Abstract
Objective:
The emergence of new drugs for managing suicidal ideation (e.g. ketamine) raises the question of whether suicidal depression (i.e. moderate to severe depression with concomitant suicidal ideation) is a specific depression phenotype. Therefore, this study characterized patients with suicidal depression (baseline clinical characteristics, suicidal ideation and depression evolutions, suicide risk) in two large cohorts of outpatients with depression.
Methods:
LUEUR and GENESE are two large, prospective, naturalistic cohorts of French adult outpatients with depression (Diagnostic and Statistical Manual of Mental Disorders, fourth edition, criteria), treated and followed up for 6 weeks. Depression severity was assessed with the Hospital Anxiety and Depression Scale, and suicidal ideation with the suicidal item of the Montgomery–Åsberg Depression Rating Scale. Patients with moderate or severe depression (Hospital Anxiety and Depression Scale–Depression subscale score >11) were selected and classified as without suicidal ideation (suicidal item of the Montgomery–Åsberg Depression Rating Scale <2), with moderate suicidal ideation (suicidal item of the Montgomery–Åsberg Depression Rating Scale [2; 3]) and with severe suicidal ideation (suicidal item of the Montgomery–Åsberg Depression Rating Scale ⩾4).
Results:
Baseline clinical features were more severe (e.g. higher anxiety and depression scores) in depressed patients with moderate/severe suicidal ideation. Depression remission after treatment was less frequent among patients with severe suicidal ideation. The risk of suicide attempt during the follow-up was threefold higher in patients with suicidal ideation among those 10% had persistent suicidal ideation.
Conclusion:
Suicidal depression could be a specific depression phenotype with more severe clinical characteristics, less frequent depression remission, suicidal ideation persistence and higher suicide attempt risk, despite antidepressant treatment. It seems that novel therapeutic strategies could be needed.
Keywords
Introduction
With more than 800,000 deaths by suicide worldwide each year, and 20 times more suicide attempts (SAs) (World Health Organization 2019), suicidal behavior (SB) (i.e. completed suicide or SA) is a major public health problem. One way to prevent SB is to focus on suicidal ideation (SI) because SI is a major risk factor of future death by suicide (Franklin et al., 2017). Patients with a major depressive episode (MDE) (another important risk factor for SB) report SI in 45–70% of cases (Sokero, 2006). SI has been mainly considered a symptom or a consequence of a psychiatric disorder (Courtet and Nobile, 2020). Consequently, clinicians use psychotherapy (e.g. cognitive behavioral therapy [CBT], dialectical behavior therapy) (Ducasse et al., 2018) and non-SI-specific drugs (antidepressants, clozapine and lithium) to reduce SI and prevent SB (Courtet et al., 2020). Antidepressants represent the main drugs used in patients with MDE to reduce SI. Evidence on efficacy of antidepressant in suicidal patients is not convergent. On one hand, antidepressant reduces SI (notably by decreasing depression severity) (Weitz et al., 2014) and decreases SB risk by 40–60% (Rihmer and Gonda, 2013). On the other hand, depression remission after antidepressant treatment seems to be less frequent in depressed patients with SI or lifetime SA (Lopez-Castroman et al., 2016), and many of them report SI persistence (Seo et al., 2014). Moreover, most randomized controlled trials on antidepressants excluded suicidal patients (current SI and/or past history of SA) and a large part of studies were focused only on treatment-related emergence or worsening of SI. Similarly, clinical trials on CBT failed to show an improvement of suicidal outcome when CBT targeted mental illness, whereas CBT significantly reduced suicidal risk when it specifically targeted SI and SB rather than mental illness (Mewton and Andrews, 2016). Thus, further studies are needed to assess the course of pre-existing SI and the baseline clinical characteristics of depressed patients with SI (Courtet et al., 2020; Courtet and Nobile, 2020). Finally, some patients with MDE will never experience SI. Altogether, this raises the question of whether suicidal depression (i.e. moderate to severe depression with SI) could be a specific phenotype of depression needing specific treatments.
Depressed patients with SI have more severe baseline sociodemographic and clinical characteristics (e.g. more often single, higher depression and hopelessness severity, more lifetime SA), and less frequent remission of depression after antidepressant treatment (Lopez-Castroman et al., 2016; Trivedi et al., 2013; Zimmerman et al., 2018). SI course is related to, but separated from that of, depression. For instance, Batterham et al. (2019) recently described two trajectory types with different outcomes for SI and for depressive symptoms. They found that class membership for the two outcomes was quite independent. Another study showed that depressive symptomatology and its course were significantly different in suicidal and non-suicidal depressed patients (van Ballegooijen et al., 2019). About 20% of patients present persistent SI despite the improvement or remission of depression (Batterham et al., 2019; Nobile et al. 2021; Seo et al., 2014). Finally, growing evidence suggests SB has its own physiopathology (e.g. opioid system dysfunction) (Nobile et al. 2020), suggesting that suicidal depression could have a different physiopathology than depression without SI. A depressive episode with SI would thus need targeted treatments (van Ballegooijen et al., 2019).
To determine whether suicidal depression is a specific phenotype is important because promising new drugs are emerging for SI, particularly opiates (e.g. buprenorphine, tianeptine) (Nobile et al., 2018; Yovell et al., 2016) and the N-methyl-
We aimed to determine whether suicidal depression was a specific depression phenotype by characterizing patients with suicidal depression (baseline clinical characteristics, SI and depressive symptom changes over time, suicidal risk) identified in two large cohorts of outpatients with an MDE. We decided to conduct our analyses in the two independent cohorts separately because of the different eligibility criteria, (i.e. alcohol and substance dependence; co-treatment with mood stabilizer or antipsychotic) that could impact depression and SI courses. Moreover, this allowed observing whether main results in one cohort were replicated in the second cohort and thus to test the replication of our main hypothesis (i.e. suicidal depression could be a specific subtype of depression) in two large independent cohorts of depressed outpatients recruited throughout France.
Material and methods
Participants’ recruitment
LUEUR and GENESE are two large, prospective, naturalistic cohorts of more than 3000 French adult outpatients with a diagnosis of MDE according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition, (DSM-IV) criteria. These cohorts were created to study depression and SB. This study is not a post hoc analysis from another study (Courtet et al., 2014; Voegeli et al., 2016). Participants were recruited by general practitioners or psychiatrists at primary and psychiatric care clinical centers throughout France. The same physician assessed the patient at the first (baseline) and second visit (after at least 6 weeks). In both studies, non-inclusion criteria were younger than 18 years of age, bipolar disorder, schizophrenia, primary substance misuse, primary organic disease and pregnancy or lactation. Additional non-inclusion criteria in the GENESE study were non-Caucasian ethnicity, and alcohol and substance dependence. Current treatment with an antipsychotic or a mood stabilizer was an additional non-inclusion criterion in the LUEUR study.
In the GENESE cohort, all patients were treated with tianeptine (between 12.5 and 37.5 mg/day). Tianeptine was systematically prescribed as the first antidepressant medication or as a change of antidepressant molecule. In the LUEUR cohort, all patients were treated with antidepressants approved by the European Medicines Agency, although most patients received tianeptine. Of note, tianeptine was one of the most frequently prescribed antidepressants in France at the time of the trials (Gusmão et al., 2013). In these studies, the goal was not to assess the effectiveness of one particular antidepressant. In both cohorts, other concomitant treatments for current somatic problems or sleep or anxiety disorders were allowed, based on clinical judgment. These studies were performed according to the French regulatory guidelines and the current Good Clinical Practice, and were approved by local independent ethics committees. All patients were informed about the study aims and procedures and signed a written informed consent.
Clinical assessment
Demographic data, number of depressive episodes and lifetime history of SA were assessed at baseline. Physicians evaluated depression severity and SI at baseline and at week 6. Patients self-rated depression severity and SI at baseline, week 1, week 2 and week 6 in the LUEUR study, and at baseline, week 2, week 4 and week 6 in the GENESE study.
Depression severity was assessed with the French self-rated version of the Hospital Anxiety and Depression Scale (HADS). In addition to Anxiety (HADS-A) and Depression (HADS-D) subscales, an agitation subscore of the HADS was calculated (sum of items 1, 7 and 11) (Lopez-Castroman et al., 2020). This scale was chosen for its simplicity of use, good comprehensibility and robust psychometric properties, demonstrated also in outpatient groups (Demyttenaere et al., 2009). In this scale, depressive symptom severity is assessed independently of any SI/SB.
SI was evaluated using the suicidal item of the self-rated Montgomery–Åsberg Depression Rating Scale (MADRS) (item number 10, or MADRS-SI). The ratings range from 0 to 6 (0–1: enjoys life or takes it as it comes; 2–3: weary of life, only fleeting suicidal thoughts; 4–5: probably better off dead, suicidal thoughts are common and suicide is considered a possible solution, but without specific plans or intention; and 6: explicit plans for suicide when there is an opportunity, active preparations for suicide). A previous study showed that a single suicide item from a depression rating scale, either clinician-rated or self-reported, is a valid approach to assess SI compared with the Beck’s scale (Desseilles et al., 2012). This method was used in large clinical studies, such as the STAR*D (Zisook et al., 2009) and also in more recent studies (Ballard et al., 2017; Bernert et al., 2017; Nobile et al., 2018).
SA occurrence was assessed at the end of the study (week 6).
Hopelessness was assessed using the self-report version of the Beck Hopelessness Scale in the LUEUR cohort at baseline, week 1, week 2 and week 6. Impulsivity and sleep disturbance were assessed using the self-report version of the Plutchik’s questionnaire (Anon, n.d.) and the item on sleep of the MADRS, respectively, in the GENESE study at baseline, week 2, week 4 and week 6.
Definition of suicidal depression
In line with the inclusion criteria of recent clinical trials on antisuicidal agents (that used either Beck Depression Inventory; Hamilton Rating Scale for Depression; MADRS; Question B5 and B9 of the mini international neuropsychiatric interview (MINI) or Suicidal Ideation Scale) (Canuso et al., 2018; Grunebaum et al., 2018), only patients with moderate to severe depression were included (i.e. patients with HADS-D score >11 at baseline, according to the definition of the French High Authority of Health). Thus, 3969 patients (81.9%) from the LUEUR cohort and 2537 patients (71.1%) from the GENESE cohort were selected. The baseline MADRS-SI score was used to classify patients into three groups: (1) depression without SI (score <2), (2) depression with moderate SI (score between 2 and 3) and (3) depression with severe SI (score ⩾4), as done in previous studies (Lopez-Castroman et al., 2016; Reeves et al., 2008). A MADRS-SI score between 2 and 3 can be indicative of passive suicidal thoughts, and a score ⩾4 of active SI (Ballard et al., 2017).
Definition of response to treatment and remission of depression
Response was defined by a decrease by at least 50% of their baseline HADS-D score. Depression remission was defined by a final HADS-D score of ⩽7 (at week 6).
Definition of severe SI persistence and of SI remission
SI persistence was defined by a MADRS-SI score of ⩾4 at week 6 in patients who had a score of ⩾4 at baseline.
SI remission was defined by a MADRS-SI score of <2 at week 6 in patients who had a score of ⩾4 at baseline.
Lost to follow-up
Some patients were lost to follow-up in both cohorts. In the LUEUR cohort, 289 (7.3%) patients were lost to follow-up (7.3% of patients with depression without SI, 7.8% of patients with moderate suicidal depression and 6.1% of patients with severe suicidal depression). In the GENESE cohort, 71 (2.8%) patients were lost to follow-up (3.5% of patients with depression without SI, 1.9% of patients with moderate suicidal depression and 4% of patients with severe suicidal depression). These patients were included in the comparison of baseline sociodemographic and clinical data, but excluded from all other analyses.
Statistical analysis
Categorical variables were presented as percentages, and quantitative variables as means with standard deviation (SD). The group with depression without SI was chosen as reference for all analyses. The demographic and clinical characteristics of the three groups were compared using a multinomial logistic regression model adjusted for depression severity (HADS-D score) and the likelihood-ratio test (LRT). The adjustment on depression severity was previously used to assess the association between SI and other variables independently of depression (Perroud et al., 2010; Rogers et al., 2018; Zisook et al., 2011). Bonferroni correction was applied for post hoc analyses.
Curves to describe the HADS-D and MADRS-SI score changes during the follow-up were created, and a linear regression model was used to test whether intra-group score changes were significant. The slopes (β) and p values of the different curves are indicated in the graphs. A generalized least squares (GLS) linear regression model was used to test for global and longitudinal between-group differences at the depression level. First-order autoregressive temporal correlation structure was used to take account to the intra-individual correlation of the residual. Significances were established from LRT in the adjusted GLS model. Adjustment variables were chosen according to their association with depression score changes (variables associated with a value of p < 0.10 were included in the model). Patients with missing data for one of the adjustment variables were excluded from this analysis.
Treatment response and depression remission during the follow-up were compared using multivariate logistic regression model adjusted for variables associated with depression remission and treatment response.
Risk factors of SI persistence and predictive factors of SI remission at the end of the follow-up period in patients with severe SI at baseline were evaluated with a logistic regression. All variables associated with SI persistence and SI remission with a value of p < 0.10 were included in a multivariate model to find the strongest predictors. Adjusted odds ratios (ORs) and 95% confidence intervals (CIs) were estimated. To test whether SI changes were associated with changes in depression, anxiety, impulsivity (GENESE), sleep (GENESE) and hopelessness (LUEUR), their respective temporal variations were estimated from the slopes of simple linear regression models fitted on each patient. Then, the association between these variations was tested with analysis of variance (ANOVA) type III, first in an unadjusted model and then after adjustment for depression changes. The R2 change was also examined to quantify how much of SI variance was explained by anxiety, impulsivity, sleep and hopelessness when the depression score changes were included in the model.
SA risk during the follow-up was compared using a multivariate logistic regression model. Adjustment variables were chosen according to their association with SA during the follow-up. Sensitivity analyses were performed by excluding patients with lifetime SA (see Supplemental Material).
All multinomial logistic regression models were performed with SPSS software (version 23.0.0.2; IBM SPSS Statistics for Windows. Armonk, NY: IBM Corp), and all linear regression analyses and the GLS model were performed with R software (version 3.6). The significance level was set at p < 0.05.
Results
Comparison of the baseline sociodemographic and clinical data
In the LUEUR cohort, 671 patients had depression without SI (16.90%), 2292 had depression with moderate SI (57.75%) and 1006 had depression with severe SI (25.35%) (Table 1). In the GENESE cohort, 370 patients had depression without SI (14.58%), 1408 had depression with moderate SI (55.50%) and 759 had depression with severe SI (29.92%). About 20% of outpatients from both cohorts had depression with severe SI, 60% were women and the mean age was around 45 years.
Association between sociodemographic and clinical data and suicidal depression.
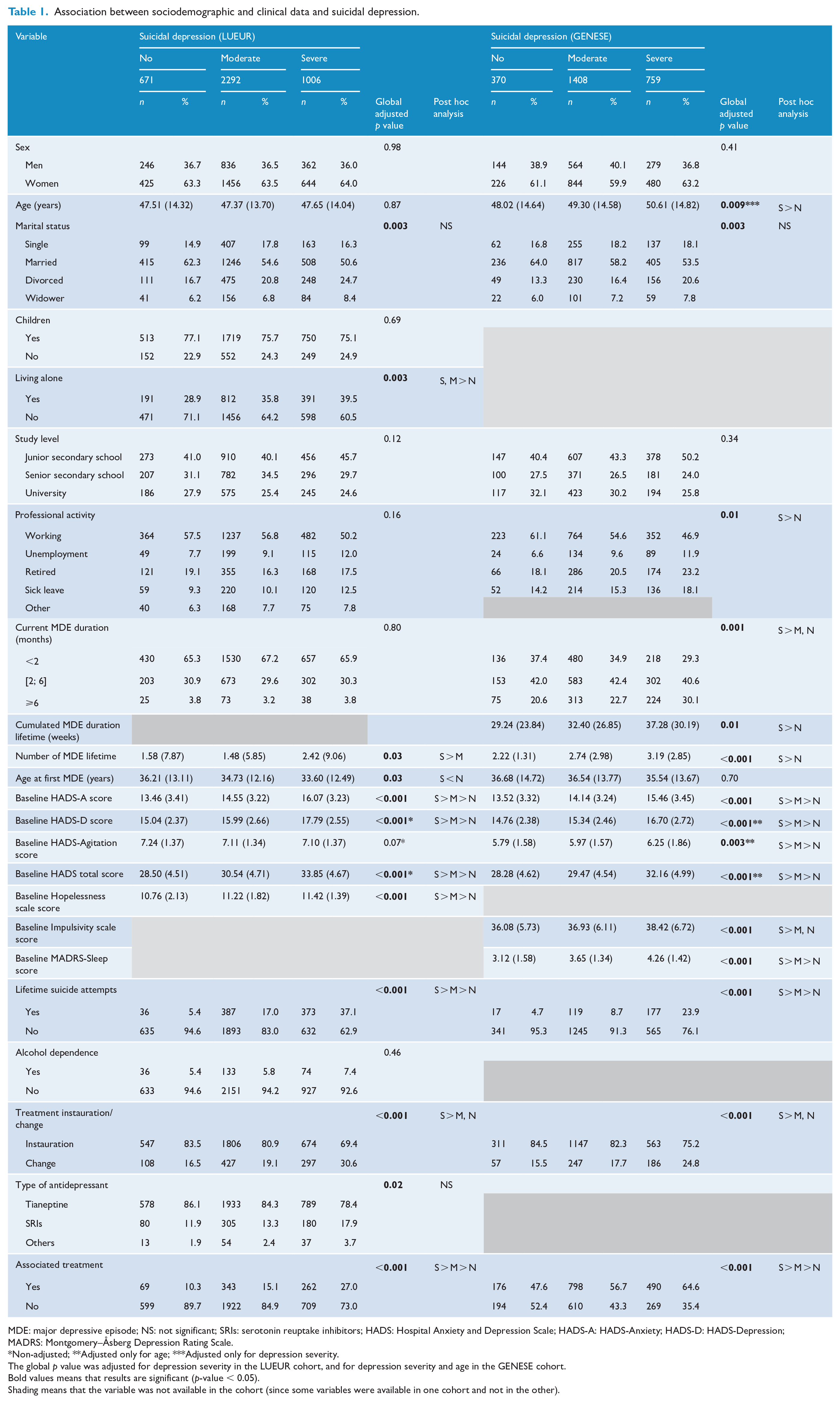
MDE: major depressive episode; NS: not significant; SRIs: serotonin reuptake inhibitors; HADS: Hospital Anxiety and Depression Scale; HADS-A: HADS-Anxiety; HADS-D: HADS-Depression; MADRS: Montgomery–Åsberg Depression Rating Scale.*Non-adjusted; **Adjusted only for age; ***Adjusted only for depression severity.
The global p value was adjusted for depression severity in the LUEUR cohort, and for depression severity and age in the GENESE cohort.Bold values means that results are significant (p-value < 0.05).Shading means that the variable was not available in the cohort (since some variables were available in one cohort and not in the other).
In both cohorts, patients with severe SI were more likely to be living alone. In the GENESE cohort, patients with severe SI were older than patients with moderate or without SI.
In both cohorts, lifetime MDE number was higher, and lifetime history of SA and associated treatments (e.g. anxiolytics, hypnotics) were more frequent in patients with moderate or severe SI than in patients without SI. Moreover, the HADS-A and HADS-D scores and HADS total score were higher in patients with moderate and severe SI. Then, the levels of hopelessness (LUEUR cohort), impulsivity (GENESE cohort) and the MADRS-Sleep score (GENESE cohort) were higher in patients with moderate and severe SI than in patients without SI. It is important to point out that antidepressant type did not differ among patients’ group in the LUEUR cohort.
To identify differences related to suicidal phenotype, analyses to find variables associated with suicidal depression were adjusted for depression severity in both cohorts, and for age in the GENESE cohort.
When patients with lifetime history of SA were removed from the analysis, results did not change (except for sociodemographic data which were no longer different among groups) (Supplemental Table S1).
Depression score changes during the follow-up
In both cohorts, HADS-D scores significantly decreased in each group during the follow-up (Figure 1). In the LUEUR cohort, but not in the GENESE cohort, the HADS-D score decrease over time was different according to the patients’ groups (respectively, global p < 0.0001 for the unadjusted and adjusted models and global p = 0.07 for the unadjusted and adjusted models; Table 2).
Multivariate analysis of HADS-D total score changes during the follow-up.
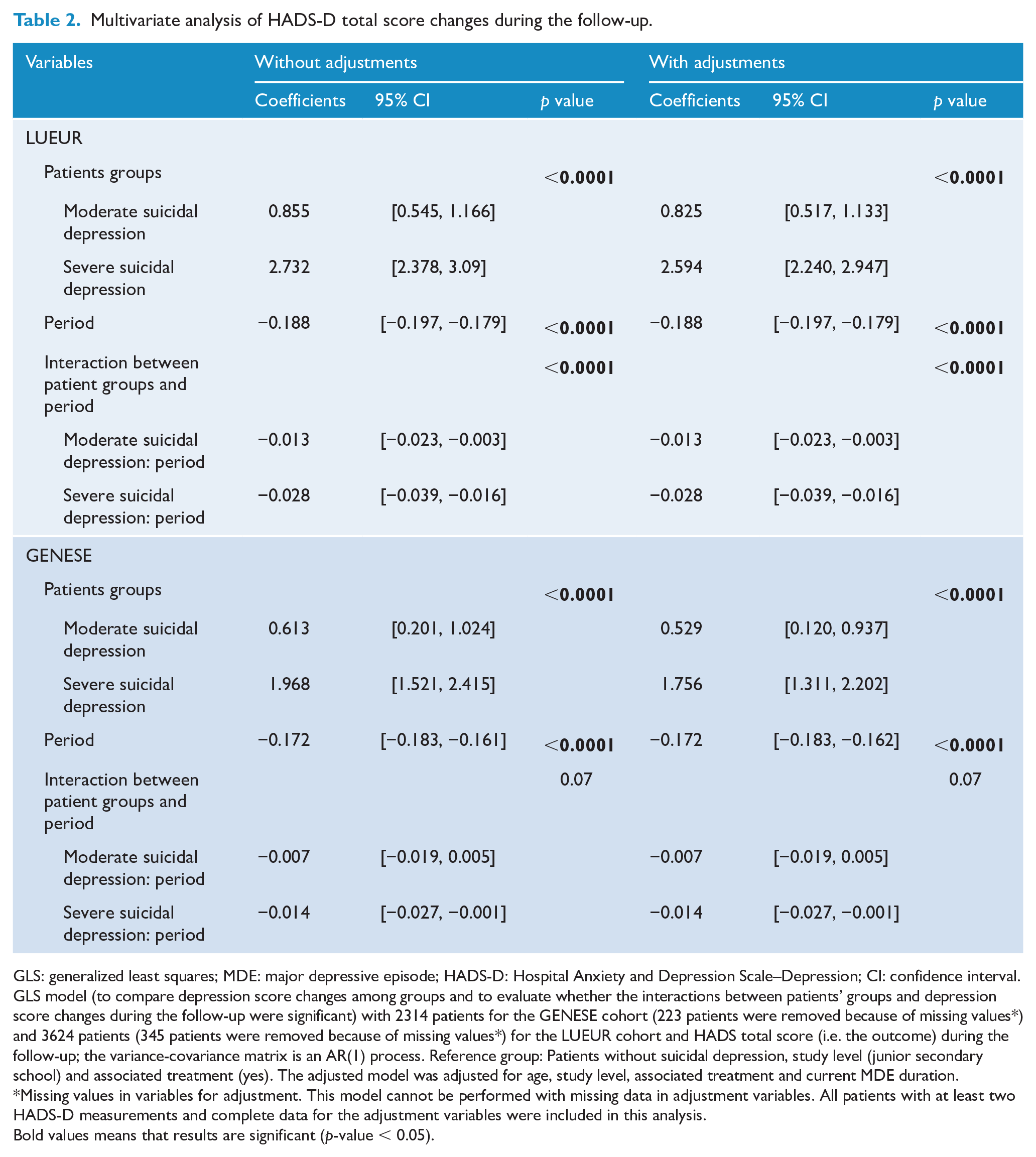
GLS: generalized least squares; MDE: major depressive episode; HADS-D: Hospital Anxiety and Depression Scale–Depression; CI: confidence interval.
GLS model (to compare depression score changes among groups and to evaluate whether the interactions between patients’ groups and depression score changes during the follow-up were significant) with 2314 patients for the GENESE cohort (223 patients were removed because of missing values*) and 3624 patients (345 patients were removed because of missing values*) for the LUEUR cohort and HADS total score (i.e. the outcome) during the follow-up; the variance-covariance matrix is an AR(1) process. Reference group: Patients without suicidal depression, study level (junior secondary school) and associated treatment (yes). The adjusted model was adjusted for age, study level, associated treatment and current MDE duration.
Missing values in variables for adjustment. This model cannot be performed with missing data in adjustment variables. All patients with at least two HADS-D measurements and complete data for the adjustment variables were included in this analysis.Bold values means that results are significant (p-value < 0.05).
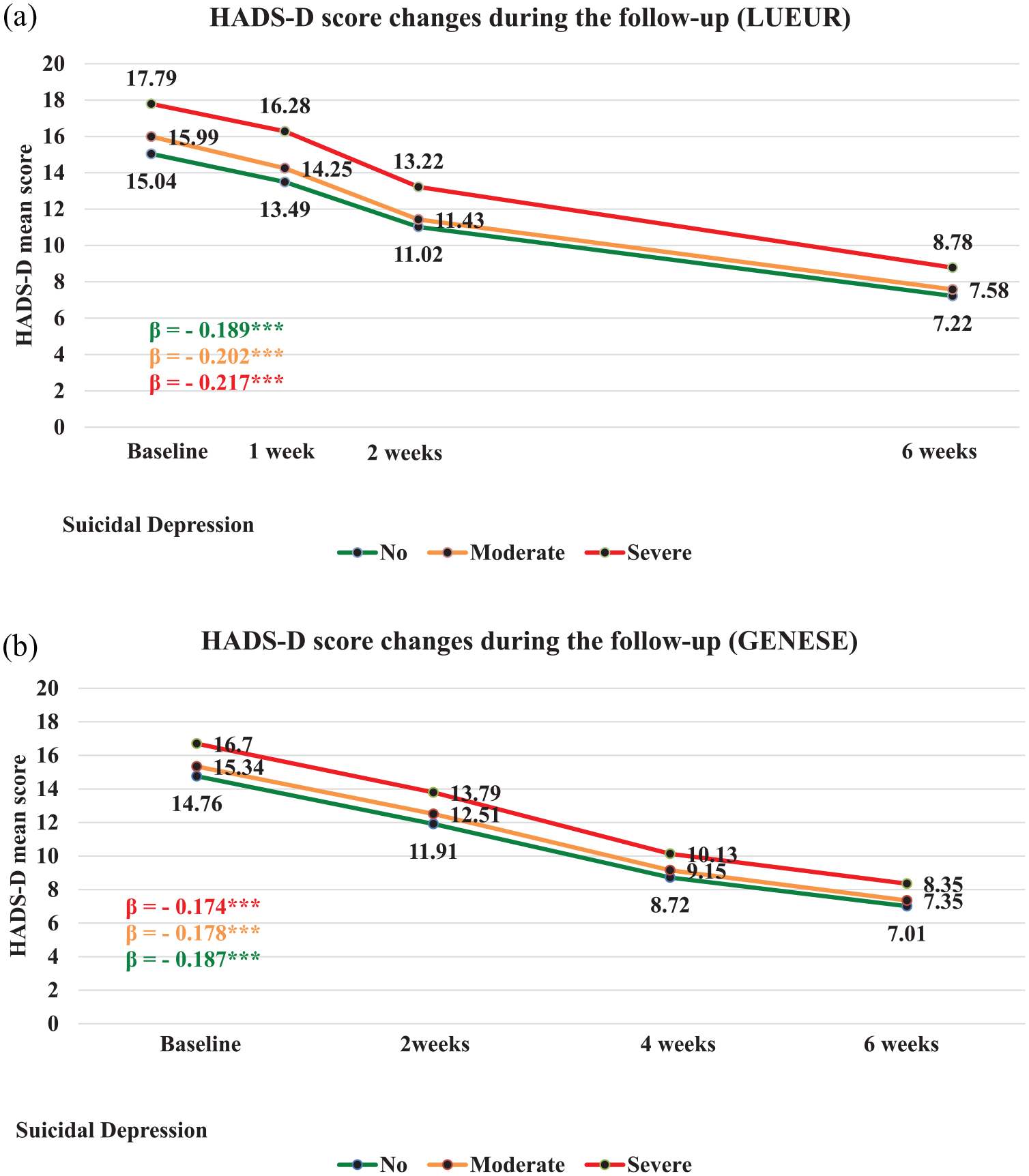
HADS-D score changes during the follow-up in the three groups of the (A) LUEUR and (B) GENESE cohorts.
Response to treatment (i.e. 50% reduction of the baseline HADS-D score) was not significantly different among groups in unadjusted models. However, when the analysis was adjusted for potential cofounders (listed in Table 3), there were significant differences at week 6 in the LUEUR cohort (OR = 0.77; 95% CI = [0.61, 0.97]; global p = 0.04) (Table 3) and a trend for an association in the GENESE cohort.
Response to treatment during the follow-up.
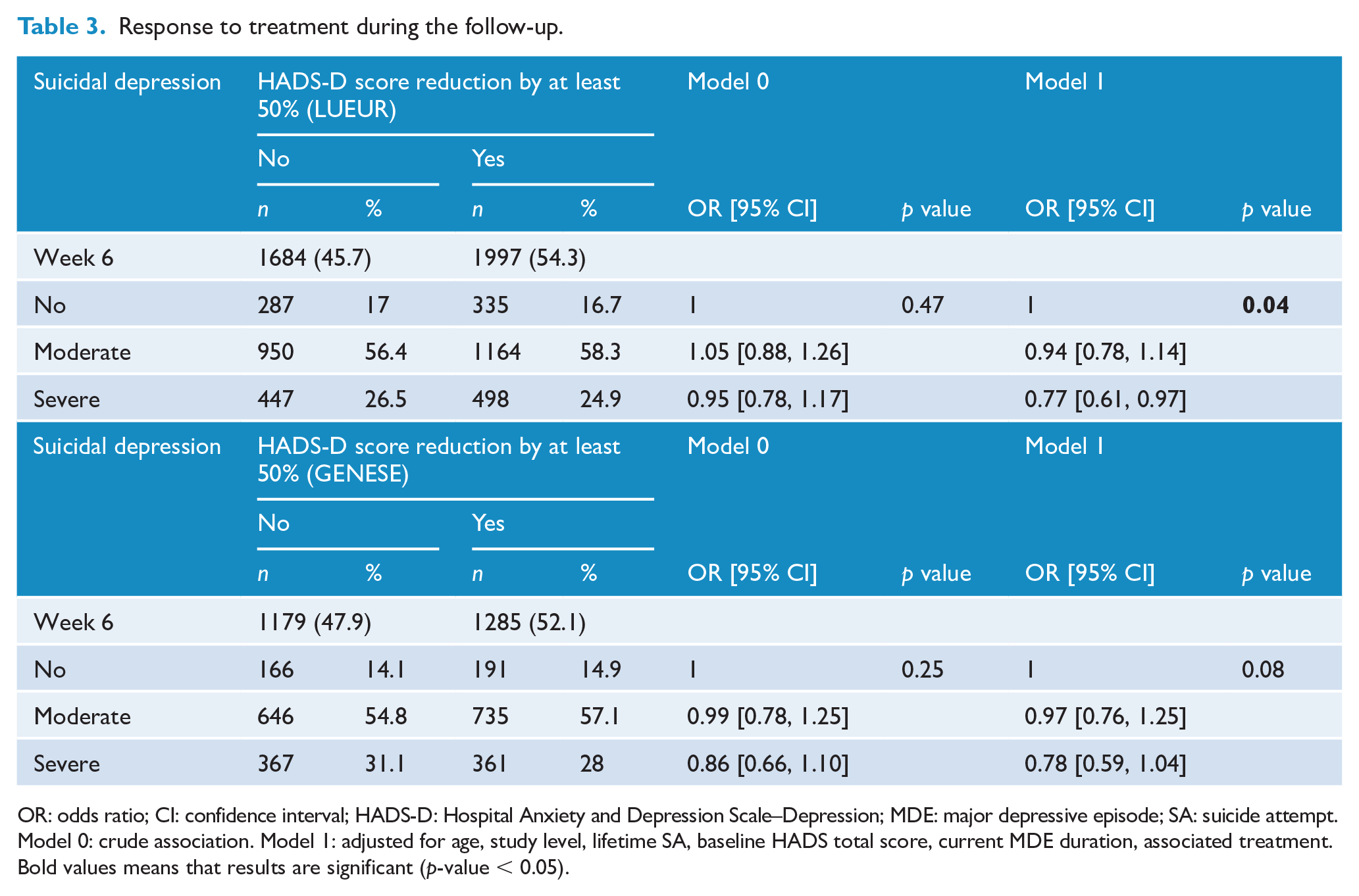
OR: odds ratio; CI: confidence interval; HADS-D: Hospital Anxiety and Depression Scale–Depression; MDE: major depressive episode; SA: suicide attempt.
Model 0: crude association. Model 1: adjusted for age, study level, lifetime SA, baseline HADS total score, current MDE duration, associated treatment.
Bold values means that results are significant (p-value < 0.05).
At week 6, depression remission (HADS-D score ⩽7) was observed in 53.5% (LUEUR) and 53.5% (GENESE) of depressed patients without SI, 50.1% (LUEUR) and 51% (GENESE) of patients with moderate SI and 39.7% (LUEUR) and 42.6% (GENESE) of patients with severe SI. Depression remission rates were significantly lower in patients with severe SI than in patients without SI in both cohorts (univariate analyses). This difference remained significant in the adjusted model in the LUEUR cohort (OR = 0.62; 95% CI = [0.44, 0.88]; global p = 0.01), but not in the GENESE cohort (OR = 0.80; 95% CI = [0.61, 1.05]; global p = 0.16) (Table 4). When patients with lifetime history of SA were removed from the analysis, the rate of depression remission remained lower in patients with severe SI than in the other two groups in the unadjusted and adjusted models (Supplemental Table S2).
Remission of depression in each group at week 6.
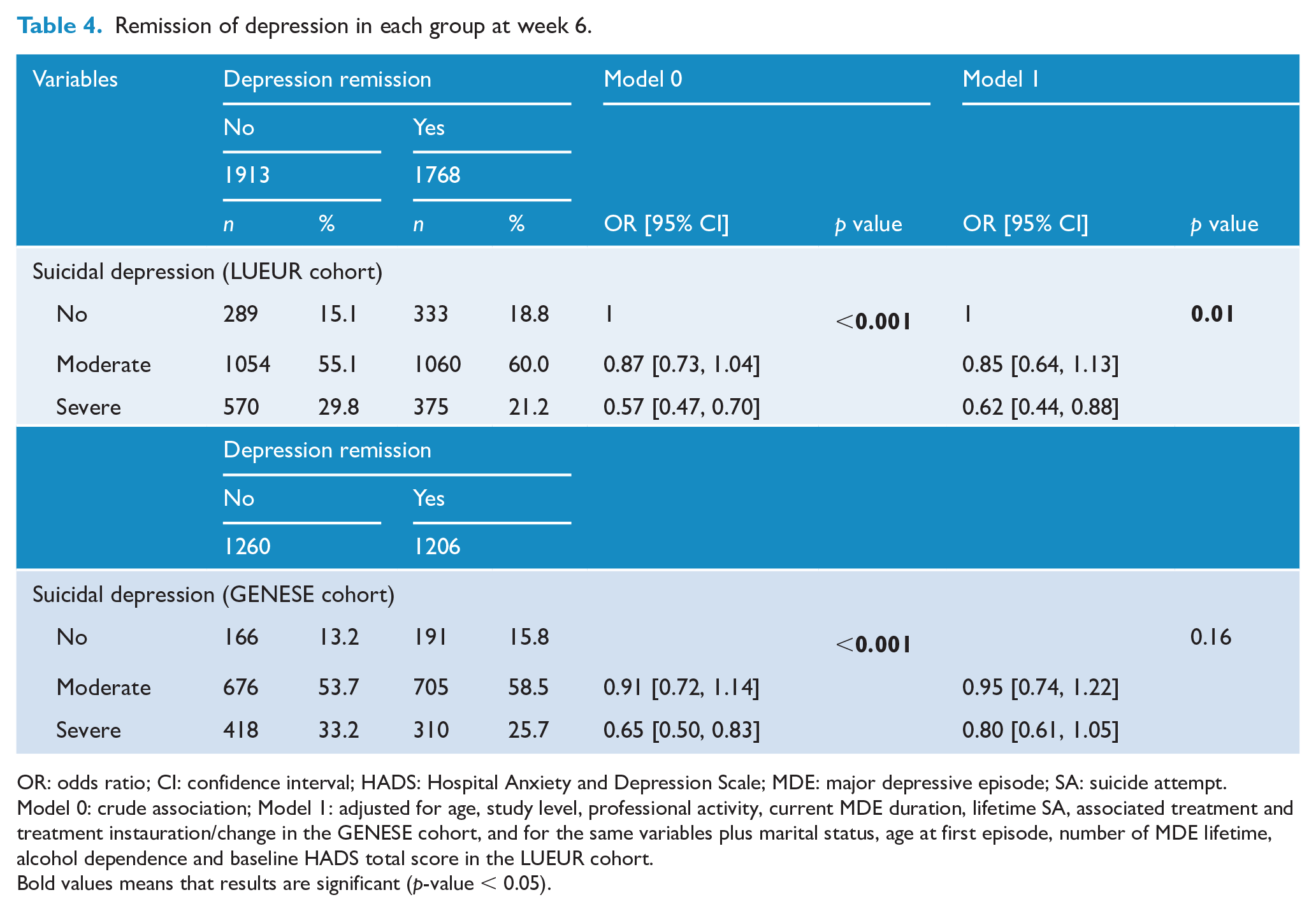
OR: odds ratio; CI: confidence interval; HADS: Hospital Anxiety and Depression Scale; MDE: major depressive episode; SA: suicide attempt.
Model 0: crude association; Model 1: adjusted for age, study level, professional activity, current MDE duration, lifetime SA, associated treatment and treatment instauration/change in the GENESE cohort, and for the same variables plus marital status, age at first episode, number of MDE lifetime, alcohol dependence and baseline HADS total score in the LUEUR cohort.Bold values means that results are significant (p-value < 0.05).
SI changes during the follow-up
SI significantly decreased in all three patient groups during the follow-up (Figures 2 and 3). Nevertheless, SI (MADRS-SI score ⩾2) occurrence remained higher in patients with moderate and severe SI than in patients without SI in both cohorts. At week 6, 12.2% (GENESE cohort) and 7.4% (LUEUR cohort) of patients with severe SI still reported severe SI (MADRS-SI score ⩾4).
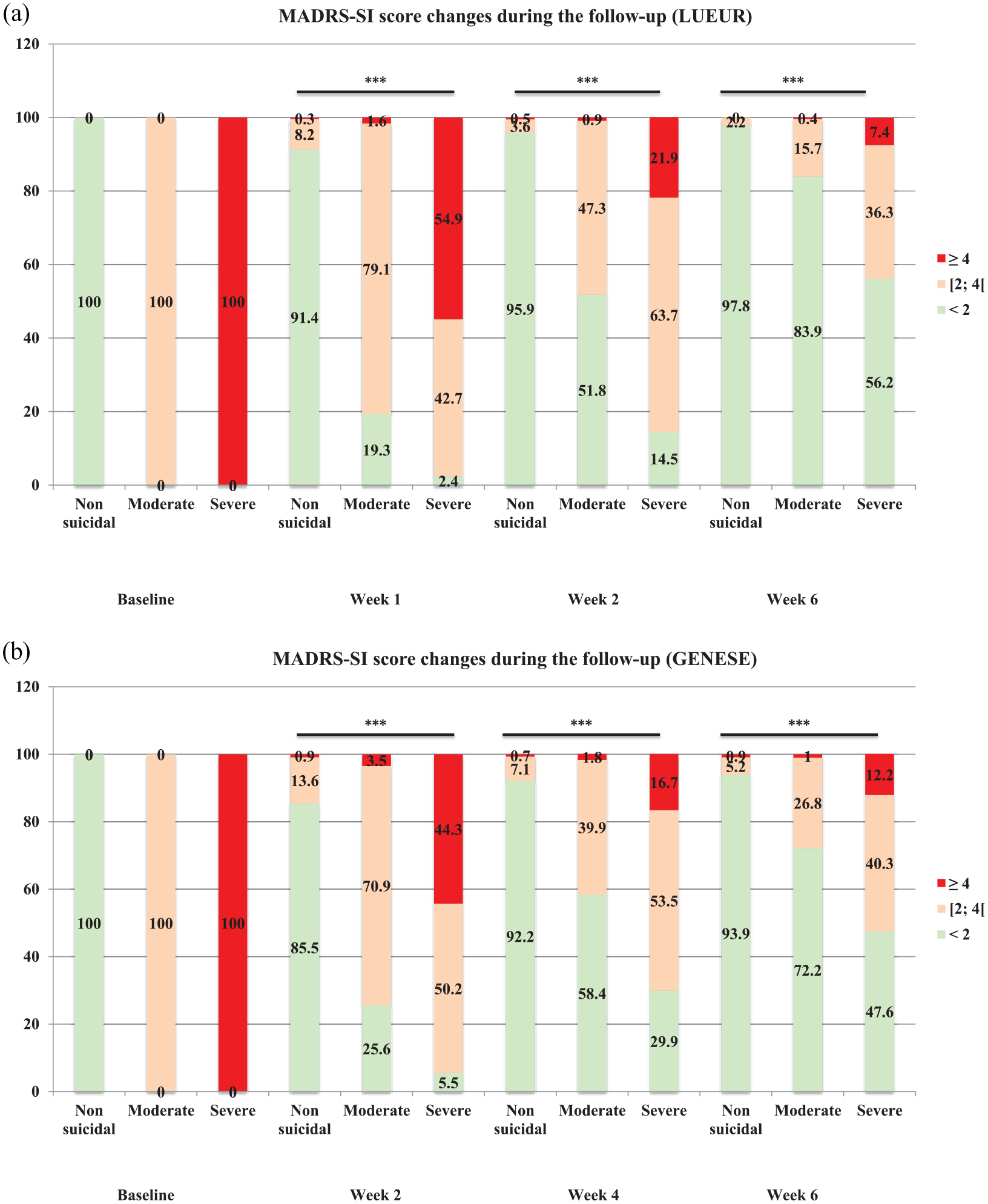
MADRS-SI score changes during the follow-up in the three groups of the (A) LUEUR and (B) GENESE cohorts.
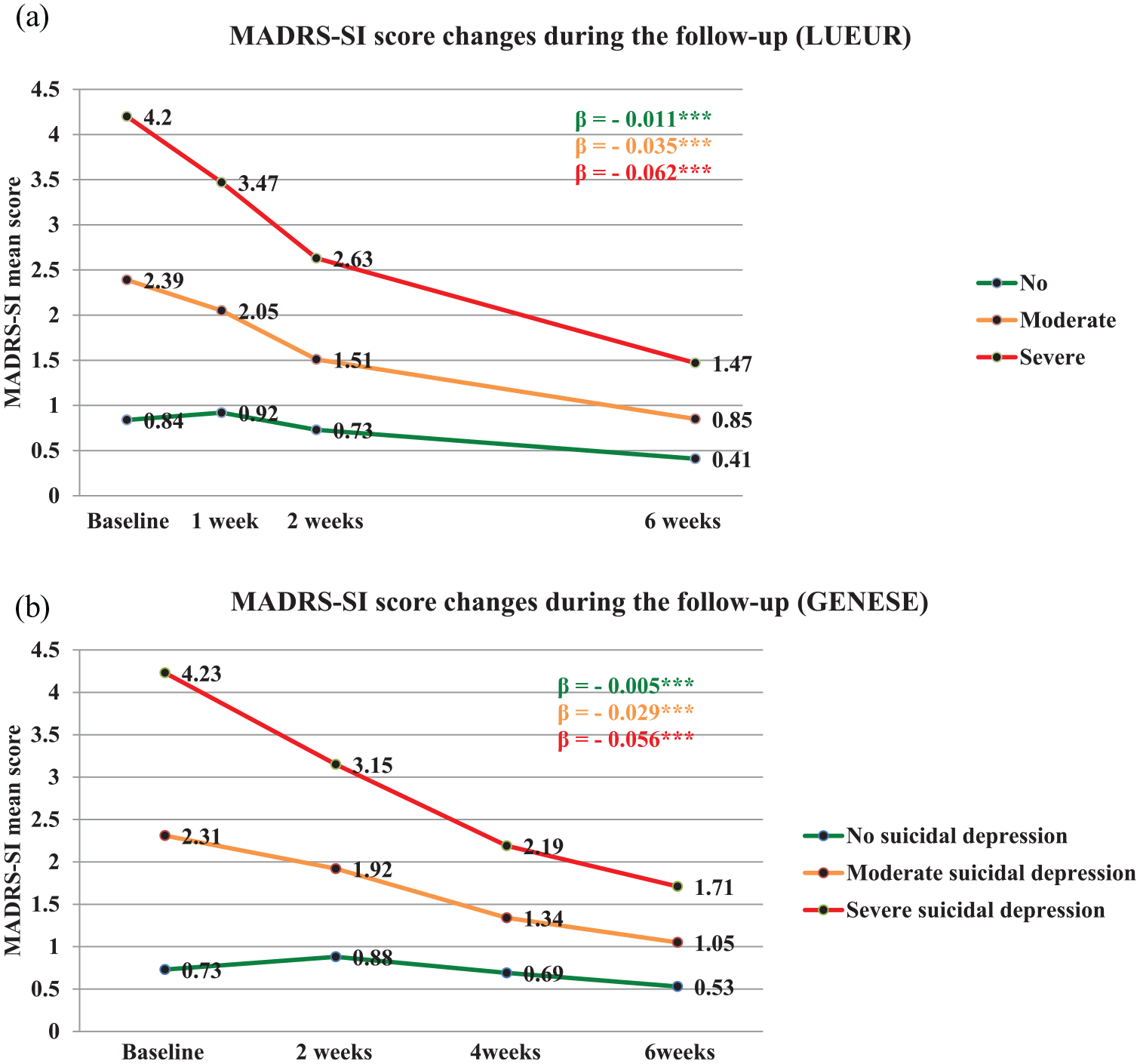
MADRS-SI score changes during the follow-up in the three groups of the (A) LUEUR and (B) GENESE cohorts.
In both cohorts, the predictors of persisting severe SI at week 6 were longer MDE duration, lifetime history of SA, higher baseline MADRS-SI score and lower HADS score decrease, as well as lower decreases of Hopelessness scale (LUEUR cohort), Impulsivity scale and MADRS-Sleep scores (GENESE cohort) (Supplemental Table S3). In the multivariate model, the main predictors were higher baseline MADRS-SI score, lower improvement of the HADS-D score and lower decrease of the MADRS-Sleep score (Table 5).
Risk factors for persistent severe SI at the follow-up end in patients with severe suicidal depression at baseline (multivariate analysis).
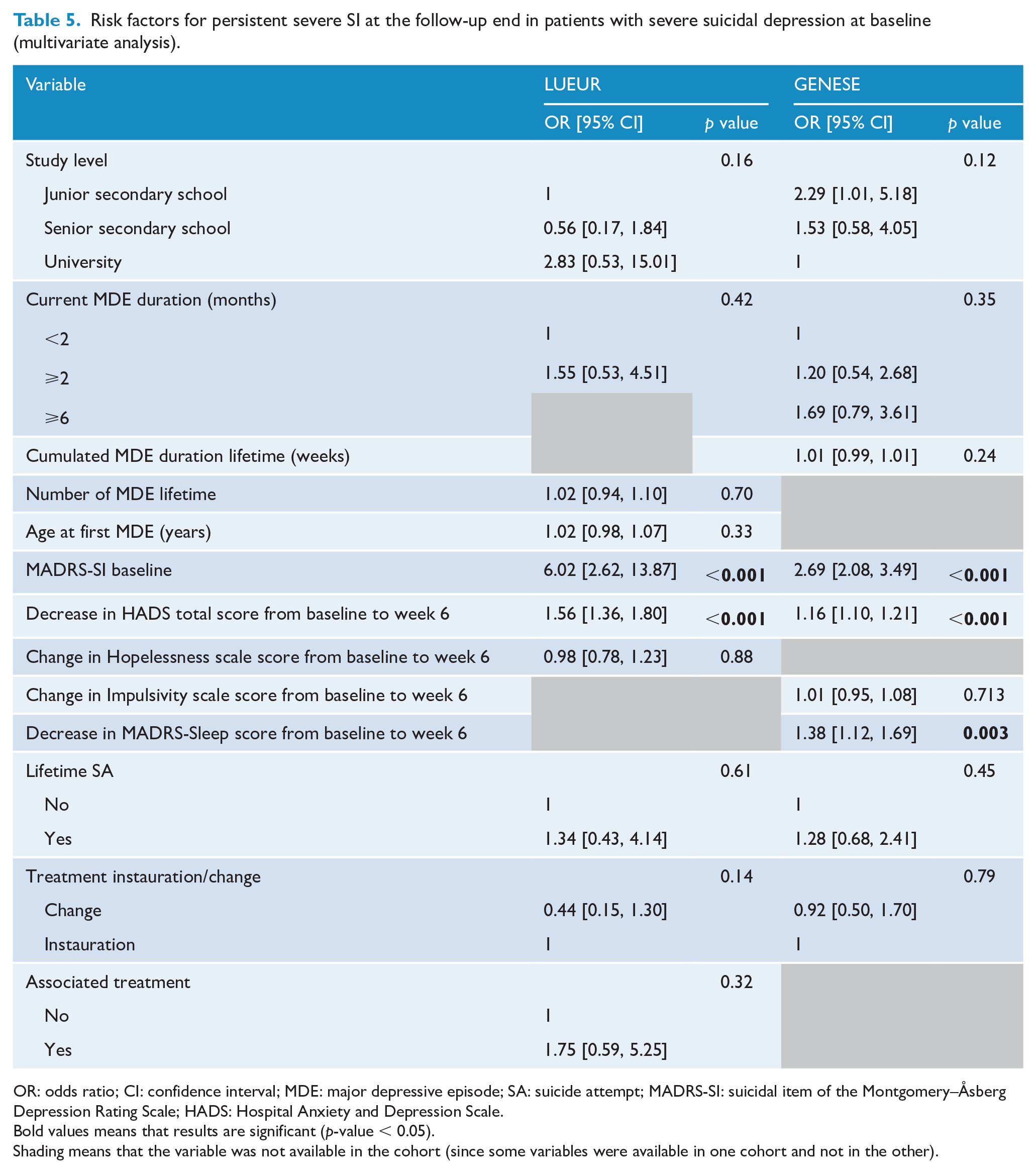
OR: odds ratio; CI: confidence interval; MDE: major depressive episode; SA: suicide attempt; MADRS-SI: suicidal item of the Montgomery–Åsberg Depression Rating Scale; HADS: Hospital Anxiety and Depression Scale.Bold values means that results are significant (p-value < 0.05).Shading means that the variable was not available in the cohort (since some variables were available in one cohort and not in the other).
SI remission at week 6 in patients with severe SI at baseline was predicted by shorter MDE duration; higher decrease in the first 2 weeks of depression, anxiety, hopelessness, impulsivity, sleep disturbances and SI; fewer lifetime SA; lower baseline MADRS-SI score; and instauration rather than a change of antidepressant treatment in both cohorts (Supplemental Table S4). In the multivariate model, fewer lifetime SA and higher early decrease of the MADRS-SI score were the only predictive factors of SI remission that remained significant for both cohorts (Table 6).
Predictors of SI remission (MADRS-SI score <2) at the follow-up end in patients with severe suicidal depression at baseline (multivariate analysis).
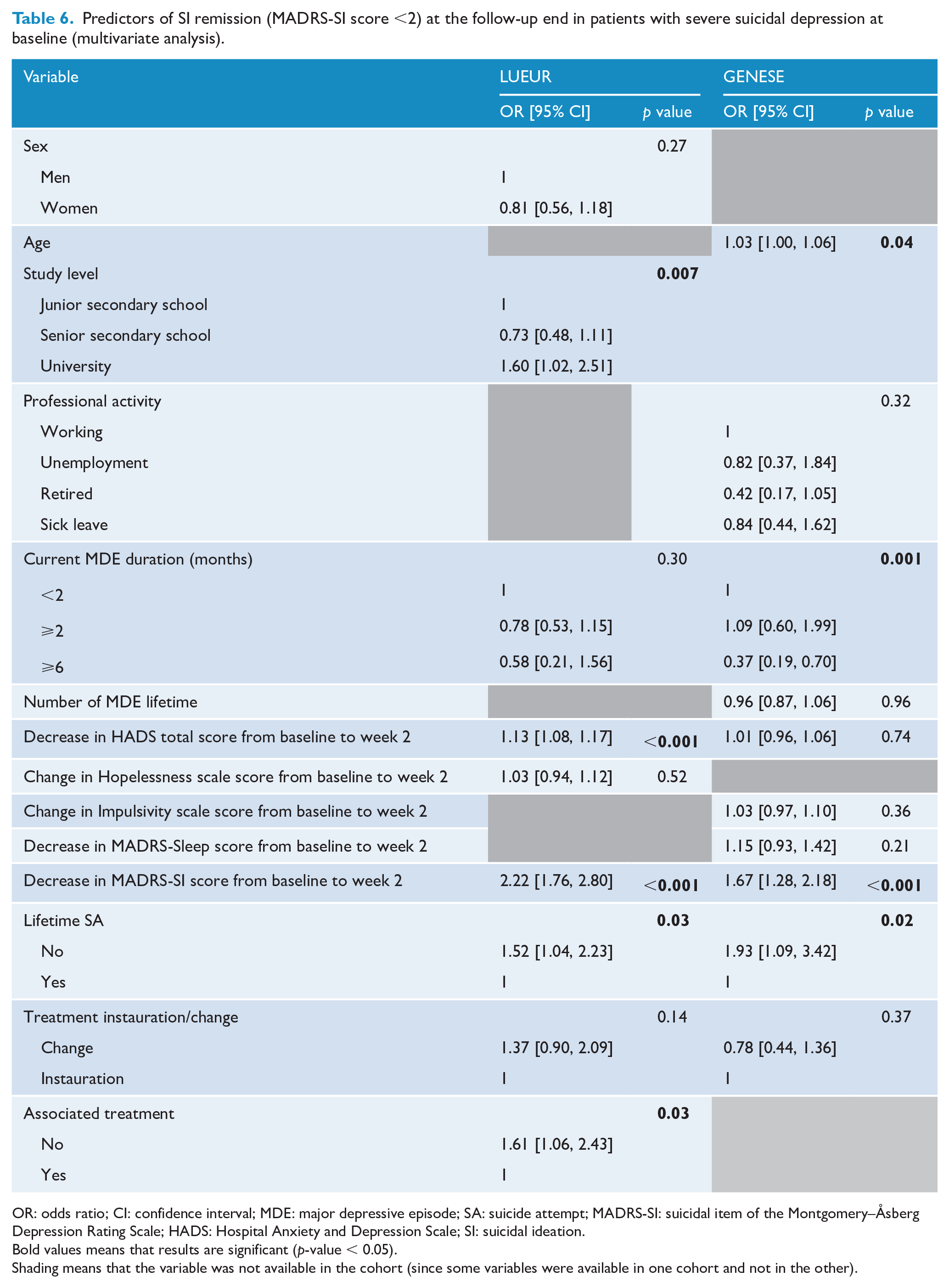
OR: odds ratio; CI: confidence interval; MDE: major depressive episode; SA: suicide attempt; MADRS-SI: suicidal item of the Montgomery–Åsberg Depression Rating Scale; HADS: Hospital Anxiety and Depression Scale; SI: suicidal ideation.Bold values means that results are significant (p-value < 0.05).Shading means that the variable was not available in the cohort (since some variables were available in one cohort and not in the other).
The analysis of variables that explained SI variation over time showed that 23% (LUEUR) and 25% (GENESE) of SI variances were explained by changes in the HADS-D score, 1.2% (LUEUR) and 1% (GENESE) by changes in the HADS-A score, 4.5% (GENESE only) by changes in the sleep score and less than 1% by changes in the Hopelessness (LUEUR) and Impulsivity (GENESE) score (Table 7). When considering only patients with severe SI at baseline, depression score changes explained 39% (GENESE) and 46% (LUEUR) of SI variance (Supplemental Table S5).
Linear regression analysis of MADRS-SI score variation in all patients.
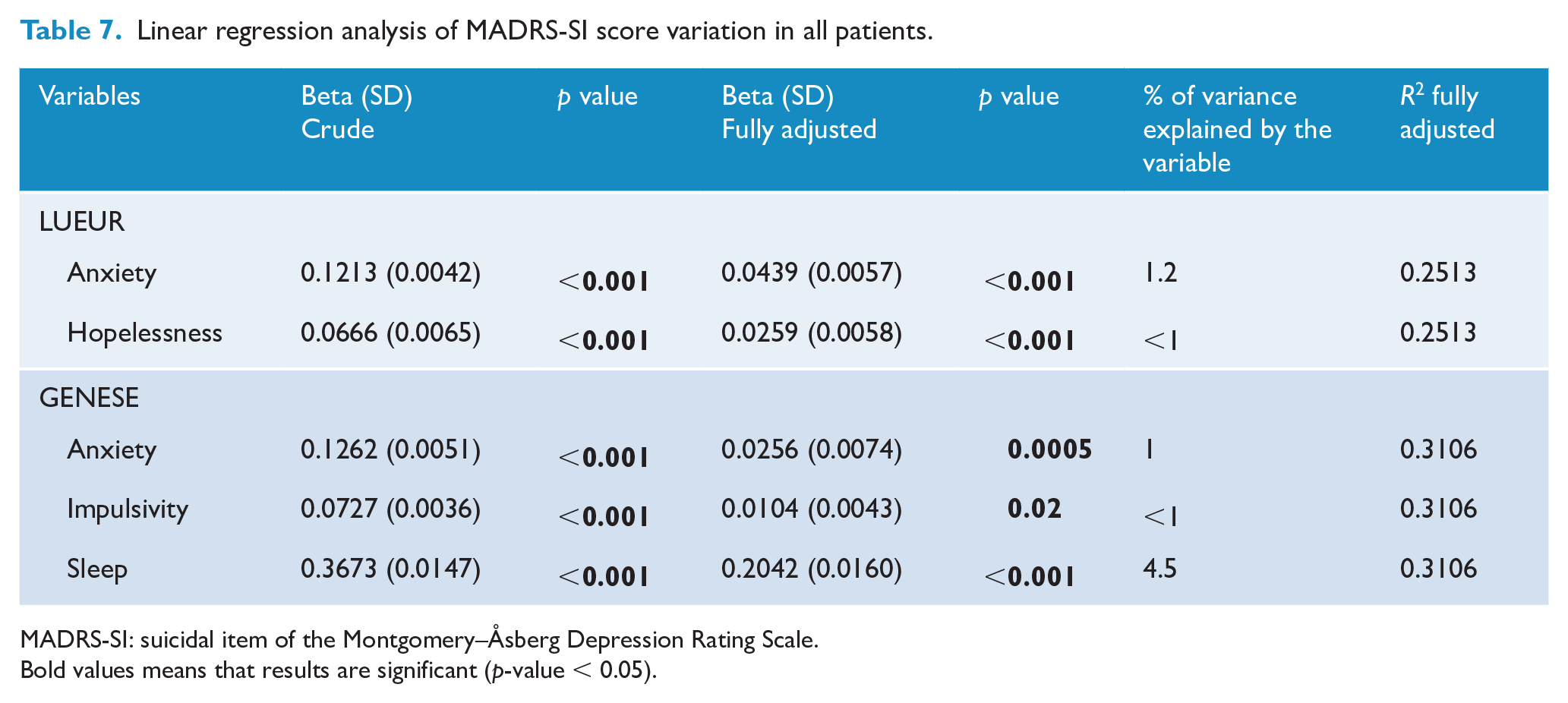
MADRS-SI: suicidal item of the Montgomery–Åsberg Depression Rating Scale.Bold values means that results are significant (p-value < 0.05).
At week 6, among all patients with SI at baseline (MADRS-SI ⩾2), SI (but not depression) was still present in 46 (1.5%) and 115 (5.6%) patients, whereas depression (but not SI) was still present in 900 (39.2%) and 434 (21.2%) patients in the LUEUR and GENESE cohorts, respectively. Among the patients with severe SI at baseline (MADRS-SI ⩾4), SI (but not depression) was still present in 22 (2.4%) and 56 (7.9%) patients, whereas depression (but not SI) was still present in 178 (19.2%) and 93 (13.2%) patients in the LUEUR and GENESE cohorts, respectively.
SA during the follow-up
During the follow-up, 68 (1.9%) patients in the LUEUR cohort and 15 (0.6%) in the GENESE cohort reported SA. The risk of SA during the follow-up was higher for patients with severe SI. In the LUEUR cohort (not applicable in the GENESE cohort due to the too small effective of SA), the number of patients with SA during the follow-up was significantly higher in patients with severe SI than in the other two groups in unadjusted and adjusted models (OR = 3.17; 95% CI = [1.80, 5.57], p < 0.001 for the adjusted model, Table 8).
SA occurrence during the follow-up in the three groups (binary logistic regression).
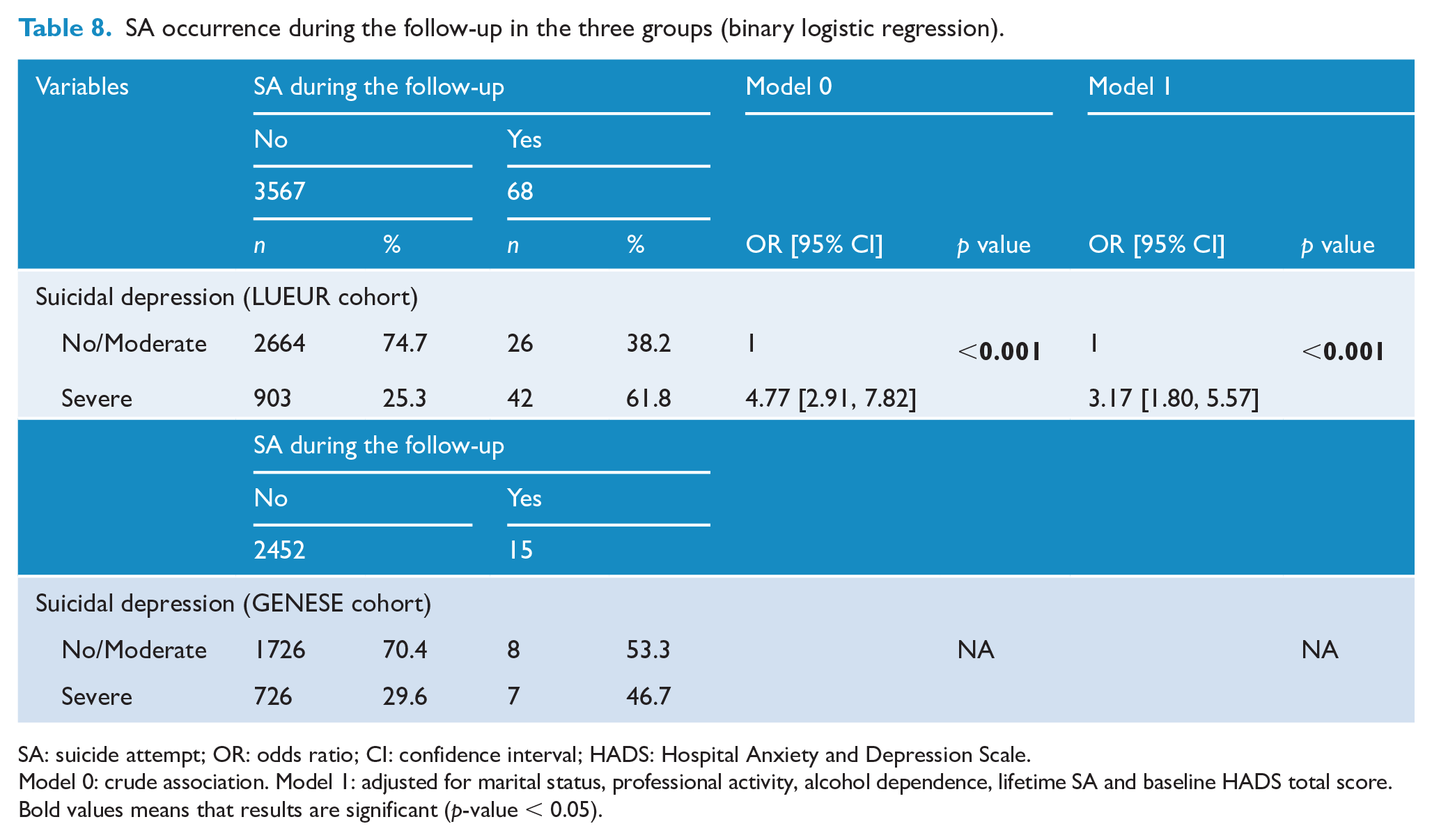
SA: suicide attempt; OR: odds ratio; CI: confidence interval; HADS: Hospital Anxiety and Depression Scale.
Model 0: crude association. Model 1: adjusted for marital status, professional activity, alcohol dependence, lifetime SA and baseline HADS total score.Bold values means that results are significant (p-value < 0.05).
Discussion
To our knowledge, this is the first study that characterized specifically suicidal depression in two large cohorts of depressed outpatients. Our results strengthen the hypothesis that suicidal depression could be a specific phenotype of depression, especially in the presence of severe SI. Indeed, baseline clinical features were more severe in patients with suicidal depression, and not only the severity of the current depressive episode. Interestingly, some of these features are considered to increase suicidal risk: greater number of lifetime MDEs (Trivedi et al., 2013), higher levels of anxiety and depression (Hoertel et al., 2018), hopelessness (Hawton et al., 2013), impulsivity (Coryell et al., 2018), more sleep disturbance (Bernert et al., 2015), more frequent history of SA (Hawton et al., 2013) and higher number of associated treatments and antidepressant treatment changes for the current MDE. These differences mostly persisted after adjusting for depression severity, suggesting that SI might help to define a more homogeneous type of depressive illness. Moreover, a gradient of SI severity was observed. Clinical features were more severe in depressed patients with moderate SI than in patients without SI, and in patients with severe SI than in patients with moderate SI.
Second, depression changes over time were different in the three groups of patients. Although depression severity decreased more in severe suicidal depressed patients than in the two other groups (significant difference of the slope with a higher slope for patients with severe SI, as shown by the GLS), this reduction was not sufficient to obtain depression remission at the end of the follow-up. Indeed, analysis of the depression changes during the 6 weeks of treatment showed that the number of responders to antidepressant treatment was lower. Remission at the end of the follow-up was less frequent in patients with severe SI than in the other two groups. Lower depression remission rate despite a greater decrease of depression severity was previously observed in a population of elderly depressed patients (Bogers et al. 2017). Despite the important decrease of depressive symptomatology in suicidal patients after antidepressant therapy initiation, these patients might have more persisting ‘residual’ symptoms. By assessing depression changes in different ways, our study strengthens previous results suggesting that in patients with depression at high risk of suicide (patients with current severe SI and/or lifetime SA), depression course is different than in non-suicidal patients and with a lower remission rate at the end of the follow-up (Lopez-Castroman et al., 2016; Pompili et al., 2010). This is not a real surprise when considering that patients with SI/SB are regularly excluded from clinical trials of antidepressants, and consequently, the real efficiency of antidepressants in this population remains unclear (Courtet and Nobile, 2020; Iltis et al., 2020).
The percentage of patients with SI during the follow-up remained higher in the group with severe SI, and the risk of SA during the follow-up was also threefold higher in this group. Moreover, SI remission (MADRS-SI score <2) was observed in 56.2% (LUEUR) and 47.6% (GENESE) of patients in the severe SI at week 6, and in 10% (GENESE) and 25% (LUEUR) at week 2 of treatment. Although not directly comparable, it is interesting to observe that with two new potential antisuicidal agents, SI decreased in about 60% of patients at day 1 with ketamine (Grunebaum et al., 2018) and in about 54% of patients at day 2 with esketamine (Canuso et al., 2018).
In our study, SI persistence was observed in 10% of patients with severe SI, as previously shown (Kasckow et al., 2016). Importantly, these patients were more at risk of SA during the follow-up than those without SI persistence. The best predictors of SI persistence were high baseline MADRS-SI score, lower decrease of HADS scores and sleep disturbances. Patients with severe SI who respond less to treatment could thus be at higher risk of SI persistence and SA. Therefore, it is primordial to strengthen the follow-up of patients with SI, sleep disturbances and no depression improvement after antidepressant therapy (McCall et al., 2019).
Furthermore, greater early SI decrease (during the first 2 weeks of treatment) and absence of lifetime history of SA were the two main predictors for SI remission in patients with severe SI at baseline in both cohorts. This suggests that the best predictors of SI remission are linked to the patients’ suicidal status rather than to their depression status. Therefore, SI decrease might be related not only to a decrease of depression severity. Indeed, depression changes explained about 25% of SI variation in the entire population and 45% when including only patients with severe SI. This result reinforces the hypothesis that patients with severe SI are different. Nevertheless, depression changes over time explained only less than 50% of SI variation, suggesting that other variables may interact with SI (i.e. sleep that explained almost 5% of SI variance). Only targeting depression is not sufficient to decrease SI as shown by SI persistence despite depression remission in some patients. Thus, depression improvement does not ensure SI resolution (Seo et al., 2014).
There are some discrepancies in results between both cohorts, mainly differences in remission of depression rates between patient’s groups according to studies. This may be explained by the two main differences in exclusion/inclusion criteria between cohorts. Indeed, patients on antipsychotics or mood stabilizers could be included in the GENESE, but not in the LUEUR cohort. It is known that antipsychotic drugs in combination with antidepressants and lithium can improve the efficacy of treatments for depression and SI in suicidal patients (Smith and Cipriani, 2017; Vermeulen et al., 2019). Therefore, the treatment might have influenced depression changes between cohorts. Moreover, alcohol abuse or dependence was an exclusion criterion in the GENESE but not in the LUEUR cohort; such comorbidity may impact the course of depression and SI.
Although specific treatments are lacking for suicidal patients, new promising drugs (such as ketamine or esketamine) are currently tested. Ketamine reduces SI in depressed patients (Grunebaum et al., 2018). Importantly, ketamine acts rapidly, unlike classical antidepressants, and reduces SI in at least half of the patients in the 24 hours following its administration (Ballard et al., 2014; Grunebaum et al., 2018). Therefore, it could be used as interventional treatment in patients with depression and severe SI to reduce the immediate risk of suicide and because antidepressants do not seem to affect SI in this group of patients. Development of specific psychotherapies to target SI is also justified in this context.
Our study has some limitations. First, a single item of the MADRS was used to define suicidal depression. However, self-report has been suggested to better catch SI than clinician reports (Czyz et al., 2016), and using a single item is a valid approach for SI evaluation (Desseilles et al., 2012). Second, the follow-up lasted only 6 weeks. However, this is the usual time needed to observe a response to antidepressant treatment. Furthermore, the interval between SI onset and SA is often very short and it is primordial to act on SI rapidly (Courtet et al., 2020; Glenn and Nock, 2014). Moreover, a longer follow-up could have increased the number of patients lost to follow-up.
Our study has some strengths. Suicidal depression was characterized in two large cohorts of outpatients with depression and with few individuals lost to follow-up (<10%). Almost all the results were replicated in the two cohorts (more than 2000 patients in each cohort), reinforcing their validity. Finally, these cohorts are representative of the population of depressed patients because they were recruited throughout France by general practitioners or psychiatrists and were outpatients. Interestingly, about 20% of patients included in the two cohorts met the inclusion criteria of clinical trials on new medications, leaving a large space for offering new therapeutic options to these patients.
Our results suggest that suicidal depression could be a specific phenotype of depression with more severe clinical characteristics, less frequent clinical remission of depression, SI persistence and higher risk of SA, despite antidepressant treatment. This suggests that SI may be a marker of severity of the depressive illness and of persistence of the suicide risk. Then, clinicians should implement personalized care with specific strategies and continuous monitoring of suicide risk in patients presenting with suicidal depression. This phenotype mainly concerns patients with severe SI because the characteristics of patients with moderate SI were closer to those of patients without SI. Our results contribute to the hypothesis that depression and SI are related but could also have independent physiopathology because remission of depression is not always associated with remission of SI. Moreover other factors might explain SI changes over time. Antidepressant drugs do not seem to be sufficient in patients with suicidal depression. These patients should be closely monitored. Combined treatments with antidepressants (pharmacological or non-pharmacological) and novel therapeutic strategies could be needed.
Supplemental Material
sj-docx-1-anp-10.1177_00048674211025697 – Supplemental material for Characteristics and treatment outcome of suicidal depression: Two large naturalistic cohorts of depressed outpatients
Supplemental material, sj-docx-1-anp-10.1177_00048674211025697 for Characteristics and treatment outcome of suicidal depression: Two large naturalistic cohorts of depressed outpatients by Bénédicte Nobile, Emilie Olié, Jonathan Dubois, Sebastien Guillaume, Philip Gorwood and Philippe Courtet in Australian & New Zealand Journal of Psychiatry
Footnotes
Author Contributions
B.N. performed statistical analyses and wrote the paper. J.D. contributed to the statistical analysis. P.G., S.G., E.O. and P.C. contributed to the data interpretation and to manuscript writing. All authors have contributed to the manuscript and have accepted the final version of the paper.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: The authors have interests in relation with one or more organization that could be perceived as a possible conflict of interest in the context of the subject of this manuscript. The relationships are summarized as follows. B.N., E.O. and J.D. report no financial relationships with commercial interests. S.G. received honoraria or research or educational conference grants from Bristol-Myers Squibb, Otsuka, Servier, Lundbeck, AstraZeneca and Janssen. P.G. reports no shares; has paid positions at University of Paris Descartes & Hospital Sainte-Anne; is on the advisory board at AstraZeneca, Janssen, Servier and Wyeth; and has no other involvement. P.C. reports no shares; has paid positions at University of Montpellier and CHU Montpellier; is on the advisory board at Janssen; and received honoraria or research or educational conference grants from Otsuka, Lundbeck, Exeltis, Pfizer and Janssen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a research grant from Servier that had no involvement in the study design, organization, analysis or preparation for publication.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author, B.N. The data are not publicly available due to restrictions (their containing information that could compromise the privacy of research participants).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
