Abstract
Bipolar disorder (BD) is one of the leading causes of disability worldwide. While many recent advances have occurred in understanding the disorder, its pathophysiology remains elusive. Recent advances in neuroimaging technologies have made it possible to investigate structural and functional abnormalities in the brain of bipolar patients
The inconsistency in the volumetric studies of brain structures is quite striking. For example, temporal lobe volumes of BD patients vary considerably across different studies [2–5]. This inconsistency also applied to amygdale volume [6, 7]. The discrepancy may in part be attributable to different methods and insufficient image quality used to study brain volumes in early studies. Regions of interest (ROI)-based structural magnetic resonance imaging (MRI) approaches have been widely used in morphometric studies, but measurement methods differ, and bias is unavoidable because of rater subjectivity in measuring ROI. Furthermore, the selection of ROI is based on assumptions that may not have empirical validity.
Voxel-based morphometry (VBM) is an assumption-free method [8], which applies voxel-wise comparisons throughout the brain to detect differences in gray matter (GM) concentration in different study groups, providing objective and operator-independent results. Optimized VBM analysis has been developed to allow comparisons of volumes [9] rather than concentrations of brain regions that are used in traditional VBM protocol, by incorporating a modulation step and improving the quality of segmentation with a customized template.
A number of studies of BD using VBM have been published [10–16]. Conflicting results suggest that although regional brain changes are present in bipolar patients, they can be affected by different populations and sample sizes of the study, as well as different clinical features of BD patients studied. Given the lack of consistent findings, we undertook this study to replicate some of the reported findings using optimized VBM in closely matched patients and controls. We broadly hypothesize that GM abnormalities will be present in the temporal lobes of BD patients. Further, we wished to examine the relationship between abnormalities in GM and the clinical features of mood disorder.
Method
Subjects
Twenty-four patients (six male) aged 19–59 years (mean = 38.21 years, SD = years) with DSM-IV bipolar I disorder were recruited from the Prince of Wales Hospital in Sydney and a specialist Bipolar Disorder Clinic. Diagnoses of the patients were made by research psychiatrists based on the Structured Clinical Interview for DSM-IV Patient Edition (SCID-P) [17], supplemented by case note review. Subjects were excluded if they had a history of ongoing substance misuse, neurological disease or closed head injury, or had an additional Axis-I or Axis-II DSM-IV diagnosis, or a medical disorder currently necessitating treatment. Patients were compared with volunteers recruited through local advertisement. Twenty-five control subjects were screened for personal or family history of neurological or psychiatric disorder by Structured Clinical Interview for DSM-III-R Nonpatient version (SCID-NP) [17], which has been used previously. They underwent the same clinical assessments and self-report questionnaires as the patients immediately prior to scanning. The Prince of Wales Hospital and University of New South Wales human research ethics committees approved the study and all participants provided written informed consent.
All participants were interviewed and clinical information regarding age, sex, years of education, medical history, duration of illness and medications was collected. Patients who had at least one first-degree relative with an affective disorder were defined as having a positive family history. Psychosis was defined if hallucinations and/or delusions had occurred during at least one affective episode for BD patients. At the time of MRI scanning, 12 patients were taking lithium (mean daily dose = 975.1±213.2 mg) with a mean plasma level of 0.77±0.1 mmol L−1. Four of those patients taking lithium were also receiving sodium valproate. Eight patients were taking valproate, carbamazepine or a combination of the two, and the remaining four patients were unmedicated.
MRI acquisition
Imaging was conducted on a 1.5 T GE Signa scanner (GE Medical Systems, Milwaukee, WI, USA). A 2-D scout mid-sagittal cut for anterior commissure-posterior commisure (AC-PC) plane alignment was first acquired. 3-D structural MRI was acquired using a T1-weighted fast spoiled gradient echo (FSPGR) sequence (TR/TE = 12.2ms/5.3ms; flip angle = 25o; matrix size = 256×256; field of view = 250×250 mm, yielding coronal slices 1.6 mm thick with no gaps and an in-plane spatial resolution of 0.977×0.977 mm pixel−1.
Voxel-based morphometry
Optimized VBM analysis [9] was applied using SPM2 (Wellcome Department of Cognitive Neurology, Institute of Neurology, London, UK) on Matlab 6.5 (Math Works, Natick, MA, USA). All imaging analyses were performed on a Windows 2000 workstation. Before VBM analysis was conducted, all images were inspected to ensure their quality for further analyses. A customized template was created from all included images by normalizing, averaging and smoothing. The structural MRI images were spatially normalized and segmented to create a GM template. Raw structural images were segmented into GM, white matter (WM) and cerebrospinal fluid (CSF) partitions. The total GM volumes, WM volumes and CSF volumes were computed by adding the GM, WM and CSF segments of images in native space for each subject. GM partitions were then normalized into the GM template to estimate optimal normalization parameters, which were reapplied to the original raw images so as to generate whole brain images in stereotactic space. These optimally normalized images were segmented to create GM partitions, which were modulated by multiplying voxel values with the Jacobian determinants derived from spatial normalization, to obtain absolute volume of GM [9]. Finally, to satisfy the Gaussian distribution assumption for statistical analysis to test regional differences, images were smoothed with a 12 mm FWHM Gaussian kernel.
Statistical analysis
Total GM volumes of the BD and control groups were compared using ANCOVA, controlling for age, sex and total intracranial volume (TIV). Regional GM volume differences were detected by applying an ANCOVA to form statistical parametric maps. Age, sex and TIV were included in the model as covariates to avoid confounding effects of these variables. VBM analyses were applied further to examine regional GM abnormalities in relation to clinical features, for example family history of mood disorder, psychotic symptoms, lithium taken or not, episodes of mania and depression and duration of disease. For each clinical feature, ANCOVA models were used to investigate effects of discrete variables on GM, and multiple regression models were used to examine effects of continuous variables on GM. Statistical threshold was set initially at p < 0.001, uncorrected with extended threshold of 200 contiguous voxels [10, 16] and further at p < 0.05, family-wise corrected for multiple comparisons over the whole brain [18], which was chosen to control type I error rate in multiple comparisons conservatively. Anatomical localization of results such as cerebral lobes and Brodmann areas (BA) were determined by an automatic labelling method [19].
Results
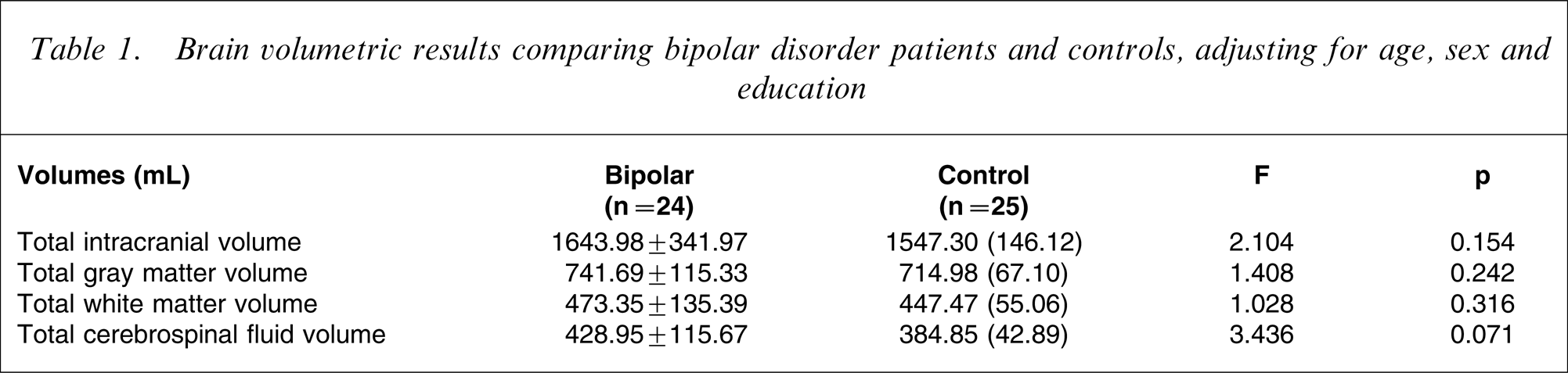
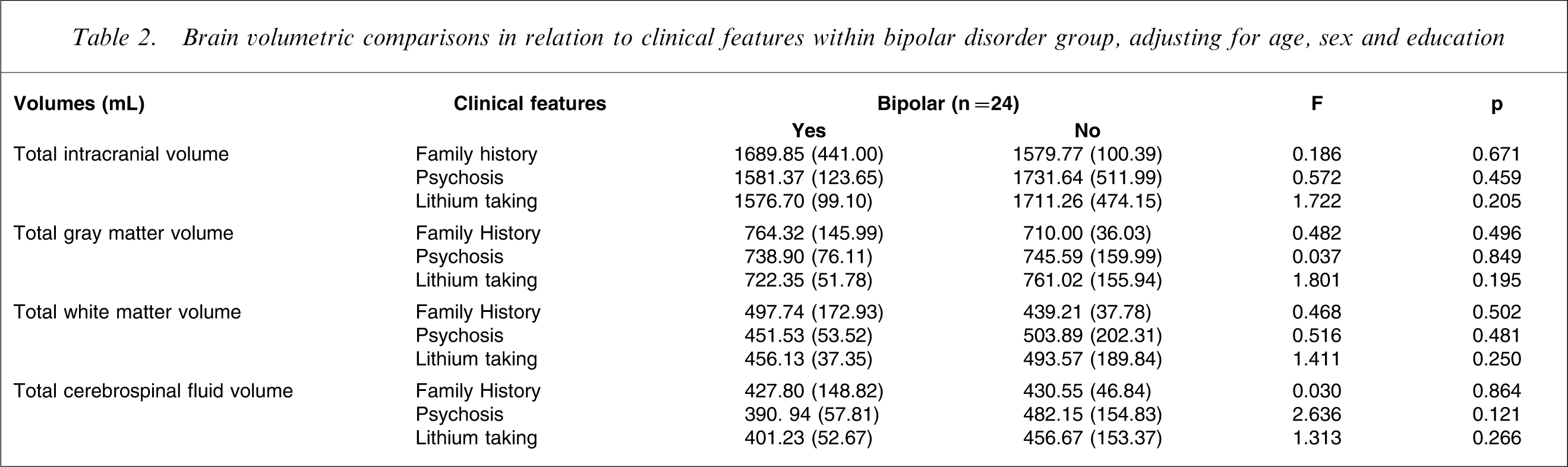
Twenty-four BD patients (mean age = 38.21 years, SD = 11.04 years) and 25 control subjects (mean age = 38.44 years, SD = 11.05 years), including six and seven male patients, respectively, were recruited into the study. BD group and control group did not differ in age, sex or years of education (p < 0.05). All subjects were right-handed. Fourteen patients in the BD group (58.33%) had positive family history, and the same number presented with psychotic features. BD patients had an average illness duration of 14.17±10.26 years, with 20.92±18.56 episodes of mania or depression. At the time of writing, 12 of them (50%) were on lithium and 21 (87.5%) were on mood stabilizing psychotropic medication. Total brain measures of BD patients were not different from the control group before or after controlling for age, sex and education (Table 1). The comparison of total brain measures within subgroups in BD and controls is given in Table 2. There was no significant difference in total brain volumetric measures in relation to clinical features between BD group and controls.
Brain volumetric results comparing bipolar disorder patients and controls, adjusting for age, sex and education
Brain volumetric comparisons in relation to clinical features within bipolar disorder group, adjusting for age, sex and education
The results of VBM analyses are presented in Table 3, Table 4 and Figure 1. Compared to healthy subjects, patients with BD had greater left parahippocampal gyrus GM volume (x = − 17, y = − 19, z = − 22, puncorrected<0.001) and less left middle temporal gyrus GM volume (x = − 44, y = − 74, z = 13, pcorrected<0.05). No increase in GM volume was found in BD subjects as compared to controls.
SPM (T) map depicted in a glass brain, showing (a) reduced gray matter volumes and (b) greater gray matter volumes in bipolar subjects compared with controls, after controlling effects of age, total intracranial volume and sex. Cluster extension set at 200 contiguous voxel, significance threshold of p < 0.001, uncorrected for multiple comparison.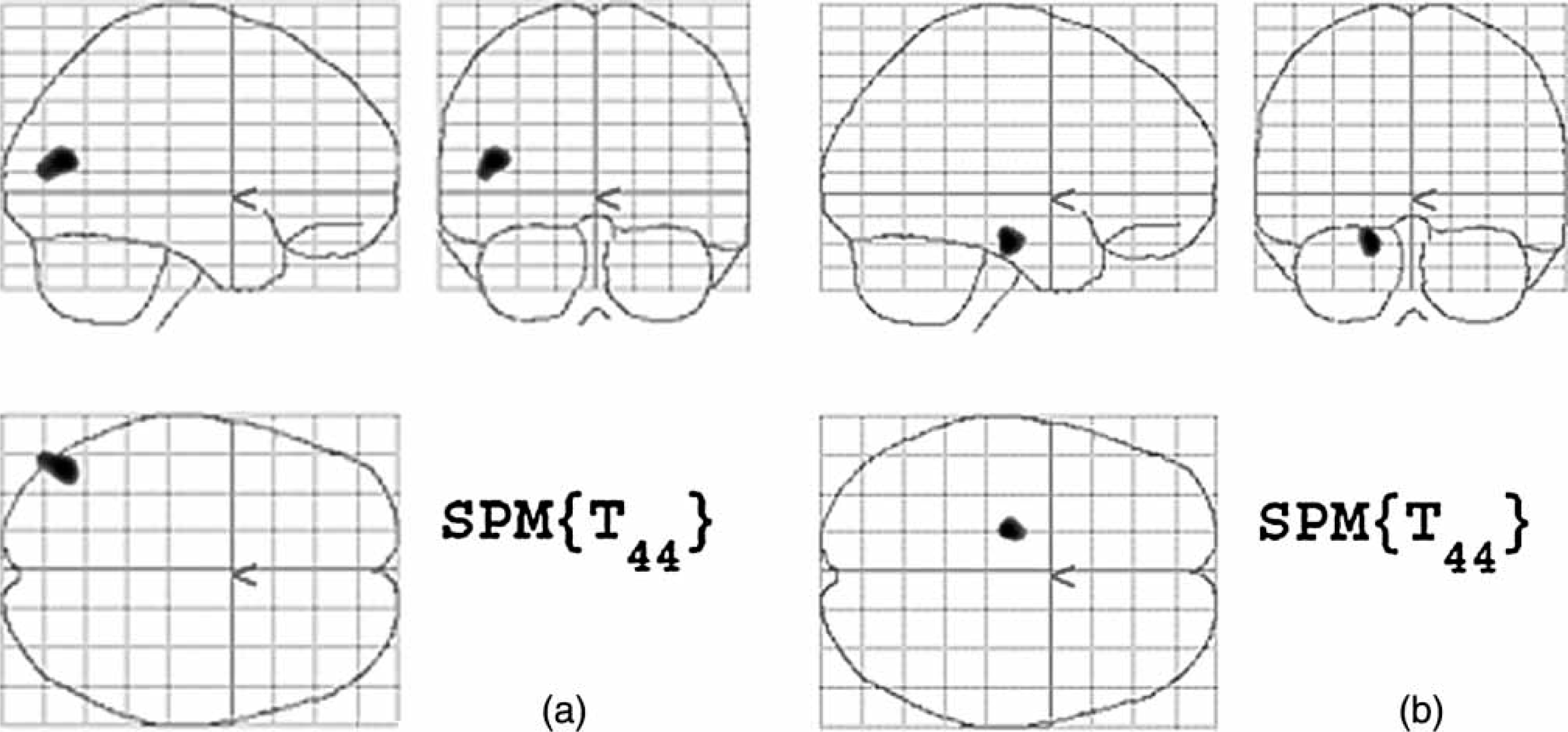
VBM analyses of patients with different clinical features vs control subjects
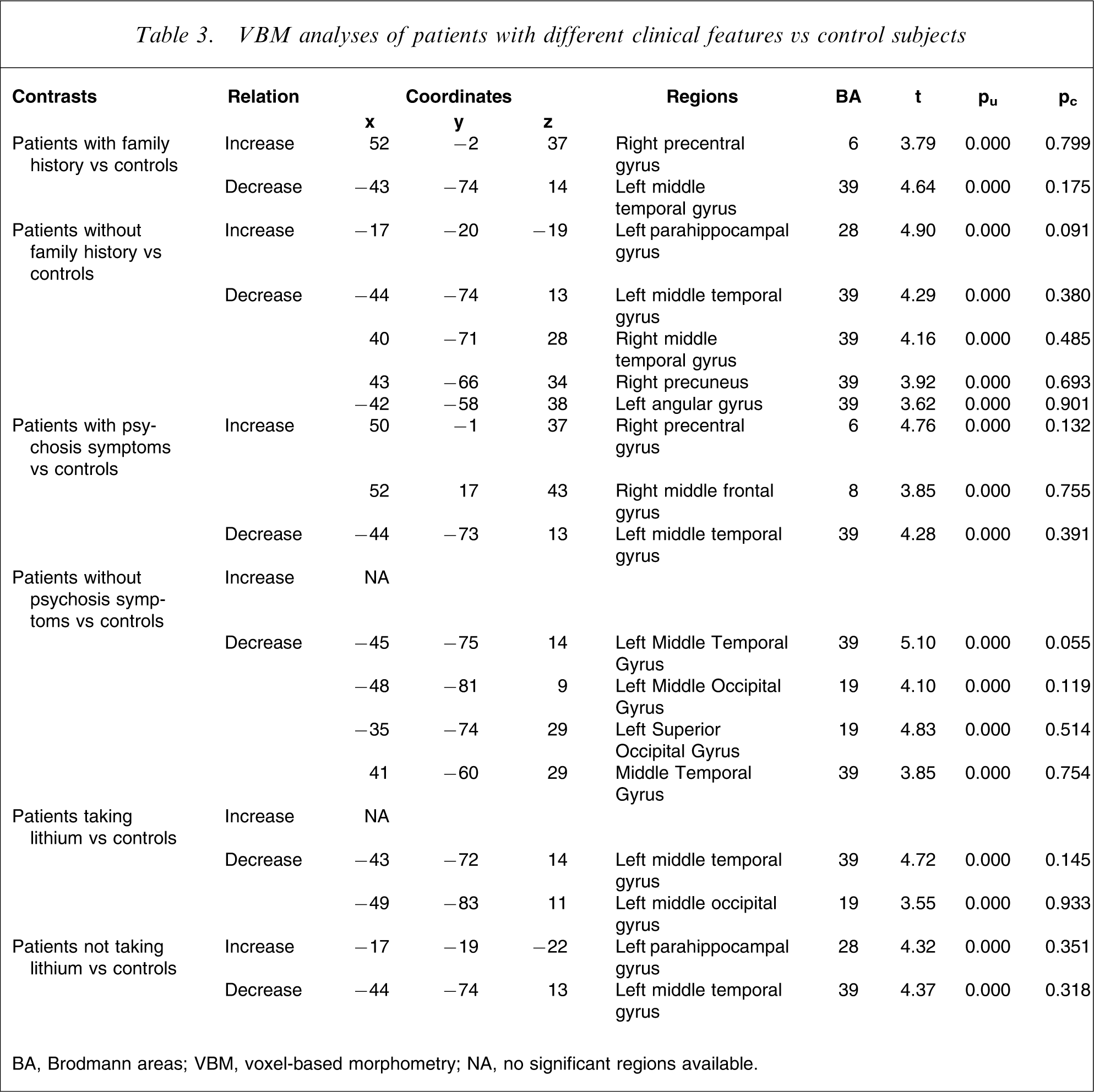
BA, Brodmann areas; VBM, voxel-based morphometry; NA, no significant regions available.
VBM analyses of clinical features within BD patients
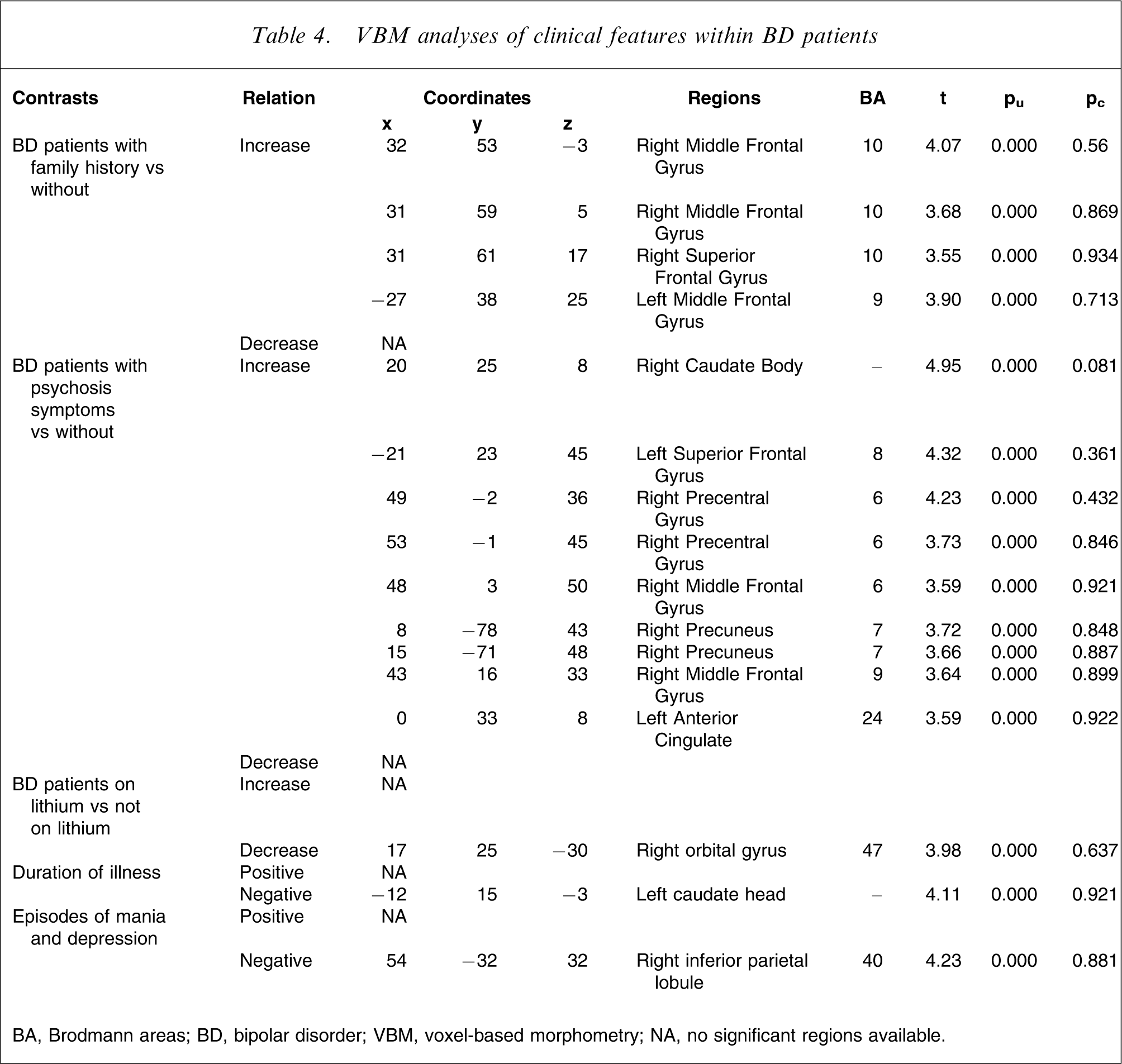
BA, Brodmann areas; BD, bipolar disorder; VBM, voxel-based morphometry; NA, no significant regions available.
In the VBM analyses of BD patients with different clinical features compared with healthy controls, family history, psychotic symptoms and being on lithium had various effects on regional GM abnormalities (Table 4). Reduced GM in left middle temporal gyrus was found in all comparisons between subgroups of patients and healthy controls. Analyses within the BD patients found different patterns of GM changes in relation to clinical characteristic variables where initial uncorrected threshold was used. However, differences shown in these regions were not significant after stringent threshold corrections for multiple comparisons over the whole brain (pcorrected<0.05) were applied. We therefore report only in Table 4 clusters thresholded by uncorrected p< 0.001, and only if a cluster consists of >200 voxels, to include clusters that had a low likelihood of being detected by chance. Similar approaches have previously been used in the published literature [10, 16].
Discussion
Global volumetric measures
The present study shows that there was no difference in total intracranial volume, total GM volume and total WM volume between BD patients and healthy controls. This is consistent with previous studies in which generalized changes of brain volumes were not shown by volumetric methods in subjects with affective disorder [20–22], with the exception of one report of ventricular enlargement in BD [23]. A noteworthy aspect of these associations is that the relationship between increased ventricular volumes and BD has been observed mostly in subjects with psychotic symptoms [24], which were not a homogenous characteristic of the present group, with only 58.33% in the present study having had psychotic symptoms. Increased CSF volumes in relation to psychotic features were not observed in subgroup analyses either, and in fact those with psychosis had a trend toward reduced CSF volume. A linear trend for a relationship between ventricular enlargement and number of previous episodes of illness [25, 26], as well as number of hospital stays has been reported [27], suggesting the role of severity of illness and its duration on ventricular size. However, this trend was not observed in the current study. It should be noted that the total volume of CSF space used in the present study, included volumes of ventricular space as well as subarachnoid space, which were examined separately in previous studies. Sulcal widening was not a significant change in BD patients in previous studies [13], which might partly explain the discrepancy between the present study and some others.
Regional abnormalities in BD patients
The main finding of the present study was the GM reduction in the left middle temporal gyrus (BA 39) in BD patients as compared with control subjects. This finding remained significant even after a conservative statistical threshold was applied. Furthermore, it was consistently significant in the VBM comparisons between subgroups of the BD and control groups. Regional abnormalities of GM volume in temporal lobes have been described in previous VBM studies of BD, although anatomical locations had varied. GM reductions were found in bilateral medial temporal lobe structures [10], left ventromedial temporal cortex [11], left amygdala [12] and left superior temporal gyrus [28] of BD patients as compared with controls. For some of these regions, for example the amygdala, there are conflicting findings from ROI methods showing either increased [6, 29, 30] or decreased volume [31, 32]. It seems that there is diversity of change in temporal structures in relation to BD, although different methods might also account for this. In the present study BD patients had reduced GM in the left middle temporal gyrus (BA 39) as well, a novel finding that has not been reported by other VBM studies. Of interest is the positron emission tomography report that this region was activated during both positive and negative emotional processing [33]. Also, healthy subjects, but not BD depressed patients, show activation on functional MRI (fMRI) in this region during positive and negative emotional changes when presented with designed captioned pictures [34]. Clearly, further study is necessary to investigate the role of this region in the development and progression of BD.
Interestingly, increased GM volume was detected in left parahippocampal gyri of BD subjects in the present study, although this result was not significant if a conservative statistical threshold was applied. Frazier
Volumetric morphology in relation to clinical features of BD
As far as we know, this study is the first one that systematically investigated regional GM abnormality patterns in BD patients in relation to a variety of clinical features, using VBM. As suggested by previous studies [38, 39], the use of ‘corrected p’ under SPM for multiple comparisons is a very stringent criterion when applied to structural analyses. In the subgroup analyses, thereafter, we used a more liberal statistical threshold that has been used previously in BD study [16] to increase the sensitivity of detecting GM abnormality in this sample.
Family history
The present study found that patients with positive family history had an increase in right precentral gyrus compared to healthy subjects. This was not present in patients without family history compared to healthy subjects. There are other areas that had decreases in GM in family history-negative patients when compared with controls. Family history-positive patients had more GM bilaterally in the middle and right superior frontal gyri.
Previous studies about volumetric changes in familial BD have reported inconsistent results in subgenual prefrontal cortices [40, 41]. Family history was negatively correlated with right prefrontal cortex in female subjects but positively in male subjects in that study [40]. Increased sulcal and ventricular CSF volumes were also found to be characteristic of male familial BD patients [42]. However, the study did not provide effects of volumetric abnormalities independent of sex or effects of BD patients without familial history [42], which contributed to the ambiguity when explaining their findings.
No previous VBM study has compared regional GM changes in relation to family history in BD. The present study showed that patients with or without family history of BD may have different changes of brain structure. Particularly, the prefrontal region seems to be better preserved in patients with family history than those without. However, whether the changes are related to different genetic factors in these two groups or whether they reflect different illness effects is not known. There was no association between genetic liability and brain structure abnormalities found in a recent genetics study [43]. Another possibility is that effects of family history may lead to an illness with particular vulnerability of certain brain regions, which might be difficult to detect in the early stages.
Psychotic symptoms
It has been suggested that BD and schizophrenia shared some characteristics in clinical symptoms and pathogenesis[44]. BD patients with psychotic symptoms might be a particular subgroup that shows this overlap clinically and neuroanatomically. Early research using MRI showed similar volumetric changes such as decreased cortical GM and increased cortical sulci in schizophrenia as well as in BD, which differed quantitatively [45]. BD patients with psychosis and schizophrenia patients shared some brain morphological changes, for example increase in ventricular size [24], which were not found in non-psychotic BD patients. This result was not repeated in the present study.
We found that psychotic BD patients had more GM in the right precentral gyrus than non-psychotic patients or controls. Moreover, psychotic BD patients had more GM in the right precuneus, middle frontal gyrus, caudate body, left superior frontal gyrus, and left anterior cingulate than non-psychotic BD patients. Although there have been many volumetric studies about BD and schizophrenia, direct comparisons between psychotic and non-psychotic BD are rare. It is difficult to determine if psychotic features imply a brain abnormality that lies in between schizophrenia and BD. However, psychotic and non-psychotic BD present distinct patterns of GM abnormality, which need to be considered when looking into volumetric changes in BD.
Lithium
Neurotrophic effects of mood stabilizers such as lithium and valproate have been reported previously with evidence in brain volumes [46, 47] and molecular changes [48]. A quantitative MRI study detected an average 3% increase in total GM volume in eight BDI patients (history of manic and depressive edisodes) after 4 weeks of lithium treatment [47], but these changes were not replicated in another study [20]. In one VBM study, both medicated BD and control subjects had more GM in the posterior cingulate cortex than unmedicated BD subjects [28], supporting the neurotrophic effects of medication. However, medicated BD, if compared to healthy controls in the same study, had less GM in the lateral orbital cortex, which is not likely be explained by neurotrophic effects. Although this GM loss might come from other confounding factors, for example severity of illness, it could possibly be due to various effects from different medication. We particularly compared BD patients taking lithium with those not, and found regional GM loss in left middle occipital gyrus in patients taking lithium compared with control, and decreased GM in right orbital gyrus compared with patients not taking lithium. The results might suggest regional effects exerted by lithium unrelated to the neurotrophic effects. It should be noted that we did not have sufficient medication-naive subjects in our sample to investigate this effect conclusively. Further studies are needed to elucidate the effect when controlling for other factors in a relatively homogenous group of subjects.
Duration of illness and episodes of mania and depression
In the present subgroup analysis, duration of illness was negatively correlated with GM volume in the left caudate head. Numbers of episodes of mania and depression were negatively correlated with GM in the right inferior parietal lobule. Both findings suggested effects of illness and symptoms on the brain volumetric measures, and imply that discrepancy can potentially arise if illness severity is not taken into account. Previous VBM studies in BD addressed the issue, although one gave a negative result for illness duration [14] and the other reported reduced GM in relation to prior manic episodes in left middle frontal gyrus and right inferior frontal gyrus [15]. Adler
Previous ROI studies have produced discrepant results about brain abnormalities in relation to the course of illness. One study showed increased left temporal lobe in BD with duration of illness [49]. However, cortical thinning of the left middle frontal cortex was found to be related to the increase of duration of illness in BD [50]. The present study found that illness course was related to GM loss in the caudate, which was consistent with some previous results, that showed a larger caudate in male adult BD patients [51] but decreased in older BD patients [52]. Likewise, one VBM study in BD adolescents also found increased GM in caudate head, thereby supporting the hypothesis of caudate decline in the course of the illness [10]. Although the caudate was reportedly enlarged in several studies [26, 51], basal ganglia have been shown to be correlated with BD inconclusively in terms of volumetric changes of putamen, caudate or globus pallidus [4, 29]. Additionally, functional activities in these regions are observed to be associated with affective states of BD patients, indicating the role of basal ganglia in affective disorder.
Limitations
There are several limitations to the current study. First, the relative small sample size prevented us from further investigating the interactions of contributing factors such as duration of illness and type of medication. Second, the statistical power of the results is limited, given the number of factors being studied within a moderate sample size. We therefore chose a conservative statistical threshold (pcorrected<0.05). Although in subgroup analyses we used an uncorrected statistical threshold, we reported only clusters with >200 voxels. The findings in subgroup analyses should nevertheless be seen as exploratory. Third, as mentioned here, we need to be cautious when explaining observations of GM differences in bipolar subjects in a cross-sectional study, and the causative deductions are tentative. Although some affected regions are compatible with results from functional studies in fMRI studies, the cause–effect relationship remains uncertain. We also acknowledge that other factors, for example genetic polymorphism and environmental heterogeneity, may be involved in this association. A longitudinal study on a large sample would be crucial in elucidating the neuropathophysiological mechanisms of BD, as well as the influence of mood states, disease progression, and medication on the pattern of regional GM volumes.
Conclusion
The current study using the optimized VBM method presents evidence of structural abnormalities in GM in adult BD, with decreased GM in left middle temporal gyrus and increased GM in left parahippocampal gyrus. Regional variation in relation to different clinical factors was also seen, suggesting a neurobiological basis for symptom profiles in BD patients. From the present study it is not possible to state whether these abnormalities are present early in the course of the disease, or are secondary to disease duration and the effects of medication.
Footnotes
Acknowledgements
The authors wish to thank all the staff of the Neuropsychiatric Institute and the Black Dog Institute who participated in this study, and the patients and volunteers for their generous contributions.
