Abstract
Correlative microscopy is an important approach for bridging the resolution gap between fluorescence and electron microscopy. We have employed FluoroNanogold (FNG) as the detection system in these types of studies. This immunoprobe consists of a gold cluster compound to which a fluorochrome-labeled antibody is covalently linked. In these preparations, the fluorescence signal from FNG is first recorded then the gold cluster compound is subjected to a silver enhancement reaction before examination by electron microscopy. Potential complications are those associated with photochemical reactions that occur during fluorescence microscopy. We have evaluated this and some anti-photobleaching agents (i.e., 1,4-diazabicyclo[2.2.2]octane [DABCO],p-phenylenediamine [PPD], and N-propyl gallate [NPG]) for their utility with FNG in correlative microscopy. When DABCO was employed, the gold signal from FNG was dramatically diminished but the fluorescence signal was unaffected. The gold signal of DABCO-treated samples decreased to approximately 30% of that of the other samples. On the other hand, PPD and NPG did not adversely affect the FNG labeling. We recommend that either PPD or NPG be used and that DABCO be avoided as an antiphotobleaching reagent for this technique.
C
Materials and Methods
Human neutrophils were isolated and prepared for ultrathin cryosectioning and immunolabeling as previously described (Takizawa and Robinson 1994a, b; Takizawa et al. 1998). Reagents were the same as reported previously, except as noted. Grids bearing ultrathin cryosections were incubated for immunocytochemical localization of myeloperoxidase (MPO) with anti-MPO (clone MPO-7, 1.3-2.6 μg/ml; Dako, Glostrup, Denmark) for 3-4 hr at 22C. The grids were then incubated with fluorescein isothiocyanate (FITC)-FNG (Fab' fragment) (4-16 μg/ml; NanoProbes, Stony Brook, NY) for 3-4 hr at 22C. After immunostaining, the FNG-labeled grids were immersed in 50% glycerol-PBS, pH 8, with or without the antiphotobleaching reagents, 1,4-diazabicyclo[2.2.2]octane (DABCO; Sigma, St Louis, MO), p-phenylenediamine (PPD; Sigma), or N-propyl gallate (NPG; Wako, Osaka, Japan). Concentrations of the antifading reagents DABCO, PPD, and NPG were 25 mg/ml, 1 mg/ml, and 10 mg/ml, respectively. The grids were temporarily mounted in a very thin layer of the different antifading media (6 μl/grid) between a coverslip (18 mm in diameter; #1 thickness) and a glass microscope slide without any special spacers. The specimens were immediately examined by fluorescence microscopy with an Olympus Provis AX80TR equipped with a 100-W mercury lamp. Photomicrography, image preparation, and electron microscopy were as described previously (Takizawa et al. 1998). A summary of the preparative methods employed is shown in Figure 1.
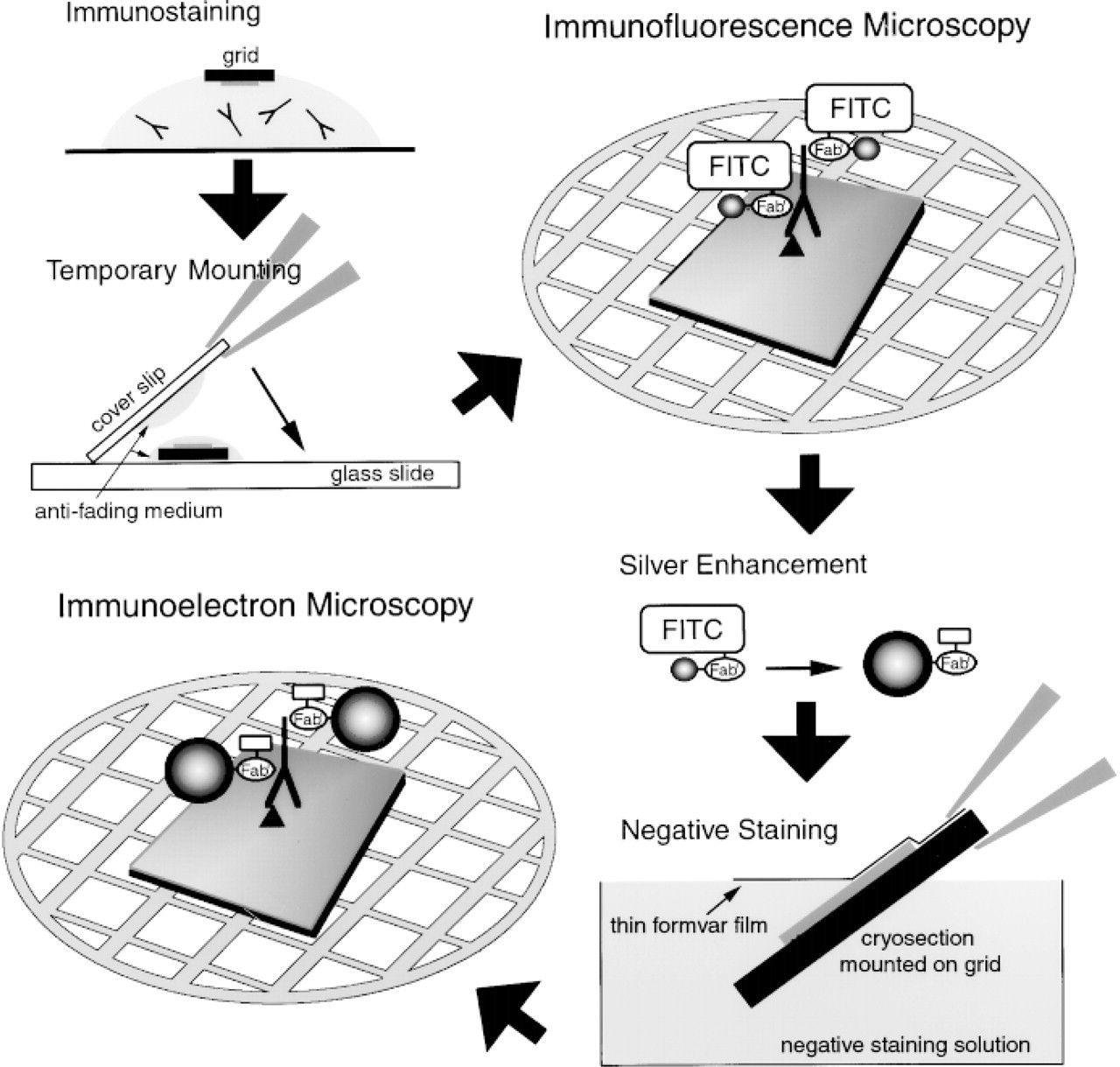
A schematic representation of procedures for correlative fluorescence and electron microscopy using ultrathin cryosections and FNG. Initially, the FNG-labeled ultrathin cryosection is mounted between a slide and a coverslip for viewing by immunofluorescence microscopy (IFM). The temporary slide is then disassembled and the grid is subjected to the silver enhancement reaction. The sections are negatively stained before examination by immunoelectron microscopy (IEM).
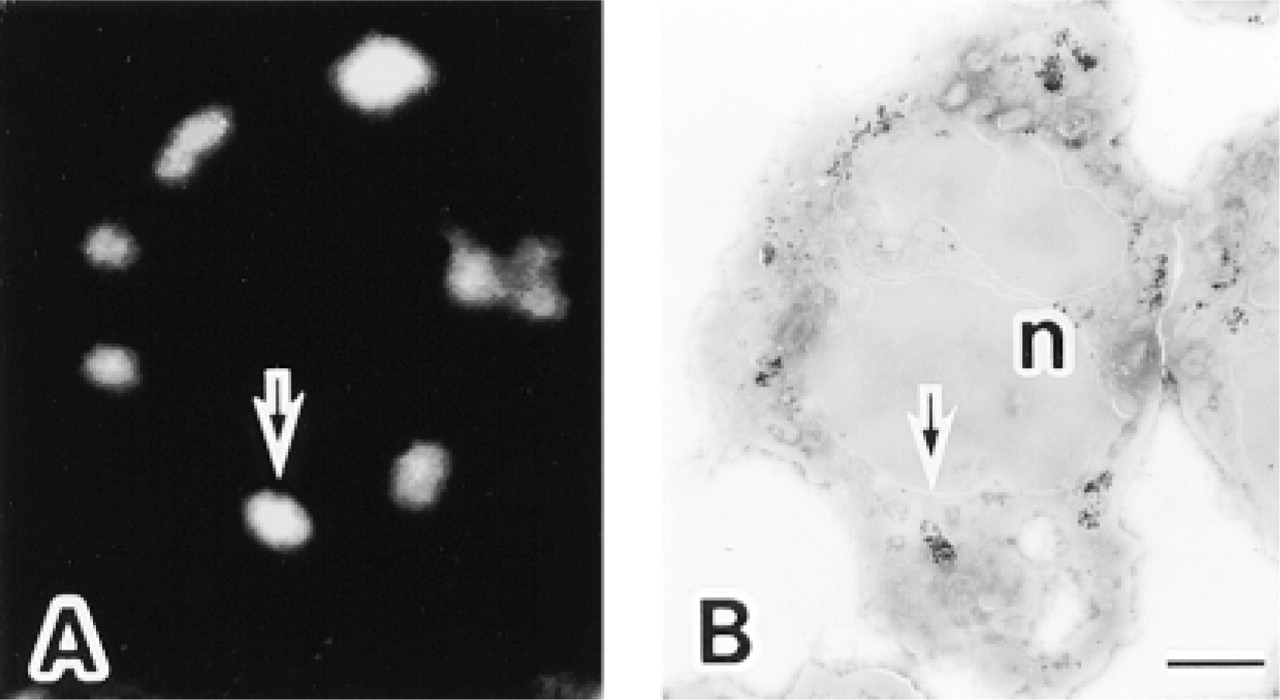
Localization of myeloperoxidase (MPO) in the same ultrathin cryosection of a human neutrophil by fluorescence and electron microscopy using anti-MPO and the FNG as the reporter system. (
Morphometric analysis of the labeling density of FNG in electron micrographs was determined. For each of samples treated with the different mounting media for IFM, at least four neutrophils were examined. Eighteen negatives of electron micrographs were printed at the same magnification (X41,000). The areas of granule profiles were determined with NIH Image 1.62 software and the number of gold particles was determined by manual counting. A total of 71 MPO-positive granules were analyzed.
Results
The immunocytochemical localization of MPO in human neutrophils, using FNG as the detection system, was initially determined by fluorescence microscopy (Figure 2A). The higher-resolution localization of MPO in the same ultrathin cryosection was then determined by EM after silver enhancement (Figure 2B). There was a one-to-one relationship between the fluorescent structures and the granule profiles labeled with silver-enhanced gold and observed by electron microscopy (Figures 2 and 3). Unlabeled granule profiles were more numerous than labeled ones and represent the MPO-negative granules characteristic of these cells (e.g., the specific granules) (Figure 3). Control specimens lacking primary antibody displayed neither fluorescence nor silver-enhanced gold labeling (not shown).
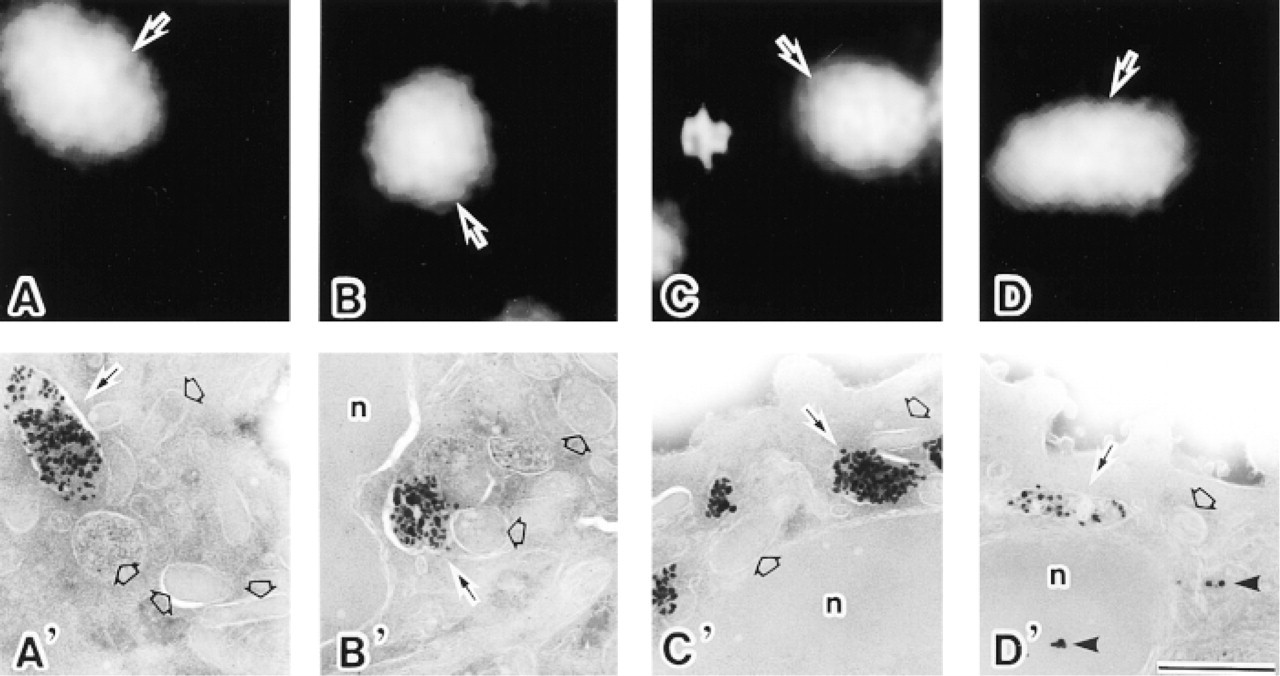
A comparison of the different antifading reagents used for FNG detection in ultrathin cryosections of neutrophils during IFM and subsequent IEM observation. (
All the antifading media tested were capable of reducing the photobleaching of the FNG-associated FITC during IFM observation. However, there was a remarkable difference in the quality of the results of subsequent IEM. When DABCO was employed as an antifading reagent, silver-enhanced 1.4-nm gold particles in the azurophilic granules were dramatically diminished even when strong fluorescence signals from the same FNG probes were associated with the same structures (Figures 3D and 3D'). Moreover, there was an increase in background signals in IEM (Figure 3D'). On the other hand, PPD and NPG did not adversely affect the labeling intensity of silver-enhanced FNG particles in the granules (Figures 3B, 3B', 3C, and 3C'). From the quantitative point of view, the gold labeling of DABCO-treated samples was significantly lower than that of the samples without antifading reagents (p<0.001), with PPD (p<0.001), and with NPG (p<0.001) (Figure 4). In addition, DABCO itself did not cause the reduction in silver-enhanced gold unless cryosections mounted in media containing DABCO were exposed to excitation light (not shown).
Discussion
In IFM, one often encounters the problem of fading of the fluorochrome due to photobleaching. One of the most convenient remedies for photobleaching of fluorescently-labeled samples in IFM is to use antifading reagents, which delay the loss of fluorescence. Compounds such as PPD, NPG, and DABCO have been widely employed for this purpose (Valnes and Brandtzaeg 1985; Longin et al. 1993). PPD is reportedly the most efficient antifading agent for samples stained with FITC-conjugated immunoprobes (Johnson and De C Nogueira-Araujo 1981; Krenik et al. 1989). NPG also can be applied to reduce photobleaching of FITC and tetramethylrhodamine isothiocyanate (Giloh and Sedat 1982; Valnes and Brandtzaeg 1985; Krenik et al. 1989). DABCO is an alternative to reduce FITC fading (Johnson et al. 1982). There are several studies comparing the efficiency of the retardation of fluorochrome fading in IFM and fluorescence in situ hybridization (e.g., Johnson et al. 1982; Valnes and Brandtzaeg 1985; Krenik et al. 1989; Longin et al. 1993; Florijn et al. 1995).
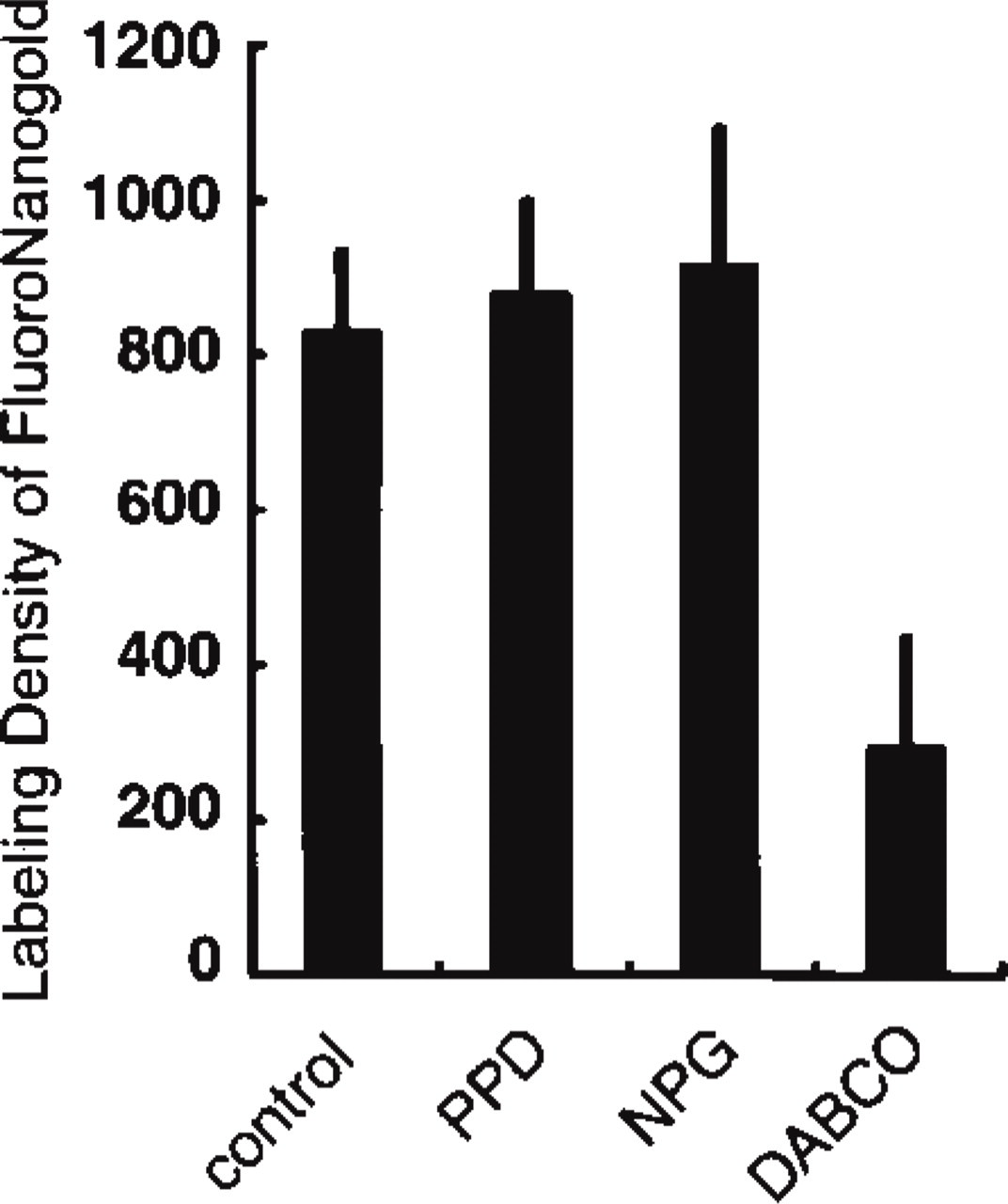
Morphometric analysis comparing the labeling density of silver-enhanced FNG after the use of different antifading reagents during IFM. Control shows the results from sections mounted in a glycerol-PBS medium without antifading reagents. PPD, NPG, and DABCO indicate the results from sections mounted in media containing those reagents. The FNG labeling of DABCO-treated samples decreases to approximately 30% of that of the other samples. Each bar is the mean + SD. The unit of labeling density is the number of gold particles/μm2 of azurophilic granule profiles.
The possibility that prior exposure of FNG to excitation light reduces the amount of gold detected by subsequent silver enhancement has been reported (Baschong and Stierhof 1998). However, we did not observe a diminution in silver-enhanced gold after fluorescence microscopy. In our preparations demonstrating MPO, we observed the fluorescence from FNG for no more than about 30 sec using a 100-W mercury lamp as the light source. Baschong and Stierhof (1998) report that “intense illumination/observation in the fluorescence microscope strongly reduces the efficiency of silver enhancement.” Although prolonged exposures to excitation light apparently can diminish the silver enhancement of FNG, judicious use of this illumination does not appear to present a problem.
Relatively brief excitation of FNG in the absence of antiphotobleaching agents does not appear to diminish subsequent silver enhancement of FNG. However, antiphotobleaching agents were very useful for fluorescence imaging of FNG. Nevertheless, the use of DABCO as the antiphotobleaching agent had a deleterious effect and resulted in a dramatic reduction in the amount of silver-enhanced gold. There was also an increase in the gold background staining when DABCO was used. However, the fluorescence signal in the presence of DABCO was not diminished in comparison to PPD or NPG. On the other hand, use of PPD or NPG did not lower the level of silver-enhanced gold compared to the absence of an antiphotobleaching agent.
The “DABCO effect” appears to be due to a photochemical reaction because exposure of FNG-labeled cryosections to DABCO in the absence of excitation light does not result in loss of silver-enhanced gold particles. The mechanism by which DABCO plus excitation light diminishes the specific silver-enhanced gold signal (but does not appear to alter the fluorescence signal) from FNG is unclear. However, this result is consistent with a DABCO-mediated, photochemically induced disassociation of the gold cluster compound from the parent FNG. This could explain our observation that the silver-enhanced gold background was higher when DABCO was used than when either PPD or NPG was employed to retard photobleaching. We recommend that either PPD or NPG be used and that DABCO be avoided as an antiphotobleaching agent when FNG is used in correlative fluorescence and electron microscopy. In addition, if commercial antifading agents are used, they should be tested for their compatibility with the silver enhancement of FNG.
Footnotes
Acknowledgments
Supported by grants-in-aid for Scientific Research and project grants of the Center for Molecular Medicine of Jichi Medical School from the Ministry of Education, Science, Sports, and Culture of Japan (TT) and by a grant from the American Heart Association, Ohio Valley Affiliate (JMR).
We thank Ms Kiyomi Inose, Ms Megumi Yatabe, and Ms Michiyo Soutome of Jichi Medical School for their help.
