Abstract
E-cadherin, a calcium-dependent cell-cell adhesion molecule, is expressed in highly specific spatiotemporal patterns throughout metazoan development, notably at sites of embryonic induction. E-cadherin also plays a critical role in regulating cell motility/ adhesion, cell proliferation, and apoptosis. We have used the continuously erupting rat incisor as a system for examining the expression of E-cadherin and the associated catenins [α-, β-, γ-catenin (plakoglobin) and p120ctn] during amelogenesis. Using immunhistochemical techniques, we observed expression of α-catenin and γ-catenin in ameloblasts throughout amelogenesis. In contrast, expression of E-cadherin, β-catenin, and p120ctn was strong in presecretory, transitional, and reduced stage ameloblasts (Stages I, III, and V) but was dramatically lower in secretory and maturation stage ameloblasts (Stages II and IV). This expression alternates with the expression pattern we previously reported for the adenomatous polyposis coli protein (APC), a tumor suppressor that competes with E-cadherin for binding to β-catenin. We suggest that alternate expression of APC and the cadherin-catenin complex is critical for the alterations in cell-cell adhesion and other differentiated cellular characteristics, such as cytoskeletal alterations, that are required for the formation of enamel by ameloblasts.
T
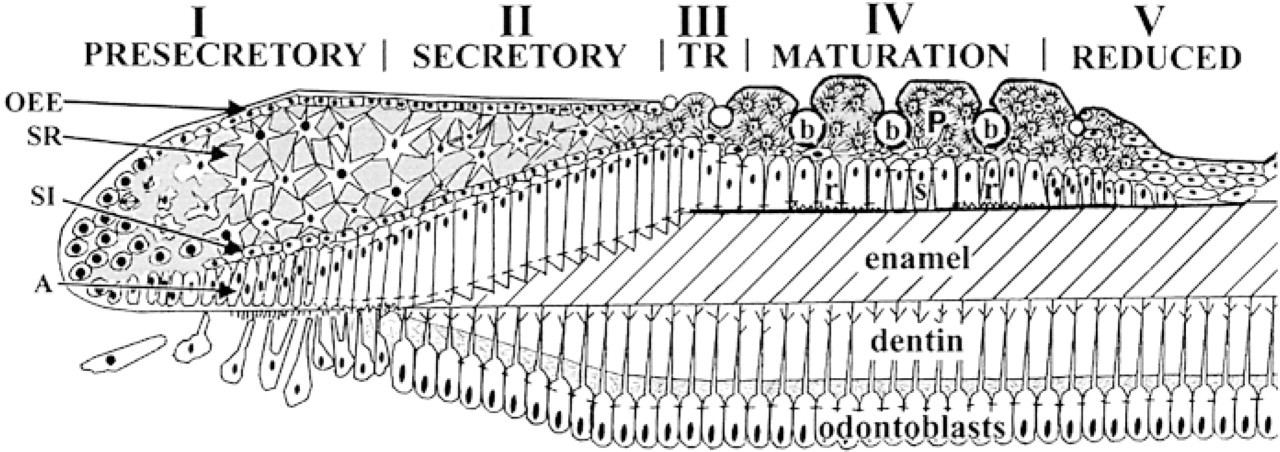
Schematic diagram of the rat incisor enamel organ. The five stages of the ameloblast life cycle are indicated. A, ameloblast layer; SI, stratum intermedium; SR, stellate reticulum; OEE, outer enamel epithelium; b, blood vessels; P, papillary cells; r and s, ruffle-ended and smooth-ended ameloblasts.
The epithelial Cadherins, E- and P-cadherin, have been reported to be expressed early in amelogenesis (Palacios et al. 1995). Most Cadherins, including E-and P-cadherin, are single-pass transmembrane glycoproteins with extracellular amino termini mediating their adhesive interactions (Gallin et al. 1983; Nose et al. 1990; Ozawa et al. 1990), and cytoplasmic carboxy termini (Sorkin and Cunningham 1989; Takeichi 1991). The cytoplasmic domains of the cadherins, although not directly involved in adhesive interactions, are required for interaction with the actin cytoskeleton. These cytoskeletal interactions, in turn, are required for cadherin-mediated cell adhesion (Jaffe et al. 1990; Kintner 1992; Takeichi 1991). The cytoplasmic domain of each cadherin interacts directly with one molecule of either β- or γ-catenin (the latter is identical to plakoglobin) (Piepenhagen and Nelson 1993; Nathke et al. 1994). These catenins, in turn, interact with α-catenin, which interacts directly (Huber et al. 1997) or indirectly (Rimm et al. 1995) with the actin cytoskeleton.
Dramatic changes in E-cadherin expression occur at sites of embryonic induction (Thiery et al. 1984; Takeichi 1988; Burdsal et al. 1993), and both β-catenin and E-cadherin play critical roles in morphogenesis (Funayama et al. 1995; Larue et al. 1996; Miller and Moon 1997). Although the cadherin-catenin interaction is clearly critical for morphogenetic signaling (Heasman et al. 1994; Hermiston and Gordon 1995), there is also evidence that each protein has signaling functions independent of the other (Orsulic and Peifer 1996; Chen et al. 1997). E-cadherin, for example, can also bind to p120ctn, which is a substrate for p60
β-Catenin can interact with a variety of proteins in addition to the cadherins and α-catenin (Miller and Moon 1996). The interactions of β- and γ-catenin (Shibata et al. 1994) with the adenomatous polyposis coli gene product (APC) are likely to be highly significant for the current study. APC protein, a tumor suppressor, competes with E-cadherin for β-catenin binding (Hulsken et al. 1994). Wild-type APC protein downregulates β-catenin (Rubinfeld et al. 1996) by targeting β -catenin to the proteasomal protein degradation pathway (Aberle et al. 1997; Salomon et al. 1997). Loss of APC function results in increased levels of cytoplasmic β-catenin (Munemitsu et al. 1995), stabilized cell adhesion, and decreased cell migration (Wong et al. 1996; Barth et al. 1997). APC also plays a role in microtubule organization, promoting microtubule assembly in vitro (Munemitsu et al. 1994) and appearing at membrane sites involved in cell migration (Nathke et al. 1996) or tubulogenesis (Pollack et al. 1997).
The interaction of β-catenin with the transcription factor LEF-1 may also be important in tooth development. β-Catenin interacts with LEF-1 to alter transcriptional activation by LEF-1 (Behrens et al. 1996; Molenaar et al. 1996). LEF-1 has been reported to regulate inductive interactions during tooth development (Kratochwil et al. 1996).
The importance of E-cadherin, β-catenin, and APC in cell differentiation and morphogenesis in a variety of systems suggested that these three proteins might also play important roles in tooth development and amelogenesis. A study of early first molar development in the mouse showed that E-cadherin is expressed in cells of the outer enamel epithelium, stellate reticulum, and stratum intermedium in bud, cap, and early bell stages, and P-cadherin is expressed in inner enamel epithelium (Palacios et al. 1995). Actin and its associated proteins, α-actinin and vinculin (Kubler et al. 1988), were first detected late in the presecretory stage (Stage I). Their expression then decreased so that, in mouse secretory-stage ameloblasts (Stage II), E-cadherin, actin, α-actinin, and vinculin were localized only in basal (proximal) and apical (distal) terminal web complexes (Kubler et al. 1988; Palacios et al. 1995; Terling et al. 1998). At the distal terminal web, cytoskeletal components, including actin, myosin, tropomyosin, α-actinin, vinculin, and desmoplakin I/II, are oriented primarily perpendicular to the longitudinal axis of the incisor (Nishikawa et al. 1988). APC, on the other hand, was strongly expressed throughout ameloblasts and stratum intermedium cells at this stage, but not in transition-stage ameloblasts (Stage III) (Wang et al. 1998). APC expression was again strong in maturation-stage ameloblasts (Stage IV) and in papillary cells contiguous with ameloblasts. In the final phase of the ameloblast life cycle (Stage V), the cells of the enamel organ became reduced and there was no detectable APC expression (Wang et al. 1998).
Given the dramatic alterations in APC expression during the ameloblast life cycle, the known ability of APC to compete with E-cadherin for β-catenin binding, and the roles of all three proteins in regulation of cell motility/adhesion and in morphogenetic activities, including differentiative signaling, it seemed likely that the cadherin-catenin complex, as well as APC, would play a critical role in amelogenesis. As a first step towards investigating this possibility, we describe here the expression patterns of E-cadherin and the catenins in the continuously erupting rat incisor.
Materials and Methods
All animals used in this work were housed in an AAALAC-approved facility and all operations were performed in accord with protocols approved by The Forsyth Institute's IACUC.
Antibodies
Anti-E-cadherin (clones 34 or 36) and monoclonal antibodies to α-catenin (clone 5), β-catenin (clone 14), γ-catenin (clone 15), and p120ctn (clone 98) were obtained from Transduction Laboratories (Lexington, KY). Anti-APC antibody (CC-1) was obtained from Calbiochem (Cambridge, MA). Peroxidase-conjugated anti-mouse secondary antibody and the Vectastain Elite ABC kit were obtained from Vector Laboratories (Burlingame, CA).
Electrophoresis and Western Blotting
Human gingival tissue was waste tissue from patients undergoing surgery for periodontal disease, obtained under an IRB-approved protocol. Enamel organs (four maxillary and four mandibular) were dissected from euthanized adult inbred Rowett rats.
A-431 cells were obtained from the ATCC (Rockville, MD) and were grown in Dulbecco's modified Eagle's medium containing 10% heat-inactivated fetal calf serum. Immortalized human gingival keratinocytes (IHGK; Kandikonda et al. 1996) were obtained from Dr. Dolphine Oda (Oda et al. 1996) and grown in keratinocyte growth medium. Cells or tissue were washed at 4C with phosphate-buffered saline (PBS = per liter: 8 g NaCl, 0.2 g KCl, 0.2 g KH2PO4, 0.15 g Na2HPO4, pH 7.4) containing 35 μg/ml PMSF and were homogenized in Laemmli sample buffer (Laemmli 1970) containing the following protease inhibitors: antipain 20 μg/ml; aprotinin 20 μg/ml; chymostatin 20 μg/ml; leupeptin 20 μg/ml; pepstatin A 20 μg/ml; soybean trypsin inhibitor 20 μg/ml; PMSF 35 μg/ml. Extracts were heated at 95C for 5 min, and centrifuged for 5 min at 14 krpm and ambient temperature before fractionation on polyacrylamide gels (Laemmli 1970). The Coomassie blue staining pattern of the enamel organ and IHGK extracts is shown in Figure 2. For Western blotting, fractionated proteins were transferred to Hybond ECL membranes (Amersham; Arlington Heights, IL) at 40 V constant voltage overnight in 20% methanol in Laemmli gel running buffer (Laemmli 1970). After transfer, membranes were stained for 3 min with Ponceau red stain (0.1% w/v in 5% acetic acid), destained with PBS, and the migrations of the protein standards marked. The membranes were then blocked at ambient temperature for 1 hr in Western blocking buffer [5% Carnation nonfat dry milk powder, 0.1% Tween-20 in Tris-buffered saline (TBS = 137 mM NaCl, 20 mM TrisCl, pH 7.6)]. Western blots were incubated with antibodies diluted as indicated in Western blocking buffer for 1 hr at ambient temperature and then washed five times over 30 min with Western blocking buffer. Blots were then incubated with peroxidase-conjugated rabbit anti-mouse Ig (ICN; Costa Mesa, CA) 0.67 μg/ml for 1 hr at ambient temperature, washed as before, and antigens detected by incubation with ECL chemiluminescent peroxidase substrate (Amersham) for 1 min, followed by exposure to film for the indicated times.
Immunohistochemistry
Maxillary and mandibular incisors obtained from euthanized adult inbred Rowett rats were fixed in 5% neutral formal/saline overnight, incubated in PBS containing 0.1% Triton X-100 for 8 hr, rinsed overnight with running water, and decalcified in 20% sodium citrate/45% formic acid for 2 weeks. This and all subsequent incubations were at ambient temperature unless otherwise indicated. The incisors were dehydrated in a graded series of ethanols and embedded in paraffin for sectioning. Deparaffinized and rehydrated sections were quenched with 0.3% H2O2 in methanol for 30 min, rinsed with PBS, and blocked with 5% horse serum in PBS for 30 min. The sections were then incubated with primary antibody (see above) at concentrations of either 1 μg/ml or 5 μg/ml for 1 hr at room temperature (anti-E-cadherin, anti-β-catenin, and anti-p120ctn) or overnight at 4C (anti-α-catenin, and anti-γ-catenin). Sections of human and rat gingivae were used as positive control (Figure 3). Sections incubated without primary antibody were used as negative controls, and consistently did not show any staining. Antibody binding was visualized using the Vectastain ABC Elite Universal kit (Vector Laboratories) with Sigma Fast DAB (Sigma; St Louis, MO) as the substrate. Sections were counterstained with 0.1% Fast Green in PBS for 2 min, dehydrated, and mounted. Sections for double staining were rinsed in water and PBS after DAB staining following the first primary antibody, blocked with 5% normal horse serum, and incubated with the second primary antibody overnight at 4C. The sections were rinsed in PBS and developed using the Vectastain Elite ABC kit with Vector VIP as the enzyme substrate, counterstained, dehydrated, and mounted.
Results
Antibody Specificity and Western Blotting
Enamel organ extracts were subjected to Western blotting to verify the antibody specificities and the presence of the antigens in the enamel organ. As shown in Figure 2, Coomassie blue staining (CB) detected a large number of high molecular weight proteins in extracts of both enamel organ and immortalized human gingival keratinocytes (IHGK). Similar results were obtained for the A-431 cell extract. The antibodies detected bands with the expected migrations, confirming that the antibody specificities were as indicated by the manufacturers and that the antigens were present in the enamel organ. Anti-E-cadherin recognized a single band migrating at 124 kD in extracts of both rat enamel organ and A-431 cells. Anti-β-catenin recognized a single band migrating at 99 kD. The anti-p120ctn antibody recognized bands migrating at 110 and 120 kD. The more rapidly migrating form is probably one of the alternatively spliced forms of p120ctn (Mo and Reynolds 1996) rather than a proteolytic breakdown product, because no major faster-migrating bands were observed with the other monoclonal antibodies used. Anti-α-catenin recognized the expected band migrating at 102 kD, and anti-γ-catenin recognized a single band migrating at 85 kD.
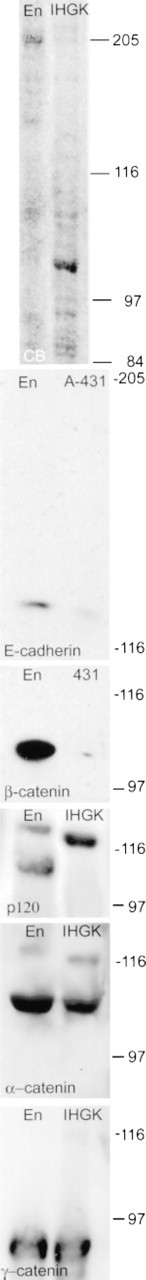
Western blots. Enamel organ extract (En), A-431 cell extract, or extract of immortalized human gingival keratinocytes (IHGK; the last two served as positive controls) containing 15 μg of protein/lane were resolved by SDS-PAGE and proteins detected by Coomassie blue staining or by Western blotting with monoclonal antibodies. CB. Coomassie blue-stained 7.5% PAG. E-cadherin. Extracts were resolved on a 5% PAG and E-cadherin was detected by incubation with monoclonal anti-E-cadherin clone 34 at a final concentration of 1 μg/ml. The blot was exposed for 1 sec after being developed. β-Catenin. Extracts were resolved on a 5% PAG and β-catenin was detected by incubation with monoclonal anti-β-catenin clone 14 at a final concentration of 0.5 μg/ml. The blot was exposed for 6 sec. p120ctn. Extracts were resolved on a 7.5% PAG and p120ctn was detected by incubation with monoclonal anti-p120cts clone 98 at a final concentration of 0.25 μg/ml. The blot was exposed for 6 sec. α-Catenin. Extracts were resolved on a 7.5% PAG and α-catenin was detected by incubation with monoclonal anti-α-catenin clone 5 at a final concentration of 1 μg/ml. The blot was exposed for 1 min. γ-Catenin. Extracts were resolved on a 7.5% PAG and γ-catenin was detected by incubation with monoclonal anti-γ-catenin clone 15 at a final concentration of 0.125 μg/ml. The blot was exposed for 6 sec. The migrations of molecular weight standards (205, 116, 97, and 84 kD) are indicated to the right of each blot.
Immunohistochemistry
No reaction was observed in control sections incubated without primary antibody. Moderate staining with the antibodies against α-catenin and γ-catenin was seen in ameloblasts at all stages of amelogenesis (Figure 4). Immunohistochemical staining using the monoclonal antibodies against E-cadherin, β-catenin, or p120ctn resulted in intense staining (brown/black) of cell contact sites in the middle layers of human oral epithelium (Figure 3). Similar reaction was observed in rat epithelium (not shown) in sections of forming rat incisors, confirming reactivity of the antibody with both species. Antibodies to α-catenin and γ-catenin moderately stained cell-cell surfaces most strongly in middle layers of human epithelium, with weaker staining of basal cells (Figure 3).
Positive controls. Immunohistochemical staining of human oral epithelium with antibodies to E-cadherin (
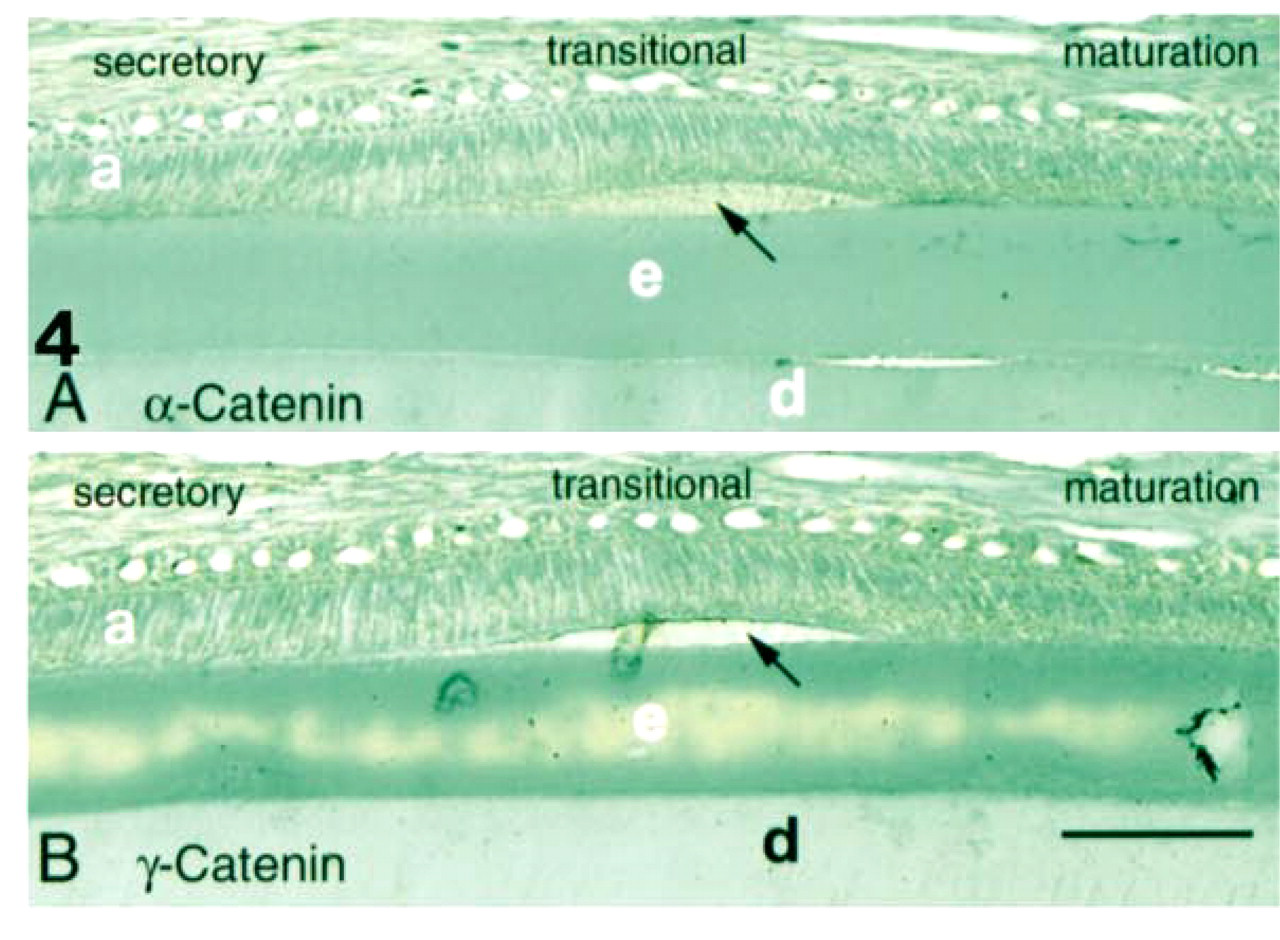
Staining of enamel organ with the anti-α-catenin (
Presecretory stage ameloblasts (Stage I) and stratum intermedium cells showed strong expression of E-cadherin (Figure 5A) and β-catenin (Figure 5B). Ameloblasts, but not stratum intermedium cells, bound anti-p120ctn at this stage (Figure 5C). Although diffuse staining was observed throughout the cytoplasm of ameloblasts, staining was concentrated along basolateral cell membranes. Odontoblasts weakly expressed β-catenin (Figure 5B) but not E-cadherin (Figure 5A).
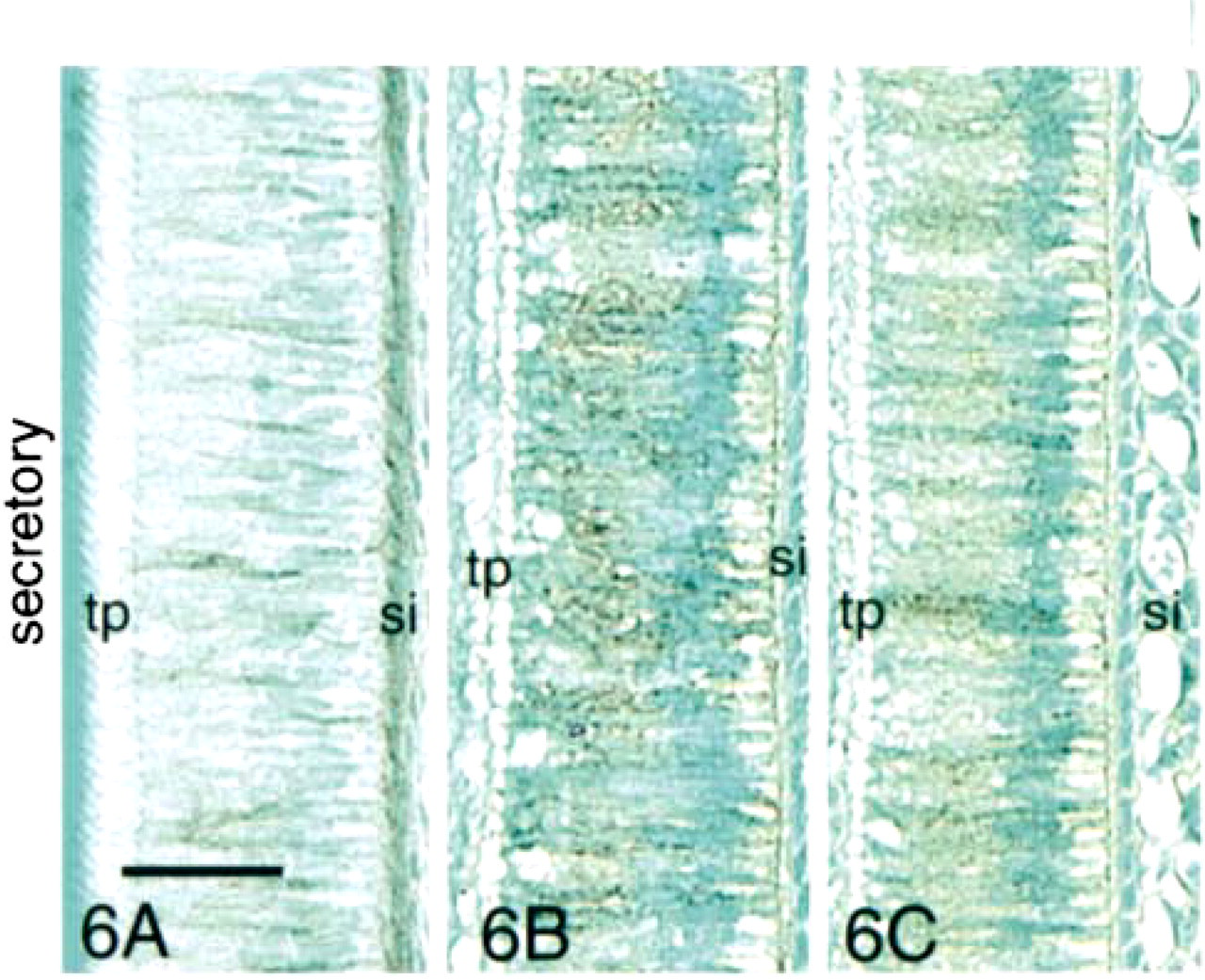
In early secretory-stage ameloblasts, the expression of E-cadherin, β-catenin, and p120ctn decreased at the stage where Tomes' processes were seen in the sections (Figure 5). Expression of E-cadherin and β-catenin persisted longer in stratum intermedium cells than in ameloblasts (Figures 6A and 6B), but staining of the former was greatly reduced in the late secretory stage (Figures 7A and 7B).
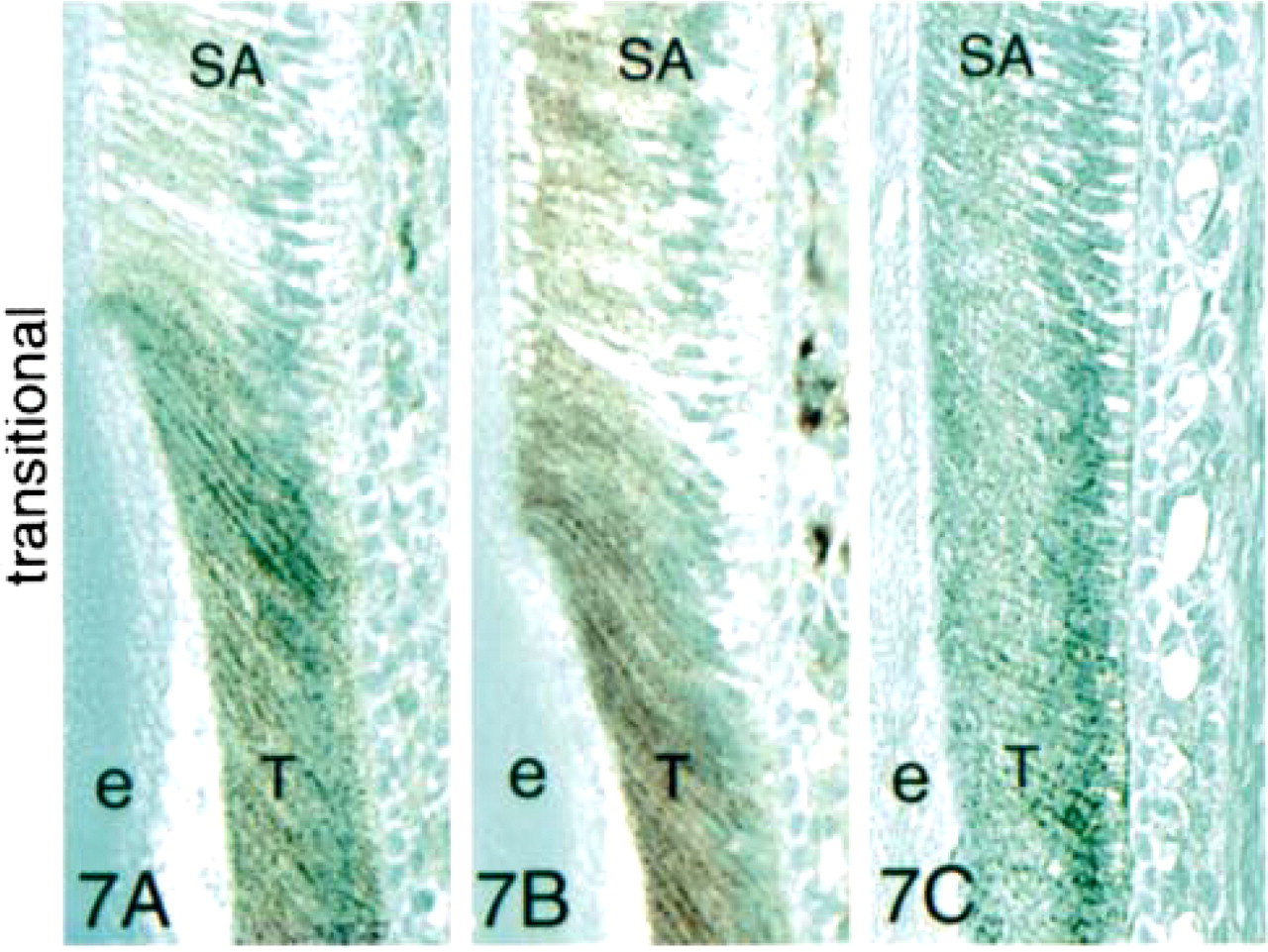
Transition-stage ameloblasts (Stage III) showed strong reactions with the anti-E-cadherin (Figure 7A), anti-β-catenin (Figure 7B), and anti-p120ctn (Figure 7C) antibodies, with the most intense staining along the lateral cell membranes. We observed no staining with these antibodies of any other cell type in the transition-stage enamel organ.
In Stage IV (maturation stage), weak expression of E-cadherin was observed along lateral borders of ameloblasts and in the apical and terminal web junctional complexes (Figure 8A). At this stage, the only significant reaction observed with the anti-β-catenin antibody was with the distal papillary cells (Figure 8B). Proximal papillary cells (those adjacent to the ameloblasts) showed little or no reaction with either the anti-E-cadherin or the anti-β-catenin antibody (Figures 8A and 8B). A slight reaction was observed with the anti-p120ctn antibody in maturation-stage ameloblasts and distal papillary cells.
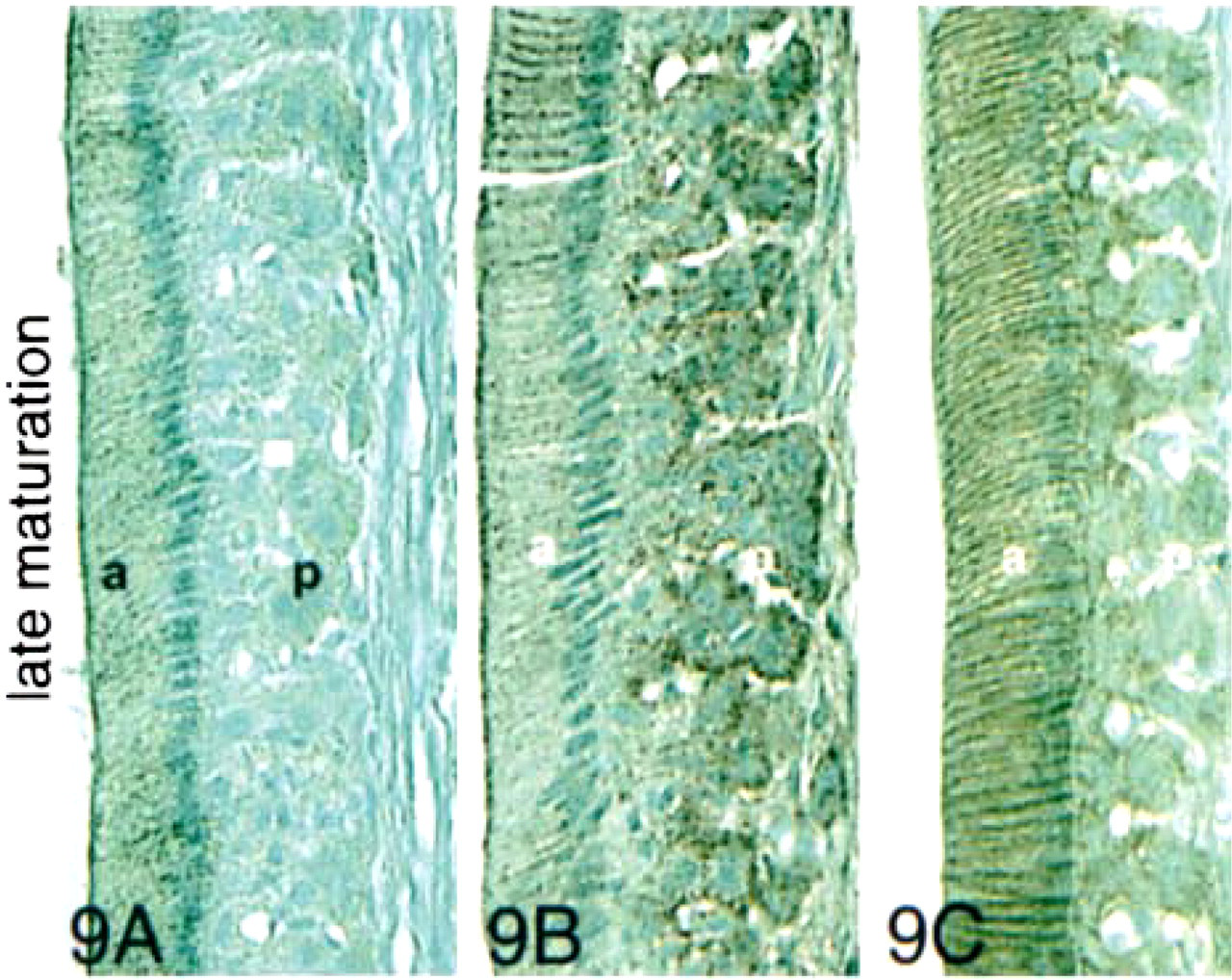
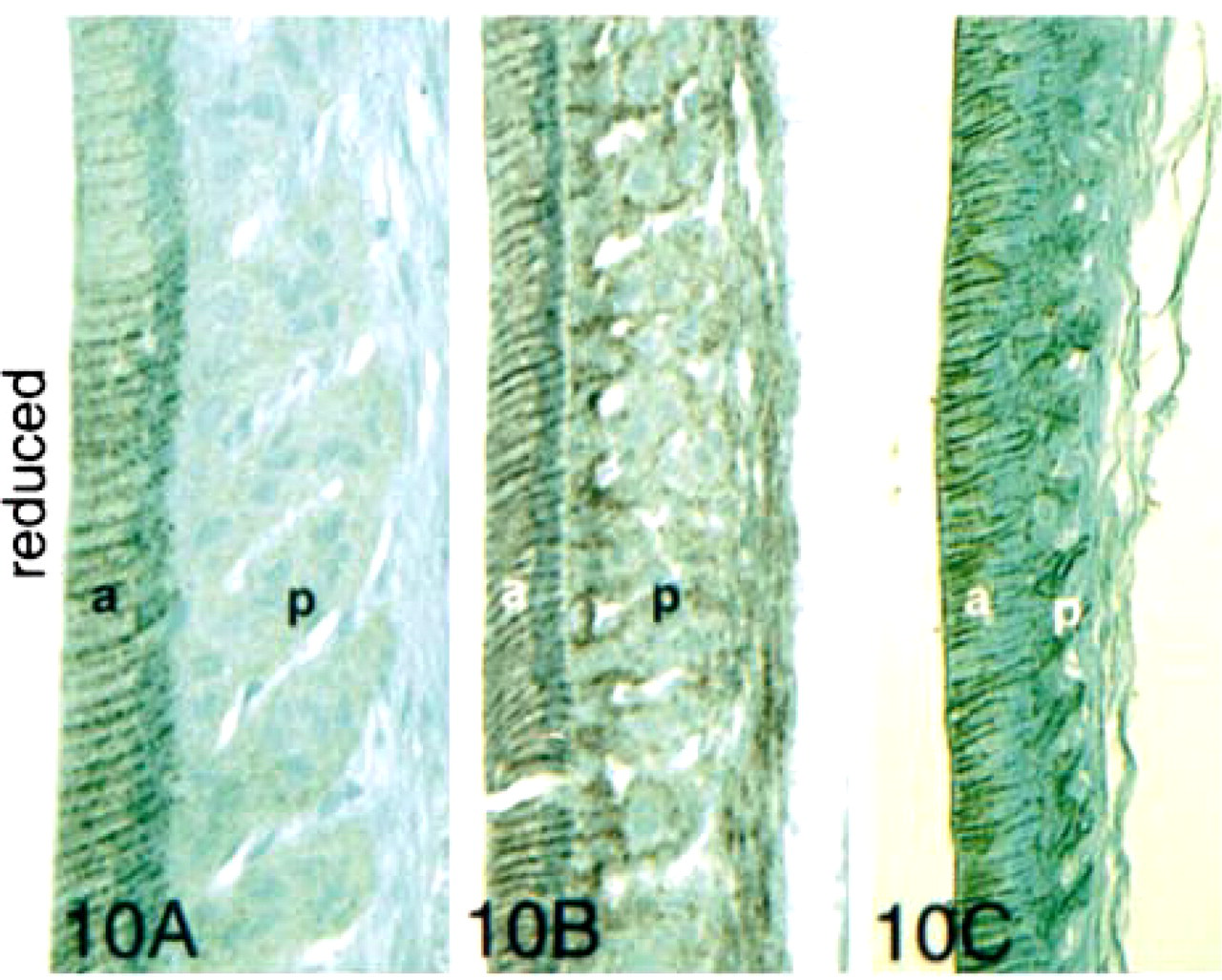
In the late maturation stage (Figure 9A) and in the reduced stage (Figure 10A), ameloblasts showed strong membrane-localized expression of E-cadherin, but the papillary cells reacted weakly. In late maturation and the reduced stage, both ameloblasts and papillary cells reacted with anti-β-catenin (Figures 9B and 10B) and p120ctn (Figures 9C and 10C).
Double staining with anti-E-cadherin (purple) and anti-APC (dark brown) confirmed the alternating expression of these two proteins (Figure 11). E-cadherin was expressed in ameloblasts in the early presecretory stage, and APC was expressed as ameloblasts differentiated into the secretory stage, at the site where dentin formation is first seen in the sections.
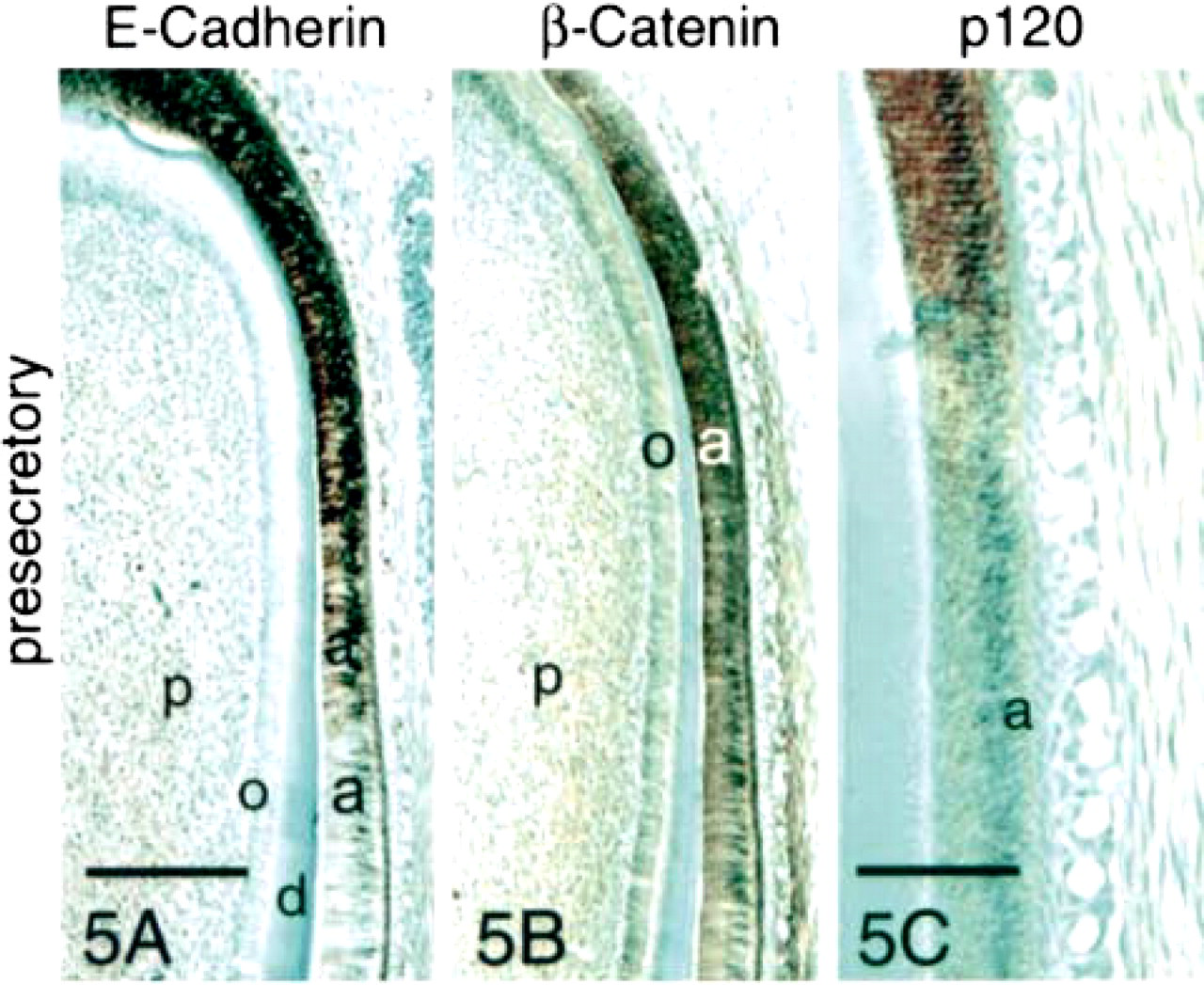
Immunohistochemical staining of presecretory-, secretory-, and transition-stage enamel organ sections with antibodies to E-cadherin, β-catenin, and p120ctn. Strong reaction of all three antibodies was observed with presecretory- and transition-stage ameloblasts. The reactions were localized at cell surfaces of both ameloblasts and stratum intermedium cells (si). Little expression of E-cadherin or these two catenins was observed in the secretory stage. However, all three proteins persisted in the basal terminal web complexes and in stratum intermedium cells in the secretory stage before fading. Odontoblasts (o) did not react with the anti-E-cadherin antibody but did express β-catenin. a, ameloblasts; d, dentin; e, enamel; p, pulp; tp, Tomes processes; SA, secretory ameloblasts; T, transition-stage ameloblasts. Bar in 5A = 200 μm and also applies to 5B. Bar in 5C = 100 μm. Bar in 6A = 50 μm and also applies to Figures 6 and 7.
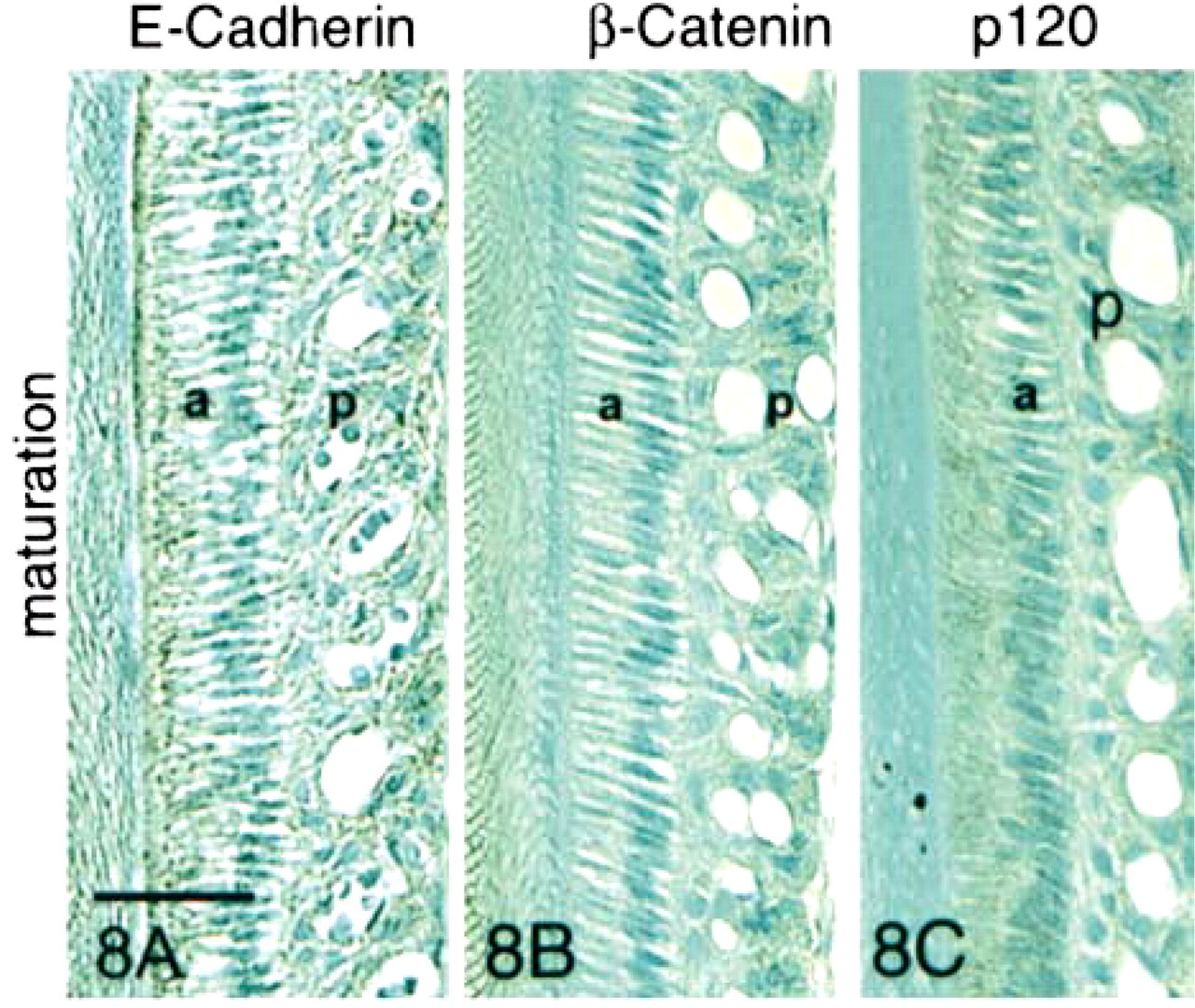
Immunohistochemical staining of maturation- and reduced-stage enamel organ sections with antibodies to E-cadherin, β-catenin, and p120ctn. In the early maturation stage, very little E-cadherin was expressed. The observed E-cadherin expression was localized at the basal junctional complexes of smooth-ended ameloblasts and the apical borders of ruffle-ended ameloblasts. The reaction was also seen along cell membranes of distal, but not proximal, papillary cells (p). In the late maturation stage, expression of E-cadherin and both catenins reappeared strongly in ameloblasts (a) and papillary cells (p). Staining was particularly intense at the periphery of the cells and at the apical and basal junctional complexes of the ameloblasts. In the papillary cells, E-cadherin expression persisted in the distal cells only, and β-catenin expression was strongly upregulated at the surfaces of all papillary cells. In the reduced-stage enamel organ, expression of all three proteins was intense at lateral cell surfaces. Bar = 50 μm.

Double staining of late presecretory-stage ameloblasts for E-cadherin (purple) and APC (dark brown). Early presecretory-stage ameloblasts stained with the antibody to E-cadherin. At the site where dentin (d, pale blue layer) was first deposited (arrow), dark brown staining (APC) began to appear. Odontoblasts (o) did not stain with either antibody. Bar = 100 μm.
Discussion
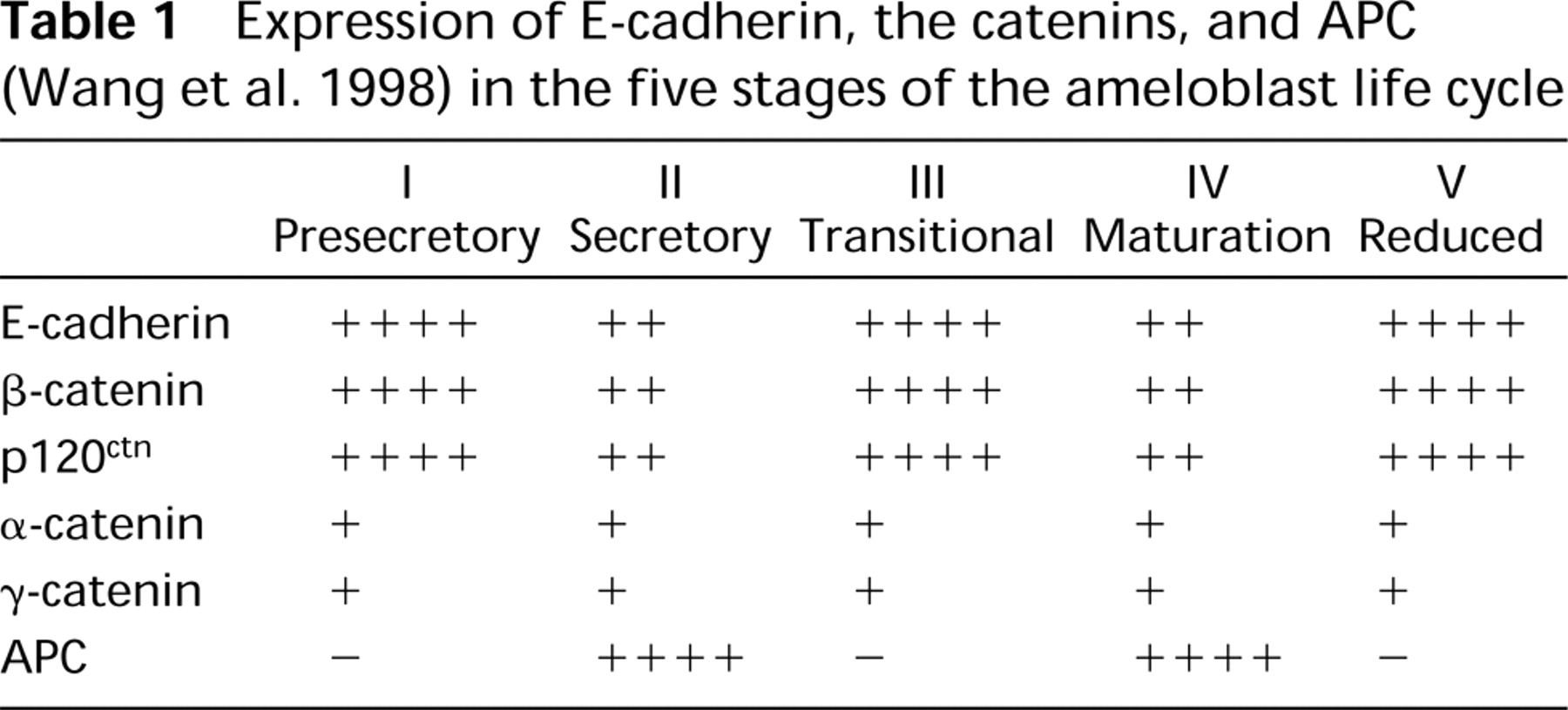
E-cadherin expression in mouse embryo craniofacial development has been reported as early as Day 14 (Luning et al. 1994). Patterns of expression in early mouse tooth development (bud, cap, and bell stages) have also been reported (Palacios et al. 1995). Here we report on the expression of E-cadherin and the catenins at later stages of tooth development in the continuously erupting rat incisor. We found that E-cadherin, β-catenin, and p120ctn were expressed at high levels in presecretory, transitional and reduced enamel organ stages, but at much lower levels in the intervening secretory and maturation stages (Figures 5-11; Table 1). In contrast, APC, which competes with E-cadherin for β-catenin binding (Hulsken et al. 1994), is expressed at high levels only in secretory (Figure 11) and maturation-stage ameloblasts (Wang et al. 1998). We detected no E-cadherin expression in odontoblasts, consistent with the observation that E-cadherin is generally expressed only in epithelia in the adult (Thiery et al. 1984; Levi et al. 1987). Changes in expression of α-catenin and γ-catenin during amelogenesis were less dramatic (Figure 4).
We observed high levels of E-cadherin and β-catenin expression in cells altering their differentiation status but participating in relatively stable adhesion (presecretory, transition, and reduced stages, or Stages I, III, and V). This is consistent with the importance of E-cadherin and β-catenin in mediating the formation of relatively stable junctions between epithelial cells and in transmitting differentiative signals. It contrasts sharply with the low expression of E-cadherin and β-catenin, but high levels of APC expression were observed in secretory-stage cells (Stage II) that are actively moving relative to their neighbors or in maturation-stage cells (Stage IV) that frequently alter their apical and basal junctions (Wang et al. 1998). One of the known functions of APC is downregulation of β-catenin. This may limit the availability of β-catenin to stabilize E-cadherin-dependent cell-cell junctions (Hulsken et al. 1994; Rubinfeld et al. 1995). Another reported effect of decreasing cytoplasmic β-catenin levels is decreased nuclear translocation and transmission of differentiative signals by transcriptional activation (Heasman et al. 1994; Funayama et al. 1995; Miller and Moon 1997; Fagotto et al. 1998).
APC has been reported to perform more than one function within the same cell (Senda et al. 1996). Therefore, APC may also be important in nucleation of microtubule formation in ameloblasts. Microtubules are critical for ameloblast function in secretory (Karim and Warshawsky 1979) and maturation stages (McKee and Warshawsky 1986; Takuma et al. 1987). This is clearly consistent with the intense expression of APC we reported in secretory- and maturation-stage ameloblasts (Wang et al. 1998) and with the alternating pattern of E-cadherin and APC expression we report here. The decreased expression of β-catenin observed at stages when APC expression is high is consistent with the reported function of APC in down-regulating β-catenin levels via proteasomal proteolysis (Aberle et al. 1997; Salomon et al. 1997).
Expression of E-cadherin, the catenins, and APC (Wang et al. 1998) in the five stages of the ameloblast life cycle
In summary, α-catenin and γ-catenin are present in ameloblasts throughout amelogenesis. The staining intensity of the other components of the cadherin-catenin complex alternates through the stages of the enamel organ life cycle. Presecretory-stage (I) ameloblasts, undergoing their initial differentiation, express E-cadherin strongly (Figure 5A) but have no detectable APC expression (Wang et al. 1998). In the secretory stage (II), when cell-cell adhesion must be downregulated to allow the ameloblasts to slide past each other, an action that requires microtubule function (Nishikawa and Kitamura 1982, 1985), APC expression is upregulated (Wang et al. 1998) and E-cadherin is dramatically downregulated (Figure 6A). In the transition stage (III), where the ameloblast morphology changes again, E-cadherin is strongly re-expressed (Figure 7A) and APC expression disappears (Wang et al. 1998). In the maturation stage (IV), when junction formation alternates between the apical and basal terminal web complexes, E-cadherin is once more downregulated (Figure 8A) and strong APC expression reappears (Wang et al. 1998). In the maturation-stage enamel organ, distal papillary cells stained with the anti-E-cadherin and anti-β
The rat incisor enamel organ is the only tissue we are aware of in which the expression of APC and the cadherin-catenin complex alternates several times during the life cycle of the cells. The continuously erupting rat incisor thus offers obvious advantages for studying both the mechanisms of regulation of expression of these proteins during development and their functions.
Footnotes
Acknowledgments
Supported by The Forsyth Institute and by the Colgate Palmolive Company.
