Abstract
CREB-binding protein (CBP) was examined in ameloblasts and in other enamel organ-derived cells of the rat incisor, using Western blotting analysis and immunocytochemistry by specific antibodies. Western blotting of labial tissues, including ameloblasts of the incisors, detected a single band with a molecular weight equivalent to the reported value of CBP. In immunocytochemistry, CBP was localized in ameloblast nuclei in the maturation zone but not in the secretion and transition zones. The nuclei of the other enamel organ-derived cells were also positive. Because this protein is suggested to take part in c-Jun-mediated transcription, the present study and the results of a previous report showing c-Jun localization in the nuclei of enamel organ-derived cells suggest that the enamel organ-derived cells, including maturation ameloblasts, undergo active transcriptional regulation.
I
It was shown in transfection experiments that influxes of calcium by opening of L-type calcium channels represent a pathway for c-Jun activation and that stimulation of c-Jun-mediated transcription requires CREB-binding protein (CBP) function (Cruzalegui et al. 1999). CBP is a general transcriptional co-activator known to interact with a wide variety of transcriptional factors, including CREB and AP-1 (Shikama et al. 1997; Chan and La Thangue 2001). The nuclei of the maturation ameloblasts exhibit strong c-Jun im-munoreactivity in the rat incisor (Nishikawa 2000). Therefore, anti-CBP immunohistochemistry was performed in this study. Smith et al. (1987) demonstrated that post-secretory ameloblasts exhibit two different appearances in the maturation zone. They are referred to as ruffle-ended ameloblasts (RA, major cell appearance with ruffled border at apical cell end) and smooth-ended ameloblasts (SA, minor cell appearance occupying one fifth of the total maturation zone) (Josephsen and Fejerskov 1977). Moreover, c-Jun labeling is stronger in RA than in SA (Nishikawa 2000). A large part of RA is considered to contribute to mineral uptake in the enamel matrix, although SA contributes to some passive calcium uptake (Reith and Boyde 1981; Takano et al. 1982). These patterns imply that different transcriptional regulation occurs in RA and SA regions. In this study, transcriptional co-activator CBP was localized in the rat incisor cell nuclei of the enamel organ, including ameloblasts (mostly in the maturation zone) by immunocytochemistry, and the presence of CBP in the incisor labial tissues, including ameloblasts, was confirmed by Western blotting analysis. The results suggest that strong transcriptional regulation occurs in maturation ameloblasts and the other enamel organ cells of the rat incisor.
Materials and Methods
Sixteen male Wistar rats (7–8 weeks, 160–260 g) (Jcl Wistar; Clea Japan, Tokyo, Japan; institutional guidelines were followed) were used for immunocytochemistry. The animals were perfused under sodium pentobarbital (Nembutal; Abbott, North Chicago, IL) anesthesia with 4% paraformaldehyde in 0.1 M phosphate buffer solution (PB), pH 7.2, via the left ventricle at room temperature (RT) for 10 min. They were immersed in the same fixative at 4C for 2 hr and the maxillary and mandibular incisors were dissected. After being washed with 0.1 M PB, the teeth were demineralized with 5% disodium EDTA solution adjusted to pH 7.3 with sodium hydroxide solution and were placed at 4C for 3–4 weeks. Demineralized incisors were cut transversely into two segments. Both segments were immersed in 25% sucrose in PBS overnight, rapidly frozen, and then cut longitudinally (6–8 μm thick) using a cryotome.
Two different antibodies against CBP were used in this study: a monoclonal antibody (MAb CBP(C-1) against the carboxy terminus of human CBP, and a polyclonal antibody (PAb) CBP(A-22) against the amino terminus of human CBP. The closely related protein p300 was also examined by a PAb p300(C-20). Because CBP is known to be a CREB-binding protein, CREB immunocytochemistry was performed with a PAb CREB-1(C-21) (Table 1). All were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). The polyclonal c-jun/AP-1(Ab-1) antibody was purchased from Oncogene Research Products (Cambridge, MA) (Table 1).
Specificity of the antibodies used in this study
Cryosections on glass microscopic slides were incubated for 20 min with 0.6% H2O2 in 80% methanol to inhibit endogenous peroxidase activity. These were labeled with the appropriate antibody, as described above, diluted with 1% bovine serum albumin in PBS (BSA–PBS) at RT for 1 hr. They were then labeled with HRP-conjugated anti-rabbit or anti-mouse IgG (Cappel; West Chester, PA) diluted 1:100 with 1% BSA-PBS for 30 min. In addition, some specimens were labeled with FITC-conjugated anti-mouse IgG (Cappel). HRP-labeled sections were incubated with a solution composed of 0.5 mg/ml diaminobenzidine (DAB) and 2 mg/ml (NH4)2 Ni(SO4)2 6H2O in Tris-HCl (pH 7.6) for 10 min, then with the DAB solution containing 0.005% H2O2 for 2–12 min. Some FITC-labeled sections were further labeled with rhodamine–phalloidin (Molecular Probes; Eugene, OR) to detect RA and SA (Nishikawa and Josephsen 1987). Control sections were processed in the same manner, with the exception of omission of the primary antibodies.
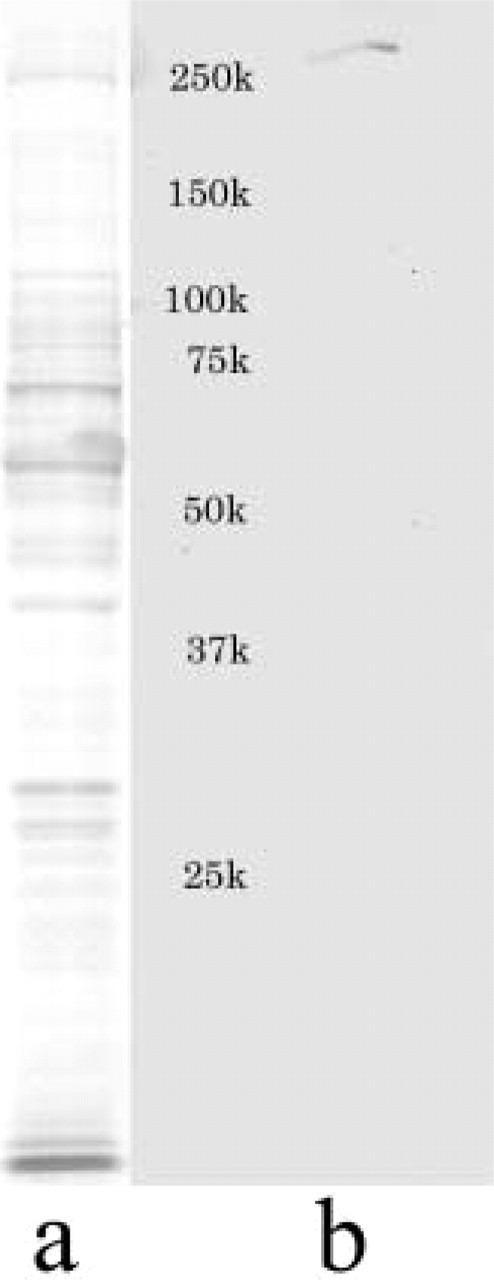
Western blotting analysis of CBP. SDS-PAGE pattern (
For Western blotting analysis, maxillary and mandibular incisors were dissected from three adult male Wistar rats (7–10 months, 480–580 g) under diethyl ether inhalation. The labial surface of the incisors containing ameloblasts and other enamel organ epithelia was scraped with a razor blade. Tissues including enamel organ epithelia were dissolved in a sample buffer composed of 10 mM Tris-HCl (pH 8.0), 1 mM EDTA, 25% glycerin, 1% SDS, and 5% 2-mercaptoethanol, and then they were incubated at 100C for 5 min. They were subjected to SDS-PAGE using a 5–20% gradient slab gel and the procedure of Laemmli (1970) under reducing conditions. Electrotransfer of proteins onto Durapore GVHP membrane (Millipore; Yonezawa, Japan) was performed at 4C by a transblot apparatus (Marysol; Tokyo, Japan) for 60 min at 100 mA, according to Towbin et al. (1974). The membrane was immersed in 1 X casein solution (Vector Labs; Burlingame, CA) and incubated with anti-CBP (A-22) antibodies, diluted 1:200 with 1 X casein solution at RT for 60 min. Then the membrane was incubated with biotinylated goat anti-rabbit IgG, followed by an avidin-biotin-peroxidase complex (Vector) according to the manufacturer's instructions (Vector ABC kit). Finally, the labeled peroxidase was developed in a solution composed of 0.5 mg/ml diaminobenzidine (DAB), 2 mg/ml (NH4)2Ni(SO4)26H2O and 0.005% H2O2 in 0.05 M Tris-HCl (pH 7.6) for 20 min. After transblotting, some of the slab gels were stained with 0.5% Coomassie Brilliant Blue R250 for detection of separated protein pattern. Molecular weights were estimated by Bio Rad Precision Protein Standards (broad range; Bio Rad, Hercules, CA).
Ameloblasts in the late maturation zone from two neighboring sections of the same incisor were labeled with the antibodies against CBP(C-1) (
Results
In Western blotting analysis using the anti-CBP (A-22) antibody and epithelial tissues of rat lower incisors, including ameloblasts, a single band was detected slightly above 250 kD on the membrane (Figure 1), which is equivalent to the reported electrophoretic value of CBP (265 kD) (Chrivia et al. 1993; Arias et al. 1994).
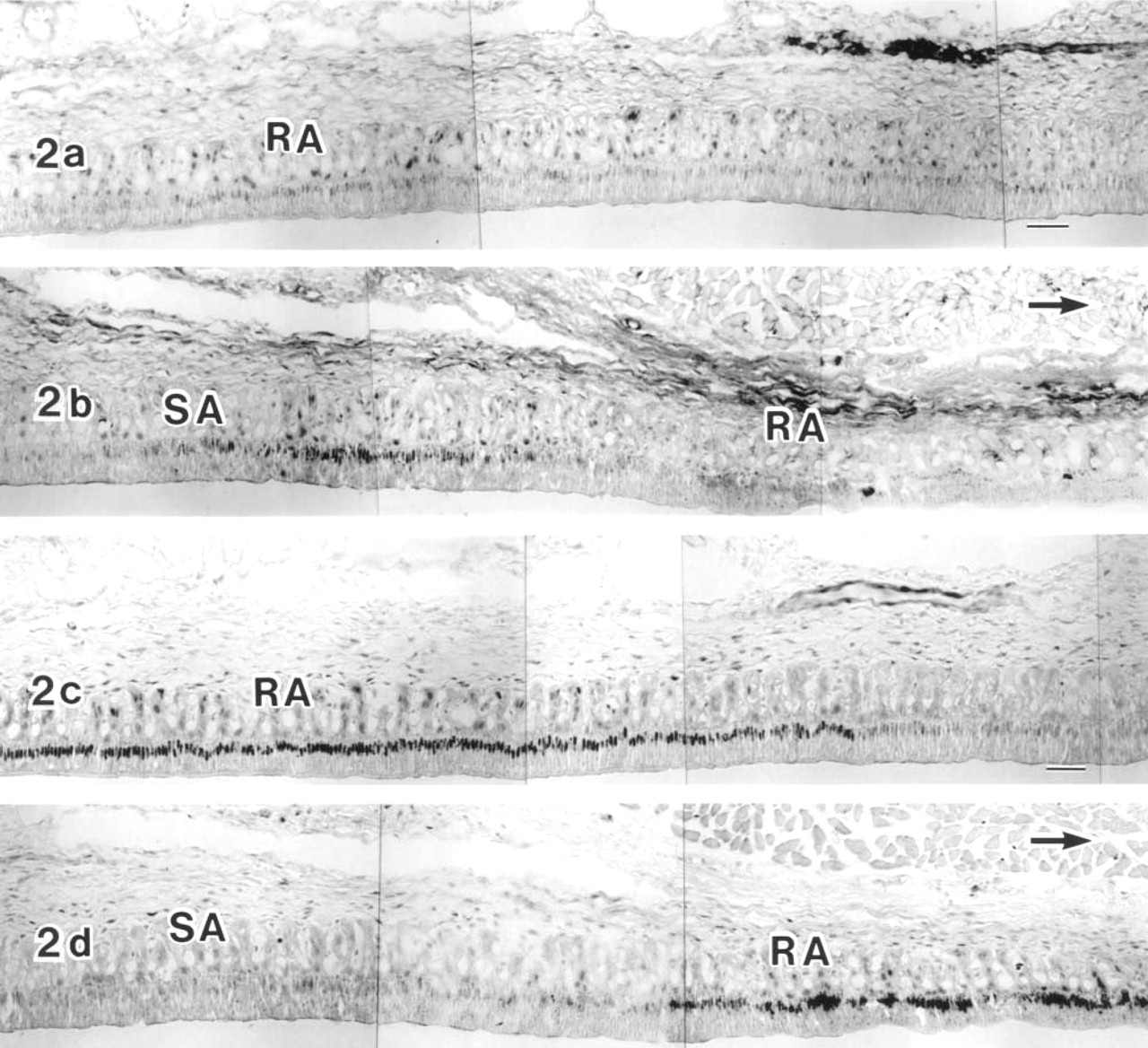
In immunocytochemistry, CBP (C-1) was detected in the ameloblast nuclei of the maturation zone (Figures 2–4), but not of the secretion and transition zones (Figure 4b), although CBP (A-22) showed slight labeling in the secretion zone (Figure 4a) and the maturation zone. Because the overall labeling intensity of anti-CBP (C-1) was higher than that of anti-CBP (A-22), anti-CBP (C-1) was used for immunocytochemistry. In some specimens the labeling intensity was not uniform throughout the entire maturation zone. The intensity in the SA region tended to be stronger than in the adjacent RA region (Figures 2 and 3). Comparison of two neighboring sections of the same tooth, labeled either with anti-CBP (C-1) or anti-c-Jun antibody, showed that the labeling pattern of either antibody was complementary to that of the other. SA had stronger CBP reaction and weaker c-Jun reaction, whereas RA had stronger c-Jun reaction and weaker CBP reaction (Figures 2a–2d). Even in an RA zone, CBP labeling varied considerably in the ameloblast nuclei but exhibited no regular pattern. To confirm the preference of the CBP labeling in the SA zone, double labeling with anti-CBP (C-1) and rhodamine–phalloidin (Rh–Ph) was performed. The results showed that stronger CBP reactivity in the nuclei was related to ameloblasts with strong basal fluorescence of Rh–Ph (Figures 3a and 3b), which has been previously shown to be SA (Nishikawa and Josephsen 1987). In other specimens, however, the RA zone had CBP reactivity equivalent to that of SA, showing rather homogeneous labeling intensity throughout the maturation zone (Figures 4d and 4e). Nuclei of enamel organ-derived cells other than ameloblasts were also labeled with anti-CBP antibody (Figures 4a–4e), but the labeling intensity was variable.
Another transcriptional co-activator, p300, closely related to CBP, was examined immunocytochemically but no specific labeling was detected in the ameloblasts and the other enamel organ-derived cells. The prototypical target transcription factor CREB was not detected immunocytochemically in the ameloblasts and the other enamel organ-derived cells (data not shown). Because the antibody used also reacted to CREM and ATF-1, these two CREB family proteins were also below detectable limits in the ameloblasts. Control sections did not show any specific labeling.
Discussion
In the maturation zone of amelogenesis, there is strong immunoreactivity of several AP-1 members (Nishikawa 2000). Fra-2, JunD, and c-Jun are remarkable in the ameloblast nuclei. Whereas Fra-2 and JunD are homogeneous through the long maturation zone of the incisor, c-Jun is stronger in the RA and weaker in the SA. Considering that AP-1 is regulatory for transcriptional factors induced by various stimuli, such as growth factors and UV irradiation (Angel and Karin 1991; Karin et al., 1997), it is suggested that ameloblasts in the maturation zone of the rat incisor undergo active transcriptional regulation and, furthermore, that RA and SA undergo a different transcriptional regulation (Nishikawa 2000). The morphological characteristics of maturation ameloblasts support several functions, i.e., active or passive mineral transport and reabsorption followed by degradation of transitory enamel proteins, although matrix removal is performed in the early phase of the maturation zone (Takano et al. 1982; Robinson et al. 1995; Smith and Nanci 1995; Smith 1998). AP-1 in the ameloblast nuclei throughout the long maturation zone is therefore likely to relate to the elevation of mineral content in enamel. In this study, to understand AP-1-related transcriptional regulation, the transcriptional co-activator CBP was localized in the ameloblasts of the rat incisor. In the transfection experiment, c-Jun-mediated transcription required cytoplasmic influx of calcium ions, calcium–calmodulin-dependent protein kinase activation, and CBP function (Cruzalegui et al. 1999). The present study showed that CBP did localize in the nuclei of the ameloblasts mostly in maturation zone, whereas the possibility exists that loss of cytoplasmic CBP occurred during long sample incubation time in a EDTA solution for demineralization. Considering that c-Jun labeling is strong in the maturation zone (Nishikawa 2000), it is reasonable to speculate that CBP and c-Jun interact with each other, directly or indirectly, although the localization of CBP and c-Jun was complementary in some specimens in this study and this study did not provide any evidence suggesting any interactions of two proteins. Furthermore, CBP was also detected in the nuclei of enamel organ-derived cells other than ameloblasts. Because AP-1 components such as c-Jun were detected in these cells, active transcriptional regulation of these cells may be performed during amelogenesis, which implies the involvement of these cells in enamel formation.
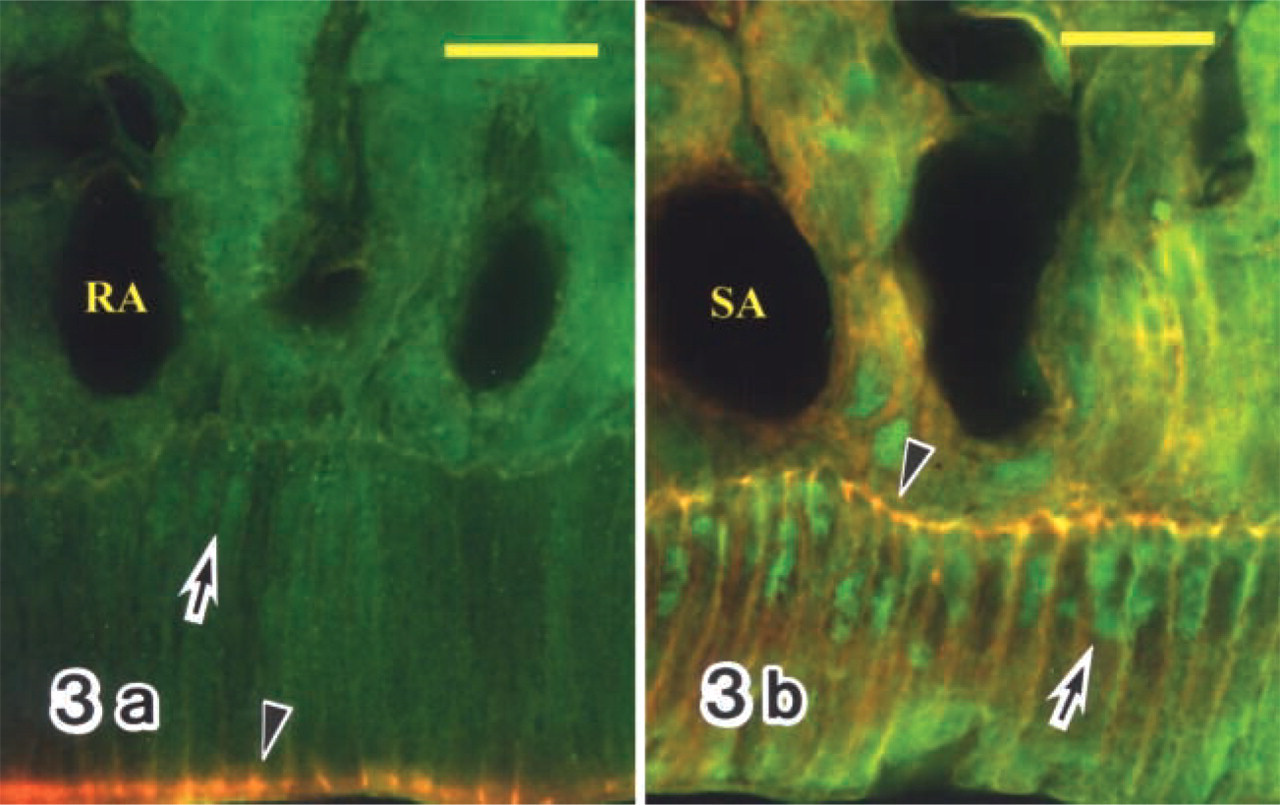
Fluorescence micrographs double labeled with anti-CBP antibody (C-1) followed by FITC-conjugated secondary antibody and rhodamine-phalloidin (Rh-Ph). RAs exhibit lower CBP labeling intensity in their nuclei (
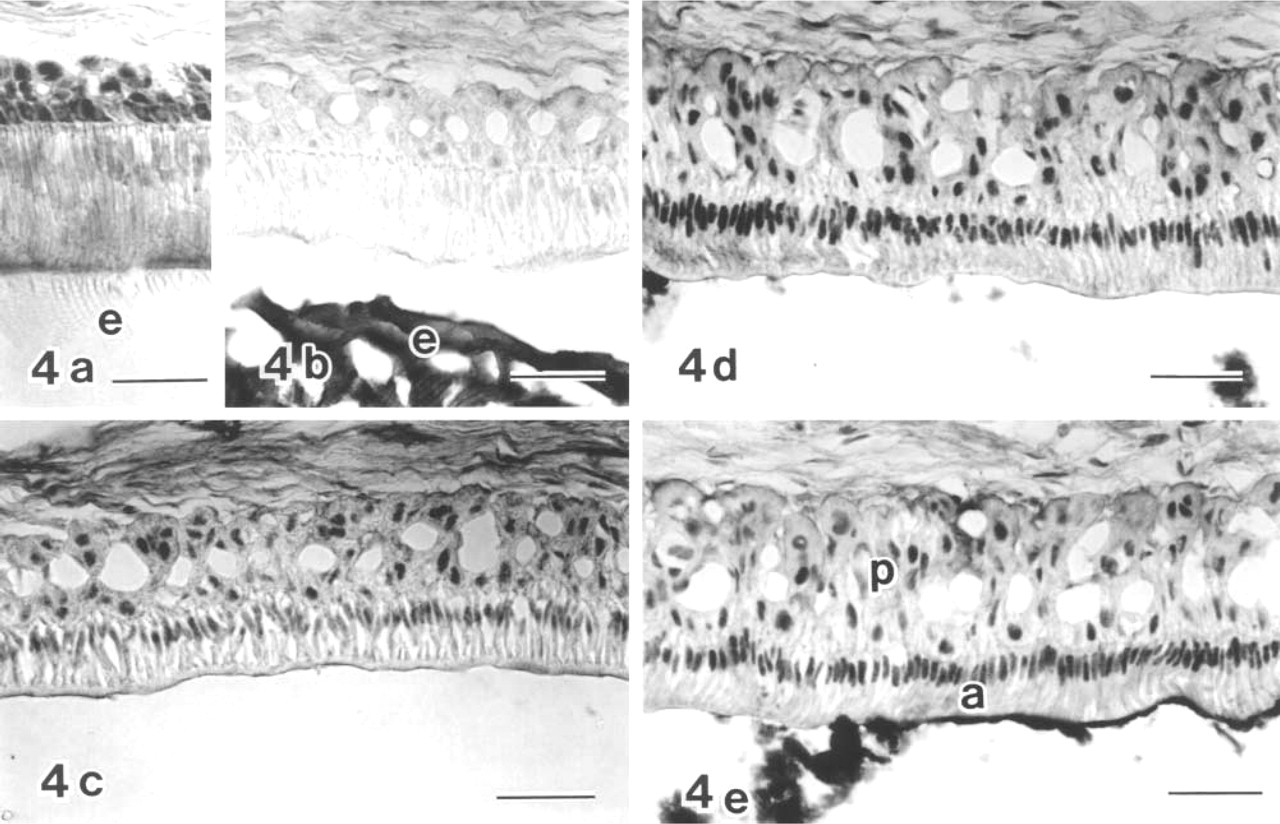
Anti-CBP (A-22) (
CBP is a transcriptional co-activator that binds to CREB phosphorylated by protein kinase A (Shaywitz and Greenberg 1999). However, it has been shown that CBP is able to bind diverse transcription factors such as c-Fos, c-Jun, NF-γB, and nuclear receptors, leading a role of CBP as a more general co-activator than was previously expected (Bannister et al. 1995; Kamei et al. 1996; Shikama et al. 1997; Zhong et al. 1998). In conclusion, CBP may function as a co-factor that participates in transcriptional activation of AP-1 and/or other factors in the maturation ameloblasts and the other enamel organ-derived cells. It is suggested that the maturation ameloblasts and the other enamel organ-derived cells of the rat incisor undergo active transcriptional regulation.
Footnotes
Acknowledgements
Supported in part by a grant-in-aid for scientific research (no. 11671824) from the Ministry of Education, Culture, Sports, Science and Technology of Japan, and by a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan to promote 2001-Multidisciplinary Research Projects in 2001–2005.
I thank Drs T. Tanabe and M. Fukae (Department of Biochemistry, Tsurumi University School of Dental Medicine) for Western blotting analysis.
