Abstract
Objective:
Current evidence from neuroimaging data suggests possible dysfunction of the fronto-striatal-limbic circuits in individuals with bipolar disorder. Somatosensory cortical function has been implicated in emotional recognition, risk-taking and affective responses through sensory modalities. This study investigates anatomy and function of the somatosensory cortex in euthymic bipolar women.
Methods:
In total, 68 right-handed euthymic women (bipolar disorder = 32 and healthy controls = 36) between 16 and 45 years of age underwent high-resolution anatomical and functional magnetic resonance imaging during the mid-follicular menstrual phase. The somatosensory cortex was used as a seed region for resting-state functional connectivity analysis. Voxel-based morphometry was used to evaluate somatosensory cortical gray matter volume between groups.
Results:
We found increased resting-state functional connectivity between the somatosensory cortex and insular cortex, inferior prefrontal gyrus and frontal orbital cortex in euthymic bipolar disorder subjects compared to healthy controls. Voxel-based morphometry analysis showed decreased gray matter in the left somatosensory cortex in the bipolar disorder group. Whole-brain voxel-based morphometry analysis controlled by age did not reveal any additional significant difference between groups.
Conclusion:
This study is the first to date to evaluate anatomy and function of the somatosensory cortex in a well-characterized sample of euthymic bipolar disorder females. Anatomical and functional changes in the somatosensory cortex in this population might contribute to the pathophysiology of bipolar disorder.
Keywords
Introduction
Bipolar disorder (BD) is a chronic mental illness characterized by the presence of episodic mood symptoms (depression and hypomania/mania) with a prevalence of 1–4% in the general population (American Psychiatric Association [APA], 2013; Merikangas et al., 2011). Despite there being a similar transcultural presentation and its high heritability rates (Kieseppä et al., 2014; McGuffin et al., 2003; Rasic et al., 2014; Wray and Gottesman, 2012), the exact pathophysiology of BD is still undetermined. Probably, the most accepted and replicated neurobiological model from neuroimaging studies suggests possible dysfunction of the fronto-striatal-limbic circuits in the brains of individuals who suffer from BD (Blond et al., 2012; Phillips et al., 2008; Strakowski et al., 2005, 2012; Townsend and Altshuler, 2012).
Resting-state functional connectivity (Rs-FC) is an imaging technique that measures spontaneous low-frequency oscillations of blood–oxygen level–dependent (BOLD) signal to determine points of high temporal correlation between two brain regions in the absence of a specific task (Van den Heuvel and Pol, 2010). Resting-state seed-based analysis provides a functional connectivity map of brain voxels that are correlated through resting-state time series using a predefined ‘seed’ region (Buckner and Vincent, 2007). Investigating spontaneous brain activity in remitted BD subjects (euthymia) might provide an important understanding into the pathophysiology of the disease. Previous studies of Rs-FC in euthymic BD subjects have mainly studied connections between limbic and cortical regions (Reinke et al., 2013) and used the amygdala as a seed point. Studies have shown abnormal connectivity between amygdala and prefrontal regions (Anticevic et al., 2013; Torrisi et al., 2013), amygdala and posterior cingulate cortex (Vargas et al., 2014), as well as, amygdala and supplemental motor area and Brodmann area (BA) 5 (Brady et al., 2016).
Our group recently studied a well-characterized sample of females with BD during euthymia to investigate Rs-FC of cortical brain networks including the default mode, frontoparietal and meso-paralimbic networks (Syan et al., 2017). Here, we set out to further investigate potential anatomical and functional changes of the somatosensory cortex in euthymic BD individuals using voxel-based morphometry (VBM) and seed-based Rs-FC. The somatosensory cortex or postcentral gyrus corresponds to BAs 3, 1 and 2 (Brodmann, 1994) and not only receives and integrates sensory information from the body (Borich et al., 2015) but is also involved with the capacity to sense internal bodily responses (Critchley et al., 2004), recognition of emotional facial expression (Adolphs et al., 2000) and risk-taking behavior in females (Zhou et al., 2014). Notably, a recent large neuroimaging meta-analysis of over 6000 subjects revealed that the somatosensory cortex is one of the sensory regions involved with affective-related activity, suggesting that brain regions originally thought to be involved solely with sensory processes may be also involved with affective regulation (Satpute et al., 2015). Abnormal anatomical volume or function of the somatosensory cortex has been reported in depression (Qi et al., 2014; Schmaal et al., 2016; Tadayonnejad et al., 2015), anxiety disorders (Cui et al., 2016), schizophrenia (Koutsouleris et al., 2015; Woodward et al., 2012) and obesity (Contreras-Rodríguez et al., 2015). Our group has recently reported increased cortical thinning in the somatosensory cortex of high-risk adolescents with a parent with BD (Hanford et al., 2016). However, to the best of our knowledge, no imaging study has specifically focused on somatosensory structure and function in individuals with BD.
Aims of the study
The objective of this study was to evaluate the gray matter (GM) volume and Rs-FC of the somatosensory cortex in a well-characterized sample of euthymic women with BD. We hypothesized that women with BD will display (1) decrease in GM volume in the somatosensory cortex and (2) abnormal Rs-FC between somatosensory cortex and limbic regions associated with affective regulation compared to age-matched controls.
Material and methods
Participants
This study was approved by the Hamilton Integrated Research Ethics Board (HiREB) and was conducted in accordance with the Helsinki Declaration. All participants provided written consent to participate in the study. Participants were recruited through community-based advertisements in the Hamilton and Halton regions, Ontario, Canada. Subjects were 68 right-handed women (32 subjects diagnosed with BD and 36 healthy controls [HCs]) between 16 and 45 years of age, with regular menstrual cycles (25–32 days). Exclusion criteria for BD subjects included (1) current mood episode (depression, hypomania/mania), (2) current psychiatric comorbidities, (3) changes in psychotropic medications or mood state within 2 months prior to study entry and (4) presence of unstable medical conditions. Exclusion criterion for HCs was the presence of any lifetime psychiatric disorders and unstable medical conditions. Exclusion criteria for all women included (1) current or recent (last 3 months) use of any systemic hormonal treatment, (2) pregnancy, (3) contraindications for magnetic resonance imaging (MRI), (4) history of head trauma resulting in a loss of consciousness, (5) neurological disorders affecting cognition and (6) current or recent (in the last 6 months) alcohol or drug abuse or dependence.
Psychiatric history was evaluated using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders (4th ed., text rev.; DSM-IV-TR) Axis I Disorders (SCID-I; First et al., 2007) followed by a gynecological clinical history. MRI data were acquired during the mid-follicular phase (days 5–10) of the menstrual cycle to avoid potential premenstrual worsening of mood (Frey and Minuzzi, 2012; Smith and Frey, 2016). Menstrual cycle phase was confirmed by two consecutive months of prospective charting using the Daily Record of Severity of Problems (Endicott et al., 2006) and sex hormonal assays (Syan et al., 2017). Clinical psychiatric questionnaires included the Montgomery–Asberg Depression Rating Scale (MADRS; Montgomery and Asberg, 1979) to assess severity of depressive symptoms, the Young Mania Rating Scale (YMRS) to assess severity of hypomanic/manic symptoms (Young et al., 1978) and the State Trait Anxiety Inventory (STAI) to assess anxiety symptoms (Spielberger et al., 1983).
Data acquisition
MRI data were acquired using a GE whole-body short-bore 3T scanner with eight parallel receiver channels (General Electric, Milwaukee, WI). Anatomical images were acquired with high-resolution T1-weighted images (gradient-echo inversion-recovery sequence, repetition time [TR] = 1.6 seconds, echo time [TE] = 5 milliseconds, matrix 256 × 256 × 128, field of view [FOV] = 220 × 220 mm2, slice thickness 1 mm). Eyes-open functional resting-state imaging data were acquired using a T2* interleaved echo-planar imaging (EPI) sequence with TR = 2000 milliseconds, TE = 40 milliseconds, flip angle = 60°, 4-mm-thick, 29 axial slices, matrix 64 × 64 resolution over 256-mm FOV. Once positioned in the scanner, subjects were instructed to ‘lay still, relax and try not to think about anything in particular’ as they looked at a fixation point. The full imaging scan lasted 10 minutes.
Data analysis
Resting-state seed-based analysis
The anatomical and functional resting-state MRI data were preprocessed using the Statistical Parametric Software SPM12 (www.fil.ion.ucl.ac.uk/spm). Imaging data were obtained in DICOM file format and converted to NIFTY-1. High-resolution T1-weighted anatomic data were segmented into GM, white matter (WM) and cerebrospinal fluid (CSF) using affine regularization (light bias regularization = 0.001, bias Full width AT half maximum [FWHM] = 60 mm cutoff), according to the International Consortium for Brain Mapping (ICBM) space template for European Brains (Ashburner and Friston, 2015). Subsequently, images were normalized into standard space using a fourth-degree B-spline interpolation and resampled to 1 mm × 1 mm × 1 mm voxels (Friston et al., 1995a). Deformation fields created during the segmentation process were used during normalization for increased precision in the alignment of images (Ashburner and Friston, 1997). Resting-state time series were then realigned to correct for minor head motions during scan session (motion greater than 3 mm in the translational plane and 3° in the rotational plane were discarded). Realignment parameters included second-degree B-spline interpolation for estimation and fourth-degree B-spline interpolation for reslicing (Friston et al., 1996). Co-registration of functional images with anatomical images was estimated using the normalized mutual information function (Ashburner and Friston, 1997). Images were spatially smoothed to increase the signal-to-noise ratio with a 8-mm FWHM Gaussian filter (Friston et al., 1995b).
After preprocessing, resting-state functional magnetic resonance imaging (Rs-fMRI) seed-based analysis was carried out using the CONN toolbox v.15.d (www.nitrc.org/projects/conn) with diagnosis as a factor and age as a covariate. Subject-specific maps of WM and CSF were used as nuisance regressors. The aCompCor strategy was employed within CONN to control for the effects of physiological motion and residual head movement (Behzadi et al., 2007; Whitfield-Gabrieli and Nieto Castañón, 2012). The first two volumes were discarded to account for T2 stabilization effects. The function images were then temporally band-pass filtered (0.008–0.09 Hz). The primary somatosensory cortex (defined as BAs 3, 1 and 2) and the somatosensory association areas (BA5) were used as seed points in a region-of-interest (ROI) analysis using the BA and Harvard-Oxford Atlas available with CONN (Desikan et al., 2006). The first- and second-level statistical analyses were completed using CONN using two-sample t-tests comparing functional activation between groups. Brain Rs-FC was corrected for multiple comparisons using the false discovery rate (FDR; p < 0.05).
Since BD and HC subjects differed in body mass index (BMI), MADRS, YMRS and STAI scores (Table 1), we tested any potential effects of BMI and these clinical variables on Rs-FC by adding BMI and MADRS, YMRS and STAI scores as second-level covariates. BD subtype and number of comorbidities were also added as second-level covariates.
Demographic and clinical characteristics of study sample (N = 68).
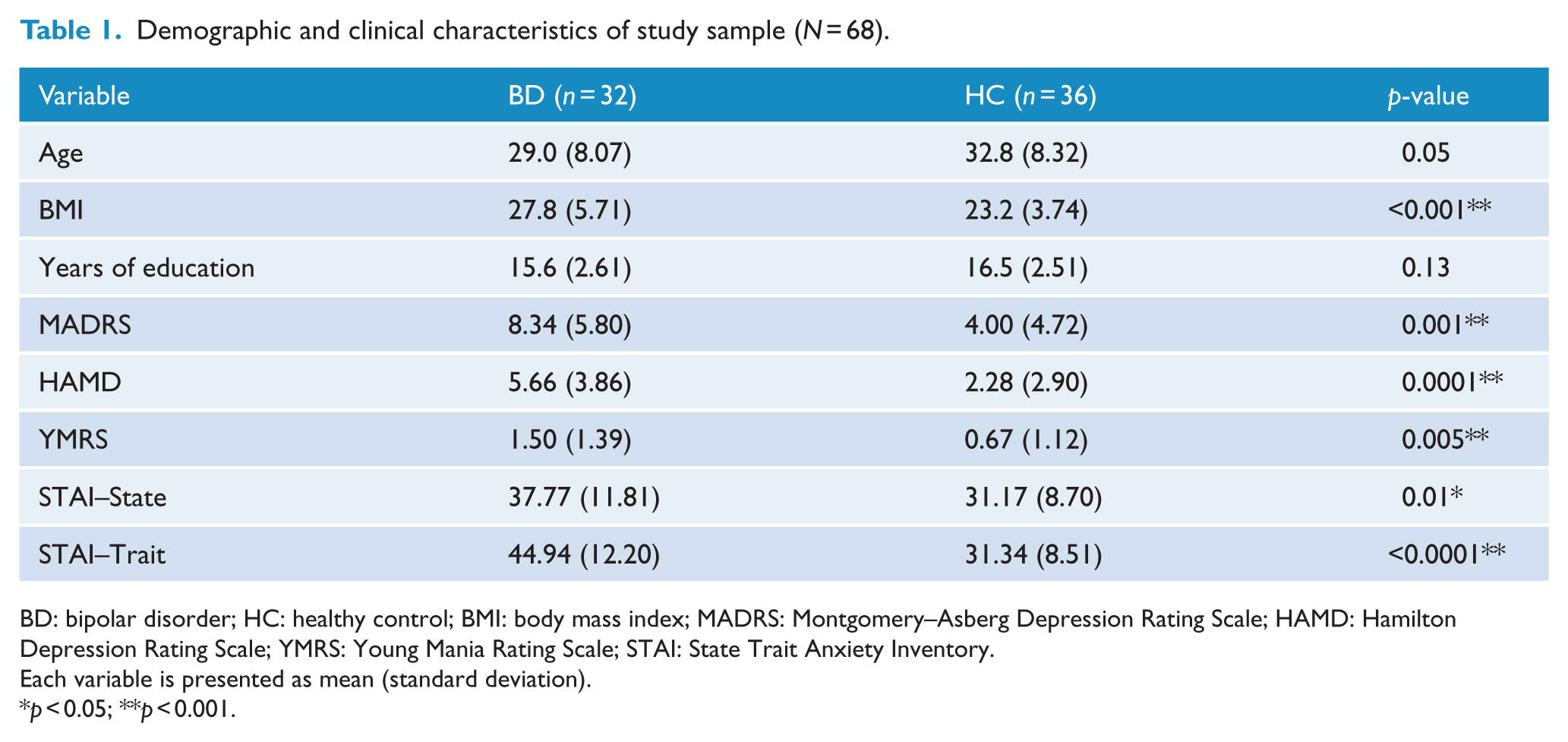
BD: bipolar disorder; HC: healthy control; BMI: body mass index; MADRS: Montgomery–Asberg Depression Rating Scale; HAMD: Hamilton Depression Rating Scale; YMRS: Young Mania Rating Scale; STAI: State Trait Anxiety Inventory.
Each variable is presented as mean (standard deviation).
p < 0.05; **p < 0.001.
In order to evaluate the potential effect of medication on Rs-FC in BD subjects, medication load was analyzed according to the method described by Hassel et al. (2008). Briefly, psychotropic medication load was calculated by categorizing each medication according to its class (lithium, anticonvulsants, antidepressants, antipsychotics and anxiolytics) and dose (Davis and Chen, 2004; Hassel et al., 2008; Reference, 2016; Sackeim, 2001). Then, the composite score of total medication load for each BD subject was added as a covariate in the Rs-fMRI second-level analysis.
Additionally, one study reported an association between Rs-FC in somatosensory cortex and current or past history of psychosis in BD subjects (Anticevic et al., 2013). To examine the potential effect of history of psychosis on Rs-FC in our sample, we also added the presence of psychotic episode in the past as a second-level covariate.
VBM analysis
GM VBM analysis was conducted using the VBM (VBM8) toolbox (www. neuro.uni-jena.de/vbm/) in SPM 12. During preprocessing, high-resolution T1-weighted anatomical images were bias-corrected, registered into a standard space (MNI template) using linear (12-parameter affine) and nonlinear transforms (Ashburner and Friston, 2015), and cerebral tissue was segmented into GM, WM and CSF. Quality control of preprocessed data was carried out by visually inspecting for artifacts and assessing the homogeneity of variance and covariance matrices using VBM8 toolboxes. Images were then spatially smoothed using an 8-mm FWHM Gaussian filter. The volume of the somatosensory cortex was compared between groups using a general linear model (GLM) using group as a factor and age as a covariate. GM volumes were corrected for multiple comparisons using the FDR (p < 0.05).
Statistical analysis
Statistical analysis of demographic and clinical variables was carried out using R (version 3.1.2, www.r-project.org). Shapiro–Wilk’s test and Bartlett’s test were used to evaluate normal distribution of continuous variables and homogeneity of variances between groups, respectively. Clinical domains were analyzed using two-sample t-tests or a Mann–Whitney U test when applicable. A p-value of <0.05 was considered statistically significant.
Results
Demographics and clinical data
Demographics and clinical data are presented in Table 1. There was no difference in age (p > 0.05) or years of education (p = 0.13) between BD subjects and HCs. Although BD subjects were clinically stable and euthymic for a minimum of 2 months, they showed higher depressive and hypomanic/manic symptoms. In addition, BD subjects had higher average BMI than controls (p < 0.001). In total, 18 BD subjects had a diagnosis of BD type I and 14 were BD type II. The average age of illness onset was 18.63 years (±6.8 years). Furthermore, the average number of psychotropic medications used was 1.8 medications. The most common psychotropic was atypical antipsychotics (used by 16 subjects), followed by anticonvulsants (15), antidepressants (12), anxiolytics agents (6), lithium (3) and sleeping aids (2).
Seed-based functional connectivity
Using the somatosensory cortex as a seed point, euthymic BD subjects showed an increased Rs-FC between right somatosensory cortex (BA3) and insular cortex (BA 13, beta coefficient = 0.25, t = 4.41, p = 0.008, FDR corrected), inferior prefrontal gyrus (BA 47, beta coefficient = 0.21, t = 4.10, p = 0.012, FDR corrected) and orbitofrontal cortex (BAs 10, 11 and 47, beta coefficient = 0.20, t = 3.74, p = 0.028, FDR corrected; Figure 1). No changes in Rs-FC were observed in other primary somatosensory areas (BAs 1 and 2) or in the somatosensory association areas (BA 5). There was no effect of BMI, MADRS, YMRS or STAI scores on Rs-FC. There was no effect of BD subtype or number of comorbidities on Rs-FC.
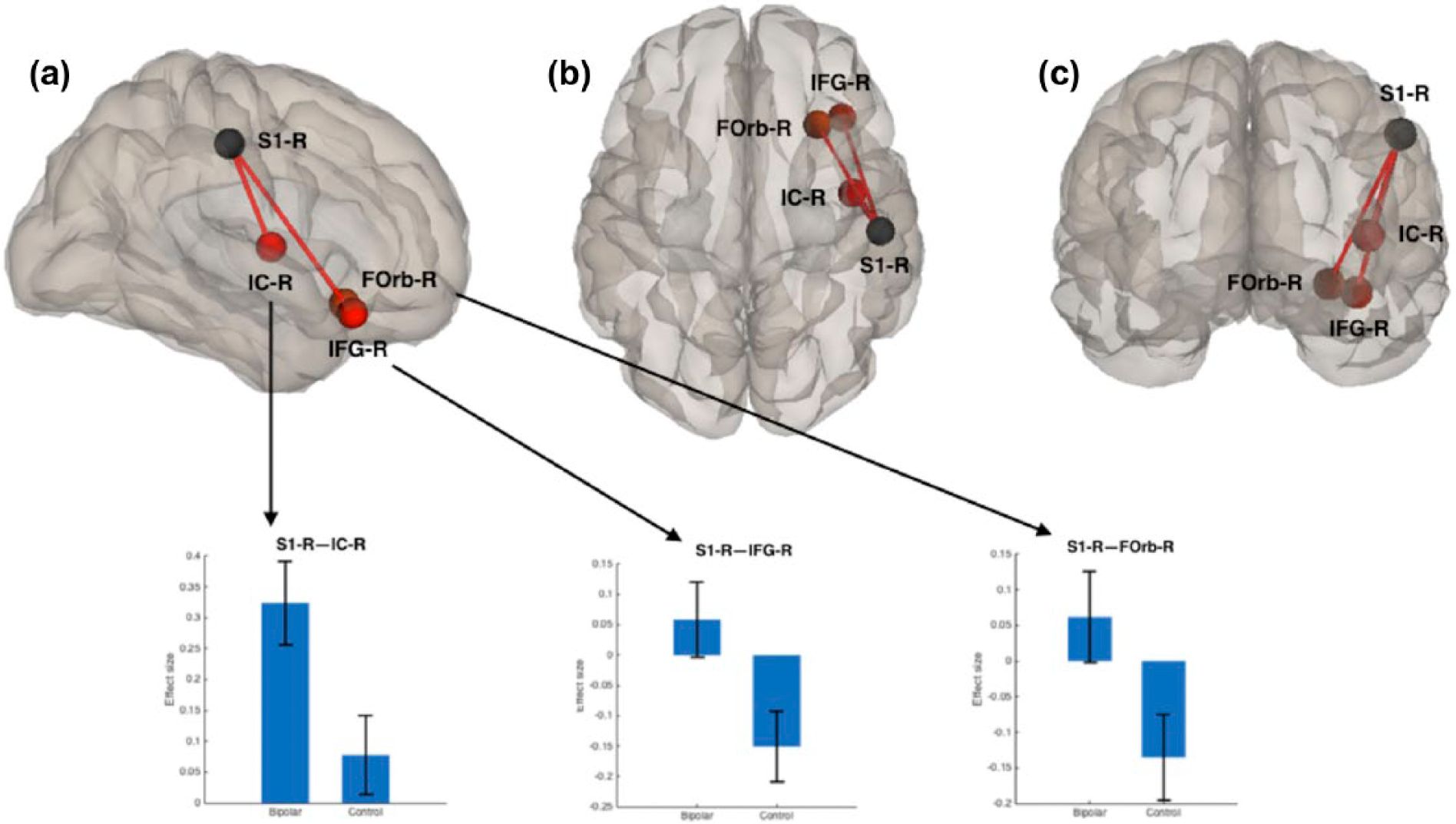
Schematic representation of the resting-state fMRI analysis using the somatosensory cortex as seed point (S1-R). Euthymic BD subjects showed an increased functional connectivity between right somatosensory cortex (S1-R) and right insular cortex (IC-R), inferior prefrontal gyrus (IFG-R) and frontal orbital cortex (Forb-R; all analysis p < 0.05, FDR corrected). Lower row shows the effect size of the connectivity between the sensorimotor cortex and (a) the insular cortex, (b) the inferior prefrontal gyrus and (c) the frontal orbital cortex in bipolar subjects and healthy controls.
VBM
VBM analysis showed decreased GM volume in the left somatosensory cortex (BA1) in the BD group compared to controls (MNI coordinates: −62, −13, 28; cluster size = 298 voxels; peak-level punc < 0.001; cluster-level punc < 0.05; Figure 2).
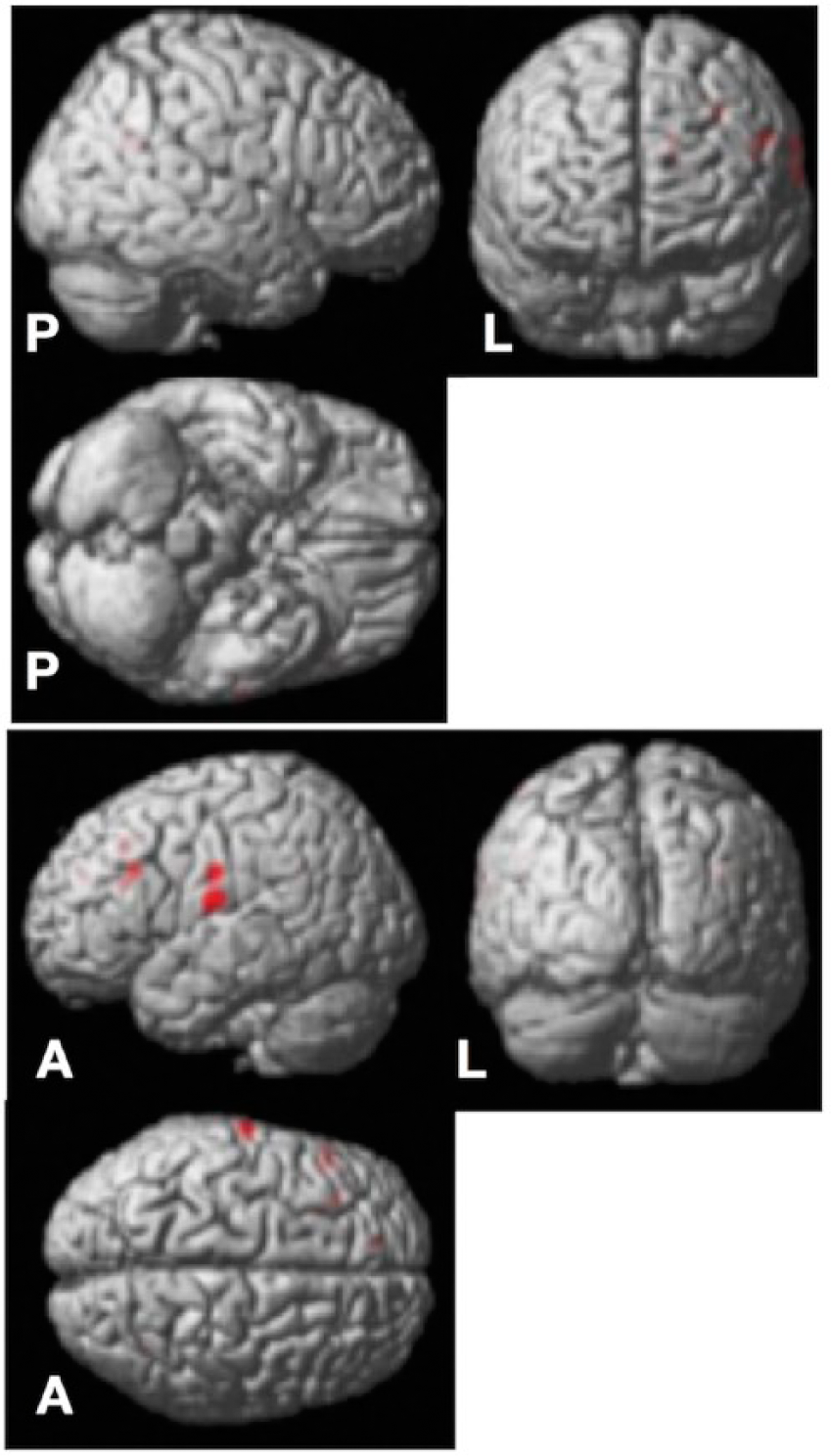
Voxel-based morphometry analysis showed decreased gray matter in the left postcentral gyrus of individuals with bipolar disorder compared to healthy controls (peak-level punc < 0.001; cluster-level punc < 0.05). Another region that showed decreased gray matter in individuals with BD included the left BA9 (MNI coordinates: −10, 45, 28; cluster size = 113 voxels; peak-level punc < 0.001; cluster-level punc < 0.05), which is part of the left dorsolateral prefrontal cortex (PFC).
Discussion
The main findings of this study were that euthymic BD females showed increased Rs-FC between the right somatosensory cortex and frontolimbic regions involved with affective regulation (insular cortex, inferior frontal gyrus and orbitofrontal cortex). Furthermore, VBM revealed decreased GM volume in the left somatosensory cortex in the BD group compared to controls.
Most seed-based Rs-FC studies in euthymic BD have focused on functional connectivity using the amygdala as a seed point. Overall, these studies support the view that euthymic BD subjects present abnormal Rs-FC between amygdala and prefrontal cortical regions (Anticevic et al., 2013; Brady et al., 2016; Torrisi et al., 2013; Vargas et al., 2014). Functional changes in somatosensory cortex in BD have been demonstrated using task-based fMRI techniques. Malhi et al. (2017) used similar inclusion criteria (i.e. only euthymic female BD subjects) to investigate brain changes using a modified word-based memory task to implicitly affective changes. They found less activation in the right somatosensory cortex, left inferior parietal lobule, right thalamus and left putamen in euthymic BD subjects compared to controls when induced to negative affect, which supports that changes in somatosensory activation occur during affective/emotional processing. In another study, task-based fMRI for attention with emotional distracters was used to evaluate depressed BD participants, individuals with major depressive disorder (MDD) and HCs. The depressed BD group showed decreased BOLD activation in the occipital lobes, lingual gyrus and middle temporal gyri compared to the MDD group and HC. However, during the attentional task, depressed BD subjects showed less activation in the somatosensory cortex compared to MDD. Interestingly, increased activation in somatosensory cortex was the only finding that differentiated MDD and HC (Cerullo et al., 2014). Thus, both task-based fMRI studies in BD subjects suggest changes in activation of the somatosensory cortex in BD during verbal memory and attentional tasks.
Functional changes in somatosensory cortex were also found during mood episodes in BD. One study investigated the correlation between whole-brain Rs-FC, default mode network (DMN) and the bipolarity index (BI; Sachs, 2004) in 15 subjects with MDD and 15 with BD type I. They found a negative correlation with BI and DMN in the left postcentral gyrus (Ford et al., 2013). A study which included 26 depressed BD participants using amplitude of low-frequency fluctuations (ALFF; Zang et al., 2004) of Rs-fMRI showed individuals with BD depression had decreased ALFF in the left postcentral gyrus, the left parahippocampal gyrus and the cerebellum (Liu et al., 2012). Altinay et al. (2016) evaluated Rs-FC using striatal regions as seed points in unmedicated BD subjects (during episodes of depression and hypomania/mania) and HCs. They found that only in the BD depression group, there was an increased Rs-FC between the putamen and somatosensory areas (Altinay et al., 2016). Overall, Rs-FC changes in somatosensory cortex in BD subjects during a depressive episode have been correlated to abnormal connectivity with the striatum and other limbic regions specifically during depressive episodes.
Studies of GM volume in adult euthymic BD subjects have mainly reported decreased GM volume in frontolimbic regions in BD, with the exemption of a single study that did not find any difference in GM volume between euthymic BD participants and HCs (Scherk et al., 2008). Almeida et al. (2009) found reduced GM volume in the rectal gyrus, parahippocampal gyrus and left putamen in euthymic BD subjects (Almeida et al., 2009). Interestingly, GM in the rectal gyrus had a significant group by gender by trait anxiety interaction. Lyoo et al. (2006) enrolled 25 BD participants and evaluated GM cortical thickness using voxel-wise and ROI analyses. Using a voxel-wise approach, they found cortical thinning in the somatosensory cortex, middle frontal cortex, pregenual and dorsal anterior cingulate cortex, posterior cingulate cortex and middle occipital cortex compared to HCs. ROI analysis confirmed findings from voxel-wise analysis and revealed a negative correlation between cortical thickness and illness duration on the right somatosensory cortex and in the left middle frontal cortex (Lyoo et al., 2006). One study reported that BD subjects showed decreased GM volume in the ventromedial PFC compared to controls. However, only the BD type I group presented widespread GM reductions in the frontal, temporal, parietal and parahippocampal cortices compared to controls. Interestingly, this study also reported decreased GM volume in the right postcentral gyrus (somatosensory cortex) in BD compared to controls (Ha et al., 2009). Adler et al. (2005) found increased GM volume in a BD participant group in several regions including the anterior cingulate, ventral PFC, fusiform gyrus and primary and supplementary motor cortex. They also found decreased GM volume in the superior parietal lobule in the euthymic BD group. One study in a pediatric BD population in different mood states (BD = 32) using ROI analysis showed decreased GM volumes bilaterally in the parietal lobe and on the left temporal lobe. Within the parietal lobe, the somatosensory cortex was significantly smaller in children with BD compared to HCs (Frazier et al., 2005). Pharmacotherapy with lithium was also suggested to have an effect in the somatosensory cortex in one study: GM volume of the somatosensory cortex, the subgenual anterior cingulate gyrus, hippocampus, amygdala complex and the insula was greater in individuals with BD on lithium treatment compared to other mood stabilizers (Germaná et al., 2010). In summary, consistent with our finding, a number of previous studies have consistently found decreased GM volume or thinning in the somatosensory cortex of individuals with BD. However, most of these studies seem to have ignored the potential relevance or this particular brain region in the neurobiology of BD despite positive findings.
The somatosensory cortex has been directly implicated in the recognition of emotions from facial expressions. A study including 108 subjects with focal brain lesions assessed the recognition and naming of six basic emotions from facial expressions. The right somatosensory cortex was found to be a critical component along with the amygdala and right visual cortices in retrieving socially relevant information from faces (Adolphs et al., 2000). Zhou et al. (2014) evaluated the risk propensity in males and females using Rs-FC and found that general risk propensity was different between the sexes, with the right secondary somatosensory cortex being involved in risk processing in women only (Zhou et al., 2014). The somatosensory cortex was also found to be involved in decision-making processes when using emotional and value-based tasks compared to a mathematical approach (Hsu and Goh, 2016). A recent neuroimaging meta-analysis examined the connection between affective responses through sensory modalities. They observed that in the somatosensory cortex, the affect-related activity was greater for auditory, olfactory, gustatory and somatosensory inputs (Satpute et al., 2015). Taken together, these results strongly suggest that the somatosensory cortex plays an important role in emotional processing and regulation and perhaps in impulse control.
We found increased Rs-FC between the somatosensory cortex and insular cortex, inferior frontal gyrus and orbitofrontal cortex. The insular cortex has been associated with several functions such as emotional processing, speech and pain perception (Nieuwenhuys, 2012). Changes in Rs-FC between somatosensory and insular cortices have been reported in subjects with BD during a mood episode (Altinay et al., 2016). In that study, the increased Rs-FC between those two regions was possibly associated with increased salience of internal and external negative events present during the mood episode (Altinay et al., 2016). In our study, the participants with BD were all euthymic, which may indicate a trait related rather than mood state related. Although no study to our knowledge has investigated Rs-FC between the somatosensory cortex and orbitofrontal cortex in BD, a study has found increased Rs-FC between the orbitofrontal cortex and visual cortex (BA 18) in mania relative to euthymia (Brady et al., 2016). This was not seen between individuals with BD during euthymia and HCs (Brady et al., 2016). Aberrant functional connectivity at rest and during task-based activity (Townsend and Altshuler, 2012) in various mood states may provide evidence for its role in the trait-based pathology of BD.
Our findings of increased spontaneous Rs-FC between the somatosensory cortex and frontolimbic regions (insular cortex, inferior frontal gyrus and orbitofrontal cortex) might also be associated with changes in associative WM tracks in BD. The somatosensory cortex is anatomically connected with frontocortical regions and the cingulate cortex mainly through the cingulum tract, the superior longitudinal fasciculus and anterior arcuate fasciculus (Catani and De Schotten, 2015). Interestingly, these association tracks have been consistently shown to be disrupted in BD (Duarte et al., 2016). We speculate that the changes we found in the somatosensory cortex in this study may in part contribute to the symptomatology or represent part of the psychopathology of BD.
Our anatomical and functional results in euthymic subjects with BD were located in contralateral hemispheres. While we found increased Rs-FC with the right somatosensory cortex seed points, the decrease in GM volume was located in the left somatosensory cortex. A possible explanation for our contralateral findings might be related to the functional interhemispheric connections between somatosensory cortices. Disbrow et al. (2001) used fMRI and magnetoencephalography to evaluate the integration of unilateral and bilateral stimulation in the human somatosensory cortex, and they found evidence of strong functional intrahemispheric processing between the somatosensory cortices. Lipton et al. (2006) used a very high-field fMRI (7T) in primates to evaluate somatosensory cortex activation after unilateral stimulation. They found not only the expected contralateral fMRI response but also an ipsilateral inhibitory response in the somatosensory cortex (BA3b) suggesting a modulatory inhibitory effect on contralateral input processing in the somatosensory cortex.
The limitations of this study include the cross-sectional design that prevents any interpretation related to cause and effect. Although we did not find an effect of medication load on Rs-FC, it is important to note that medication sub-analysis was assessed using composite medications scores. Thus, we were not able to evaluate the influence of specific (individual) medications on Rs-FC due to the limited sample size. A further limitation is that the population is composed of only females. Whether or not our results are also applicable to males, it remains to be determined.
This study is the first to specifically examine the structure and function of the somatosensory cortex in a well-characterized sample of euthymic BD females compared to age-matched controls. We found increased Rs-FC between the somatosensory and insular cortex, inferior prefrontal gyrus and frontal orbital cortex in euthymic BD subjects compared to controls. We also found decreased GM volume in the left somatosensory cortex in the BD group. Future research should consider using emotional regulation tasks along with fMRI to evaluate the impact of changes in somatosensory connectivity and the inclusion of male subjects to investigate sex differences. The somatosensory cortex may be an interesting target for future research using neurostimulation, such as repetitive transcranial magnetic stimulation, in the treatment of BD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.N.F. has received grant/research support from Alternative Funding Plan Innovations Award, Brain & Behavior Research Foundation, Canadian Institutes of Health Research, Hamilton Health Sciences Foundation, J. P. Bickell Foundation, Ontario Brain Institute, Ontario Mental Health Foundation, Society for Women’s Health Research, Teresa Cascioli Charitable Foundation, Eli Lilly and Pfizer and has received consultant and/or speaker fees from AstraZeneca, Bristol-Myers Squibb, Canadian Psychiatric Association, CANMAT, Daiichi Sankyo, Lundbeck, Pfizer, Servier and Sunovion, outside the submitted work. L.M. reports grants from Ontario Mental Health Foundation, J. P. Bickell Foundation, Alternative Funding Plan Innovations Award, Brain & Behavior Research Foundation, Canadian Institutes of Health Research, Hamilton Health Sciences Foundation and Ontario Brain Institute and personal fees from Bristol-Myers Squibb, Lundbeck, Sunovion Pharmaceuticals, Canadian Psychiatric Association and Canadian Network for Mood and Anxiety Treatments. S.K.S., G.B.C.H., A.H. and M.S. all declare that they have no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Ontario Mental Health Foundation (type A grant—Dr Frey) and from J. P. Bickell Foundation (medical research grant—Dr Frey).
