Abstract
Objective:
Previous studies showed alterations of brain function in the ventromedial prefrontal cortex of schizophrenia patients. Also, neurochemical changes, especially GABA level alteration, have been found in the medial prefrontal cortex of schizophrenia patients. However, the relationship between GABA level in the ventromedial prefrontal cortex and brain functional activity in schizophrenia patients remains unexplored.
Methods:
In total, 23 drug-naïve, first-episode psychosis patients and 26 matched healthy controls completed the study. The single voxel proton magnetic resonance spectroscopy data were acquired in ventromedial prefrontal cortex region, which was used as the seed region for resting-state functional connectivity analysis. The proton magnetic resonance spectroscopy data were processed to quantify the concentrations of GABA+, glutamine and glutamate, and N-acetylaspartate in ventromedial prefrontal cortex. Spearman correlation analysis was used to examine the relationship between metabolite concentration, functional connectivity and clinical variables. Pearson correlation analysis was used to examine the relationship between GABA+ concentration and functional connectivity value.
Results:
In first-episode psychosis patients, GABA+ level in ventromedial prefrontal cortex was higher and was positively correlated with ventromedial prefrontal cortex-left middle orbital frontal cortex functional connectivity. N-acetylaspartate level was positively correlated with positive symptoms, and the functional connectivity between ventromedial prefrontal cortex and left precuneus was negatively associated with negative symptoms of first-episode psychosis patients.
Conclusion:
Our results indicated that ventromedial prefrontal cortex functional connectivity changes were positively correlated with higher local GABA+ level in first-episode psychosis patients. The altered neurochemical concentration and functional connectivity provide insights into the pathology of schizophrenia.
Keywords
Introduction
Prefrontal cortex dysfunction plays an important role in the pathophysiology of schizophrenia (SCZ), especially at early stage (Zhang et al., 2015). A meta-analysis of resting-state functional magnetic resonance imaging (fMRI) studies has shown hypoactivation in ventromedial prefrontal cortex (vmPFC) of SCZ patients (Kühn and Gallinat, 2013). The vmPFC activity has also been closely related to various cognitive functions compromised in SCZ patients (Hiser and Koenigs, 2018), including theory of mind (Zemánková et al., 2018), decision-making (Ritter et al., 2004) and self-reflection (Van Der Meer et al., 2010).
Proton magnetic resonance spectroscopy (1H-MRS) provides a useful tool to detect and quantify the neuronal metabolite and neurotransmitter concentrations in vivo noninvasively. A large number of 1H-MRS studies have shown neurochemical changes in the prefrontal regions of SCZ patients. Specifically, higher γ-aminobutyric acid (GABA) level has been observed in medial prefrontal cortex (MPFC) of unmedicated SCZ patients (De La Fuente-Sandoval et al., 2018; Kegeles et al., 2012) as well as in the vmPFC area of drug-naïve, first-episode SCZ patients (Yang et al., 2015). Abnormal GABA synthesis and uptake were shown from a post-mortem study in SCZ patients (Simpson et al., 1989), suggesting the important role of GABAergic neurotransmission underlying the pathology of SCZ.
Increasing evidence has suggested the important role of GABA in modulating brain functional activity (Alcaro et al., 2010; Enzi et al., 2012). Through multimodal neuroimaging studies, it has been shown that a negative correlation exists between GABA concentration and stimulus-induced intra-regional activity (Duncan et al., 2014). Combining 1H-MRS and fMRI, it has been demonstrated that the GABA level within MPFC was inversely correlated with the negative functional coupling between vmPFC and amygdala in healthy subjects (Delli Pizzi et al., 2017a). Chen et al. (2019) found that GABA concentrations in the MPFC were positively associated with default mode network (DMN) deactivation during working memory task and positively correlated with anti-correlation between DMN and control network (CN) at rest in healthy adults. In first-episode psychosis (FEP) patients, GABA level in the anterior cingulate cortex (ACC) was positively correlated with local blood oxygen level–dependent BOLD response during Stroop task (Overbeek et al., 2019). However, the relationship between vmPFC GABA levels and brain functional activity in FEP patients remains unexplored.
In this exploratory study, we aimed to investigate the relationship between the GABA+ concentration in vmPFC and its functional connectivity (FC) in FEP patients. It has been shown that the antipsychotic medications could act as antagonists at dopamine D2 receptors, which may affect extracellular GABA level (Bourdelais and Deutch, 1994) and neurometabolic concentration (De La Fuente-Sandoval et al., 2018; Kegeles et al., 2012). Therefore, we only included first-episode, drug-naïve patients with SCZ to rule out the medication effect in the results. We hypothesized that the GABAergic signal in the vmPFC was correlated with the brain FC alteration in FEP patients.
Materials and methods
Participants
In total, 23 drug-naïve FEP patients (10 with first-episode SCZ, 13 with schizophreniform disorder) were recruited from Shanghai Mental Health Center. The patients diagnosed with schizophreniform disorder at the time of study enrollment were subsequently diagnosed with SCZ 6 months later. The inclusion criteria for patient group were age between 18 and 40 years, Han Chinese ethnicity, right-handedness, first-episode and drug-naïve. All of them fulfilled the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) diagnostic criteria for SCZ or schizophreniform disorder. The diagnosis was confirmed by a research psychiatrist (Z.Y.) using the Mini International Neuropsychiatric Interview (MINI 7.0) (Sheehan et al., 1998). Exclusion criteria were inability to provide informed consent, substance abuse (including nicotine), mental retardation, significant medical conditions including severe cardiovascular, hepatic and renal diseases and pregnancy or breastfeeding.
Clinical symptoms were assessed using the Positive and Negative Syndrome Scale (PANSS) (Kay et al., 1987) and the Clinical Global Impression (CGI) severity scale (Guy, 1976).
In total, 26 healthy controls were recruited from the local community via advertisement. All of them were interviewed by a research psychiatrist (Z.Y.) using the MINI 7.0 (Sheehan et al., 1998). Those with other mental disorders, neurological diseases, or a positive family history of mental illness were excluded.
This study was approved by the Institutional Review Board of the Shanghai Mental Health Center. Written informed consent was obtained from all the participants.
Data acquisition and processing
All magnetic resonance imaging (MRI), magnetic resonance spectroscopy (MRS) and fMRI data were collected on a 3.0T Siemens Verio MR Scanner (Siemens AG, Erlangen, Germany) with a 32-channel head coil at the Shanghai Mental Health Center. During MR scanning, the participants were asked to lay supine, keep quiet and remain awake with their eyes closed. Anatomical T1-weighted images were acquired using a magnetic preparation rapid gradient echo (MPRAGE) sequence with the following parameters: echo time (TE) = 3.65 ms, repetition time (TR) = 2530 ms, field of view (FOV) = 256 mm × 256 mm, slice thickness = 1.0 mm and slice number = 224. The resting-state fMRI data were collected with gradient echo pulse echo planar imaging (GRE-EPI) sequence (TE = 30 ms, TR = 2000 ms, flip angle [FA] = 90°, FOV = 22 cm, matrix = 64 × 64, slice thickness = 4 mm, slice gap = 0 mm, slice number = 30, voxel size = 3.4 mm × 3.4 mm × 4 mm, scan duration = 6 minutes).
For MRS data acquisition, the voxel was placed in the vmPFC (Figure 3(A)). The 30 mm × 30 mm × 30 mm voxel was centered in the interhemispheric fissure. The single voxel 1H-MRS data were acquired using a MEGA-PRESS spectral editing sequence on Siemens system (Mescher et al., 1998). The spectra with or without a frequency-selective inversion pulse were subtracted to obtain the difference spectrum to eliminate creatine signal. The following acquisition parameters were used: TR = 1500 ms, TE = 68 ms, number of averages = 128, edit pulse frequency = 1.90 ppm, edit pulse bandwidth = 45 Hz, region of interest (ROI) = 30 mm × 30 mm × 30 mm.
FC analysis
The fMRI data preprocessing was carried out using the Data Processing Assistant for Resting-State fMRI (DPARSF) (http://www.restfmri.net). A seed-based approach, which is based on Statistical Parametric Mapping (SPM8) (http://www.fil.ion.ucl.ac.uk/spm) and Resting-State fMRI Data Analysis Toolkit (REST; http://www.restfmri.net), was utilized for FC analysis. The first 10 volumes were discarded to avoid the influence of subject adaptation to the surroundings and magnetic equilibrium effect. Slice timing and head motion corrections were performed for the remaining data volumes. The exclusion criteria were over 2.5 mm translation or over 2.5° rotation in any direction. Spatial normalization was performed using the standard EPI template from the Montreal Neurological Institute (MNI). Imaging smoothing were performed with Gaussian kernel (FWHM = 4 mm). The data preprocessing also included linear detrending and filtering with band-pass filter (0.01–0.08 Hz).
The vmPFC was used as the seed ROI in the FC analysis. We defined the ROI on the standard MNI template (x = 0, y = 48, z = −8) as a sphere with 10 mm radius (Figure 1). The REST toolkit was used to correlate the averaged signal within the vmPFC with voxel-wise whole brain signals.
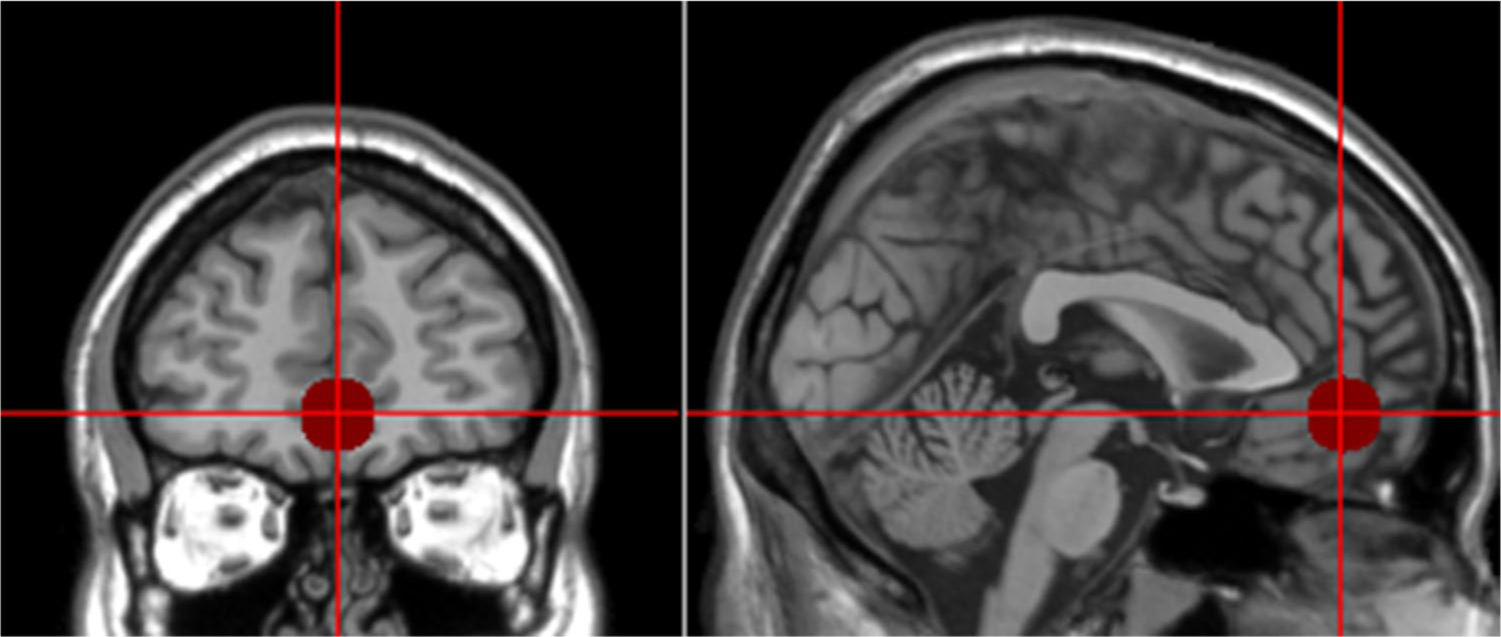
The ventromedial prefrontal cortex (vmPFC) seed region of interest (ROI) in resting-state functional connectivity analysis.
MRS data analysis
The MRS data were processed using the frequency-domain spectral fitting software LCModel (Provencher, 1993; Rosenberg et al., 2005). The metabolites quantified using LCModel include γ-aminobutyric acid (GABA+), the compound of glutamine and glutamate (Glx) and N-acetylaspartate (NAA) (Figure 3(C)). Since the signal around 3 ppm has a significant contribution from macromolecules as well as GABA, this apparent GABA concentration is often called GABA+ (O’Gorman et al., 2011). The quantification results with relative Cramer–Rao lower bounds less than 20% were accepted for further analysis (Quadrelli et al., 2016). No subject was excluded based on Cramer–Rao lower bound criteria. In order to correct for the partial volume effect, we performed tissue segmentation on the T1-weighted image using SPM8. The individual gray matter (GM), white matter (WM), and cerebrospinal fluid (CSF) maps were then registered and the fraction of each in the spectroscopic ROI was calculated. We then utilized the fractions of GM, WM and CSF to correct the partial volume effect and obtain the water-referenced quantification results (Bustillo et al., 2011).
Statistical analysis
All the statistical analysis was performed using SPSS (version 18.0; SPSS Inc., Chicago, III, USA). Demographic measures of age, gender and education years were compared between FEP patients and healthy controls using independent-sample t test or chi-square test. We used independent-sample t test to compare the metabolite concentration and FC value between FEP patients and healthy controls. The alteration of FC was considered significant at corrected p < 0.05 and cluster size >2295 mm3 (85 voxels) after AlphaSim multiple comparison correction (1000 iterations) using REST (Ledberg et al., 1998). Spearman correlation analysis was performed to examine the relationship between metabolite concentration, FC and clinical variables. We also examined the relationship between FC value and metabolite concentration with Pearson correlation analysis in both FEP patients and healthy controls. The results are considered statistically significant at p < 0.05.
Results
Demographics and clinical measures
The demographic and clinical information are shown in Table 1. No significant difference was found in age, gender and education level between the FEP patients and healthy controls.
Demographic and clinical characteristics of the participants.
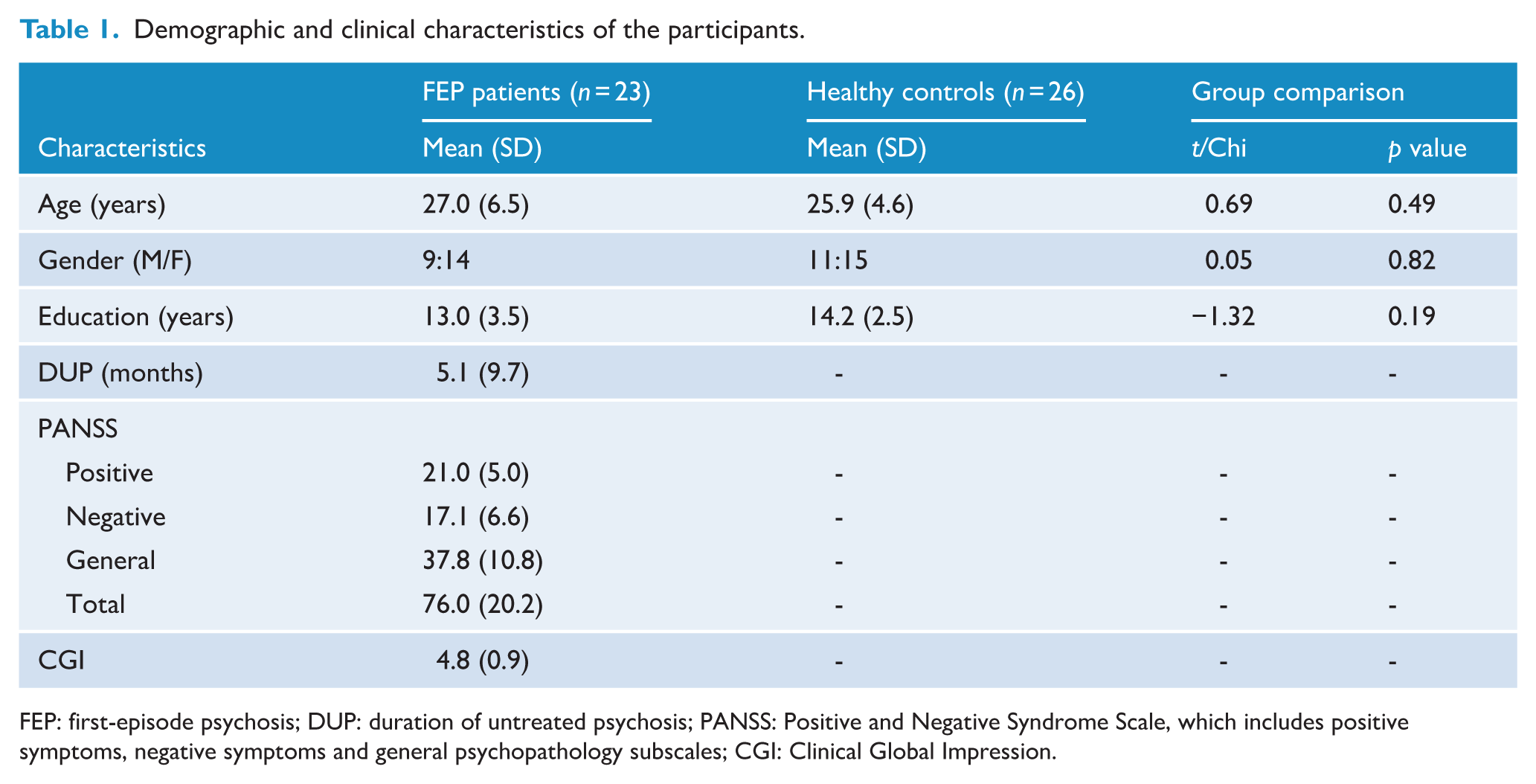
FEP: first-episode psychosis; DUP: duration of untreated psychosis; PANSS: Positive and Negative Syndrome Scale, which includes positive symptoms, negative symptoms and general psychopathology subscales; CGI: Clinical Global Impression.
Altered resting-state FC in FEP patients
Compared with healthy controls, we found increased FCs between vmPFC and left middle temporal, left precuneus, left middle orbital frontal (MOF) and right middle temporal cortex in FEP patients. On the other hand, we observed decreased FCs between vmPFC and left supplementary motor area (SMA), left dorsal anterior cingulate cortex (dACC), right precentral gyrus, right superior frontal gyrus and right hippocampus in FEP patients (Figure 2). The detailed FC alteration information is presented in online supplementary table S2.
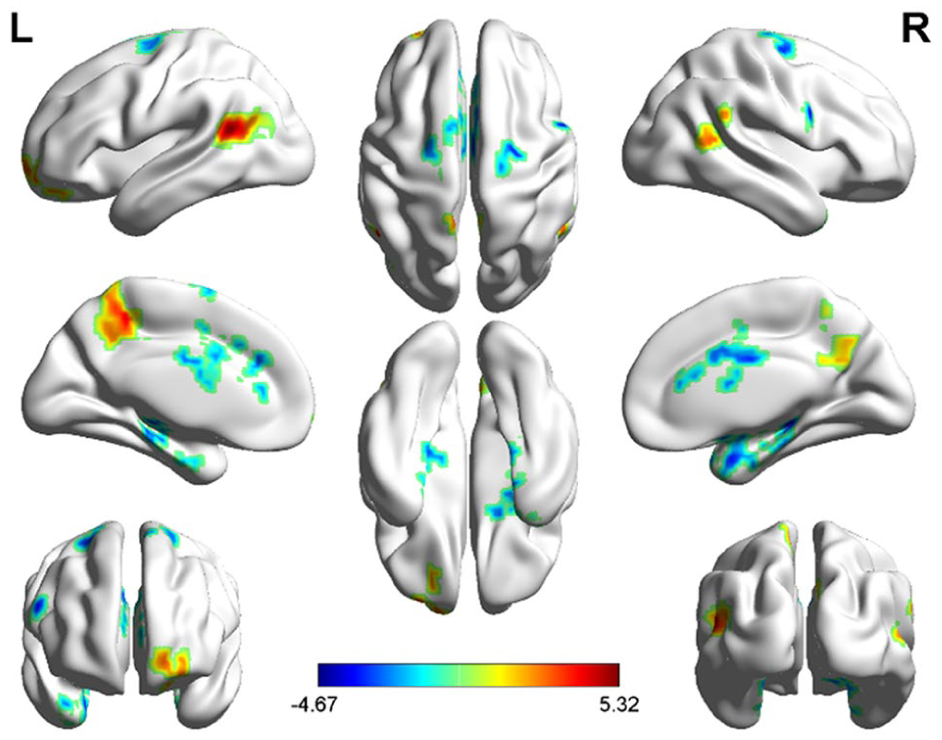
Brain regions showing altered FCs in FEP patients compared with healthy controls.
GABA, NAA and Glx changes in FEP patients
A significant difference in GABA+ concentration was found in the vmPFC between FEP patients and healthy controls (p = 0.046) (Figure 3(B)); however, there was no significant difference in NAA (p = 0.762) or Glx (p = 0.351) level between the two groups. The comparison of all metabolites concentrations is illustrated in Table 2. Full-width at half maximum, Cramer-Rao lower bounds, signal-to-noise ratios and tissue segmentation data for each group are presented in online supplementary table S1.
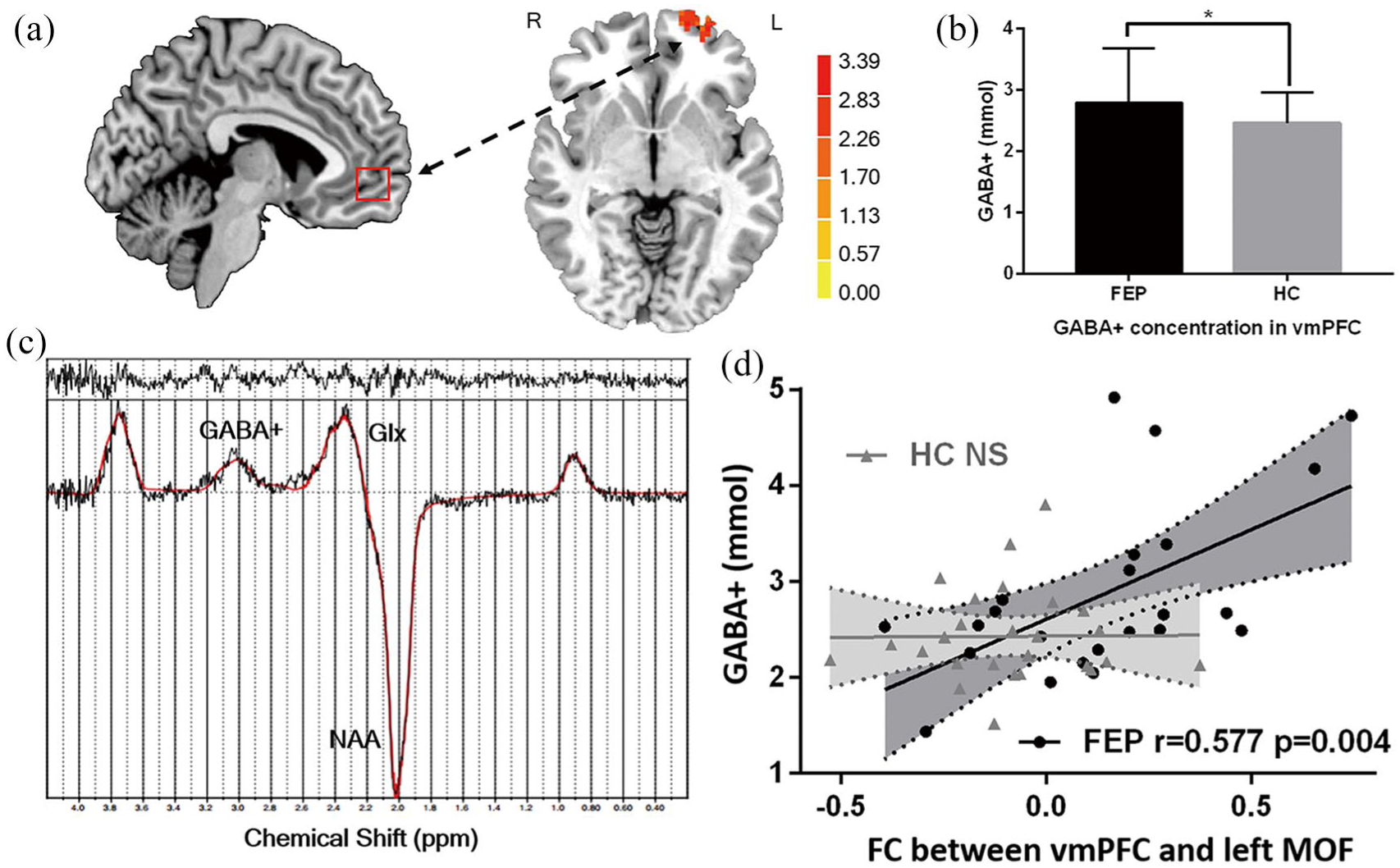
Increased ventromedial prefrontal cortex (vmPFC) to left middle orbital frontal (MOF) cortex functional connectivity (FC) was correlated with higher GABA+ concentration in first-episode psychosis (FEP) patients: (A) localization of vmPFC voxel for 1H-MRS data acquisition and the increased vmPFC-left MOF FC, (B) GABA+ level in vmPFC was significantly higher in FEP patients, and (C) representative 1H-MRS spectrum fitted by LCModel. The quantified metabolites are GABA+, Glx and NAA (GABA+: γ-aminobutyric acid, Glx: glutamine and glutamate, NAA: N-acetylaspartate) and (D) scatter plots of vmPFC-left MOF FC in correlation with GABA+ level in patients and healthy control groups, respectively.
Metabolite concentration in vmPFC between FEP patients and healthy controls.
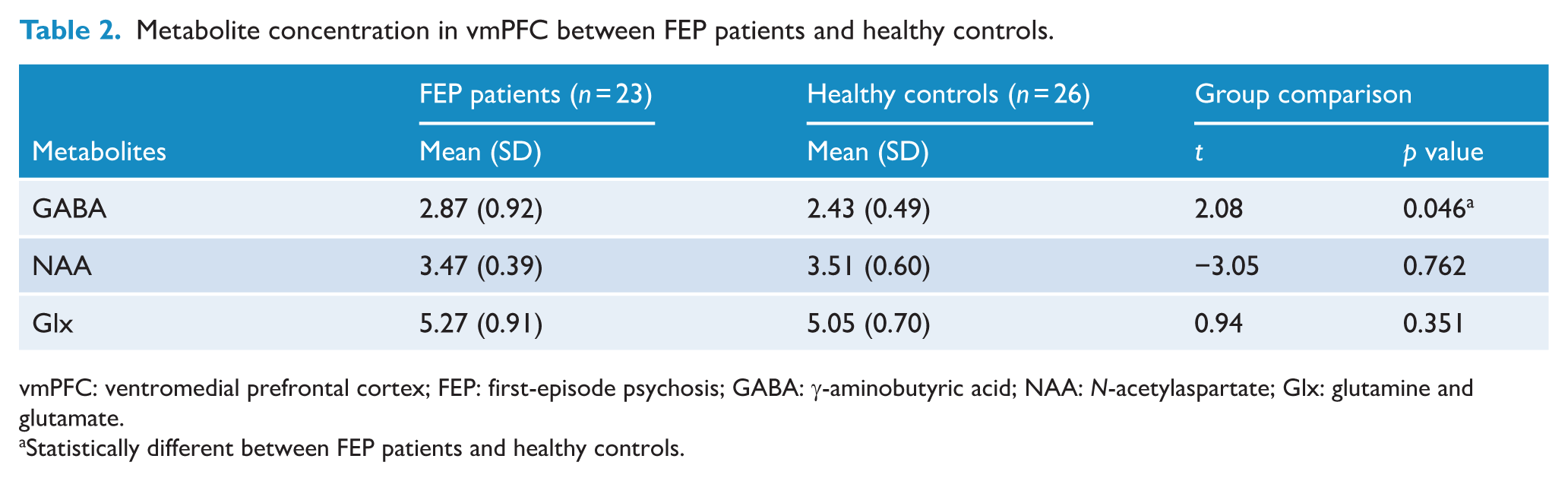
vmPFC: ventromedial prefrontal cortex; FEP: first-episode psychosis; GABA: γ-aminobutyric acid; NAA: N-acetylaspartate; Glx: glutamine and glutamate.
Statistically different between FEP patients and healthy controls.
Correlation between metabolic concentration in vmPFC and FC
In FEP patients, the GABA+ concentration in vmPFC was positively correlated with the FC between vmPFC and left MOF (r = 0.577, p = 0.004) (Figure 3(D)).
Relationship between altered FC, metabolite concentration and clinical symptomology in FEP patients
Duration of untreated psychosis (DUP) was positively correlated with FC between vmPFC and left SMA (r = 0.621, p = 0.002).
Moreover, NAA level in the vmPFC was positively correlated with positive symptoms (r = 0.477, p = 0.02) (Figure 4(A)), and the FC between vmPFC and left precuneus was negatively correlated with negative symptoms in FEP patients (r = −0.422, p = 0.045) (Figure 4(B)).
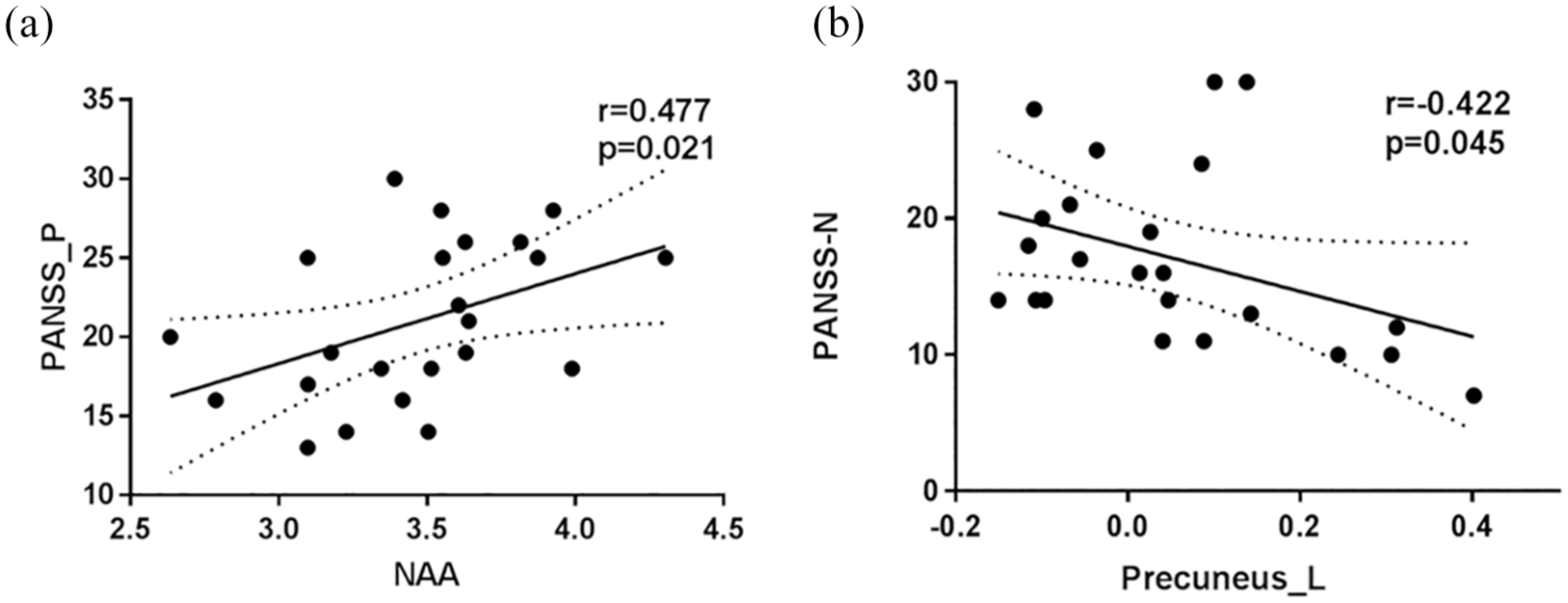
Correlation between metabolite concentration, FC and symptomology in FEP patients: (A) Spearman correlation analysis shows a positive relationship between NAA concentration and PANSS positive score and (B) a negative relationship between vmPFC-left precuneus FC and PANSS negative score.
Discussion
In this study, we used a combined fMRI-MRS methodology to investigate the neurochemical metabolite concentration in vmPFC and its association with brain functional activity in FEP patients. The main findings in this study are as follows: (1) GABA+ concentration in the vmPFC was significantly higher in FEP patients compared to healthy controls (2) and GABA+ level within vmPFC was positively correlated with its FC to the left MOF. To our knowledge, this is the first brain neurochemical–functional coupling study in first-episode, drug-naive SCZ patients. These findings indicate a putative mechanism between brain functional activity at network level modulated by regional neurochemical changes within the network. It may add new insights into the pathology of SCZ.
In our work, we observed higher GABA+ level in the vmPFC of drug-naïve FEP patients, which was consistent with previous studies (Kegeles et al., 2012; Yang et al., 2015). Decreased level of 67 kDa isoform of glutamic acid decarboxylase (GAD67), the enzyme responsible for most GABA synthesis, and reduced activity of the reuptake transporter of GABA have been found in the prefrontal cortex in SCZ patients (Hashimoto et al., 2008; Volk et al., 2001). Also, increased level of postsynaptic GABA-A receptor α2 subunit has been found across cortical regions (including the prefrontal cortex) in patients with SCZ (Volk et al., 2002). Moreover, increased GABA level in MPFC has been found in subjects at ultra-high risk of psychosis (De La Fuente-Sandoval et al., 2016). It is possible that, as a compensatory response of GABA synthesis deficiency, reduced GABA reuptake and upregulation of GABA-A receptors lead to excessive GABA accumulation. Our finding suggests the pathogenetic role of GABAergic dysfunction in vmPFC of FEP patients.
GABA is the main inhibitory neurotransmitter in the brain, and GABAergic dysfunction may lead to abnormal brain activity. Several lines of evidence have indicated that GABA concentrations in a variety of brain regions, including vmPFC (Jocham et al., 2012), amygdala (Levar et al., 2017), ACC (Northoff et al., 2007) and visual cortex (Muthukumaraswamy et al., 2009), predicted BOLD responses within the regions. Recent fMRI studies have shown that GABA level was inversely correlated with the strength of FC within motor and frontal-limbic networks (Delli Pizzi et al., 2017b; Levar et al., 2017; Stagg et al., 2014). It was suggested that higher GABA concentration in one region enhanced the inhibitory signaling within it (Delli Pizzi et al., 2017b). However, we found a positive association between GABA+ level in the vmPFC and FC to left MOF in FEP patients. This may suggest the disruption of inhibitory signaling in the vmPFC of FEP patients.
The N-methyl-
NAA is synthesized in neuronal mitochondria and associated with energy consumption in the brain. It is also the precursor for N-acetyl-aspartyl-glutamate (NAAG) that acts to regulate Glu and dopamine release (Moffett et al., 2007). Thus, NAA is not only a putative marker of neural integrity, but also a measure of neuronal metabolism and an indirect reflection of excitatory signaling in the brain. Reduction of cortical NAA concentration has consistently been found in chronic SCZ patients (Liemburg et al., 2016; Natsubori et al., 2014), while NAA level alteration in FEP patients remains inconclusive (Ohrmann et al., 2005). In this study, no significant difference of NAA concentration in vmPFC was found between FEP patients and healthy controls.
Compared with the healthy controls, FEP patients showed increased FCs between vmPFC and left middle temporal, left MOF and right middle temporal cortex as well as left precuneus. Lesion or abnormal functional activity in the MOF and MPFC has been correlated with reduced social and emotional functioning in SCZ (Shamay-Tsoory et al., 2009). Significantly reduced FCs between the vmPFC and left SMA, left dACC, right precentral gyrus, right superior frontal gyrus and right hippocampus were also shown in this study. The SMA, precentral gyrus and superior frontal gyrus are components of the premotor area in brain, which is engaged in motor planning and execution (Walther and Strik, 2012). Previous neuroimaging studies have found that deactivation in the SMA was associated with motor dysfunction in SCZ (Scheuerecker et al., 2009).
It is of great interest for clinicians to link neurochemical and functional activities in vmPFC with clinical characteristics of patients with SCZ. We found a negative correlation between vmPFC-left precuneus FC and negative symptom in this study. The vmPFC and left precuneus are both involved in emotional processing (Northoff et al., 2006), which is compromised in SCZ (Van Der Meer et al., 2010). The altered connectivity between vmPFC and left precuneus may underly the modulatory mechanism in emotional processing of SCZ patients. Moreover, the NAA concentration in vmPFC was positively correlated with positive symptoms in FEP patients in our work, in agreement with previous studies (Psomiades et al., 2018; Seese et al., 2011).
There are some limitations in our study. First, the correlation between FC value and metabolite level was not statistically significant after false discovery rate correction, which might be due to the small sample size. Second, MRS cannot differentiate intracellular, extracellular or vascular contributions to the measured GABA or Glx signals. A combination of pharmacological and neuroimaging studies may better unveil neurotransmission and synaptic abnormalities in SCZ. Moreover, we should interpret the GABA results with caution since we measured GABA+ which includes GABA and macromolecule signals (O’Gorman et al., 2011). In addition, the measured Glx level included both glutamate and glutamine signals, which was desirable if we could separate them out. Finally, the educational level was matched between groups in this study, which might be a confounding factor, since SCZ was associated with cognitive deficits (including educational performance) (Welham et al., 2008). However, it is the first neurochemical–functional coupling study in first-episode, drug-naïve SCZ patients, which provide new perspectives in the pathology of SCZ.
In this exploratory work, we performed a combined fMRI-MRS study, to investigate the relationship between GABA+ level and brain FC of first-episode, drug-naïve psychosis patients. We demonstrated a positive correlation between increased vmPFC GABA+ concentration and vmPFC-left MOF FC, indicating that inter-regional activity could be affected by local GABAergic metabolism in patients with FEP. Moreover, the aberrant FC and NAA concentration in vmPFC were correlated with the severity of schizophrenic symptoms of FEP patients. Our findings have provided new insights into the pathology of first-episode SCZ.
Supplemental Material
Supplemental_Materials – Supplemental material for Neurochemical and brain functional changes in the ventromedial prefrontal cortex of first-episode psychosis patients: A combined functional magnetic resonance imaging—proton magnetic resonance spectroscopy study
Supplemental material, Supplemental_Materials for Neurochemical and brain functional changes in the ventromedial prefrontal cortex of first-episode psychosis patients: A combined functional magnetic resonance imaging—proton magnetic resonance spectroscopy study by Haixin Cen, Jiale Xu, Zhilei Yang, Li Mei, Tianyi Chen, Kaiming Zhuo, Qiong Xiang, Zhenghua Song, Yingchan Wang, Xiaoyun Guo, Jinhong Wang, Kaida Jiang, Yifeng Xu, Yao Li and Dengtang Liu in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to acknowledge those patients and healthy controls taking part in this study and the support of the funding sources.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (81871047, 81871083), Key Program of Multidisciplinary Cross Research Foundation of Shanghai Jiao Tong University (YG2017ZD13), Key Program of SMHC Clinical Research Center (CRC2017ZD03), Shanghai Key Laboratory of Psychotic Disorders (13dz2260500) and Early Psychosis Program of Shanghai Mental Health Center (2013-YJTSZK-05).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
