Abstract
Objective:
Schizophrenia is a devastating mental disorder accompanied by aberrant structural brain connectivity. The question whether schizophrenia is a progressive brain disorder is yet to be resolved. Thus, it is not clear when these structural alterations occur and how they develop over time.
Methods:
In our selective review, we summarized recent findings from longitudinal magnetic resonance imaging studies investigating structural brain alterations and its impact on clinical outcome at different stages of the illness: (1) subjects at ultra-high risk of developing psychosis, (2) patients with a first episode psychosis, and (3) chronically ill patients. Moreover, we reviewed studies examining the longitudinal effects of medication on brain structure in patients with schizophrenia.
Results:
(1) Studies from pre-clinical stages to conversion showed a more pronounced cortical gray matter loss (i.e. superior temporal and inferior frontal regions) in those individuals who later made transition to psychosis. (2) Studies investigating patients with a first episode psychosis revealed a decline in multiple gray matter regions (i.e. frontal regions and thalamus) over time as well as progressive cortical thinning in the superior and inferior frontal cortex. (3) Studies focusing on patients with chronic schizophrenia showed that gray matter decreased to a greater extent (i.e. frontal and temporal areas, thalamus, and cingulate cortices)—especially in poor-outcome patients. Very few studies reported effects on white matter microstructure in the longitudinal course of the illness.
Conclusion:
There is adequate evidence to suggest that schizophrenia is associated with progressive gray matter abnormalities particularly during the initial stages of illness. However, causal relationships between structural changes and illness course—especially in chronically ill patients—should be interpreted with caution. Findings might be confounded by longer periods of treatment and higher doses of antipsychotics or epiphenomena related to the illness.
Introduction
Schizophrenia has been conceptualized as a psychiatric disorder of disrupted structural brain connectivity accompanied by a functional miscommunication between different brain areas (Friston and Frith, 1995; Pettersson-Yeo et al., 2011; Wernicke, 1906). A plethora of neuroimaging studies have shown structural brain alterations in patients with schizophrenia (Fusar-Poli et al., 2013; Haijma et al., 2013; Olabi et al., 2011; Vita et al., 2012). Currently, there is some debate between conflicting models of brain pathology related to the disorder. With respect to the neurodevelopmental model of schizophrenia, these structural brain abnormalities are thought to stem from (early) developmental processes that are linked to genetic, prenatal and environmental factors (Kochunov and Hong, 2014). The post-onset progression model implies that structural brain abnormalities progress over time after illness onset (Andreasen, 2010; Meyer-Lindenberg, 2011). The question whether brain structure is already altered before illness onset might be valuable for prognosis, intervention and treatment, and lead to a better understanding of disorder-related pathophysiological mechanisms (McGuire et al., 2015).
Although numerous structural neuroimaging studies in schizophrenia have been conducted so far, most of them have employed a cross-sectional study design (Haijma et al., 2013). Therefore, it is not clear when these structural alterations occur and how they develop over time. Moreover, the relationship between structural brain alterations and clinical outcome measures has hardly been investigated (Hulshoff Pol and Kahn, 2008). Exploring the dynamics of brain structure is best addressed through longitudinal studies, since it allows for the assessment of the dynamically changing brain throughout the lifespan. Thus, it is conceivable that structural brain alterations might be more pronounced during certain stages of the disorder or are different in specific subgroups (e.g. patients with good- vs. poor-outcome) (Pantelis et al., 2005). In this context, longitudinal studies in individuals at ultra-high risk (UHR) of developing psychosis and patients with first episode psychosis (FEP) are of importance. Studying patients at these stages provide an opportunity to examine the brain structure without the potential confounding effects of antipsychotic medication, or secondary effects such as social deprivation, which might affect longitudinal brain changes in and of themselves (Gong et al., 2016). Therefore, the majority of longitudinal neuroimaging studies have especially focused on individuals with preclinical symptoms (i.e. UHR) or patients with FEP (Fusar-Poli et al., 2012; Olabi et al., 2011). In contrast to these illness stages, studying patients with chronic schizophrenia allows to investigate whether ongoing pathophysiological processes may underlie brain changes. However, effects of antipsychotic medication and factors secondary to the illness—such as substance abuse or lifestyle—on brain structure remain difficult to control (Weinberger and Radulescu, 2016).
To dissect the longitudinal course of the illness into different stages (UHR, FEP, and chronic schizophrenia) might disclose different dynamical and pathophysiological processes that occur in the brain at different times. It has been argued that brain changes appear to be especially prominent in the first years of illness compared to changes in later stages of the illness (Kochunov and Hong, 2014; Schnack et al., 2016; Van Haren et al., 2016).
In our selective review, we included studies exploring brain structure and its impact on clinical or functional outcome in the longitudinal course of the illness. We included studies using longitudinal designs and performing at least two magnetic resonance imaging (MRI) assessments in the same subjects, thus, investigating effects on gray or white matter (WM) over time. First, we reviewed studies that investigated brain structure before and after transition to psychosis in subjects at UHR of developing psychosis. Second, we reviewed studies assessing brain structure at multiple time points after illness onset and examined whether progressive brain changes predict illness outcome in patients with a first episode of schizophrenia as well as in patients with chronic schizophrenia. Finally, we reviewed studies examining the longitudinal effects of medication on brain structure in patients with schizophrenia. The aim of the current review was to disclose different trajectories that occur in the brain at different stages of the illness (Figure 1).
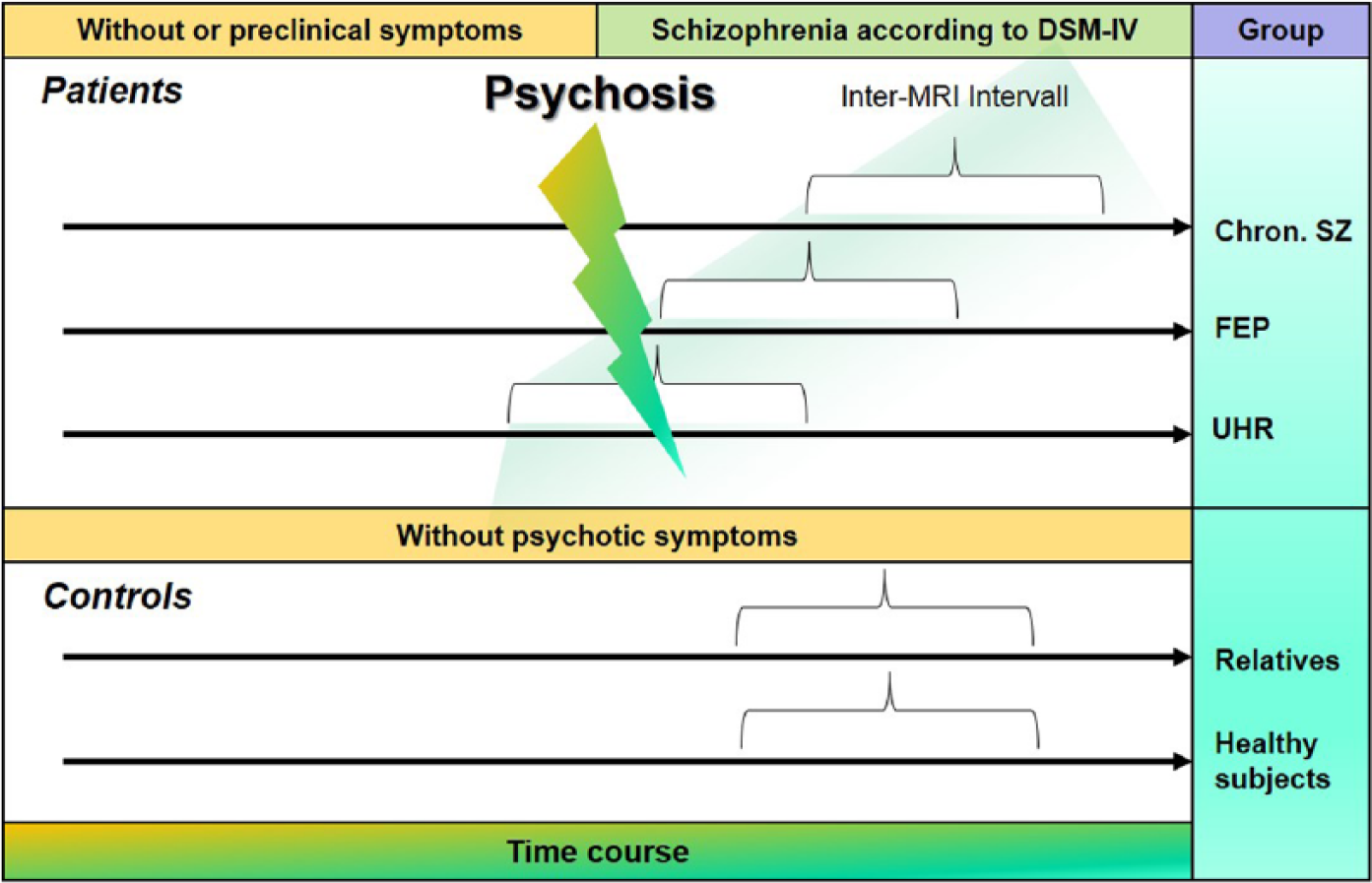
Different stages to explore structural brain changes in the longitudinal course of schizophrenia and in healthy subjects performing at least two magnetic resonance imaging (MRI) assessments in the same subject.
Brain structure before and after transition to psychosis in individuals at UHR of developing psychosis
Progressive gray matter changes as psychosis develops
Of all morphological changes demonstrated by structural neuroimaging studies of patients with schizophrenia, enlarged lateral ventricles (LVs) and reduced regional brain volume in the frontal operculum and lateral temporal lobes (Fornito et al., 2009; Shenton et al., 2001) have been among the most consistent ones. Although these abnormalities are well-established in patients with schizophrenia, the timing of their manifestation is debated. Structural MRI studies in individuals at UHR of developing psychosis provide important insight into the neurobiological basis of the risk for and the conversion to the disorder. Several prospective longitudinal studies have tracked changes in neuroanatomical parameters in UHR subjects from pre-clinical stages through to conversion to manifest illness (Borgwardt et al., 2008; Cannon et al., 2015; Pantelis et al., 2003; Sun et al., 2009; Takahashi et al., 2009a, 2009b; Walter et al., 2012; Ziermans et al., 2012). In all these studies, more pronounced cortical gray matter loss has been detected in those UHR individuals who later made a transition to psychosis relative to healthy controls and relative to non-converters (Borgwardt et al., 2008; Cannon et al., 2015; Pantelis et al., 2003; Sun et al., 2009; Takahashi et al., 2009a, 2009b; Walter et al., 2012; Ziermans et al., 2012). Sun et al. (2009) found the onset of psychosis to be significantly associated with progressive cortical volume loss in the right prefrontal cortex within a 1-year follow-up interval. The significant decrease in gray matter volume in prefrontal regions was confirmed in other studies (Borgwardt et al., 2008; Pantelis et al., 2003; Ziermans et al., 2012), and these findings have been extended to include other regions such as the inferior temporal, medial and superior parietal cortex, left fusiform gyrus, precuneus, the cingulate gyri and cerebellar cortices (Borgwardt et al., 2008; Pantelis et al., 2003; Ziermans et al., 2012). One study also reported longitudinal changes in gray matter volume in UHR subjects who do not convert after at least 12 months; however, this was restricted to the cerebellum (Pantelis et al., 2003). The variations in results between these studies may be related to methodological factors including relatively small sample sizes (Ns of 8 to 12 converters), use of varying inter-scan interval (1–2 years) and different approaches to correcting for multiple comparisons voxel-wise throughout the brain.
Taken together, these studies provide consistent evidence for greater gray matter decline over time in at-risk individuals who go on to develop the full-blown disorder relative to those who don’t. Meta-analytical evidence suggests that in particular baseline gray matter reductions in superior temporal and inferior frontal areas are associated with later transition to psychosis (Fusar-Poli et al., 2011). However, the underlying mechanisms of these progressive gray mater changes have thus far not been sufficiently explored. Follow-up scans for converting UHR cases in all longitudinal MRI studies were performed after subjects had made a transition to psychosis. Most of the converters and some of the non-converters received antipsychotic drug treatment during the interscan interval. Gray mater volume reductions may thus be a secondary phenomenon caused by factors related to the pathophysiology of schizophrenia or related to use of antipsychotics or to other factors (Frick et al., 2013). However, in a recent large-scale multi-center study (
Longitudinal effects on white-matter and outcome in in individuals at UHR of developing psychosis
Schizophrenia has been found to involve cortico-cortical dysconnectivity, both in terms of functional connectivity between different brain regions (McGuire and Frith, 1996) and in terms of neuroanatomical alterations underlying the functional changes, particularly in WM tracts (Ellison-Wright and Bullmore, 2009). In line with the neurodevelopmental hypothesis of schizophrenia, reduced WM volume and integrity has also been found in early and subclinical stages of the disorder, that is, in first-episode patients (Gasparotti et al., 2009) and subjects at ultra-high risk (Carletti et al., 2012).
To date, only one longitudinal study investigated the time course of WM changes by using diffusion tensor imaging (DTI) to study UHR subjects before and after the onset of illness. They compared WM DTI properties of UHR subjects (
Assessment of brain structure at multiple time points after illness onset
Assessment of brain structure alterations in patients with a FEP in the longitudinal course
The debate as to whether or not schizophrenia is a progressive brain disorder is yet to be resolved (Fusar-Poli et al., 2013; Zipursky et al., 2013). Studies of longitudinal brain changes in patients with FEP using MRI techniques have yielded inconsistent results. Some studies have shown significant decrease in multiple gray matter regions, including frontal regions, the thalamus and total brain volume over time in first-episode schizophrenia patients (up to 15 years) (Andreasen et al., 2011). Others found further progressive cortical thinning in the superior and inferior frontal and, to a lesser extent, superior temporal cortex in patients with FEP within a 2-year follow-up interval (Gutiérrez-Galve et al., 2015). Gray matter loss, resulting from cortical thinning, not from surface shrinkage (cf. Kahn and Sommer, 2015), in FEP has been linked to clinical and cognitive outcomes (Hulshoff Pol and Kahn, 2008; Van Haren et al., 2008, 2012, 2013; Vita et al., 2012). Yet other studies, however, report a lack of progression of gray matter changes (Haukvik et al., 2015; Schaufelberger et al., 2011). Such inconsistencies between studies may in part be explicable by methodological differences. For example, some longitudinal studies used rather narrow inclusion criteria, including patients meeting criteria for schizophrenia, schizoaffective disorder or schizophreniform disorder (e.g. Andreasen et al., 2011), while others included other psychotic disorders such as delusional disorder, brief psychotic disorder, and psychosis not otherwise specified (e.g. Schaufelberger et al., 2011). Further variations between studies include sample sizes, age range of participants, the duration of time that the person has been ill prior to involvement in the study, the duration of treatment prior to entering the study and length of follow-up periods. The lack of consistency between important study variables such as definition of first-episode psychosis, duration of follow-up and treatment regimens makes it hard to interpret and integrate results from different studies. It is also very likely that there are different trajectories in subgroups of patients that have yet to be disentangled.
Longitudinal effects on white-matter and outcome in FEP
WM changes over time in FEP patients have only been investigated by very few studies (Steen et al., 2006). An earlier review concluded that there were no significant longitudinal WM changes and suggested that longitudinal changes in whole-brain volume may be limited to the gray matter in FEP patients (Steen et al., 2006). Two recent studies provided contrasting results. A longitudinal DTI study of previously drug-naïve patients with first episode schizophrenia (FES;
Brain structure in patients with chronic schizophrenia in the longitudinal course
Longitudinal effects on gray matter and outcome in chronic schizophrenia
Studies that have focused on progressive structural brain changes beyond the first episode of psychosis in mostly long-term treated patients with schizophrenia are scarce. Findings from longitudinal neuroimaging studies (follow-up intervals up to 10 years) have shown that whole brain gray matter decreases to a greater extent in patients with chronic schizophrenia compared to healthy subjects. These progressive gray matter losses are most pronounced in widespread frontal and temporal areas, thalamus, caudate nucleus, insula, periventricular area, putamen and cingulate cortices (e.g. DeLisi et al., 2004; Mathalon et al., 2001; Mitelman et al., 2009a, 2009b; Van Haren et al., 2007, 2008, 2016; Veijola et al., 2014; Wood et al., 2001). Some of these studies investigated whether aging, symptom severity, level of functioning or cognition were associated with gray matter reductions over time. Mitelman et al. (2009a, 2009b) divided their sample into a good-outcome and a poor-outcome group depending on clinical and functional outcome measures at baseline. Poor-outcome patients displayed a greater gray matter loss throughout the brain compared to good-outcome patients at follow-up 4 years later. Furthermore, poor-outcome in chronic schizophrenia was associated with progressive loss of volume in the putamen (Mitelman et al., 2009b). For the whole sample, they found an association between Positive and Negative Syndrome Scale (PANSS) subscale scores and gray matter volumes. General psychopathology correlated inversely with gray matter volumes in left parietal areas over time and positive symptoms correlated with longitudinal changes in the subgenual cingulate gyrus (Mitelman et al., 2009a). Van Haren et al. (2008) found that poor-outcome patients (i.e. low level of functioning) showed greater losses of total brain volume compared to good-outcome patients after a 5-year follow-up period. Furthermore, in the same sample Van Haren et al. (2007) found an association between decreased gray matter density in the superior frontal gyrus and number of hospitalization. In another study, more pronounced superior temporal lobe gray matter volume loss was associated with more severe clinical symptoms after a 4-year follow-up interval (Mathalon et al., 2001). However, other studies failed to establish a link between structural brain decline and outcome measures. For example, Veijola et al. (2014) examined brain volume changes and its relation to symptom severity, level of functioning and cognition in participants with schizophrenia and healthy controls in a relatively long follow-up period of almost a decade. Regression models were used to examine whether brain volume changes predicted clinical and cognitive differences over time. Their results indicate that symptom severity, level of functioning and decline in cognition were not associated with brain volume reduction in schizophrenia during a very long follow-up period.
Other longitudinal studies failed to find any progressive changes in gray matter in patients with chronic schizophrenia at all (e.g. Nesvåg et al., 2012; Whitworth et al., 2005; Yoshida et al., 2009). Nesvåg et al. (2012) investigated cortical thickness and (sub-)cortical volume in a sample of 52 chronically ill and long-term treated patients with schizophrenia compared to 62 healthy subjects within a follow-up period of 5 years. In both groups, regional cortical volumes were decreased over time, but there was no significant group by time interaction for any cortical region. Yoshida et al. (2009) also reported no gray matter volume changes over time, neither for the superior temporal gyrus nor the amygdala–hippocampal complex as predefined regions of interest. They studied 16 patients with chronic schizophrenia within a follow-up interval of about 3 years. However, in the schizophrenia group, volume change in the amygdala–hippocampal complex correlated with PANSS negative symptoms. But this study has its limitations: Despite of a small number of patients, duration of illness varied between 1 and 31 years (average 16.3 years), indicating an enormously heterogenic sample. Moreover, the follow-up period differed remarkably between patients with schizophrenia and healthy controls (average scan intervals 3.1 years vs. 1.4 years, respectively). These negative findings may support the assumption that marked progression at the initial stage of schizophrenia might be less pronounced in patients with chronic schizophrenia. In line with these findings, Chiapponi et al. (2013) summarized that some areas within the amygdala–hippocampus complex are affected early during the course of schizophrenia, whereas the superior temporal gyrus appeared to be already impaired at the onset of symptoms. Moreover, the deterioration seems to be particularly prominent during the first 5 years after disease onset and becomes relative more stable after that time (Schnack et al., 2016).
Taken together, some longitudinal MRI studies in patients with chronic schizophrenia found evidence for accelerated loss of gray matter over time. In addition, progressive structural brain changes were more pronounced in poor-outcome patients compared to good-outcome patients. However, there are still inconsistencies between studies that may stem from study quality and methodological issues, sample characteristics as well as quality and frequency of the assessments during follow-up intervals.
Longitudinal effects on WM and outcome in chronic schizophrenia
Until now only very few studies have focused on longitudinal effects on WM microstructure in patients with chronic schizophrenia compared to healthy subjects. Therefore, the knowledge of long-term disorder-related WM microstructure alterations in chronic schizophrenia is still very limited.
In a recent study, Domen et al. (2017) applied DTI and performed tract-based spatial statistics (TBSS) to investigate the longitudinal course of WM microstructure in patients with chronic psychotic disorder (
These findings are partly in line with the results of a study by Mitelman et al. (2009b, 2009c), examining an older sample of patients with chronic schizophrenia (
Taken together, findings from longitudinal studies investigating WM microstructure are far too limited to draw firm conclusions on changes in WM microstructure over time in patients with chronic schizophrenia. Reduced FA in chronically ill patients observed at baseline (Domen et al., 2017; Mitelman et al., 2009b, 2009c) may reflect disturbed WM maturation from an early age, suggesting a neurodevelopmental origin, corresponding with findings from studies in UHR and FEP (see above). Again, those findings are in line with the assumption that the acceleration of structural brain decline might slow down after a critical period (i.e. <5 years of illness duration) (cf. Schnack et al., 2016; Van Haren et al., 2016).
Effects of antipsychotic medication on longitudinal brain volume changes
Antipsychotic medication might alter the natural course of volume changes, i.e., volume differences between patients and controls due to neurodevelopmental insults and/or the natural course of the illness. Although this is of great importance, it is only very rarely addressed in MRI studies, because of the difficulty to sample reliable information about compounds and doses over many years. Therefore, the available data are extremely limited, populations are usually small, and findings must be interpreted with caution. Four reviews have qualitatively compared the structural integrity of brain regions in treatment-naive patients, following short-term treatment administration and after long-term treatment (Moncrieff and Leo, 2010; Navari and Dazzan, 2009; Scherk and Falkai, 2006; Smieskova, et al., 2009). Notably, these reviews are of lower quality, predominantly because they report highly inconsistent data, often from insufficiently controlled studies or studies with small samples, heterogeneous outcome measures or drug exposures. Furthermore, these reviews are presented in a narrative format that is susceptible to reporting bias, and unable to be qualitatively assessed according to Grading of Recommendations, Assessment, Development and Evaluations (GRADE) criteria. The available evidence is particularly conflicting for comparisons across medicated and unmedicated patients in early psychosis. Most consistent evidence supports increased basal ganglia volume following treatment with typical antipsychotics; however, atypical medications have been associated with both increases and reductions of basal ganglia volume, and other inconsistent findings of increased and decreased subcortical regions (including the hippocampus) have been reported for patients taking typical antipsychotics. In patients on long-term antipsychotic treatment, effects of switching antipsychotics have been examined longitudinally. Switching from typical to atypical medication reduced basal ganglia and thalamus volume, but no difference was reported in ventricular or whole brain volume. Some evidence pointed to other class-specific changes, for example, longitudinal changes following typical antipsychotic administration but not following atypical administration; however, over time, the differences between the two types of medication become less clear (Smieskova et al., 2009). Linear associations reported between reductions of regional brain volume and increasing antipsychotic exposures could represent a modulatory effect of the medication on brain structure, but could also reflect a more severe course of illness requiring higher cumulative medication doses (Navari and Dazzan, 2009). Two more recent original studies that had not been included in the above meta-analyses also reported conflicting results, with one study showing volume increases (Van Haren et al., 2011) and the other decreases (Ho et al., 2011). A more recent meta-analysis (Fusar-Poli et al., 2013) included 30 longitudinal MRI studies with antipsychotic administration in schizophrenia patients. Brain volumes before and after antipsychotic exposure, duration of illness, severity of psychotic symptoms and demographic, clinical and methodological variables were considered in the analysis. The overall sample consisted of 1046 schizophrenia patients and 780 controls for a median duration of follow-up of 72.4 weeks. At baseline, patients showed significant whole brain volume reductions and enlarged LV volumes compared to controls. No baseline volumetric abnormalities were detected in the gray matter volumes, WM volumes, cerebrospinal fluid and caudate nucleus. Longitudinally, there were progressive gray matter volume decreases and LV enlargements in patients but not in controls. The gray matter volume decreases were inversely correlated with cumulative exposure to antipsychotic treatment, while no effects were observed for duration of illness or illness severity. Schizophrenia is characterized by progressive gray matter volume decreases and lateral ventricular volume increases. Some of these neuroanatomical alterations may be associated with antipsychotic treatment. Several other recent original studies, two from Finland on the same sample (Guo et al., 2015; Veijola et al., 2014) in 33 patients with schizophrenia form the 1966 Finland birth cohort study showed in a 10-year follow-up brain volume reduction in schizophrenia patients especially in the temporal lobe and periventricular area. Symptom severity, level of functioning and decline in cognition were not associated with brain volume reduction in schizophrenia. The amount of antipsychotic medication (dose years of equivalent to 100 mg daily chlorpromazine) over the follow-up period predicted brain volume loss (
For first episode patients, a voxel-based meta-analysis (Fusar-Poli et al., 2011) compared medication-naive patients with healthy controls, and reported gray matter reductions of superior temporal and insular cortices and cerebellum. However, another voxel-based meta-analysis explicitly considered the effect of medication status in FES (Leung et al., 2011), where reductions in insula, anterior cingulate cortex, middle and inferior frontal and precentral gyri, uncus/amygdala and superior temporal gyrus were found in both medicated and unmedicated FES patients compared to healthy controls. Medicated FES patients were additionally characterized by reductions in the superior frontal gyri, inferior temporal gyrus, posterior cingulate cortex (PCC), claustrum, cerebellum and caudate. Additional reductions in unmedicated FES patients were located in medial frontal gyri, thalamus and parahippocampus.
In summary, the evidence for effects of antipsychotic medication on MRI volume changes is unclear. Residual confounding could also account for the findings and caution must be applied before drawing causal inferences from associations demonstrated in observational studies of moderate size. Volume increases and decreases probably interact in a complicated way with the individual course of the illness. In general, it is yet not clear what kind of volume changes, decreases or increases, are potentially ‘good’ or ‘bad’ for the individual patient.
Conclusion
The current review summarized findings from longitudinal studies exploring brain structure and its association with clinical outcome in schizophrenia at different stages of this disorder. Overall, there is adequate evidence to suggest that schizophrenia is associated with progressive gray matter abnormalities. Due to very few studies that investigated WM alterations, it is not possible to draw firm conclusions on changes in WM microstructure over time.
Dissecting the longitudinal course of the illness in different stages may help to crystalize when these structural alterations occur and how they develop over time. Findings from studies investigating subjects at UHR of developing psychosis suggest that structural brain abnormalities occur prior to full-blown symptoms and progressively worsen as psychosis develops. Since individuals in this early phase of the illness are less likely on medication, there is sufficient evidence that structural brain alterations in those subjects are not confounded by exposure to antipsychotic treatment. Studies assessing brain structure at multiple time points after illness onset showed ongoing structural brain alterations in patients with FEP as well as in patients with chronic schizophrenia over time—especially in those subjects with a poor-outcome (i.e. more severe symptoms and a lower level of global functioning). However, causal relationships between structural changes and illness course should be interpreted with caution. Brain alterations could be a result from pathophysiological processes related to the severe illness. A severe illness itself could also affect brain structure due to a higher likelihood for substance abuse, worse life-style, diminished cognitive challenges or reduced levels of physical activity that are associated with this disorder (Weinberger and Radulescu, 2016). In addition, a more serve illness could be confounded by longer periods of treatment and higher doses of antipsychotics. These confounding factors may affect changes in brain structure independently to the illness. Poor-outcome patients may be more likely to have neurodevelopmental problems which also may independently be related to more pronounced brain alterations.
To clarify these issues, future studies should (1) include larger samples, ideally (2) scan people before any psychiatric symptoms occur which would only be possible by using large population based samples, or first episode patients with a wide variety of diagnoses (schizophrenia and its broadest spectrum, bipolar disorder, major depression) and symptoms, (3) measure at multiple time points (e.g. every 2–5 years) over decades, (4) control for life time risk factors (e.g. maltreatment, urbanicity, high paternal age, cannabis, etc.), (5) control for familial genetic risk, (6) control for medication, social functioning, exercise, smoking, substance use and any other factors that may have influence on the central nervous system (CNS), (7) control for cognitive functioning, (8) use ‘mulit-omics’ biological measures (genome-wide association study [GWAS], epigenetics, proteomics, etc.) to further stratify groups and (9) validate results through replication samples. All this is of course difficult and costly and disentangling the mechanisms underlying structural alterations into primary or secondary epiphenomena of the illness remains challenging. There are, however, large studies currently underway addressing some of these issues (e.g. PSYSCAN, PRONIA, MACS, B-SNIP, UK Biobank, SHIP and others).
Findings from those large-scale projects would result in multi-omics data sets—based on neuroimaging data (e.g. structural, functional, and diffusion MRI) and non-neuroimaging data (e.g. clinical, level of functioning, demographic, cognitive and genetic variables)—that provide input for multi-parametric classification (e.g. by means of graph analysis and machine learning) to stratify subsamples. This should ultimately aim at improving care of those individuals, e.g., at UHR of developing psychosis or the outcome of patients with schizophrenia (Galletly et al., 2016; McGuire et al., 2015). The aim of transferring findings from this sort of research to clinical practice should target early intervention, physical health and psychosocial outcome (Galletly et al., 2016). However, there are many challenges to master for translating these potential results into clinical patient management.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the German Research Foundation (DFG) as part of the Research Unit ‘Neurobiology of Affective Disorders’ (DFG FOR 2107; grant no.: KI 588/14-1). The DFG had no further role in the writing of the review and in the decision to submit the paper for publication.
