Abstract
Objective:
The persistent disease burden of psychotic disorders often comes from negative symptoms; however, prognostic biomarkers for negative symptoms have not been fully understood. This study investigated whether the altered functional connectivity of the striatum predicts improvement in negative symptoms and functioning after 1 year of usual treatment in patients with first-episode psychosis.
Methods:
Resting-state functional magnetic imaging was obtained from 40 first-episode psychosis patients and 40 age- and sex-matched healthy control subjects. Whole-brain functional connectivity maps were generated with subdivisions of the striatum as seed regions and compared between first-episode psychosis patients and healthy controls. In 22 patients with first-episode psychosis, follow-up assessments of negative symptom severity and general functional status were conducted after 1 year of usual treatment. Multiple regression analyses were performed to examine factors predictive of symptomatic or functional improvements over the 1-year period.
Results:
First-episode psychosis patients showed greater functional connectivity between the left dorsal caudate and left primary motor cortex, as well as between the left ventral rostral putamen and right temporal occipital fusiform cortex, than healthy controls. Lower functional connectivity between the right dorsal rostral putamen and anterior cingulate cortex was observed in the first-episode psychosis patients than in healthy controls. In multiple regression analyses, lower functional connectivity of the left dorsal caudate–left primary motor cortex/right dorsal rostral putamen–anterior cingulate cortex predicted improvement in negative symptoms. In addition, lower right dorsal rostral putamen–anterior cingulate cortex functional connectivity predicted improvement in general functioning.
Conclusion:
These results suggest that altered striatal functional connectivity can be a potent neurobiological marker in the prognosis prediction of first-episode psychosis. Furthermore, altered striatal functional connectivity may provide a potential target in developing treatments for negative symptoms.
Keywords
Introduction
Schizophrenia is often described in terms of positive and negative symptoms (Andreasen, 1982). Positive symptoms such as hallucinations and delusions have been important in diagnosing a patient with schizophrenia (American Psychiatric Association, 2013), whereas negative symptoms have been emphasized in relation to long-term prognosis, including functional recovery (Buchanan, 2007; Galderisi et al., 2013; Remington et al., 2016). Negative symptoms consist of the following: diminished emotional expression, avolition, alogia, anhedonia and asociality (Foussias et al., 2014; Kirkpatrick et al., 2006). Because these symptoms are impairments of normal functioning, which are crucial for cognitive, interpersonal and occupational performance, improving negative symptoms is essential for better quality of life as well as long-term prognosis in patients with schizophrenia (Fenton and McGlashan, 1994; Kurtz et al., 2005).
Although antipsychotic medications, which are the mainstay of treatment for schizophrenia, are highly effective for positive symptoms, most antipsychotics rarely have an effect on negative symptoms, and even clozapine shows limited efficacy (Leucht et al., 2013). Novel pharmacological agents such as glycine-site modulators or neuromodulation techniques such as transcranial direct current stimulation (tDCS) and transcranial magnetic stimulation (TMS) have been investigated as alternative treatment strategies (Palm et al., 2016; Shi et al., 2014; Umbricht et al., 2014), but the trials to date were not successful enough for application in clinical practice (Bugarski-Kirola et al., 2016; Kantrowitz, 2017). One of the major hurdles in developing new treatment methods for negative symptoms is that the neural correlates of improvements in negative symptoms, which can be a target for the intervention, have not yet been clearly identified (Remington et al., 2016). Therefore, it is necessary to investigate neurobiological predictors of improvements in negative symptoms to develop a better treatment strategy and to achieve a favorable long-term prognosis in patients with schizophrenia.
The brain regions known to be related to negative symptoms are the prefrontal cortex, anterior cingulate cortex (ACC), sensorimotor or associative cortex, and striatum (Balleine et al., 2007; Goghari et al., 2010; Sigmundsson et al., 2001), which are associated with motivation, emotion and cognition. Among those regions, the striatum has been studied as a neurobiological region underlying psychotic symptoms, both positive and negative, especially in relationship to dopamine D2 receptors. In the dopamine hypothesis of schizophrenia, hypofunction of the frontostriatal pathway has been suggested as the cause of negative symptoms and accompanying cognitive impairments (Howes and Kapur, 2009). In line with the traditional hypothesis that emphasized the striatum, disrupted functional connectivity (FC) between the cortex and the striatum has been reported not only in patients with chronic schizophrenia but also in patients with first-episode psychosis (FEP) (Fornito et al., 2013; James et al., 2016; Peters et al., 2017). The altered corticostriatal connectivity was linked with negative symptoms in schizophrenia patients (Tu et al., 2012) and was also found in healthy individuals with high social anhedonia (Wang et al., 2016). These previous findings suggested that corticostriatal FC has a close relationship with negative symptoms, and this FC may be a potential neurobiological predictor of improvements in negative symptoms and functional status in patients with schizophrenia.
Despite the potential of corticostriatal FC to be a marker for predicting improvements in negative symptoms, studies to date have investigated corticostriatal FC as a marker for improvements only in positive symptoms (Sarpal et al., 2015, 2016). For example, Sarpal et al. (2015) reported that 12 weeks of antipsychotic treatment in drug-naïve FEP patients produced increased striatal FC to the hippocampus, dorsolateral prefrontal cortex and ACC in association with improvements in positive symptoms. Their subsequent study (Sarpal et al., 2016) showed that baseline striatal FC may serve as a predictor of improvements in positive symptoms after 12 weeks of antipsychotic treatment. These studies successfully showed the possibility of striatal FC as a marker for antipsychotic effects on positive symptoms during clinical trials; however, its role in predicting prognosis during a naturalistic course, which might be more affected by negative symptoms, needs further investigation. Because current treatments of schizophrenia have limited efficacy for negative symptoms, a comprehensive approach including medication, psychotherapy and rehabilitation should be provided for a sufficient length of time to observe improvements in negative symptoms and general functioning, unlike in clinical trials designed to observe a relatively quick effect of an antipsychotic medication on positive symptoms under highly controlled conditions.
Therefore, to investigate the neurobiological correlates of improvements in negative symptoms of schizophrenia patients, a naturalistic observational study with a sufficient observational period would be crucial. Furthermore, it was necessary to study patients with FEP whose negative symptoms have more potential for improvement than patients in a chronic state with steadfast negative symptoms. Therefore, in this naturalistic observational study, we aimed to investigate whether the altered corticostriatal FC would be predictive of improvements in negative symptoms and general functional status in patients with FEP after 1 year of usual treatment. We first hypothesized that patients with FEP would show altered striatal FC to various brain regions compared to healthy control (HC) subjects. In addition, we expected that altered striatal FC found in FEP patients would be a predictor of the 1-year prognosis reflected by improvements in negative symptoms and general functional status.
Materials and methods
Participants
In total, 40 patients with FEP and 40 age-, sex-, and handedness-matched HC subjects participated in this study and underwent a resting-state functional magnetic resonance imaging (fMRI) scan. Patients with FEP were recruited from an inpatient and outpatient clinic at Seoul National University Hospital. The inclusion criteria for patients with FEP were defined as individuals aged 16–40 years who satisfied the diagnosis of schizophreniform disorder, schizophrenia or schizoaffective disorder by an assessment using the Structural Clinical Interview for DSM-IV Axis I, with a duration of illness (DUI) less than 2 years. HCs were recruited via Internet advertisement and were screened using SCID-I Non-patient Edition (SCID-NP). HCs were excluded when they had any first- to third-degree biological relatives with a psychotic disorder. In all participants, the intelligent quotient (IQ) was measured using the Korean version of the Wechsler Adult Intelligence Scale (Yum et al., 1992). The exclusion criteria were neurological disease or significant head trauma, evidence of medical illness with documented cognitive sequelae, intellectual disability (IQ <70), severe personality disorders, substance abuse (except for nicotine) and cerebral pathology on MRI.
Symptomatic severity and functional status of the patients with FEP were examined at the time of enrollment and reassessed after 1 year of usual treatment using the Positive and Negative Syndrome Scale (PANSS) and Global Assessment of Functioning (GAF). Given the growing evidence for two subdomains of negative symptoms in schizophrenia, we calculated two scores to evaluate amotivation and expression deficits (Blanchard and Cohen, 2006). An amotivation score was calculated by summation of the following items from the PANSS: emotional withdrawal, passive/apathetic social withdrawal and active social avoidance. A diminished expression score was calculated by summing the following items from the PANSS: blunted affect, poor rapport, lack of spontaneity and flow of conversation, and motor retardation. Changes in symptomatic and functional status were calculated by subtracting the scores on the clinical scales measured at the 1-year follow-up point from the scores at the baseline assessment. Duration of untreated psychosis (DUP) was defined as the time from the emergence of psychotic symptoms to the first introduction of antipsychotic treatment. Medication use at baseline and during the follow-up period was documented by a thorough review of medical records. The dose of antipsychotics at the time of fMRI scanning was converted to olanzapine equivalent dose, and during 1 year, doses were calculated as the mean daily olanzapine equivalent dose (Gardner et al., 2010).
This study was approved by the Institutional Review Board of Seoul National University Hospital. All participants in this study understood the procedures in their entirety following a detailed explanation and provided written informed consent.
Image acquisition and data preprocessing
All MRI data were obtained on a 3T Trio MR scanner (Siemens Magnetom Trio, Erlangen, Germany) using a 12-channel head coil. We acquired T1-weighted anatomical images using magnetization-prepared rapid gradient echo (echo time [TE]/repetition time [TR] = 1.89/1670 ms, field of view [FOV] = 250 mm, flip angle = 9°, matrix = 256 × 256, voxel size = 1.0 × 1.0 × 1.0 mm3, 208 sagittal slices). The functional images were collected using a gradient echo-planar imaging (EPI) pulse sequence (TE/TR = 30/3500 ms, FOV = 240 mm, flip angle = 90°, matrix = 128 × 128, voxel size = 1.9 × 1.9 × 3.5 mm3, 35 axial slices, 116 volumes). Participants were instructed to relax with eyes closed but to not fall asleep. To ensure that they had not fallen asleep, a questionnaire was completed after the scan. To minimize possible motion artifacts, head cushions were used, and the subjects were asked to move as little as possible during the acquisition. Resting-state fMRI images were acquired for 6 minutes and 58 seconds. After image acquisition, all acquired images were visually inspected and confirmed by independent radiologists for any problems that could have occurred during image acquisition.
We used the CONN toolbox v17f (www.nitrc.org/projects/conn), implemented in the Statistical Parametric Mapping software package, version 12 (SPM12; www.fil.ion.ucl.ac.uk/spm; Wellcome Department of Cognitive Neurology, London, UK), for preprocessing the images. For gradient field stabilization, the first four volumes of the EPI data were removed. The remaining 112 contiguous EPI functional images were first processed by slice-timing correction and subsequently realigned to correct for head motions. We confirmed that no subjects exceeded the head motion criteria, i.e., translation >2.0 mm and rotation >2.0°, in any direction. The functional images were spatially normalized to the Montreal Neurological Institute (MNI) standardized space. The functional images were resampled to a 2 × 2 × 2 mm3 voxel dimension and spatially smoothed using an 8-mm full-width half-maximum (FWHM) isotropic Gaussian kernel. We removed confounding variables including six head motion parameters (with their first-order derivatives) and nuisance signals from the white matter and cerebrospinal fluid. In addition, we conducted linear detrending and temporal bandpass filtering (0.008 < f < 0.09 Hz).
FC analysis
To test the FC of subregions of the striatum, we used the seed-based methods described in a previous study (Di Martino et al., 2008). The spherical regions of interest (ROIs) with a 3-mm radius were defined bilaterally as the following: dorsal caudate (DC, MNI coordinates: ±13, 15, 9), ventral caudate (VC, ±10, 15, 0), ventral caudate/nucleus accumbens (VC/NA, ±9, 9, −8), dorsal rostral putamen (DRP, ±25, 8, 6), dorsal caudal putamen (DCP, ±28, 1, 3) and ventral rostral putamen (VRP, ±20, 12, −3).
For each participant, seed-based FC analysis was performed using Pearson’s bivariate correlation between the time series of each ROI pair and those of the remaining brain voxels. We transformed the correlation coefficients to a normal distribution by Fisher’s z transformation. Using a generalized linear model, one-way analysis of covariance (ANCOVA) with the baseline olanzapine equivalent dose of antipsychotics as a covariate was performed to reveal group differences in striatal FC across the FEP and HC groups. Clusters with significant group differences were defined by a cluster-extent threshold of the corrected false discovery rate (FDR) of p < 0.05, in addition to an uncorrected voxel-level threshold of p < 0.001. The minimum cluster size to define the clusters at the corrected threshold was 40 voxels. Then, we applied the Bonferroni-corrected value of p < 0.004 to address multiple comparison issues of the 12 seed regions.
Statistical analysis
The demographic and clinical characteristics of subjects were compared using independent t-tests or Welch’s t-test if the variances were not equal. A chi-square analysis or Fisher’s exact test was used for categorical data. To examine the predictive values of baseline striatal connectivity on changes in negative symptoms and functional status in FEP patients, we performed a multiple regression analysis using the backward elimination method. Anticipated predictive factors included demographic characteristics (age, sex, IQ and years of education), DUP, baseline amotivation, diminished expression or GAF scores, medication use for 1 year (mean olanzapine equivalent dose, antidepressant use, mood stabilizer use, anxiolytic use) and striatal FC that showed significant group differences. We also performed an exploratory analysis to determine whether striatal FC could predict positive and general symptom changes in patients with FEP in the same way as above.
Results
Demographic and clinical characteristics of the participants
Demographic and clinical characteristics at baseline and at the 1-year follow-up point of the participants are summarized in Table 1. Patients with FEP had lower education levels (t = −2.127, p = 0.037) and IQ scores (t = −4.166, p < 0.001) than HC subjects. The average DUP of patients with FEP was 4.5 ± 4.1 months, and the median was 3.0 months. The FEP patients had a median of 0.6 months and a mean 1.7 ± 2.3 months of exposure to antipsychotic medication prior to the fMRI scan. The olanzapine equivalent dose of antipsychotics before the fMRI scan was 6.9 ± 6.7 mg/day.
Demographic and clinical characteristics of patients with first-episode psychosis (FEP) at baseline and after 1 year of follow-up and healthy control (HC) subjects.
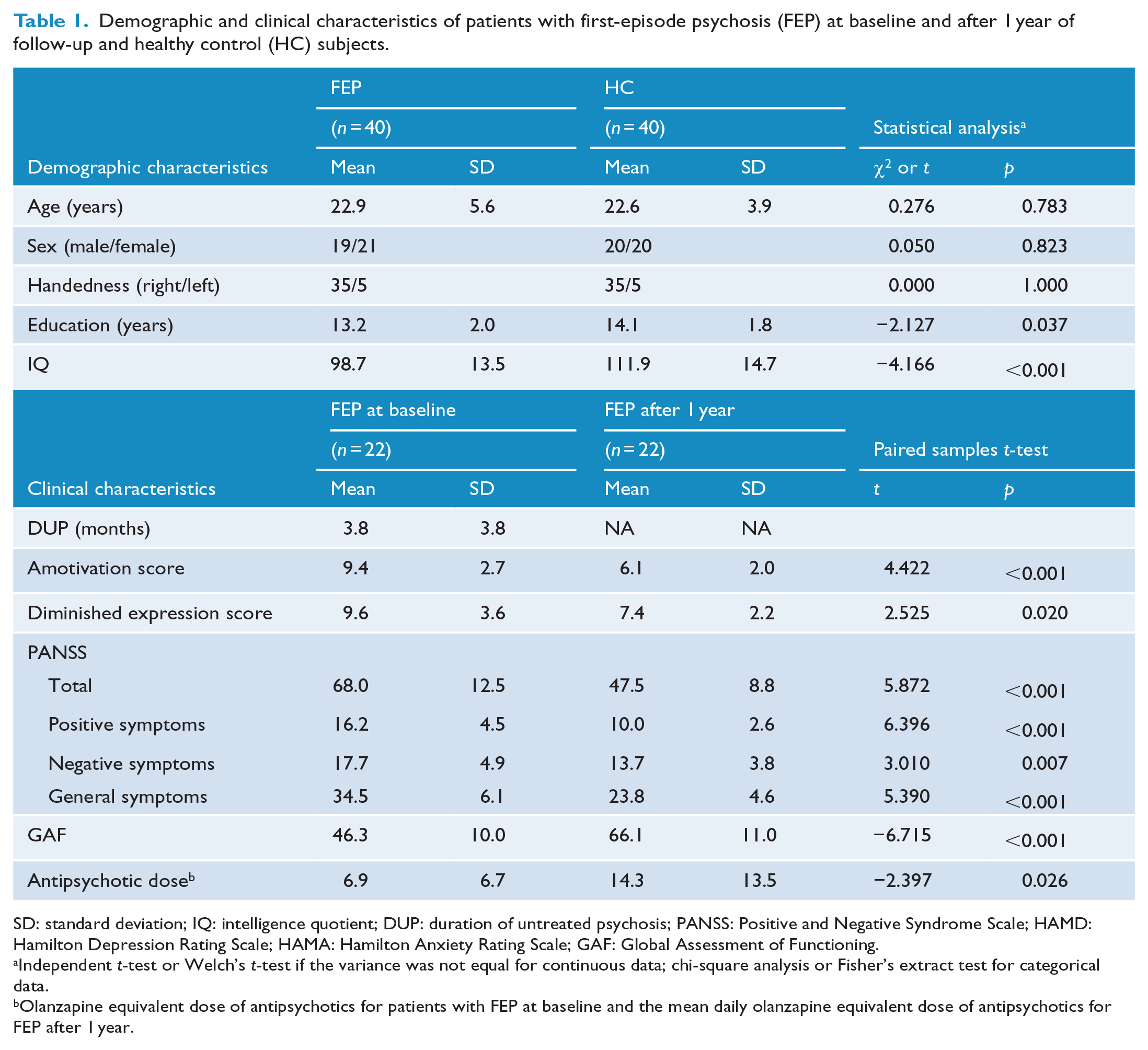
SD: standard deviation; IQ: intelligence quotient; DUP: duration of untreated psychosis; PANSS: Positive and Negative Syndrome Scale; HAMD: Hamilton Depression Rating Scale; HAMA: Hamilton Anxiety Rating Scale; GAF: Global Assessment of Functioning.
Independent t-test or Welch’s t-test if the variance was not equal for continuous data; chi-square analysis or Fisher’s extract test for categorical data.
Olanzapine equivalent dose of antipsychotics for patients with FEP at baseline and the mean daily olanzapine equivalent dose of antipsychotics for FEP after 1 year.
Among the 40 FEP participants, 22 patients completed 1-year follow-up clinical assessments. A comparison of FEP patients who completed the 1-year follow-up assessment and those who did not complete did not show any significant differences in demographic and clinical characteristics at baseline (Table S1 in the Supplementary Material).
Striatal FC results
The results of the FC analysis are summarized in Table 2. Significant group differences were observed in the left DC, right DRP and left VRP seed-to-brain networks. The FEP group exhibited greater FC between the left DC and left primary motor cortex (PMC) as well as between the left VRP and right temporal occipital fusiform cortex than the HC group. In contrast, lower FC between the right DRP and bilateral ACC was observed in the FEP group than in the HC group (Figure 1). There was no significant cluster showing a difference in striatal FC between the FEP patients who completed the 1-year follow-up assessment and those who did not.
Brain regions showing significantly different baseline connectivity with striatal seeds between first-episode psychosis (FEP) and healthy control (HC) groups.
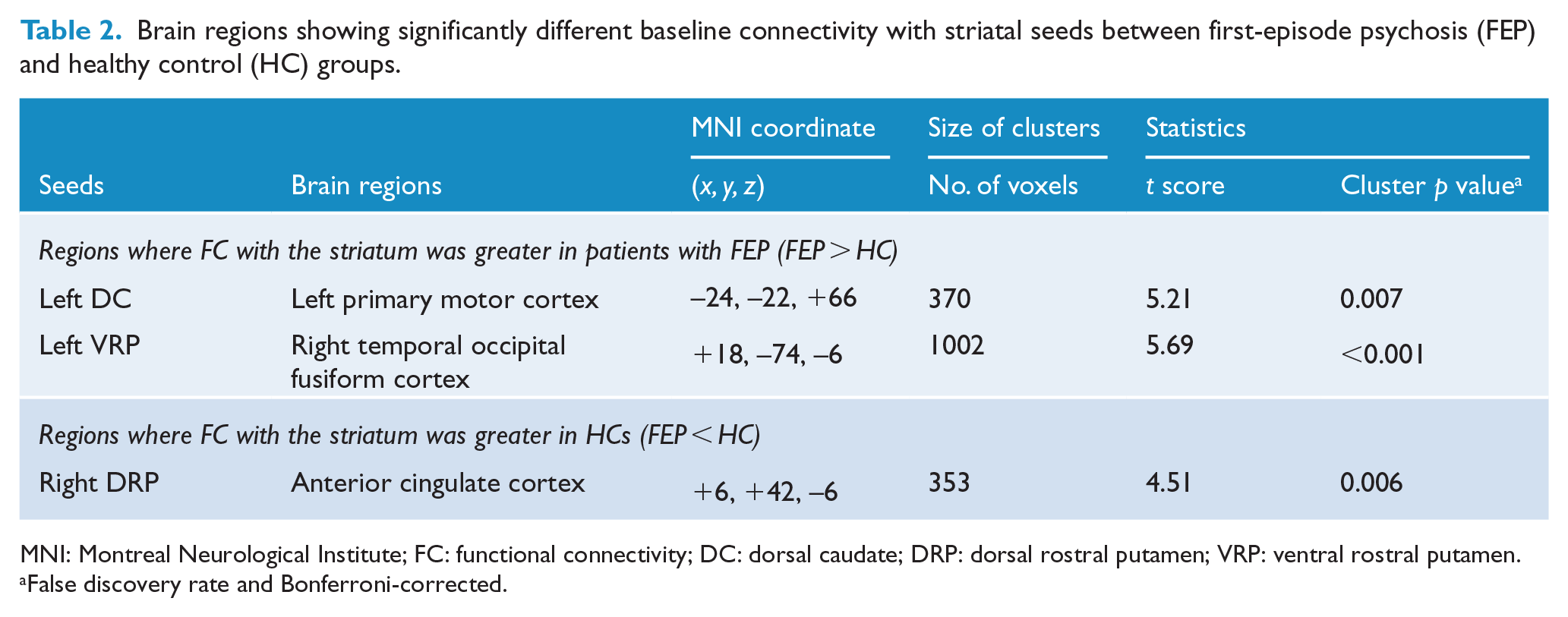
MNI: Montreal Neurological Institute; FC: functional connectivity; DC: dorsal caudate; DRP: dorsal rostral putamen; VRP: ventral rostral putamen.
False discovery rate and Bonferroni-corrected.
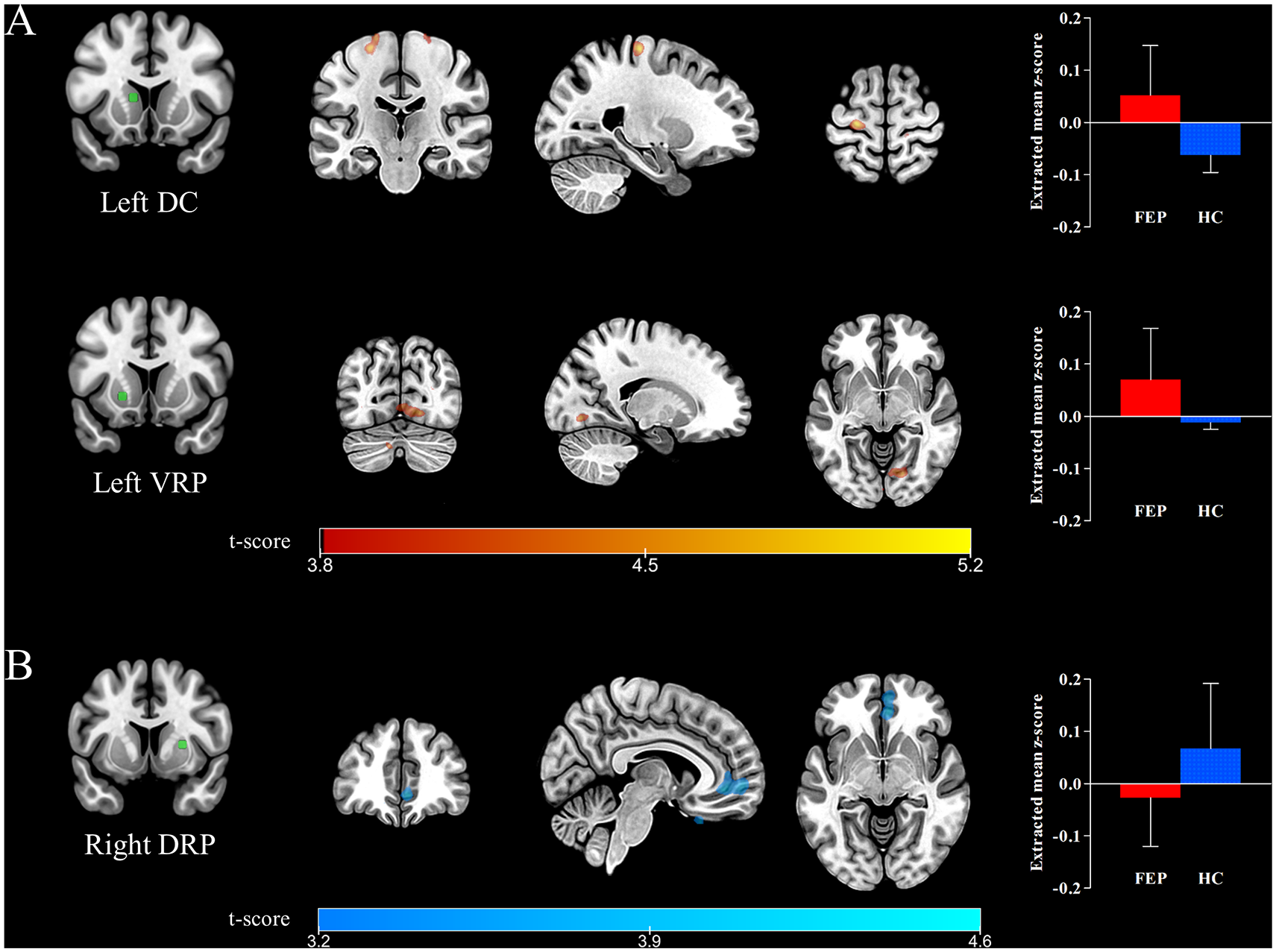
Statistical maps showing significant between-group differences in corticostriatal functional connectivity (FC) of the striatal seeds. (A) The regions where FC with the left dorsal caudate (DC) and ventral rostral putamen (VRP) was greater in patients with first-episode psychosis (FEP) than in healthy controls (HC) are marked in red-yellow and include the left primary motor cortex and right temporal occipital fusiform cortex, respectively. (B) The region where FC with the right dorsal rostral putamen (DRP) was lower in patients with FEP than in HCs is marked in blue-green and contains the anterior cingulate cortex. All results are displayed at a false discovery rate (FDR)-corrected threshold of p < 0.05. The left hemisphere is depicted on the left in the axial and coronal slices.
Multiple regression analysis
In the multiple regression analyses, improvements in amotivation scores were significantly predicted by lower FC between the right DRP and ACC at baseline (β = −11.988, 95% confidence interval [95% CI] = −21.650 to −2.326, p = 0.019), younger age (β = −0.358, 95% CI = −0.656 to −0.060, p = 0.022), not using antidepressants (β = −2.374, 95% CI = −4.397 to −0.350, p = 0.025) and higher amotivation scores at baseline (β = 1.273, 95% CI = 0.943 to 1.603, p < 0.001). Improvements in diminished expression scores were also predicted by lower left DC-PMC FC (β = −16.671, 95% CI = −25.631 to −7.711, p = 0.001), lower right DRP-ACC FC (β = −10.783, 95% CI = −19.016 to −2.550, p = 0.014), higher diminished expression scores at baseline (β = 0.962, 95% CI = 0.732 to 1.192, p < 0.001) and shorter DUP (β = −0.344, 95% CI = −0.581 to −0.107, p = 0.008). Lower FC between the right DRP and ACC (β = −75.953, 95% CI = −131.888 to −20.017, p = 0.011), not using antidepressants (β = −14.354, 95% CI = −25.223 to −20.017, p = 0.013) and lower GAF scores at baseline (β = −1.344, 95% CI = −1.903 to −0.784, p < 0.001) predicted improvements in general functional status after a 1-year follow-up period (Table 3 and Figure 2). In addition, the exploratory analysis showed that positive symptoms were predicted by striatal FC, but general symptoms were not (Table S2 in the Supplementary Material).
Multiple regression models for negative symptoms and functional improvements during 1 year of follow-up.
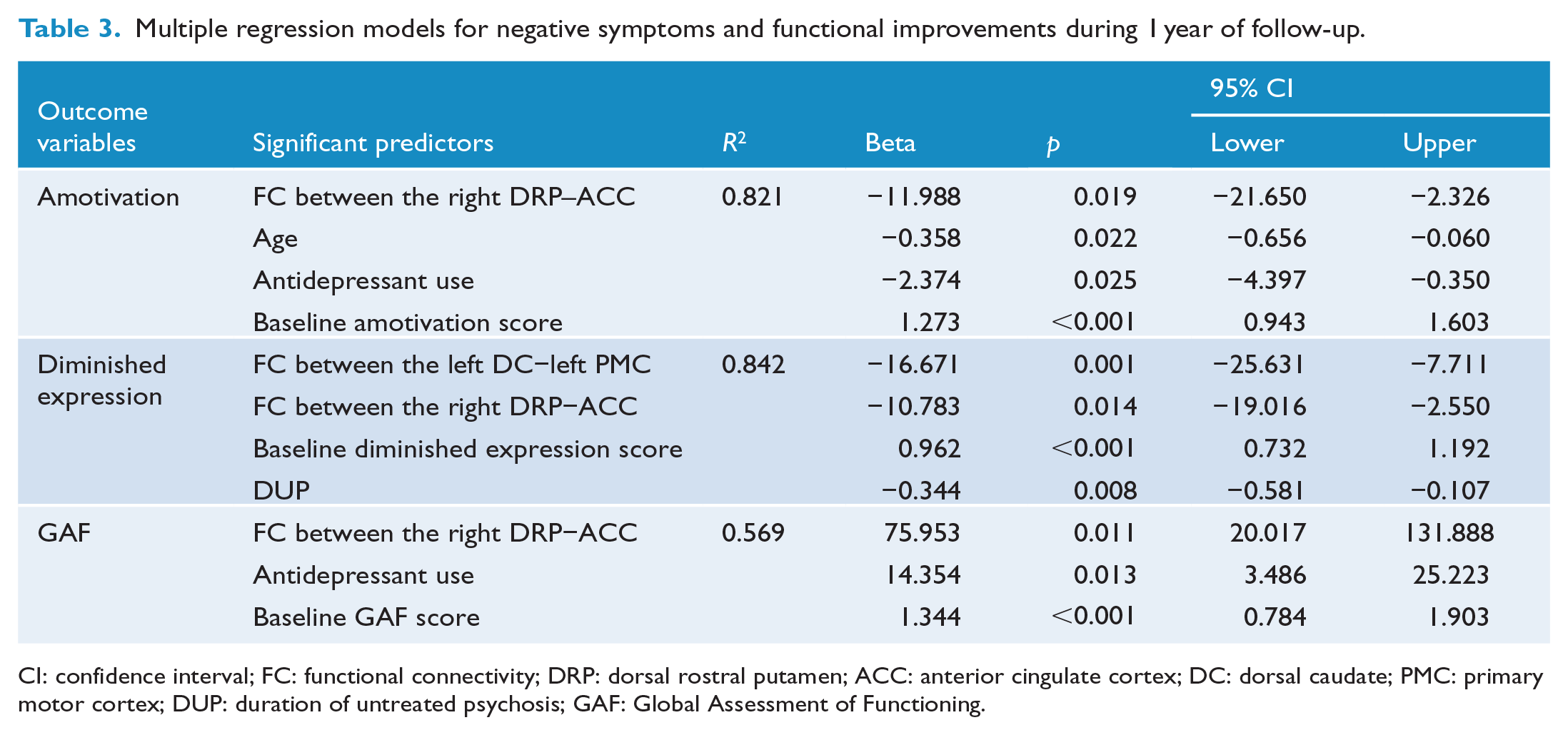
CI: confidence interval; FC: functional connectivity; DRP: dorsal rostral putamen; ACC: anterior cingulate cortex; DC: dorsal caudate; PMC: primary motor cortex; DUP: duration of untreated psychosis; GAF: Global Assessment of Functioning.
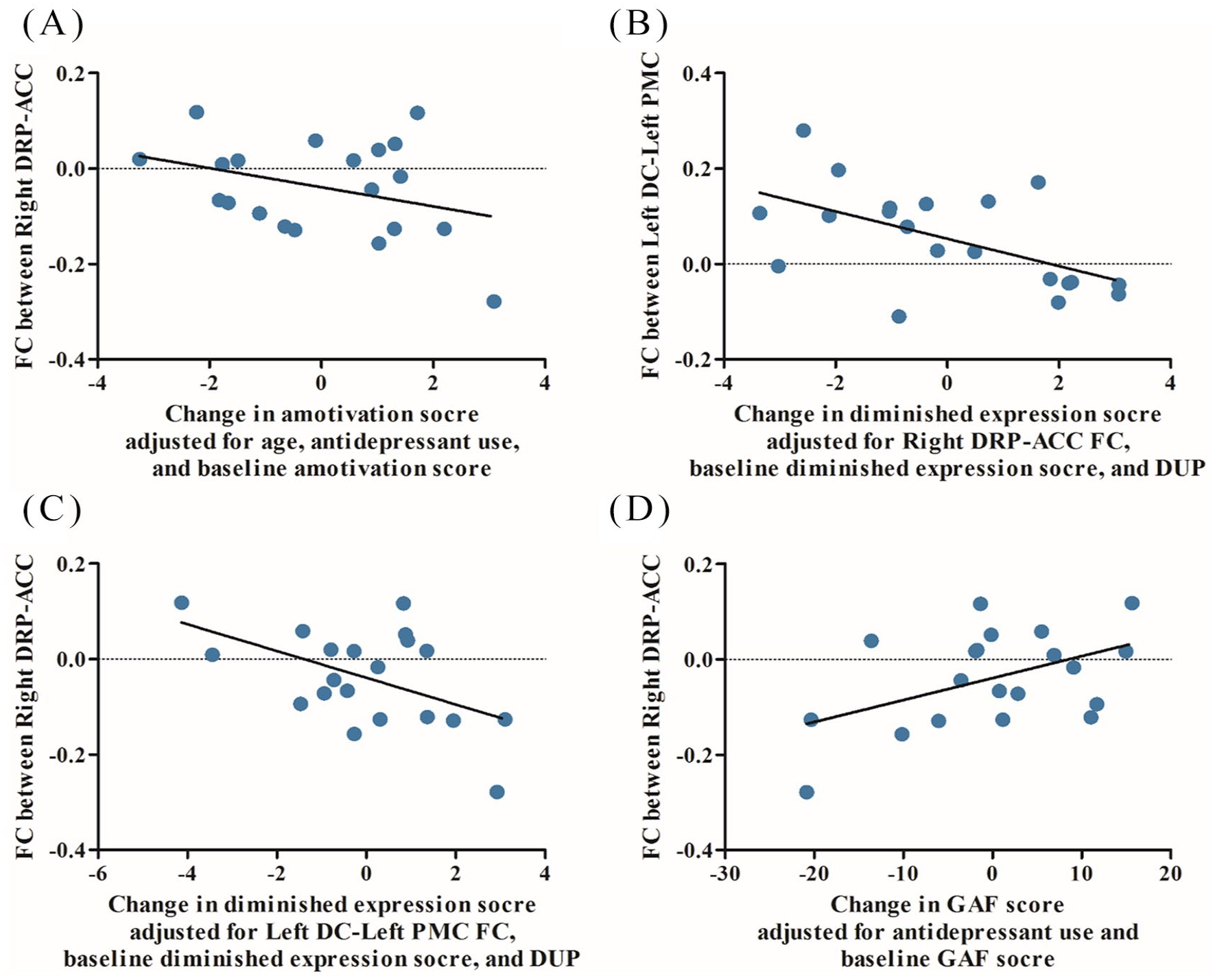
The correlation between baseline striatal functional connectivity (FC) and change in scores on (A) amotivation, (B and C) diminished expression and (D) Global Assessment of Functioning (GAF) adjusted for other significant predictors obtained from each multiple regression model.
Discussion
We investigated whether the altered baseline corticostriatal FC predicted the prognosis of FEP patients after 1 year of usual treatment, including improvements in negative symptoms and recovery of general functioning. Greater FC was observed between the left DC and left PMC and between the left VRP and right temporal occipital fusiform cortex in patients with FEP than in HCs. Conversely, FC between the right DRP and ACC was found to be lower in the FEP group than in the HC group. The right DRP–ACC FC and left DC–left PMC FC had predictive value for improvements in negative symptoms after 1 year. Improvements in general functional status were also predicted by a lower baseline FC between the right DRP and ACC. Taken together, our results suggest that alterations in corticostriatal FC are evident in FEP patients and could be a biomarker for predicting prognosis after 1 year from the beginning of treatment.
As hypothesized, altered patterns of striatal FC to various brain regions were found in the FEP group compared to those in the HC group. We found that the patients with FEP showed lower FC between the right DRP and ACC than the HCs. This result is in accordance with Yan et al. (2012), who also reported reduced connectivity between the striatum and ACC in patients with schizophrenia (Yan et al., 2012). In addition, a large body of evidence has linked the ACC with negative symptoms, especially symptoms of amotivation and apathy (Tekin and Cummings, 2002). Therefore, striatum–ACC connectivity may serve as a neurobiological underpinning of negative symptoms in patients with schizophrenia. Recently, several studies have shown altered striatal connectivity to various brain areas comprising the prefrontal cortex in patients with schizophrenia (Peters et al., 2017), their healthy relatives (Fornito et al., 2013) and subjects who have an at-risk mental state for psychosis (Dandash et al., 2014; Hubl et al., 2018). Our results extend the findings of those previous studies and give support to the traditional dopamine hypothesis for negative symptoms and the idea that a disrupted frontostriatal circuit plays an important role in the pathophysiology of schizophrenia; thus, frontostriatal FC may act as a potential marker for predicting prognosis in FEP patients.
Another interesting finding was that patients showed a greater connectivity between the left DC and left PMC than HCs. The connectivity between the dorsal striatum and sensorimotor cortex is important for action–reward association (Balleine et al., 2007), which has been suggested as the pathophysiological mechanism of motivational deficits in schizophrenia (Stepien et al., 2018). Wang et al. (2016) also reported hyperconnectivity between the dorsal striatum and motor cortex in individuals with high social anhedonia. These findings, including our own, suggest that altered FC between the striatum and sensorimotor cortex may be related to negative symptoms. In addition, we found greater FC between the VRP and temporal occipital fusiform cortex in patients with FEP than in HCs. The fusiform gyrus was involved in the perception of emotions in facial stimuli in schizophrenia (Radua et al., 2010) and was associated with ventral striatal activation in the implicit salience attribution task (Esslinger et al., 2012). The putamen also plays an important role in schizophrenia and psychosis as one of the components comprising the salience network (Farid and Mahadun, 2009; Kapur, 2003; Peters et al., 2016). Considering these early findings, our result of greater FC between the ventral putamen and fusiform cortex in FEP is not surprising, although this result has not been previously reported and thus needs to be interpreted with caution.
Along with our initial hypothesis, altered corticostriatal FC in patients with FEP measured at baseline predicted later improvements in negative symptoms and general functioning after 1 year of usual treatment. Specifically, DRP–ACC FC was predictive of improvements in both negative symptoms and general functioning. This finding suggests that frontostriatal FC may be the most promising biological predictor of prognosis in FEP patients, as it is associated with improvements in both negative symptoms and functional recovery. Improvements in amotivation scores were predicted by FC between the right DRP and ACC. In line with this finding, it has been reported that ACC activation is involved in the reduced motivation associated with impairments in effort computation in patients with schizophrenia (Lee et al., 2015). Meanwhile, improvements in diminished expression scores were predicted by FC between the left DC and left PMC. In accordance with this finding, previous studies reported that the DC and PMC were anatomically and functionally connected with each other (Balleine et al., 2007; Wang et al., 2016) and associated with negative symptoms of schizophrenia (Mucci et al., 2015; Potkin et al., 2002; Stepien et al., 2018). In addition, it was reported that the severity of affective flattening was associated with the aberrant mirror neuron system, which includes the motor cortex (Lee et al., 2014). One notable finding is that not using antidepressants predicted improvements in amotivation scores and diminished expression scores. Not using antidepressants might indicate that significant depressive symptoms were not present. Considering that previous studies reported concurrent depressive symptoms in schizophrenia as a poor prognostic indicator of recovery (Conley et al., 2007), our multiple regression analysis results may indicate that patients without significant depressive symptoms who were not using antidepressants had positive outcomes. In a current situation where there are no reliable biological measures to predict the prognosis of FEP patients (Tarcijonas and Sarpal, 2019), our results provide a potential resting-state fMRI biomarker that predicts improvements in negative symptoms and general functioning in patients during the earliest stages of psychosis.
Strengths of the current study included the observation of naturalistic changes in negative symptoms and functioning during a relatively long period of usual treatment, which was the most suitable study design to predict long-term prognosis; previous studies predicting the response to antipsychotic medications using striatal FC had a short-term observational period under highly controlled conditions (Lui et al., 2010; Sarpal et al., 2016). Although Sarpal et al. (2016) provided a valuable idea to investigate striatal FC for predicting improvements in positive symptoms, they did not report whether the striatal FC could predict a change in negative symptoms or general functioning, which is indeed important for the patient’s long-term prognosis (Birchwood et al., 1998; Harrison et al., 2001). This might have been due to the study design of Sarpal et al. (2015), which was a randomized controlled clinical trial to investigate the effects of antipsychotic medications on positive symptoms; it is not an ideal design to reveal changes in negative symptoms and functional status. This study aimed to compensate for those points by using a naturalistic cohort study design, and the results indicated that striatal FC can be a prognostic marker to predict improvements in negative symptoms and functional status. From the clinical utility perspective, the role of striatal FC as a biomarker may help clinicians make better decisions about which patients need to get more clinical attention. Our results implicate that patients with altered striatal FC may be expected to have a poor prognosis; therefore, intensive care and attention may be needed from the early course of the disease to improve negative symptoms. We expect that this finding would contribute to the development of new treatment methods targeting negative symptoms to improve long-term prognosis beyond antipsychotic medication.
This study had several limitations. First, almost all patients who participated in our study were taking antipsychotic medications. Because the striatum contains a large amount of dopamine D2 receptors and is the primary target of antipsychotics, antipsychotic medications may have affected striatal FC of the FEP patients measured at baseline (Lui et al., 2010; Sambataro et al., 2010). Although one previous study showed that striatal FC was not affected by antipsychotics in patients (Fornito et al., 2013), and we tried to control the medication effect as a covariate in the statistical analysis including group comparisons, cautious interpretation is warranted. Regarding the effect of medication, we also controlled the dose of antipsychotics taken for 1 year with one of the independent variables. However, the influence of almost doubling the antipsychotics dose during follow-up could not be completely ruled out. Second, the number of FEP patients who refused to participate in the follow-up assessment at 1 year was relatively large. However, there were no significant differences in demographic and clinical characteristics or striatal FC at baseline between the FEP patients who participated in the follow-up assessment and those who did not. Third, we do not have an independent validation set for the role of striatal FC as a biomarker. A future replication study with a large sample size is mandatory to confirm the current findings. Fourth, we could not acquire the resting-state fMRI in patients with FEP after 1 year of follow-up. The relationship between the change in striatal FC, which has been proposed as a predictor of improvement in negative symptoms and functioning in our current study, and symptomatic improvement could be further supported by a future study with longitudinal imaging data. Finally, although the 1-year follow-up period is long relative to previous studies to predict prognosis in FEP patients, it is also a relatively short time period to determine the role of striatal FC as a long-term prognostic predictor. Therefore, future studies with a longer follow-up period are needed to provide more solid evidence to support the results of the current study.
In conclusion, we found that baseline corticostriatal FC can be a potential biomarker for predicting improvements in negative symptoms and general functioning after 1 year of usual treatment in patients with FEP. Although our study results warrant verification through a future multicenter large-scale cohort study due to limited sample size, the current study provides the first evidence to predict the 1-year prognosis of FEP patients using corticostriatal FC in a naturalistic cohort study design. Corticostriatal FC may be helpful not only in predicting the prognosis of schizophrenia but also in developing new targeted treatment methods for improving negative symptoms.
Supplemental Material
R1_supplementary_FEP_striatalFC – Supplemental material for Resting-state functional connectivity of the striatum predicts improvement in negative symptoms and general functioning in patients with first-episode psychosis: A 1-year naturalistic follow-up study
Supplemental material, R1_supplementary_FEP_striatalFC for Resting-state functional connectivity of the striatum predicts improvement in negative symptoms and general functioning in patients with first-episode psychosis: A 1-year naturalistic follow-up study by Sanghoon Oh, Minah Kim, Taekwan Kim, Tae Young Lee and Jun Soo Kwon in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Brain Research Program and the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT & Future Planning (Grant Nos. 2017M3C7A1029610, 2019R1A2B5B03100844, and 2019R1C1C1002457).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
