Abstract
Objective:
Antipsychotic drugs are widely used for treating patients with first episode of psychosis, targeting threshold psychotic symptoms. The clinical high risk of psychosis is characterized as subthreshold psychotic symptoms and it is unclear whether they can also benefit from antipsychotic drugs treatment. This study attempted to determine whether initiating antipsychotic drugs treatment in the clinical high risk of psychosis phase was superior to initiating antipsychotic drugs treatment in the first episode of psychosis phase, after the 2-year symptomatic and functional outcomes.
Method:
Drawing on 517 individuals with clinical high risk of psychosis from the ShangHai At Risk for Psychosis program, we identified 105 patients who converted to first episode of psychosis within the following 2 years. Patients who initiated antipsychotic drugs while at clinical high risk of psychosis (CHR_AP; n = 70) were compared with those who initiated antipsychotic drugs during a first episode of psychosis (FEP_AP; n = 35). Summary scores on positive symptoms and the global function scores at baseline and at 2 months, 1 year and 2 years of follow-up were analyzed to evaluate outcomes.
Results:
The CHR_AP and FEP_AP groups were not different in the severity of positive symptoms and functioning at baseline. However, the CHR_AP group exhibited significantly more serious negative symptoms and total symptoms than the FEP_AP group. Both groups exhibited a significant reduction in positive symptoms and function (p < 0.001). Repeated-measures analysis of variance revealed group by time interaction for symptomatic (F = 3.196, df = 3, p = 0.024) and functional scores (F = 7.306, df = 3, p < 0.001). The FEP_AP group showed higher remission rates than the CHR_AP group (χ2 = 22.270, p < 0.001). Compared to initiating antipsychotic drug treatments in the clinical high risk of psychosis state, initiating antipsychotic drugs treatments in the first episode of psychosis state predicted remission in a regression model for FEP_AP (odds ratio = 5.567, 95% confidence interval = [1.783, 17.383], p = 0.003).
Conclusion:
For clinical high risk of psychosis, antipsychotic drugs might be not the first choice in terms of long-term remission, which is more reasonable to use at the first episode of psychosis phase.
Introduction
Since antipsychotic drugs (AP) have been widely used for the treatment of psychotic symptoms, most doctors are quick to assume they may be effective for those attenuated psychotic symptoms in individuals at clinical high risk (CHR) of psychosis (Malhi and Bell, 2019). In particular, early intervention in the first 2–5 years of psychosis has been approved for improving long-term outcomes (Correll et al., 2018; Eaton et al., 1995; Srihari et al., 2012). This critical period hypothesis (Birchwood et al., 1998) was proposed more than 20 years ago but only considers the post onset of psychosis, rather than the premorbid phase. It remains largely unknown whether this critical period should be extended to the CHR phase, and if those subthreshold psychotic symptoms should also be targeted by AP treatment.
Premorbid phase interventions that delay or prevent onset of psychosis are clearly highly valuable for the CHR population. However, paradoxically, AP treatment in this phase is controversial. Although at least three existing clinical trials (McGlashan et al., 2006; McGorry et al., 2002; Woods et al., 2003) have indicated that AP, either alone or combined with psychotherapy, may benefit individuals with CHR, in terms of reducing the risk for conversion to full threshold psychosis or delaying the onset, concerns have been raised around safety (Liu and Demjaha, 2013), stigma (Yang et al., 2010) and ethical issues (Haroun et al., 2006). Although psychological (Addington et al., 2011) and nutritional (Amminger et al., 2020) early interventions (Stafford et al., 2013) were found slightly effective in reducing risk, in order to deal with attenuated psychotic symptoms (such as subthreshold delusion and hallucination), it would be beneficial for health professionals to know whether AP treatment is also effective. There were several recent meta-analyses (Bosnjak Kuharic et al., 2019; Davies et al., 2018a, 2018b; Fusar-Poli et al., 2019) showing no superior efficacy of AP compared to other treatments in individuals with CHR. This is an especially urgent question for countries in which non-pharmacological intervention for psychosis has not been well established.
The primary aim of this study is, therefore, to determine whether initiating AP treatment in the premorbid phase of psychosis is superior to initiating treatment during the following 2 years of the post-onset phase. We took advantage of an ongoing longitudinal program that includes a 10-year naturalistic CHR cohort, named the ShangHai At Risk for Psychosis (SHARP), where three-quarters of the participants had been treated with AP after their first outpatient visit (Collin et al., 2018; Zhang et al., 2018a). Among 517 individuals with CHR, 105 of them had converted to first episode of psychosis (FEP) within the following 2 years (Zhang et al., 2018c). This offered us the unique opportunity for post hoc analysis on the impact of premorbid AP treatment on illness trajectories and long-term symptomatic and functional outcomes.
Methods
Projects and participants
The SHARP program aimed to recruit and follow up individuals with CHR in an early psychosis identification program at the Shanghai Mental Health Center (SMHC) in China. A series of longitudinal studies were conducted, starting in 2011, to explore risk factors for psychosis. The SHARP team was originally led by Dr Larry Seidman before his untimely passing in 2017. Dr Seidman was also the principal investigator of the North American Prodrome Longitudinal Study (NAPLS)-2 project at Harvard University, and he implemented methods similar to those used in the NAPLS-2 to identify individuals with CHR in China, starting in 2010 (McEvoy et al., 2017; Zhang et al., 2014, 2018a). The SHARP cohort was psychotropically naïve when recruited. Individuals had not received treatment of any kind for a psychiatric disorder, and they had no history of substance abuse or dependence, which were exclusion criteria of the SHARP program. Zhang et al. (2014, 2015), Ren et al. (2017) and Zheng et al. (2012) provided further details regarding the SHARP methodology. Furthermore, most SHARP participants began to receive AP medication after their first outpatient visit; only one participant underwent psychotherapy, on four separate occasions.
Four studies and three funded projects contributed to establish the SHARP cohort. The Research Ethics Committees at the SMHC approved these studies. All participants gave written informed consent at the recruitment stage. Participants younger than 18 years of age assented, although consent forms were signed by their parents. A total of 517 CHR individuals were identified using face-to-face interviews based on the Structured Interview for Prodromal Syndromes (SIPS; Miller et al., 2002, 2003). For this study, 105 individuals with CHR who converted to psychosis in the following 2 years were included in the analysis. The number of participants in the overall project is shown in Figure 1. Both individuals with CHR and their caregivers were informed that the study involved several naturalistic follow-ups at 2 months, 1 year and 2 years and that research procedures were independent of routine clinical treatment procedures at the SMHC. With the exception of those who desired no further contact, individuals with CHR were re-assessed by telephone or face-to-face interview after 2 months, 1 year and 2 years using the SIPS. For the 105 converters, their outcome was determined based on the face-to-face interviews. Among them, 88 (83.8%) individuals were hospitalized, either at the SMHC or in local psychiatric units, 89 (84.8%) were diagnosed with schizophrenia, 15 (14.3%) with bipolar disorder with psychotic symptoms and 1 with obsessive-compulsive disorder with psychotic symptoms.
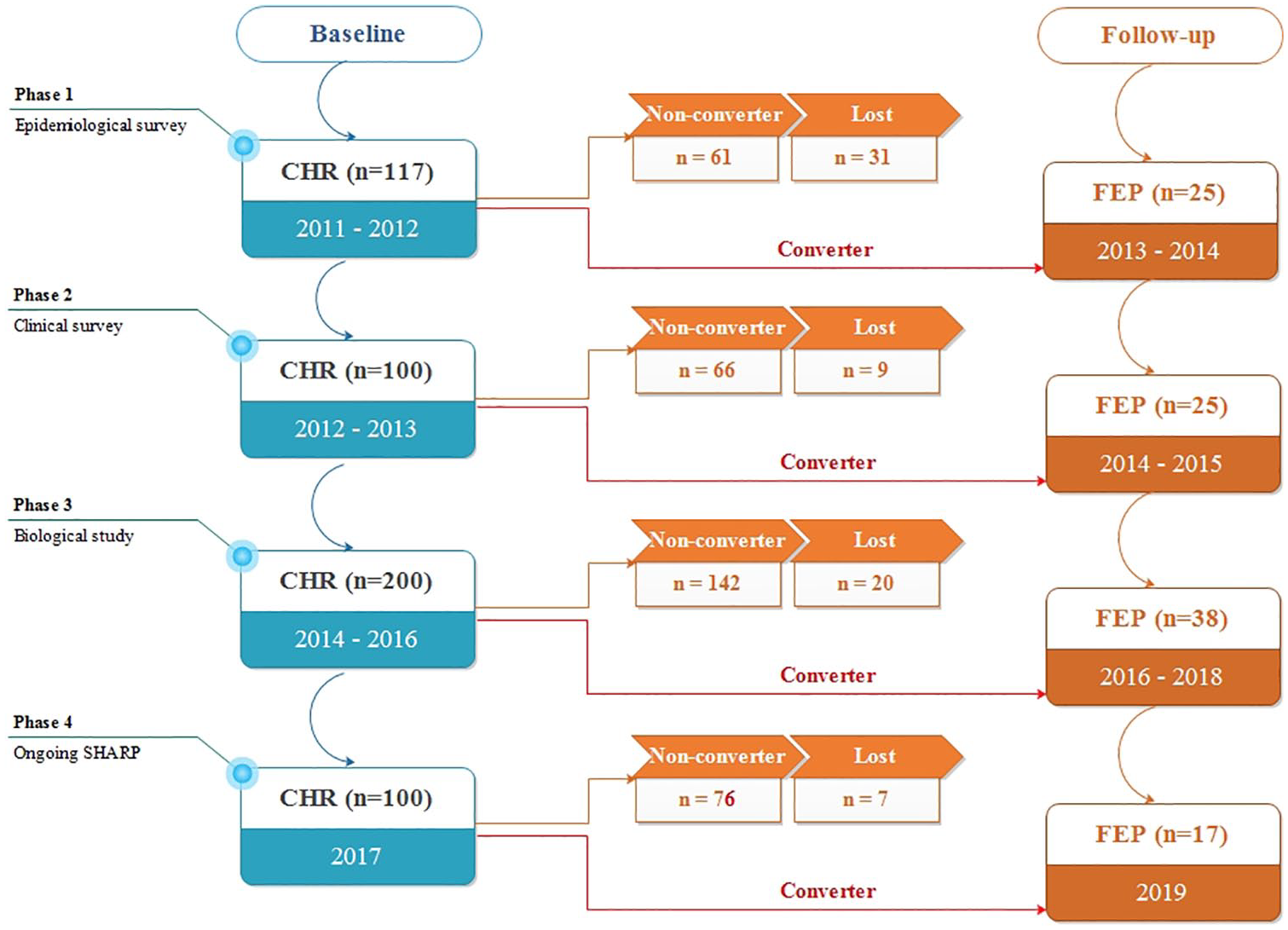
Number of participants at enrollment and follow-up.
Symptomatic and functional variables
The SIPS (Miller et al., 2003) was used to determine whether individuals met the criteria for CHR status, which consists of 19 items that assess four symptom domains: positive, negative, disorganized and general symptoms. The global assessment of function (GAF) was used to measure the global psychological, social and occupational functioning of patients in the SIPS interview. In our previous studies (Zhang et al., 2014, 2017), the Chinese version (Zheng et al., 2012) of SIPS, which was developed by the SHARP team, also demonstrated good inter-rater reliability (intraclass correlation coefficient r = 0.96, p < 0.01 for the SIPS total score) and validity (26.4% converted to psychosis in the following 2 years) in China. The first author is SIPS certified through Yale University-sponsored SIPS training.
Timeline criteria and outcome definitions
Four timeline periods were used in this study (Figure 2): (1) duration of untreated prodromal symptoms (DUPrS) is the period between the onset of attenuated psychotic symptoms and receiving appropriate diagnosis/care; (2) duration of untreated psychosis (DUP) is the period between the onset of the first frank psychotic symptoms and the receipt of AP treatment specific to this study; (3) CHR_Duration is the period between receiving CHR diagnosis and converting to FEP; and (4) AP_Duration is the period during which individuals receive AP treatment. The sample data was further divided into two groups: the CHR_AP group and the FEP_AP group. The CHR_AP group were those individuals who initiated AP treatment when they were at CHR status. Accordingly, the FEP_AP group were those individuals who initiated AP treatment after they converted to FEP.
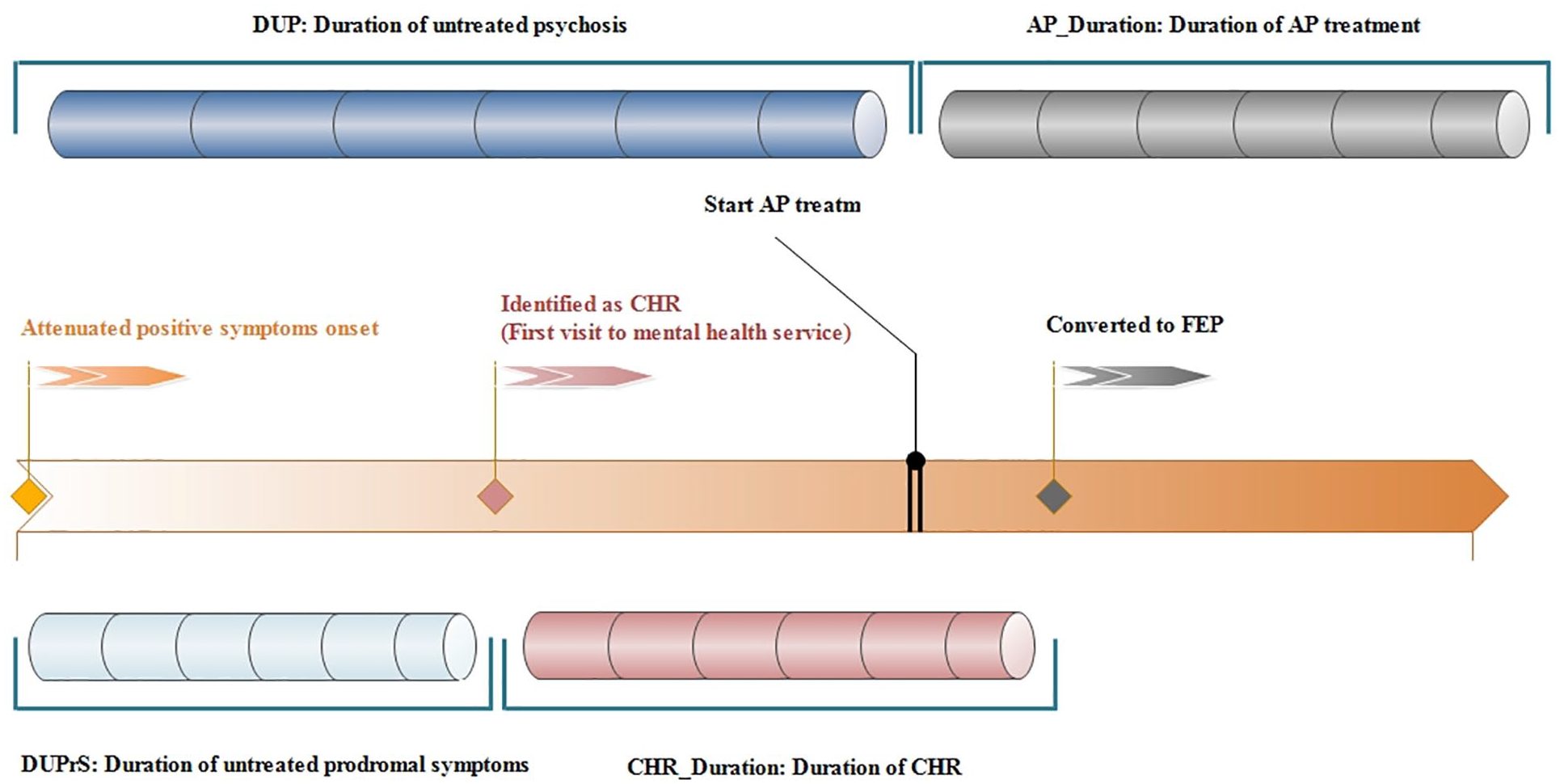
Conceptual Instruction for DUPrS, DUP, CHR_Duration and AP_Duration.
Remission was a major study outcome and determined based on both the GAF score for general function and the SIPS score for positive symptoms, at each follow-up point. Specifically, individuals with CHR who showed remission had a current GAF score of >70 (functional remission) and symptom scores of <3 for all positive symptoms (symptomatic remission). Overall remission included both symptomatic and functional remission.
Statistical analysis
Individuals with CHR were classified into CHR_AP and FEP_AP groups. Demographic and baseline clinical features were presented and compared between the two groups. Means and SDs were applied to describe continuous variables; counts and percentages were applied to describe categorical variables. Independent t-tests were conducted to measure CHR_AP vs FEP_AP group differences in continuous variables; either chi-square statistics or Mann–Whitney U test was used to examine categorical variables. The comparison of effects between the CHR_AP and FEP_AP groups on symptomatic and functional improvement during a 2-year follow-up was evaluated using repeated-measures analysis of variance (RMANOVA). Statistical significance was corrected using Bonferroni and Greenhouse-Geisser corrections were applied to correct for sphericity violations. RMANOVA was performed on the CHR_AP and FEP_AP groups to estimate and compare the trajectories of improvements in SIPS-positive symptom score and GAF score. Microsoft Excel was used to represent, as a pie chart, the proportion of individuals in each group who showed remission at their 2-year follow-up, and as a bar chart, the time points for DUPrS, DUP, CHR_Duration and AP_Duration by case. Comparisons between groups were conducted. The predictors were selected according to our a priori knowledge to mitigate model instability due to overfitting. For the same reason, we avoided including terms for the interactions among the predictors such as DUP and DUPrS. We evaluated our published papers (Zhang et al., 2014, 2017, 2018b, 2019) on psychosis and poor outcome prediction in CHR samples. As a result, seven predictors were selected including (1) age, (2) drop GAF score (GAF score baseline from highest in the past year), (3) total score of positive symptoms, (4) total score of negative symptoms, (5) DUPrS, (6) CHR_Duration and (7) CHR_AP vs FEP_AP. To identify baseline predictors of 2-year remission, a multiple logistic regression analysis was conducted that included the above seven predictors from demographic, clinical and medicinal variables.
Results
Sample characteristics
Individuals in the CHR_AP group and FEP_AP group did not differ in positive symptom severity or functioning at baseline. The CHR_AP group was younger and had significantly higher negative symptom scores and total scores on the Scale of Prodromal Symptoms (SOPS) compared to the FEP_AP group (Table 1).
Baseline demographics and clinical variables comparing CHR individuals initiated AP treatments at CHR statues (CHR_AP) and those initiated AP treatments at FEP statues.
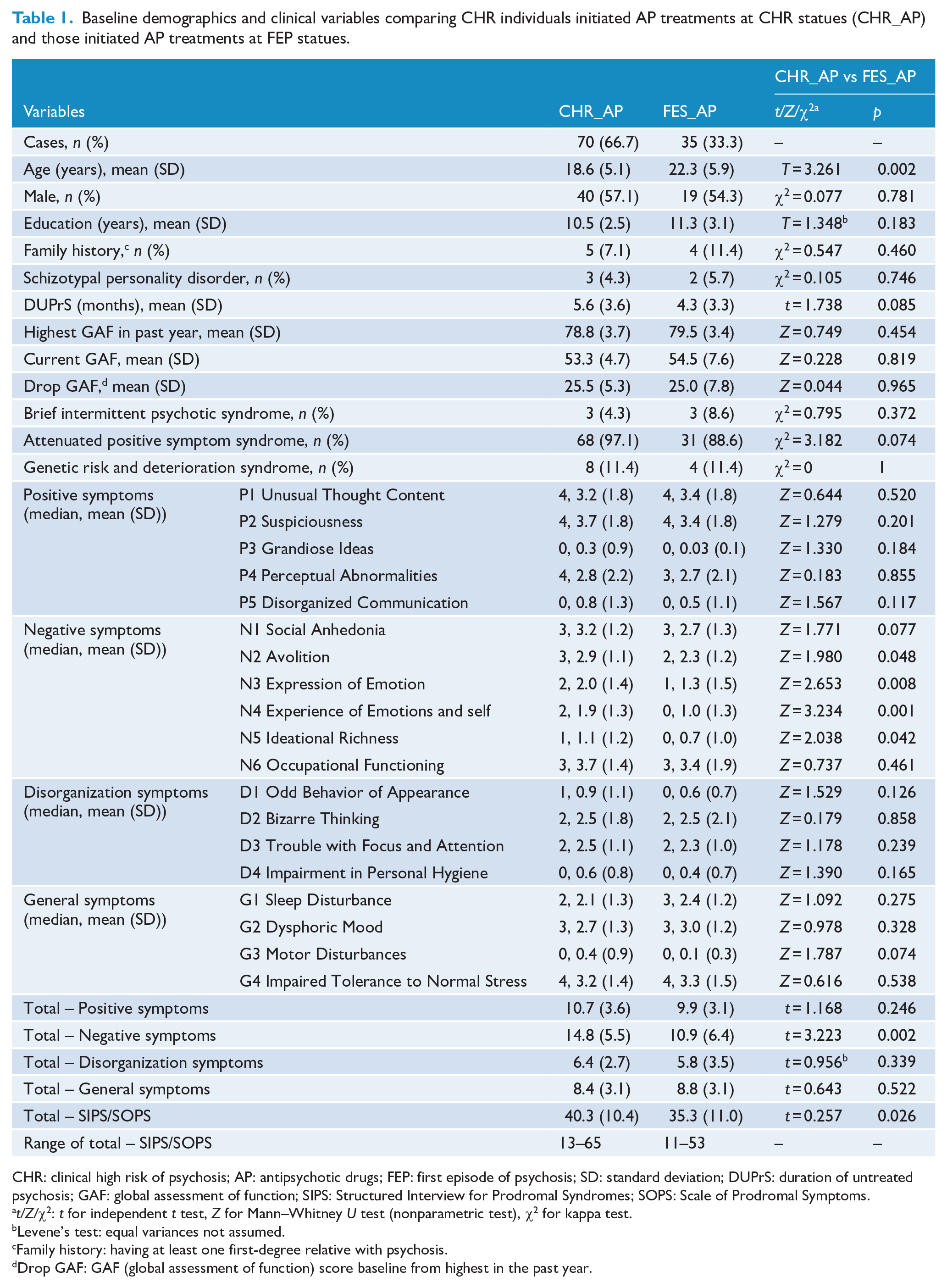
CHR: clinical high risk of psychosis; AP: antipsychotic drugs; FEP: first episode of psychosis; SD: standard deviation; DUPrS: duration of untreated psychosis; GAF: global assessment of function; SIPS: Structured Interview for Prodromal Syndromes; SOPS: Scale of Prodromal Symptoms.
t/Z/χ2: t for independent t test, Z for Mann–Whitney U test (nonparametric test), χ2 for kappa test.
Levene’s test: equal variances not assumed.
Family history: having at least one first-degree relative with psychosis.
Drop GAF: GAF (global assessment of function) score baseline from highest in the past year.
Medication exposure
Among the 105 CHR individuals, 70 received AP treatment during the CHR period (CHR_AP group) with an indicated olanzapine equivalent dose of 10.1 (SD = 6.9) mg, 52 individuals (74.3%) received AP monotherapy and 18 individuals (25.7%) received combined antidepressants with a fluoxetine equivalent dose of 19.7 (SD = 7.4) mg. For 35 individuals in the FEP_AP group, the mean olanzapine equivalent dose of AP was 10.5 (SD = 5.7) mg, 30 individuals (85.7%) received AP monotherapy and 5 individuals (14.3%) received combined antidepressants with a fluoxetine equivalent dose of 18.4 (SD = 5.1) mg. The three most commonly used AP in current sample were aripiprazole (n = 35, 33.3%), olanzapine (n = 31, 29.5%) and risperidone (15, 14.3%). Others included amisulpride (n = 13, 12.4%), ziprasidone (n = 11, 10.5%), quetiapine (n = 8, 7.6%) and paliperidone (n = 5, 4.8%). There were 40 individuals with CHR (38.1%) who changed their medication during the follow-up.
Symptom and function improvement
As shown in Figure 3, RMANOVA compared the symptom and function improvement trajectories between the CHR_AP and FEP_AP groups. Overall, both groups exhibited a significant reduction in positive symptoms and function (p < 0.001). As shown in Figure 3(A), time was a significant factor in the SIPS-positive symptom ratings (F = 19.330, df = 3, p < 0.001), and the group by time interaction significantly improved symptoms (F = 3.196, df = 3, p = 0.024). For function improvement (Figure 3(B)), time was also a significant factor in the GAF ratings (F = 40.767, df = 3, p < 0.001), and the group by time interaction significantly improved function (F = 7.306, df = 3, p < 0.001).
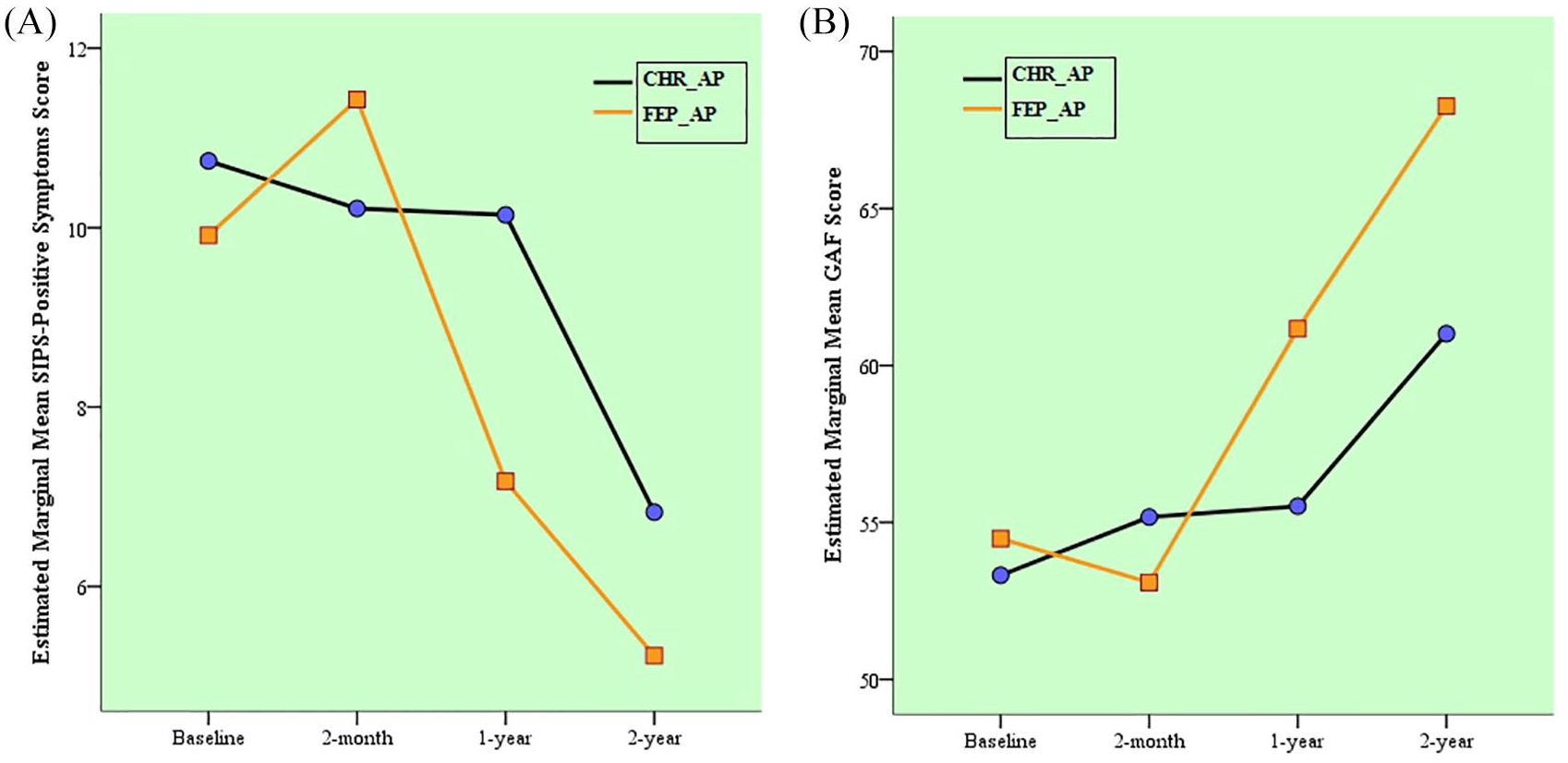
Mean score trajectories for (A) SIPS-positive symptoms and (B) GAF, based on the mixed-effects regression model.
Remission rates
The remission rates for each group are presented in Figure 4. The FEP_AP group showed the higher overall remission rates and similar patterns in symptomatic and functional remission rates.
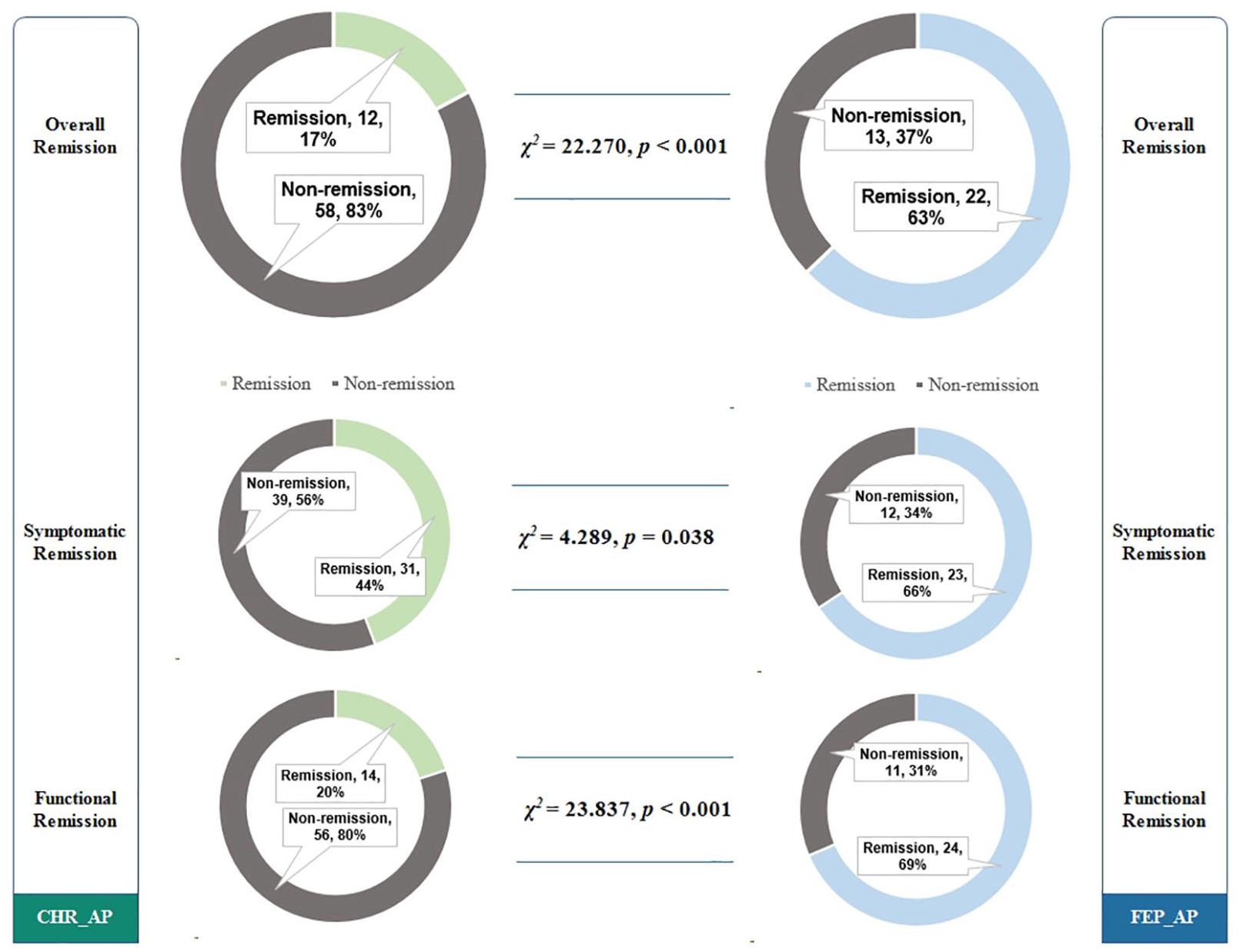
Two-year remission rates for the CHR_AP and FEP_AP groups.
Psychotic development and AP treatment timeline
On average, 105 individuals delayed 5.2 (SD = 3.568) months from attenuated symptom onset to their first visit to a professional in search of help. The mean time between being identified as CHR and developing full psychotic symptoms was 9.2 (SD = 6.388) months. As shown in Figure 5(C), compared with those individuals whose symptoms and function remitted over the course of the study, non-remission individuals had significantly longer delays on DUPrS (p = 0.028) and CHR_Duration (p < 0.001), but no difference in AP_Duration or DUP.
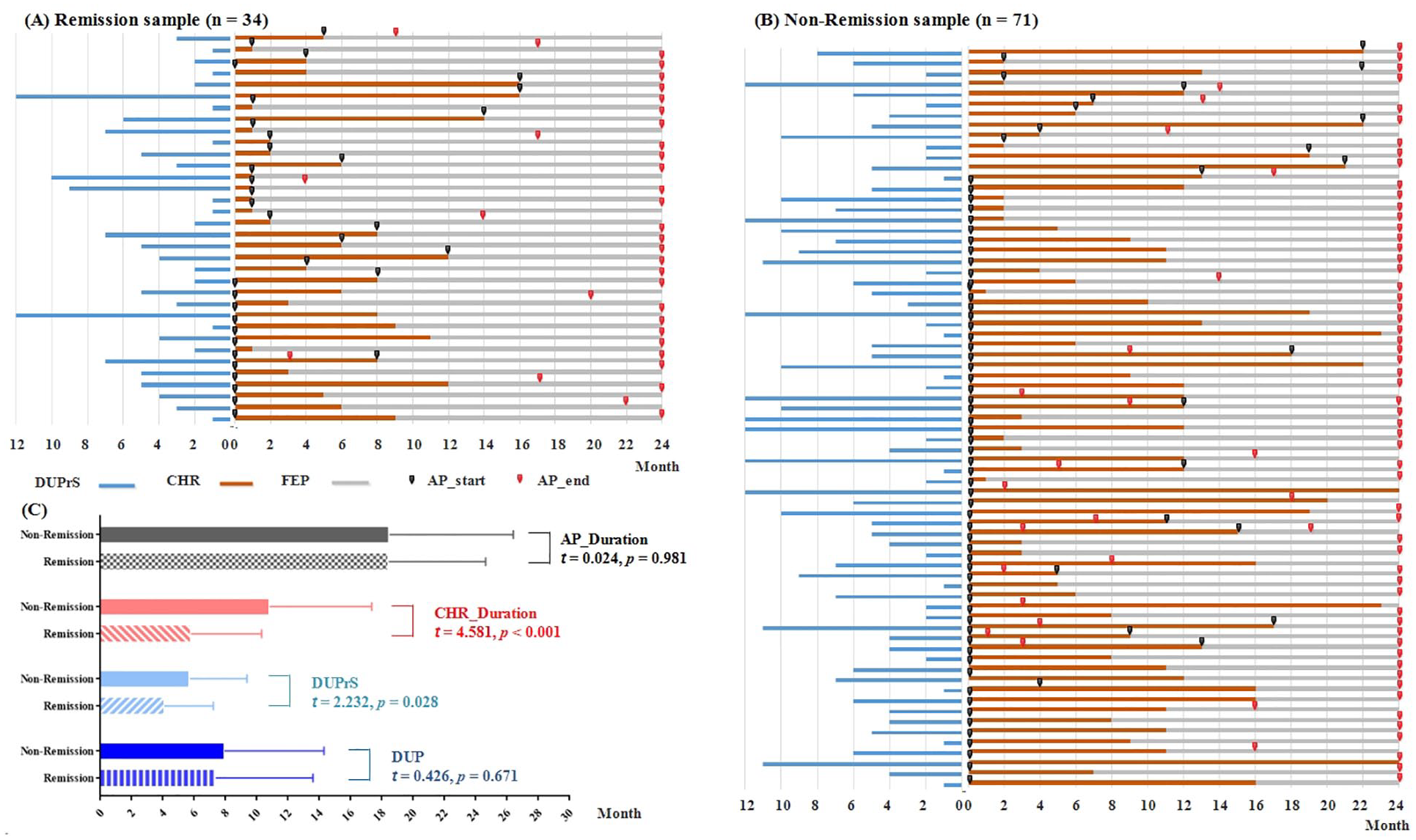
Individual psychotic development trajectory plots and antipsychotic drugs (AP) treatment timeline: (A) remission group, (B) non-remission group and (C) psychotic development timelines compared between the remission group and non-remission group.
Prediction of remission
Logistic regression was used to evaluate the effects of the seven predictors which were selected according to our a priori knowledge. Consistently, FEP_AP (odds ratio [OR] = 5.567, 95% confidence interval [CI] = [1.783, 17.383], p = 0.003), CHR_Duration (OR = 1.171, 95% CI = [1.059, 1.294], p = 0.002) and negative symptoms (OR = 1.180, 95% CI = [1.051, 1.325], p = 0.005) were all found to significantly predict remission in this model. Specifically, individuals with CHR who initiated AP during the FEP had shorter duration of CHR. Furthermore, less severe negative symptoms predicted a greater likelihood of remission.
Discussion
This study compared symptom severity and functioning in CHR individuals with AP treatment initiated at the premorbid phase with those of individuals who initiated AP treatment after conversion to FEP, at baseline and at 2 months, 1 year and 2 years. The main finding was a few differences between groups at baseline (Table 1); however, significant differences emerged in both symptomatic and functional remission after 2 years. This result is in contrast to our hypothesis. The divergent trajectories were not explained by baseline severity of positive symptoms or by AP treatment delays reflected in DUP or AP treatment duration. To our knowledge, our study is the first to report that initiating AP treatment at the premorbid phase increases the risk of poorer symptomatic and functional recovery, compared with delayed AP treatment until after FEP. Previous evidence showed that patients with schizotypal disorder treated with AP medication were at higher risk of transition than others (Albert et al., 2017). Although schizotypal disorder is not the same thing as CHR status, neither study supported the early use of AP for treatment during the premorbid phase of psychosis.
There are several reasons to explain the relatively poorer outcomes in individuals with CHR initiating AP treatment at the premorbid phase than at the post-onset phase. For example, and possibly the major reason, the outcome definition applied in this study was mainly based on long-term functioning, which is more susceptible to AP treatment (Harrow et al., 2012), not only because of the side effects of medication but also the negative ramifications in terms of stigmatization (Corcoran et al., 2005) from long-term AP use. Besides, while there were no differences in most demographic and clinical features at baseline between the CHR_AP and FEP_AP groups, individuals in the CHR_AP group were slightly younger than in the FEP_AP group. There is evidence that young people may be particularly vulnerable to various adverse effects of AP (e.g. rapid weight gain; Alvarez-Jimenez et al., 2008; Stafford et al., 2015). Adverse effects could further explain the poorer recovery in this group. Importantly, while AP drugs mainly play a role in the management of positive symptoms, for those adolescents and young adults in the critical period of physical growth, brain development and socialization, premature exposure to AP may be particularly harmful to their functional recovery.
Interestingly, the direction of improvement trajectories for positive symptoms is congruent with those regarding functioning (Figure 3). For both groups, functional improvements can reliably be interpreted as reduced severity of positive symptoms. As expected, significant time effects and group by time interaction effects were seen for both symptomatic and functional improvements. These data further supported the fact that individuals with CHR, but initiated AP at the FEP phase, would incur the benefits of AP treatment on symptoms and functioning, compared to those treated with AP at the premorbid phase.
The established illness timeline, from the onset of subthreshold symptoms to full psychosis, and treatment from the first visit to the mental health service to the end point of follow-up may shed light on the optimal time for early intervention for psychosis. Two recent interesting findings revealed that FEP patients with a prior history of CHR state are associated with poorer outcomes in psychotic symptoms and global functioning, compared with patients without such a history (Fusar-Poli et al., 2020a; Rosengard et al., 2019). The study suggested that FEP patients who experienced a CHR state may have a relatively longer DUP causing a poorer following recovery. This study also revealed that long periods of DUPrS and CHR state are significant predictors for poorer recovery. Together with our previous findings (Zhang et al., 2018b, 2018d), this evidence highlights the importance of reducing DUPrS to decrease future functional impairment in populations at the premorbid phase of psychosis. However, the major finding from this study adds a new message to remind clinicians that although shortening DUP is important, it is still necessary to pay attention in choosing appropriate, evidence-based treatments, instead of carelessly initiating AP treatment in CHR populations. Worthy of mention, if AP is the treatment for individuals with CHR (i.e. the endpoint of DUP used in this study), DUP was neither significantly different between remission and non-remission groups nor a significant predictor of remission.
The identified divergent trajectories and notable differences of long-term outcomes between the CHR_AP and FEP_AP groups are strengthened by the fact that our analyses addressed potential confounders of this comparison. The CHR_AP and FEP_AP groups were not different in severity of positive, disorganized and general symptoms and functioning at baseline, but were similar in dosage of AP, had equivalent exposure to antidepressants and demonstrated no difference in DUPrS. Our findings offer insights for the early management of CHR individuals, especially regarding the rational use of AP treatments. Although AP seems to be the most accessible form of treatment and are widely used for the Chinese CHR population, other non-pharmaceutical, evidence-based and less stigmatized early interventions such as cognitive behavioral and nutritional supplements should be considered as first-line therapy, while AP treatment can be used after the FEP.
Limitations
Generalizability of the study findings is limited by the fact that the CHR sample was recruited from the sole specialized CHR identification program. However, the SMHC is the largest psychiatric service center in China (serving over 1,000,000 outpatients per year) and provides professional treatment for patients throughout the country. About half of the current sample were not Shanghai natives. The present naturalistic and prospective observational study had the largest sample size to date. The sample included a large number of individuals with CHR who converted to FEP and had been treated with AP, allowing us to compare between CHR_AP and FEP_AP groups and formally test our a priori hypotheses with full statistical power. Second, the SHARP program was designed as a naturalistic and prospective observational study which found that many confounding factors cannot be controlled and not to address medication-related research questions. Thus, no side effect or tolerance data are available. However, since the very beginning of study, the prescription and delivery of AP were carefully recorded during the follow-up assessments. Third, our data may have been subject to inaccuracies in drug information and potential recall bias. To solve this issue, we performed tripartite checks involving the individuals with CHR, family members and medical records to confirm medical treatment details. However, our approach was less accurate than other strict methods, such as pill counts plus self-reporting. Fourth, the CHR_AP and FEP_AP groups differed in their total and negative symptom scores at baseline. Our previous study (Zhang et al., 2017) reported that negative symptoms significantly predicted conversion in the CHR sample. Thus, our results could have been confounded by the fact that negative symptoms were more severe in the CHR_AP group than in the FEP_AP group. This potential effect requires further investigation. Finally, we did not account for the effect of antidepressants, which may also prevent psychosis (Fusar-Poli et al., 2007).
Conclusion
In this longitudinal, large-scale, naturalistic CHR cohort, our findings provide novel evidence for poorer prognosis in both symptomatic and functional outcomes in individuals who had initiated AP treatment at the CHR state than those at FEP state. This study also revealed that long periods of CHR and more severe negative symptoms were significant predictors of poorer recovery. These results provide new insights into AP usage for prodromal phase psychosis, which is crucial for improving services and avoiding unnecessary harm in early intervention (Woods et al., 2020). Although no robust evidence yet exists to favor any indicated intervention over another (Fusar-Poli et al., 2020b), avoiding unnecessary harm remains a minimum requirement.
Footnotes
Acknowledgements
For the purpose of commemorate, Dr Larry J. Seidman passed away on 7 September 2017. Dr Robert W. McCarley passed away on May 27, 2017. Both were founders and core members of the SHARP (ShangHai At Risk for Psychosis) project.
Author Contributions
TH.Z. and JJ.W. conceptualized the study, wrote the first draft of manuscript and conducted the statistical analyses. LH.X., HR.C., and YY.W. interviewed participants and collected and organized the primary data. YY.T., YG.H. and XC.T. managed the literature searches, statistical analyses and edited the manuscript. CB.L., B.X. and JJ.W. designed the study and provided supervision in the implementation of the study. All authors have approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by Ministry of Science and Technology of China, National Key R&D Program of China (2016YFC1306800), National Natural Science Foundation of China (81671329, 81671332), Science and Technology Commission of Shanghai Municipality (19441907800, 17411953100, 19ZR1445200, 19411950800), Shanghai Clinical Research Center for Mental Health (19MC1911100) and The Clinical Research Center at Shanghai Mental Health Center (CRC2018ZD01, CRC2018ZD04, CRC2018YB01 and CRC2019ZD02).
