Abstract
Objective:
Drug-induced delirium has been attributed to opioid, benzodiazepine, antipsychotic, antihistaminic and anticholinergic drug groups at therapeutic doses. Delirium also occurs in hospital-treated self-poisoning (at supra-therapeutic doses), although the causative drug classes are not well established and co-ingestion is common. We tested the magnitude and direction of association of five major drug groups with incident cases of delirium.
Methods:
A retrospective longitudinal cohort (n = 5131) study was undertaken of deliberate and recreational/chronic misuse poisoning cases from a regional sentinel toxicology unit. We described ingestion and co-ingestion patterns and estimated the unadjusted and adjusted odds for developing a drug-induced delirium. We also estimated the odds of drug-induced delirium being associated with three outcomes: intensive care unit admission, general hospital length of stay and discharge to home.
Results:
Drug-induced delirium occurred in 3.9% of cases (n = 200). The unadjusted odds ratios for development of delirium were increased for anticholinergics 10.79 (5.43–21.48), antihistamines 6.10 (4.20–8.84) and antipsychotics 2.99 (2.20–4.06); non-significant for opioids 1.31 (95% confidence interval = [0.81, 2.13]); and reduced for benzodiazepines 0.37 (0.24–0.58); with little change after adjustment for age, gender and co-ingestion. Delirium was associated with intensive care unit admission, longer length of stay and discharge destination.
Conclusion:
Drug-induced delirium was uncommon in this population. Co-ingestion was common but did not alter the risk. In contrast to drug-induced delirium at therapeutic doses in older populations, opioids were not associated with delirium and benzodiazepines were protective. Drug-induced delirium required increased clinical services. Clinical services should be funded and prepared to provide additional supportive care for these deliriogenic drug group ingestions.
Introduction
Delirium is an important and common clinical entity in general hospital patients and has been associated with poorer outcomes independent of age, severity of illness or dementia (Khan et al., 2012; Siddiqi et al., 2006). Medications and other drugs may be the sole cause of delirium or part of a multifactorial aetiology; drug-induced delirium can occur in association with many different drug classes (Carter et al., 1996), whether at therapeutic or supra-therapeutic doses or during periods of withdrawal or discontinuation.
Most reports of drug-induced delirium are in therapeutic dosing and there are obvious ethical restrictions involved with recruiting volunteers to be exposed to supra-therapeutic doses of agents thought to cause adverse effects. Opioid analgesics, benzodiazepines and corticosteroids are often suggested as contributors to delirium; however, the association between psychoactive drugs and delirium cannot be confirmed with current epidemiological evidence (Gaudreau et al., 2005). A systematic review of therapeutic dosing found 14 prospective studies of delirium demonstrating increased risk with opioids, benzodiazepines, dihydropyridines and antihistamines, with no increased risk for neuroleptics or digoxin and uncertainty for histamine (H2) antagonists, tricyclic antidepressants, antiparkinson medications, corticosteroids, non-steroidal anti-inflammatory drugs and antimuscarinics (Clegg and Young, 2011). Another review suggested increased risk only for psychoactive medications (as a single variable) and opioids (Gaudreau et al., 2005).
Self-poisoning is common among general hospital presentations and deliberate self-poisoning (DSP) is the most common variant of all self-poisoning types in adults (Hiles et al., 2015; Whyte et al., 1997). Self-poisoning episodes may involve doses of medication ranging from sub-therapeutic to supra-therapeutic and sometimes toxins and substances never intended for ingestion, while the variety of agents used in self-poisoning often reflect prescribing patterns for patients being treated for mental health disorders as well as agents available over the counter (Buckley et al., 2015). Co-ingestion of more than one drug or medication in self-poisoning is common (mean co-ingestion of 1.99 agents; range 1–18, per admission) (Buckley et al., 2015). Specific drugs in overdose have been associated with delirium including olanzapine (Morgan et al., 2007), promethazine (Page et al., 2008) and baclofen (Leung et al., 2006). Other less obvious cognitive impairments have also been associated with sedative drug overdose (Dassanayake et al., 2012), an effect that may be prolonged long after recovery from the episode of DSP (Oxley et al., 2015).
However, clinical scenarios often include a complex mix of emotional distress, mental illness and co-ingestion of multiple drug classes, at a variety of doses, sometimes of unknown medications, together with co-ingestion of alcohol; all in the context of external stressful social circumstances and, especially in older patients, physical illness. In this complex clinical setting, an improved knowledge of which medications, or at least drug classes, are most likely to cause delirium might directly benefit clinical management in the emergency department (ED) and during the general hospital stay and indirectly influence drug prescribing practices in the community to reduce exposures for patients at risk of self-poisoning.
In addition to the uncertainty about which medications are more likely to cause delirium in self-poisoning patients, there might be important confounders to drug exposure associations with delirium. These include dose taken; co-ingestion of other agents; co-administration of usual prescribed agents; and age, gender and indication for use. Cross-sectional, case series and cohort designs using cases derived from clinical case registers of self-poisoning patients are a useful way to examine these associations.
It is not known whether the poor outcomes associated with delirium in general hospital patients are also true for drug-induced delirium in DSP patients. Hospital-treated self-poisoning is associated with substantial rates of intensive care treatment, and discharge to the psychiatric hospital for after-care (Buckley et al., 2015; Carter et al., 2006). For service provision requirements, it would also be useful to know the most relevant short-term outcomes associated with drug-induced delirium: intensive care unit (ICU) admission, length of stay in the general hospital and discharge destination.
Aims
This study has four aims:
To describe the clinical characteristics associated with delirium in a population of hospital-treated self-poisonings;
To determine which major drug classes ingested in an episode of hospital-treated self-poisoning are associated with delirium;
To determine which drug classes, ingested in an episode of hospital-treated self-poisoning, are independently associated with delirium after adjustment for co-ingestion and other confounding;
To determine whether delirium in self-poisoning patients is associated with clinically relevant outcomes: ICU admission, length of stay in the general hospital or discharge destination.
Methods
Setting and study design
Newcastle, 160 km north of Sydney on the East coast of New South Wales, is the seventh largest city in Australia and the second most populated area in the State. The surrounding Hunter region also includes the local government areas of Lake Macquarie, Cessnock, Maitland, Port Stephens, Muswellbrook, Dungog, Singleton and Upper Hunter.
The Hunter Area Toxicology Service (HATS) is a regional toxicology service situated at the Newcastle Calvary Mater that serves a primary referral urban population of over 410,000 (Newcastle, Lake Macquarie and Port Stephens) and is also a tertiary referral centre for a further rural population of over 243,000 (Lower and Upper Hunter Valley). All poisoning presentations in the primary referral area are directed to the Calvary Mater Newcastle ED and admitted to the clinical toxicology unit (or notified if admitted to another hospital) under the care of the attending clinical toxicologist (Buckley et al., 2015; Whyte et al., 1997). Detailed clinical information is recorded in the HATS database, a clinical case register for all poisonings. A preformatted admission sheet is used by medical and psychiatry staff to collect clinical and demographic data at admission (Buckley et al., 1999). All cases are discussed at a weekly review and quality assurance meeting attended by toxicology and psychiatry staff for review of diagnoses, clinical management and discharge destination. Data from the preformatted admission sheet, additional clinical data from the record during the admission, discharge destination and any relevant decision from the weekly review meeting are entered into the case register database by trained staff blinded to any study hypotheses.
For this study, we used a retrospective longitudinal cohort design. The inception rule for the cohort was any index episode of admission by an adult for deliberate of recreational/chronic misuse self-poisoning, during the study period, with follow-up to the end of each admission.
Participants
The database of the HATS case register was used to extract prospectively collected information for all self-poisoning presentations aged 16 years or older, during the 10-year period from 2004 to 2013. Only a patient’s first admission in the period (index admission) was included in the analysis, to remove the potential for bias brought about by lack of independence of observations. We included any deliberate and recreational/chronic misuse poisonings and excluded the iatrogenic, accidental and occupational poisonings (Whyte et al., 1997). We included alcohol as a co-ingested substance, as simple alcohol ingestion cases are not admitted under toxicology. Any recording of delirium was selected as an outcome; except where delirium was determined by a clinician to be caused by withdrawal from a substance, or was clearly present while the patient was in the community in the period before the self-poisoning ingestion.
Participant characteristics
We reported demographic variables (sex, age group and marital status), current Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) mental illness diagnoses and current substance use. The DSM-IV was the DSM version in use from 1994 to 2013, which spanned the period of the study. Individual mental illness diagnoses were grouped according to Major Diagnostic categories for DSM-IV: Any Anxiety, Mood, Personality, Substance use, Psychotic, or Adjustment disorder, any Relational problems (from V-codes) and any Dementia diagnosis of any sub-type, and any other diagnosis outside of these classifications coded as ‘Other’ disorder. Current drug use (not necessarily a substance use disorder) was identified by the treating clinician in the course of clinical assessment and coded for the following classifications: any alcohol, benzodiazepines, amphetamines, opioids, cocaine, cannabis, hallucinogen, barbiturates, volatile hydrocarbons, antihistamines, nicotine, other drug use and any intravenous (IV) drug use.
Independent variables: drugs or medications ingested in DSP event
We examined each individual agent ingested by classification into major pharmacological groups according to their pharmacological class (see Supplementary Table 1 for the classification of individual drugs and medications). When there was some overlap of possible classification, medications were allocated to the group with which their clinical effects are most commonly associated. For example, mood stabilisers from the anticonvulsant class (e.g. sodium valproate) were included in the anticonvulsant group, and antipsychotics (e.g. quetiapine) were included in the antipsychotic group even when they had prominent anticholinergic properties. The ingested major drug groups were opioids, non-opioid analgesics, antipsychotics, antidepressants, benzodiazepines (and other sedative-hypnotics), lithium, anticholinergics, antihistamines, anticonvulsants, cardiac (and antihypertensive) medications, other medical drugs, stimulants and hallucinogens. Alcohol was not considered as a single ingestion but was included to capture co-ingestion with any of the above groups. Any individual drug was only classified into one group, and multiple ingestions of the same type could be coded into the same group (e.g. quetiapine and olanzapine into antipsychotics) or into different groups (e.g. paracetamol into non-opioid analgesics and paracetamol plus codeine preparations into opioids).
Dependent variables
The primary outcome was any clinical diagnosis of delirium during the admission for DSP. For the purposes of the study, delirium was considered to be present when the diagnosis was documented in the patient’s record at any time or identified at the weekly quality assurance review meeting. Any clinical diagnosis of a withdrawal delirium and any delirium diagnosis preceding the ingestion were excluded. Any participant who simply had an impaired level of consciousness associated with the ingestion of a sedative drug was not considered to have a delirium, unless other signs consistent with a delirium were evident after the sedation had resolved.
Secondary outcomes were any ICU admission, length of stay in the general hospital and discharge destination following the poisoning episode of: discharge home vs discharge to institutional care. Institutional care was a composite endpoint which included transfer to psychiatric hospital, transfer to another service in the general hospital and discharge to ‘other’ destination, which included Nursing Home, Gaol and Police custody.
Statistical analyses
Participant characteristics, categorised by delirium outcome, were examined using descriptive statistics. The association of exposure to ingestion by a priori selected major drug groups as a predictor of delirium was examined using a series of univariate logistic regression models, with results expressed as odds ratios (ORs) with 95% confidence interval (95% CI). We specifically focused on drug exposures implicated as causes of delirium in overdose and in hospitalised patients: opioids, antipsychotics (Burns, 2001), benzodiazepines (Clegg and Young, 2011), antihistamines (Chu, 2011; Page et al., 2008; Radovanovic et al., 2000) and anticholinergics (Burns et al., 2000). We did not select antidepressants because the evidence suggesting a possible relationship was for tricyclic antidepressants (Clegg and Young, 2011), which were commonly ingested in previous decades, but are not commonly ingested in recent times (Buckley et al., 2015).
We then undertook a series of adjusted models. We report the adjusted odds ratio (AOR) with 95% CI for each of the five major drug group exposures as a predictor of delirium, sequentially adjusting for gender, age and co-ingestion by selected major drug groups (opioids, benzodiazepines, antipsychotics, antihistamines, anticholinergics and alcohol). Given the patterns of co-ingestion, we undertook initial examinations of possible collinearity among independent (predictor) and adjusting (potential confounding) variables. No collinearity issues were identified.
For the secondary outcomes of the study, we estimated the association of delirium as the independent variable predicting two categorical outcomes: ICU admission and discharge destination of home vs institutional care using unadjusted logistic regression models. For the continuous outcome of general hospital length of stay (in hours), we expected a non-normal distribution, differential by subgroup, and so we also reported the median, interquartile range (IQR) and a median test with delirium status as the predictor.
We used IBM SPSS Statistics software (2017; IBM SPSS Statistics for Windows, Version 25.0., Armonk, NY: IBM Corp) for all analyses.
Ethics approval
The HATS database and clinical case register has approval for use in quality assurance projects granted by the Hunter New England Human Research Ethics Committee (HNEHREC). This specific study was undertaken in part as a student project for the RANZCP advanced trainee scholarly project and so specific ethics approval was also secured (HNEHREC Reference No: 16/04/20/5.14).
Results
Initially, 8305 episodes were identified, with 2411 excluded for being second and subsequent presentations, 693 excluded because of poisoning type (iatrogenic, accidental or occupational), 6 excluded as cases of withdrawal delirium and 64 cases excluded for data issues (e.g. duplication, missing data), leaving 5131 cases for the analysis.
Participant characteristics
As detailed in Table 1, DSP accounted for 93% (n = 4774) and recreational/chronic misuse accounted for 7% (n = 357) of the population. The female-to-male ratio was 3:2, with a predominantly young population, 75% being aged 16–44 years and mostly single. The most common DSM groups were relationship problems 44%, substance use disorder 42% and mood disorder 39%. Current drug use was most commonly nicotine at 24%, with alcohol 22%, cannabis 6%, amphetamines 4% and benzodiazepines 4%.
Participant characteristics and outcomes.
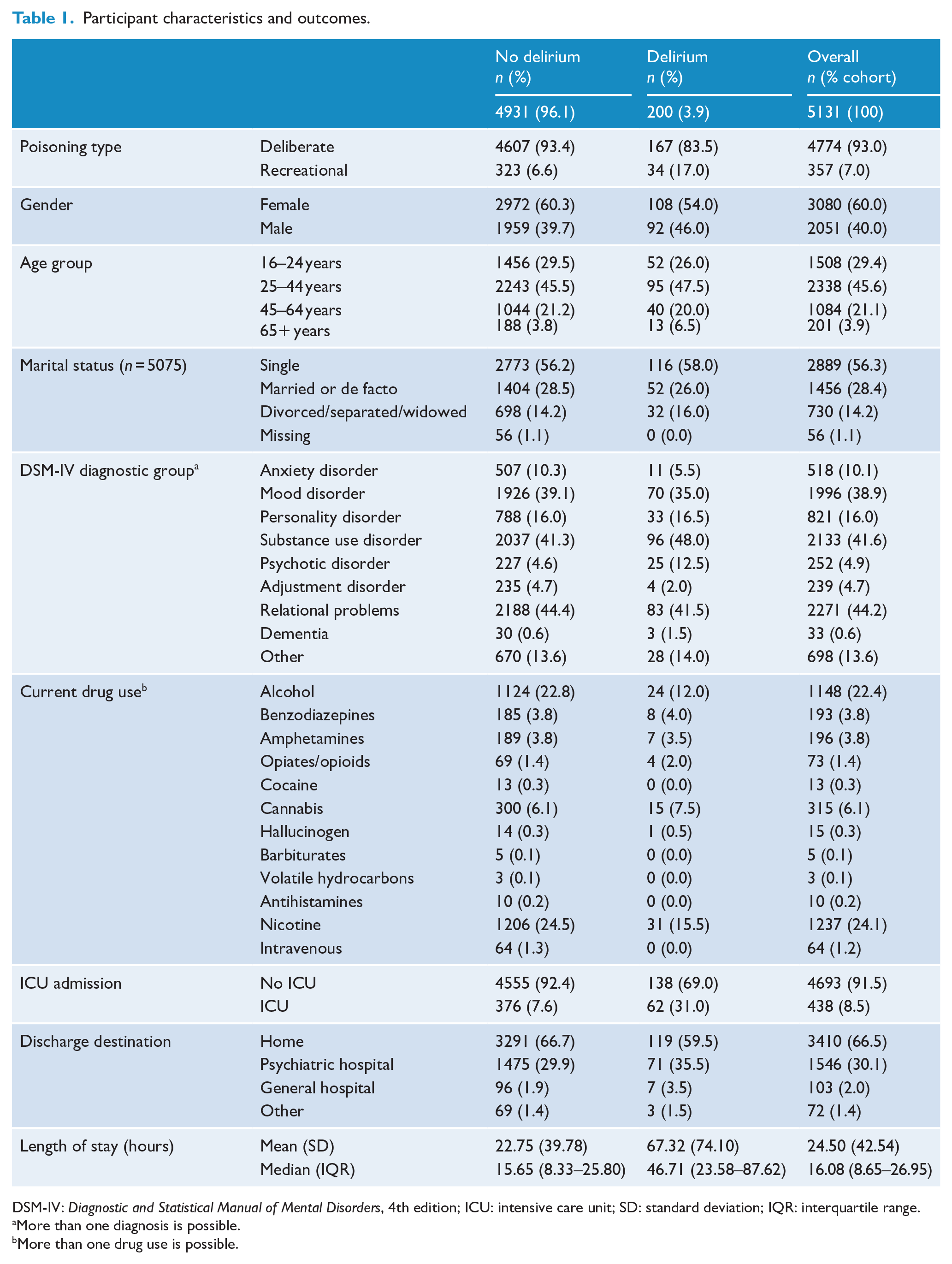
DSM-IV: Diagnostic and Statistical Manual of Mental Disorders, 4th edition; ICU: intensive care unit; SD: standard deviation; IQR: interquartile range.
More than one diagnosis is possible.
More than one drug use is possible.
Outcomes
Drug-induced delirium occurred in 3.9% (n = 200) of cases. ICU treatment was required by 8.5% (n = 438); general hospital length of stay was a mean of 24.50 hours (standard deviation [SD] = 42.54) and a median of 16.08 hours (IQR = 8.65–26.95). Two-thirds of the population were discharged home at the end of the general hospital admission and 30% were transferred to the psychiatric hospital for possible inpatient care.
Pattern of ingestion and co-ingestion by major drug group
Ingestion and co-ingestion patterns are summarised in Table 2. The most commonly ingested major drug groups were non-opioid analgesics 33%, antidepressants 25% and benzodiazepines (and other sedative hypnotics) 24%. Co-ingestion was common. The percentage of co-ingestion for each major drug group ranged from 63% (stimulants) to 85% (cardiac medications); and for the most commonly ingested major drug groups, the range of co-ingestion was similarly high, 68% for non-opioid analgesics, 78% for antidepressants and 76% for benzodiazepines. There were nearly 2000 admissions with a single major drug group ingestion, over 2000 with 2 major drug group co-ingestions and over 700 with 3 major drug group co-ingestions. The number of major drug group ingestions (single and 2,3,4,5+ co-ingestions) by admissions can be seen in Supplementary Figure 1.
Patterns of ingestion and co-ingestion (n = 5313).
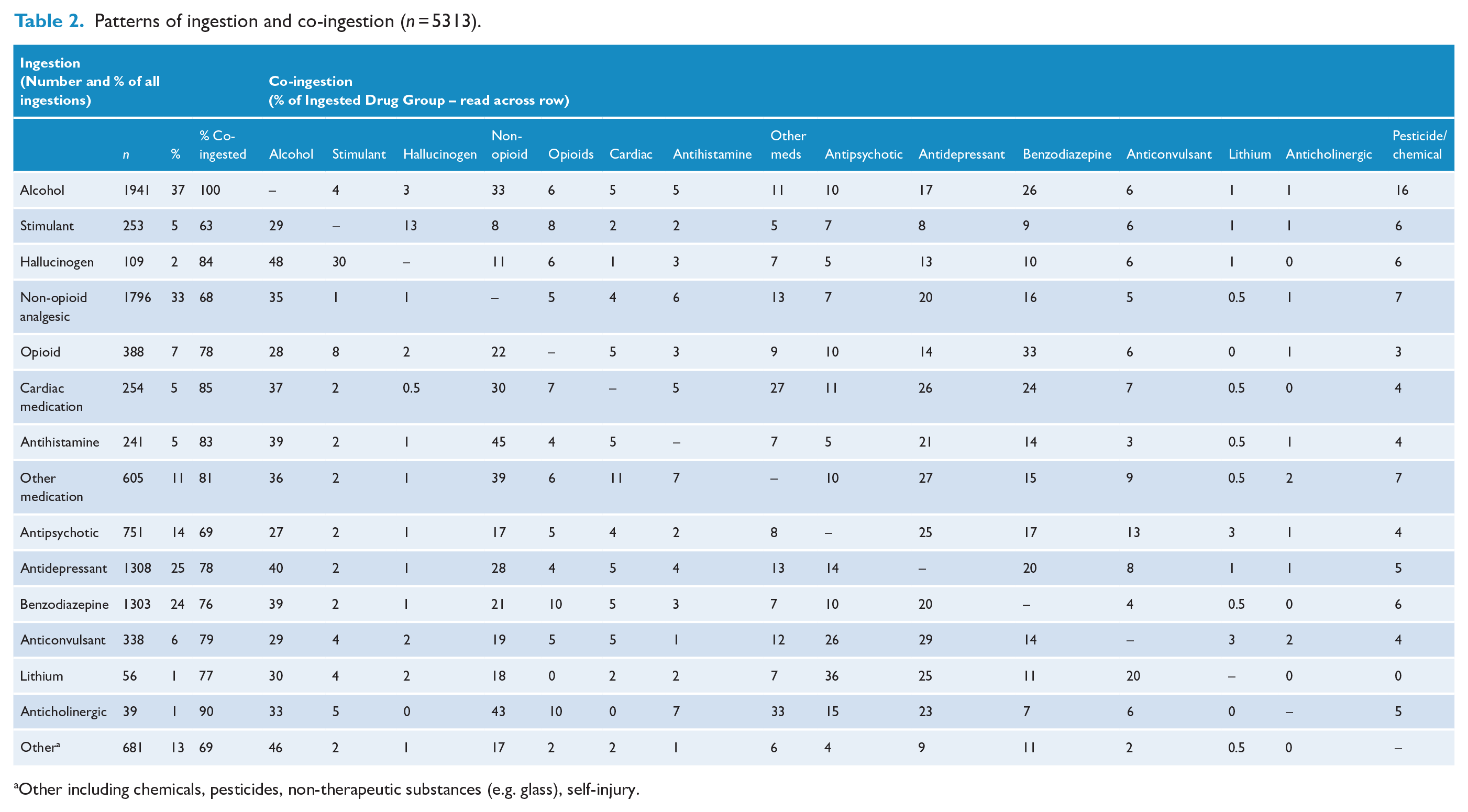
Other including chemicals, pesticides, non-therapeutic substances (e.g. glass), self-injury.
Unadjusted odds for drug-induced delirium
The unadjusted OR for any ingestion by specified major drug group vs all other ingestions (referent groups) for the development of delirium were increased for anticholinergics 10.79 (5.43–21.48), antihistamines 6.10 (4.20–8.84) and antipsychotics 2.99 (2.20–4.06); non-significant for opioids 1.31 (95% CI = [0.81, 2.13]); and reduced for benzodiazepines 0.37 (0.24–0.58). The unadjusted odds for drug-induced delirium for ingestions by all drug classes can be seen in Supplementary Table 2.
Adjusted models for drug-induced delirium
As detailed in Table 3, univariate estimates of ORs for each of the five major drug groups of interest were largely unaffected after sequential adjustment for age, gender, each of the other four co-ingestions by major drug group and alcohol co-ingestion.
Unadjusted and adjusted odds ratios for ingestions by major drug group and delirium.
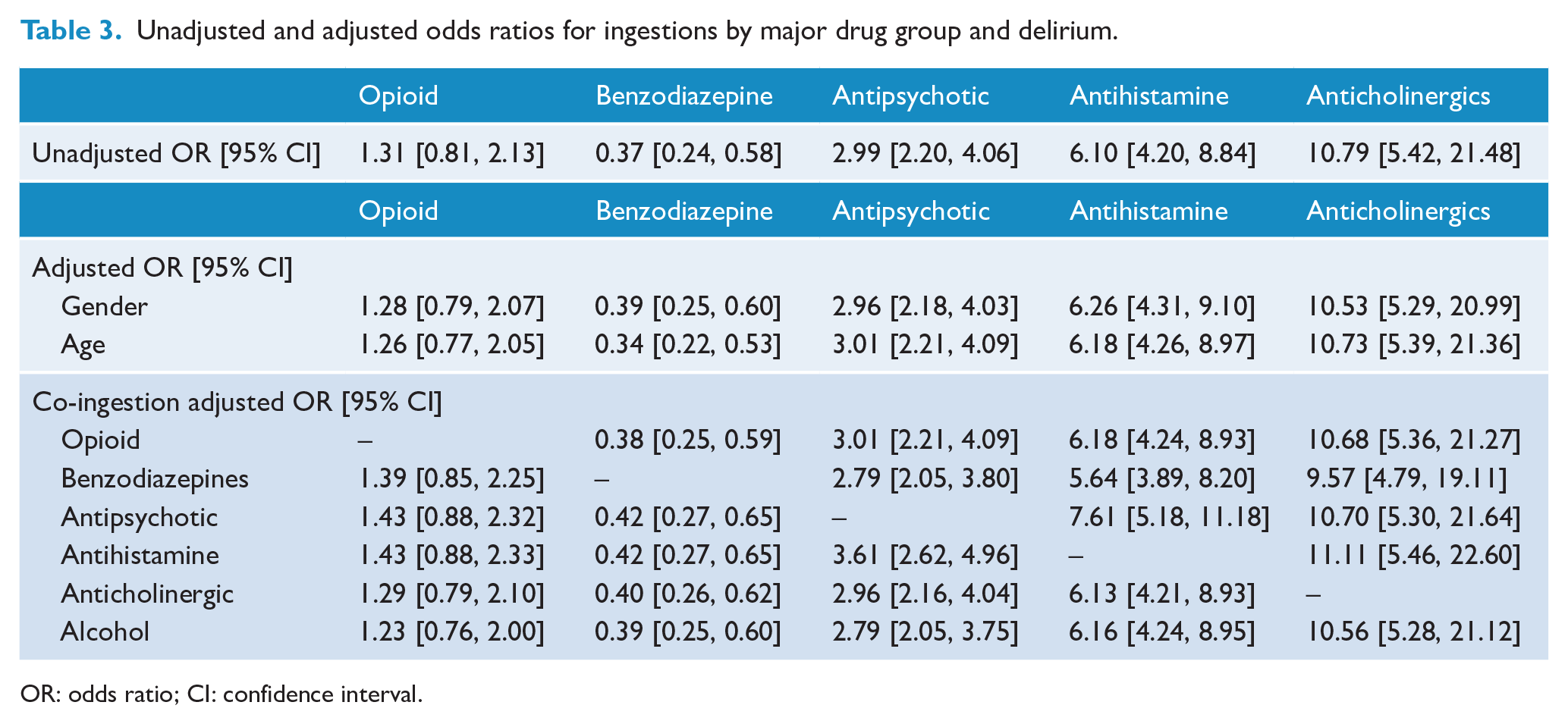
OR: odds ratio; CI: confidence interval.
Delirium predicting secondary outcomes
Delirium was significantly associated with increased odds of ICU treatment (OR = 5.40; 3.93–7.42), and longer general hospital length of stay, median 15.65 vs 46.83 hours (median test = 124.65, p = 0.001). Delirium was also significantly associated with a decreased odds of discharge to home (OR = 0.72; 0.54–0.96); or if expressed for the complementary composite endpoint of ‘institutional care’, an increased odds of 1.38 (1.04–1.84) (see Table 4).
Unadjusted odds ratios for delirium predicting clinical outcomes.
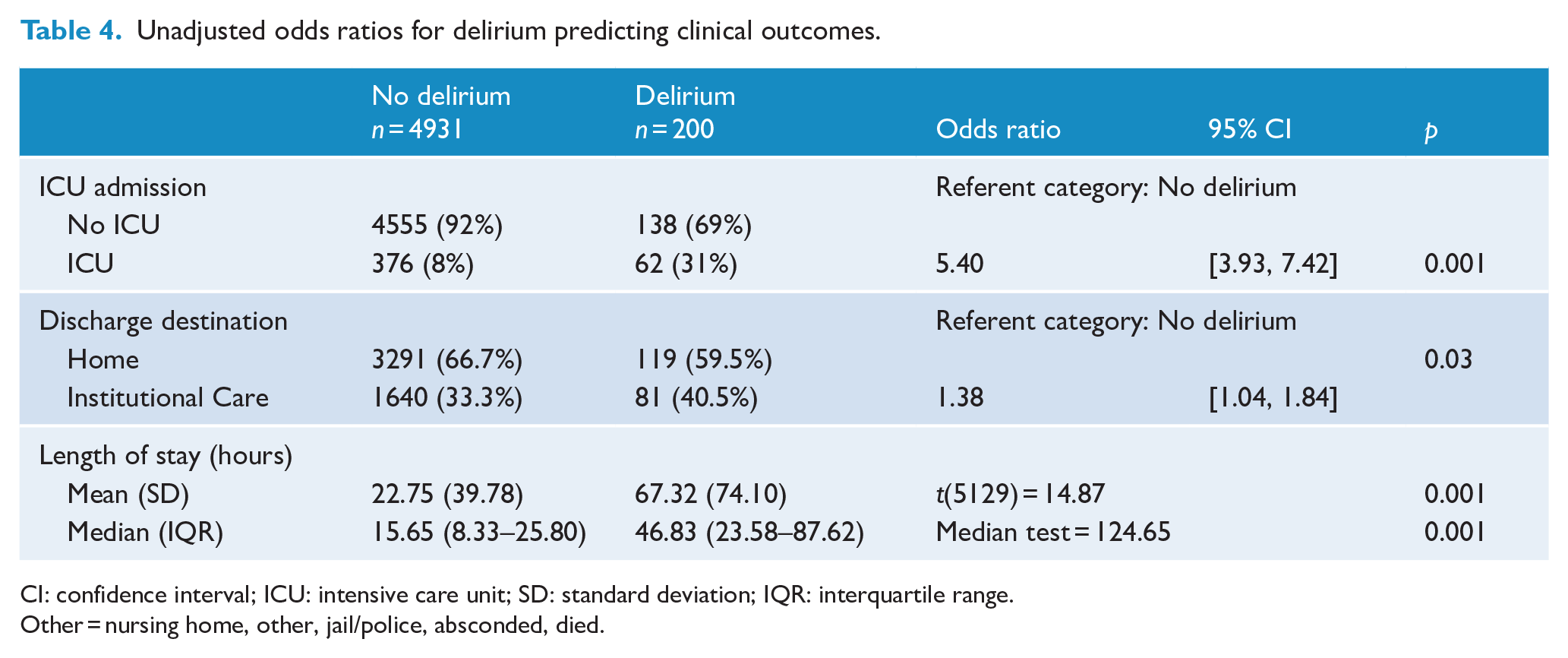
CI: confidence interval; ICU: intensive care unit; SD: standard deviation; IQR: interquartile range.
Other = nursing home, other, jail/police, absconded, died.
Discussion
Main findings
A wide range of ingestions by major drug group was observed; co-ingestion was more common than single group ingestions. Drug-induced delirium was relatively uncommon. Systematic reviews of all-cause delirium in general hospital populations have reported much higher estimates of prevalence; delirium at admission 10–31% and incidence of new delirium per admission 3–29% (Siddiqi et al., 2006), although estimates of general hospital drug-induced delirium are not well established.
Antipsychotics, antihistamines and anticholinergics all showed a significantly increased risk of association with delirium, with little change after adjustment for potential confounders, suggesting that it is reasonable to think of these medication groups as likely causative agents for drug-induced delirium in this self-poisoning population. From these likely causative major drug groups, antipsychotics were very commonly ingested and would be of most direct relevance to ED, ICU and other clinicians servicing this population. Our results show a different pattern to that reported in a systematic review of 14 studies (usually in the elderly and in therapeutic doses), which showed increased risk for opioids, benzodiazepines and possibly antihistamines, with no increased risk for antipsychotics and uncertainty for antihistamines and anticholinergics (Clegg and Young, 2011). The clinical population of hospital-treated self-poisoning is likely to be relatively young, medically fit and with less pre-morbid cognitive impairment compared to the usual general hospital populations, especially for elderly populations, which are frequently the populations of interest in delirium studies (Siddiqi et al., 2006). These differences in clinical characteristics may explain to some extent the differences found for drug groups associated with delirium.
The pharmacological mechanisms of action responsible for delirium, at least in part, are likely to be the anticholinergic and/or antihistaminic actions of these three drug groups. Surprisingly, opioids had a non-significant association with delirium, even after adjustment for confounding; and benzodiazepines (and other hypnotics) showed a significantly reduced odds for delirium, suggesting that these relatively commonly ingested medications may have been protective against drug-induced delirium in this population.
The possible reasons for a potentially protective effect of benzodiazepines are not obvious, particularly in this clinical population, i.e., relatively younger adults with supra-therapeutic dosing. Benzodiazepines have been considered to both protect against delirium and to be a risk factor for it, mostly in elderly populations (Meagher, 2001); and have been recommended as useful agents to treat delirium, especially where there is associated seizure or alcohol withdrawal (Mayo-Smith et al., 2004). In our study, benzodiazepine co-ingestion did not modify the deliriogenic effects of antipsychotics, antihistamines or anticholinergics, suggesting that benzodiazepines are not acting as just a simple treatment or pre-treatment for drug-induced delirium.
Major drug group or individual medication
Our analysis of exposure and co-ingestion was done at the level of major drug group rather than at the level of individual drug. As an example, not all antipsychotics will carry the same chance of induction of delirium. A review of overdose by atypical antipsychotics in the United States, of clozapine (n = 162), olanzapine (n = 21), quetiapine (n = 216) and risperidone (n = 48), reported adverse effects of confusion: clozapine 20%, olanzapine 38%, quetiapine 0.5%, risperidone 4%; central anticholinergic syndrome: clozapine 20%, olanzapine 43%, quetiapine 0%, risperidone 2%; and delirium: clozapine 9%, olanzapine 14%, quetiapine 0% and risperidone 0% (Burns, 2001). We plan future analyses of drug-induced delirium at the individual medication level for the antipsychotic group.
Delirium predicting adverse outcomes
Individual hospital studies have shown a longer ICU length of stay associated with delirium (Schubert et al., 2018), while our study showed a greater requirement for ICU care as a categorical variable. It may be that drug-induced delirium would be, in part, a non-specific proxy for medical seriousness requiring ICU intervention. Medically serious sedative drug poisonings, e.g., antipsychotics, opioids, antihistamines, benzodiazepines and co-ingestions, may require airway protection and mechanical ventilation in an ICU; or, e.g., antipsychotics and antidepressants may require monitoring and management of cardiac toxicity (Thanacoody, 2020). Since delirium is also commonly seen in ICU and ventilated patients (Cavallazzi et al., 2012), our classification of drug-induced delirium, in the ICU sub-group, might have been better classified as multifactorial delirium due to some contribution from end organ dysfunction, hypoxic injury, ventilation, sedation and other ICU experiences, e.g., sensory deprivation and disrupted circadian rhythms.
All-cause delirium in general hospital populations has been associated with increased length of stay and greater rates of institutional care on discharge (Siddiqi et al., 2006). Similarly, our results for drug-induced delirium showed an increased length of stay in the general hospital, a reduced likelihood of discharge to home, with a corresponding increased likelihood of discharge to the composite endpoint of institutional care. The increased risk of institutional care in previous studies is often in reference for elderly populations (Siddiqi et al., 2006) and usually means nursing home or other residential aged care home, which is not the same concept as institutional care in our study. We had a tiny minority with discharge to a nursing home for which it would not be appropriate to calculate an OR. Institutional care in our study was predominantly accounted for by discharge to a psychiatric hospital, which would not be expected to be a direct effect of drug-induced delirium. It is more likely that drug-induced delirium might be a proxy variable for the medical seriousness of the poisoning. The concept of medically serious suicide attempt has demonstrated associations with increased suicidal ideation, lethality of attempt, mental pain and communication difficulties (Horesh et al., 2012), a set of variables that could account for discharge to a psychiatric hospital.
Study limitations
The study relied on data from a single sentinel unit for the toxicological management of self-poisoning (Buckley et al., 2015; Hiles et al., 2015) and the results may not be generalisable to other places or populations. All variables recorded in the self-poisoning case register are derived from clinical assessment and not by use of standardised instruments. Cases of delirium may have been underestimated because of lack of clinical recognition, and our classification of drug-induced delirium arising in the period following medication exposure may have been overestimated by the inclusion of delirium with multifactorial causes (e.g. hypoxic brain injury suffered as a result of the poisoning and related periods of unconsciousness, aspiration or immobility and ICU intervention effects). Estimates of ORs for exposure by major drug group were derived using all other drug exposures as the referent group, where the referent group would include major drug groups with increased, neutral or decreased odds of developing delirium. This may affect the precision of the estimates reported and different values would result if alternative referent groups were used. Our exposures and co-ingestions were determined according to major drug groups, rather than individual drugs. It is likely that individual drugs within a class would have different risks for induction of delirium. We did not a priori select drug groups like non-opioid analgesics, hallucinogens, stimulants or lithium for study. We did not adjust any models for other possible confounders, e.g., co-ingestions by other major group, the amount of drug exposure by dose, exposure to regular medications, by clinical indication or for substance use. Although adjustment for these potential confounders may have altered the estimate of risk for the five major groups, we believe this is unlikely given such small changes after adjustment for age, gender and the other major drug group co-ingestions or alcohol co-ingestion. The analyses reporting increased risk for drug-induced delirium for the three secondary outcomes (ICU admission, longer length of stay and discharge destination) were exploratory, were unadjusted for potentially confounding variables and should be considered to be hypothesis generating only.
Conclusion
Non-opioid analgesics, antidepressants, benzodiazepines and antipsychotics are the most commonly ingested major drug groups in hospital-treated deliberate and recreational/chronic misuse self-poisonings. Delirium is relatively uncommon, but should be anticipated as more likely to occur after antipsychotic drug ingestion, which is common, and after antihistamine or anticholinergic ingestion, which are relatively uncommon. In contrast to reviews of drug-induced delirium at therapeutic doses, opioids were not associated with delirium and benzodiazepines appeared to be protective. Co-ingestion of drugs from other groups is common but does not substantially alter the risk of developing delirium above the ingestion of antipsychotics, antihistamines and anticholinergic drugs. Clinical services should be aware of these deliriogenic drug groups and be prepared to provide additional supportive care should delirium develop (Carter et al., 1996). Community prescribers could limit the prescribing, number of tablets and number of antipsychotic, anticholinergic and other medications dispensed to patients thought to be at risk of self-poisoning. Several antihistamines (e.g. Promethazine, Pheniramine, Cyproheptadine and Diphenhydramine) have been used in self-poisoning (Buckley et al., 2015) and are currently available without prescription as either Schedule 2 or Schedule 3 medications so limiting exposures to antihistamines may not be easily achieved.
Supplemental Material
sj-docx-1-anp-10.1177_00048674211009608 – Supplemental material for Hospital-treated deliberate self-poisoning patients: Drug-induced delirium and clinical outcomes
Supplemental material, sj-docx-1-anp-10.1177_00048674211009608 for Hospital-treated deliberate self-poisoning patients: Drug-induced delirium and clinical outcomes by Lindsay Gale, Katie McGill, Scott Twaddell, Ian M Whyte, Terry J Lewin and Gregory L Carter in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The HATS case register has had contributions from multiple clinicians (Clinical Toxicology registrars, Psychiatry registrars, Psychiatry Clinical Nurse Consultants), administrative officers and research managers over many years. We would like to thank them all for their diligence. We also thank and acknowledge all the patients who have had treatment in our clinical service and we offer our best wishes for their future lives.
Author Contributions
L.G. contributed to conceptualisation, investigation, methodology, project administration, writing – original draft, and writing – review and editing. K.M. contributed to data curation, formal analysis, visualisation, and writing – review and editing. S.T. contributed to conceptualisation, methodology, and writing – review and editing. T.J.L. contributed to formal analysis and writing – review and editing. I.M.W. contributed to resources, software, validation, and writing – review and editing. G.L.C. contributed to conceptualisation, investigation, methodology, project administration, resources, supervision, visualisation, and writing – review and editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Professors G.L.C. and I.M.W. have management and clinical responsibilities for patients treated in the Hunter Area Toxicology Service. Professor G.L.C. was Chair of the Royal Australian and New Zealand College of Psychiatry Clinical Practice Guidelines for Deliberate Self-Harm. The opinions expressed do not represent the opinions of the Calvary Mater Newcastle hospital or the Royal Australian and New Zealand College of Psychiatry.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study received no specific grant from any funding agency, commercial or not-for-profit sectors. The Hunter Area Toxicology Service case register has received financial support from various grants and other funding sources. K.M.’s position is funded by the Burdekin Suicide Prevention Program and administered by Hunter New England Mental Health Services. K.M. is also the recipient of a PhD scholarship from the Suicide Prevention Research Fund, made possible through a partnership between Suicide Prevention Australia and the Regional Australia Bank.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
