Abstract
Objective:
Lifetime depression and depression around the time of an acute coronary syndrome event have been associated with poor cardiac outcomes. Our study sought to examine the persistence of this association, especially given modern cardiac medicine’s successes.
Methods:
For 332 patients admitted for an acute coronary syndrome, a baseline interview assessed major depression status, and psychological measures were administered. At 1 and 12 months post–acute coronary syndrome event, telephone interviews collected rates of hospital readmission and/or death and major depression status, while biomarker information was examined using medical records.
Results:
The 12-month mortality rate was 2.3% and cardiac readmission rate 21.0%. Depression subsequent to an acute coronary syndrome event resulted in a threefold and 2.5-fold increase in 1-month and 12-month odds of cardiac readmission or death, respectively. No relationship with past depressive episodes was found. Poor sleep was associated with higher trait anxiety and neuroticism scores and with more severe depression.
Conclusion:
Lifetime depression may increase the risk of depression around the time of an acute coronary syndrome but not influence cardiac outcomes. We suggest that poor sleep quality may be causal or indicate high anxiety/neuroticism, which increases risk to depression and contributes to poor cardiac outcomes rather than depression being the primary causal factor.
Associations between depression and cardiovascular disease (CVD), especially acute coronary syndrome (ACS; including myocardial infarction [MI] and unstable angina), have long been described. Numerous studies have documented a lifetime (LT) history of depression as a risk factor for ACS onset and that those depressed around the time of an ACS event are more likely to be hospitalized or die within the next year. The latter have commonly been sub-divided into those who (1) are depressed prior to the ACS event, (2) develop depression concurrently within 1 month of the ACS event and (3) develop depression at least 1 month following the ACS event.
Both a systematic review of a etiological contributions to CVD in initially healthy subjects (Penninx, 2017) and a meta-analysis pooling longitudinal data from 21 prognostic cohort studies involving over 120,000 subjects (Nicholson et al., 2006) quantified that LT depression results in an estimated 80–90% increased risk of CVD. Lichtman et al. (2014) reviewed 53 studies and 4 meta-analyses, investigating the effect of depression on adverse ACS outcome. Consistent positive associations were found between depression assessed before, during or after the index admission and cardiac mortality at follow-up periods ranging from 1 month to 10 years. Despite methodological variability, the authors concluded, in a statement approved by the American Heart Association, that there is sufficient evidence supporting the position that concurrent depression is a risk factor for an adverse ACS outcome.
Despite such well-documented associations, it remains unclear whether differing time periods for depression in relation to the ACS provide differing levels of risk. Clarification remains pertinent, given the prevalence (15–31%) of depression in patients with coronary artery disease and its impact (Lesperance et al., 1996; Lichtman et al., 2014; Rudisch and Nemeroff, 2003). It is also unclear whether recent improvements in cardiovascular technology and management approaches (Forman et al., 2016; Ramakrishna et al., 2016)—which have resulted in improved cardiac outcomes (i.e. distinctive reductions in subsequent morbidity and mortality rates)—have impacted on the previously identified risk.
Our earlier studies indicated that the first-ever or recurrent depression emerging around the time of an ACS posed the greatest risk (compared to an earlier episode or extant depression) of a poor cardiovascular outcome and death (Parker et al., 2008, 2011). Such findings raise questions about possible triggering mechanisms at the time of a coronary event, with inflammatory processes akin to the ‘acute sickness response’ to infection or injury being proposed (Granville Smith et al., 2015a) as one explanatory hypothesis.
In addition, sleep is known to regulate key physiological responses such as inflammation, glucose tolerance, insulin sensitivity and appetite (Alcántara et al., 2014), and when dysregulated, can dangerously increase the heart rhythm and myocardial oxygen demand (Gabor et al., 2001). There is more robust evidence that poor sleep significantly increases the risk of fatal and non-fatal cardiovascular events (Marin et al., 2005) and that short sleep (less than 7 hours per night) results in an increased 1-year risk of ACS recurrence and mortality (Pelletier et al., 2016). Poor sleep may also reflect a higher-order variable such as high trait anxiety or neuroticism, which has previously been suggested as a risk variable (Pelletier et al., 2016).
In light of this, we undertook the current study, seeking to clarify the risk periods for depression leading to a poor ACS outcome in light of improved cardiac management and outcome, and examine a range of biomarkers and psychological factors so as to inform about underlying mechanisms.
Methods
Participants and procedure
A total of 344 participants with a confirmed ACS event (the primary admitting diagnosis) were recruited consecutively between 2010 and 2014 from cardiology wards at Sydney’s Prince of Wales Hospital. The ACS event was confirmed from patient’s medical records and verbal confirmation from a cardiac doctor or nurse on the ward at the time of recruitment. The baseline assessment occurred on average 4.5 days following admission. Inclusion criteria were the following: being 18 years or older, no visual or hearing impairment, ability to speak and understand English, no concurrent infection and no elevated mortality risk from comorbid conditions. The study protocol was approved by the university and hospital ethics committees, and informed written consent was obtained from all participants.
Clinical interview and questionnaires
At baseline assessment, the structured clinical interview (Mini International Neuropsychiatric Interview [MINI]; Sheehan et al., 1998) was conducted by a researcher to assess for any current or LT major depressive episode (MDE) and/or dysthymia according to Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) criteria. Several additional psychological measures were administered, including the Eysenck Personality Questionnaire Revised (EPQ-R) neuroticism scale (Eysenck and Eysenck, 1968) and the Costello and Comrey Anxiety Scale (CCAS; Costello and Comrey, 1967) trait anxiety measure, to assess further comorbidities. In addition, the Pittsburgh Sleep Quality Index (PSQI; Buysse et al., 1989) self-report questionnaire—which assesses sleep quality over the preceding month—was administered (and repeated at follow-up assessments), with a total score of 5 or more indicative of poor sleep quality. The MINI was repeated during the 1-month and 12-month follow-up telephone interviews. The 1-month assessment allowed determination of any depression developing following the ACS or if any earlier onset episode had continued. Participants reporting depression were classified with ‘ongoing depression’ if their depression pre-dated their ACS and continued through to the 1-month follow-up or with ‘ACS-related depression’ if depression emerged within 1 month of their cardiac event. The 1-month and 12-month follow-up telephone interviews further assessed participants’ physical status (i.e. hospital readmission or death).
Assessment of immune, autonomic and nutritional biomarkers
At baseline and as detailed previously (Vollmer-Conna et al., 2015), a blood sample was obtained to examine inflammatory biomarkers, including interleukin-6 (IL-6), interleukin-1-beta (IL-1b), interferon-gamma (IFN-γ), tumor necrosis factor alpha (TFN-α), interleukin-10 (IL-10) and C-reactive protein (CRP) and nutritional biomarkers—determining the polyunsaturated fatty acid profile of serum phospholipids (Omega-3 (N3), Omega-6 (N6), N3/N6 ratio) and quantifying serum 25-hydroxyvitamin D (25[OH]D) levels (Vitamin D). Cardiac injury markers included troponin and white cell count (WCC). Tests of autonomic functioning were undertaken, with heart rate variability measures derived from electrocardiogram data (high-frequency variability, low-frequency variability, pulse oximeter [indirect measure of oxygen saturation, via blood flow fluctuation] and electrodermal activity—physiological measure of anxiety).
Statistical analyses
Data were analyzed using the Statistical Package for Social Sciences (SPSS; Version 22.0) and interpreted with a significance level of p < 0.05. Binary logistic regression was used to assess the role of depression status in determining outcomes following the ACS event. Univariate analyses of variance and Pearson’s pairwise correlations were used to explore potential differences in biomarkers across depression sub-groups.
Results
Demographic information
Of the 344 participants initially enrolled in the study, the mean age was 62.1 years (standard deviation [SD] = 11.9 years) and 79.1% were male. The other socio-demographic variables of the sample at baseline are presented in Table 1. Depression status information was collected at 1 month for 331 (96.2%) of those patients (mean age of 62.0 years and SD of 11.8 years), of whom 79.5% were male. Most participants (n = 239; 72.0%) reported no previous history of LT depression prior to the ACS event, while 93 participants (28.0%) met criteria for an earlier (i.e. LT) depressive episode. As assessed at 1 month, 14 (4.1%) were classified as having ‘ongoing depression’, 47 (13.7%) as having ‘ACS-related depression’ and the remaining 271 as ‘not currently depressed’. A prior LT episode of depression had been experienced by 100%, 57.4% and 19.2% of those in the ‘ongoing depression’, ‘ACS-related depression’ and ‘not currently depressed’ groups, respectively.
Baseline social-demographic characteristics of those currently depressed (n = 61) or not currently depressed (n = 271).
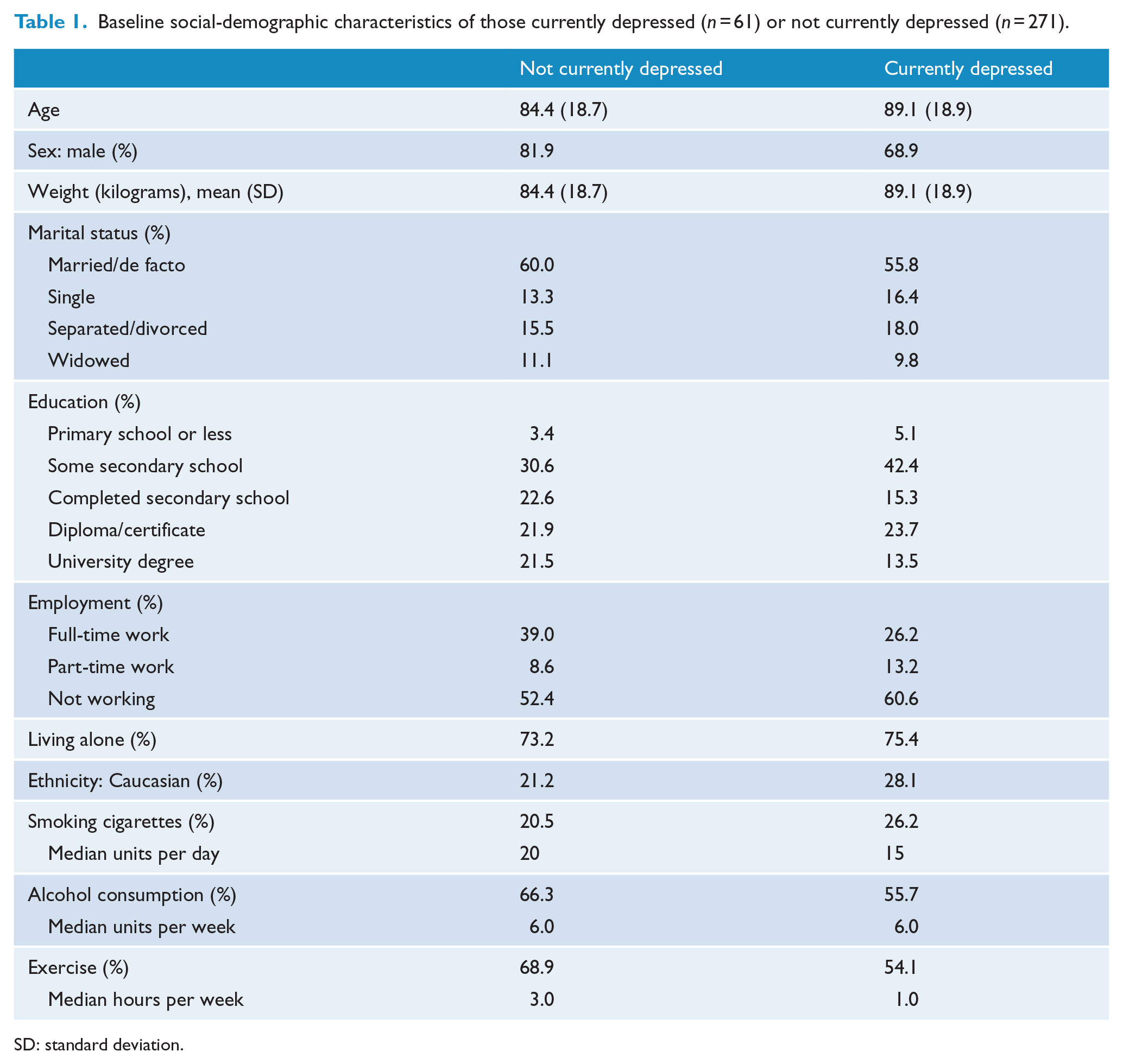
SD: standard deviation.
Outcomes 1-month post-cardiac event
Three individuals died before the 1-month follow-up clinical interview and therefore could not be assigned to a group. Consequently, analyses of cardiac readmission with or without death produced identical results. A total of 36 (10.9%) cardiac readmissions were reported at 1 month. The three event-focused depression sub-groups (ongoing depression, ACS-related depression and those not currently depressed) comprised the predictor variables in the initial binary logistic regression analyses, with results for cardiac-related hospital readmission at the 1-month follow-up shown in Table 2. The unadjusted odds ratio (OR) of cardiac readmission was 2.99 times greater for patients with ACS-related depression compared to those not currently depressed (p < 0.01). Ongoing depression was also associated with increased and comparable odds (i.e. 2.94) of cardiac readmission but did not reach statistical significance presumably due to the smaller number of patients. Including patients’ age and gender in the regression model did not have any appreciable influence.
Unadjusted odds ratios for the likelihood of cardiac readmission or death at 1-month and 12-month follow-up according to depression status around the time of the ACS event.
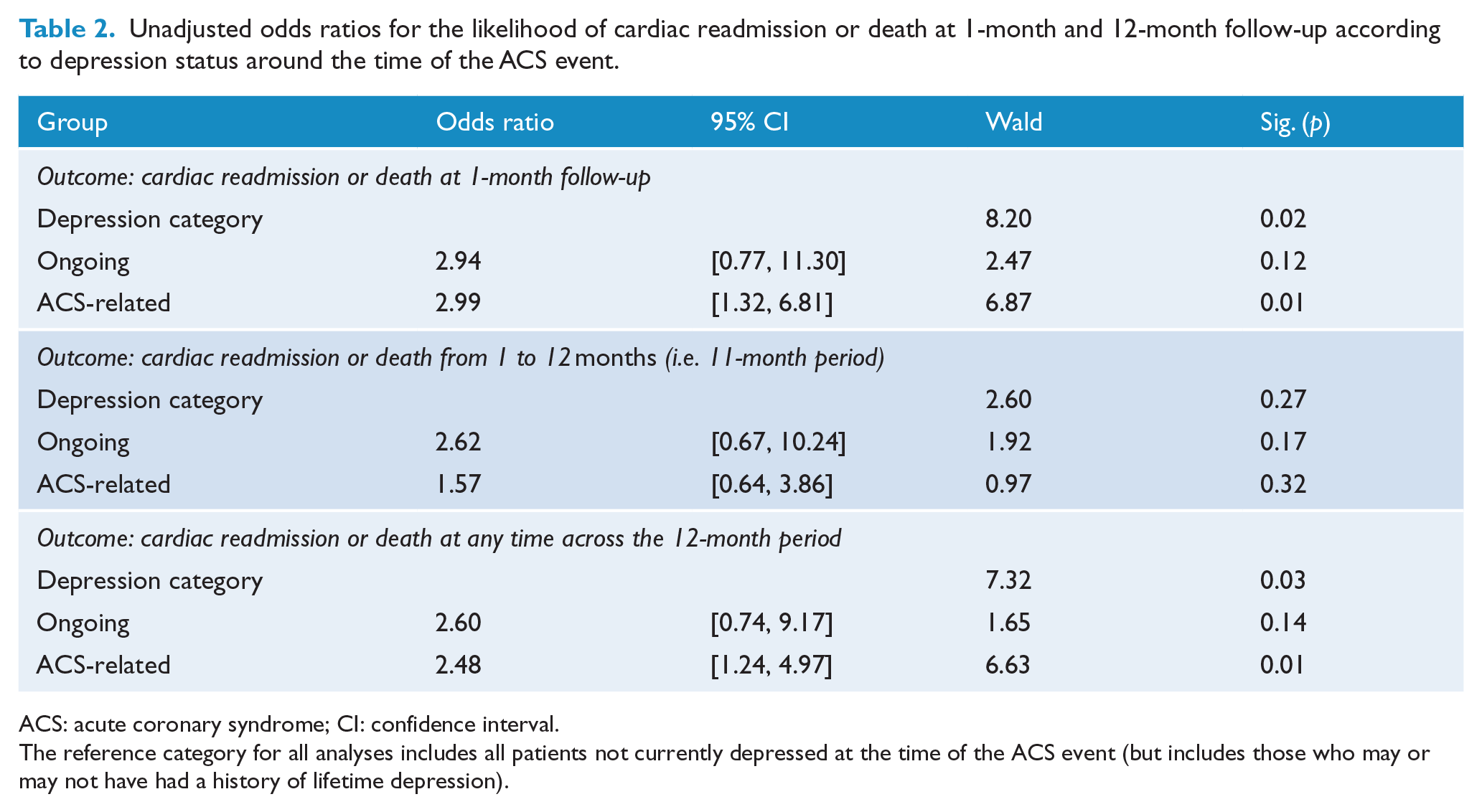
ACS: acute coronary syndrome; CI: confidence interval.
The reference category for all analyses includes all patients not currently depressed at the time of the ACS event (but includes those who may or may not have had a history of lifetime depression).
Outcomes 12 months post-cardiac event
Analysis of 12-month outcomes initially included only ‘new’ cardiac readmissions and/or deaths that occurred after the 1-month follow-up (n = 34). There were no significant differences in the unadjusted odds for cardiac readmission for those with ongoing depression or ACS-related depression compared with those not currently depressed. Inclusion of the five individuals who had died over the interval did not substantially change the unadjusted ORs (see Table 2).
Over the entire 12-month follow-up period, 70 (21.0%) individuals were readmitted for cardiac problems and, as noted, an additional five died. As shown in Table 2, the unadjusted odds of cardiac readmission (or death) were significantly higher for patients in the ACS-related depression group compared to the not currently depressed group. Those with ongoing depression also showed somewhat increased odds of cardiac readmission or death, but this did not reach formal significance. After adjusting for age, the odds of readmission or death observed for both the ACS-related depression (OR: 2.48, 95% confidence interval [CI] = [1.24, 4.97], p = 0.011) and ongoing depression (OR: 2.60, 95% CI = [0.74, 9.17], p = 0.139) groups were similar to the unadjusted odds analyses, while the inclusion of gender did not improve the model fit.
The impact of LT depression
Of the 93 patients with an LT depression, 27 (29.0%) developed an ACS-related depression, a 3.5-fold increase compared to only 20 (8.4%) of the no LT depression participants (χ2 = 31.3, p < 0.001). Of the remaining patients with an LT history of depression, 52 (55.9%) were not depressed at assessment, while a further 14 (15.1%) had experienced an ongoing depressive episode prior to, and at the time of the ACS event.
Figure 1 and Table 3 summarize the incidence rates of cardiac-related hospital readmission or death among patients occupying five depression categories over time (i.e. never depressed, LT depression only, ACS-related depression only, LT and ACS depression, and ongoing depression). No “both LT and ongoing depression” group was created as all with ongoing depression had experienced LT depression. In comparison to the low rates observed in patients who were never depressed or experienced a history of LT depression only, rates were distinctly and comparably higher for those with ACS-related depression or ongoing depression, either alone or in conjunction with LT depression.
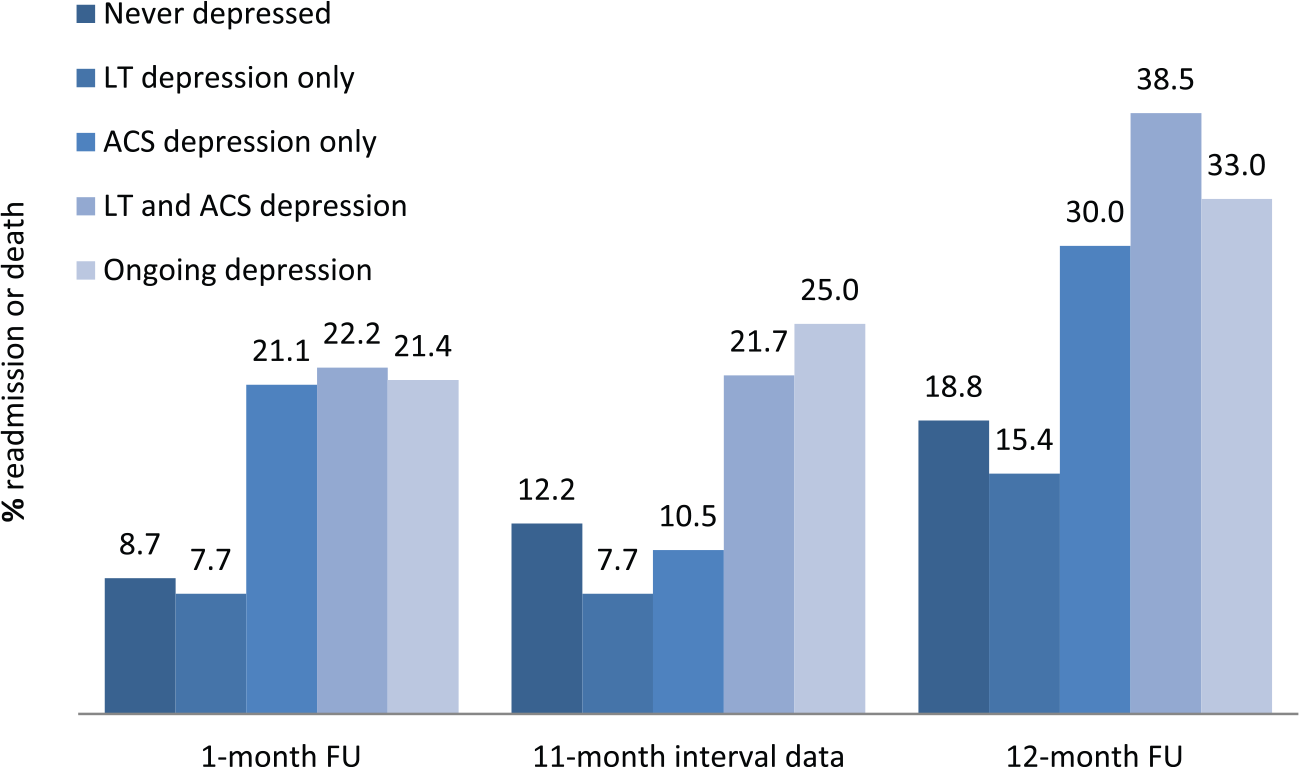
Incidence rates of cardiac-related hospital readmission or death across varying depressive groups as assessed at 1-month and across the entire 12-month follow-up period (n = 331).
Incidence rates of cardiac-related hospital readmission or death across varying depressive groups as assessed at 1-month and across the entire 12-month follow-up period (N = 331).
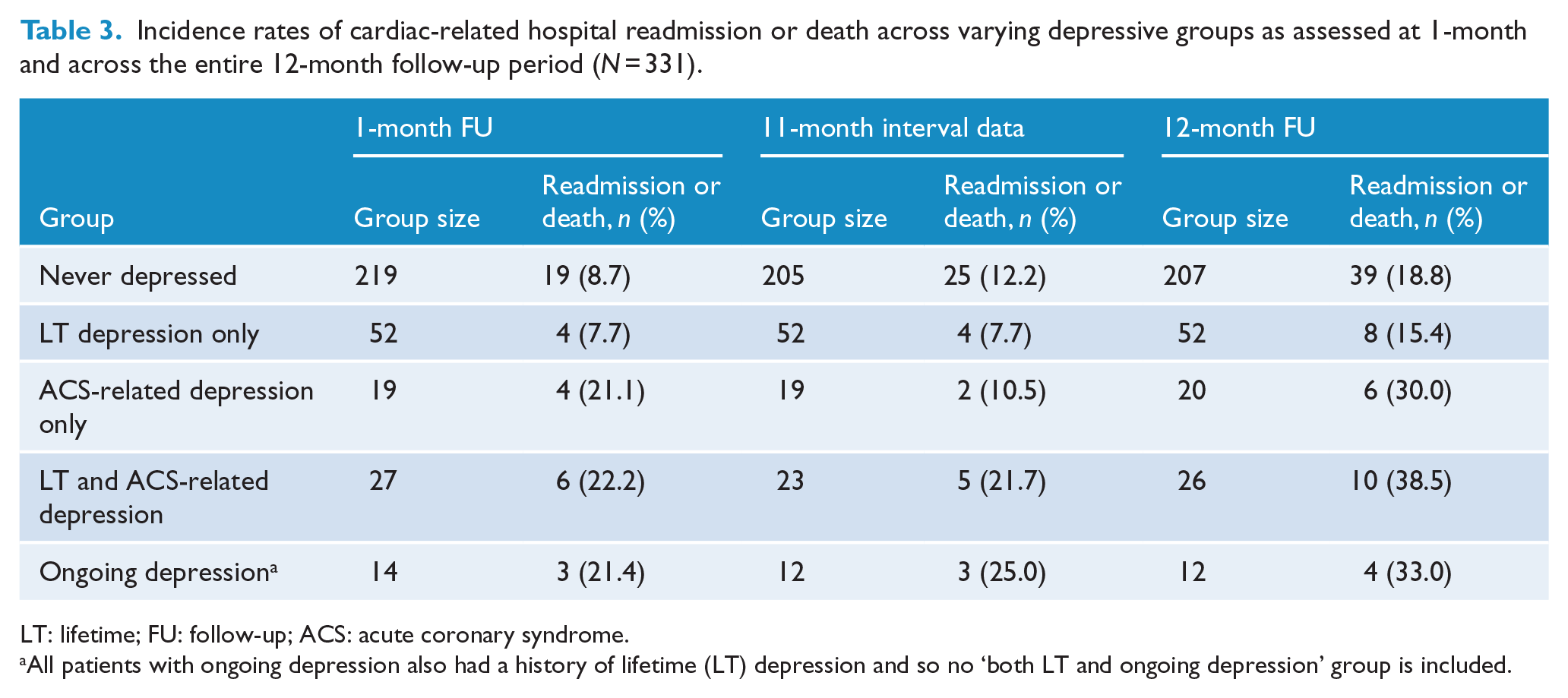
LT: lifetime; FU: follow-up; ACS: acute coronary syndrome.
All patients with ongoing depression also had a history of lifetime (LT) depression and so no ‘both LT and ongoing depression’ group is included.
Binary logistic regression analyses including all five groups, and controlling for age as a covariate, quantified that patients experiencing both LT and ACS-related depression were 3.08 times more likely to be readmitted or die within the 1-month follow-up period (95% CI = [1.10, 8.62], p = 0.032) compared to patients who were never depressed (the reference group), while a history of LT depression only (OR = 0.90, 95% CI = [0.29, 2.80], p = 0.861) or ACS-related depression only (OR = 2.8, 95% CI = [0.84, 9.34], p = 0.901) did not significantly increase the odds of poor outcome by 1 month. Patients who had experienced an ongoing depression (and, as noted, who all had experienced LT depression) were at 3.01 times increased risk of readmission or death compared to the never-depressed group at the 1-month follow-up, albeit not formally significant (95% CI = [0.76, 11.88], p = 0.115), again presumably due to the small group number.
At the 12-month follow-up, participants with both LT and ACS-related depression were 2.97 times more likely to be readmitted or die (95% CI = [1.23, 7.17], p = 0.015) compared to the never-depressed reference group. In contrast, a history of LT depression only (OR = 0.87, 95% CI = [0.38, 2.03], p = 0.755) or ACS-related depression only (OR = 1.83, 95% CI = [0.65, 5.14], p = 0.254) or ongoing depression (OR = 2.53, 95% CI = [0.71, 9.05], p = 0.152) did not significantly increase the likelihood of a poor 12-month outcome.
Examination of biomarkers and questionnaire data
As noted in the ‘Methods’ section, biomarker data were collected and analyzed. None showed group differences. Table 4 displays the total scores for sleep quality (PSQI), trait anxiety (CCAS) and neuroticism (EPQ-R) for each group across each assessment time period. Only sleep quality showed a significant group difference at the baseline assessment (F (4, 323) = 11.6, p < 0.01), 1-month follow-up (F (4, 322) = 11.6, p < 0.01) and 12-month follow-up assessments (F (4, 309) = 15.8, p < 0.01).
Sleep quality (PSQI total scores), trait anxiety (CCAS) and neuroticism (EPQ-R) scores between depressive groups (N = 328).
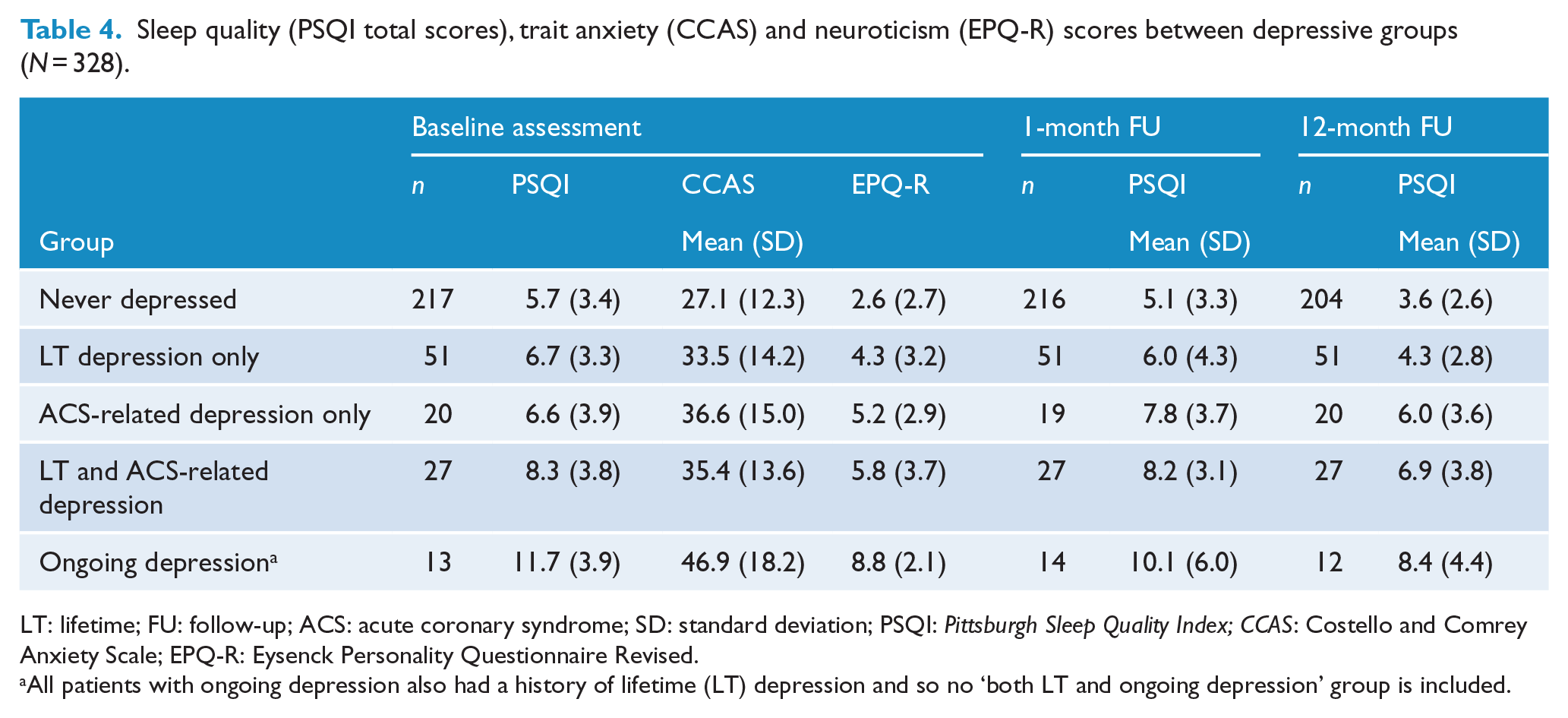
LT: lifetime; FU: follow-up; ACS: acute coronary syndrome; SD: standard deviation; PSQI: Pittsburgh Sleep Quality Index; CCAS: Costello and Comrey Anxiety Scale; EPQ-R: Eysenck Personality Questionnaire Revised.
All patients with ongoing depression also had a history of lifetime (LT) depression and so no ‘both LT and ongoing depression’ group is included.
Pairwise comparisons showed that, at baseline assessment, sleep quality was poorest in the ongoing depression group compared to those with both LT and ACS-related depression (F (4, 323) = 3.6, p < 0.05) and at the p < 0.001 level when compared to all other groups. Those with both LT and ACS-related depression showed significantly poorer sleep quality only when compared with the never-depressed group (p < 0.01).
At the 1-month follow-up, participants with ongoing depression showed significantly poorer sleep quality compared to those never depressed (p < 0.001) and those with LT depression only (p < 0.01). PSQI scores indicated poorer sleep for patients with both LT and ACS-related depression (p < 0.001) and for those with ACS-related depression only (p < 0.05) when compared with the never-depressed group. At the 12-month follow-up, the patients with ongoing depression showed significantly poorer sleep quality compared to patients never depressed (p < 0.001) and those with LT depression only (p < 0.001). Similarly, self-reported sleep quality was poorer for patients with both LT and ACS-related depression (p < 0.001) and ACS-related depression alone (p < 0.01) when compared to never-depressed patients. Those with both LT and ACS-related depression also showed poorer sleep quality compared to those with LT depression only (p < 0.01).
While ongoing poor sleep may be caused by a number of different factors, a key candidate is high trait anxiety. We therefore examined trait anxiety (CCAS) and neuroticism (EPQ-R) scores. Correlational analyses showed that, at baseline assessment, higher PSQI scores (i.e. poorer sleep quality) were significantly associated with higher trait anxiety scores on the CCAS (r (323) = 0.26, p < 0.0001) and higher neuroticism scores on the EPQ-R (r (324) = 0.33, p < 0.0001). Similar associations were quantified when baseline PSQI scores were correlated with 1-month and 12-month CCAS and EPQ-R scores, suggesting that higher anxiety and neuroticism were compromising sleep in an enduring manner.
Mean baseline trait anxiety and neuroticism scores are presented in Figure 2. Overall group differences in CCAS scores were highly significant (F (4, 320) = 11.2, p < 0.001). Bonferroni pairwise comparisons showed that CCAS trait anxiety scores were significantly higher (all p < 0.05) in all groups experiencing depression relative to patients never depressed and the highest for patients experiencing ongoing depression—who also had significantly higher trait anxiety scores compared to those with a history of LT depression only (CCAS mean difference = 13.4, p = 0.05). Mean trait neuroticism scores also showed highly significant overall group differences (F (4,321) = 21.7, p < 0.001) and were also significantly higher (p < 0.05 level) within all groups experiencing depression compared to those never depressed and were highest for those with ongoing depression—and whose trait neuroticism scores were significantly higher (p < 0.05) compared to those in each other group (Figure 2).
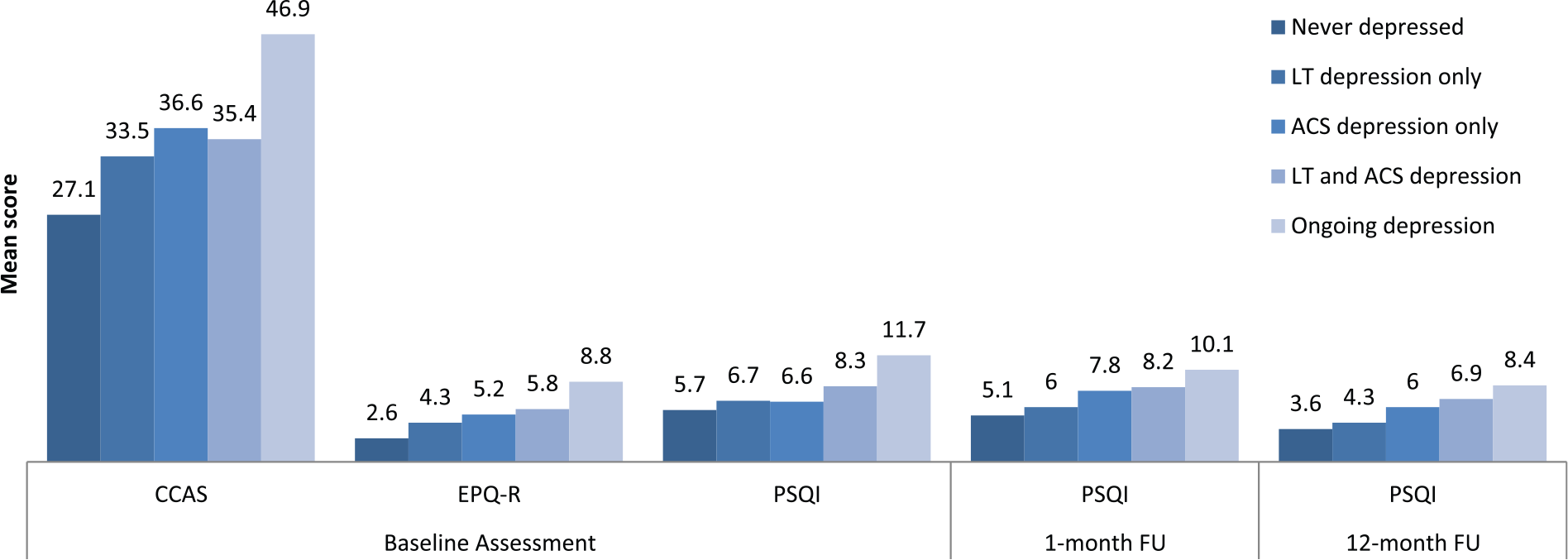
Trait anxiety (CCAS), neuroticism (EPQ-R), and sleep quality (PSQI total scores) across depressive groups (n = 328).
Discussion
This study investigated the impact of the presence and timing of depression on the likelihood of hospital readmission and/or death at 1 and 12 months following admission for an ACS event, a focus of intrinsic importance and currently salient in light of increasingly successful cardiac management (Forman et al., 2016; Ramakrishna et al., 2016) leading to decreased morbidity and mortality rates. In relation to the latter issue, only eight (2.3%) sample patients died over the 12-month follow-up period. This finding is consistent with a 12-month mortality rate of 2% recently reported in a European sample of 33,538 patients discharged from hospital with an MI (Alcántara et al., 2014) and are lower than 12-month mortality rates reported in earlier studies—for instance, of 7.7% in a 20-year-old study (Lesperance et al., 1996) and 5.6% in our own earlier study (Parker et al., 2008). Thus, examining the mortality rate as a discrete outcome variable may no longer be an appropriate research strategy due to its likely low base rate. There is, however, less evidence that 12-month readmission rates have decreased over time, with study rates ranging from 12.9% to 20.2% (Rodriguez-Padial et al., 2017). A readmission rate of 21.0% in this study indicated that this latter outcome variable may be more suitable in such studies.
Our analyses quantified that ACS-related depression resulted in significantly increased odds of a worse outcome during the 1-month period following the initial admission compared to patients not then depressed. There was a possible slight potentiating effect for patients experiencing both LT and ACS-related depression, with this group showing the highest incidence rates of cardiac-related hospital readmission or death at both follow-up periods. Despite the statistically non-significant difference in cardiac risk among patients with ongoing depression compared to those who were not depressed, they showed comparable ORs and incidence rates of poor cardiac outcome within the first year following admission to the ACS depressed group, suggesting that the non-significant finding for the latter group reflected the smaller group size. Such findings indicate that depression presenting around the time of ACS poses an increased risk of poor cardiac outcome (i.e. whether ACS-related or a pre-existing and ongoing episode).
Of key importance, our study clarifies previous interpretations of the impact of LT depression. In this study, LT depression alone appeared to make no contribution in that its rate of a poor outcome over 12 months was 15.4% compared to 18.8% for those who had never been depressed. Further analyses, however, revealed that those with LT depression were four times more likely to develop ACS-related depression compared with those never experiencing such an episode. Furthermore, those with LT depression as well as either ACS-related depression or ongoing depression were approximately twice as likely to have a poor outcome. Current findings, therefore, challenge previous studies implicating LT depression as a risk factor per se and suggest that any LT depression contribution simply reflects in increasing the chance of having depression around the time of the cardiac event—and that it is only the latter that leads to a poor outcome. Our findings are compatible with Lesperance et al. (1996) reporting a twofold increase in risk of MI-associated depression in those with a history of MDE compared to those without such a history and with a study by Sundbøll et al. (2017) reporting mortality risks as higher among patients with an MI and having a previous depressive diagnosis than among those without a previous depression diagnosis, but clarify such findings. Mediation analyses of direct and indirect effects between LT depression, ACS-related/ongoing depression and poor health outcomes in future studies—pursuing factors that determine depression at differing time points—are required, with the contribution of trait anxiety considered shortly. Thus, it would appear that if depression leads to a poor outcome, then it is limited to depression present around the time of the cardiac event.
Our results are consistent with previous findings indicating that depression commencing with or shortly following the ACS event poses a high risk to poor coronary outcome (Parker et al., 2008, 2011) and also suggest that the risk associated with depression is not restricted to depression having its onset at that proximal time—as depression present prior to the ACS event and which continued through to the ACS event was also predictive of a worse outcome in our study.
In pursuing causal factors, then the focus should be on those that are relevant to depression commencing or already present at the time of the cardiac event. Biological explanations that have been put forward invoke pathophysiological mechanisms that are shared in those experiencing an ACS event and in those who are depressed and notably inflammation and cardiac autonomic regulation (Granville Smith et al., 2015b). Psychosocial factors may include the impact of demoralization or a lack of application to cardiac rehabilitation programs and even adherence with cardiac medications.
We collected and tested a set of biological markers measured at the time of the ACS event (Granville Smith et al., 2015b) and did not find sufficient support for the possible explanation of inflammatory processes being invoked. Only poor sleep quality was identified as differentiating specific sample sub-groups, being least likely in those who were never depressed and highest in those with ongoing depression. As a seeming ongoing factor (in that it was present in the depressed groups at baseline and at follow-up periods), poor sleep might act directly or by factors contributing to poor sleep, as modeled in Figure 3. As poor sleep may also reflect a higher-order variable such as high trait anxiety or neuroticism, we analyzed data on those variables. The lowest scores on both variables were returned by those who never experienced depression, while the highest scores were returned by those in the ongoing depression sub-group, with intermediate scores in other sub-groups with depression. As those with high trait anxiety and/or high neuroticism levels are more likely to develop depression, it may well be high trait anxiety rather than depression that increases the risk of a poor cardiac outcome.
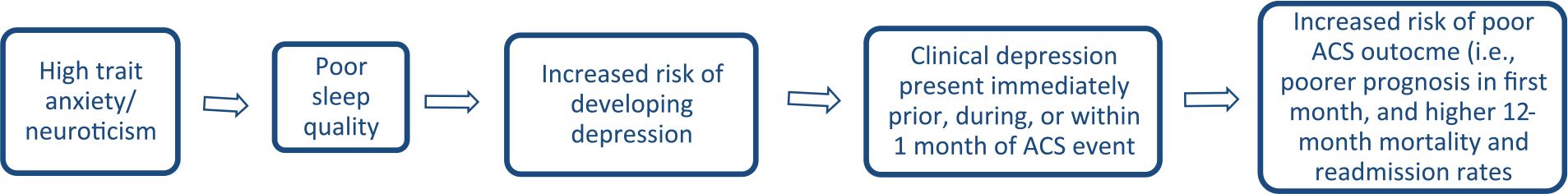
Proposed relationship between biological, psychological and physical vulnerabilities on risk of poorer cardiac outcome.
A meta-analysis (Celano et al., 2015) of 44 articles (involving 30,527 subjects), evaluating the prospective relationship between anxiety and mortality in patients with coronary artery disease, showed that anxiety was associated with a moderate increase in mortality risk (OR = 1.21 per SD increase in anxiety) and that the effects were attenuated by clinical covariates such as depression. In addition, anxiety was insufficient as a prognostic indicator when measured in the immediate post-ACS period, relative to those in a stable condition. However, the analysis examined anxiety as a non-specific construct, without differentiating varying types (e.g. phobic vs non-phobic anxiety) and therefore may not have been sensitive to potential nuances between potentially helpful and unhelpful anxious behaviors. Thus, in our earlier study of 489 post-ACS patients (Parker et al., 2010), the risk of cardiac death or readmission was halved in patients with generalized anxiety disorder, while those with agoraphobia (which is usually secondary to panic disorder) led to a nearly threefold increased risk of poorer cardiac outcome. Similarly, Ossola et al. (2016) followed 251 patients in the 2 years after their first ACS and reported that baseline anxiety was ‘protective’ and moderated the effect of incident depression on new cardiac events. Thus, anxiety when experienced as ‘apprehensive worrying’ may be beneficial so that such individuals may be more vigilant and more compliant with cardiac rehabilitation. Other expressions of anxiety (e.g. panic anxiety) may exert pathological effects implicated in worsening cardiac outcome.
Limitations
We acknowledge several study limitations including some analyses being underpowered as to not indicate statistically significant differences (i.e. generating a type II error). For instance, patients with ongoing depression showed increased odds of cardiac readmission or death and comparable to those with ACS-related depression, but only the latter group differed significantly and presumably reflecting the larger sub-group size (with the ongoing depressed group only comprising 14 subjects). Second, we did not study any impact of treatment on past or current depressive episodes. Third, the current sample was predominantly male (80%), and while men generally appear more likely to experience ACS (Worrall-Carter et al., 2016), a larger representation of women might have allowed any gender-based differences to be clarified. In addition, this paper focused on the relationship between depression and cardiac health, and while some additional variables were considered (e.g. anxiety and sleep), we did not extensively examine the impact of all potential determinants on cardiac outcome (e.g. social support).
Summary and conclusion
Our data quantified that 12-month mortality rates were strikingly and historically low (2.3%), as might now be expected in modern Western countries with sophisticated cardiac management, and therefore argues against mortality being the only or key outcome variable in such studies. Conversely, 12-month cardiac hospital readmission rates (21%) were relatively consistent with other studies undertaken over the past two decades and argue for this to be the principal outcome variable in future studies. Our analyses established that depression present immediately prior, during or within 1 month after an ACS event is the strongest predictor of a poor 12-month cardiac outcome, albeit with the risk of a poor prognosis being relatively higher in the first month. Although an LT history of depression was associated with a poor medical outcome, this association is likely to reflect those with an LT history of depression being more likely to develop depression at the time of the ACS event—and with depression at that time only (rather than its historical experience) influencing a poor outcome. Our pursuit of biomarkers identified poor sleep quality as the only candidate—which might provide a direct risk—or be a consequence of high trait anxiety and neuroticism. Thus, certain expressions of anxiety may both dispose to depression and act so as to increase the risk of a poor cardiac outcome, rather than ‘depression’ per se around the time of the cardiac event being the determining factor. The role of poor sleep quality and neuroticism on worse cardiac outcomes, independent of the effects of depression, need to be examined further.
Footnotes
Acknowledgements
The authors thank Matthew Hyett for study assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This project was funded by a grant from the New South Wales Department of Health Infrastructure funding (Australia). This study was supported by a National Health and Medical Research Council (NHMRC) Program Grant (1037196). The funding source was not involved in the design, collection, analysis, interpretation or dissemination of the presented data.
