. This makes it necessary to investigate the proliferation and apoptosis of myoepithelial cells under various conditions. Formerly, it was considered that there was no or quite low proliferative activity in myoepithelial cells in salivary glands (Batsakis et al. 1989). Redman (1994) identified mitotic figures of mature myoepithelial cells in developing rat parotid glands by transmission electron microscopy (TEM). Burgess et al. (1996), by immunohistochemical observations of rat parotid glands with atrophy induced by duct ligation, showed that myoepithelial cells proliferated both in atrophy and in normal parotid glands. However, it is not established whether myoepithelial cells are able to proliferate in atrophic submandibular glands, which differ histologically from parotid glands. Apoptosis of myoepithelial cells in salivary glands was studied in atrophic rat parotid glands with TEM by Walker and Gobe (1987), who failed to find apoptotic myoepithelial cells.
Therefore, it is both important and interesting from the clinical aspect to determine whether or not proliferation and apoptosis of myoepithelial cells occur in atrophic submandibular gland. The purpose of the present study was to clarify this problem. To achieve this, rat submandibular glands with atrophy induced by doubly ligated ducts were examined, using immunohistochemical double staining for actin and proliferating cell nuclear antigen (PCNA), double staining for actin immunohistochemistry, and nick end-labeling (TUNEL) and TEM.
Materials and Methods
Experimental Procedures
Seven-week-old male Wistar rats were used in this study. In the experimental rats, the right submandibular gland and its excretory duct were dissected under general anesthesia with ether. Then the excretory duct was doubly ligated with metal clips near the hilum of the gland (Takahashi et al. 2000). The experimental animals were sacrificed at 1, 2, 3, 4, 5, 7, 14, or 28 days after the duct ligation and control rats were not operated. The fresh submandibular glands were removed and frozen in liquid nitrogen for both kinds of double staining used here. Frozen sections were made with a cryostat, air-dried, and fixed in 4% paraformaldehyde for 2 min. After rinsing with PBS, the sections were ready for the double staining. The animals for TEM were perfused with 2% paraformaldehyde-1.25% glutaraldehyde under pentobarbital general anesthesia. The excised submandibular glands were immersed in the same fixative.
All animal experimentation followed the Guide for the Care and Use of Laboratory Animals, Hokkaido University Graduate School of Dental Medicine.
Immunohistochemical Double Staining for Actin and PCNA
The sections were exposed to an anti-PCNA mouse monoclonal antibody (MAb) (PC-10; DAKO JAPAN, Kyoto, Japan), a marker for proliferating cells (Miyachi et al. 1978), after inhibiting endogenous peroxidase with 3% hydrogen peroxide. The sections were incubated with a biotinylated anti-mouse rabbit polyclonal antibody (DAKO JAPAN), streptavidin-biotin-horseradish peroxidase (HRP) complex (DAKO JAPAN) and 3,3′-diaminobenzidine (DAB) for the visualization in turn. After rinsing the sections with PBS, they were incubated with HRP-labeled anti-a-smooth muscle actin (1A4; DAKO JAPAN), a marker for myoepithelial cells. Actin localization was developed with a VIP Substrate Kit (Vector Laboratories; Burlingame, CA) and the sections were counterstained with methyl green.
Normal mouse serum was substituted for both primary antibodies as a negative control.
Single immunohistochemistry for actin was also performed, omitting the part with PCNA from the above immunohistochemical double staining.
Double Staining for Actin Immunohistochemistry and TUNEL
The apoptotic cells were identified by the modified TUNEL method (Gavrieli et al. 1992) using an Apop Tag Peroxidase In Situ Apoptosis Detection Kit (Intergen; New York, NY). Endogenous peroxidase activity in the sections was quenched with 3% hydrogen peroxide. The sections were incubated with TdT and dUTP-digoxigenin with no proteinase K pretreatment and next with anti-digoxigenin-peroxidase sheep polyclonal antibody. The reaction was visualized by the DAB method and then the sections with TUNEL reaction were immunostained with anti-actin antibody as described above and counterstained with methyl green.
Negative control sections were incubated with distilled water and normal mouse serum in the absence of TdT for TUNEL and anti-actin antibody for immunohistochemistry, respectively.
In both types of double staining, the actin-positive reaction was realized as purple cytoplasm and the PCNA- and TUNEL-positive reaction as brown nuclei.
Quantification
The labeling indices of PCNA and TUNEL in myoepithelial cells were calculated. In doubly stained sections, approximately 200 myoepithelial cells were randomly counted at a magnification of ×400 (BH-2; OLYMPUS, Tokyo, Japan) and the percentage of double-positive cells was calculated. The mean of the percentages from four stained sections was used as a representative value for that animal. The means and standard error of the mean (SEM) were calculated for four experimental rats at each time point and four control rats, and then one-way analysis of variance (ANOVA), followed by the Fisher's protected least significant difference (PLSD) post-hoc test, were performed with StatView 4.5 statistical software (Abacus Concepts; Berkeley, CA).
Transmission Electron Microscopy
The fixed tissue was immersed in 1% osmium tetroxide, stained en bloc with 4% uranyl acetate, and embedded in Epon 812. Ultrathin sections were cut, stained with uranyl acetate and lead citrate, and observed with a transmission electron microscope (H-7000 electron microscope; HITACHI, Tokyo, Japan).
Results
Histologically, ducts were dilated in the edematous glandular tissue and mitotic figures of duct cells were identified at 2 days after duct ligation. At 3 and 4 days, apoptosis of acinar cells was frequently observed and acinar cell numbers decreased. After 7 days most acinar cells had disappeared and the connective tissue with lymphoid cell infiltration increased around the residual ducts (Takahashi et al. 2000).
PCNA and TUNEL Observations
Single immunohistochemical staining for actin showed the distribution of myoepithelial cells in the submandibular glands. In the controls, myoepithelial cells surrounded acini and intercalated ducts. However, they were not observed at the periphery of striated ducts (Figure 1A). In the experimental glands, the atrophic acini were still surrounded by myoepithelial cells at 3 and 4 days (Figure 1B). With the disappearance of acini, many dilated ducts were surrounded by myoepithelial cells (Figure 1C).
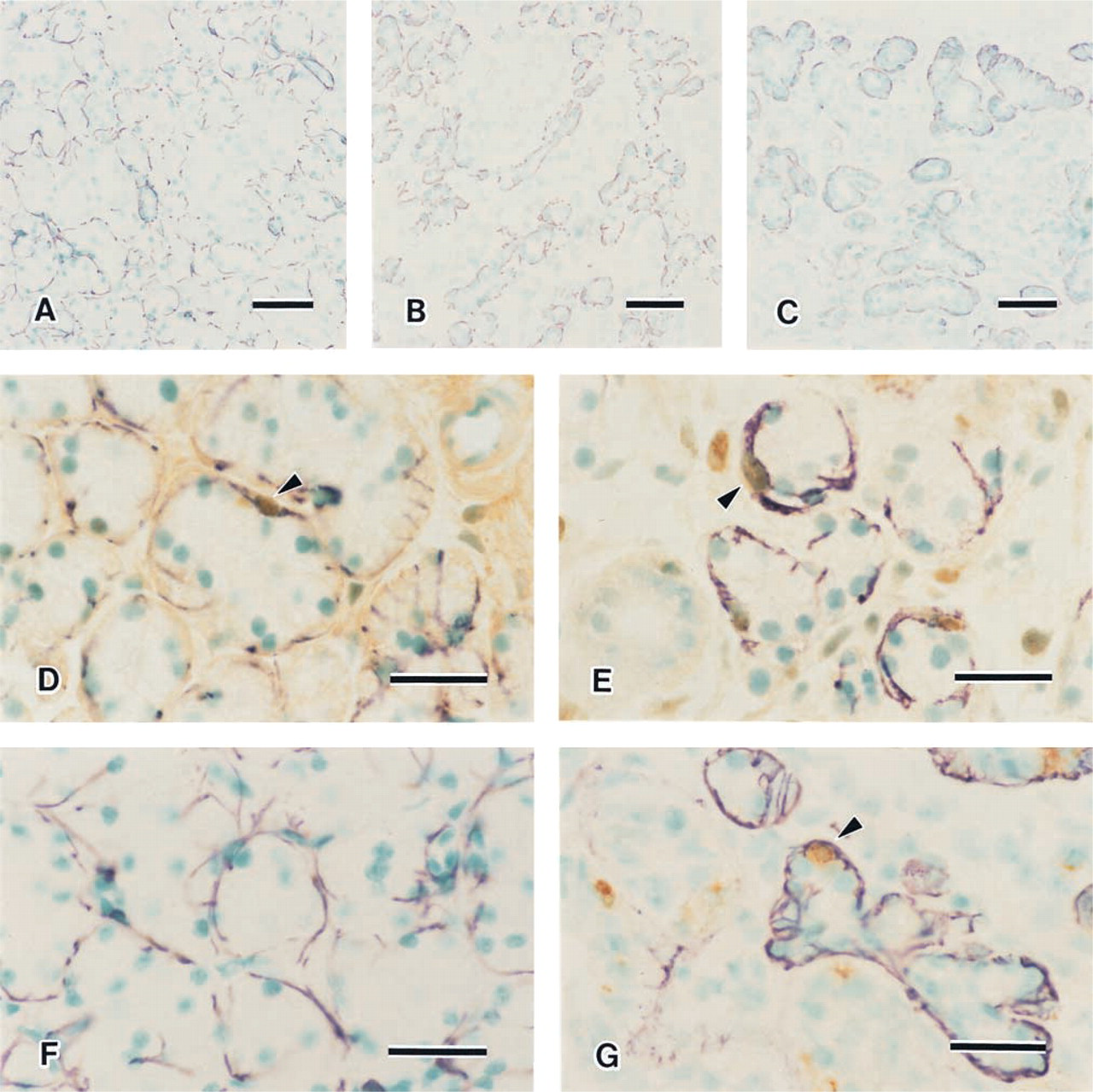
Single immunohistochemistry for actin (
A few PCNA-positive myoepithelial cells were identified in the control glands (Figure 1D). In the duct ligated gland, PCNA-positive myoepithelial cells were often observed, especially at 2 and 3 days (Figure 1E). No TUNEL-positive myoepithelial cells were identified in the control glands (Figure 1F). However, in the experimental glands TUNEL-positive myoepithelial cells were observed at 2 days and later they were identified in all experimental specimens (Figure 1G). Negative control sections for both kinds of double staining showed no reaction.
Labeling indices of PCNA- and TUNEL-positive myoepithelial cells after duct ligation (± SEM)
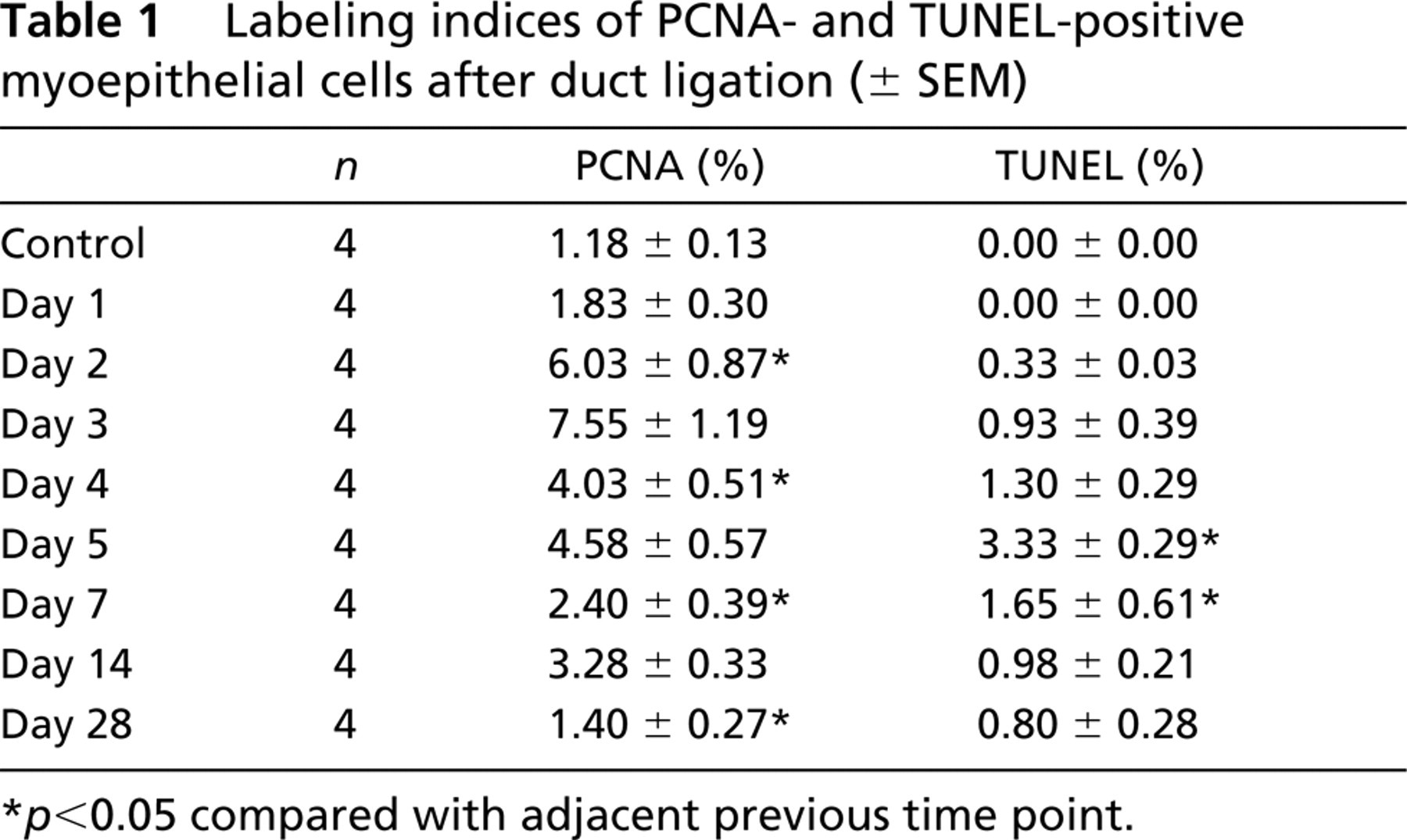
p<0.05 compared with adjacent previous time point.
The labeling indices of PCNA and TUNEL for myoepithelial cells are shown in Table 1 and Figure 2. The PCNA labeling index increased significantly between 1 and 2 days (p<0.0001) after duct ligation, reached a peak of 7.55% at 3 days, and significantly declined between 3 and 4 days (p<0.0005). The TUNEL-positive myoepithelial cells first appeared at 2 days. The TUNEL labeling index increased significantly between 4 and 5 days (p<0.0001) and was highest, 3.33%, at 5 days, after which it declined significantly between 5 and 7 days (p<0.0005). The PCNA and TUNEL indices in the control glands were 1.18% and 0%, respectively.
TEM Observations
In the control glands, myoepithelial cells were located between the basal lamina and acinar or intercalated duct cells. In the cytoplasm, microfilaments, sometimes associated with dense bodies, were observed. There were caveolae along the basal plasma membrane (Figure 3A).
In the experimental glands, ultrastructural changes of myoepithelial cells were remarkable from 3 days after duct ligation. Myoepithelial cells often showed bizarre structures, with cell processes protruding into interstitial spaces. The basement membrane of such myoepithelial cells tended to be thickened and arranged in many irregular folds (Figure 3B). Apoptosis of myoepithelial cells was rarely identified. In apoptotic myoepithelial cells, nuclear chromatin was aggregated into a dense mass and the electron density of the cytoplasm was increased (Figure 3C). Some myoepithelial cells had phagocytosed apoptotic bodies (Figure 3D).
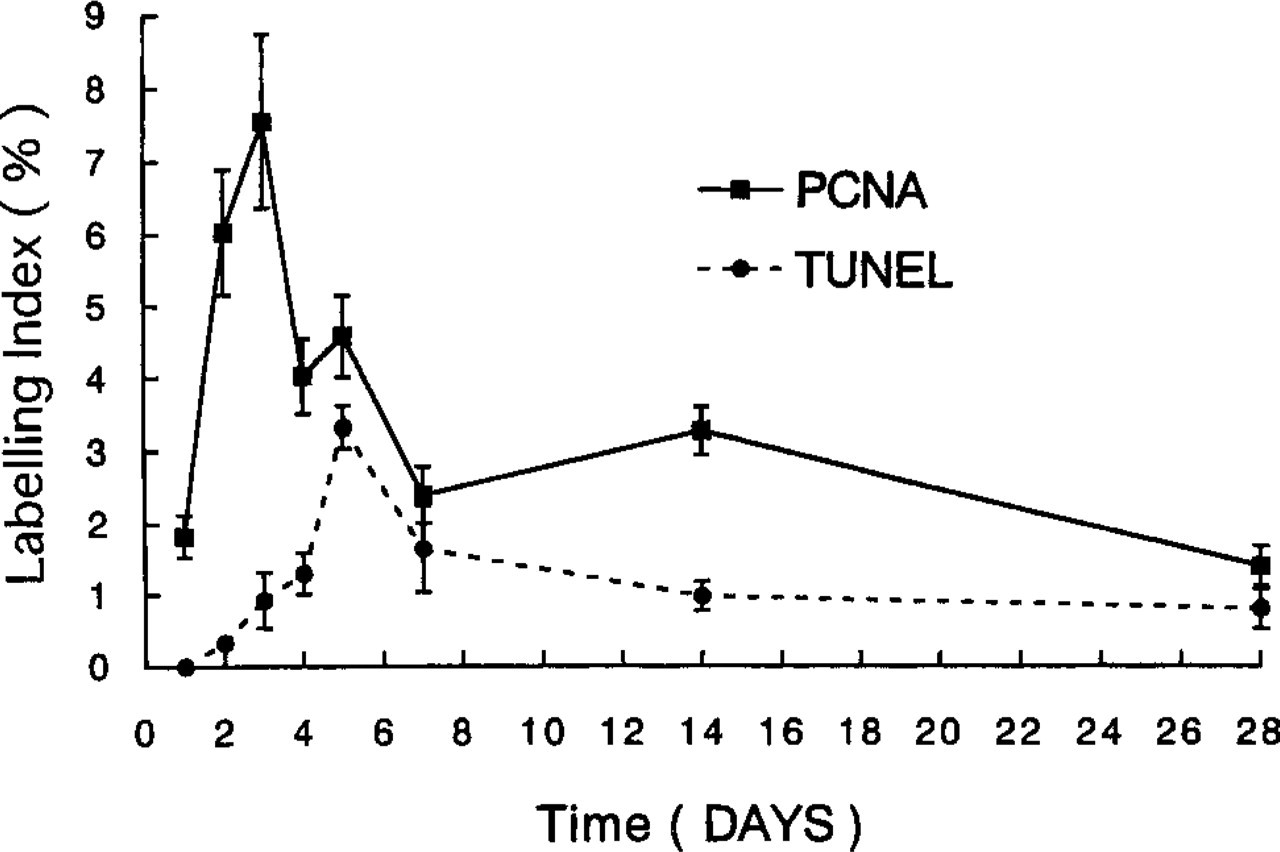
Changes in labeling indices of PCNA (solid line)- and TUNEL (broken line)-positive myoepithelial cells in experimental animals. Error bars show SEM (n=4 at each time point).
Discussion
In atrophy of the parotid gland (Emmelin et al. 1974; Garrett and Emmelin 1979) and involution of mammary glands (Radnor 1972; Walker et al. 1989), myoepithelial cells often show a bizarre structure, with protrudent cellular processes and irregular folds of the basement membrane thickening, probably due to new synthesis by myoepithelial cells (Emmelin et al. 1974; Garrett and Emmelin 1979). These ultrastructural changes are common morphological changes of myoepithelial cells in regressive exocrine glands. The myoepithelial cells in this study also often showed similar changes, and this finding demonstrates that atrophy was induced in the submandibular gland. This atrophy was induced both by obstruction of the excretory duct and by damage to the chorda tympani on the excretory duct that would have been included in the duct ligation (Harrison and Garrett 1972; Harrison et al. 2001).
It has been shown that precursors of or immature myoepithelial cells are able to proliferate in cultured and transplanted human mammary glands (Joshi et al. 1986) and during development of hamster Harderian (Lopez et al. 1992), rat parotid (Taga and Sesso 1979; Redman et al. 1980), and rat submandibular (Cutler and Chaudhry 1973) glands. In contrast, mature myoepithelial cells have been considered unable to proliferate (Cutler and Chaudhry 1973; Joshi et al. 1986; Batsakis et al. 1989) because they were end-differentiated cells (Batsakis et al. 1989). However, mitosis of myoepithelial cells was observed in developing rat parotid glands (Redman 1994), and cycling myoepithelial cells were identified in mouse mammary glands stimulated by hormone (Sapino et al. 1990) and in atrophic rat parotid glands (Burgess et al. 1996). In the present study, PCNA-positive myoepithelial cells were also found in atrophic rat submandibular glands. This suggests that mature myoepithelial cells in the rat submandibular gland possess proliferative ability that is especially active under some experimental conditions. Further study will be needed to investigate other exocrine glands under various conditions.
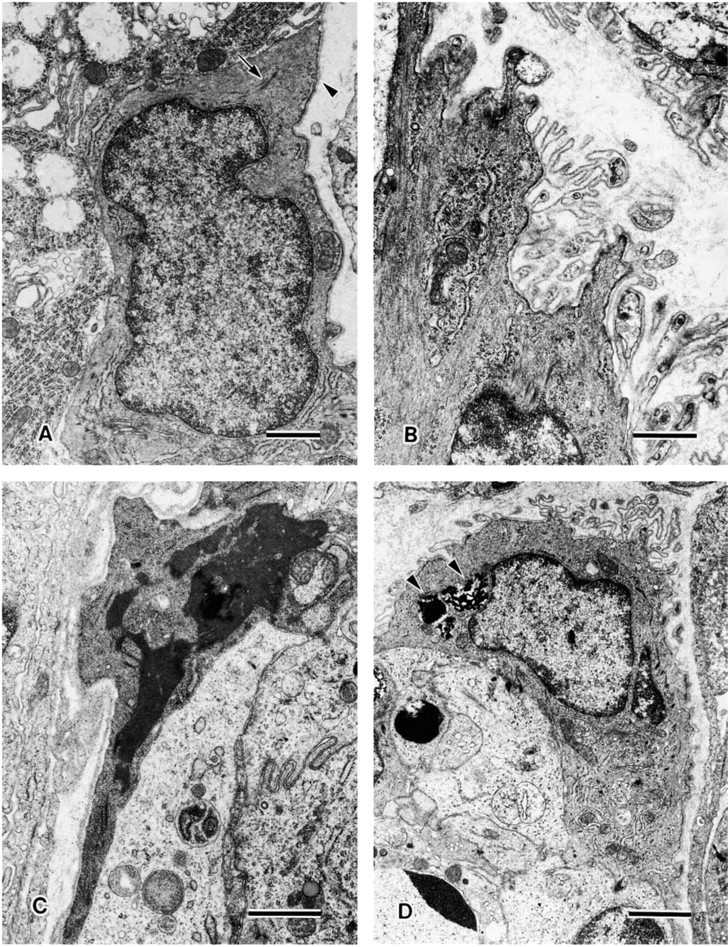
Ultrastructure of myoepithelial cells in control (
The maximum of the PCNA index of myoepithelial cells in the present study (7.55%) is much lower than that in atrophic parotid gland (23.1%) (Burgess et al. 1996). This may arise from differences in the myoepithelial cell distribution between submandibular and parotid glands in the rat. Myoepithelial cells are situated at the periphery of both acini and intercalated ducts in rat submandibular glands and mainly of intercalated ducts in rat parotid glands. Thus, there are more myoepithelial cells in submandibular glands than in parotid glands (Dewey 1958; Bogart 1971; Garrett and Emmelin 1979; Redman 1994). Therefore, it may be unnecessary for myoepithelial cells in submandibular glands to proliferate as actively as in parotid glands during atrophy.
Using TEM and double staining for actin immunohistochemistry and TUNEL, this study demonstrates that apoptosis of myoepithelial cells occurs in atrophic rat submandibular glands. This is the first report to identify apoptotic myoepithelial cells in the salivary gland, although apoptosis of myoepithelial cells was reported in involution of lactating breasts in mice (Walker et al. 1989). There is a difference in the occurrence of apoptosis of myoepithelial cells between atrophic submandibular glands and parotid glands, in that no apoptosis of myoepithelial cells was observed by TEM (Walker and Gobe 1987). There are two possible reasons for this. One is that deletion of myoepithelial cells is not necessary in atrophic parotid glands because of the relatively smaller number of myoepithelial cells (Dewey 1958; Bogart 1971; Garrett and Emmelin 1979; Redman 1994). The other is that apoptosis of myoepithelial cells was not detectable by TEM alone, regardless of its occurrence. Although TEM is useful to investigate apoptotic cells morphologically, it is very difficult to observe them in a wide area with TEM. Therefore, using TUNEL together with TEM is more useful to identify apoptotic cells. It would be desirable if atrophic parotid glands are investigated with both methods as in the present study.
It has been understood that apoptotic bodies are phagocytosed by intraepithelial macrophages, duct cells, and acinar cells in atrophic salivary glands (Walker and Gobe 1987; Takahashi et al. 2000). In addition, apoptotic bodies phagocytosed by myoepithelial cells were observed in this study. This finding suggests that myoepithelial cells are also able to participate in the disposal of apoptotic bodies. These apoptotic bodies may derive from acinar cells adjacent to phagocytosing myoepithelial cells.
Recently, P2Y2 receptor, one of the receptors for extracellular nucleotides (P2 receptors), was reported to be an important component of the response to injury in submandibular gland from the fact that P2Y2 receptor activity and mRNA levels were increased in duct ligated submandibular gland (Turner et al. 1997; Ahn et al. 2000). In the duct ligated submandibular gland, P2Y2 receptor mRNA was mainly detected in acini and intercalated ducts by in situ hybridization (Ahn et al. 2000). However, it is still not known whether P2Y2 receptor mRNA of myoepithelial cells surrounding acini and intercalated ducts is upregulated in atrophic submandibular gland.
In conclusion, the present study demonstrates that both apoptosis and proliferation of myoepithelial cells occur in atrophy of rat submandibular glands.
Footnotes
Acknowledgments
Supported by Grants-in-Aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science and Technology (no. 13671961).
We gratefully acknowledge the assistance of Dr Neal I. Walker (Brisbane, Australia) with the manuscript.
