Abstract
Objectives:
Effective treatment of depression is a key target for suicide prevention strategies. However, only around one-third of young people with suicide risk respond to evidence-based treatments. Understanding the trajectory of suicidal ideation, as a marker of suicide risk, over the course of evidence-based treatment for depression might provide insight into more targeted and effective treatments.
Methods:
This is a secondary analysis of data from the multicentre Youth Depression Alleviation–Combined Treatment trial. A total of 153 young people aged 15–25 years diagnosed with major depressive disorder were randomly assigned in this double-blind, placebo-controlled trial to either cognitive behavioural therapy plus fluoxetine or cognitive behavioural therapy plus placebo. Participants were assessed for depression and suicidal ideation at baseline and at weeks 4, 8 and 12.
Results:
Using group-based trajectory modelling, we identified two distinct depression trajectories. The first (Improving; 54.9%; n = 83) comprised those who experienced a consistent decline in depression symptoms. The second (Persisting; 45.1%; n = 70) comprised those who, despite treatment, still had clinically significant levels of depression by the end of treatment. For suicidal ideation, we identified four distinct trajectories: Non-clinical (15.5%; n = 20), Low Improving (47.1%; n = 75), High Improving (24.8%; n = 38) and High Persisting (12.7%; n = 20). Treatment allocation was not significantly associated with trajectory membership for either depression or suicidal ideation.
Conclusion:
Understanding the course of depression and suicidal ideation during treatment has important implications for managing suicide risk. The findings suggest that there is an identifiable group of young people for whom enhanced psychological and/or pharmacological intervention might be required to ensure a better treatment response. Specific interventions for those with suicidal ideation may also be prudent from the outset.
Clinical trial registration:
The Youth Depression Alleviation–Combined Treatment trial was prospectively registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12612001281886).
Introduction
Suicide is a leading cause of death in young people internationally (World Health Organization, 2014). Given that major depression represents a significant population attributable risk for self-harm and suicide in young people (Witt et al., 2018), effective treatment of depression is therefore an important aspect of suicide prevention. Current evidence-based clinical practice guidelines recommend psychological therapies as first-line treatment for depression in young people (National Institute for Health and Clinical Excellence [NICE], 2019), with depression-focused cognitive behavioural therapy (CBT) the most commonly used therapy in high-income countries (Callahan et al., 2012).
Suicidal ideation is often a feature of depression, with suicidal ideation a risk factor for self-harm (Ribeiro et al., 2016; Witt et al., 2018) and suicide (Ribeiro et al., 2016). Suicidal ideation severity is known to fluctuate (Selby and Yen, 2014), including over the course of evidence-based treatment for depression (Vitiello et al., 2009), and treatment-mediated improvements in depression symptomatology do not necessarily equate to improvements in suicidal ideation severity (Batterham et al., 2017, 2019; Madsen et al., 2016a, 2016b; van Spijker et al., 2014). Given most effects of current evidence-based psychological and medication treatments for depression are modest (Locher et al., 2017; Weisz et al., 2017), earlier identification of clinically relevant subgroups of patients who are likely to benefit from alternative or additional treatment might facilitate tailored and more effective intervention to improve outcomes. This is particularly important for those who are experiencing suicidal ideation, and therefore are potentially at greater risk of self-harm and suicide, as interventions directly targeting suicidal ideation and self-harm might be more effective than treatments focused more broadly on depressive symptomatology (Meerwijk et al., 2016).
A key challenge for clinicians working with these young people is that the nature, frequency and level of intent of suicidal ideation and the urge to self-harm fluctuate are situation dependent and can be difficult for young people to disclose. Few clinical trials of depression-focused treatments have assessed suicidal ideation and self-harm as outcomes in young people (Hetrick et al., 2012) or how treatment for depression might affect suicidal ideation. Depression-focused psychological treatment has been found to have inconsistent effects on suicidal ideation (Cuijpers et al., 2013). Understanding the trajectory of suicidal ideation, as a marker of potential risk for self-harm and suicide, over the course of evidence-based treatment for depression might provide insight into more effective treatment.
We have therefore used a latent variable classification method to define trajectories of change in depression severity and suicidal ideation over the course of 12 weeks of gold standard treatment for young people diagnosed with major depression and to investigate the factors associated with membership to these trajectories.
Methods
This study involves secondary analysis, designed post-data collection, of data from the Youth Depression Alleviation–Combined Treatment (YoDA-C) trial. This was a 12-week randomised, double-blind, placebo-controlled, multicentre clinical trial prospectively registered with the Australian and New Zealand Clinical Trials Register (ACTRN12612001281886). The study was approved by the Melbourne Health Human Research Ethics Committee (HREC/12/MH/151). Detailed methodological information on this trial is reported elsewhere (Davey et al., 2014).
Setting and participants
Young people seeking treatment from one of four primary care community-based youth mental health treatment centres or one specialist youth mental health service located in the north-western region of Melbourne, Victoria, were consecutively enrolled into the trial between February 2013 and December 2016 and were sequentially randomised into either cognitive behavioural therapy (CBT) plus fluoxetine (CBT + FLX) or CBT plus placebo (CBT + PLO) on a 1:1 basis.
Eligible participants were (1) aged between 15 and 25 years (inclusive) at trial commencement; (2) diagnosed with major depressive disorder (MDD) according to the Structured Clinical Interview for the Diagnostic and Statistical Manual for Mental Disorders (DSM), Axis I Disorders (SCID-IV) (First et al., 1997); (3) scored 20 or greater on the Montgomery–Åsberg Depression Rating Scale (MADRS) (Montgomery and Asberg, 1979) at baseline, indicating at least moderate levels of depression; and (4) able to provide written informed consent, including both an adequate intellectual capacity and English language fluency.
Young people were excluded from the trial if they: (1) had a lifetime or current diagnosis of psychosis according to the SCID-I/P; (2) had a lifetime or current diagnosis of bipolar I or II disorder; (3) had a diagnosis of an acute or unstable medical disorder that would interfere with treatment; (4) were currently pregnant; (5) had a severe disturbance such that the young person would be unable to comply with the requirements of informed consent or with the study protocol; (6) received at least 2 weeks of treatment with an antidepressant medication immediately prior to baseline; or (7) had any previous treatment with fluoxetine that had been either ineffective or poorly tolerated. Participants were also withdrawn from the trial if treatment with antipsychotic medications was required, although treatment with benzodiazepines or benzodiazepine-like medications was permitted if clinically indicated.
Intervention and control conditions
Young people allocated to the CBT + FLX condition received a daily 20 mg tablet of fluoxetine. Dosage could be increased up to 40 mg if clinically indicated. Those allocated to the CBT-PLO group received one pill identical in packaging, appearance, colour and taste to the trial medication.
All participants were concurrently offered weekly sessions of CBT for 12 weeks. These sessions were delivered by Masters- or higher-level clinical psychologists, occupational therapists or social workers with additional training, all of whom had experience treating young people with depression. Sessions covered seven core components of manualised CBT: (1) psychoeducation, (2) understanding and monitoring emotions, (3) behavioural activation, (4) chain analysis, (5) identifying automatic thoughts, (6) working with unhelpful thoughts and (7) relapse prevention.
Measures
Past week depression symptom severity was measured using the interview-rated MADRS (Montgomery and Asberg, 1979), a 10-item clinician-rated scale in which total scores can range from 0 to 60. Higher total scores indicate greater depression severity.
Past month suicidal ideation was measured using the self-reported 30-item Suicidal Ideation Questionnaire (SIQ) (Reynolds, 1987). Items are scored on a 7-point Likert-type scale ranging from 0 (never had the thought) to 6 (had the thought almost every day) (Reynolds, 1987). Total scores can therefore range from 0 to 180. Higher total scores on the SIQ are therefore indicative of greater suicidal ideation. A cut-point of 41 is typically used to indicate clinically significant suicidal ideation (Reynolds, 1987).
Outcomes were assessed at baseline and again at weeks 4, 8 and 12.
Covariates
Demographic covariates were modelled as time invariant factors and included (1) male sex and (2) older age (i.e. 18 years and older at baseline). This latter covariate was included as results from the main study indicated there were significant differences in treatment outcomes for adults as compared to younger adolescents (Davey et al., 2019).
A number of clinical covariates measured at baseline were also included: (1) number of previous episodes of depression; (2) duration of the current episode of depression (in weeks); (3) scores on the Alcohol Use Disorders Identification Test (AUDIT) (Saunders et al., 1993); (4) scores on the Generalized Anxiety Disorders – 7-item scale (GAD-7) (Spitzer et al., 2006); and (5) and past month self-harm, which included all intentional acts of self-poisoning, intentional drug overdose and/or self-injury irrespective of suicidal intent or other types of motivation in line with international definitions (Hawton et al., 2003). Self-harm was measured using the Columbia Suicide Severity Rating Scale by combining ratings from the ‘suicidal behaviour-actual attempts’ and ‘non-suicidal self-injury’ subscales (Posner et al., 2007).
We also investigated the extent to which treatment allocation to the CBT + FLX arm influenced trajectory membership and the extent to which depression-trajectory membership influenced suicidal ideation membership.
Analyses
Latent group-based trajectory modelling was used to identify subgroups that differ in trajectories of depression and suicidal ideation severity over the course of the 12-week treatment period and to characterise the factors associated with membership to these trajectories. Group-based trajectory modelling is a flexible, semi-parametric approach that allows for the identification of distinct subgroups of patients following distinct longitudinal trajectories in either depression or suicidal ideation symptom evolution over time (Nagin and Odgers, 2010).
Trajectory enumeration was assessed using the recommended two-stage approach (Nagin, 2005). First, the optimal number of trajectories was determined by fitting a null model and progressively adding additional trajectories until no further improvement in model fit was observed (Twisk and Hoekstra, 2012). Second, the shape of the individual trajectories retained was determined by fitting cubic, quadratic and linear terms and progressively dropping non-significant higher order polynomials until no further improvement in model fit was observed (Nagin and Odgers, 2010).
Model fit was guided by the sample size-adjusted Bayesian information criterion (BIC) and Bayes factor. Given that the BIC can, in some instances, continue to demonstrate model improvement with the addition of trajectories (Nagin, 2005), we also used a number of other fit statistics as recommended (Hensen et al., 2007; Nagin and Odgers, 2010; Nyland et al., 2007). Specifically, for each identified trajectory, we ensured that the Odds of Correct Classification exceeded five (Nagin, 2005), entropy was greater than 0.70 (Nagin, 2005), the average posterior probability for each group exceeded 0.80 and each trajectory group contained no fewer than 10 participants to guard against model over-fit. Trajectory selection was also guided by clinical acumen to ensure identified trajectories meaningfully described patterns of change in depression symptomatology and suicidal ideation severity during CBT treatment.
Multinomial logistic regression modelling was next used to investigate the extent to which a number of demographic and clinical covariates (as described above) were associated with membership to each identified trajectory. These models were weighted by the inverse of the probability of trajectory membership derived from the posterior probability estimates.
Data from all participants were incorporated into these models. Missing data were handled by applying the full information maximum-likelihood approach, as recommended (Muthén and Muthén, 2012). All analyses were conducted in Stata for Windows, version 14.2, using the traj plug-in (Jones and Nagin, 2013).
Results
A total of 153 young people aged 15–25 years (M = 19.6, SD = 2.7 years) diagnosed with MDD were randomly assigned to receive either CBT + FLX (n = 76) or CBT + PLO (n = 77) (see CONSORT flow diagram in the original trial report) (Davey et al., 2014). Scores at baseline indicated the sample had moderate to severe levels of depression (M = 32.9, SD = 5.4). The median number of depressive episodes was 2.0 (interquartile range: 1.0–4.0), and over one-third (n = 61; 39.9%) were experiencing their first episode of depression. Most participants reported suicidal ideation in the month preceding recruitment (75.0%; n = 114), and the mean score on the SIQ at baseline was 72.1 (SD = 42.4), indicating clinically significant levels of suicidal ideation. Just over one-quarter (26.8%; n = 41) had engaged in an episode of Non suicidal self injury (NSSI) and/or suicide attempt within the month preceding trial entry, according to scores on the Columbia-Suicide Severity Rating Scale (C-SSRS). The trial arms were well balanced with respect to a number of key demographic and clinical characteristics (Table 1). Further information on the characteristics of the sample is reported in a related publication (Davey et al., 2019).
Key demographic and clinical characteristics of the sample at baseline.
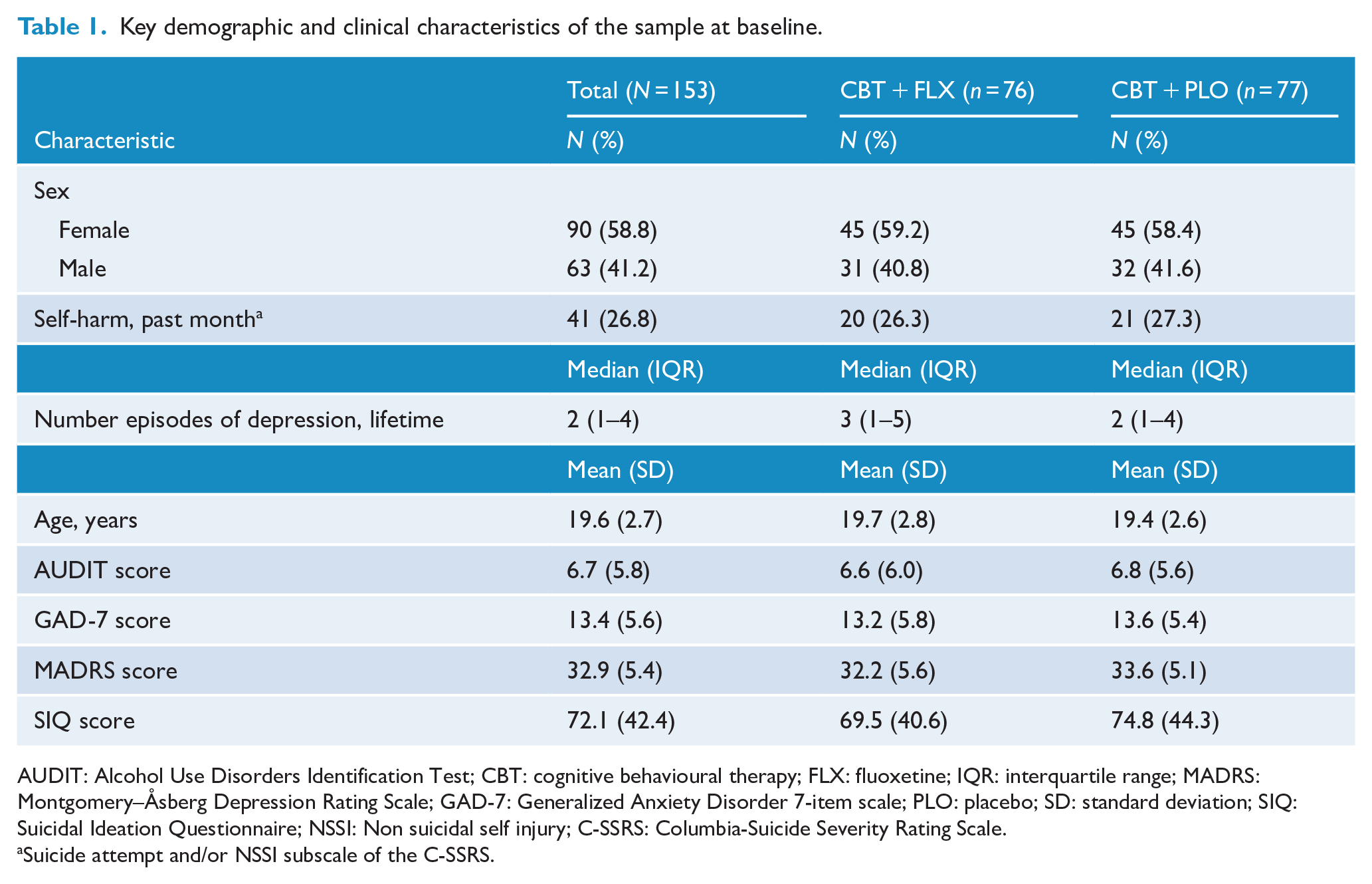
AUDIT: Alcohol Use Disorders Identification Test; CBT: cognitive behavioural therapy; FLX: fluoxetine; IQR: interquartile range; MADRS: Montgomery–Åsberg Depression Rating Scale; GAD-7: Generalized Anxiety Disorder 7-item scale; PLO: placebo; SD: standard deviation; SIQ: Suicidal Ideation Questionnaire; NSSI: Non suicidal self injury; C-SSRS: Columbia-Suicide Severity Rating Scale.
Suicide attempt and/or NSSI subscale of the C-SSRS.
We have previously reported that adding fluoxetine to CBT did not appear to be beneficial in reducing depressive symptoms. Similarly, while there were reductions in suicidal ideation scores across treatment, no significant between-group differences were observed (Davey et al., 2019). In this secondary analysis, we use a latent variable classification method to define trajectories of change in depression severity and suicidal ideation over the course of treatment and to investigate the factors associated with membership to these trajectories.
Depression trajectories
In this population, while the addition of a third MADRS trajectory appeared to improve statistical fit, there were fewer than 10 participants in this third trajectory group suggesting model over-fit. We therefore chose to retain the more parsimonious two-trajectory model for MADRS depression scores (sample size-adjusted BIC: −1904.68; Table 2).
Fit statistics and class membership for the trajectory models.
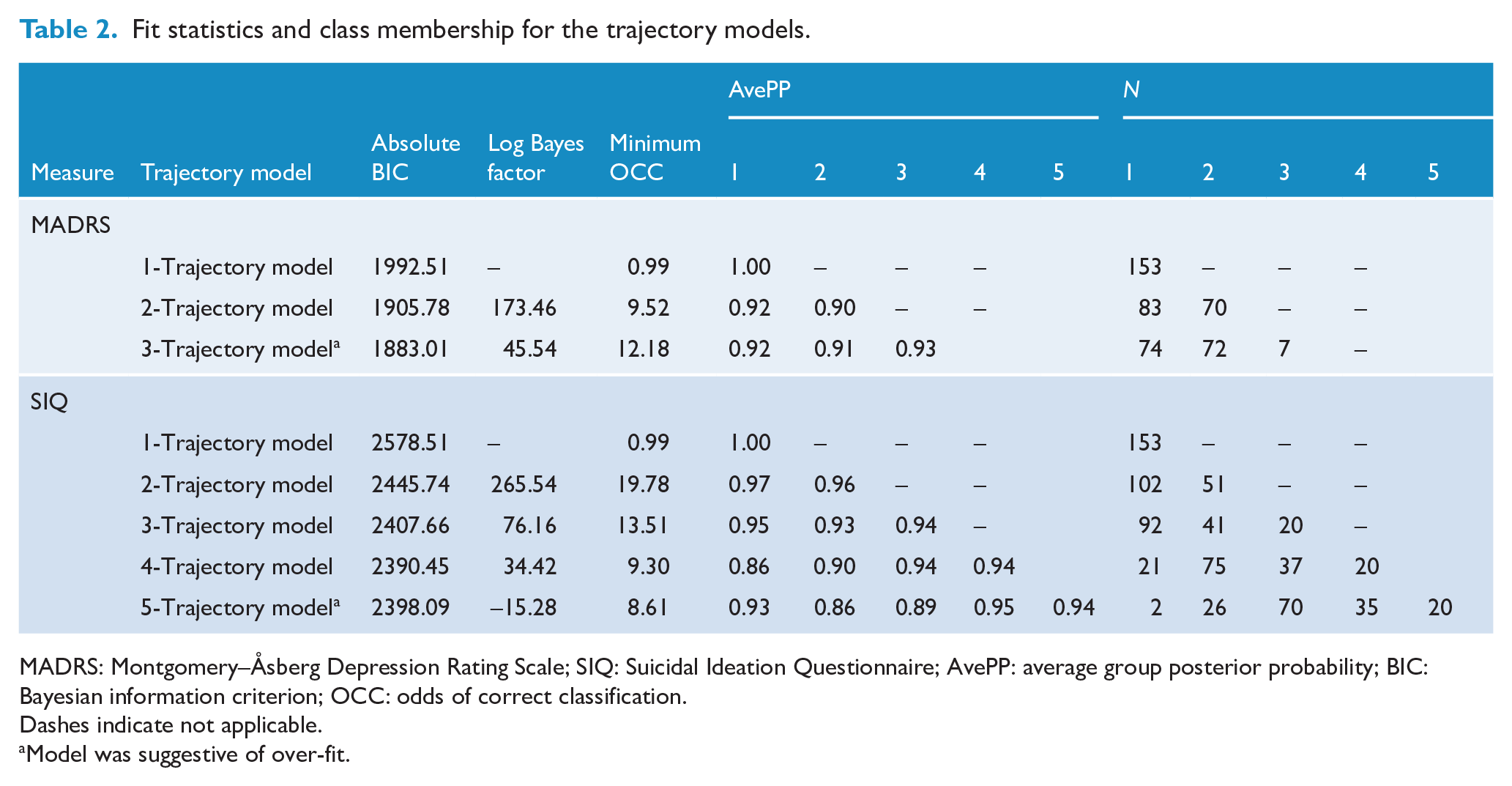
MADRS: Montgomery–Åsberg Depression Rating Scale; SIQ: Suicidal Ideation Questionnaire; AvePP: average group posterior probability; BIC: Bayesian information criterion; OCC: odds of correct classification.
Dashes indicate not applicable.
Model was suggestive of over-fit.
The first trajectory group (Improving; 54.9%; n = 83), described by a quadratic polynomial, comprised those who experienced a significant decline in depression symptoms across the 12-week trial (Mean difference [MΔ] = −21.9, 95% confidence interval [CI] = [−23.7, −20.2]; Table 3; Figure 1). The second trajectory group (Persisting; 45.1%; n = 70), described by a linear polynomial, comprised those who, despite treatment, still had clinically significant levels of depression by the end of treatment (MΔ = −6.5, 95% CI = [−8.7, −4.4]; Table 3; Figure 1).
Estimated means and change scores on the MADRS and SIQ at baseline, week 4 (mid-treatment), week 8 (mid-treatment) and week 12 (end of treatment) for the MADRS and SIQ trajectory groups.
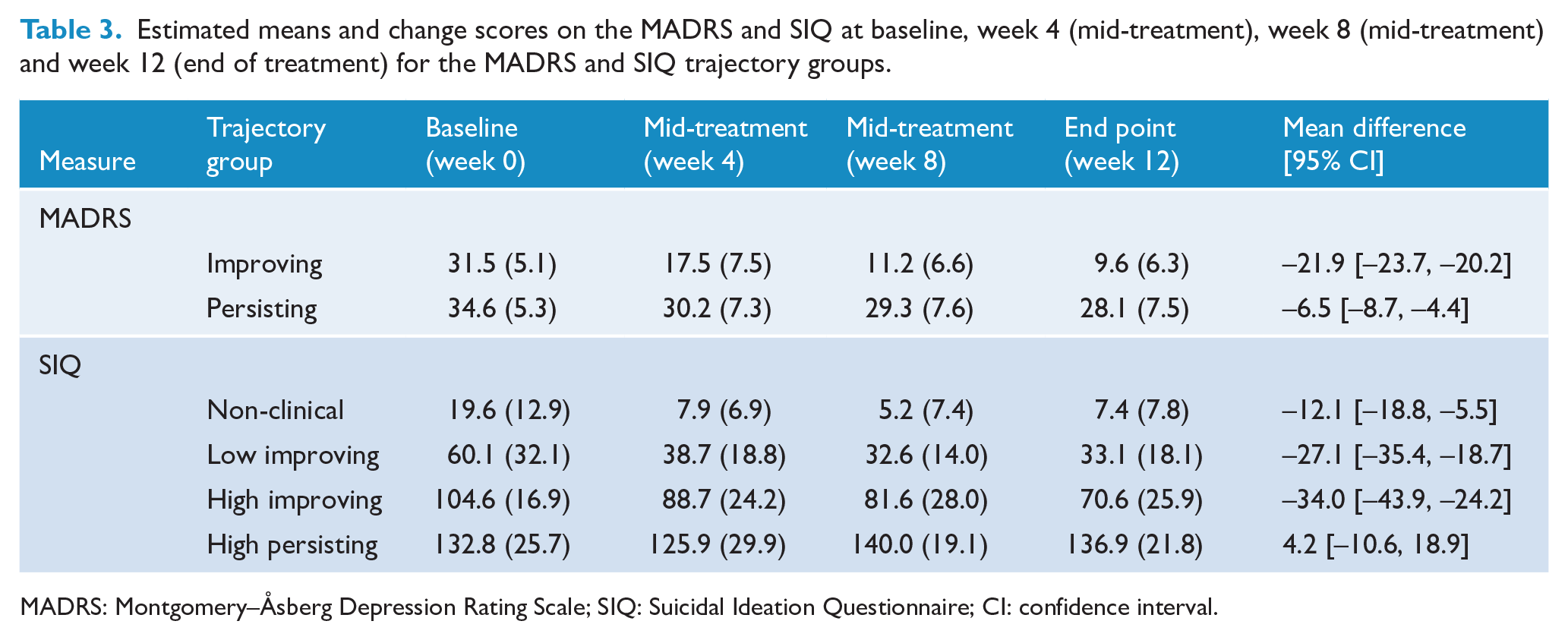
MADRS: Montgomery–Åsberg Depression Rating Scale; SIQ: Suicidal Ideation Questionnaire; CI: confidence interval.
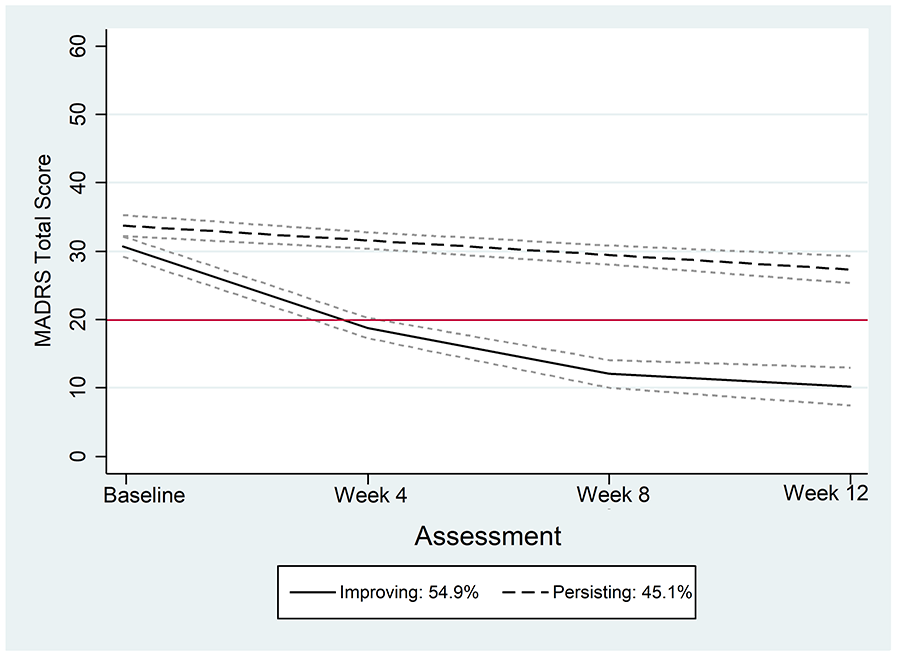
Two-group trajectory model of depression symptoms, as measured by the MADRS, across assessment time points.
There were no significant demographic or clinical predictors, including treatment allocation to fluoxetine, associated with MADRS trajectory group membership (Table 4).
Multinomial logistic regression model predicting membership to MADRS and SIQ trajectory groups.
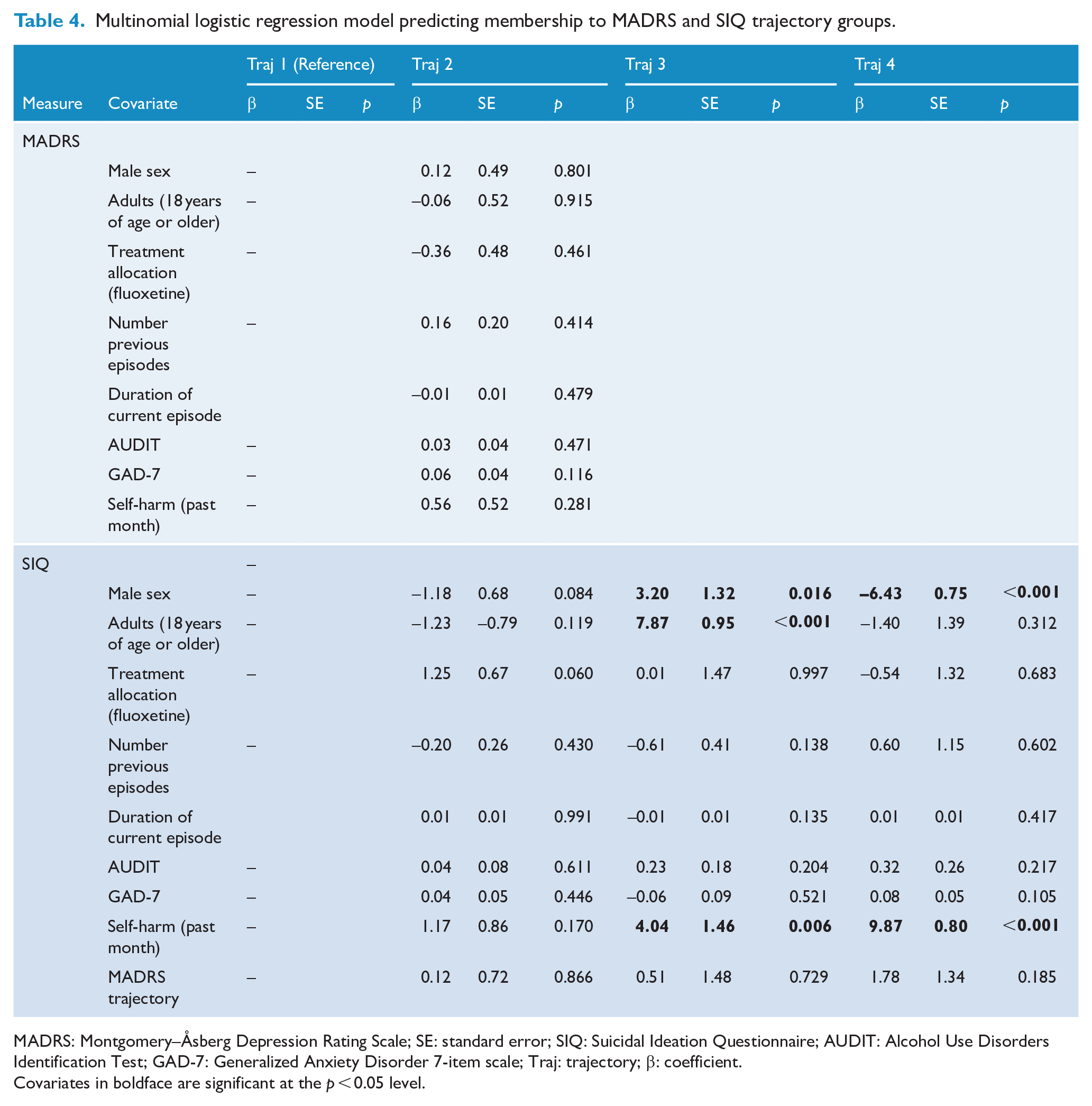
MADRS: Montgomery–Åsberg Depression Rating Scale; SE: standard error; SIQ: Suicidal Ideation Questionnaire; AUDIT: Alcohol Use Disorders Identification Test; GAD-7: Generalized Anxiety Disorder 7-item scale; Traj: trajectory; β: coefficient.
Covariates in boldface are significant at the p < 0.05 level.
Suicidal ideation trajectories
For suicidal ideation, we chose a four-group trajectory model (sample size-adjusted BIC: −2383.85; Table 2). The first trajectory group (Non-Clinical; 15.5%; n = 20), described by a linear polynomial, comprised those who scored below the clinical cut-point on the SIQ at baseline and end of treatment (MΔ = −12.1, 95% CI = [−18.8, −5.5]; Table 3, Figure 1). The second trajectory group (Low Improving; 47.1%; n = 75), described by a quadratic polynomial, comprised those who scored above the clinical cut-point on the SIQ at baseline, but experienced meaningful decline such that scores were no longer in the clinically concerning range at end of treatment (MΔ = −27.1, 95% CI = [−35.4, −18.7]; Table 3). The third group (High Improving; 24.8%; n = 38), described by a linear polynomial, comprised those who scored very high on the SIQ at baseline and experienced the largest decline in suicidal ideation by end of treatment. Despite this, their scores remained above the clinical cut-off (MΔ = −34.0, 95% CI = [−43.9, −24.2]; Table 3). The final and smallest class (High Persisting; 12.7%; n = 20), described by a linear polynomial, had very high suicidal ideation scores that increased, albeit non-significantly, over the course of treatment (MΔ = 4.2, 95% CI = [−10.6, 18.9]; Table 3; Figure 2).
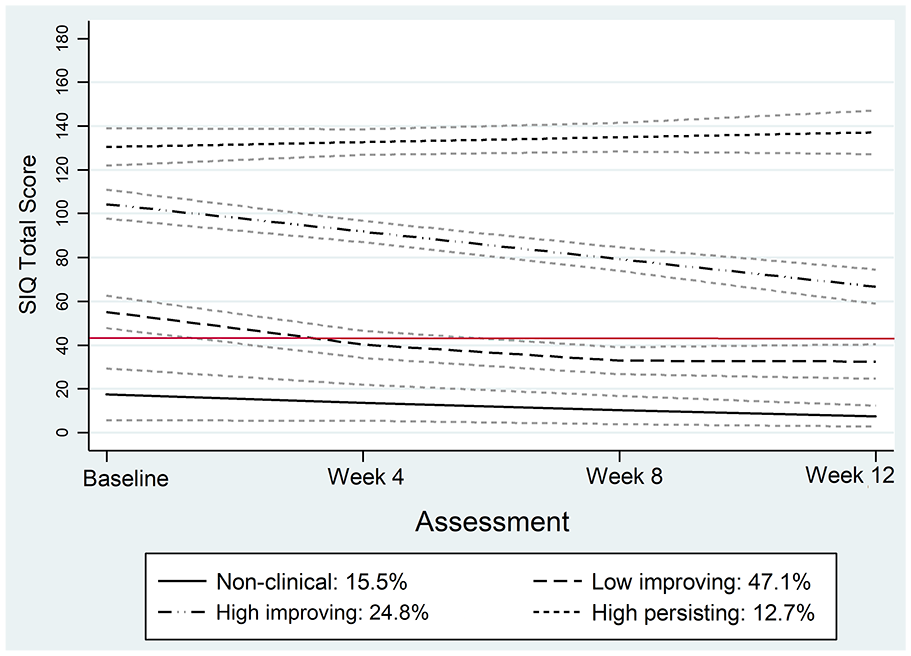
Four-group trajectory model of suicidal ideation severity, as measured by scores on the SIQ, across the assessment time points.
Compared with the Non-Clinical trajectory group (8.9% female, 19.1% male), males were more likely to be assigned to the High Improving Suicidal Ideation trajectory group (27.0%), while females were more likely to be assigned to the High Persisting Suicidal Ideation trajectory group (15.6%). Those over 18 years of age at baseline were more likely to be assigned to the High Improving Suicidal Ideation trajectory group (24.0%) as compared with the Non-Clinical trajectory group (16.4%). A recent (1 month) self-reported history of self-harm was also significantly associated with an increased likelihood of being assigned to the High Improving (41.5%) and the High Persisting (19.5%) Suicidal Ideation trajectory groups, as compared with the Non-Clinical trajectory group (2.4%). However, no other clinical factors were significantly associated with suicidal ideation trajectory group membership. Importantly, this includes treatment allocation (i.e. fluoxetine vs placebo) and MADRS trajectory group (Table 4).
Discussion
This study was a secondary analysis of data from a randomised controlled trial of CBT plus fluoxetine (CBT + FLX) versus CBT plus placebo (CBT + PLO) for the treatment of 15- to 25-year-old young people diagnosed with major depression (Davey et al., 2019). Two distinct trajectories for clinician-rated depression symptoms were identified over the course of the 12-week trial, in line with trajectory analysis of data from clinical trials undertaken with community-based adults (Batterham et al., 2019) and adolescents treated in a tertiary clinic setting (Davies et al., 2019). Half were assigned to the Improving group. These young people had severe depression at baseline and experienced early significant improvement in their symptoms such that, by week 4 of the trial, their scores on the MADRS were below the cut point used for entry into the trial, indicative of treatment response (Hawley et al., 2002). This early improvement is consistent with what has been observed in previous similar studies in adults (Batterham et al., 2017) and adolescents (Davies et al., 2019; Scott et al., 2019). However, in this study, almost one-half of those who began the trial with severe depression experienced only slight improvement in depression symptomatology over the course of the trial, continuing to experience clinically significant levels of depression, despite treatment. In contrast, in a recent similar analysis, albeit from a trial of psychotherapeutic interventions, data from 465 adolescents showed significant reductions in depression over 18 weeks in both of the two trajectories identified in that study, but with one group showing no further improvement after 18 weeks. In a second recent secondary analysis of 12-week data from the Treatment for Adolescent Depression trial that randomised to CBT, CBT + FLX, FLX or PLO (Treatment for Adolescents with Depression Study [TADS], n = 439) (Scott et al., 2019), three trajectories were identified. Two were similar to those shown in this study: a group with high-severity depression and early significant improvement; a group with high-severity depression and limited symptom change. A third trajectory group was identified who had moderate-severity depression and late significant improvement in depression. The findings across these studies are in contrast to studies in adults in which those with higher baseline severity of depression experienced greater improvements in symptoms following CBT treatment (Fournier et al., 2010; Kirsch et al., 2008).
The findings across these studies highlight the need for effective early intervention for this group, with vigilant and ongoing monitoring of symptoms to ensure those for whom a poor outcome is indicated are identified and a responsive treatment plan is in place.
Four distinct trajectories of suicidal ideation were identified, consistent with previous trajectory analysis in clinical trials in community-based adult populations (Allan et al., 2018; Kasckow et al., 2016; Madsen et al., 2016b). Almost two-thirds of participants were assigned to trajectories (i.e. Non-Clinical and Low Improving) categorised by no to very low suicidal ideation. A third trajectory (High Improving) comprised almost one-quarter and was characterised by young people with severe suicidal ideation that improved throughout the course of treatment, although levels remained within the clinically concerning range. A fourth trajectory (High Persisting) was also identified, comprising 12.7% of the sample, characterised by those experiencing severe suicidal ideation that did not improve.
As in similar analyses in samples of adults (Batterham et al., 2019), we found no relationship between trajectories of depression and suicidal ideation, suggesting that remission in depressive symptomatology might not be related to changes in suicidal ideation, particularly for those experiencing very high levels of suicidal ideation at baseline. For this group in particular, achieving meaningful reductions in suicidal ideation appears to be challenging (Batterham et al., 2019). It might be that specific interventions aimed at reducing suicidal ideation and self-harm are required in order to obtain greater changes than those achieved with treatments that focus on depressive symptomatology alone (Meerwijk et al., 2016). The current findings highlight that there might be factors other than depression symptoms alone that are important to address in order to reduce suicidal ideation. This raises the possibility that separate biopsychosocial processes might underlie the manifestation of depressive symptoms compared with suicidal ideation. Further development and evaluation of interventions specifically focused on reducing suicidal ideation, such as Living with Deadly Thoughts (van Spijker et al., 2014) and Reframe-IT (Hetrick et al., 2017), should therefore be prioritised, alongside further investigation into the mechanisms underlying the effectiveness of these interventions. This will help to clarify the relationship between remission in depressive symptomatology and suicidal ideation (Witt et al., 2019).
Clinical practice guidelines suggest the addition of a serotonin-specific reuptake inhibitor (SSRI), such as fluoxetine, when there is poor response to psychotherapy (CBT is typically recommended) (NICE, 2005; Vitiello, 2019) The YoDA-C trial aimed to understand whether, in young people with moderate to severe depression, immediate combination treatment might improve outcomes. Allocation to either CBT + FLX or CBT + PLO was not associated with trajectory group membership for either the depression or suicidal ideation trajectories identified in this study, highlighting the need for continuing efforts to develop and to evaluate adjunctive interventions (Quinn et al., 2018; Rice et al., 2016). The evidence base for combination treatments therefore remains limited (Cox et al., 2014).
Consistent with previous research (Allan et al., 2018), multinomial regression modelling suggested that young people with a recent (past month) history of self-harm were more likely to be assigned to either the High Improving or High Persisting Suicidal Ideation trajectories. There have been some attempts to elucidate the factors that might be involved with the progression to self-harm in those with suicidal ideation (Branley-Bell et al., 2019). However, this work is preliminary, and current findings are a reminder of how commonly they co-occur, particularly for those with very severe suicidal ideation (Witt et al., 2018). Understanding the nature of, and fluctuation in, the underlying distress and suicidal intent, and the function that self-harm might have in this clinical picture, is critical to informing ongoing efforts to develop and test interventions for this group. This is particularly the case for those whose suicidal ideation does not improve and who might require enhanced treatment. In the meantime, ensuring a good assessment of self-harm and targeting adapted CBT or Dialectical Behaviour Therapy (DBT) may be a better treatment approach for these young people (Hawton et al., 2015).
Limitations
Findings should be considered in light of the following limitations. First, latent class trajectory analysis constitutes a data-driven technique. Despite this, it is notable that both the trajectories of depression symptom severity and suicidal ideation identified in this study were similar to those previously found in clinical trials of community-based adults with a range of conditions (Batterham et al., 2017, 2019; Madsen et al., 2016b).
Second, the overall YoDA-C trial was underpowered to reliably detect small to medium effect sizes. One-fifth of included participants were either lost to follow-up or discontinued treatment prematurely. While there is no clear guidance on how to achieve adequate statistical power for latent trajectory analysis, we have used a number of fit statistics as recommended, to determine model fit in this study. However, it should be noted that by week 12, there was a higher rate of drop-out in the group who received placebo (n = 20), compared with those who received fluoxetine (n = 15), indicating that data may not be missing at random but is influenced by treatment allocation. However, as we implemented the full information maximum-likelihood approach to account for missing data, we nevertheless believe our models are robust. It is the case that the analysis is limited to data over relatively brief window of observation and possible that the addition of data from long-term follow-up could alter the results. Finally, the results are not generalisable to young people treated in other settings.
Implications
Understanding the course of suicidal ideation during evidence-based treatment for depression in young people has important implications for improving interventions to address suicidal ideation, with potential downstream implications for preventing self-harm and suicide (Meerwijk et al., 2016) in this population. These findings generate important hypothesis for further examination.
Previous work has suggested that end-of-treatment response might predict long-term outcomes (Emslie et al., 2010; Kennard et al., 2009). Consequently, ‘treatment-resistant’ depression is defined as failure of a full course of treatment (Fava, 2003; Hetrick et al., 2011). However, we found that there was an identifiable group of young people for whom enhanced intervention might be required to ensure a better treatment response. This may be particularly so for those following the Persisting Depression trajectory. For these young people, waiting until the full course of treatment has been completed before implementing enhanced psychological and/or pharmacological intervention only risks further symptomatic and functional deterioration, including self-harm and suicide. Similarly, for those with very high suicidal ideation scores at baseline and little initial improvement, adjunctive or alternative interventions might be offered earlier to facilitate a more rapid resolution of symptoms and improved rates of recovery.
Three-quarters had suicidal ideation at baseline, making delivery of an intervention that targets this symptom from the outset important, particularly given evidence that interventions that focus on self-harm and suicidal ideation may be more effective in reducing suicide risk than those that focus on depression alone (Comtois and Linehan, 2006; Tarrier et al., 2008) and may also affect depression severity (Hawton et al., 2015). Increasing rates of recovery in this group might avert the potentially negative effects of depression and suicidal ideation and self-harm on key developmental outcomes for this age group (Davey and McGorry, 2019). However, it is also important to note that a minority (12.7%) of young people in this trial experienced severe suicidal ideation that worsened over the course of CBT treatment. For these young people, close monitoring of any potential adverse effects of treatment is warranted.
Conclusion
In this exploratory study, we identified two distinct trajectories for interview-rated depression symptoms, of which almost half of the young people experienced only slight improvement in depression severity. We also identified four trajectories for suicidal ideation. Allocation to either CBT + FLX or CBT + PLO was not associated with class membership. Depression trajectories were also not associated with class membership for suicidal ideation, suggesting that improvement in depression symptoms might not be associated with improvement in suicidal ideation. While effective treatment of depression is an important component of a multi-level intervention strategy for suicide prevention, suicidal ideation might be driven by factors beyond depression severity and it is important to intervene directly to address these factors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: O.D. has received grant support from the Brain and Behavior Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Deakin University, Lilly, NHMRC and ASBDD/Servier. She has also received in kind support from BioMedica Nutraceuticals, NutritionCare and Bioceuticals. All other authors declare no relevant conflicting interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The YODA-C trial received funding from the Australian National Health and Medical Research Council (NHMRC). Individual authors also report the following funding: M.B. is supported by two NHMRC Senior Principal Research Fellowships (1059660 and 1156072). O.D. is a R.D. Wright Biomedical NHMRC Career Development Fellow (1145634). S.C. is supported by a NHMRC Senior Research Fellowship (APP1136344). S.H. is funded by an Auckland Medical Research Foundation Douglas Goodfellow Repatriation Fellowship and is the CureKids Research Fellow. K.W. is funded by an Emerging Leadership 1 Fellowship awarded by the NHMRC.
