Abstract
The pediatric early warning score (PEWS) is a tool used to predict clinical deterioration. Referenced vital sign parameters are based on expert opinion but heart rate and respiratory rate percentiles in hospitalized children have been published. This retrospective case-control study of unplanned intensive care unit (ICU) transfers compares evidence-based vital signs (EBVS) effect on PEWS sensitivity and specificity, determines the impact of age categories on PEWS deterioration prediction, and evaluates whether EBVS PEWS is associated with need for invasive ICU supports. EBVS PEWS improved sensitivity (43%-71% vs 30%-63%) for unplanned transfers with slightly decreased specificity (88%-98% vs 93%-99%). Logistic regression analysis and odds ratios (ORs) demonstrated EBVS PEWS was associated with increased risk for ICU-specific supports (OR = 1.16, 95% confidence interval [CI] = 1.0-1.34, P = .0498). Evidence-based vital signs can improve PEWS sensitivity to identify unplanned ICU transfers and identify patients requiring ICU-specific interventions.
Keywords
Introduction
Early identification of clinical deterioration is critically important to decrease pediatric morbidity and mortality. Early warning systems (EWS) are tools comprised of objective vital signs and routine nursing assessments designed to predict clinical deterioration and possible need for intensive care unit (ICU) level care.1,2 Composite scores trigger escalation policies, including thresholds for multidisciplinary bedside huddles and rapid response team (RRT) activation. The PEWS is widely utilized in acute inpatient settings as a predictor of deterioration2-5 on both general medical and subspecialty services. 6
Heart rate (HR) and respiratory rate (RR) measurements are essential to clinical assessments in acute illnesses and are key components of composite PEWS. Accurate reference ranges are essential to identifying abnormal values. While there exist widely referenced resources for pediatric vital sign ranges,7-10 supporting evidence for these data is generally weak 11 and sources are not well defined. Moreover, there are discrepancies between these commonly referenced sources and vital sign data derived from patients admitted to acute care units. 12 Even among early studies, there is variability in the vital signs utilized for PEWS scoring, if these values are listed at all.1,2,5,13
Bonafide et al determined standardized HR and RR percentile curves based on data collected from 116, 000 vital signs of 14, 000 hospitalized children. 12 This study illustrates the discrepancy between EBVS reference ranges for hospitalized children compared with those previously described in healthy pediatric outpatients. 11 Although a previous study showed no significant improvement on bedside PEWS predictions of deterioration using EBVS, 14 no study to date has compared the impact of these vital sign parameters on the Brighton PEWS model. Therefore, we aimed to assess whether PEWS calculated using EBVS was better able to identify patients at risk for deterioration compared with vital signs derived from published reference ranges (institution-based vital signs, IBVS) in children. We sought to determine whether more refined age categories, which would better account for physiologic changes across the pediatric age spectrum, would increase PEWS prediction of deterioration independent of vital sign criteria. Finally, we aimed to demonstrate whether there was an association between a need for ICU-specific therapies following transfer and a change in PEWS calculated using EBVS.
Methods
Study Design
We conducted a retrospective case-control study of patients who experienced unplanned ICU transfers between April 2016 and February 2017 at a 323-bed stand-alone, quaternary care urban, academic children’s hospital. This study was reviewed and approved by the Institutional Review Board at our institution.
Study Population and Design
Cases were defined as patients experiencing unplanned escalations of care from acute care medical and surgical units to the ICU via RRT or code team activation. We included patients transferred to the neonatal, pediatric, and cardiovascular ICUs. We selected cases from a patient database compiled by the Pediatric Early Recognition and Resuscitation Committee (PERRC) at our institution. We excluded cases aged >18 years, second ICU transfer during a hospitalization, length of stay >60 days at time of transfer, or planned ICU transfer (eg, expected post-operative care). The highest total PEWS in the 24 hours prior to ICU transfer was rescored. If multiple scores with equivalent values were present one score was selected at random for rescoring. Controls were randomly matched by primary admission service, age, hospital length of stay, and date of transfer (within 2 weeks). Cases that could not be matched exactly to hospital day at ICU transfer were instead matched to controls of equal or greater hospital length of stay to avoid replacement. Controls were selected from a Research Electronic Data Capture (REDCap)15,16 database compiled by PERRC and hosted by our institution, which stores maximum PEWS values for all hospitalized patients each day.
Late rescues were defined as patients who received ICU-specific therapies including intubation, mechanical ventilation, vasopressor initiation, and/or cardiopulmonary resuscitation within 12 hours of ICU transfer. These patients were detected with the use of an automated trigger tool 17 that screens for unplanned ICU transfers. Each unplanned transfer was manually screened for presence of late rescue criteria. However, all cases received ICU-level care following transfer, which may include intubation or initiation of inotropic medications after the 12-hour late rescue window.
Vital Sign and Pediatric Early Warning Score Criteria
PEWS in our institution is based on the Brighton PEWS model 1 and has been in use since 2008. Current IBVS have been in use since 2012. They were determined by an expert panel of experienced nurses and critical care physicians who referenced vital sign parameters from other institutions in addition to Pediatric Advanced Life Support (PALS) vital sign guidelines (Table 1). PEWS is utilized exclusively on acute care medical and surgical units and is based on vital sign and routine nursing assessments in 3 domains: behavior, respiratory, and cardiovascular. Possible scores range from 0 to 3 in each domain based on degree of abnormality. Additional points are added to summative PEWS for presence of tracheostomy, continuous albuterol therapy for ≥12 hours, and/or for persistent post-operative emesis (Supplemental Table 1). Summative PEWS of 0 to 2 results in routine monitoring at 4-hour intervals; scores of 3 require physician notification and re-evaluation within 2 hours; scores of 4 or individually scoring 3 in a single category require prompt bedside evaluation by the primary team, bedside nurse, and charge nurse; and scores of 5 or greater mandate RRT activation. The RRT is composed of a PICU fellow or advanced practitioner, PICU charge nurse, and a PICU-based respiratory therapist. PEWS was calculated automatically by the electronic health record once all components were documented by the bedside nurse. The automated process for calculating PEWS within the electronic medical record has been previously published. 18
Institution-Based Vital Sign Criteria for Hospitalized Children.
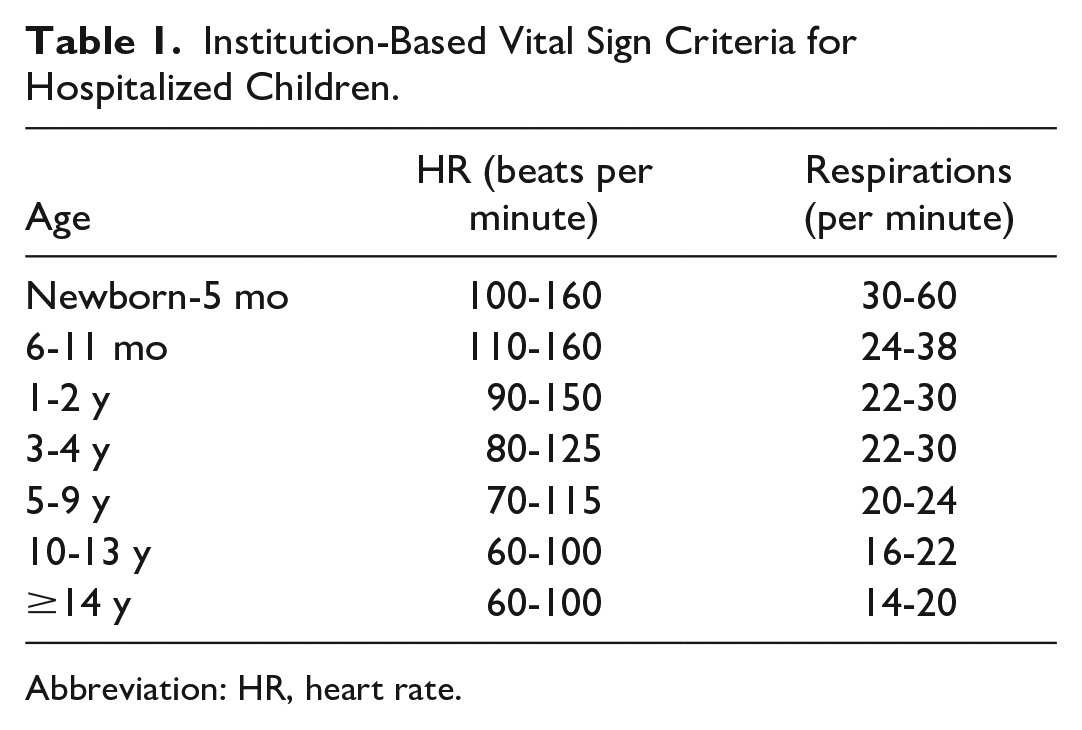
Abbreviation: HR, heart rate.
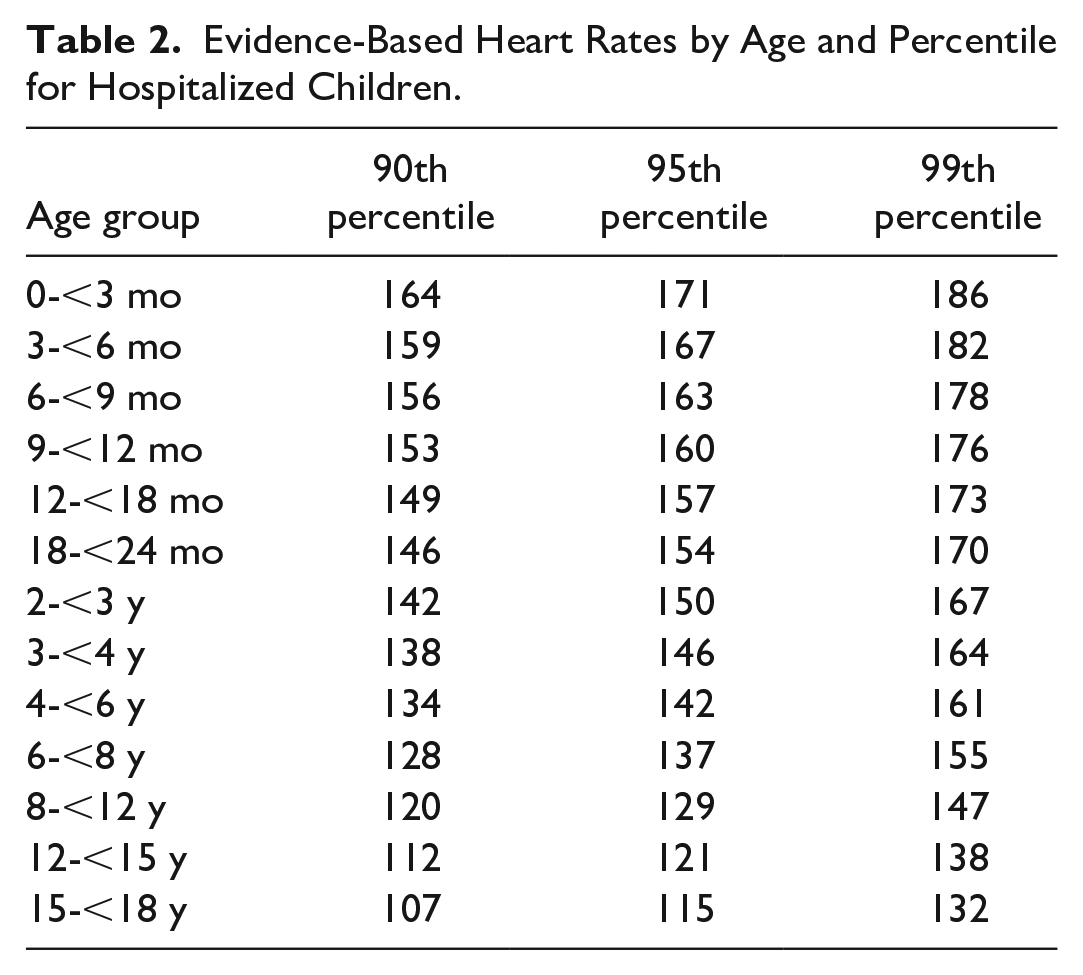
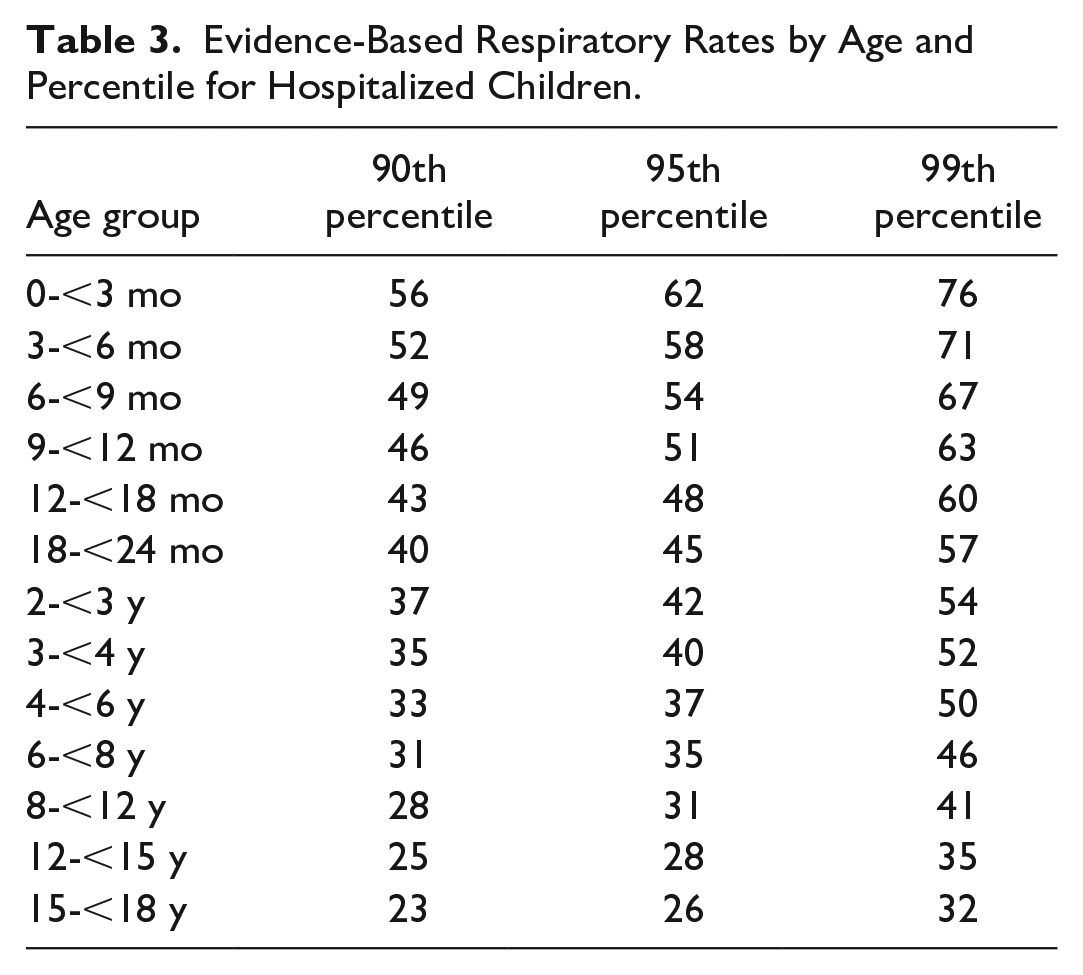
Evidence-based vital signs used in the comparison for this study were based on normal distribution curves for vital signs presented by Bonafide et al 12 (Tables 2 and 3). Pediatric early warning score values for EBVS were assigned as follows: <90th percentile = 0; 90th to 94th percentile = 1; 95th to 98th percentile = 2; and ≥99th percentile = 3. Subjective nursing assessments of mental status, perfusion, and respiratory effort were unchanged in their contribution to revised PEWS. The 5 age groupings used in the current IBVS system were, in addition, compared with the 13 subdivided groups presented by Bonafide in the EBVS system.
Evidence-Based Heart Rates by Age and Percentile for Hospitalized Children.
Evidence-Based Respiratory Rates by Age and Percentile for Hospitalized Children.
Statistical Methods
Characteristics data were reported in summary statistics as mean and standard deviation (SD) or median and interquartile range (IQR) for continuous variables, and frequency and proportion for categorical variables. Primary admission service, age, gender, hospital length of stay, and date of transfer were compared between case and control groups using fixed effects regression. Paired t-tests compared the differences between IBVS and EBVS PEWS within each case and control group. Sensitivity and specificity with 95% confidence intervals (CI) for unplanned transfer were calculated for IBVS and EBVS PEWS. To account for matching in this case-control study, conditional logistic regression including matching variables as covariates was used to calculate the receiver operating characteristic (ROC) area under the curves (AUCs) and reported with the corresponding 95% CIs, and differences between AUCs were compared using the chi-square test. Logistic regression was applied to examine the influence of the breakdown of age categories by including the interaction term of age category and PEWS, and was run separately for case and control groups. Late rescue case analysis was carried out by logistic regression for IBVS and for EBVS, respectively. Odds ratio and 95% CI were used to assess the effect of PEWS to predict patients at risk for late rescues. All data analyses were carried out by SAS software version 9.2 (SAS Institute Inc., Cary, NC). A P value of ≤.05 was considered statistically significant.
Results
Study Population Characteristics
During the study period, PEWS of 400 unique ICU transfers and 800 controls was analyzed and rescored. Characteristics of this population are shown in Table 4. There were no statistically significant differences in patient sex (P = .86), mean age (P = .25), or primary service at the time of ICU transfer (P = .58). There was a statistically significant difference in hospital day at transfer as some controls were not able to be matched exactly to length of hospital stay at case transfer while preserving other matching criteria (P = .02). Table 5 shows the distribution of patients by age category in both institutional and data-driven models.
Characteristics of the Study Population.
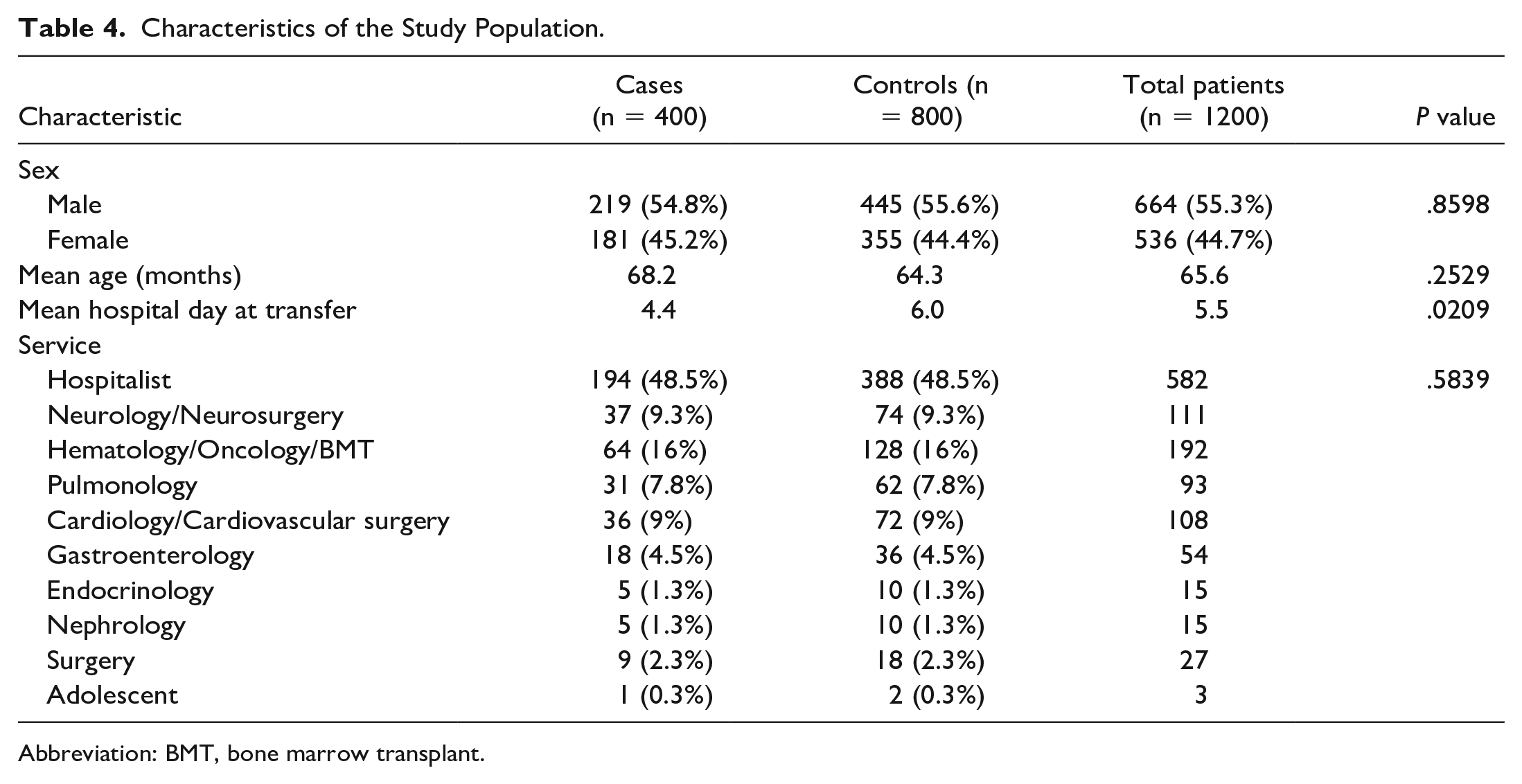
Abbreviation: BMT, bone marrow transplant.
Study Population Distribution by Age Category.
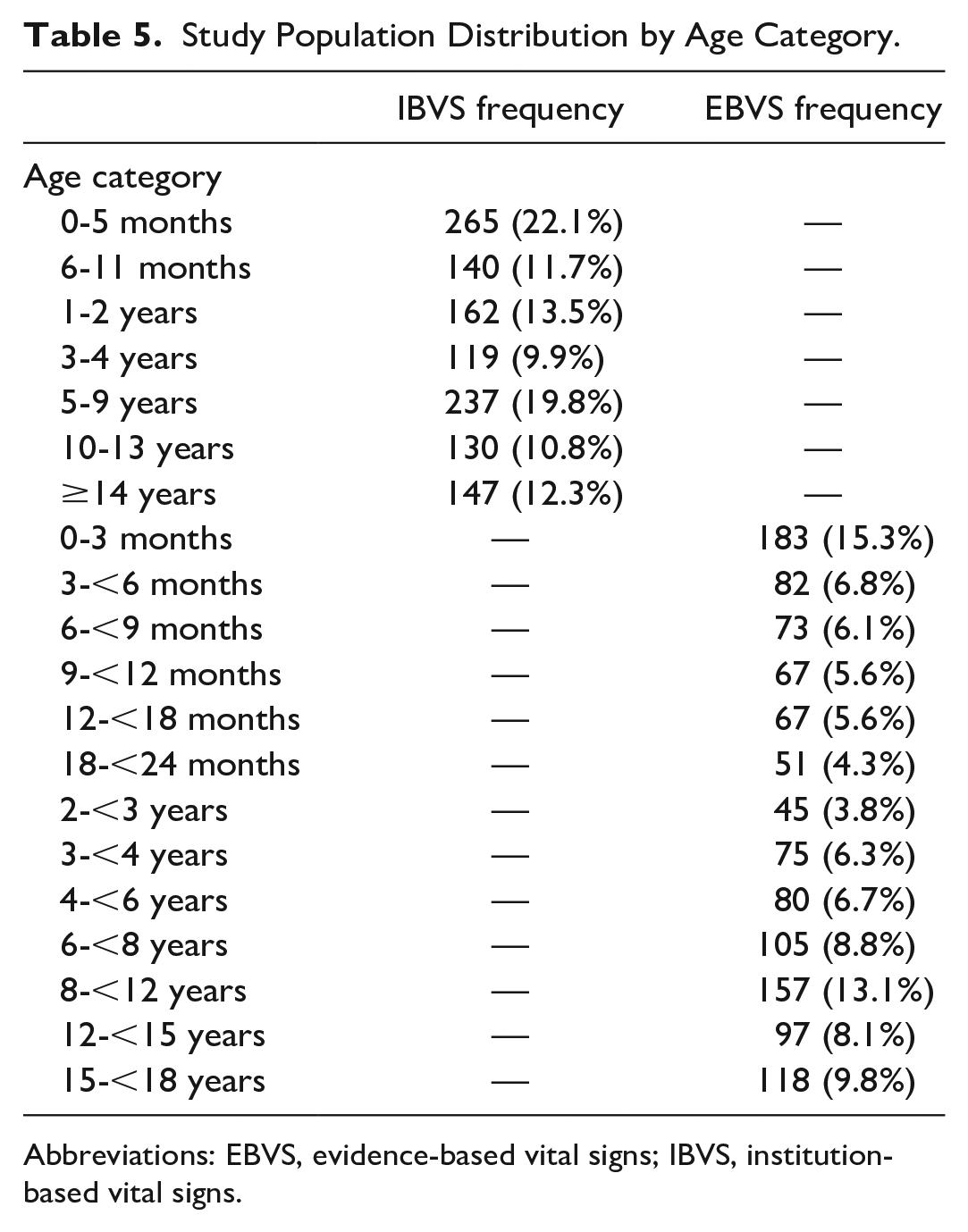
Abbreviations: EBVS, evidence-based vital signs; IBVS, institution-based vital signs.
Supplemental Table 2 demonstrates the non-normal distribution of PEWS by IBVS and EBVS among the total study population. In total, 42.8% of cases and 4.6% of controls had increased scores using EBVS (P ≤ .001). Among cases and controls with initial PEWS of 1 or higher, 4.6% of cases and 31.9% of controls had a reduced score (P ≤ .001).
Evidence-Based Vital Signs Improve the Sensitivity of Pediatric Early Warning Score to Predict Intensive Care Unit Transfer
Table 6 compares the IBVS and EBVS PEWS in case group and control groups. The median PEWS for EBVS was statistically significantly higher than IBVS among cases (4.0, IQR: 2.0, 5.0 vs 3.0, IQR: 2.0, 5.0, P < .0001) and similar in control groups (0.0, IQR: 0, 1.0 vs 0.0, IQR: 0, 1.0). The mean time from the highest recorded PEWS to ICU transfer is 4.67 ± 0.23 hours.
Differences in Pediatric Early Warning Score Among Cases and Controls After Evidence-Based Vital Signs Rescoring.
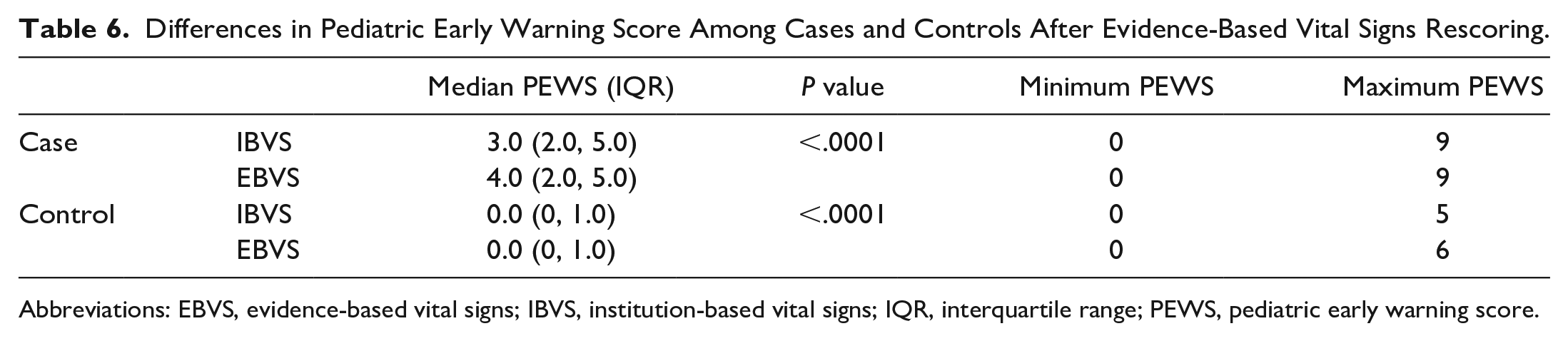
Abbreviations: EBVS, evidence-based vital signs; IBVS, institution-based vital signs; IQR, interquartile range; PEWS, pediatric early warning score.
Table 7 displays the sensitivities, specificities, and AUCs for IBVS and EBVS by the three clinically significant PEWS cutoffs. The ranges for sensitivity were improved across the three PEWS cutoff points for EBVS compared with IBVS (43%-71% vs 30%-63%) with only slightly decreased specificity (88%-98% vs 93%-99%). There was no statistically significant difference in the AUC for EBVS and IBVS across PEWS thresholds.
Sensitivities and Specificity of Pediatric Early Warning Score to Predict Unplanned Intensive Care Unit Transfer Using Evidence-Based Vital Signs versus Institution-Based Vital Signs.
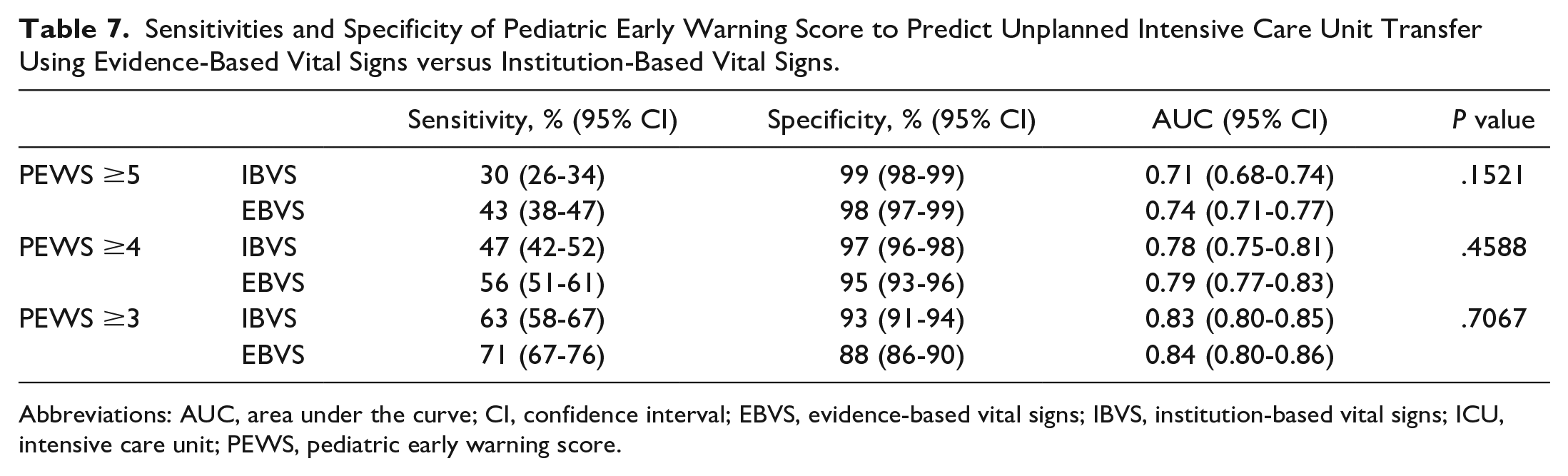
Abbreviations: AUC, area under the curve; CI, confidence interval; EBVS, evidence-based vital signs; IBVS, institution-based vital signs; ICU, intensive care unit; PEWS, pediatric early warning score.
We next evaluated the impact of EBVS PEWS on positive predictive value (PPV) and number of patients needed to evaluate (NNE) for ICU transfer (Table 8). We extrapolated the data to calculate PPV under the EBVS PEWS system by utilizing an unplanned transfer incidence of 0.67% in our institution. As anticipated, there was a slight decrease in the PPV and subsequent increase in the number of patients requiring evaluation for transfer at each PEWS threshold.
Impact of Evidence-Based Vital Signs on Positive Predictive Value and Number Needed to Evaluate.
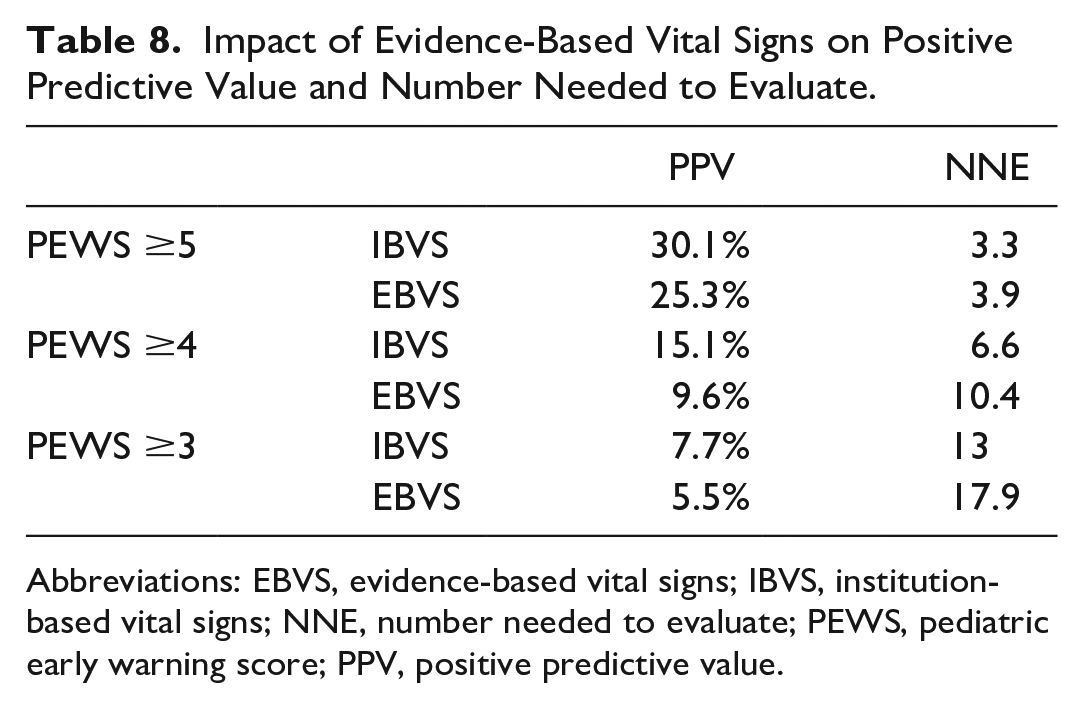
Abbreviations: EBVS, evidence-based vital signs; IBVS, institution-based vital signs; NNE, number needed to evaluate; PEWS, pediatric early warning score; PPV, positive predictive value.
More Refined Age Categories Do Not Improve Pediatric Early Warning Score Prediction of Deterioration
Table 9 presents the Wald tests for the interaction terms of PEWS with age categories for each scenario including IBVS PEWS versus IBVS age, IBVS PEWS versus EBVS age, EBVS PEWS versus IBVS age, and EBVS PEWS versus EBVS age. In each scenario, none of the p values for PEWS ≥5, PEWS ≥4, and PEWS ≥3 were statistically significant, indicating that more granular age categories do not influence the tool’s prediction of deterioration.
Association Between Vital Sign Criteria and Age Categories on Pediatric Early Warning Score Prediction of Deterioration.

Abbreviations: EBVS, evidence-based vital signs; IBVS, institution-based vital signs; PEWS, pediatric early warning score.
Late Rescue Intensive Care Unit Transfers and Patients Requiring Intensive Care Unit -Specific Therapies Have Higher Pediatric Early Warning Score Using Evidence-Based Vital Signs
Table 10 describes the ICU-specific therapies cases received following transfer and the influence of EBVS on the PEWS of these patients. The median PEWS was increased across all groups when comparing EBVS and IBVS PEWS, and all changes were statistically significant. There was a small proportion of patients whose PEWS was decreased with EBVS ranging from 1% to 11%. In contrast, a substantial proportion of each ICU-specific intervention group had increased PEWS with EBVS, ranging from 49% to 88%. “Other” therapies included continuous insulin infusions, continuous electrolyte infusions, exchange transfusion, and therapies requiring intensive monitoring such as anticoagulation infusions, antimalarial agents, or specialized respiratory supports (eg, Heliox). Patients who did not receive one of the listed ICU-specific therapies in the first 12 hours still received critical care support and monitoring following their transfer.
Change in Pediatric Early Warning Score by Intensive Care Unit -Specific Therapies.
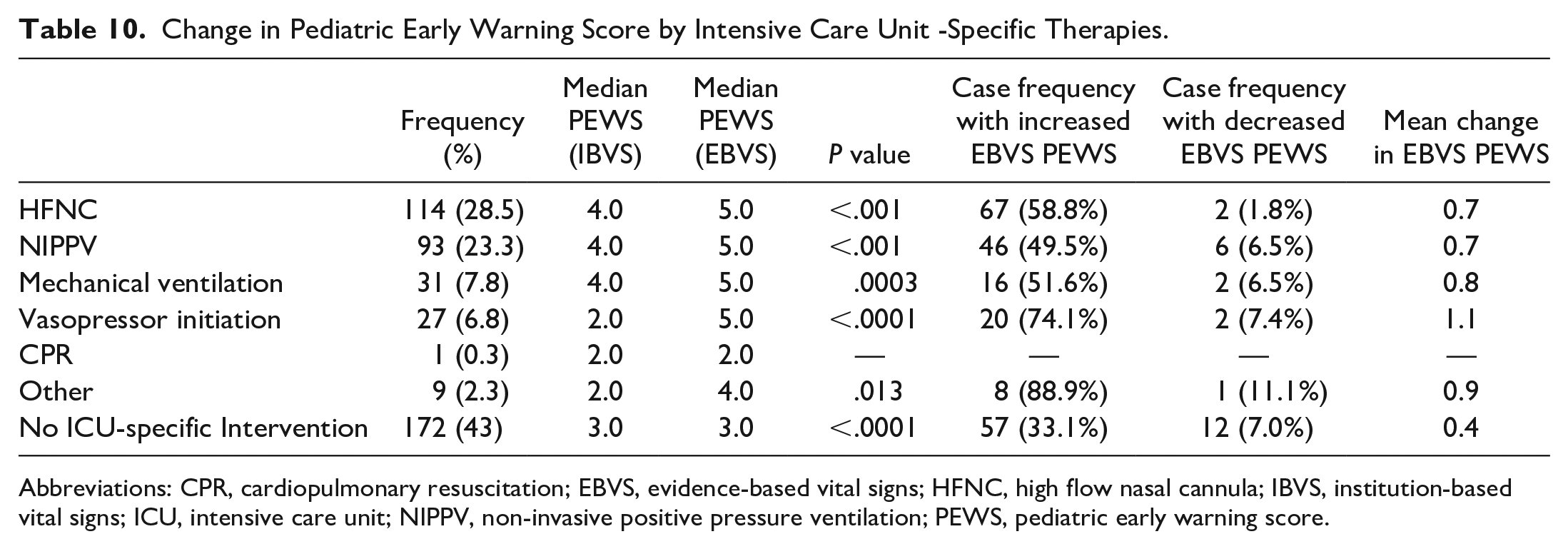
Abbreviations: CPR, cardiopulmonary resuscitation; EBVS, evidence-based vital signs; HFNC, high flow nasal cannula; IBVS, institution-based vital signs; ICU, intensive care unit; NIPPV, non-invasive positive pressure ventilation; PEWS, pediatric early warning score.
Among the 400 cases, 47 cases were identified as late rescues (Table 11). Mean PEWS for late rescues was 2.0 with SD of 3.0 for IBVS and 5.0 with SD of 3.0 for EBVS. Compared with non-late rescue cases, EBVS PEWS was statistically associated with the risk of late rescues with OR of 1.16 (95% CI = 1.0-1.34, P = .0498). There was no significant association between IBVS PEWS and the risk of late rescues with the OR of 1.05 (95% CI = 0.91-1.22, P = .502). There was no statistically significant difference when adjusting for age and gender covariates among late rescue and non-late rescue groups.
Association Between Pediatric Early Warning Score and Late Rescue Intensive Care Unit Transfers.
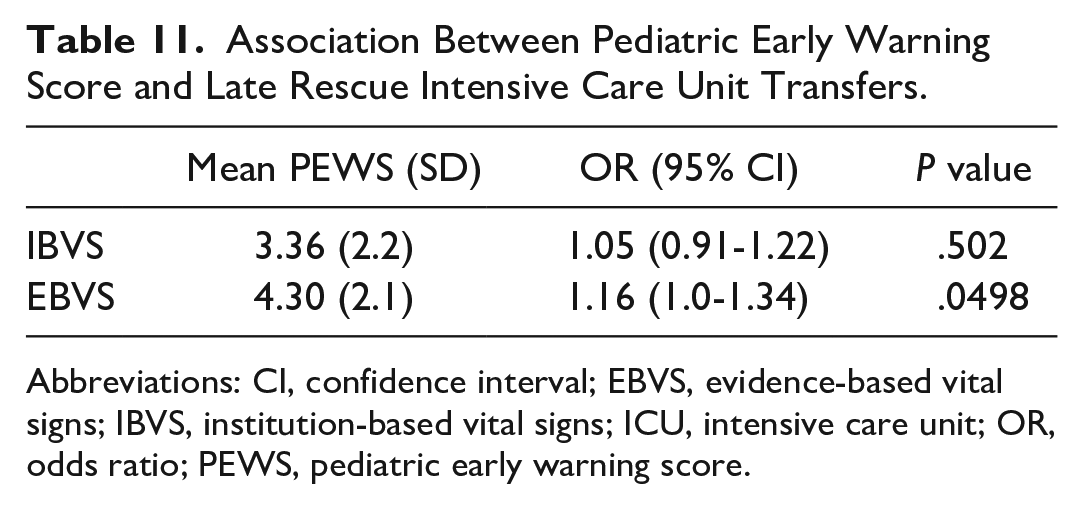
Abbreviations: CI, confidence interval; EBVS, evidence-based vital signs; IBVS, institution-based vital signs; ICU, intensive care unit; OR, odds ratio; PEWS, pediatric early warning score.
Discussion
To our knowledge, this is the first study to investigate the effect of EBVS on the Brighton PEWS model. Our retrospective analysis demonstrated that EBVS resulted in a small but non-statistically significant improvement in the sensitivity of PEWS to identify risk for clinical deterioration in patients admitted to acute care units who experienced unplanned ICU transfer. EBVS PEWS did not significantly improve the PPV of the tool. Patients meeting late rescue criteria and requiring ICU-specific therapies were more likely to have had elevated PEWS using EBVS. This suggests that the degree of vital sign abnormality may have been underestimated by current parameters in patients who received escalations of care and that EBVS may have a greater impact to detect more severe deterioration events. In addition, more granular age categories do not significantly improve the sensitivity of PEWS prediction of deterioration.
Previous research has similarly demonstrated no significant improvement of EBVS using the bedside PEWS model. 14 Both studies share similarities in study methodology, although Ross et al converted historical data for subjective scoring of the respiratory data set to generalize to their population. In contrast, we preserved the patient-level data collected for each member of the study population, which included an assessment of respiratory effort at the time of data collection. In both cases, application of EBVS did not significantly alter the overall predictive utility of either PEWS model. In addition, the case population studied by Ross et al excluded patients requiring ICU transfer exclusively for intensive nursing care. While this allows for analysis of a larger population requiring ICU-specific medical therapies, it is not entirely representative of the population that receives care in many critical care settings, particularly in those larger academic settings that provide care for a high proportion of children with medical complexity and intensive nursing needs. Together, these studies reinforce the limitations of existing EWS tools to reliably predict deterioration in the pediatric population.
There is significant variability among the vital sign parameters currently implemented in PEWS scoring, with previously little evidence to support the use of these criteria. Textbooks and reference range resources7-9 including PALS guidelines 10 that currently inform vital sign parameters for PEWS are largely based on consensus opinion. A meta-analysis 11 of vital signs from well children in outpatient settings highlights that these reference ranges correspond poorly to data-driven assessments of percentile curves in the well pediatric population. Bonafide et al have demonstrated even more substantial discrepancies among hospitalized non-ICU patients, citing that 38% of vital sign values measured in inpatient settings would be assigned points in PEWS using parameters developed by expert opinion. The original Brighton PEWS publication intentionally omitted specific vital sign parameters for ease of manual score calculations; however, the introduction of automated PEWS calculations in electronic medical records has removed this labor-intensive barrier. While there may be limited effectiveness of EBVS on PEWS systems, there is still value in utilizing evidence-based parameters for monitoring hospitalized children during routine care and in periods of clinical deterioration.
The physiologic variability of HR and RR by age in children adds additional layers of complexity to PEWS. While EBVS percentile curves demonstrate subtle variations with age on HR and RR, we have demonstrated that this variable is not independently associated with deterioration and does not significantly alter PEWS sensitivity. The reason for this lack of effect is not clearly understood, however may be related to broad definitions of “normal” vital signs for age group in previous PEWS models as a means to simplify calculations prior to automation. This data may have other applications outside of PEWS, however, including in assigning physiologic alarm parameters that may impact alarm fatigue. The cognitive burden on physicians and nurses to recognize abnormal vital signs should be minimized as safely as possible without sacrificing the sensitivity of the system to detect abnormalities.
Integration of vital sign criteria that are more specific to inpatient settings should lead to earlier recognition of decompensation and more timely initiation of goal-directed therapies. This may result in differences in clinical management that may mitigate or promote earlier ICU team involvement. In addition, a more sensitive EWS system may facilitate clinical interventions in patients at high risk of clinical decompensation and ultimately may decrease the rate of preventable ICU transfers or the intensity of therapies to rescue patients transferred to the ICU. Given the retrospective application of EBVS in this study, it is difficult to fully quantify implications of our results on clinical management in real time. Follow-up studies will be necessary to assess outcome measures related to rates of ICU transfers and patient morbidity.
This study has several limitations. First, while PEWS models across institutions share similarities, there are variations in system design and specific vital sign criteria that may impact the generalizability of this study. The magnitude of the impact of EBVS PEWS in a given institution may vary compared with our results. However even among the initial Brighton PEWS validation studies, there is variation among the vital sign criteria listed,2,5 if listed at all.1,13 The lack of validated and universally utilized vital signs is a major limitation to PEWS research on the whole and is one which this study seeks to address. Second, our model for PEWS only takes into account vital sign abnormalities ≥90th percentile when calculating total score and does not incorporate criteria for severe bradycardia or bradypnea, which may signal impending cardiac or respiratory arrest. Additional studies are needed to determine the effect that incorporating these parameters would have on PEWS sensitivity and specificity and clinician notification fatigue. Third, transfers meeting criteria for late rescues were rare, and therefore, we may lack statistical power to detect significant differences in the IVBS regression analysis for severe deterioration. Dedicated studies in this population will be beneficial moving forward and may include analysis of other metrics of deterioration.19,20 Finally, this retrospective study analyzes a single timepoint in a patient’s hospital course prior to ICU transfer. The highest IBVS PEWS does not necessarily correlate with the highest possible EBVS PEWS within the given time period, and these data do not inform time from a clinician’s first recognition of decompensation in patients transferred to the ICU. Moreover, a single PEWS value does not, in isolation, hold any specific predictive value for ICU transfer in the absence of clinical context. Additional research will provide valuable data to characterize the effect of RRT utilization and post-transfer morbidity following EBVS implementation.
Conclusion
In summary, we have demonstrated that EBVS resulted in small though non-statistically significant improvements in Brighton-based PEWS sensitivity to identify unplanned ICU transfers from acute care units. We demonstrated that EBVS were better able to identify patients requiring ICU-specific interventions. However, EBVS did not significantly impact PEWS sensitivity by age. EBVS PEWS may be beneficial to facilitate directed interventions in early clinical decompensation to reduce unplanned ICU transfers, and may be used in conjunction with other track and trigger tools to minimize preventable morbidity.
Author Contributions
Dr LR and Dr ND: Conceptualized and designed this study. Dr LR: Collected and scored the data. Dr AZ: Performed statistical analysis of the data. Dr LR: Drafted and edited the manuscript with assistance from Dr AZ. Dr ND: Provided mentorship, oversight, and critical review of the data and manuscript. All authors revised and reviewed the manuscript and approved the final work as submitted.
Supplemental Material
sj-pdf-1-cpj-10.1177_00099228231166264 – Supplemental material for Use of Evidence-Based Vital Signs in Pediatric Early Warning Score to Predict Clinical Deterioration on Acute Care Units
Supplemental material, sj-pdf-1-cpj-10.1177_00099228231166264 for Use of Evidence-Based Vital Signs in Pediatric Early Warning Score to Predict Clinical Deterioration on Acute Care Units by Lisa Rickey, Anqing Zhang and Nathan Dean in Clinical Pediatrics
Supplemental Material
sj-pdf-2-cpj-10.1177_00099228231166264 – Supplemental material for Use of Evidence-Based Vital Signs in Pediatric Early Warning Score to Predict Clinical Deterioration on Acute Care Units
Supplemental material, sj-pdf-2-cpj-10.1177_00099228231166264 for Use of Evidence-Based Vital Signs in Pediatric Early Warning Score to Predict Clinical Deterioration on Acute Care Units by Lisa Rickey, Anqing Zhang and Nathan Dean in Clinical Pediatrics
Footnotes
Acknowledgements
We acknowledge the assistance of Ms Susan Keller in a review of the literature for this study and thank Ms Parissa Safari who maintains the PERRC patient database.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
