Abstract
Introduction:
Schizophrenia is increasingly conceived as a disorder of brain network connectivity and organization. However, reports of network abnormalities during the early illness stage of psychosis are mixed. This study adopted a data-driven whole-brain approach to investigate functional connectivity and network architecture in a first-episode psychosis cohort relative to healthy controls and whether functional network properties changed abnormally over a 12-month period in first-episode psychosis.
Methods:
Resting-state functional connectivity was performed at two time points. At baseline, 29 first-episode psychosis individuals and 30 healthy controls were assessed, and at 12 months, 14 first-episode psychosis individuals and 20 healthy controls completed follow-up. Whole-brain resting-state functional connectivity networks were mapped for each individual and analyzed using graph theory to investigate whether network abnormalities associated with first-episode psychosis were evident and whether functional network properties changed abnormally over 12 months relative to controls.
Results:
This study found no evidence of abnormal resting-state functional connectivity or topology in first-episode psychosis individuals relative to healthy controls at baseline or at 12-months follow-up. Furthermore, longitudinal changes in network properties over a 12-month period did not significantly differ between first-episode psychosis individuals and healthy control. Network measures did not significantly correlate with symptomatology, duration of illness or antipsychotic medication.
Conclusions:
This is the first study to show unaffected resting-state functional connectivity and topology in the early psychosis stage of illness. In light of previous literature, this suggests that a subgroup of first-episode psychosis individuals who have a neurotypical resting-state functional connectivity and topology may exist. Our preliminary longitudinal analyses indicate that there also does not appear to be deterioration in these network properties over a 12-month period. Future research in a larger sample is necessary to confirm our longitudinal findings.
Introduction
The disconnection hypothesis of schizophrenia suggests that dysfunctional integration and communication among brain regions is central to illness pathology (Friston and Frith, 1995), a notion that continues to gain support across the psychosis field. Resting-state functional magnetic resonance imaging (rs-fMRI) studies have identified abnormal functional connectivity (rs-FC) at various stages of psychosis. The majority of past research has been conducted in established schizophrenia patient cohorts and, despite somewhat mixed results, the prevailing findings support decreased rs-FC in patients, primarily involving frontal and temporal brain regions (for review, see Yu et al. (2012)). However, fewer studies have examined the nature of rs-FC disturbances during the first-episode illness stage of psychosis.
In individuals experiencing first-episode psychosis (FEP), reports of abnormal rs-FC are divergent, partially due to the widely varied methods and lack of a data-driven approach across research groups (Greicius, 2008). Only four studies have conducted whole-brain analyses of rs-FC networks in FEP and reported widespread FC reductions (Argyelan et al., 2015), reduced inter-hemispheric rs-FC (Guo et al., 2014, 2017), and both increased and decreased rs-FC between various frontal, temporal and subcortical regions in FEP groups relative to controls (Li et al., 2016b). FEP studies that have constrained the investigation of rs-FC to hypothesized region(s) of interest (ROI) vary greatly in the specified ROI and, unsurprisingly, have reported diverse results of both increased rs-FC (Cui et al., 2015; Fornito et al., 2013; Gong et al., 2016; Guo et al., 2015; Li et al., 2016b) and decreased rs-FC (Cui et al., 2015; Fornito et al., 2013; Gong et al., 2016; Guo et al., 2014, 2015; Li et al., 2016b) in FEP participants relative to controls.
In addition to the employment of widely varying methodologies to map rs-FC, it is possible that the heterogeneity of findings at the earlier stages of illness reflect differing stages of progressive structural brain changes on a background of dynamic maturational brain changes (Cropley and Pantelis, 2014; Pantelis et al., 2009). In light of the highly varied rs-FC literature, it appears that the classic approaches of characterizing rs-FC cross-sectionally are not sensitive or specific enough to elucidate network-level abnormalities that might underpin the onset of psychosis. Thus, mapping brain function more extensively and longitudinally presents an important way forward.
In addition to employing a longitudinal design, another way to enhance our understanding of the neurobiological functional differences and changes that are present during the FEP illness phase, and which may help explain the inconsistent rs-FC findings, is by broadening our scope to explore other features of connectivity using graph theory. Recently, in conjunction with measures of FC, graph-theoretical methods have been applied to fMRI data in an attempt to understand the topology and efficiency of brain networks. The advantage of such an approach is that it goes beyond connectivity analyses and facilitates the exploration of complex networks and metrics, such as efficiency of information transfer on whole-brain and regional scales (Rubinov and Sporns, 2010). Previous studies have reported abnormal structural (Fornito et al., 2012; Zalesky et al., 2011) and functional brain network topology in established schizophrenia (Hadley et al., 2016; Lo et al., 2015; Lynall et al., 2010); however, few studies have used graph theory to understand network organization in FEP. While structural network topology has been found to be abnormal in early illness stages (Crossley et al., 2017; Hu et al., 2016; Palaniyappan et al., 2016; Zhang et al., 2015), to our knowledge, no study has examined functional network topology during resting state in FEP longitudinally. Investigating functional brain networks in psychosis using graph-theoretical methods may better elucidate the interplay between network arrangement and communication during the early stages of the disorder and how network properties may change over time.
To bring clarity to the disparate research on rs-FC in FEP we adopted a novel, complementary data-driven approach focusing on FC and well-established graph metrics to characterize both local and global brain network properties in FEP individuals. We mapped resting-state functional brain networks in FEP individuals and healthy controls at baseline and at 12 months follow-up to address the following three aims: (1) to investigate whether rs-FC and topological descriptors of functional brain networks significantly differed between FEP individuals and healthy age- and sex-matched controls; (2) to test whether any longitudinal rs-FC changes in FEP individuals between baseline and 12 month follow-up were more pronounced than expected due to healthy development and (3) to investigate whether any longitudinal changes in rs-FC correlated with measures of clinical characteristics. We hypothesized that FEP participants would show reduced rs-FC and reduced network efficiency relative to controls at both time points and that network properties would change abnormally over a 12-month period.
Methods
Participants
A total of 29 FEP participants (mean age = 19.5 ± 2.3 years, 22 males) were recruited through Orygen Youth Health (OYH) clinical sites in Victoria, Australia. Inclusion criteria for FEP participants consisted of meeting the
General exclusion criteria for all participants included any history of significant head injury, previous or current history of neurological disease, the presence of standard magnetic resonance imaging (MRI) contraindications (e.g. metal implants, claustrophobia, current pregnancy or breastfeeding), premorbid IQ < 70 and a documented history of developmental delay or intellectual disability. Clinical symptoms were assessed using the Brief Psychiatric Rating Scale (BPRS; Andersen et al., 1989), the Scale for the Assessment of Negative Symptoms (SANS; Andreasen, 1989) and the Montgomery–Åsberg Depression Rating Scale (MADRS; Montgomery and Åsberg, 1979). In addition, all participants were evaluated using the Social and Occupational Functioning Assessment Scale (SOFAS; Goldman et al., 1992). Due to participants who withdrew or did not complete follow-up assessments, 14 FEP and 20 control participants had 12-month imaging and clinical data available. The study was approved by the Melbourne Health Human Research Ethics Committee (MHREC ID 2012.069); and all participants provided written informed consent prior to participation.
Imaging data acquisition
Magnetic resonance images were acquired on a Siemens Avanto 3T Magnetom Trio Tim scanner. T1-weighted images were acquired using an optimized Magnetization-Prepared Rapid Acquisition Gradient Echo (MPRAGE) sequence. The sagittal imaging parameters were 176 sagittal slices of 1 mm thickness without gap, field of view (FOV) = 250 × 250 mm2, repetition time (TR) = 1980 ms, echo time (TE) = 4.3 ms, flip angle = 15°, using an acquisition matrix of 256 × 256 resulting in a final reconstructed voxel resolution of 0.98 × 0.98 × 1.0 mm3. Resting-state fMRI was acquired using T2*-weighted echo-planar imaging (TE = 40 ms; TR = 2.4 s; voxel dimensions = 3.3 × 3.3 × 3; matrix size = 64 × 64). Resting-state fMRI data were acquired for 8 minutes, resulting in 200 volumes. During the acquisition, participants were asked to close their eyes and let their minds wander without going to sleep.
fMRI data preprocessing
Data preprocessing was performed using FSL (FMRIB Software Library, www.fmrib.ox.ac.uk) and SPM8 (www.fil.ion.ucl.ac.uk/spm). For each subject, echo-planar images were slice-time corrected, realigned to the mean functional image to correct for motion, co-registered to their structural T1-weighted scan via rigid-body registration and then spatially normalized by non-linear registration to the Montreal Neurological Institute (MNI) 152 template with 2 mm resolution. Data were spatially smoothed using a Gaussian kernel of full width at half maximum 4 mm and bandpass filtered (0.01–0.1 Hz). Head motion was controlled with the Friston 24-parameter model (Friston et al., 1996), and signals from white matter and the ventricles were regressed to account for physiological noise. The global signal was not regressed due to ongoing controversy surrounding whether this step is warranted when mapping rs-FC (Yang et al., 2014). Given that measures of rs-FC may be influenced by head motion (Power et al., 2012), each individual’s movement during scanning was quantified using framewise displacement (FD; Power et al., 2013). FD is a compressed single index calculated from derivatives of the six rigid-body realignment parameters. Volumes exceeding an FD of 0.5 mm, a commonly used threshold (Power et al., 2012), were eliminated, otherwise known as scrubbing.
Whole-brain functional network mapping
See Figure 1 for an overview of the methodology used. For each individual, a whole-brain resting-state functional network was mapped using established methods (Fornito et al., 2016). In brief, regionally averaged fMRI signals were determined for the
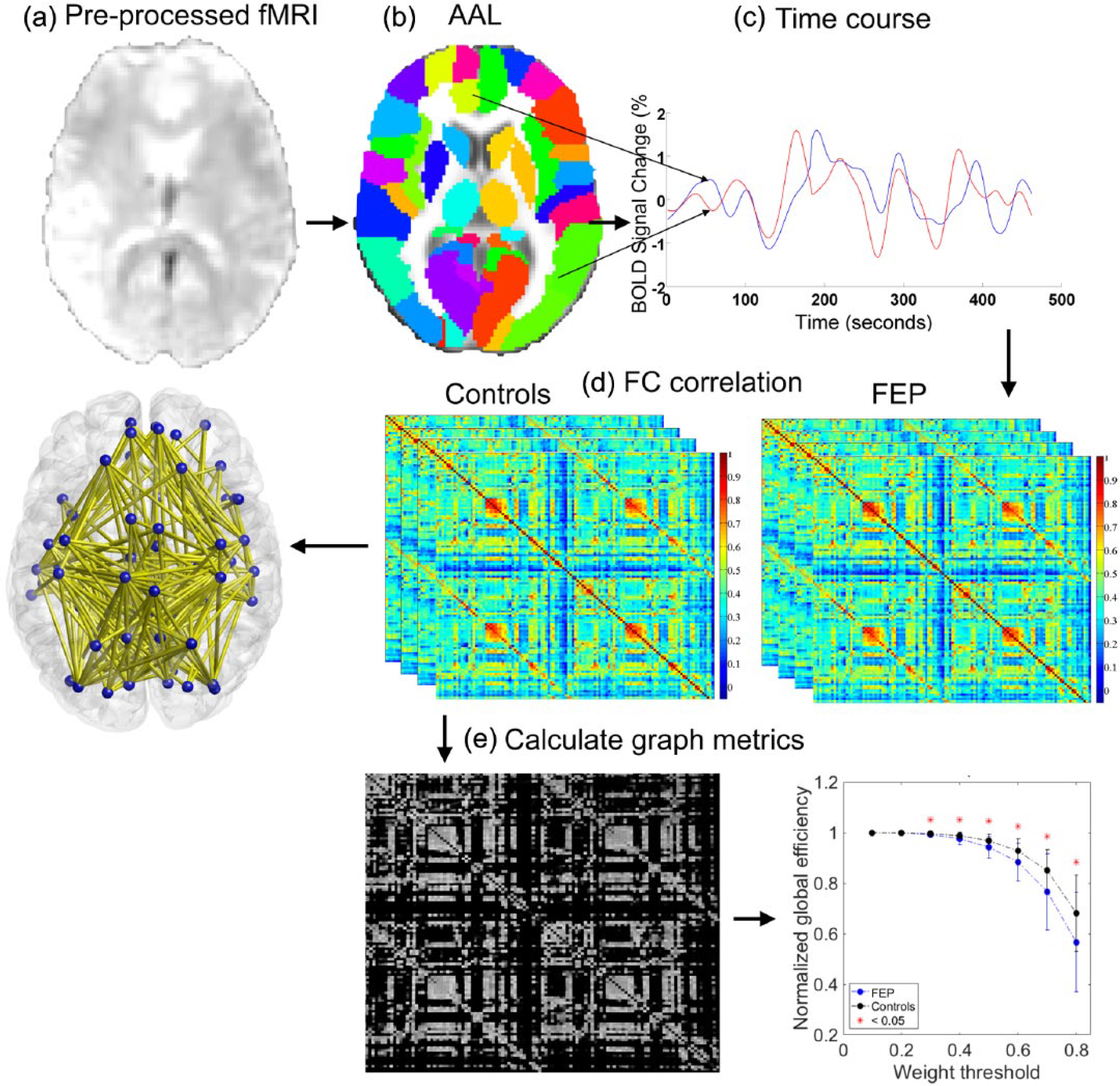
(a) Pre-processed and registered fMRI data were parcellated using (b) the AAL template. (c) The time course was extracted from each anatomical node. (d) A 116 × 116 connectivity matrix was then generated for each participant quantifying the extent of inter-regional FC between each pair of regions. The NBS was then used to identify the network of connections that significantly differed in FC between patients and controls. (e) Weight thresholding was performed by eliminating all connections below the chosen threshold and binarising the resulting network. Graph metrics including normalized global and local efficiency were then calculated.
Graph-theoretical analyses
Seven commonly studied graph-theoretical measures were computed: (1)
Statistical analyses exploring rs-FC and graph theory measures
We tested the following two hypotheses: (1) whether there was a difference in rs-FC and network topology between FEP individuals and healthy controls at baseline as well as at follow-up and (2) whether there was a difference between the two groups in the 12-month
Functional network properties and symptomatology/functioning
Pearson correlation was used to test for an association between rs-FC and graph theory measures with the five BPRS subscales (positive, agitation/mania, negative, depression/anxiety and total score; Ventura et al., 2000) and illness duration in the FEP group and four SANS subscales (anhedonia/avolition, flat affect/alogia, attention and total score), MADRS total score and SOFAS score in both FEP and control groups (groups analyzed separately) at both baseline and at follow-up. A repeated measures
Results
Demographics
Demographic information is shown in Tables 1 and 2. FEP individuals were statistically matched in terms of mean age and gender. No network measure was significantly correlated with age, duration of illness, medication dosage (chlorpromazine equivalent dosage) or IQ (
Demographic and clinical information at baseline.
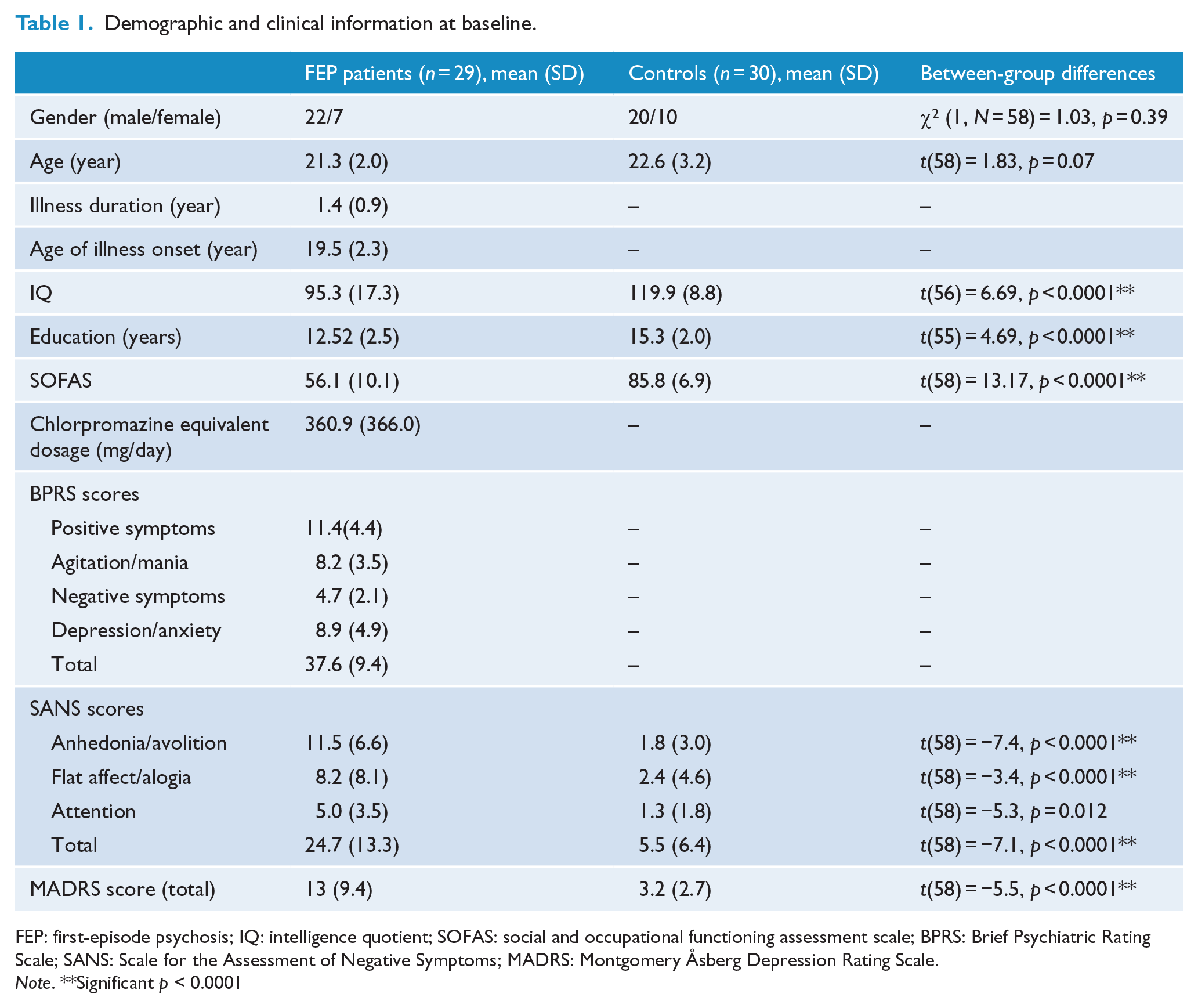
FEP: first-episode psychosis; IQ: intelligence quotient; SOFAS: social and occupational functioning assessment scale; BPRS: Brief Psychiatric Rating Scale; SANS: Scale for the Assessment of Negative Symptoms; MADRS: Montgomery Åsberg Depression Rating Scale.
Demographic and clinical information at 12-month follow-up.
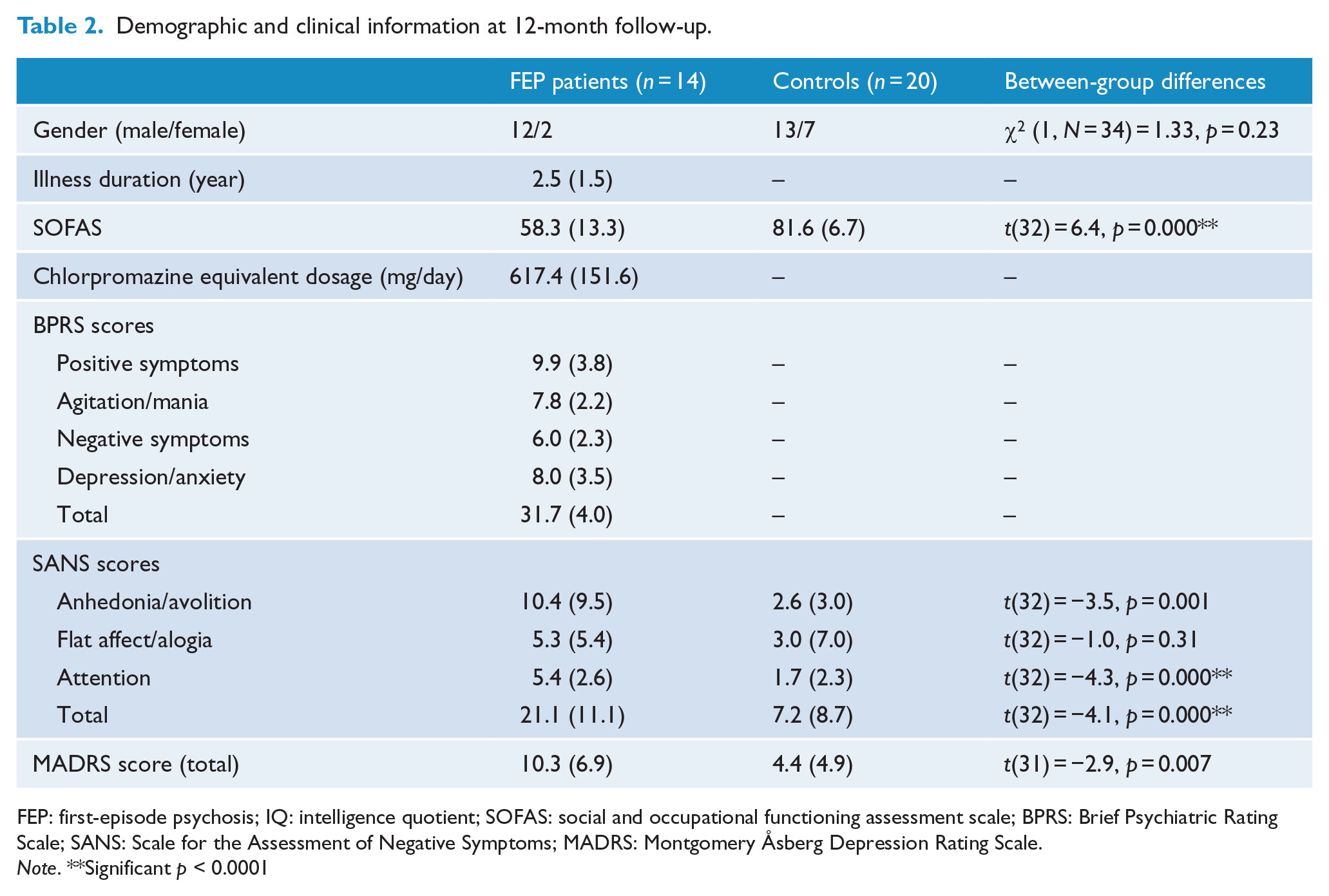
FEP: first-episode psychosis; IQ: intelligence quotient; SOFAS: social and occupational functioning assessment scale; BPRS: Brief Psychiatric Rating Scale; SANS: Scale for the Assessment of Negative Symptoms; MADRS: Montgomery Åsberg Depression Rating Scale.
Whole-brain FC
Analyses confirmed the null hypothesis of equality in rs-FC between FEP participants and healthy controls for all pairs of brain regions (
Functional connectivity and network topology metrics at baseline and at 12-month follow-up.
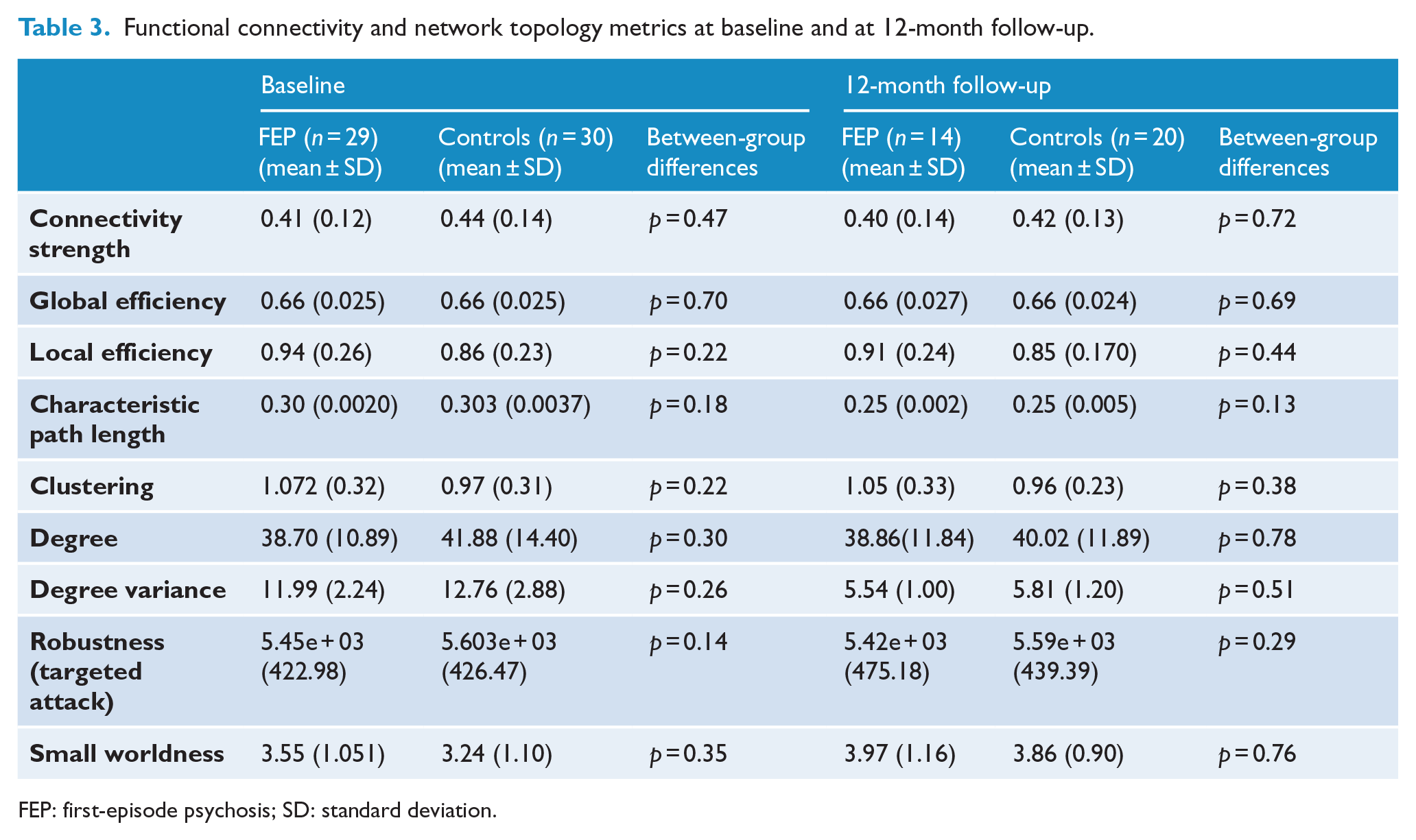
FEP: first-episode psychosis; SD: standard deviation.
Functional connectivity and network topology metrics change over 12 months.
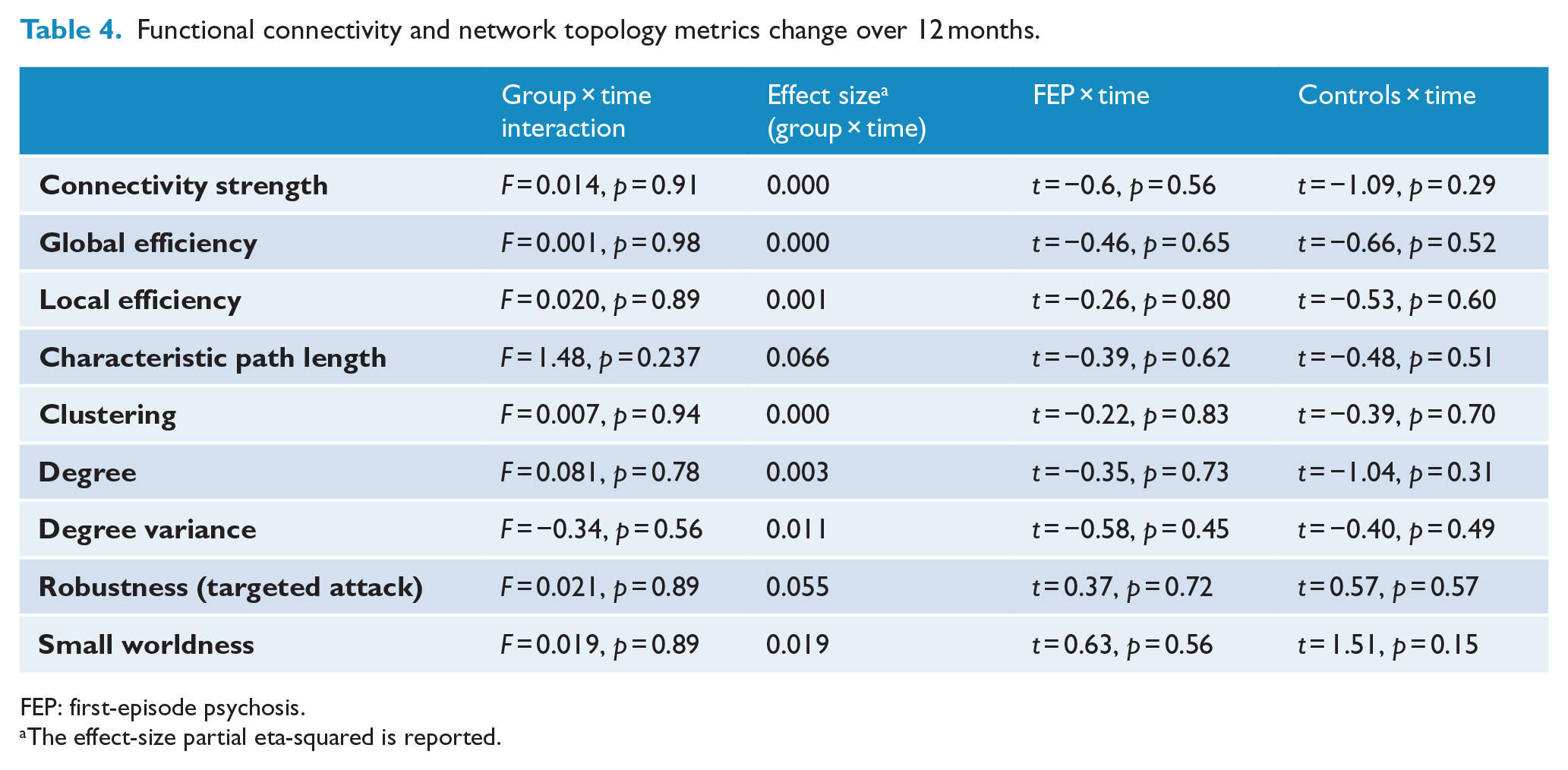
FEP: first-episode psychosis.
The effect-size partial eta-squared is reported.
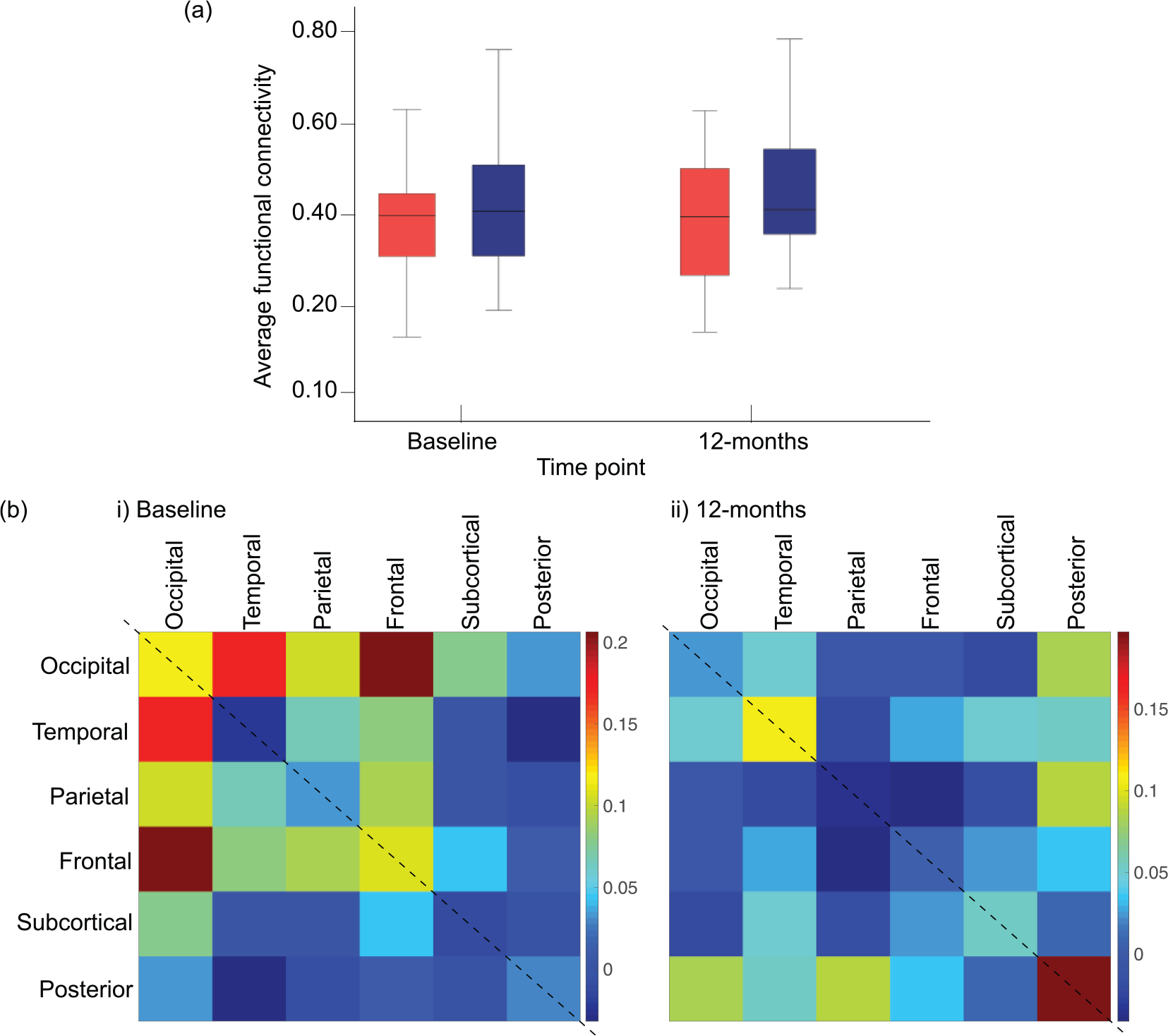
(a) Group differences in mean rs-FC across the whole brain between FEP (red) and control (blue) groups at baseline and 12-month follow-up. (b) Matrices showing the effect size of rs-FC between-group differences at (i) baseline and (ii) at 12-month follow-up for each brain lobe. Effect sizes quantified with Cohen’s
Network topology
Analyses confirmed the null hypothesis of equality between FEP participants and healthy controls in the seven graph measures tested at baseline and at 12-month follow-up (
Longitudinal changes
We found no evidence of an interaction effect between time and group on rs-FC and graph measures over a 12-month follow-up period (see Table 4). There was also no evidence of a main effect of time on rs-FC and graph measures over the 12-month follow-up period in either the controls or the FEP participants (see Table 4).
Relationships with symptomatology/functioning
There were no significant associations between symptom severity, social functioning or illness duration and measures of rs-FC strength and network topology at baseline or at 12-month follow-up. Symptom scores did not significantly change over the 12-month follow-up period in FEP participants, and there was no significant association between symptoms change and change in network measures over time.
Discussion
This study found no significant difference in rs-FC and network topology in FEP participants relative to healthy controls, and no evidence of change over a 12-month period. We also found no significant associations between network measures, social functioning and clinical characteristics. With an abundance of evidence reporting rs-FC abnormalities during the early illness stage of psychosis, the present findings provide novel insights regarding rs-FC integrity and topology in FEP both cross-sectionally and longitudinally.
Rs-FC in FEP is comparable to healthy individuals
To our knowledge, this is the first study to report no significant difference in rs-FC in FEP individuals relative to healthy controls. Although the current results do not support the prevailing findings of aberrant rs-FC, they add valuable insight to the existing pool of mixed literature. Our study adopted a data-driven approach to maximize inference, minimize bias and add to the handful of previous studies that used similar brain-wide analyses. Previous whole-brain rs-FC studies have reported both increased (Argyelan et al., 2015; Li et al., 2016b) and decreased (Guo et al., 2014, 2017; Li et al., 2016a) rs-FC in FEP cohorts relative to controls. Despite these studies adopting a similar data-driven approach, results are inconsistent, both in the direction of abnormal rs-FC reported and the specific brain regions implicated. These mixed findings in addition to the current finding of no impairment in FEP suggest that many factors are likely playing a role in mediating and/or moderating rs-FC during the early illness phase. For example, illness duration can vary across studies from months (Li et al., 2016b) to multiple years (Argyelan et al., 2015; Guo et al., 2017); consequently, moderating factors such as duration of medication use and illness are highly varied across cohorts, both of which have been shown to have an effect on rs-FC in FEP (Sarpal et al., 2015, 2017). Interestingly, this study found no significant relationship between network measures and IQ, education, medication use or illness duration both cross-sectionally and over time. Relatedly, our FEP sample had lower total symptom scores relative to previous whole-brain studies (Guo et al., 2014; Li et al., 2016b), placing them in a mild to moderately ill bracket (Leucht et al., 2005). This may be indicative of effective response to antipsychotic medication and/or psychosocial therapy, and our finding of no rs-FC impairment may reflect this. Results from longitudinal rs-FC studies in drug naïve FEP participants following their first antipsychotic medication administration indicate a progressive increase in rs-FC strength that correlated with symptom improvement (Sarpal et al., 2015). It is therefore possible that prior to the commencement of this study, effective antipsychotic treatment normalized any rs-FC or topological abnormalities that were present in this FEP cohort to levels observed in controls. If this is the case, it is an extremely encouraging notion that early intervention and/or effective pharmacological treatment can either reverse neural abnormalities (if they were ever present) or prolong/prevent their emergence over the observed 12-month period.
This, however, is speculative, and there is no way of testing this within the design of this study. Although our FEP cohort did not have the shortest illness duration compared to other studies (mean 1.5 years), they were all recruited from a specialized early intervention service, which likely provided swift and targeted treatment that may have minimized the duration of untreated psychosis (DUP). DUP has been associated with a number of outcomes, including increased symptom severity, poor prognosis (Penttila et al., 2014) and reduced rs-FC between regions of the striatum (Sarpal et al., 2017). Unfortunately, this study did not have information pertaining to DUP, and thus, any inferences made are speculative. However, it is possible that early and effective treatment from a specialized psychosis intervention service played a role in these individuals continuing to develop along a neurodevelopmental trajectory similar to that observed in controls.
Functional network topology in FEP is comparable to healthy controls
Contrary to our hypothesis, we found no evidence that FEP networks differed topologically relative to controls in any of the graph-theoretical measures tested. This finding is in line with results by Fornito et al. (2011), who investigated functional topology during a cognitive control task and found intact functional network architecture in FEP individuals relative to controls (Fornito et al., 2011). Taken together, these results suggest that the functional efficiency of brain-wide information exchange, and local integration and communication of information at the nodal level (Fornito et al., 2012), is not impaired in FEP relative to controls. Given that previous studies have shown abnormal structural topology in FEP (Crossley et al., 2017; Hu et al., 2016; Palaniyappan et al., 2016; Zhang et al., 2015), future research should explore white matter connectivity in addition to structural network topology in this and other FEP samples to further determine whether structural network architecture is also intact. Again, our finding of no functional topological impairment in FEP may reflect their relatively good clinical state and prognosis and/or the efficacy of their treatment.
Change in rs-FC over time in FEP was comparable to controls
There is an ongoing question surrounding whether psychosis is a progressive brain disorder involving trait neural abnormalities that are present at illness onset and progressively worsen as the illness becomes more established (Pantelis et al., 2009). To further explore this theory, we investigated whether rs-FC and topology changed abnormally in FEP individuals relative to controls over a 12-month follow-up period, and contrary to our hypothesis, we found no evidence of aberrant rs-FC change relative to that of normal development in controls. It is worth mentioning that our FEP group did meet criteria for FEP at baseline. However, there would be increased diagnostic stability in terms of type of psychosis at 12-month follow-up, given the average illness duration is now >2 years. Thus, recent-onset psychosis may be considered a better categorization for this sample at follow-up. Research has shown that roughly 55–60% of individuals who experience FEP go on to have a long-term functional disability (Menezes, Arenovich, & Zipursky, 2006). The notion that 40–45% of FEP individuals do not, questions the theory that early progressive neuropathological deficits are expected to be present during the first episode of psychosis. FEP individuals are at a point along the psychosis trajectory when schizophrenia is not a stable diagnosis nor is it an inevitable outcome; thus, it is perhaps unsurprising that we found little evidence of a stable neural abnormality associated with the disorder within a 1 year period. Given the clinical heterogeneity of early-onset psychosis, it is possible that different pathological mechanisms are operating at different stages of the illness that this study did not encompass (Bartholomeusz et al., 2017; Pantelis et al., 2005). Furthermore, at baseline, FEP participants who went on to complete a follow-up assessment had significantly lower BPRS total symptoms scores relative to FEP participants that withdrew from the study. It is therefore possible that our follow-up early-onset psychosis cohort are a subgroup of individuals who experienced less severe symptomatology and in turn are more neurobiologically similar to healthy controls and subsequently followed a similar neurodevelopmental trajectory over the 12-month period. Longitudinal designs with longer follow-up periods and larger samples are needed.
Limitations
The stringent brain-wide corrections for multiple comparisons provided excellent control for false-positive results; however, it was consequently more difficult to avoid false negatives (Radua et al., 2012). In addition, the high attrition rate meant we were unable to assess all FEP participants at both time points, resulting in a relatively small follow-up cohort and in turn, this limits the conclusions and inferences that can be made regarding longitudinal changes in rs-FC. We therefore cannot exclude the possibility that we were unable to detect some group differences because of limited statistical power in the longitudinal analyses.
Furthermore, our approach to investigating neural abnormalities in FEP, which largely focused on FC and network efficiency, did not identify potential brain network alterations in this sample, suggesting that clinical presentation may not be driven by aberrant connectivity at this illness stage. However, this was not directly examined in this study, and future studies should consider investigating connectivity (including white matter connectivity), gray matter volumetric alterations and multi-modal approaches and how this correlates with symptomatology and functioning in the early illness stages.
Conclusion
The pathophysiology of schizophrenia is heterogeneous and has been demonstrated to involve multiple brain regions and systems. The early stage of the disorder is most likely a time when neural correlates and mechanisms of psychosis are most varied and difficult to characterize, and the highly mixed literature speaks to this. Early identification and intervention are increasingly viewed as a critical process in order to alter the trajectory of psychosis, and as a field, there is still much to be learned about the neurobiology of this complex and sometimes impermanent illness stage. From a functional perspective, our null findings are encouraging and suggest a more optimistic perception of the illness. It is important for the schizophrenia field to be informed that our results are indicative of a group of FEP individuals who show no observed abnormalities in rs-FC and topology and no evidence of deterioration in the network properties measured over a 12-month period. With this said, a larger sample with a longitudinal design is needed to confirm whether a subgroup of FEP patients who show no rs-FC abnormalities at baseline do display a neurotypical trajectory over time. While our 12-month data might be an indication, it is difficult to make conclusions and inferences with limited statistical power. Further to this, this study does not have any long-term follow-up data to confirm the prognosis of these FEP participants, as some may have essentially recovered, while others may have gone go on to develop a more chronic stable psychotic illness beyond the 12-month follow-up. Therefore, longer follow-up periods are needed to identify the underlying protective mechanisms driving unaffected rs-FC and topology at baseline in early psychosis cohorts.
Given the field is moving toward personalized medicine, the logical next step is to identify under what specific circumstances do a comparatively healthy FC and topological network occur in early-stage psychosis and what neuroprotective factors may be contributing to this neurotypical picture.
Footnotes
Acknowledgements
The authors wish to acknowledge the contributions of study participants, clinicians at recruitment services, staff at the Murdoch Children’s Research Institute and research staff at the Melbourne Neuropsychiatry Centre, including coordinators Phassouliotis, C. and Merritt, A. and research assistants Jones, H., Di Biase, M. and Tahtalian, S. We would also like to thank Chester Kang for IT support. Andrew Zalesky and Cali F Barthomolomeusz share joint last authorship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
E.P.G. was supported by the University of Melbourne and CRC for Mental Health PhD top-up scholarship. C.P. was supported by NHMRC Senior Principal Research Fellowship (628386 and 1105825). A.Z. was supported by a NHMRC Career Development Fellowship (GNT1047648) S.W. was supported by a NHMRC Career Development Fellowship (ID: 1007716).
