Abstract
Objective:
Shared neuropathological features between schizophrenia patients and their siblings may represent intermediate phenotypes of schizophrenia and can be used to investigate genetic susceptibility to the illness. This study aimed to discover regional white matter abnormalities in first-episode schizophrenia (FES) patients and their unaffected siblings compared to healthy subjects in the Chinese Han population using optimized Voxel-Based Morphometry (VBM).
Method:
A total of 51 drug-naive, FES patients, 45 of their unaffected siblings and 59 healthy comparisons were studied with magnetic resonance imaging (MRI).
Results:
FES patients exhibited significant regional white matter deficits in the left inferior frontal gyrus and left joint of external capsule and internal capsule compared with healthy subjects (corrected FDR, p<0.005). The sibling group also showed significant white matter deficits in these two regions compared with the healthy comparison group (uncorrected, p<0.001). White matter deficits with a less stringent threshold for significance in the left cerebellum anterior lobe, left middle frontal gyrus, left hippocampus, right anterior cingulate and right internal capsule were observed in patients compared to their siblings.
Conclusions:
Our findings extend those from previous VBM analyses showing that FES patients and their unaffected siblings may share white matter deficits in the left inferior frontal gyrus and the left joint of external capsule and internal capsule. These regional white matter deficits may be related to genetic factors related to schizophrenia susceptibility.
Introduction
The onset of schizophrenia has been associated with interactions between environmental and genetic factors (Rapoport et al., 2005). Previous studies have found brain abnormalities in the early stage of schizophrenia (Sprooten et al., 2013; Whitford et al., 2007; Guo et al., 2014a; Guo et al., 2014b). Given that the complex interactions between environmental influences and genetic factors may alter the shape and size of developing neural structures (Boos et al., 2007), it is reasonable to hypothesize the existence of structural brain abnormalities in siblings of schizophrenia patients (Zorrilla et al., 1997). In our previous study (Hu et al., 2013), it was found that patients with schizophrenia and their unaffected siblings shared gray matter volume decreases in the left middle temporal gyrus.
Magnetic resonance imaging(MRI) studies have found that schizophrenia patients relative to healthy subjects have white matter abnormalities. Volumetric studies of chronic schizophrenia patients have found a decreased white matter volume in diverse regions including temporal (Okugawa et al., 2002), prefrontal (Wible et al., 2001; Sanfilipo et al., 2000) and occipital cortices (Mitelman et al., 2003). Separately, diffusion tensor imaging (DTI) studies have found that schizophrenia patients showed reduced or increased white matter fractional anisotropy (FA) in different brain regions (Brambilla et al., 2005; Guo et al., 2012; Jones et al., 2006). In contrast to chronic schizophrenia patients, however, relatively few MRI studies have been conducted to identify white matter abnormalities in first-episode schizophrenia (FES) patients. Studies of FES are especially important, since FES is much less confounded by drug effects, and effects of aging, as well as long-term effects of having a chronic illness. However, previous studies of white matter abnormalities produced inconsistent results. For example, many studies have reported white matter abnormalities in FES, including reduced FA in frontotemporal white matter (Szeszko et al., 2005), irregular shape and impaired myelination of the corpus callosum (Flynn et al., 2003; Frumin et al., 2002), and a decreased magnetization transfer ratio in the uncinate fasciculus (Bagary et al., 2003), while other studies have failed to observe any white matter irregularities (Cahn et al., 2002; Salokangas et al., 2002). Furthermore, it remains uncertain whether there are white matter abnormalities in FES that could be revealed with voxel-based morphometry.
Both demyelination and axonal elimination resulting from neuronal death may contribute independently to white matter deficits (Boos et al., 2007). However, it is still unclear that these abnormalities are associated with increased risk of developing the illness and to what extent. This issue can be partially overcome by studying brain structures in siblings of schizophrenia patients. Studies in families of schizophrenia patients have shown that this disorder has a heritability estimates between 80% and 86%, including interactions between environmental and genetic factors (Cardno et al., 1999; Tsuang et al., 2001). Thus, the presence of white matter deficits in siblings of patients is likely related to the shared genetic vulnerability of developing schizophrenia. A meta-analysis has indicated that structural brain volumes in relatives of schizophrenia patients differ from those of healthy subjects (Boos et al., 2007). Several studies in schizophrenia show that a variety of white matter abnormalities might be heritable (Harrison and Weinberger, 2005; Reveley et al., 1982). Thus, neuroanatomical features can be considered as good candidates for identifying endophenotypes of schizophrenia. Endophenotypes are heritable, associated with the illness, primarily state independent, co-segregate within families, and found in unaffected relatives of patients at a higher prevalence than in the general population (Gottesman and Gould, 2003). Determining the nature of brain structural abnormalities in siblings of schizophrenia patients would be useful for endophenotypic research.
Voxel-based morphometry (VBM) is a neuroimaging analysis technique to assess structural differences between MRI images (Good et al., 2001). VBM, a voxel-based method, avoids operational bias in the selection of brain regions and can be used to capture subtle changes in brain structures effectively. Although VBM has been widely applied to study structural brain changes in schizophrenia, the exact nature of detected white and gray matter changes remains poorly understood (Mechelli et al., 2005). The procedure of VBM involves performing spatial normalization, segmenting the white matter from the normalized images, and smoothing white matter segments. As a result of non-linear spatial normalization, the volumes of certain brain regions are either stretched or shrunk. However, what VBM is actually testing for is regional differences in the density of white or gray matter (Ashburner and Friston, 2000). Density should not be confused with cell packing density measured cytoarchitectonically. A meta-analysis of VBM findings by Di et al. (2009) identified white matter deficits in multiple brain regions in schizophrenia, including the right and medial frontal white matter regions and the bilateral internal capsule as regions most commonly found to be abnormal. The results of another meta-analysis suggest that structural brain changes in schizophrenia are partially associated to the risk of developing it and these brain abnormalities may therefore predate the onset of schizophrenia (Boos et al., 2007). However, it is still unclear whether the observed brain white matter changes in recent-onset schizophrenia is a pre-existing state or an acquired damage.
Few VBM studies of white matter in patients who are drug-naive, less than 30 years of age, and experiencing their first episode of schizophrenia have been conducted. Here, we conducted a VBM study to examine whether or not regional white matter abnormalities are present in drug-naive patients with FES and their unaffected siblings. This study aimed to identify neuroanatomical endophenotypes in schizophrenia to further understand genetic contributions to the neuroanatomical characteristic of this disorder and its early identification.
Methods and materials
Subjects
Fifty-one drug-naive, FES patients and 45 of their healthy siblings (HS) were recruited from the Mental Health Central, the Second Xiangya Hospital of Central South University, Hunan province, China. To confirm the diagnosis of schizophrenia, patients were evaluated by the Chinese translation of the Structured Clinical Interview from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) (SCID) (First, 1997), which has been shown to be reliable and valid in China (Zhou et al., 1997). The patients were right-handed, at their first-episode and drug naive. None of the patients had received any other type of treatment such as psychological therapy or electroconvulsive therapy. The duration of untreated psychosis was less than two years. The Chinese version of the Positive and Negative Symptom Scale (PANSS) (Kay et al., 1987; Phillips et al., 1991) was used to assess symptom severity by a senior psychiatrist at the scan time. Fifty-nine healthy subjects were recruited from the community. None of them had a psychiatric history or a family history of schizophrenia and related psychosis and all were matched by age, sex, handedness, and education level to the patients and siblings to the extent possible. Structured interviews based on the Chinese version of SCID were performed to screen the healthy siblings and comparison group to rule out individuals who had mental illness. All of the participants were of Han Chinese ethnicity, aged 16 years to 30 years, and underwent more than nine years of formal education. The exclusion criteria included neurological illness, history of head injury, severe organic disorders, substance-related disorders, family history of mental illness (except for schizophrenia in the unaffected siblings), mental retardation and contraindications for MRI scanning.
The Ethics Committee in the Second Xiangya Hospital of Central South University reviewed and approved the study. For those who agreed to participate in the study, a written informed consent was obtained.
Imaging acquisition
The scan was performed on a 3.0 T Philips scanner (Philips Medical Systems). High-resolution T1-weighted images were acquired with a three-dimensional magnetization prepared rapid acquisition gradient echo sequence (3D-MPRAGE). The whole brain volumetric images were obtained in a sagittal orientation with the following parameters: repetition time=7.4ms, echo time=3.4ms, inversion time=875ms, flip angle=9°, acquisition matrix=228×228, field of view=250 mm×250 mm, slice thickness=1.1 mm, gap=0 mm and slices=301.
MRI data analysis
Structural MRI data were preprocessed and analyzed using the Statistical Parametric Mapping 8 software package (SPM8, http://www.fil.ion.ucl.ac.uk/spm), in particular the VBM8 toolbox (http://dbm.neuro.uni-jena.de/vbm). All structural MRI images were normalized to the same standard stereotactic space by registering each of the images to the same template image. Then those normalized images were segmented for signal intensity and previous probability information. Next the white matter segmented images were normalized to the customized template. The use of intensity modulation on the normalized segmented white matter MRI images depending on whether the surrounding voxels were compressed or expanded in the normalization process. The white matter images were then smoothed using an 8-mm full width at half-maximum FWHM Gaussian kernel, which was made to increase the signal to noise ratio. The anatomical localization of the most significant clusters was determined by using the automated anatomical labeling atlas software and anatomical atlases (Tzourio-Mazoyer et al., 2002).
Statistical analysis
Group comparisons of demographic characteristics were performed by using the Chi-square test or ANOVA, and these analyses were conducted with the Statistical Package for Social Sciences, version 17.0 (SPSS Inc., Chicago, Illinois).Voxel-based group comparisons were carried out using analysis of covariance (ANOVA) in SPM8. An overall F-test was performed first and then followed by post hoc t-tests on significant regions from the F-test analysis. Voxel-based t-tests were computed among the groups for white matter deficits in regions with a significant F-test, contrasting FES versus HC, FES versus HS, and HS versus HC. Statistical inferences were made with a voxel level statistical threshold (P<0.005) after false discovery rate (FDR) correction for multiple comparisons and/or an extent threshold (P<0.001) (uncorrected) with a minimum cluster size of 30 voxels (1 voxel = 1.5×1.5×1.5 mm3).
Results
Demographic and clinical characteristics
Demographic variables and clinical symptoms of study participants are summarized in Table 1. There was no significant difference in sex, age, or years of education among the patient group, the sibling group, and the comparison group.
Demographic and clinical characteristics of three groups.
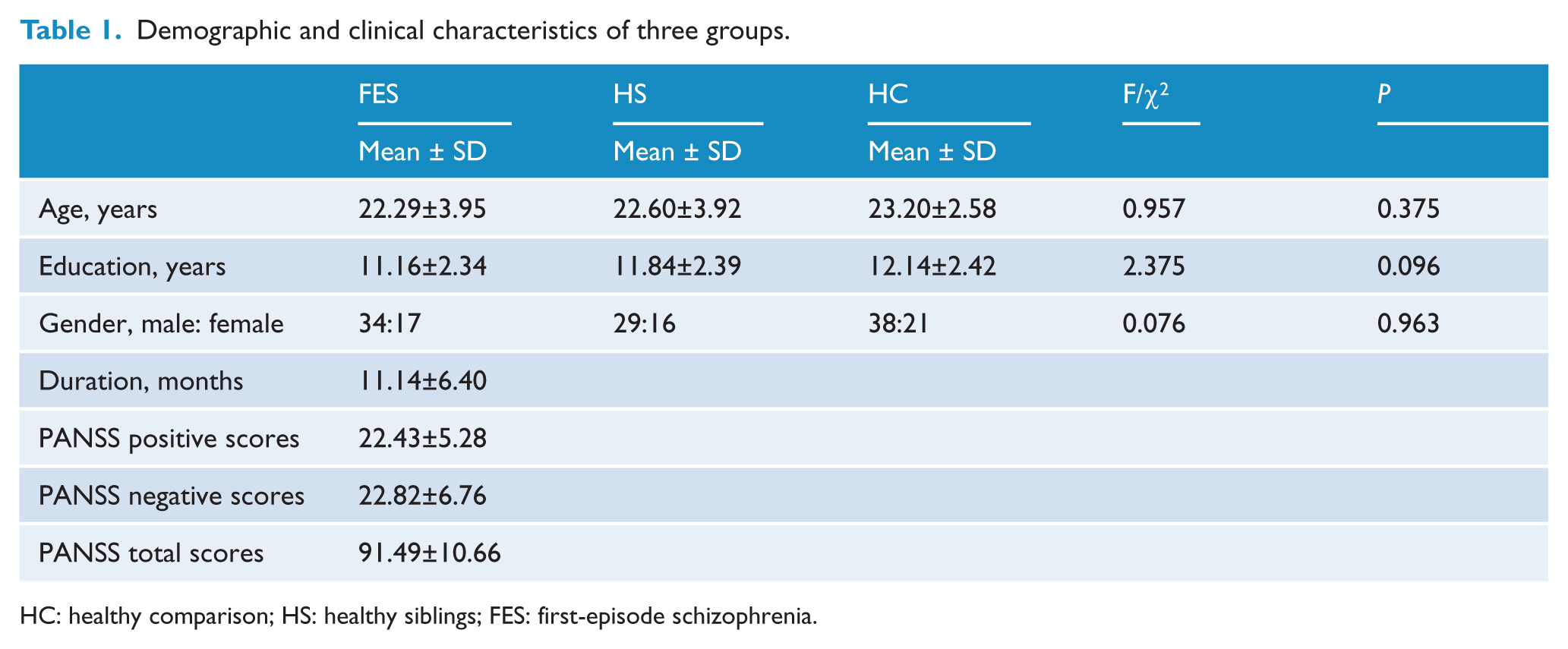
HC: healthy comparison; HS: healthy siblings; FES: first-episode schizophrenia.
Group differences in white matter abnormalities
The VBM analysis showed that white matter abnormalities were significantly different among the three groups in the left inferior frontal gyrus (IFG), left joint of external capsule and internal capsule, left cerebellum anterior lobe, left middle frontal gyrus, left hippocampus, right anterior cingulate and right internal capsule (corrected FDR, P<0.005; Figure 1).
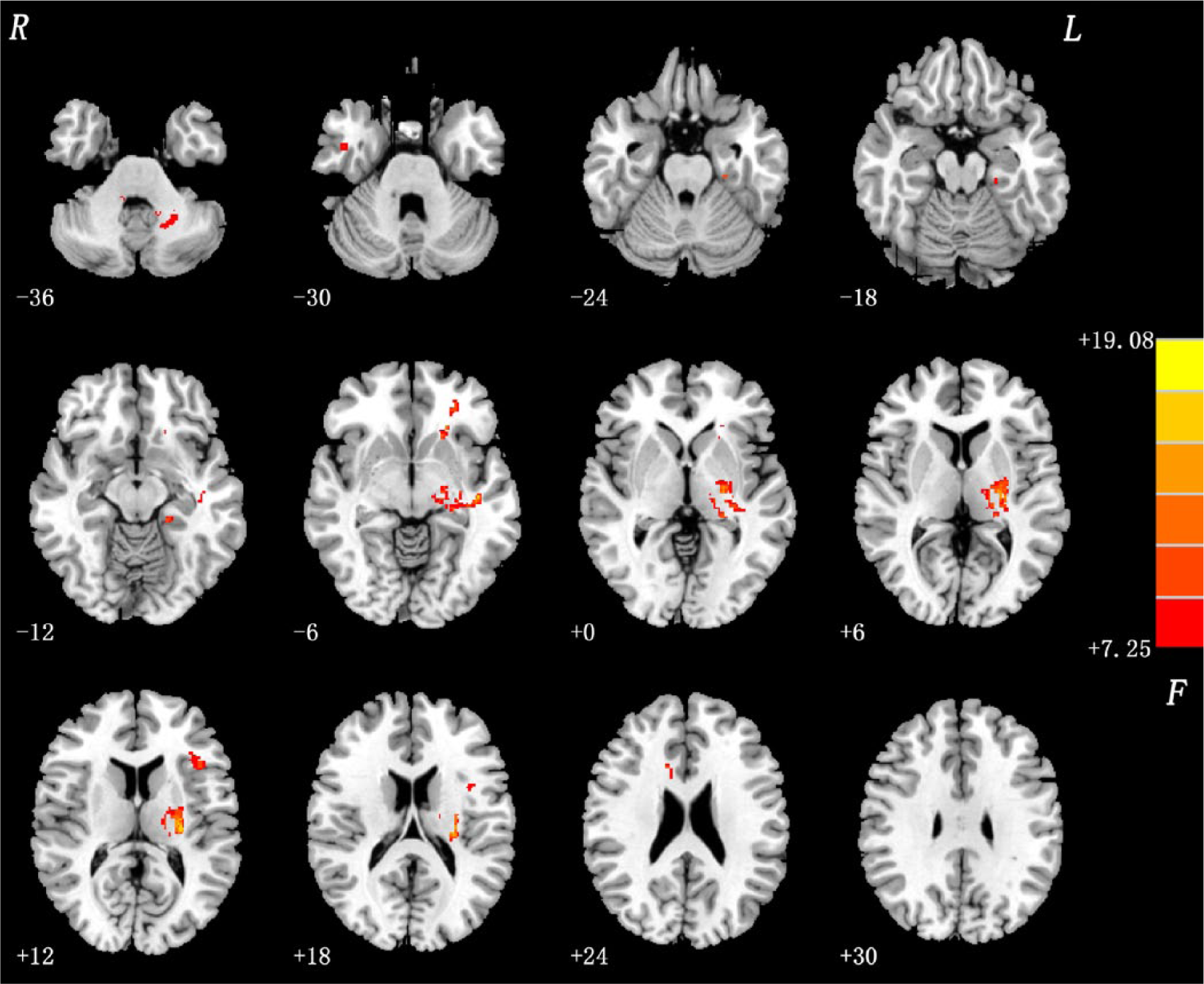
Group difference: ANOVA results. White matter abnormalities between three groups were detected in the left inferior frontal gyrus (IFG), left joint of external capsule and internal capsule, left cerebellum anterior lobe, left middle frontal gyrus, left hippocampus, right anterior cingulate, and right internal capsule. Statistical inferences were made with a voxel-level statistical threshold (P<0.005) after false discovery rate (FDR) correction for multiple comparisons.
The t-test comparing FES versus healthy subjects revealed highly significant white matter deficits in the left IFG and the left joint of external capsule and internal capsule in patients when compared to healthy subjects (P<0.005, corrected FDR; Table 2, Figure 2).
Brain regions showing significant white matter differences between the three study groups.
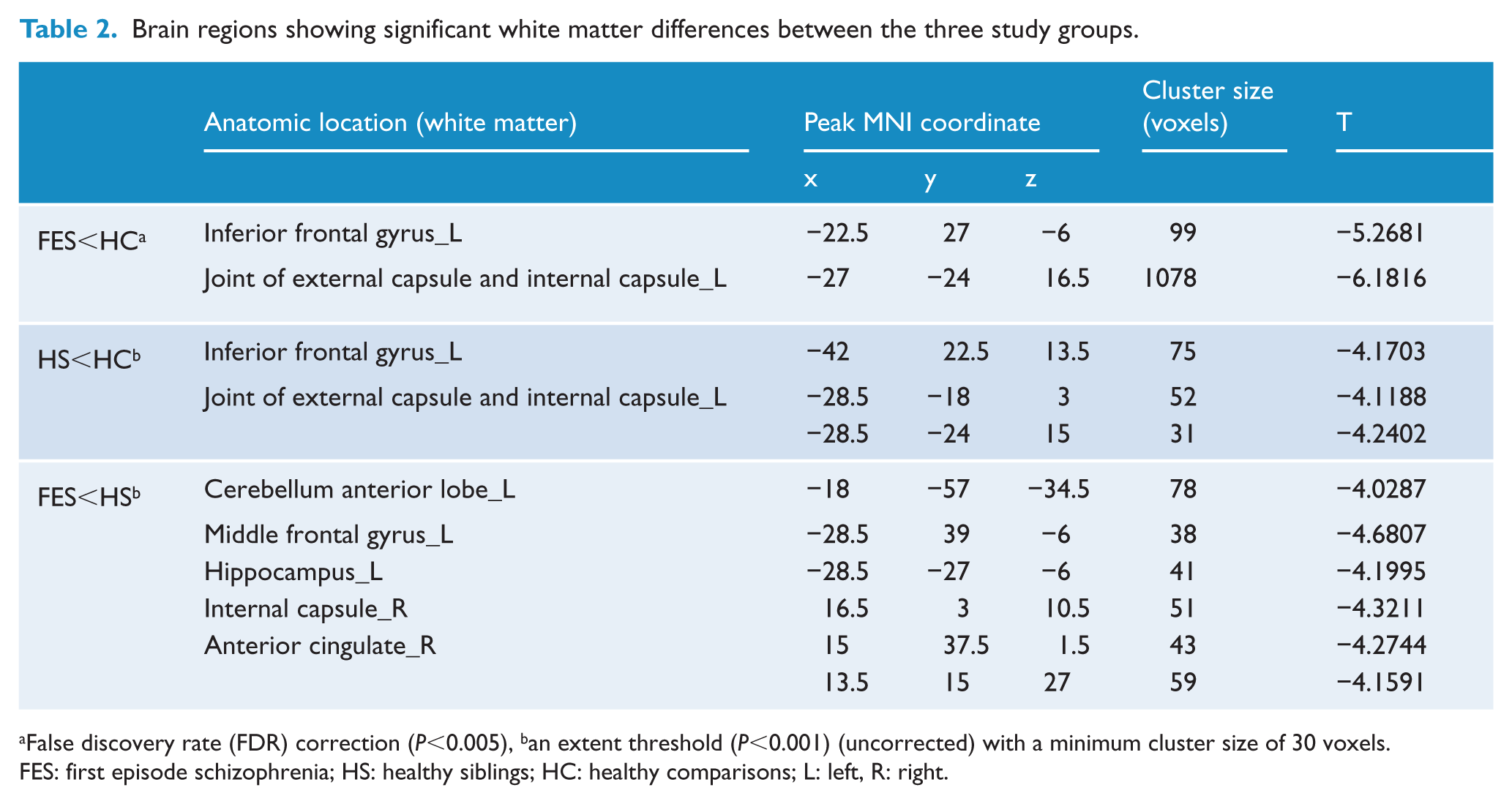
False discovery rate (FDR) correction (P<0.005), ban extent threshold (P<0.001) (uncorrected) with a minimum cluster size of 30 voxels.
FES: first episode schizophrenia; HS: healthy siblings; HC: healthy comparisons; L: left, R: right.
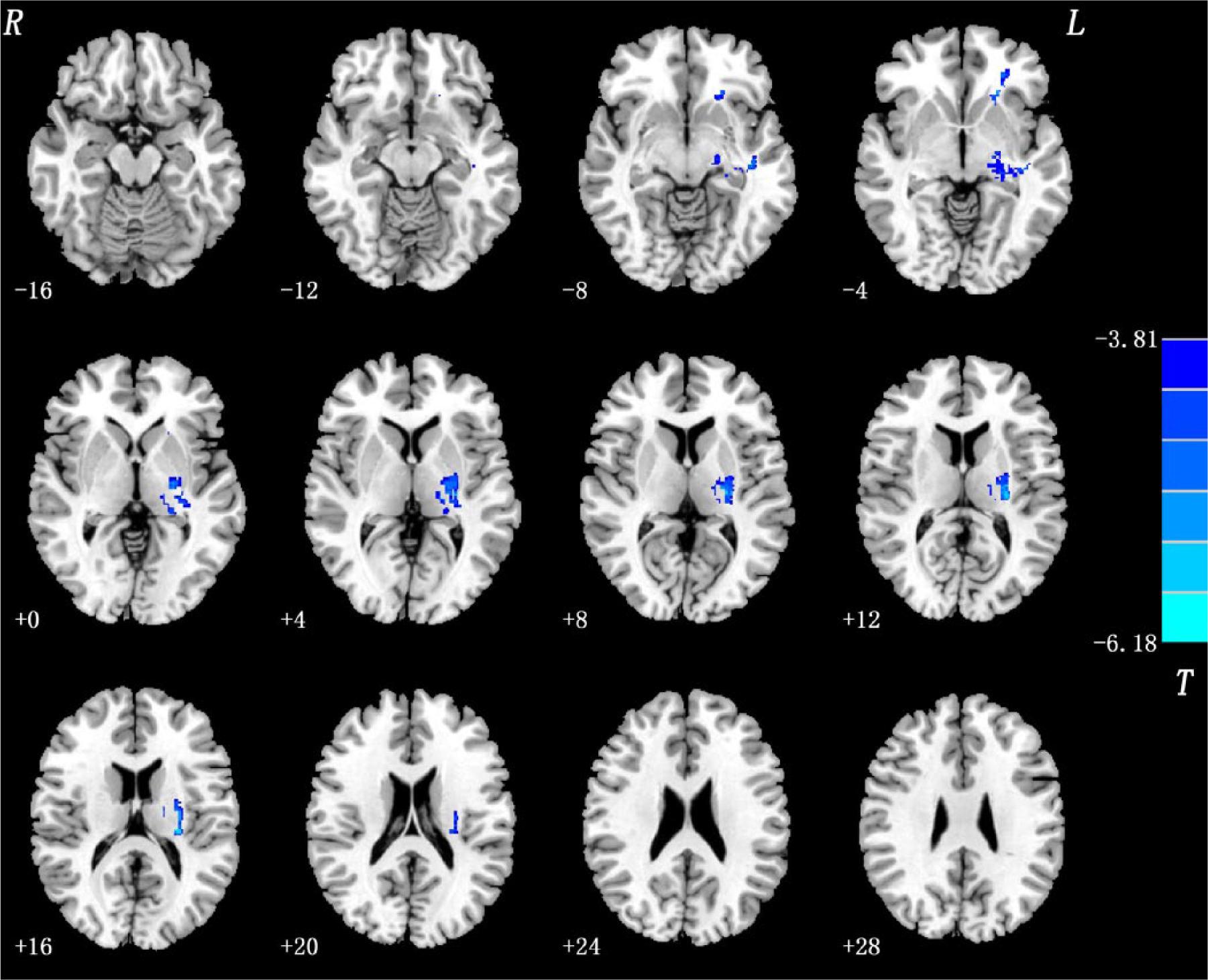
Group difference: Patients versus controls. In FES relative to healthy subjects, significant white matter deficits were detected in the left IFG and the left joint of external capsule and internal capsule. Statistical inferences were made with a voxel-level statistical threshold (P<0.005) after FDR correction for multiple comparisons.
The t-test comparing HS versus healthy subjects revealed significant white matter deficits in the above-mentioned two regions in the HS than healthy subjects (p<0.001, uncorrected). However the significant differences were not observed after FDR correction for multiple comparisons was performed (Table 2, Figure 3).
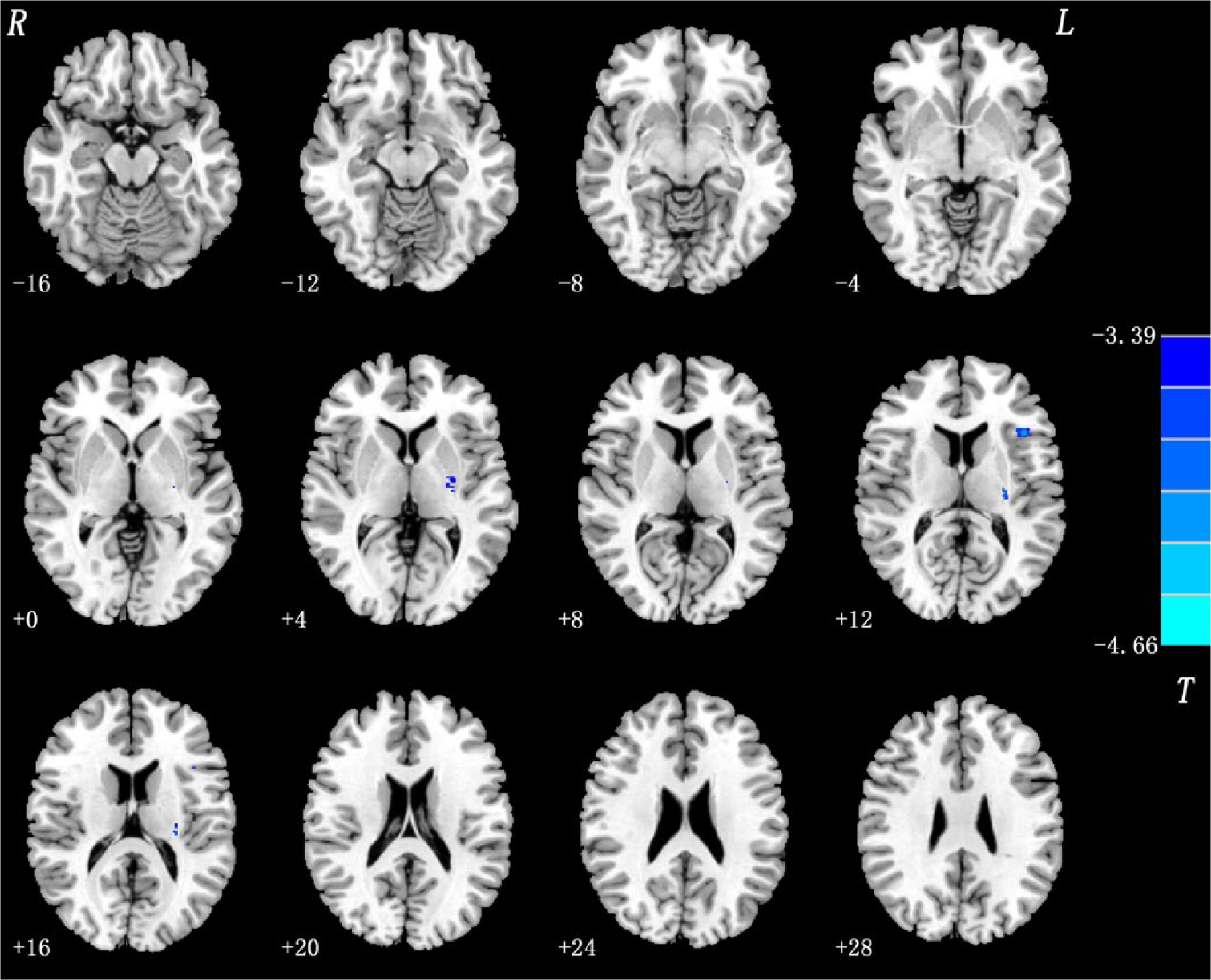
Group difference: Siblings versus controls. In HS relative to healthy subjects, significant white matter deficits were detected in the left IFG and the left joint of external capsule and internal capsule. Statistical inferences were made with an extent threshold (P<0.001, uncorrected) with a minimum cluster size of 30 voxels.
The t-test comparing FES versus HS revealed no suprathreshold voxels for increases or decreases at the corrected P<0.005 level. At a less stringent P-value (P<0.001, uncorrected), areas of less white matter in the patients compared to their siblings were observed in the left cerebellum anterior lobe, left middle frontal gyrus, left hippocampus, right anterior cingulate, and right internal capsule (Table 2, Figure 4).
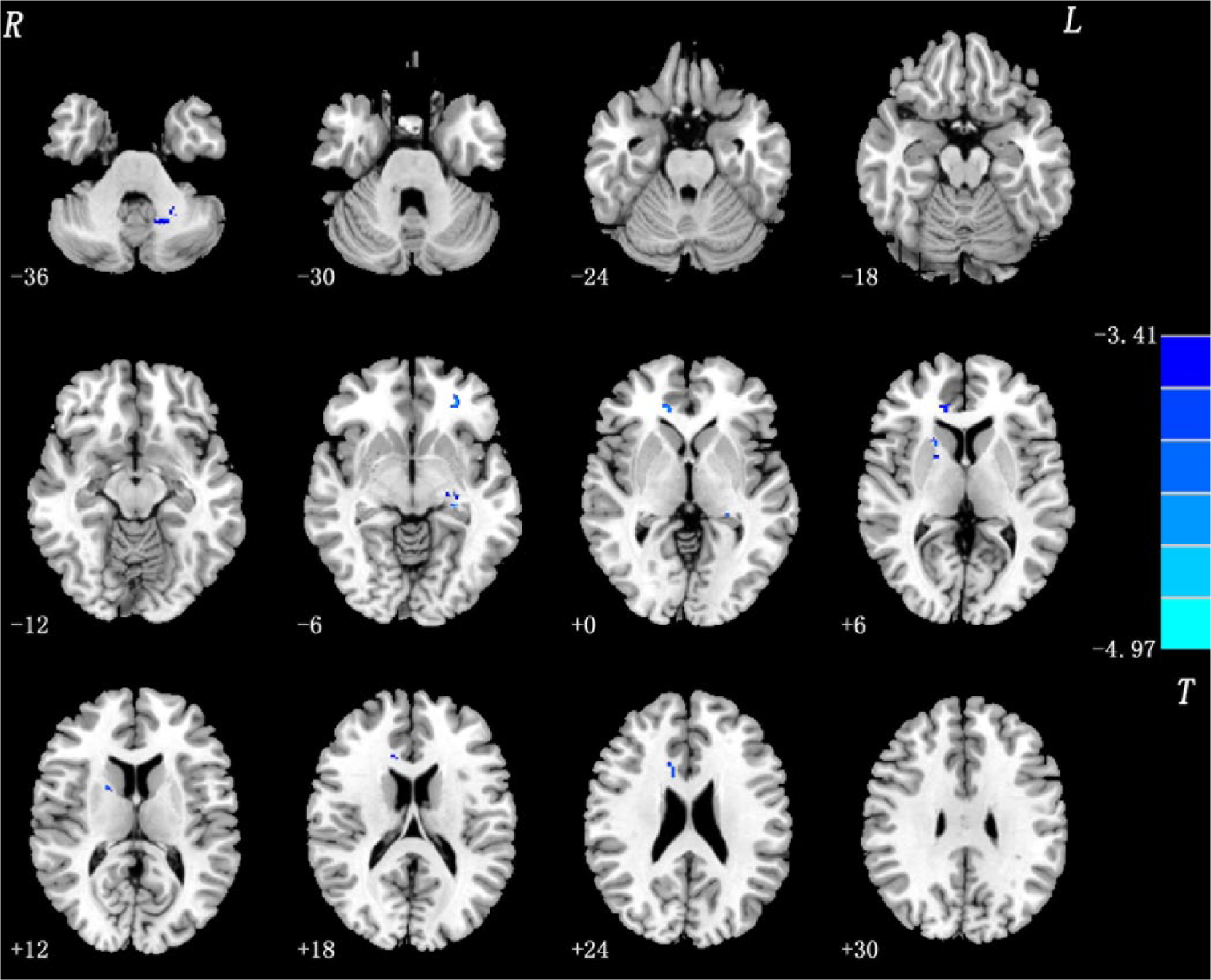
Group difference: Patients versus siblings. In FES relative to HS, areas of less white matter were observed in the left cerebellum anterior lobe, the left middle frontal gyrus, the left hippocampus, the right anterior cingulate and the right internal capsule. Statistical inferences were made with an extent threshold (P<0.001, uncorrected) with a minimum cluster size of 30 voxels.
Discussion
We report the findings of a VBM study investigating white matter deficits in drug-naive patients with FES and their unaffected siblings. The VBM analysis results indicated that FES patients and their unaffected siblings shared white matter deficits in the left IFG and the left joint of external capsule and internal capsule. Our study supports the findings that brain structure is heritable and siblings may share regional white matter deficits (Bartley et al., 1997; Weinberger et al., 1981).
In the present study, schizophrenia patients exhibited highly significant white matter deficits in the left IFG and the left joint of external capsule and internal capsule, this result is consistent with that described in previous studies. Whitford et al. (2007), for example, studied patients with FES and healthy subjects with MRI using VBM and found that FES patients exhibit white matter deficits in the frontal and temporal lobes. Similar results were reported by Hao et al. (2006), who used DTI and observed that FES patients exhibit significant reduced FA relative to healthy subjects in the white matter of the IFG, orbitofrontal cerebrum and cerebral peduncle. Furthermore, Bagary et al. (2003) used magnetization transfer imaging and observed reduced magnetization transfer ratio (which is likely related to abnormalities in myelin or reduced axonal density) in patients with FES in the white matter of the uncinatus fasciculus, prefrontal lobe, anterior cingulate and insula. These results suggested that white matter abnormalities are present in schizophrenia at illness onset.
Importantly, we also observed white matter deficits in siblings compared to healthy subjects in the left IFG and left joint of external capsule and internal capsule, although these deficits were smaller than those detected in FES patients. Our VBM findings are consistent with those shown by previous studies using diverse methods. Hao et al. (2009), for example, used a DTI method and found schizophrenia patients and their unaffected siblings share significant reduced white matter fractional anisotropy in the hippocampus and the left prefrontal cortex compared with healthy participants. However, the regional white matter differences between siblings and healthy subjects failed the FDR correction for multiple comparisons; these differences are likely related to our sample selection factors, such as age and drug-naive factors. Furthermore, a meta- analytic study (Boos et al., 2007) showed that white matter deficits is smaller and milder in first-degree relatives of schizophrenia patients compared with healthy comparison participants.
White matter plays an important role in connecting disparate regions of neural tissue and in modulating the transmission velocities of action potentials (Bartzokis, 2002). Therefore, the regional white matter abnormalities we observed in the present study could be expected to result in a disintegration of neural activity and a dysfunction in neural communication. A study has suggested that such disintegration in neural activity can be accounted for the cognitive impairment characteristic of schizophrenia (Bartzokis, 2002). The left IFG is associated with many cognitive processes such as semantic processing (Devlin et al., 2003) and verbal fluency (Hirshorn and Thompson-Schill, 2006). White matter abnormalities in the left joint of external capsule and internal capsule may reflect the white matter tract deficits in cortical and subcortical neural circuits; as a result, executive function, emotional stability, and motivation are impaired in schizophrenia patients. A recent DTI study found that white matter tract deficits between the left IFG and striatum are present in FES and appeared significantly associated with executive dysfunction (Quan et al., 2013). Thus, the shared regional white matter deficits in FES patients and their siblings could explain why unaffected siblings share a similar cognitive impairment with patients (Hu et al., 2011; Snitz et al., 2006).
White matter deficits can lead to alterations in behavior and dopaminergic function related to schizophrenia (Roy et al., 2007). Interestingly, both patient group and sibling groups displayed significant white matter deficits in the left IFG and the left joint of external capsule and internal capsule, but only the patient group was suffering from psychosis. This result could be partly explained by other risk factors, such as cannabis use and stressful life events (Lawrie et al., 2008) that may be more likely to act nearer to the onset time. Another possible explanation is that structural brain abnormality could be more extensive among patients compared to the siblings. In the present study, we did observe significant white matter deficits in the left cerebellum anterior lobe, left middle frontal gyrus, left hippocampus, right anterior cingulated, and right internal capsule in patients compared to their siblings, although significant differences failed the multiple comparison correction. The shared neuropathological features of schizophrenia patients and their siblings might represent meaningful intermediate phenotypes and the use of those intermediate phenotypes may help to investigate genetic susceptibility to the illness (Honea et al., 2008). It is very difficult to assess the individual risk of developing schizophrenia in a clinical setting. However, our findings suggest that brain structural MRI findings may reflect some common genetic vulnerability to developing schizophrenia. Thus, studies searching for susceptibility genes for psychosis can benefit from the use of neuroimaging findings, such as white matter deficits in IFG, reported in our study.
The present study acknowledges potential limitations. For instance, our study is a cross-sectional study. Thus, we may be unable to identify more subtle endophenotypes and to detect the ones that involve progressive changes. A longitudinal study should be conducted to confirm progressive white matter pathology in schizophrenia. VBM has also exhibits standard methodological limitations, including its vulnerability to normalization and smoothing errors (Crum et al., 2003). Considering these issues, we used a custom template to reduce ventricle-related artifacts and applied a moderate smoothing kernel to reduce statistical noise.
In conclusion, our findings extend those from previous VBM analyses showing that FES patients and their unaffected siblings might share white matter deficits in the left IFG and the left joint of external capsule and internal capsule; these regional white matter deficits may be related to genetic vulnerability to schizophrenia.
Footnotes
Acknowledgements
We gratefully acknowledge the participation of the study subjects and investigators.
Funding
The study was supported by Grants from the National Natural Science Foundations of China (Grant nos. 81260210, 81360211, and 30900483); the Natural Science Foundations of Guangxi (Grant no. 2013GXNSFAA019107); and the special funding by the Ministry of Health of the Peoples’ Republic of China (Grant no. 201002003).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
