Abstract
Objective:
Ketamine may reduce suicidal ideation in treatment-resistant depression. But it is not known how quickly this occurs and how long it persists. We undertook a systematic review and meta-analysis to determine the short- and long-term effectiveness of ketamine for suicidality.
Method:
CENTRAL, EMBASE, Medline, and PsycINFO were searched until 12 December 2018. Randomised controlled trials of ketamine or esketamine reporting data on suicidal ideation, self-harm, attempted or completed suicide in adults diagnosed with any psychiatric disorder were included. Two reviewers independently extracted data, and certainty of evidence was assessed using the Grading of Recommendations Assessment, Development and Evaluation tool. Standardised mean difference was used for continuous outcomes.
Results:
Twenty-five reports from 15 independent trials, with a total of 572 participants diagnosed with predominately affective disorders, were included. The evidence was rated moderate to low. In most trials, ketamine was administered at 0.5 mg/kg via a single intravenous infusion over a 30- to 45-minute period. Only a single trial of intranasal esketamine was identified. At 4 hours post-infusion, treatment with ketamine was associated with a significant reduction in suicidal ideation scores (standardised mean difference = −0.51, 95% confidence interval = [−1.00, −0.03]), which persisted until 72 hours post-infusion (time points between 12 and 24 hours: standardised mean difference = −0.63, 95% confidence interval = [−0.99, −0.26]; between 24 and 72 hours: standardised mean difference = −0.57, 95% confidence interval = [−0.99, −0.14]), but not thereafter. However, there was marked heterogeneity of results. In a single trial of esketamine, marginal effects on suicidal ideation were observed. In terms of actual suicidal behaviour, there were virtually no data on effects of ketamine or esketamine.
Conclusion:
A single infusion of ketamine may have a short-term (up to 72 hours) beneficial impact on suicidal thoughts. While confirmation of these results in further trials is needed, they suggest possible use of ketamine to treat acute suicidality. Means of sustaining any anti-suicidal effect need to be found.
Introduction
Suicidal ideation and self-harm are frequent and potentially life-threatening major public health problems, especially for people with severe mental health issues and mood disorders (Malhi et al., 2015). People presenting to hospital services for self-harm have a considerable risk of new episodes of self-harm (an average of 16.3% within a year in one review [Carroll et al., 2014], but well over 20% in other recent studies [Geulayov et al., 2016]) and also of completed suicide (1.6%) within a year (Carroll et al., 2014). Although fewer studies have focused on those presenting to clinical services for suicidal ideation only (i.e. without concurrent self-harm), patients hospitalised for clinically significant levels of suicidal ideation are also at increased risk of suicide (Hubers et al., 2018). Effective interventions in people presenting with suicidal ideation or self-harm are therefore a priority in order to contribute to efforts to reduce rates of self-harm and completed suicide.
Although there is currently no evidence that psychological treatments reduce suicide rates in self-harm patients (Crawford et al., 2007), brief cognitive-behavioural-based psychological therapy has been shown to be effective in reducing repetition of self-harm at 6- and 12-month follow-up (Hawton et al., 2016a, 2016b). However, psychological treatments for suicidal patients, although they are brief, require between 3 and 10 sessions (Hawton et al., 2016a), making them unsuitable for emergency treatment of suicidal patients. Currently, the only potential treatment available for acutely suicidal patients is electroconvulsive therapy (ECT) (Malhi et al., 2015), but there is little evidence to support this (Bergfeld et al., 2018). Otherwise treatment relies on close monitoring or hospitalisation (Griffiths et al., 2014).
Recent studies in both treatment-resistant unipolar and bipolar patients showed promising effects of ketamine in reducing depression scores (Caddy et al., 2015; McCloud et al., 2015). Although some reviews investigating the effectiveness of ketamine as an emergency treatment have been conducted, often they include only brief mention of the efficacy of ketamine in reducing suicidal ideation in these populations of patients without specific details (Caddy et al., 2015; McCloud et al., 2015; Naughton et al., 2014).
One small systematic review, including data from nine studies, showed that overall ketamine appeared to be associated with a reduction in suicidal ideation at 40 minutes with effects appearing to persist up to 10 days post-infusion (Reinstatler and Youssef, 2015). A second older review also found that ketamine may be associated with beneficial effects on suicidal ideation (Fond et al., 2014). However, as both of these reviews included studies of diverse designs, the authors did not calculate an overall pooled effect of ketamine on suicidal ideation. A further systematic review did not consider effects for ketamine beyond the 4-hour post-administration window (Bartoli et al., 2017b). A fourth systematic review (Xu et al., 2016) and an individual patient data meta-analysis (Wilkinson et al., 2018) both found that ketamine was associated with a reduction in suicidal ideation, but that the effect was less durable at the 7-day follow-up assessment.
Recently, there has been attention to intranasal treatment with esketamine for patients with treatment-resistant depression. This has recently received approval from the Food and Drug Administration (FDA) in the United States for use as an adjunctive treatment to antidepressant therapy (https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm632761.htm).
Because of the increasing emphasis on finding effective interventions to reduce suicides (Zalsman et al., 2016), and the publication of new trials designed a priori to include patients with clinically significant levels of suicidal ideation and using specific suicidal ideation rating scales, we undertook a systematic review to identify all studies reporting information on the effect of ketamine or esketamine on suicidality and conducted a meta-analysis for a number of time points between an immediate effect and up to more than 1 month.
Methods
The protocol of this systematic review was registered with PROSPERO (CRD42017070591).
Search strategy and inclusion/exclusion criteria
We searched CENTRAL, EMBASE, Medline, and PsycINFO until 12 December 2018 using the strategy outlined in Supplemental Table 1 in the Appendix. All published and unpublished randomised controlled trials (RCTs), comparing ketamine or esketamine as monotherapy or add-on treatment with placebo or active comparator in adult patients (i.e. 18 years and over) with a primary diagnosis of any major psychiatric disorder, were eligible for inclusion in this review. Cross-over trials were also included; however, to protect against the carry-over effect, only data for the period prior to cross-over were considered. Studies were excluded if the drugs under investigation were administered as an analgesic and/or anaesthetic agent, or data on suicidality were not available.
Data extraction
Abstracts and full texts were screened independently by two authors (K.W. and J.P.) and any disagreements were discussed with a third member of the team (A.C. and K.H.). Two reviewers (K.W. and J.P.) independently extracted data from included trials using a standardised form. We extracted outcome data at seven clinically relevant time points, as follows: (1)
Outcome measures
The primary outcome was the mean change in suicidal ideation as assessed by the Beck Scale for Suicidal Ideation (BSSI), the Columbia Suicide Severity Rating Scale (C-SSRS) or from the suicidal ideation item of a depression rating scale such as the Montgomery-Åsberg Depression Rating Scale (MADRS), Hamilton Depression Rating Scale (HDRS), Beck Depression Inventory (BDI, versions 1 or 2) or the (Quick) Inventory of Depressive Symptomatology (IDS/QIDS).
We also extracted data on the following secondary outcomes where these were reported in the included studies: (1) repeat self-harm episodes, (2) further episodes of attempted suicide, (3) suicide deaths and (4) deaths from any cause.
Statistical analysis
Where possible, we synthesised quantitative data between studies using meta-analysis (Witt et al., 2017). For continuous outcome data, we calculated the standardised mean difference (SMD) with corresponding 95% confidence intervals (CIs). For dichotomous outcome measures, we calculated the pooled odds ratio (OR) with corresponding 95% CI (Higgins et al., 2011). We used data from the intention-to-treat analyses for both continuous and dichotomous outcomes. The DerSimonian–Laird random-effects model, as implemented by RevMan version 5.3 (http://handbook-5-1.cochrane.org/), was used to synthesise both continuous and dichotomous outcome data.
We also planned, a priori, a series of subgroup and sensitivity analyses. For details of these, see the review protocol in PROSPERO (CRD42017070591).
Risk of bias assessment and the Grading of Recommendations Assessment, Development and Evaluation
The risk of bias of included studies was assessed by two reviewers independently (K.W., J.P.) using the Cochrane Risk of Bias Tool (https://methods.cochrane.org/bias/resources/cochrane-risk-bias-tool). Any disagreements were discussed and, if necessary, involved a third reviewer (K.H., A.C.). The GRADE tool was also used to assess the overall certainty of evidence (Schünemann et al., 2013). Further information is available in the Supplemental Appendix.
Role of the funding source
The funders had no role in study design, data collection, data analysis, data interpretation, or writing of the report. K.H., A.C., J.P. and K.W. had full access to all the data in this study and all authors had final responsibility for the decision to submit for publication.
Results
A total of 5547 potentially relevant records were obtained from the electronic search, while a further 30 were identified by hand search and personal correspondence; 3962 records remained after removing duplicates. Following a review of their titles and abstracts, a total of 3898 records were excluded, while a further 39 were excluded following a review of their full texts for the reasons as outlined in Figure 1.
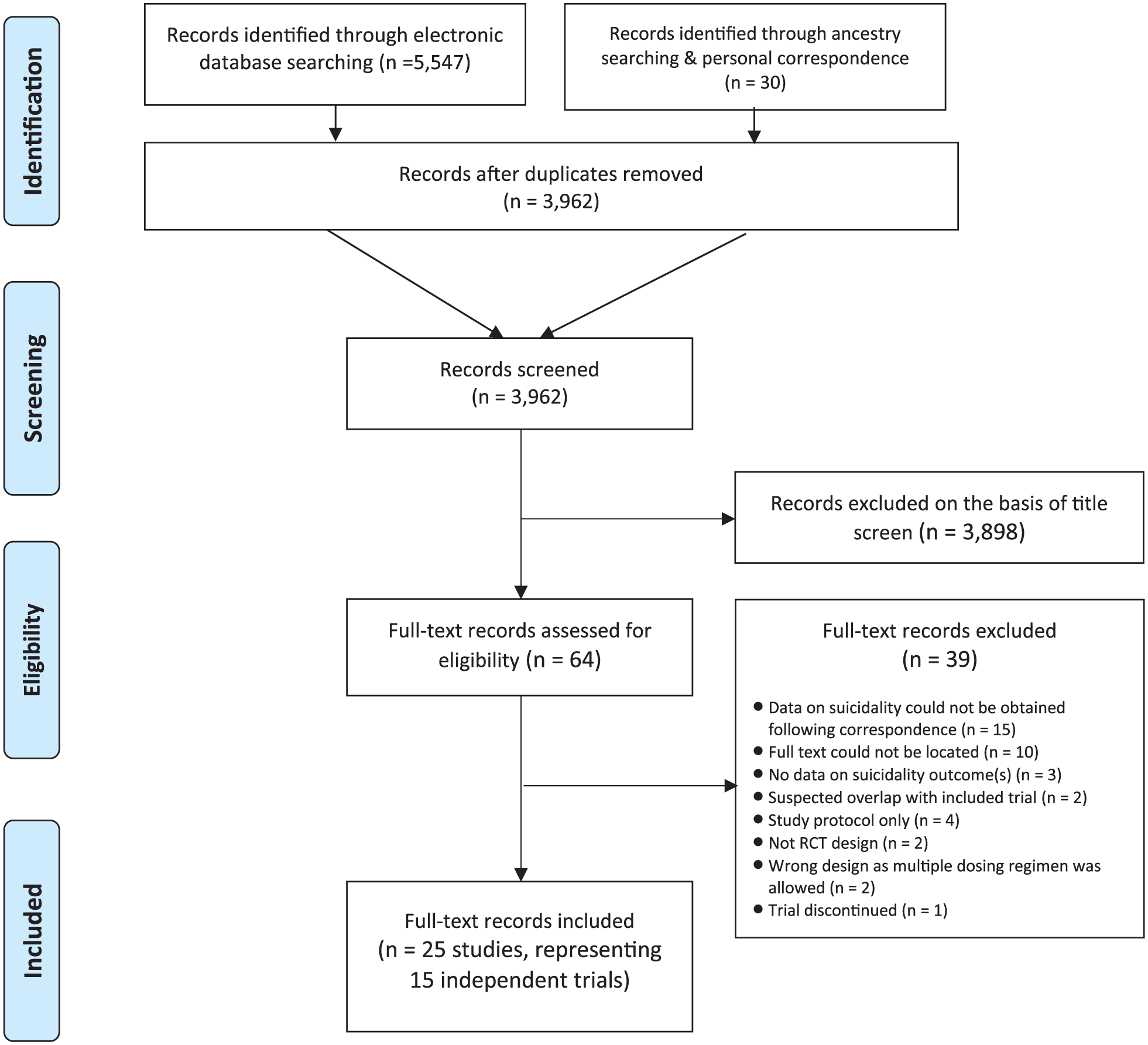
Selection of studies for inclusion.
Where necessary, we clarified methodological or data queries with study authors; however, not all replied (Burger et al., 2016). One further trial was excluded due to the study design as trial participants were randomised to one of two different dosing regimens (either two doses per week or three doses per week) (Singh et al., 2016). As participants therefore received multiple doses, and data could not be obtained prior to the second dose, we concluded that the results could not be pooled with the results of the other trials in a meta-analysis. One further trial identified as awaiting assessment at the time of the initial systematic search was subsequently published and we were therefore able to include this study from personal correspondence. This therefore left a total of 25 studies, representing 15 independent trials, eligible for inclusion in the review (see Table 1 for study characteristics and the Supplemental Appendix for full reference lista1–a15).
Study characteristics for all trials included in the review.
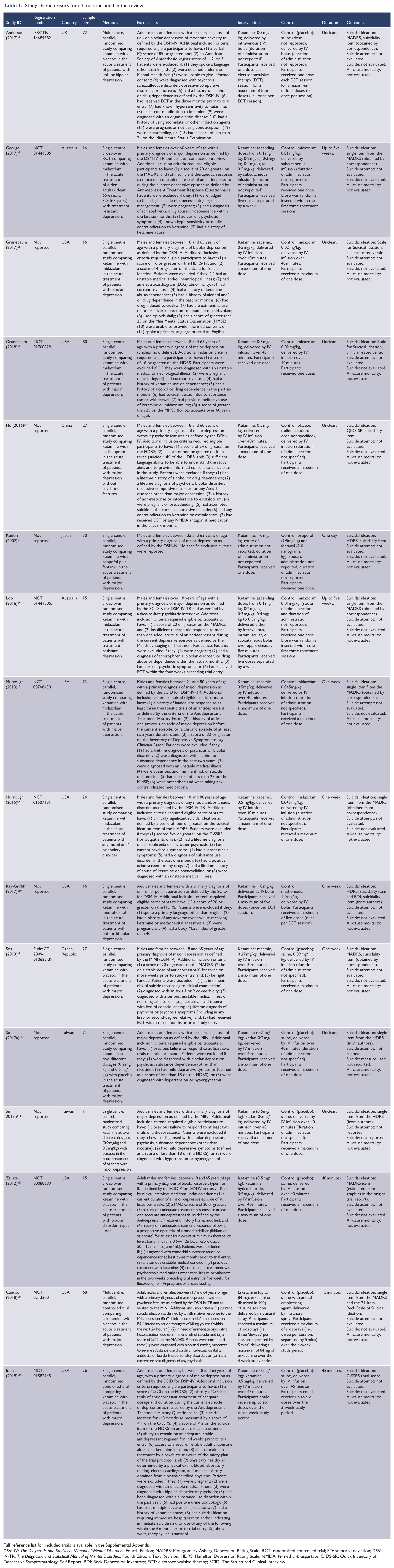
Full reference list for included trials is available in the Supplemental Appendix.
Overall, 572 adult participants (range: 15–80) between 37.7 and 65.6 years of age at randomisation (mean: 46.1 years, standard deviation [SD]: 11.2 years) were analysed. Most studies recruited participants with a diagnosis of unipolar major depression (10 studies),a2,a4–a8,a11,a12,a14,a15 followed by uni- or bipolar depression (2 studies),a1,a10 bipolar depression (2 studies)a3,a13 and any mood and/or anxiety disorder (1 study).a9 Of the 14 trials included, only six included patients with clinically significant levels of suicidal ideation.a3–a5,a9,a14,a15 In three other trials, patients at high risk of suicide were excluded from participation.a2,a8,a11
Over half of the sample was female in the 8 of the 13 trials (61.5%) that included information on gender. Scores on a measure of suicidal ideation at baseline (reported in nine trials) indicated moderate levels of suicidal thoughts. In the nine trials that reported information on suicidal behaviour, between 11.1% and 62.5% of the sample had a history of self-harm and/or attempted suicide prior to randomisation.
Interventions and comparisons
Most trials included in this review (14 trials) investigated the impact of ketamine on suicidal ideation. Ketamine was administered as a primary treatment for the acute relief of depression and/or suicidal ideation in all but three of these trials, where ketamine was used alongside ECT for the rapid relief of treatment-resistant depression.a1,a6,a10 In most trials, ketamine was administered at 0.5 mg/kg (eight trials;
In nine trials, including one in which ketamine was administered alongside ECT, ketamine was administered on one occasion only,a3–a6,a8,a9,a11–a13 while in two further trials in which ketamine was used alongside ECT, participants could receive up to five infusions of ketamine (i.e. once per ECT session).a1,a10 In the remaining four trials, participants could receive multiple doses across the intervention period.a2,a7,a14,a15 To protect against potential effects of repeated doses, for those trials in which the drug was administered on more than one occasion, only data prior to the second dose were extracted and included in any meta-analysis.
Ketamine was administered intravenously in all but three trials, where it was either administered subcutaneously,a2 or where intravenous, intramuscular and subcutaneous routes of administration were possible.a7 For most studies (11 trials), ketamine was administered via an infusion over a 30- to 45-minute period. For two trials, ketamine was infused via bolus,a1,a10 and for one trial the duration of infusion was not clearly reported.a6 In a single trial of esketamine, only the intranasal route of administration was used.a14
The majority of included studies compared ketamine with another drug (nine trials), most commonly midazolam as active placebo,a2–a4,a7–a9 or propofol and fentanyla6 and methohexitala10 as active comparators. The remainder compared the effects of ketamine with a placebo control (i.e. saline solution).a1,a5,a11–a15
Effect of ketamine on suicidal ideation scores
By the immediate effect time point (i.e. within 4 hours), ketamine was associated with a significant reduction in suicidal ideation scores (SMD = −0.51, 95% CI = [−1.00, −0.03], 10 studies,
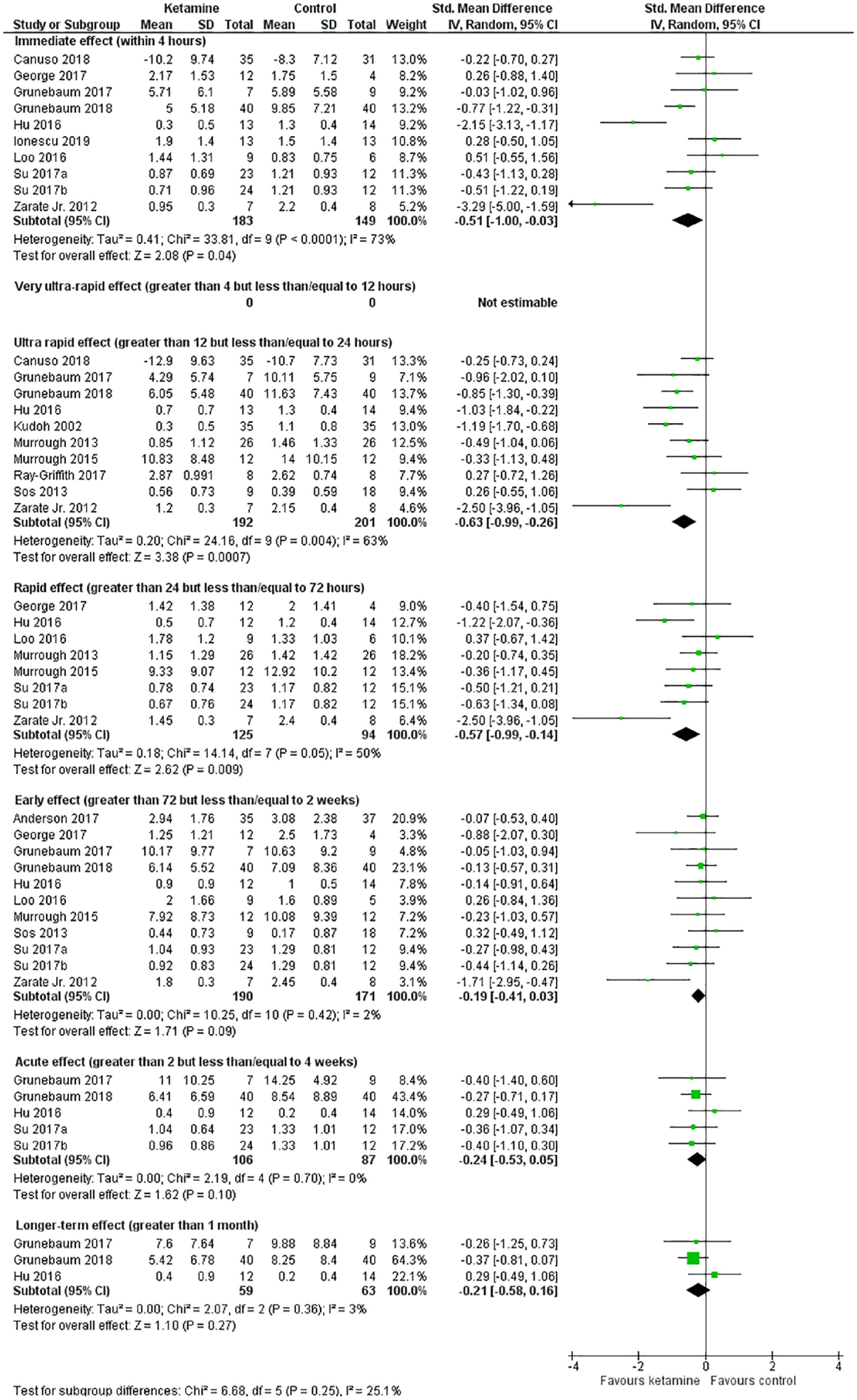
Suicidal ideation scores across all time points.
By the early effect time point (i.e. >72 hours but ⩽2 weeks), ketamine was no longer associated with a significant treatment effect for suicidal ideation (Figure 2).
Effect of ketamine on self-harm, attempted suicide, suicide and all-cause mortality
One study of two different doses of ketamine (i.e. 0.2 vs 0.5 mg/kg) found that no participant in either the ketamine or control group engaged in a further episode of self-harm in either dosing group over the course of the follow-up period (up to 28 days).a12
This same study also reported data on the proportion with a suicide attempt by the longest time point (up to 28 days); no participant attempted suicide in either the ketamine or control arms in this trial.a12 There were also no suicide deaths in this trial.a12 No included trial reported data on all-cause mortality.
Risk of bias and GRADE
An overall summary of the risk of bias for all 15 trials is provided in Supplemental Figures 1 and 2 in the Appendix alongside an assessment of overall outcome quality. Overall, the certainty of the evidence was low to moderate (Supplemental Table 2 in the Appendix).
Subgroup and sensitivity analyses
Results from the sensitivity analyses excluding those studies in which a full-scale measure of suicidal ideation, as opposed to a single item measure, was used did not show material differences in terms of the observed effect of ketamine at any time point (Supplemental Figure 3 in the Appendix). We also added one further (post hoc) subgroup analysis. Specifically, given recent findings that ketamine may accelerate the effect of ECT in patients with treatment-resistant depression (Jankauskas et al., 2018), we excluded those studies in which ketamine was administered as part of ECT treatment.a1,a6,a10 Excluding these trials did not materially affect either the direction or magnitude of effect for ketamine for any time point. Given their influence on the results, we also conducted a further post hoc sensitivity analysis excluding the Ionescu and Zarate trials which showed the strongest effects in favour of ketamine,a13,a15 but found no material difference in either the direction or magnitude of the effect for ketamine for any time point.
Small study effects
The sample sizes of some of the included trials were relatively small, with eight trials (53.3%) including under 30 participants. We therefore performed Egger’s test to examine the impact of small study effects on the results. There was no evidence of small study effects for the immediate (i.e. within 4 hours;
Discussion
This review, combining data from 15 independent RCTs involving 572 participants, shows evidence of effects in favour of a single treatment of ketamine for suicidal ideation in adult psychiatric patients predominately diagnosed with mood disorders, and principally unipolar major depression. These effects were apparent up to 72 hours post-infusion, in line with some previous work (Wilkinson et al., 2018; Xu et al., 2016), although considerable heterogeneity was associated with the estimates. The results suggest that ketamine can potentially be used in the emergency treatment of acute suicidal ideation. It is notable that effects in favour of ketamine for suicidal ideation would appear to be more time limited than its effects on depression more broadly, as recent reviews have indicated positive effects of ketamine on depression scores up to 1 week post-infusion (Caddy et al., 2015; Xu et al., 2016).
These results are broadly in line with those of a recent individual participant data (IPD) meta-analysis in which ketamine was found to be associated with significant reductions in suicidal ideation in patients who were predominately diagnosed with major depression, although most of the eight trials analysed did not specifically target patients with clinically significant suicidal ideation (Wilkinson et al., 2018). However, we included 15 trials and doubled the number of participants (
In terms of diagnosis, while we included studies of participants diagnosed with any psychiatric disorder, the diagnoses of participants were almost always mood disorders. This raises the issue of whether ketamine might have benefits for suicidality in patients with non-affective disorders, especially those with borderline personality disorder, in whom repeated self-harm is a common feature (Hawton et al., 2016b). The average levels of suicidal ideation at baseline in our review were moderate in those trials in which it was reported. However, in the nine trials with information about actual suicidal acts, substantial proportions of the participants had a history of self-harm and/or attempted suicide before trial entry, suggesting that they constituted patients at considerable risk. There is some evidence suggesting that the anti-suicidal effect of ketamine is independent from the antidepressant effect (Sanacora et al., 2017; Zalsman et al., 2016), but larger studies including a variety of disorders are needed before we can draw clinically meaningful conclusions.
A number of the analyses included in this review were associated with high levels of heterogeneity owing to differences in dosing and route of administration. Ketamine dosing in the trials included in this review ranged from 0.27 to 1.0 mg/kg, although we would note that higher doses were not necessarily associated with a superior effect of ketamine. In terms of route of administration, in most of the trials included in the review, ketamine was administered intravenously. Although we identified three recently published trials investigating the effectiveness of esketamine delivered via intranasal administration as an adjunct to oral antidepressant therapy, two of which suggested a significant treatment effect in favour of esketamine for symptoms of depression at up to 2 months post-treatment (Daly et al., 2018),a14 we were able to include data from only one of these trials,a14 finding only a modest and non-significant effect at the immediate time point (i.e. up to 4 hours) in reducing suicidal ideation (and suicide risk as defined by a score of between 0 and 1 on the clinician-administered Suicide Ideation and Behaviour Assessment Tool).a14 We were unable to obtain data on suicidal ideation to include the second of these trials in the present review (Daly et al., 2018), and the third trial was discontinued prematurely due to tolerability problems (Gálvez et al., 2018). Absorption via the nasal mucosa was variable, leading to high peak serum levels of esketamine in some participants. Notably, due to adverse effects, no participant was able to achieve the necessary 10 pump actions required to administer the full esketamine dose in the treatment protocol. There is therefore a need to evaluate both the safety and effectiveness of esketamine post-market before recommendations can be made as to both the safety and effectiveness of esketamine as an emergency treatment for suicidality. While giving ketamine intravenously to suicidal patients in an accident and emergency department complicates the logistics of the administration of the intervention, it can guarantee that the drug is properly delivered. Other routes of delivery (such as intramuscular) also require further research.
In terms of mechanisms of the effect of ketamine on suicidality, the most obvious is through its impact on depression, although, as noted above, the two may not be completely correlated (Wilkinson et al., 2018), and the treatment effect for depression appears to last longer than that for suicidality (Caddy et al., 2015; McCloud et al., 2015), although it is still relatively short term (Ibrahim et al., 2012). Other potential mechanisms of the anti-suicidal ideation effect of ketamine includes amelioration of symptom clusters associated with reduction of suicidal thoughts such as subjective symptoms of depression (e.g. sadness, pessimism, loss of interest) (Keilp et al., 2018) or improvement in neurocognitive domains such as memory (Gorlyn et al., 2015). These potential mediators require further research.
Limitations
The main limitation in our review is related to an intrinsic limitation in the available information from included studies. The assessment of suicidal ideation mainly relied on use of a single item on a depression rating scale, and the scales used varied across studies. While there is some evidence of excellent correlation (around
A second limitation relates to the choice of the control group in the majority of included studies. The trials either used another anaesthetic agent (most commonly midazolam) or placebo, which are not effective in the treatment of depression or suicidality. Therefore, it may be that the effect of ketamine observed here could be masked if future trials were to compare the effectiveness of ketamine against other active antidepressant agents. In addition, ethical considerations relating to the off-label use of ketamine, including questions around the representativeness of participants included in these trials to date, and potential long-term side effects of ketamine use, including the potential for rapid rebound of suicidal ideation after ketamine treatment, require further consideration before ketamine can be recommended for its anti-suicidal potential (Singh et al., 2017).
A further limitation is that in the included trials there was no reporting of potential adverse effects (e.g. dissociation, blurred vision, dizziness, malaise, nausea or possible emergence psychosis) beyond the few hours after treatment, even in studies where ketamine was administered at relatively high doses (e.g. 1.0 mg/kg). This problem was also highlighted in a recent review, which found limited reporting of safety or tolerability outcomes in trials of ketamine for the treatment of depression (Short et al., 2018). This is a key point also considering that previous studies have shown that ketamine effects on depression might be mediated by, or related to, both dissociative (Luckenbaugh et al., 2014) and psychotomimetic (Sos et al., 2013) effects. The need for strategies to mitigate adverse effects of ketamine has recently been highlighted (Bartoli et al., 2017a; Cooper et al., 2017). This is an issue that requires careful consideration in future trials, especially when ketamine is used as an off-label treatment (Sanacora et al., 2017).
We also had to exclude some studies either because they did not report information on suicidality (e.g. Abdallah et al., 2012; Alizadeh et al., 2015; Berman et al., 2000; Daly et al., 2018; Feder et al., 2014; Gálvez et al., 2017; Lai et al., 2014; Lapidus et al., 2014; Li et al., 2016; Ochs-Ross et al., 2019; Phillips et al., 2017; Popova et al., 2018; Rodriguez et al., 2013; Yoosefi et al., 2014; Zhong et al., 2016) or because they were secondary publications of studies we had already included in the review (e.g. DiazGranados et al., 2010; Lally et al., 2014; Zarate et al., 2006). Inconsistent reporting of clinical trials numbers in these secondary publications complicates the identification of duplicated studies in this field. In addition, for studies in which information on suicidal ideation was not reported, although we attempted to contact corresponding authors on three occasions to obtain these data, we received responses from few. These problems highlight the need for greater transparency in trials of ketamine or esketamine on adverse outcomes, and particularly those relating to suicidal ideation and behaviour. Stricter adherence and enforcement of CONSORT (Consolidated Standards Of Reporting Trials) reporting guidelines in these trials would represent an important first step to help overcome these problems.
Sample sizes of the included trials were also relatively small, with eight trials including under 30 participants. This contributed to heterogeneity and uncertainty of the estimated effects in individual trials, as there were often too few participants to detect clinically significant effects of ketamine, particularly for rarer outcomes such as suicidal behaviour.
Finally, while we had planned, a priori, secondary analyses relating to possible differential effect in males compared with females and by suicidal ideation severity at baseline, the trials included in our analysis did not report disaggregated data by these factors so we could not carry out these subgroup/sensitivity analyses.
Clinical implications
The results of this review suggest that ketamine may have a role in acute treatment for suicidality. However, there is clearly a need for clinical measures to ensure persistence of any benefits. One possibility is combining some sort of acute psychosocial intervention, such as brief psychological therapy with ketamine treatment. Adaptations of psychosocial treatments which have been shown to have impacts on suicidality, for example, cognitive-behavioural therapy (CBT) approaches (Hawton et al., 2016b), could provide a basis for the development of acute therapy in this setting. Another is the use of multiple treatments with ketamine, which in practice is common (Caddy et al., 2015), and yet most studies included in the current review evaluated the effects of ketamine as a single treatment. These possible approaches to maximising the potential benefits of ketamine in the clinical management of people with suicidal ideation are well worth pursuing in order to determine the place of ketamine in contributing to current efforts to reduce suicide.
Conclusion
The results of this review suggest that a single dose of ketamine (0.5 mg/kg) may have short-term benefits in the acute treatment of suicidal ideas, but that ways, including possible psychosocial interventions, need to be found to maintain these effects. There is a clear need for more research focused on the effects of ketamine in suicidal patients, including in those with psychiatric disorders other than affective disorders. In such research, there needs to be close monitoring of suicidal ideation, perhaps using digital and frequently repeated recording of patient’s thoughts (e.g. ecological momentary assessment). There is also a need to closely monitor side effects of the medication to ensure patient safety, including beyond the immediate post-treatment period. We conclude that currently there is considerable uncertainty about the use of ketamine specifically as a treatment for suicidal ideas, but that current results of trials suggest that this drug may have a potential place in the clinical care of suicidal patients.
Supplemental Material
Online_Appendix – Supplemental material for Ketamine for suicidal ideation in adults with psychiatric disorders: A systematic review and meta-analysis of treatment trials
Supplemental material, Online_Appendix for Ketamine for suicidal ideation in adults with psychiatric disorders: A systematic review and meta-analysis of treatment trials by Katrina Witt, Jennifer Potts, Anna Hubers, Michael F Grunebaum, James W Murrough, Colleen Loo, Andrea Cipriani and Keith Hawton in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors wish to thank the following for providing raw data on suicidal ideation in relation to their trials: Ian Anderson, Shona Ray-Griffith, Peter Sos and Tung-Ping Su. We also wish to thank Toshi Furukawa for assistance with data extraction.
Declaration of Conflicting Interests
The following authors were involved in trials included in this review: M.F.G., J.W.M. and C.L. All other authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.C. is supported by the National Institute for Health Research (NIHR) Oxford Cognitive Health Clinical Research Facility in his role as NIHR Research Professor, by the NIHR Research Professorship (RP-2017-08-ST2-006) and by the NIHR Oxford Health Biomedical Research Centre (BRC-1215-20005). Personal funding from the NIHR was awarded to K.H. in his role as an Emeritus NIHR Senior Investigator. The views expressed are those of the authors and not necessarily those of the UK National Health Service, the NIHR or the UK Department of Health.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
