Abstract
Objective:
Bipolar disorder is one of the most frequent psychiatric disorders among suicidal patients. A large part of patients with bipolar disorder (30–50%) will attempt suicide. Suicidal ideation being a major risk factor of suicidal act, it is crucial to better characterize patients with suicidal bipolar depression (i.e. depression with current suicidal ideation). The aim of this study was to characterize suicidal bipolar depressed patients in comparison with non-suicidal depressed patients in terms of clinical characteristics, evolution of depression and suicidal ideation course over time, and risk of suicide attempt during follow-up.
Methods:
Among patients with bipolar disorder recruited from the network of FondaMental expert centres for bipolar disorder between 2009 and 2017, we selected patients with at least mild depression (Montgomery–Åsberg Depression Rating Scale total score >11) and without current manic symptomatology (Young Mania Rating Scale total score <7) at baseline (N = 938). Suicidal depression was defined by a baseline score ⩾2 for item 12 of the Quick Inventory of Depressive Symptomatology–Self Report (N = 271, 28.9%). Non-suicidal depression was defined by a baseline item 12 of the Quick Inventory of Depressive Symptomatology–Self Report score <2 (N = 667, 71.1%). A subsample of about 300 patients (with or without suicidal ideation at baseline) was followed up for 2 years.
Results:
Baseline clinical features (e.g. depression severity, childhood trauma, global functioning) were more severe in patients with than without suicidal depression. Suicidal patients tended to remain more suicidal throughout the follow-up than patients without suicidal ideation at baseline (3.4-fold higher risk of persistent suicidal ideation at the 2-year visit despite an improvement in depressive symptomatology).
Conclusions:
Depressed bipolar disorder patients reporting suicidal ideation had more severe clinical features at baseline and were more prone to report persistent suicidal ideation during the follow-up, independently of thymic state. Clinicians should closely monitor this subgroup of patients.
Introduction
Suicide is a major public health problem. More than 800,000 persons die by suicide worldwide each year, and there are 20–30 more suicide attempts (Zalsman et al., 2016). The inclusion of ‘suicidal behaviour disorder’ in Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) as a ‘condition for further study’ is the first step in the recognition that suicidal behaviour has its own physiopathology. It is crucial to better understand suicidal behaviour in order to find new therapeutic targets and to offer more personalized care to suicidal patients.
More than 90% of individuals who died by suicide suffer from a psychiatric disorder (Tondo et al., 2016). Bipolar disorder (BD) is one of the most represented psychiatric disorders among suicidal patients. Indeed, approximately 30–50% of patients with BD will attempt suicide (Ducasse et al., 2017; Miller and Black, 2020). Moreover, suicide attempt lethality is higher in BD than in other psychiatric disorders. A recent meta-analysis by Franklin et al. (2017) found that suicidal ideation (SI) is the third most potent predictor of future death by suicide, behind prior psychiatric hospitalizations and prior suicide attempts. This suggests that suicidal acts could be prevented by reducing SI especially since a large part of patients with BD report SI (Köhler-Forsberg et al., 2017). Even if antidepressants are suggested to reduce SI in depressed subjects, use of antidepressant still remains a source of debate in BD. In order to reduce SI within patients with BD, the use of lithium or electroconvulsive therapy (ECT) should be considered. Indeed, lithium is known to significantly reduce SI, and it significantly reduces suicidal risk more than some mood stabilizers (e.g. carbamazepine) (Smith and Cipriani, 2017). ECT has also shown good results in reducing SI within bipolar patients (Liang et al., 2018). Nevertheless, delay of action of lithium and ECT is quite long (some weeks). Moreover, necessity of a pre-therapeutic assessment delays and complicates their initiation. It is thus important to better understand the physiopathology of SI and find efficient therapeutics that act rapidly and that can be initiated rapidly. Interestingly, growing evidence suggests that SI and depression are related but may have a different physiopathology (Jobes and Joiner, 2019). In a cohort of depressed outpatients, Batterham et al. have identified different types of trajectories of SI and depressive symptoms. The characteristics of class membership were different for the two outcomes (Batterham et al., 2019). Some factors could thus impact changes of SI independently of depression. Furthermore, several studies showed that SI was persistent in a high proportion of individuals with a mental disorder, despite the improvement in depressive symptomatology, both at short (some weeks) and at long term (up to 10 years) (Madsen et al., 2016; Seo et al., 2014).
Thus, new potential therapeutic agents to target acute suicidal risk are currently developed. For instance, it has been demonstrated that ketamine efficiently reduces SI in depressed patients with BD (Grady et al., 2017; Kraus et al., 2017; Lee et al., 2015) with short-term efficacy (significant reduction in SI in half of the patients in the first 24 hours). But the emergence of such therapeutics targeting patients with SI involves confirming whether patients with suicidal depression (depression with SI) are a specific subgroup of depressed patients. The aim of this study was twofold: (1) transversal analysis to characterize suicidal bipolar depressed patients in terms of clinical characteristics in a cohort of 938 outpatients with BD; (2) longitudinal analysis to assess evolution of depression, SI course over time and risk of suicide attempt; in a subsample of this cohort, about 300 outpatients (with or without SI at baseline) with BD were followed for 2 years.
Methods
Participant enrolment
The methods are presented briefly below and have been extensively described elsewhere (Henry et al., 2011, 2017). The network of expert centres dedicated to BD is an innovative healthcare system in France and consists of 12 centres in different regions of the country (Henry et al., 2011) coordinated by Fondation FondaMental (www.fondation-fondamental.org). This network provides support to clinicians for the management of patients with BD and advice on personalized treatment strategies. It also generates clinically relevant data that can be used for research. A web application, E-bipolar©, is used to record each patient’s data in a computerized medical file. Anonymized data are stored in a national database FACE-BD (FondaMental Advanced Centres of Expertise for Bipolar Disorder) that was approved by the French body overseeing the safety of computerized databases (Commission Nationale de l’Informatique et des Libertés, DR-2011-069). Access to this web-based system for research purposes is regulated by a scientific committee.
Patients with BD are referred by a general practitioner or a psychiatrist to the expert centre where they are evaluated and followed. The same evaluation package is used by all centres. The assessment protocol was approved by the relevant ethical review board and requires only a letter of information to the patients (CPP-IIe de France IX, 18 January 2010).
Assessments
Patients were interviewed by a senior psychiatrist specialized in BD to clinically determine the diagnosis. The Structured Clinical Interview for DSM-IV Axis I Disorders was used to confirm the diagnosis and type of BD and to assess the presence of comorbid psychiatric disorders (e.g. anxious disorder, eating disorders, substance abuse) (Røysamb et al., 2011). Depressive symptomatology was assessed with two scales for all patients: the Montgomery–Åsberg Depression Rating Scale (MADRS) (Montgomery and Asberg, 1979) rated by clinicians and the Quick Inventory of Depressive Symptoms–Self Report (16-item QIDS-SR) (Rush et al., 2003) fulfilled by patients. Manic symptomatology was assessed by clinicians with the Young Mania Rating Scale (YMRS) (Young et al., 1978). The number of previous manic, hypomanic and depressive episodes and the presence of lifetime rapid cycling (defined by four or more thymic episodes within 1 year) were also recorded. The patient predominant polarity was determined according to the criteria defined by Colom et al. (2006): depressive or manic predominant polarity was defined when at least two of three past episodes fulfilled the criteria for major depressive episode or manic or hypomanic episode, respectively. Moreover, at each follow-up visit, the number of manic, hypomanic and depressive episodes since the last visit was recorded as well as the number of psychiatric hospitalizations and healthcare use since last visit.
SI was assessed with SI items from two depressive scales for all patients: item 12 of the QIDS-SR (QIDS–suicidal ideation item [QIDS-SI]) fulfilled by the patients and item 10 of the MADRS (MADRS–suicidal ideation item [MADRS-SI]) rated by clinicians. SI presence during the interval between visits was assessed with the following question: ‘Since the last visit did you think about death/taking your life even if you will never do it?’ Lifetime history of suicide attempt, number of lifetime suicide attempts and lifetime history of violent suicide attempt (e.g. hanging) were systematically recorded by a clinician. At each follow-up visit, the occurrence of suicide attempt since last visit was recorded.
Sociodemographic characteristics (age, gender, study level, marital status) and tobacco status were recorded. Current psychotropic treatments were also recorded. Adherence to treatment was assessed using the French version of the Medication Adherence Rating Scale (MARS) (Fond et al., 2017). In this 10-item questionnaire, a low score is linked to low medication adherence and a total score ⩾8 characterizes good medication adherence.
Global social functioning was assessed with the Functioning Assessment Short Test (FAST) (Rosa et al., 2007). The FAST is an interview specifically developed to assess the disability level in patients with BD and includes items on autonomy, occupational functioning, cognitive functioning, financial issues, interpersonal relationships, and leisure time. High total score indicates bad functioning.
Sleep disturbances were assessed with the Pittsburgh Sleep Quality Index (PSQI) (Buysse et al., 1989), a 19-item self-completed questionnaire that asks participants to describe their sleep patterns, such as usual bedtime and waking time, sleep latency and sleep time. A high score indicates bad sleep quality.
Anxiety was assessed using the State Trait Anxiety Inventory (STAI-A) (Borteyrou et al., 2008), a 20-item self-report on current anxiety symptoms. High scores indicate higher current anxiety.
The Childhood Trauma Questionnaire (CTQ) was used to assess the history of childhood trauma (Paquette et al., 2004). This scale evaluates five dimensions: emotional abuse, physical abuse, sexual abuse, emotional neglect and physical neglect. A total score >36 reveals the presence of childhood trauma (Vahapoglu et al., 2018).
Sample
Since this cohort consists of routine care patients, they are not solicited as much as prospective non-observational study. Thus, a large part of those patients are seen only at the inclusion visit. Consequently, we decided to perform two types of analysis: transversal analysis with all patients seen at the inclusion visit and longitudinal analysis with patients with at least two visits. Among the 3305 outpatients included in the 12 French FACE-BD centres between January 2009 and January 2017, 938 (28%) individuals with BD type I, II or not otherwise specified, with at least a mild depressive symptomatology (MADRS total score >11) (Kjaergaard et al., 2014; Schulte-van Maaren et al., 2013) and without manic symptomatology (YMRS total score <7) (Bonnín et al., 2018), were selected for the transversal analysis of this study. Among them, 435 (46.4%) had a second visit at 1 year and 268 (28.6%) had a third visit at 2 years. Those patients were selected for the longitudinal analysis. The severity of depression and SI at inclusion was not significantly different between patients selected for the transversal analysis only and patients selected for the longitudinal analysis only (Supplementary Table 1).
Definition of suicidal depression
Suicidal depression was defined by an MADRS total score >11 and QIDS-SI score ⩾2 (i.e. (2) ‘I think of suicide or death several times a week for several minutes’; (3) ‘I think of suicide or death several times a day in some detail, or I have made specific plans for suicide or have actually tried to take my life’); non-suicidal depressed patients were defined by an MADRS total score >11 and QIDS-SI score <2 (i.e. (0) ‘I do not think of suicide or death’; (1) ‘I feel that life is empty or wonder whether it’s worth living’). The suicidal item of QIDS-SR was chosen because patients are more likely to disclose SI in self-reported tests than to a clinician (Meyer et al., 2010). A previous study showed that a single suicide item from a depression rating scale is a valid approach to assess SI compared with the Scale for Suicide Ideation (Desseilles et al., 2012). This method was previously used in large clinical studies, such as the STAR*D (Zisook et al., 2009), and also in more recent studies (Bernert et al., 2017; Nobile et al., 2019).
Definition of thymic state during the follow-up
During the follow-up, the current patient mood state was defined as follows: manic episode (YMRS and MADRS total scores >7 and <12, respectively), mixed episode (YMRS and MADRS total scores >7 and ⩾12, respectively), depressive episode (YMRS and MADRS total scores <7 and ⩾12, respectively), and euthymia (YMRS and MADRS total scores <7 and <12, respectively).
Statistical analysis
Categorical variables were presented as percentages, and quantitative variables were presented as means with standard deviation (SD). At baseline, univariate binomial logistic regression models and the likelihood-ratio test (LRT) were used to assess the association between independent variables (sociodemographic and clinical variables) and the presence of suicidal depression. Then, multivariate binomial logistic regression models were used to assess the association between all independent variables associated with suicidal depression with a value of p < 0.10, independently of depression severity (measured with score of the QIDS-SR without the SI item).
During the follow-up, univariate binomial logistic regression models and LRT were performed, and the adjusted odds ratios (OR) and 95% confidence intervals (CI) were also estimated to quantify the risk of persistence of different symptoms (e.g. depression, SI) according to the patient groups. Then, models were adjusted for the trait variables associated with suicidal depression at baseline (age at BD onset, lifetime suicide attempts, and CTQ score).
A linear mixed model was used to determine whether depression severity decreased significantly in each group during the follow-up and to test whether the interaction between patient groups and period was significantly associated with depression changes during the follow-up.
The risk of suicide acts during the follow-up was assessed using a binomial logistic regression model and LRT.
Risk factors of SI persistence at the 1-year follow-up visit in patients with suicidal depression were assessed using a binomial logistic regression model and LRT (except for qualitative variables with not enough effectives, where the Fisher’s exact test was used when possible). The significance level was set at a value of p < 0.05. Analyses were performed using the SPSS software (version 26.0.0.0; IBM SPSS Statistics for Windows. Armonk, NY: IBM Corp), and the R software (version 3.6) was used for regression analyses and linear mixed models.
Results
Transversal analysis: sociodemographic and clinical comparison between suicidal and non-suicidal patients
Among the 938 at least mildly depressed BD patients, 585 (62.4%) were women, with a mean age of 40.69 (12.37) years. Most patients had type II BD (49.7%) and a depressive predominant polarity (58.5%). In total, 271 patients (28.9%) had baseline suicidal depression (MADRS >11 and QIDS-SI ⩾2); among them, 20.7% were actively suicidal (QIDS-SI ⩾3).
Sociodemographic variables (e.g. gender, age, marital status) and BD characteristics (e.g. type, predominant polarity, history of rapid cycling) were not significantly different between suicidal and non-suicidal patients (Table 1) as well as the frequency and type of psychotropic drug intake (Supplementary Table 2). Suicidal patients had more severe depression according to the QIDS-SR and MADRS total scores and related modified scores (total score minus SI item score). They had significantly worse baseline clinical features, more sleep disturbances and worse global functioning. After controlling for depression severity, patients with suicidal depression had significantly more often a lifetime history of suicide attempt and violent suicide attempt, alcohol and substance use disorder, and childhood trauma, They were younger at BD onset, more anxious and less adherent to treatment than non-suicidal patients (Table 1). According to the clinicians, patients with suicidal depression had more often SI (according to MADRS-SI ⩾2) than non-suicidal patients. Nevertheless, it is important to point out that 25% of suicidal depressed patients (QIDS-SI ⩾2), among those 17% actively suicidal (QIDS-SI ⩾3), were non-suicidal according to the clinician (MADRS-SI <2).
Association between sociodemographic and clinical data and suicidal depression in patients with BD with and without suicidal depression at baseline.
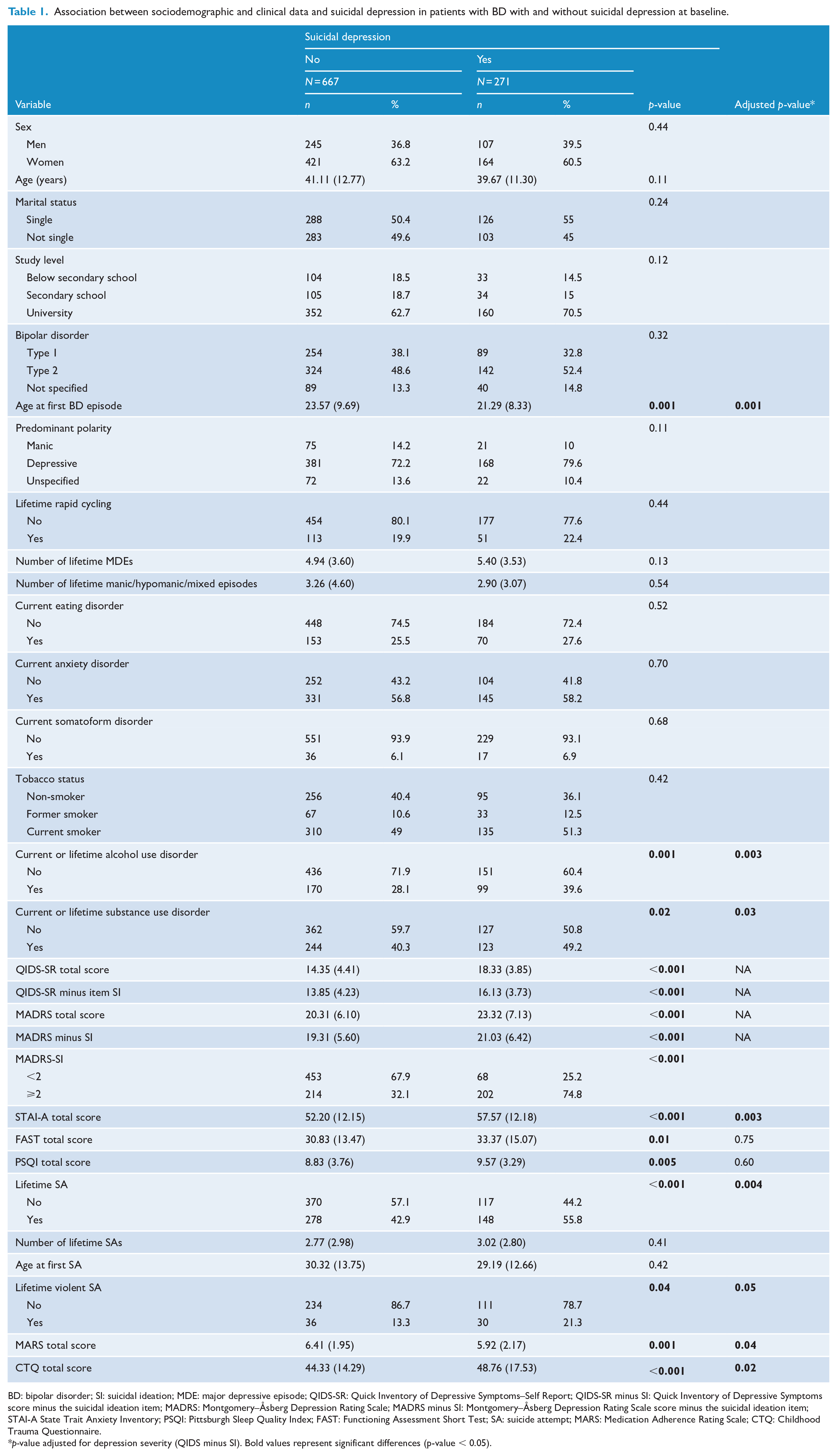
BD: bipolar disorder; SI: suicidal ideation; MDE: major depressive episode; QIDS-SR: Quick Inventory of Depressive Symptoms–Self Report; QIDS-SR minus SI: Quick Inventory of Depressive Symptoms score minus the suicidal ideation item; MADRS: Montgomery–Åsberg Depression Rating Scale; MADRS minus SI: Montgomery–Åsberg Depression Rating Scale score minus the suicidal ideation item; STAI-A State Trait Anxiety Inventory; PSQI: Pittsburgh Sleep Quality Index; FAST: Functioning Assessment Short Test; SA: suicide attempt; MARS: Medication Adherence Rating Scale; CTQ: Childhood Trauma Questionnaire.
p-value adjusted for depression severity (QIDS minus SI). Bold values represent significant differences (p-value < 0.05).
Longitudinal analysis: evolution of depression and SI across time
At the 1-year follow-up visit, SI (QIDS-SI and MADRS-SI) was more frequent in patients who were suicidal at baseline (QIDS-SI ⩾2) than in non-suicidal patients, even after adjustment for trait variables associated with suicidal depression at baseline (age at BD onset, lifetime suicide attempts, and CTQ score). Similarly, reporting SI between visits (OR = 2.20; 95% CI = [1.35, 3.58]; p = 0.001) was associated with baseline suicidal depression according to QIDS-SI. Moreover, SI at baseline according to QIDS-SI was significantly associated with a 1.7- and 2.6-fold higher risk of persistent SI according to clinician (with MADRS-SI) and patient (with QIDS-SI), respectively, at the 1-year visit (Figure 1; Table 2).
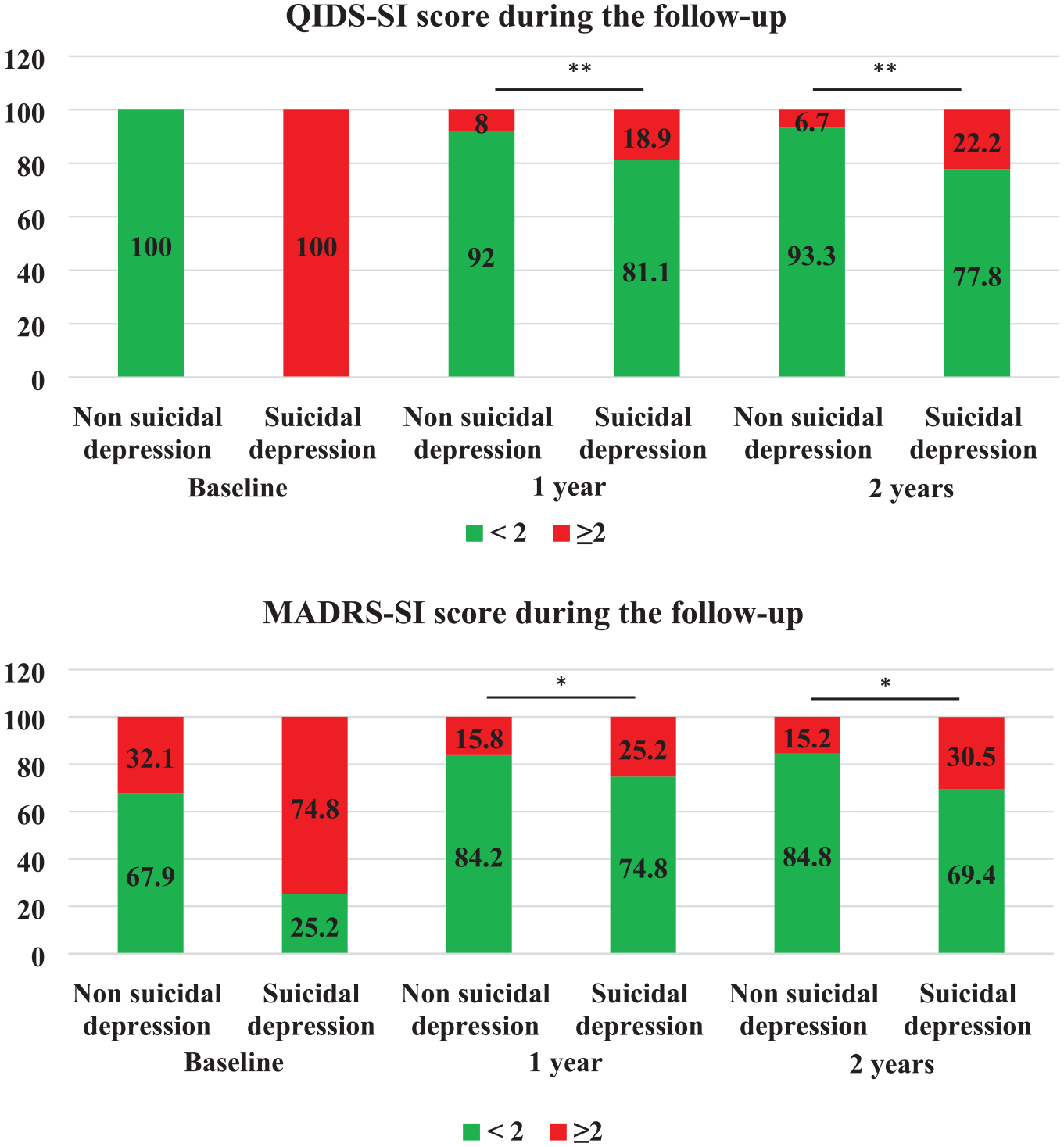
Suicidal ideation changes during the follow-up.
Association between clinical data and suicidal depression in patients with BD with and without baseline suicidal depression at the 1-year follow-up visit.
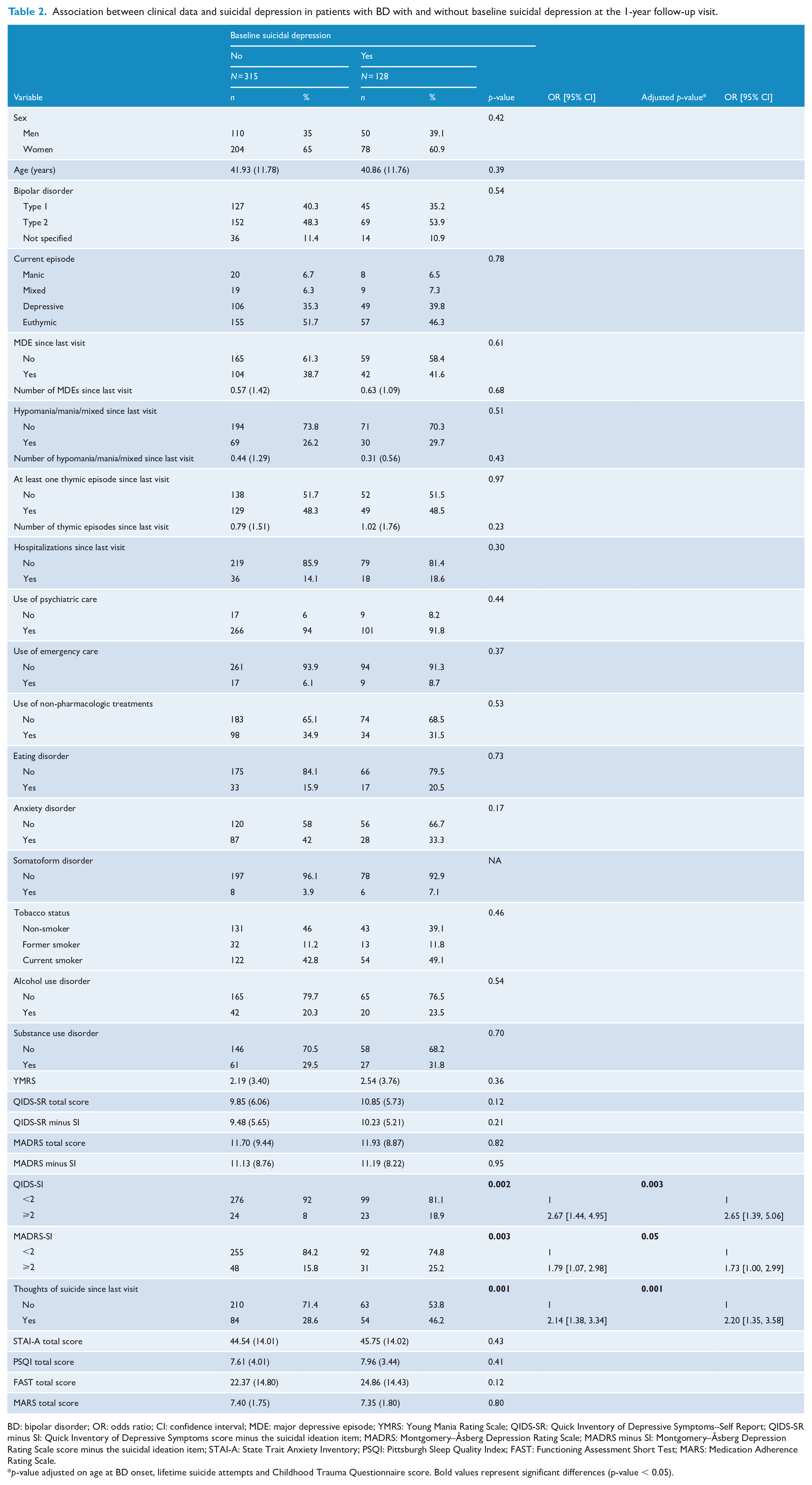
BD: bipolar disorder; OR: odds ratio; CI: confidence interval; MDE: major depressive episode; YMRS: Young Mania Rating Scale; QIDS-SR: Quick Inventory of Depressive Symptoms–Self Report; QIDS-SR minus SI: Quick Inventory of Depressive Symptoms score minus the suicidal ideation item; MADRS: Montgomery–Åsberg Depression Rating Scale; MADRS minus SI: Montgomery–Åsberg Depression Rating Scale score minus the suicidal ideation item; STAI-A: State Trait Anxiety Inventory; PSQI: Pittsburgh Sleep Quality Index; FAST: Functioning Assessment Short Test; MARS: Medication Adherence Rating Scale.
p-value adjusted on age at BD onset, lifetime suicide attempts and Childhood Trauma Questionnaire score. Bold values represent significant differences (p-value < 0.05).
Type of current mood episode, healthcare use, number and type of thymic episodes between visits, frequency of alcohol and substance use disorder, depression severity, anxiety, sleep disturbances, global functioning and treatment adherence were not significantly different between groups at the 1-year visit (Table 2). Among euthymic patients at the 1-year follow-up visit, suicidal patients at baseline had significantly more depressive residual symptoms than non-suicidal patients at baseline (Supplementary Table 3).
At the 2-year follow-up visit, there were no between-group differences for all clinical variables except for SI in the adjusted model (Supplementary Table 4). Baseline SI according to QIDS-SI was significantly associated with a 3.4-fold higher risk of persistent SI at the 2-year visit (Figure 1) according to patient (with QIDS-SI) but not clinician (with MADRS-SI) (p = 0.09).
Depression severity reduction (according to QIDS-SR and MADRS total scores) during follow-up was significant in both groups (Figure 2). The interaction between group and period was not significantly associated with depression changes according to the QIDS-SR and MADRS scores (p = 0.31 and p = 0.80, respectively). This indicates that depression changes over time were not different between groups.
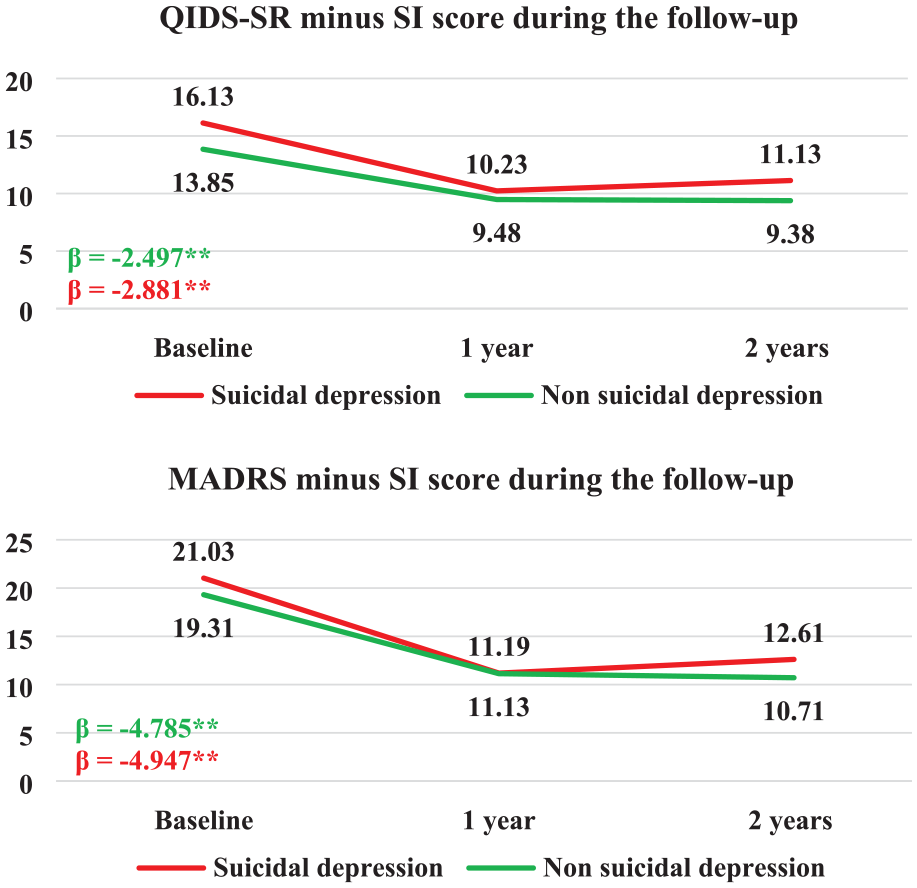
Depression score changes during the follow-up.
Interestingly, it is observed that between the first and the second years, mean of depression scores and proportion of patients with SI slightly increased in the suicidal depressed patients’ group but decreased in the non-suicidal group (Figures 1 and 2). An increase of about 1 point is observed for mean of depression scores and an increase of about 5% of proportion of patients with SI is observed in the suicidal depressed patients’ group.
The frequency of suicide attempts during the follow-up was not significantly different between groups (13.1% vs 8.7% at the 1-year follow-up and 24.2% vs 17.7% at the 2-year follow-up visit) (Supplementary Table 5).
Risk factors of SI persistence in suicidal depressed patients
None of the tested variables was significantly associated with the risk of SI persistence at the 1-year follow-up visit, except for lifetime history of violent suicide attempt (Supplementary Table 6).
Discussion
Suicide is a complex phenomenon. There is no single, simple way to elucidate its physiopathology. Moreover, even if SI is a major risk factor of death by suicide, people experiencing SI would not systematically attempt suicide. Moreover, a part of people dead by suicide did not experience SI. Consequently there is a growing interest in the ideation to action framework. The ‘bipolar suicidality model’ (BSM) (Malhi et al., 2018) is derived from the diathesis-stress model applied to BD and incorporates the recent ‘ideation to action’ models, namely, the interpersonal theory of suicide model, the integrated motivational and volitional model and the three-step theory model. This model is complex and takes into account a large panel of component of suicidal behaviour that can contribute to the understanding of genesis of suicidal act within bipolar patients. Importantly, BSM provides a framework for investigations. Among those investigations, it is possible to evaluate and characterize clinical specificities of bipolar patients with SI. Indeed, this could help to determine whether bipolar patients with SI could be a specific phenotype of bipolar patients with higher risk of suicidal behaviour and thus identify specific targets for suicide prevention intervention.
Here, we compared the clinical characteristics of suicidal and non-suicidal patients and investigated the course of SI and depression in a large cohort of depressed BD outpatients during 2-year follow-up. Our study indicates that baseline clinical features were more severe in patients with SI than those without SI. Most of these clinical characteristics are known to increase suicidal behaviour: young age at BD onset (Miller and Black, 2020), lifetime history of suicide attempt, comorbid substance use disorder (O’Rourke et al., 2017), history of childhood trauma (Etain et al., 2017), sleep disturbances (Stanley et al., 2017), high level of anxiety (Ott, 2018) and low treatment adherence (Gonzalez-Pinto et al., 2006). Moreover, history of suicide attempts, comorbid substance use disorder, childhood trauma, younger age at BD onset, anxiety and lower adherence to treatment were strongly associated with being suicidal independently of depression severity. As previously done in studies characterizing anxious depressed patients (Fava et al., 2006), our results were adjusted on depression severity, suggesting that more severe clinical features are linked to suicidal status rather than severity of the depressive episode. During the follow-up, patients becoming euthymic during follow-up having reported SI at baseline had more depressive resid-ual symptoms than non-suicidal patients at baseline. Furthermore, even if we selected patients without mixed features at baseline in order to characterize specifically depressed suicidal patients (based on YMRS and MADRS scores that can be equivalent to new DSM-5 criteria), some of them developed mixed features during follow-up. There were no differences between baseline suicidal and non-suicidal patients (may be explained by the very few numbers of patients with mixed features during follow-up). Nevertheless, it is important to keep in mind that patients with history or current mixed features are at high suicidal risk and that their suicidal state should be closely evaluated by clinicians. Finally, even if a non-significant difference was observed regarding risk of suicide attempt during follow-up between patients’ groups, due to a possible lack of statistical power and to the fact that those patients are referred to FACE-BD expert centres thus may reducing risk of suicide attempt, we suggest that suicidal patients at baseline were more prone to attempt suicide during the follow-up.
The main finding of our study is that patients with suicidal bipolar depression at baseline tended to remain suicidal throughout the follow-up (at each visit and also between visits) compared with patients without suicidal depression at baseline. This is independent of baseline suicidal risk factors because these results were adjusted for history of suicide attempt, childhood trauma, and age at BD onset. This was also independent of clinical state since there were no more significant differences between patients’ groups (i.e. depressive symptomatology, level of anxiety, sleep disturbances) during follow-up. Almost 20% of patients with suicidal depression at baseline reported SI during the follow-up. Among the patients who reported SI at baseline, history of violent suicide attempts predicted SI persistence during the follow-up. Previously, SI persistence was detected in 10–15% of individuals from psychiatric and non-clinical samples, respectively (Kasckow et al., 2016; Madsen et al., 2016). In a study that included BD patients, Köhler-Forsberg et al. (2017) found that 14% of outpatients experienced SI over 6 months. We could thus hypothesize that SI is related to but independent of depression and deserves a specific management. Indeed, we found that depression course was not different in the two patient groups. Conversely, in patients with baseline suicidal depression, SI persisted despite optimized treatment, as suggested by the improvement in depressive symptomatology and by their referral to FACE-BD expert centres that has been shown to significantly improve BD symptomatology (Henry et al., 2017). Finally, we observed a small increase in proportion of patients with SI and in depression scores between the first and second year in the suicidal depressed patients’ group. Since suicidal depressed patients are not different from non-suicidal patients on all variables tested at 1 and 2 years of follow-up (even depression scores despite a little increase) except for SI, this phenomenon reinforces the hypothesis that depressed bipolar patients with SI at baseline are more severe than depressed bipolar patients without SI and that they are more at risk of relapse of SI. Interestingly, new potential therapeutic strategies to target SI are emerging, such as ketamine that can rapidly reduce SI in patients with BD. In patients with unipolar depression, ketamine anti-suicidal effect is only partially explained by its antidepressant effect, suggesting an improvement in SI independent of improvement in depressive symptomatology. Additional studies on ketamine effects are needed especially in suicidal patients with BD.
Our results also emphasize the discrepancy between the patient’s and clinician’s ratings of SI (25% of suicidal depressed patients according to self-report at baseline were non-suicidal according to the clinician), in agreement with previous literature data (Samuel et al., 2013). In a study on veterans, the psychiatrist versus patients’ discrepancy for SI assessment was around 44% (McClure et al., 2018). Furthermore, in some studies, self-rating of SI was more predictive of a future suicide attempt than the clinician rating (Czyz et al., 2016; Gao et al., 2015). In the daily practice, clinicians should systematically assess SI, even in euthymic patients with BD, and complete their observation with the patient’s rating.
Our study has some limitations. First, only about 50% of patients selected for transversal analysis could have been selected for the prospective analysis. This could be explained by the fact that this cohort consists of routine care. Thus, patients are not solicited as much as prospective non-observational study. It thus reflects the difficulty of some bipolar patients to adhere to care at long term. However, baseline depression severity and SI were not different between patients who were followed up and those who were not. Second, we used a single item of the QIDS-SR to define suicidal depression. However, it has been shown that using a single item of a depression scale is a valid approach to evaluate SI. Moreover, it has been suggested that self-reports better catch SI than the clinician’s evaluation (Gao et al., 2015). Finally, our sample may not be fully representative of the BD population. Indeed, in general, patients referred to FACE-BD expert centres have a better access to healthcare and more years of education than other patients with BD.
Our study also has some strengths. Indeed, for the transversal analysis a huge sample of depressed bipolar patients was included. Then, we also included a longitudinal study for half of those patients, since the others were not seen again reflecting the real life numbers of patients who dropped out from care (Wells et al., 2013). Furthermore, a large panel of clinical variables (e.g. anxiety, sleep quality, global functioning) that are related to suicidal behaviour was assessed. It is important and necessary to evaluate the predictive value of such variables in a longitudinal way in order to better prevent suicide. It could have been interesting to also assess family history of suicidal behaviour within those patients to improve risk stratification of bipolar depressed patients. Studies including the assessment of potential endophenotypes integrating genetics, neuroimaging and neuropsychological constructs are needed in order to better understand suicidal behaviour and prevent suicide (Fornaro et al., 2018).
To conclude, our results highlight that patients with BD and suicidal depression have more severe clinical features and are more prone to SI, independently of their thymic state, and to have depressive residual symptoms during euthymia. These patients should be closely monitored by clinicians and may benefit from the development of specific therapeutic strategies to specifically target SI.
Supplemental Material
Suppl_Table_1 – Supplemental material for Characterization of depressed bipolar patients with current suicidal ideation
Supplemental material, Suppl_Table_1 for Characterization of depressed bipolar patients with current suicidal ideation by Bénédicte Nobile, Jonathan Dubois, Bruno Aouizerate, Valerie Aubin, Joséphine Loftus, Frank Bellivier, Raoul Belzeaux, Caroline Dubertret, Sebastien Gard, Emmanuel Haffen, Raymund Schwan, Pierre-Michel Llorca, Christine Passerieux, Paul Roux, Mircea Polosan, Bruno Etain, Marion Leboyer, Philippe Courtet and Emilie Olié in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank the FondaMental Foundation (![]() ), a private foundation supporting research in mental health that is developing a new model of translational research in psychiatry in France and supports the infrastructure of Bipolar Expert Centres. We express all our thanks to the nurses and to the patients who were included in this study. We thank Hakim Laouamri and his team (Seif Ben Salem, Karmène Souyris, Victor Barteau and Mohamed Laaidi) for the development of the FACE-BD computer interface, data management, quality control and regulatory aspects.
), a private foundation supporting research in mental health that is developing a new model of translational research in psychiatry in France and supports the infrastructure of Bipolar Expert Centres. We express all our thanks to the nurses and to the patients who were included in this study. We thank Hakim Laouamri and his team (Seif Ben Salem, Karmène Souyris, Victor Barteau and Mohamed Laaidi) for the development of the FACE-BD computer interface, data management, quality control and regulatory aspects.
FACE-BD Clinical Coordinating Center (Fondation FondaMental); B. Etain, E. Olié, M. Leboyer, E. Haffen and PM Llorca
FACE-BD Data Coordinating Center (Fondation FondaMental); V. Barteau, S. Bensalem, O. Godin, H. Laouamri, and K. Souryis;
FACE-BD Clinical Sites and Principal Collaborators in France.
AP-HP, Département Médico-Universitaire de psychiatrie et d’addictologie, DMU IMPAACT, Hôpitaux Universitaires H Mondor, Créteil; S. Hotier, A. Pelletier, N. Drancourt, JP. Sanchez, E. Saliou, C. Hebbache, J. Petrucci, L. Willaume and E. Bourdin.
AP-HP, GHU Paris Nord, DMU Neurosciences, Hôpital Fernand Widal; F. Bellivier, M. Carminati, B. Etain, J. Maruani, E. Marlinge, and J. Meheust.
Hôpital C. Perrens, Centre Expert Trouble Bipolaire, Service de Psychiatrie Adulte, Pôle 3-4-7, Bordeaux; B. Antoniol, A. Desage, S. Gard, A. Jutant, K. Mbailara, I. Minois, and L. Zanouy.
Département d’Urgence et Post Urgence Psychiatrique, CHRU Montpellier, Montpellier; L. Bardin, A. Cazals, P. Courtet, B. Deffinis, D. Ducasse, M. Gachet F. Molière, B. Noisette, E. Olié and G. Tarquini.
Pôle de Psychiatrie, addictologie et pédopsychiatrie, Hôpital Sainte Marguerite, Marseille; R. Belzeaux, N. Correard, F. Groppi, A. Lefrere, L. Lescalier, I. Muraccioli, M. Rebattu and N. Viglianese.
Service de Psychiatrie et Psychologie Clinique, CHU de Nancy, Hôpitaux de Brabois, Vandoeuvre Les Nancy; T. Schwitzer, R. Cohen, M. Milazzo, and O. Wajsbrot-Elgrabli.
Service Universitaire de Psychiatrie, CHU de Grenoble et des Alpes, Grenoble; T. Bougerol, B. Fredembach, A. Suisse, B. Halili, A. Pouchon, and M. Polosan.
Centre Hospitalier de Versailles, Service Universitaire de Psychiatrie d’adultes, Le Chesnay; A.M. Galliot, I. Grévin, A.S. Cannavo, N. Kayser, C. Passerieux, and P. Roux; Service de Psychiatrie.
Centre Hospitalier Princesse Grace, Monaco; V. Aubin, I. Cussac, M.A. Dupont, J. Loftus, and I. Medecin.
Service de psychiatrie et addictologie, Hôpital Louis Mourier, Colombes, AHPH, Groupe Hospitalo-universitaire AP-HP Nord, DMU ESPRIT France: C. Dubertret, N. Mazer, C. Portalier, C. Scognamiglio, and A. Bing.
Service de Psychiatrie de l’adulte B, Centre Expert Trouble Bipolaire, CHU de Clermont-Ferrand, Clermont-Ferrand, France: PM. Llorca, L. Samalin, L. Foures, D. Lacelle, S. Pires, C. Doriat and O. Blanc.
Author Contributions
E.O., P.C. and B.N. formulated the hypothesis, designed the study, interpreted results and wrote the paper. J.D. and B.N. performed statistical analysis. B.A., V.A., J.L., F.B., R.B., C.D., S.G., E.H., R.S., P.-M.L., C.P., P.R., M.P., B.E., M.L., E.O. and P.C. included and assessed patients. All authors contributed to revise and comment the paper. All authors approved the submitted version of the article.
Data Availability Statement
Due to ethical and legal restrictions, data involving clinical participants cannot be made publicly available. All relevant data are available upon request from the Fondation FondaMental for researchers who meet the criteria for access to confidential data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was funded by Fondation FondaMental (RTRS Santé Mentale), by the Investissements d’Avenir programme managed by the ANR under reference ANR-11-IDEX-0004-02 and ANR-10-COHO-10-01, and by INSERM (Institut National de la Santé et de la Recherche Médicale).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
